Abstract
Glutamate (Glu) neurotoxicity is an important element of a number of neurological disorders including central nervous system (CNS) ischemia. We evaluated the effects of the novel AMPA Glu antagonist LY293558 on functional neurological outcome in two rabbit stroke models. In the reversible spinal cord ischemia model, ischemia of the caudal lumbar spinal cord was produced by temporary occlusion of the abdominal aorta. LY293558 was administered 5 min after recirculation as a 16 mg/kg i.v. bolus followed by 2.2 mg/kg infused over 1 h. Control animals received saline. LY293558 significantly increased the duration of ischemia required to produce paraplegia, from 30.5 ± 15.8 min (mean ± SD) controls to 50.1 ± 11.5 in treated animals (p < 0.01). In an irreversible model of cerebral ischemia, 50 μm plastic microspheres were injected into the carotid artery and lodged in the cerebral microvasculature. LY293558 did not significantly reduce neurological damage in this model. These data suggest that LY293558 may have therapeutic benefit following some types of ischemic injury.
The amino acid glutamate (Glu) is the principal excitatory neurotransmitter in the mammalian brain (Fonnum, 1984). Excessive neuronal release of Glu may be pivotal early on in the sequence of events resulting in ischemic cell death in the central nervous system (CNS). Several lines of evidence support this “excitotoxic hypothesis.” The application of Glu to neurons in culture (Prehn et al., 1993) or intracerebrally in rats (Beal et al., 1991) results in neuronal degeneration similar to that in ischemic injury. Excitatory amino acids, including glutamate, are elevated in ischemic brain (Benveniste et al., 1984; Matsumoto et al., 1993; Zini et al., 1993; Takagi et al., 1993), and brain regions particularly susceptible to ischemia express a high level of Glu receptors (Cotman and Iversen, 1987). The infarct volume resulting from an ischemic insult is significantly correlated with the magnitude of Glu release in brain (Takagi et al., 1993), and a Glu release inhibitor reduced ischemic brain injury (Leach et al., 1993). Glu neurotoxicity is thought to be a feature of a number of pathological conditions including epilepsy (Wuarin et al., 1992; Dingledine et al., 1990; Steen et al., 1983; Martin et al., 1992), Alzheimer's disease (Advokat and Pellegrin, 1992), and Parkinson's disease (Jacewicz et al., 1990; Smith et al., 1988; Turski et al., 1991).
Glu receptors may be divided pharmacologically into a number of receptor subtypes, including the N-methyl-D-aspartate (NMDA) receptor and the AMPA receptor (McLennan, 1983; Monaghan et al., 1983; Petralia and Wenthold, 1992). Competitive (Sauer et al., 1993; Madden et al., 1992b; Gotti et al., 1990; Nellgard and Wieloch, 1992a) and noncompetitive (Kochhar et al., 1988; Ozyurt et al., 1988; Gotti et al., 1990; Nellgard and Wieloch, 1992a) NMDA antagonists reduce ischemic injury, although this neuroprotectant effect has been attributed to drug-induced hypothermia by some authors (Buchan, 1992; Hayward et al., 1993). Although NMDA antagonists consistently reduce neural injury following irreversible focal ischemia, these compounds have a variety of undesirable side effects, including sedation (Kochhar et al., 1991), psychotomimetic properties (Koek et al., 1988), cardiac (Soltis and DiMicco, 1992) and respiratory dysfunction (Foutz et al., 1988), and potential interference with learning and memory (Advokat and Pellegrin, 1992). These side effects may limit the potential therapeutic usefulness of MK-801 and similar compounds in CNS ischemia.
Compounds that block the AMPA Glu receptor also appear to possess neuroprotectant properties (Nellgard and Wieloch, 1992b; Sheardown et al., 1990; Gill et al., 1992; Bullock et al., 1994a), and it has been reported that simultaneous blockade of both NMDA and AMPA receptors decreases damage in retina (Mosinger et al., 1991) and brain (Nellgard and Wieloch, 1992a). Although numerous reports have described the reduction of CNS injury by AMPA antagonists using histological measures, there were few data relating these findings to neurological outcome. In the present experiments, we have examined the ability of the novel AMPA antagonist LY293558 to improve functional outcome following CNS ischemia. Unlike most previously examined AMPA antagonists (e.g., CNQX, NBQX), LY293558 is highly water-soluble and is systemically active (Ornstein et al., 1993). Two ischemia models were used, a rabbit spinal cord ischemia model (RSCIM), (Zivin and DeGirolami, 1980) in which the abdominal aorta is reversibly occluded with a snare ligature, and the microsphere cerebral embolization model (MCEM) (Zivin et al., 1987), in which cerebral ischemia is induced by embolization with numerous small plastic microspheres injected into the carotid circulation.
In addition to the effects of LY293558 on neurological function, we also measured the time course of plasma concentrations of the compound in ischemic and nonischemic animals, and the effects of LY293558 on blood pressure (BP).
MATERIALS AND METHODS
AMPA antagonist
LY293558 [(–)-3S,4aR,6S,8aR-6-(2(1(2)H-tetrazol-5-yl)eth-1,2,3,4,4a,5,6,7,8,8a-decahydroisoquinoline-3-carboxylic acid] is a selective antagonist for the excitatory amino acid AMPA receptor. LY293558 is a highly water-soluble drug suitable for i.v. administration, and its properties have recently been described in detail (Ornstein et al., 1993).
Animals
Male New Zealand white rabbits (2–3 kg) were individually housed and provided food and water ad libitum before surgery.
RSCIM
Animals were anesthetized with halothane. Using a paramedial incision, the abdominal aorta was exposed at the level of the renal arteries. Small-diameter plastic tubing was placed around the aorta just distal to the left (more caudal) renal artery. The ends of the tubing were threaded through a small plastic button and then through a larger-diameter plastic tube, forming a snare ligature. The incision was closed around the tube so that the free ends of the tubing were accessible externally. Animals were allowed to recover for at least 3 h and appeared to behave normally prior to the initiation of ischemia. Aortic occlusion was performed by pulling and clamping the small tube around the aorta. Complete paraplegia was observed in all animals within 2 min of occlusion. Occlusion durations encompassing all grades of neurological function, from complete recovery to permanent hindlimb paralysis, were selected. At the end of the ischemic period, the tubing was released to restore blood flow through the aorta. The tubing was removed and the abdominal wall closed with wound clips without further anesthesia. Animals were returned to their home cages and maintained for 4 days. Animals dying within this period were excluded from the study to ensure that no animals with aortic thrombosis were included in the data analysis. Neurological function was evaluated 18 h and 4 d after ischemia/reperfusion by two observers blind to animal treatment and duration of ischemia. Animals were classified by the presence or absence of paraplegia. Paraplegic animals showed no motor response to noxious stimuli in the hind limbs and were totally incontinent. Rabbits that were not paraplegic were either normal or had some motor function of the hind limbs, even if voluntary movement was only barely detectable. Bowel and bladder function were variable. If it was difficult to ascertain whether an animal was totally paraplegic, the animal was classified as paraplegic.
At 5 min after the onset of reperfusion, animals received saline (n = 11) or LY293558 (n = 14) as a 16 mg/kg initial bolus followed by 2.2 mg/kg infused at a constant rate for 1 h.
MCEM
An incision was made lateral to the trachea to expose the right common carotid artery. The external carotid was ligated just rostral to the carotid bifurcation. A catheter, filled with heparinized saline and sealed with an injection cap, was inserted anterograde into the common carotid artery and secured with ligatures. The incision was closed with the distal end of the catheter accessible externally. Animals were allowed to recover from anesthesia for at least 2 h and appeared to behave normally prior to embolization. Plastic microspheres, 50 μm, (New England Nuclear, Boston, MA, U.S.A.) were carefully weighed for each animal and mixed with a tracer quantity of 15 μm microspheres labelled with 51Cr. Microspheres were suspended in a solution of 0.05% polysorbate-80 in normal saline and sonicated at high power for 10 min. The amount of radioactivity present in each microsphere dose was measured using a gamma counter, and microspheres were transferred to individual 0.5-ml gas-tight syringes (Hamilton Co., Reno, NV, U.S.A.) and sonicated at low power until embolization. The quantity of microspheres injected was varied from animal to animal to provide a range of microsphere doses encompassing all grades of neurological function. Animals were restrained briefly during embolization. The heparinized saline was withdrawn from the carotid artery catheter and the microsphere suspension was rapidly injected. The injection system was flushed with 3 ml normal saline, avoiding the formation of air bubbles within the catheter or injection cap. Animals were returned to their home cages for subsequent neurological evaluation.
Neurological function was evaluated 18 h after embolization by two observers blind to animal treatment and microsphere dose. Each animal was evaluated as either functional (alert and able to right itself) or abnormal (dead or having unequivocal neurological deficit such as greatly reduced level of spontaneous activity, inability to stand, or markedly uncoordinated movements). Following evaluation, animals were killed using Beuthanasia-D (Schering-Plough, Kenilworth, NJ, U.S.A.) and their brains removed. Radioactivity trapped in the brain was measured using a gamma counter. The total radioactivity recovered in the brain was compared to the specific activity of the injected microsphere dose to determine the weight of microspheres trapped in the brain.
Three groups of animals received i.v. infusions of LY293558. In the first group (n = 23), animals received a 16 mg/kg dose followed by 2.2 mg/kg infused at a constant rate over 1 h, initiated 5 min after embolization. In the second group (n = 10), animals received 8 mg/kg followed by 1.1 mg/kg infused over 1 h. In the third group (n = 8), animals received 16 mg/kg, followed by bolus administration of an additional 2.2 mg/kg once an hour for 18 hours. Each treatment group was compared with a group (of similar n) of saline-treated control animals.
Data analysis
Neurological damage as a function of ischemic insult was analyzed using quantal dose-response analysis techniques described previously (Waud, 1972; Zivin and Waud, 1992). A computer was used to fit logistic (S-shaped) curves to the fraction of abnormal animals as a function of ischemia duration (RSCIM) or weight of microspheres trapped in the brain (MCEM). Using this technique, the ischemic duration or microsphere weight necessary to produce clinically apparent neurologic deficit in 50% of a group of subjects may be computed for each experimental condition (ET50 in the RSCIM, for effective time; ES50 in the MCEM, for effective stroke). Pharmacological manipulations that improve neurological outcome will increase the ET50 or ES50, implying a shift of the dose-response curve to the right. Control and treatment animals were compared using t-tests, with p < 0.05 considered significant for all comparisons. This quantal bioassay technique allows high efficiency determination of dose-response curves using a relatively small number of subjects.
Concentration of LY293558 in plasma
Blood plasma levels of LY293558 were evaluated using a separate group of 10 animals. Three animals received abdominal aorta occluders as previously described, and each animal was subjected to 25 min occlusion of the abdominal aorta. Four animals received carotid artery catheters and were embolized with radiolabeled microspheres as described previously. Nonischemic animals underwent abdominal surgery, and snare ligatures were placed but not occluded. At 5 min after reperfusion (spinal cord ischemia) or microsphere injection (cerebral embolization animals), animals received LY293558, 16 mg/kg i.v., followed by 2.2 mg/kg infused over 1 h. Blood samples were collected at 15, 30, 60, and 90 min after embolization or reperfusion, and plasma was analyzed for LY293558 concentration by capillary gas chromatography (GC)/mass spectrometry (MS). Briefly, the method involved extraction of LY293558 from plasma samples with anion exchange cartridges, followed by derivatization of the carboxylic acid in LY293558 to its methyl ester, and acylation of the secondary amines with trifluroacetic anhydride. Derivatized products were then quantified using capillary GC/MS, with positive electron impact detection. The limit of detection for the assay was 0.05 μg/ml. The plasma concentrations were evaluated using a mixed-factorial design analysis of variance (ANOVA).
Effects of LY293558 on blood pressure
The effects of LY293558 on BP were evaluated in three animals. BP was monitored using a carotid artery catheter oriented retrograde and connected to a physiograph. BP was monitored for 20 min prior to drug treatment. LY293558 (16 mg/kg initial bolus followed by 2.2 mg/kg infused over 1 h) was administered i.v. The effects of LY293558 on BP were evaluated using repeated-measures ANOVA.
RESULTS
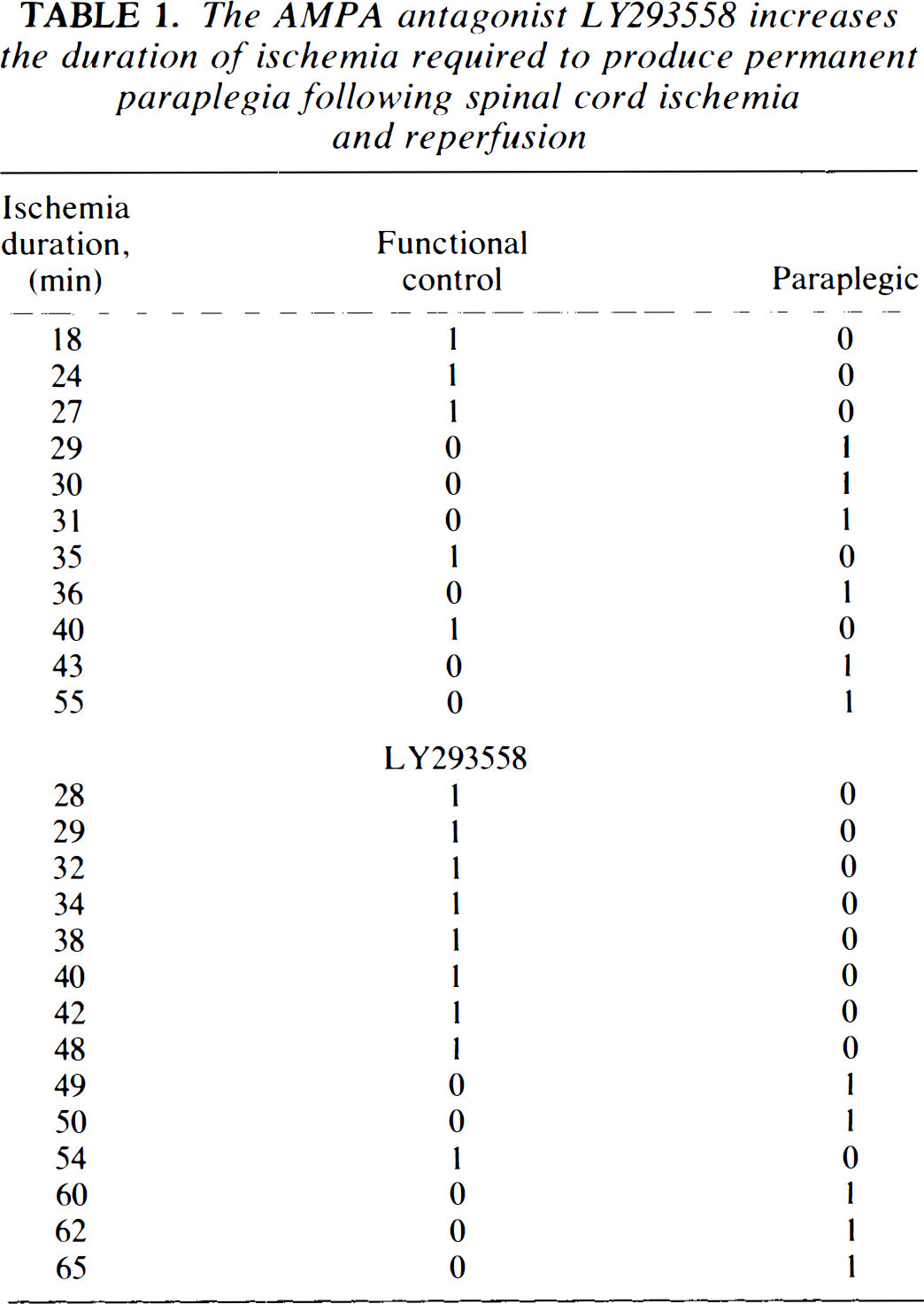
LY293558 substantially improved neurological outcome following reversible ischemia/reperfusion of the rabbit spinal cord. The duration of ischemia and neurological outcome at 18 h for the two groups are presented in Table 1. In the control animals, the duration of ischemia associated with a 50% probability of paraplegia was 30.5 ± 15.8 min (ET50 ± SD). In treated animals, this increased significantly to 50.1 ± 11.5 min (p < 0.01). These data are illustrated in Fig. 1. This significant increase in ET50 was maintained 4 days following ischemia/reperfusion.
The AMP A antagonist LY293558 increases the duration of ischemia required to produce permanent paraplegia following spinal cord ischemia and reperfusion
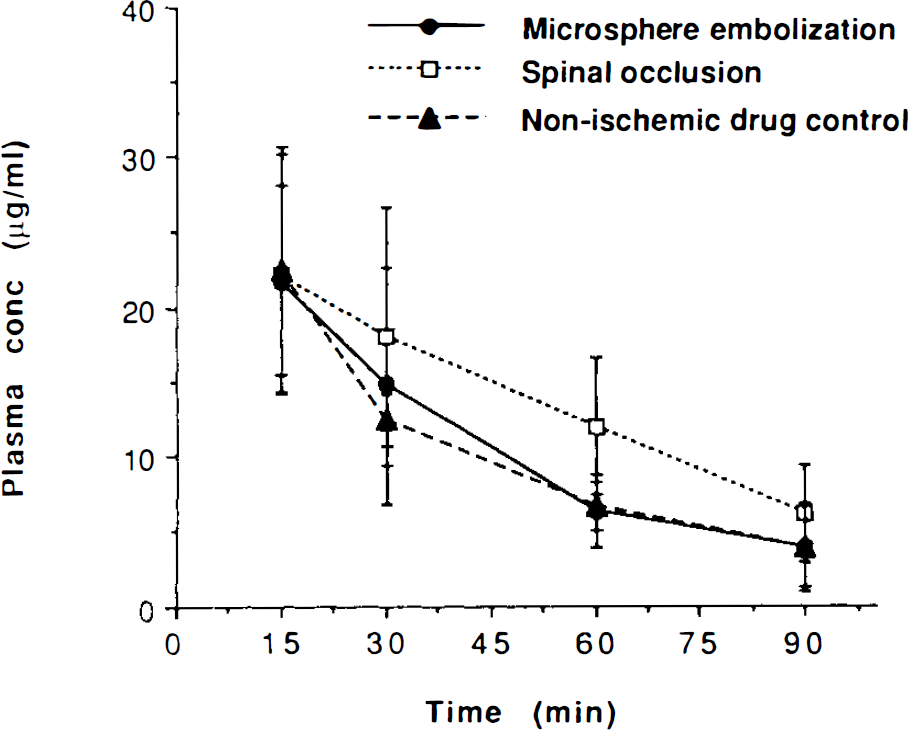
Plasma concentrations of LY293558 in microsphere embolization, spinal occlusion, and nonischemic control animals. Each point represents the mean of four samples, and the bars indicate the standard deviations. Plasma concentrations among the three groups were not statistically significant.
In the cerebral embolization model, no significant neuroprotectant effect was observed. In the first experiment, the quantity of microspheres associated with a 50% likelihood of permanent neurological injury in the control group was 0.50 ± .40 mg (ES50 ± SD). In animals receiving a LY293558 16 mg/kg bolus followed by a 2.2 mg/kg infusion for 1 h, the ES50 was 0.64 ± .30, not significantly different from control. In the second experiment, when the dose was halved, the ES50 for the control and treated groups were, respectively, 0.49 ± .21 and 0.34 ± .30 mg. Repeated administration for 18 h also failed to significantly improve neurological outcome in this model.
LY293558 significantly increased both systolic and diastolic BP. BP increased rapidly following drug administration, peaking within 5 min, and gradually returning to baseline over the next 20 min. Systolic BP increased from a baseline of 115 ± 8.6 (mean ± SD) to a peak 130 ± 5.0 [F(1,2) = 27, p = 0.04]. Diastolic BP also increased significantly from 78.33 ± 2.9 to 98.33 ± 7.6 [F(1,2) = 48, p = 0.02].
Plasma LY293558 declined significantly (p < 0.0001) over 90 min, despite the 16.0 mg/kg bolus followed by 2.2 mg/kg infusion for 1 h. Clearance of LY293558 was not significantly different in microsphere embolization, spinal cord occlusion, and nonischemic control animals (see Fig. 1).
Animals treated with LY293558 displayed mild sedation and ataxia for several hours after drug administration, although the sedative effect of this compound appeared less severe than we have observed with NMDA antagonists (e.g., MK-801) (Kochhar et al., 1988; Madden et al., 1992a). All animals had fully recovered by the 18-h evaluation.
DISCUSSION
AMPA antagonists have reduced infarct volume (Smith and Meldrum, 1992; Gill et al., 1992; Buchan et al., 1991; Bullock et al., 1994b) and neuronal degeneration in hippocampus, striatum, cortex, and cerebellum (Sheardown et al., 1990; Diemer et al., 1992; Nellgard and Wieloch, 1992b; Buchan et al., 1991; Baichen and Diemer, 1992; Nellgard and Wieloch, 1992a) in several animal models. De Graba and coworkers (1994) recently demonstrated improved functional outcome following administration of 2,3-dihydroxy-6-nitro-7-sulfamoyl-benzo(F)-quinoxaline (NBQX). Here, we show that administration of the AMPA antagonist LY293558 significantly improved neurological function following reversible spinal cord ischemia but not irreversible cerebral ischemia. Neuroprotection was observed at both 18-h and 4-day evaluations, indicating that the neuroprotective effects of LY293558 persist after the compound has been cleared from circulation after transient ischemia. LY293558 improved neurological outcome in the reversible spinal cord ischemia model but not in the irreversible cerebral embolization model. This may be attributable to differing sensitivities of the two models. In this RSCIM, a difference in the survival of a relatively small number of motor neurons may produce a detectable improvement in hindlimb function, whereas in the MCEM, survival of a large number of neurons is probably necessary to detect neuroprotection. It is also possible that the short half-life of the compound and the failure to reach a steady-state drug concentration, despite the continuous infusion procedure, merely delayed the development of neural injury in the irreversible ischemia model. The decline in circulating LY293558 may not have been important in the spinal cord model, in which ischemia is followed by reperfusion, but may have been critical in the microsphere model. Bullock et al. (1994b) recently reported that this compound reduced the volume of ischemic injury following irreversible cerebral ischemia in cats, when the animals were killed after 6 h of ischemia. Comparison of these two studies must be made with caution, however, because there were a number of methodological differences. In that study, animals were anesthetized and their BPs and pHs manipulated, and a constant drug level was maintained, throughout the 6-h period until killing. It should also be noted that we have previously demonstrated significant neuroprotection following administration of NMDA receptor antagonists in this model (Kochhar et al., 1988; Madden et al., 1992b), and it is unclear whether the present results indicate that AMPA antagonists are not effective in this type of embolic ischemia or whether the dose was insufficient.
Administration of 16 mg/kg LY293558 rapidly increased BP by an average of 15 mm Hg (systolic) and 20 mm Hg (diastolic). The effectiveness of this compound in the reversible spinal cord ischemia model, but not in the irreversible cerebral embolization model, may reflect enhanced reperfusion of the spinal cord resulting from elevated BP. The effects of hypertension in irreversible cerebral ischemia are controversial. A large body of evidence from the spontaneously hypertensive rat indicates that hypertension at embolization may exacerbate ischemic injury. Induced hypertension, however, has reduced neurological injury in some CNS ischemia preparations (Sterz et al., 1990; Cole, Drummond et al., 1990; Patel et al., 1991). We have recently observed a similar pattern of results with a hemoglobin solution that elevates BP (Bowes et al., 1994). At the moment, the precise relationship between BP and neurological injury is unclear. Other physiological variables, e.g., cerebral blood flow, should also be examined.
AMPA antagonists as a treatment for CNS ischemia are attractive because they do not have the potential for psychiatric side effects, as do NMDA antagonists. Because AMPA antagonists do not bind to the phencyclidine receptor, they may prevent excitotoxic injury with fewer psychiatric side effects, and, in animals, drugs that block the AMPA receptor appear less sedating than NMDA antagonists (Nellgard and Wieloch, 1992a). It is also possible that combined NMDA and AMPA therapy may improve neurological function to a greater extent than either substance alone. This has been observed histologically in neurodegeneration of retina and brain (Mosinger et al., 1991; Nellgard and Wieloch, 1992a), but not, to our knowledge, in a functional neurological assay. The present experiments suggest that AMPA antagonists may improve neurological function following some types of CNS ischemia. In contrast to most other CNS ischemia models, these studies have assessed neural injury in awake and unanesthetized animals, limiting the effects of confounding variables such as temperature or anesthesia. These results suggest a potentially beneficial role of AMPA antagonists in the pharmacological treatment of CNS ischemia, although cardiovascular, blood flow, and potential side effects require further study.
Footnotes
Acknowledgment:
The authors wish to thank Sonia Núñez and James Wright for their excellent technical assistance, and Dr. Derryle Schoepp for his comments. The physiograph was provided by Dr. H. Kirk Hammond.
