Abstract
Therapeutic hypothermia is neuroprotective after brain injury, reducing metabolic demand, inflammation, and secondary injury cascades. However, clinical application is limited by systemic side effects, and selective brain cooling without systemic hypothermia has not been demonstrated in large mammals. We aimed to demonstrate the feasibility of selective brain cooling using cerebrospinal fluid (CSF) exchange with cooled saline via a double-lumen external ventricular drain (EVD) system in a porcine model. Using 12 pigs under general anesthesia, we placed a double-lumen EVD in the lateral ventricle and a lumbar spinal drain to enhance CSF outflow. Cooled NaCl (13 °C) was infused at 120, 360, or 720 mL/h while maintaining intracranial pressure. After reperfusion, mean arterial pressure was increased to 140 mmHg. Brain temperatures were recorded bilaterally; core temperature was monitored. Ipsilateral brain temperature decreased by 7.5 °C, contralateral cooled by 3.1 °C. Core temperature remained stable. Target temperature was reached in 1.5 h at 120 mL/h, and within 10–15 min at 360–720 mL/h. Arrhythmias occurred with NaCl but not with Ringer’s acetate. We demonstrate that selective brain cooling is feasible in a large mammal model via CSF exchange without systemic hypothermia. This method may offer a safe, controllable approach for neuroprotection.
Introduction
Hypothermia has been studied and been observed as a neuroprotective after brain injury (BI).1,2 In acute phase, after BI, hypothermia affects the brain metabolism by decreasing oxygen and glucose consumption. Apoptosis and inflammatory mediators are also impacted. In the later stage of BI, enzyme activation stimulates protein and lipid breakdown, which can be slowed and decreased with hypothermia. In the later stages of brain injury, hypothermia has been associated with enhanced angiogenesis, neurogenesis, and synaptogenesis. 3 Moreover, hypothermia has demonstrated a reduction in vasogenic edema and blood brain barrier dysfunction. 4
Despite hypothermia’s neuroprotective effect, randomized controlled trials and international guidelines have demonstrated its benefits primarily in global cerebral ischemia, such as after cardiac arrest and neonatal hypoxic–ischemic encephalopathy.3,5–9 Systemic hypothermia has been associated with coagulopathy, infection, and cardiac arrhythmias.10,11 Furthermore, current technologies have well established complications.3,5–10 The majority of complications related to hypothermia have been due to systemic hypothermia and selective brain cooling has never been demonstrated in large mammals without changes in core temperature of subject. Given this, there is a need to perform selective brain cooling that alleviates systemic complications. We are reporting utilizing a novel fluid exchange device (IRRAflow® from IRRAS Corporation, Stockholm, Sweden) in a porcine model to selectively induce hypothermia with minimal systemic core temperature change.
Materials and methods
We hypothesized that by actively exchanging cerebrospinal fluid (CSF) with cooled solution we can selectively reduce brain temperature without changes in core temperature of the porcine model body. All experiments were performed using female domestic pigs and were approved by the national Animal Experiment Board (Hankelupalautakunta ELLA). This proof-of-concept study was conducted in 12 female domestic pigs (3 months old, ~30 kg). Approval was granted under protocol number ESAVI-2022-006345 by the Animal Experiment Board in Finland. All procedures complied with Finnish national legislation and the European Directive 2010/63/EU on animal welfare. ARRIVE guidelines were followed. An initial cohort of six pigs underwent endovascular occlusion to induce transient global cerebral ischemia. Bilateral vertebral and common carotid arteries were occluded using balloon catheters for 20 min, followed by angiographic confirmation of ischemia and reperfusion.
Ultimately, 12 pigs were included in the final analysis. The animals were divided into four experimental groups based on irrigation rate, ischemia status (Table 1). In ischemic model after reperfusion a mean arterial pressure (MAP) increased to 140 mmHg during the occlusion phase.
Experimental groups and irrigation parameters. Summary of porcine groups used in the study, including ischemia status, irrigation rate, duration, MAP, and number of animals per group.

The porcine model was put under general anesthesia, and in supine position. On the porcine model Kocher point (above the lateral ventricle in mid pupillary line) incision of skin was performed and bone was drilled. After puncture of dura, we inserted a double lumen external ventricular drainage catheter (EVD) to lateral ventricle 3–4.5 cm from skin incision. The proximal lumen was used for infusion and the distal lumen for drainage, enabling continuous CSF exchange. Lumbar puncture was performed, and a catheter was inserted in the lumbar CSF space for CSF removal and to increase CSF exchange. Since cranial and spinal CSF pathways are connected this allows to accelerated removal of warm CSF for exchange to cooled saline or ringer acetate.
This concept is based on the patent pending by corresponding author WO2024097946A1, which describes an extracorporeal CSF circulation and cooling system using an external dual-lumen catheter. The system allows continuous controlled infusion and drainage of CSF, with integrated temperature regulation. By cooling the irrigation fluid and simultaneously removing endogenous CSF, it is possible to reduce brain temperature selectively without affecting systemic core temperature. In addition to cooling, the system supports simultaneous monitoring of intracranial pressure and drainage rate.
After the catheters were in the correct position, a commercially available digital pump, with ability to irrigate, drain, and measure intracranial pressure was connected to the double lumen catheter in the ventricle (Figure 1). The system included a dual-lumen EVD (IRRAflow®), a digital pump with ICP monitoring (IRRAS), and continuous ECG and ABP monitoring via Philips IntelliVue MX700. The digital pump operates in a cycle fashion, irrigation, drainage, and monitoring. Moreover, the digital pump operates within a high and low intracranial pressure (ICP) parameters set by the treating physician. This ensures that too much fluid is not delivered nor taken out. The device is FDA-cleared and CE-marked.12–14 Once the treating parameters were set on the digital pump, the tubing of the system was inserted into two ice baths to ensure the saline/ringer acetate temperature was cooled to its maximum in this setup (13 °C; Figure 2). The system was then programed to irrigate at a volume of 120–720 mL/h (software was updated to use higher volume for irrigation of CSF space with cooled saline/ringer acetate to lateral ventricle while controlling intracranial pressure) and operate between an ICP range of 0 and 20 mmHg. The infusion temperature of 13 °C was achieved by cooling Ringer’s acetate/NaCl to 4 °C and using ice packs. Due to transit time and ambient conditions, the fluid reached the brain at ~13 °C. This temperature was achieved based on technical reasons. 15 Once the system was irrigating the spinal catheter was allowed to drain (Figure 3).

Double lumen catheter with ability to do CSF exchange.
Experiment set-up where cooled ringer acetate goes through ice bath before enter in to CSF space.
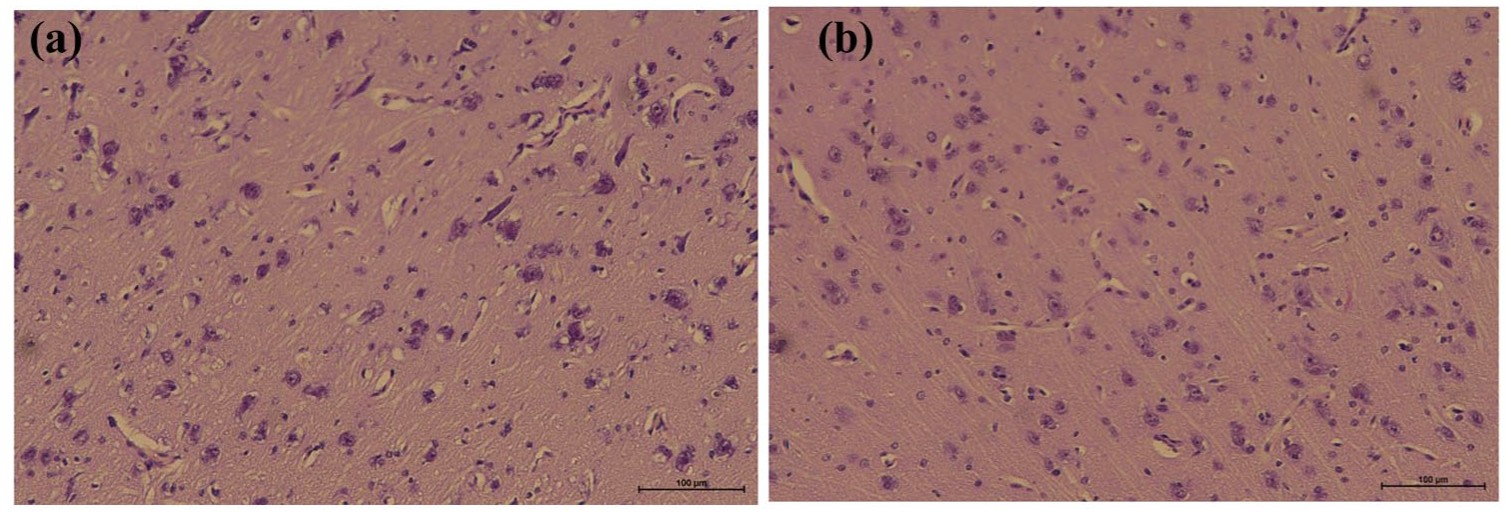
The cooler (a) hemisphere is less edematic compare to warmer (b) hemisphere. This is due to catheter location.
Results
In pigs subjected to endovascular occlusion, angiographic confirmation demonstrated successful transient ischemia. After removing of endovascular balloons, cooled solutions were irrigated according to the Table 1, where results can be seen in relation to irrigation volume.
While the system was irrigating the cooled saline (one pig) or ringer acetate (11 pigs), brain parenchymal temperature was measured from the contralateral brain hemisphere (from the double lumen catheter) and the ipsilateral brain hemisphere (all temperature measurements were done from new drilled sections). Intraparenchymal temperature probes were placed bilaterally in the frontal and posterior lobes (two ipsilateral, two contralateral). Contralateral brain hemisphere temperature dropped by 3.1 °C from core temperature while core temperature changed only by 0.5 °C. Ipsilateral temperature cooled by 7.5 °C from core temperature (38.3 °C) to 30.8 °C, while core temperature dropped to 37.7 °C. Selective cooling to 30.8 °C was initially achieved in 1.5 h (irrigation rate 120 mL/h). However, after increasing the irrigation rate to 360 and further to 720 mL/h, the same temperature was reached within 15–10 min accordingly, indicating that higher flow rates significantly accelerate cooling.
One porcine started to have arrhythmias when NaCl saline was used, this is most likely due to the hyperventilation due to changes of CSF composition previously reported in infection cases (Rezai Jahromi et al.12,16). After this observation NaCl was changed to ringer acetate and arrythmias was not seen in any other animals (n = 11). At the completion of the study, animals were sacrificed with intravenous KCl injection under general anesthesia.
These results demonstrate that selective brain cooling is feasible with elevated MAP (140 mmHg), which supports stable perfusion during irrigation. In histological analysis, the brain lobes that reached lower temperatures showed visibly less edema compared to the contralateral lobes that remained warmer after ischemia (Figure 3). This suggests that localized hypothermia may reduce tissue injury and improve post-ischemic outcomes.
Discussion
In literature, there are different systems and techniques utilized to cool the brain, including external cooling, intranasal cooling, and endovascular cooling.6,11,15 External cooling, such as the use of ice packs or cooling blankets, is a non-invasive and readily available method of cooling the brain. However, this method may not be effective in achieving deep brain cooling, and it may be limited by the need for patient cooperation and comfort. Intranasal cooling, which involves the use of a cooling device inserted into the nasal cavity, is relatively non-invasive and has been shown to be ineffective in lowering brain temperature. Similarly, noninvasive head and neck surface cooling has been explored in patients with traumatic brain injury, achieving modest reductions in brain temperature without systemic hypothermia. 17 Endovascular cooling, which involves the insertion of a catheter into the femoral vein to circulate cooled saline, is a more invasive but effective method of cooling the brain. However, this technique has several reported systemic issues.
During the study, one of the porcines experienced cardiac arrhythmia. Arrhythmias were observed in one of 12 pigs, which received NaCl irrigation. The remaining pigs, treated with Ringer’s acetate, did not exhibit arrhythmias. While electrolyte imbalance is a plausible cause, autonomic dysregulation or direct cooling of brainstem cardiac centers cannot be excluded. Before advancing to human studies, the optimal timing, cooling rate, and controlled rewarming protocols must be determined, especially given the arrhythmias occasionally observed during rewarming phases.
Conclusion
In this large-animal proof-of-concept study, we demonstrate that selective brain cooling is feasible through CSF exchange using a double-lumen EVD, without systemic hypothermia. This technique allows precise, reproducible modulation of brain temperature, and represents a promising foundation for future translational studies targeting neuroprotection. Further optimization of cooling dynamics and safety parameters is required prior to clinical application.
Footnotes
Acknowledgements
Not applicable.
Author contributions
BRJ, NJ, JK, and SY-H. conceived the study, designed the experimental protocol, and performed data analysis. BRJ, NJ, SY, JK, NB, MN, and ARB. prepared the manuscript, critical revisions, and intellectual input. All authors reviewed and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: IRRAS Corporation provided the IRRAflow® device used in this study but did not fund the research. The study was funded by the Finnish government fund(TYH20244). The company had no role in study design, data collection, analysis, or manuscript preparation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Behnam Rezai Jahromi has received grants from IRRAS to run ARCH clinical trial. All other authors declare no conflicts of interest.
Ethical considerations
All experiments were approved by the Animal Experiment Board in Finland under protocol number ESAVI-2022-006345. All procedures complied with Finnish national legislation and the European Directive 2010/63/EU on animal welfare. ARRIVE guidelines were followed.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data availability statement
Raw data supporting the findings of this study are available from the corresponding author upon reasonable request.
