Abstract
Acute ischemic stroke is a leading cause of death and disability worldwide. Therapeutic hypothermia has long been considered as one of the most robust neuroprotective strategies. Although the neuroprotective effects of hypothermia have only been confirmed in patients with global cerebral ischemia after cardiac arrest and in neonatal hypoxic ischemic encephalopathy, establishing standardized protocols and strictly controlling the key parameters may extend its application in other brain injuries, such as acute ischemic stroke. In this review, we discuss the potential neuroprotective effects of hypothermia, its drawbacks evidenced in previous studies, and its potential clinical application for acute ischemic stroke especially in the era of reperfusion. Based on the different conditions between bench and bedside settings, we demonstrate the importance of vascular recanalization for neuroprotection of hypothermia by analyzing numerous literatures regarding hypothermia in focal cerebral ischemia. Then, we make a thorough analysis of key parameters of hypothermia and introduce novel hypothermic therapies. We advocate in favor of the process of clinical translation of intra-arterial selective cooling infusion in the era of reperfusion and provide insights into the prospects of hypothermia in acute ischemic stroke.
Introduction
Acute ischemic stroke (AIS) is the leading disease causing human death and disability worldwide. 1 It brings huge burden on health-care systems and socio-economic development. 2 However, it is regrettable that the current therapies for AIS are relatively limited and most of them failed to reduce mortality or improve neurological prognosis. In addition, due to the narrow therapeutic time window and the strict eligibility criteria, 3 very few patients can receive these effective treatments clinically. For example, only 3–7% AIS patients have been actually treated with intravenous thrombolysis in Western countries. 4 Therefore, it is urgent to find other new therapies, so as to truly achieve the goal of improving the prognosis of AIS patients.
For a long time, people have been keen to explore the effective neuroprotective strategies in AIS. A large number of neuroprotective agents have been tested for its efficacy in the last few decades. 5 Disappointingly, neuroprotective agents, regarded as effective drugs in pre-clinical studies, have been proved ineffective in clinical trials unexceptionally. 5 This leaves the neuroprotection translation in a quandary. Given the huge sum of R&D expenditure and the lack of promising positive results, many pharmaceutical companies have abandoned the development of neuroprotective agents.
Although the neuroprotection translation in AIS has gone through the tribulation, therapeutic hypothermia is still thought to be one of the most robust neuroprotective strategies. 6 The application of hypothermia as a therapeutic agent has a long history. The earliest record of hypothermia can even be traced back to 3500 BC. 7 Then, after sleeping for centuries, hypothermia rekindled our interest in the late 1980s when Busto and colleagues reported that lowering brain temperature by only a few degrees could confer marked protection. 8 In the past few decades, great breakthroughs have been made in the field of hypothermia. The neuroprotective effects of hypothermia has been demonstrated by several clinical trials in resuscitation after cardiac arrest9,10 and hypoxic ischemic encephalopathy in neonates, 11 and is thus recommended in the guidelines for clinical use.12,13 However, less encouraging progress has been made in treating AIS despite continuous efforts.
In view of the great progress made in the field of therapeutic hypothermia and endovascular therapy in recent years, 14 summarizing the existing knowledge will better guide the future development of hypothermia, especially when combined with wide application of endovascular therapy in clinical practice. In this review, we mainly focus on the role of hypothermia and its potential translations in AIS. First, we summarize the neuroprotective mechanisms of hypothermia and make an analysis of mismatches between research and clinical applications. Then, we demonstrate the importance of vascular recanalization for neuroprotective effects of hypothermia in AIS, analyze the key parameters impeding hypothermic therapy in AIS, introduce novel hypothermic therapies that have emerged in recent years, and advocate in favor of the process of future clinical translation of intra-arterial selective cooling infusion (IA-SCI). Finally, we discuss the perspectives of hypothermia in AIS, in the hope to evoke inspirations for future studies in the field.
Neuroprotective mechanisms of hypothermia
The neuroprotective mechanisms of hypothermia in AIS have been deeply studied (Figure 1). Numerous pre-clinical studies based on vascular recanalization models have shown that hypothermia can affect multiple aspects of stroke pathophysiology in different stages, including but not limited to cell metabolism, cell death, inflammation, and white matter integrity. 15 Recent findings also hint that a relatively fast and mild hypothermia exert greater and longer-term benefits in both cell culture and rodent models.16–18 Similar to cocktail drugs, hypothermia targets multiple injury phases as well as different cell types.
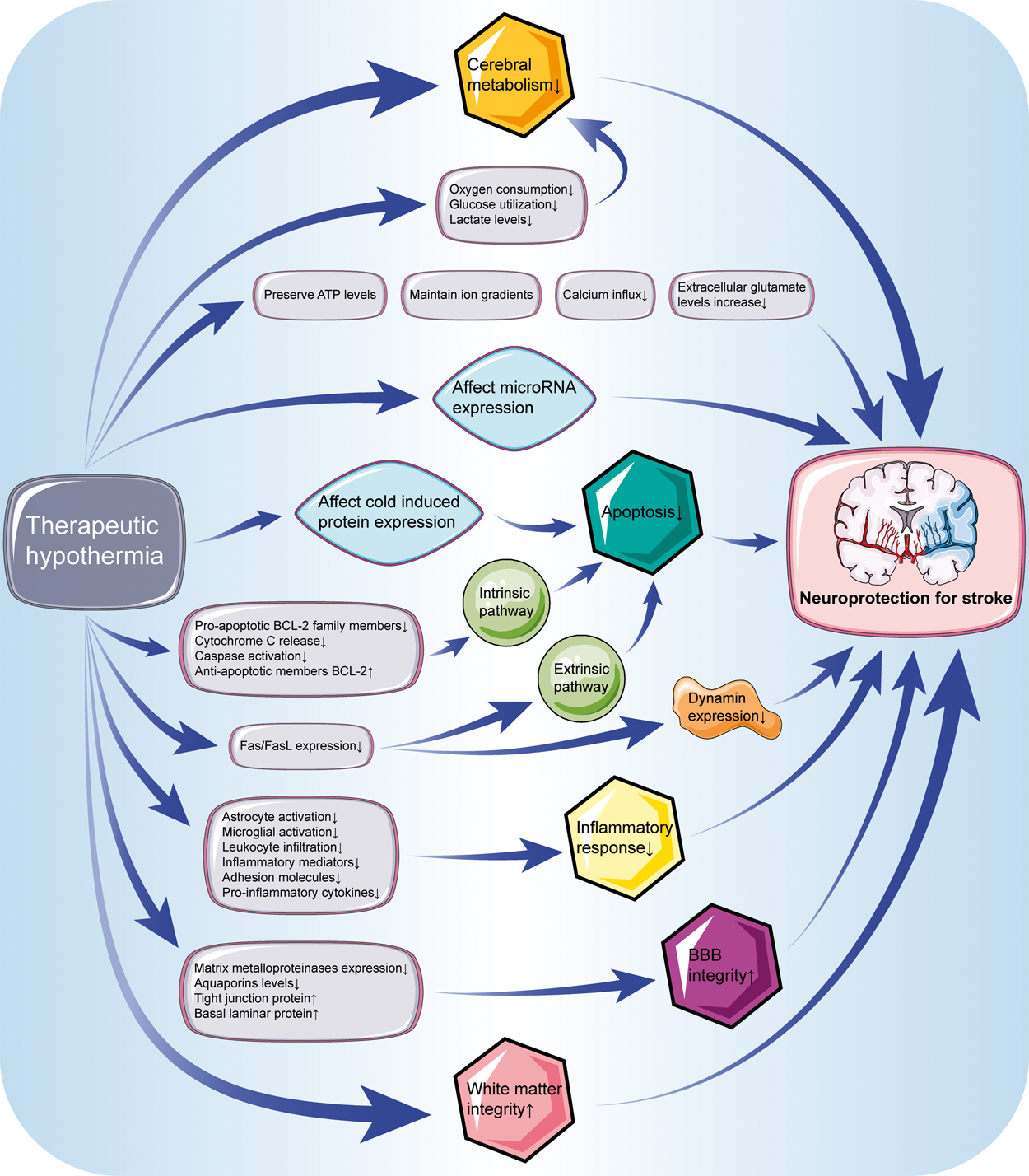
Neuroprotective mechanisms of hypothermia on stroke. Hypothermia can affect multiple aspects of stroke physiology in different stages. Neuroprotective mechanisms of hypothermia on stroke include decreased cerebral metabolism, inhibition of apoptosis, anti-inflammatory effects, maintenance the integrity of blood-brain barrier and white matter, etc. Abbreviations: ATP: adenosine triphosphate; BBB: blood-brain barrier.
Reducing cerebral metabolic rate is believed the major mechanism for hypothermia to exert neuroprotective effects, as cerebral metabolic rate decreases by 7–10% for every degree Celsius reduction in body temperature. 19 Reduction in oxygen consumption, glucose utilization, and lactate levels indirectly reflect the decrease of cerebral metabolism in hypothermia.20,21 Meanwhile, the accumulation or release of excitatory amino acids plays an important role in neuronal damage. 22 Hypothermia can maintain ion gradients by preserving constant ATP levels, thereby avoiding calcium influx and subsequent increase in extracellular glutamate levels. 23 Additionally, microRNA expression is also closely related to brain injury and has become a hotspot of current research. 24 Several experimental studies based on cerebral ischemia or traumatic brain injury (TBI) model have found that hypothermia also affects the microRNA expression, such as miR-15b and miR-874.25,26
Cold induced protein is another factor that may affect the neuroprotective effects of hypothermia. Cold-inducible RNA-binding protein (CIRBP) and cold-inducible RNA-binding motif protein 3 (RBM3) are the major cold induced proteins studied. Hypothermia activates the expression of CIRBP, 27 which could exert protective effects against oxidative stress and apoptosis.28,29 Overexpression of RBM3 could rescue neuronal cells from forced apoptosis, while the neuroprotection of mild hypothermia are diminished after blocking RBM3 expression.30,31 More work is needed to better understand the mechanisms of cold induced protein in hypothermia.
Apoptosis is extensively affected by hypothermia. Hypothermia is found to inhibit neuronal apoptosis in two pathways, namely, mitochondrial based intrinsic pathway and receptor-mediated extrinsic pathway. Hypothermia could suppress pro-apoptotic BCL-2 family members, cytochrome c release and caspase activation, while increasing anti-apoptotic members BCL-2 in intrinsic pathway.32–34 Apoptotic death in extrinsic pathway can be triggered by Fas/FasL. Hypothermia has an inhibitory effect on the expression of Fas/FasL. 35 The inhibition of Fas further decreases dynamin expression, which is also confirmed to produce neuroprotective effects. 36
It is widely recognized that there is immune surveillance in the brain and a strong immune response would be induced after ischemia. Both pre-clinical and clinical studies indicate that the inflammatory response mediated by inflammatory cells and cytokines aggravate the injury.37–39 Therapeutic hypothermia has anti-inflammatory effects by suppressing astrocyte and microglial activation as well as leukocyte infiltration, accompanied reduced levels of inflammatory mediators, adhesion molecules and pro-inflammatory cytokines.40–42 At present, there is sufficient evidence that hypothermia acts on multiple levels of inflammatory response in brain ischemia to achieve neuroprotection, although detailed mechanism is not yet fully elucidated.
Maintaining the integrity of blood-brain barrier (BBB) is critical for brain homeostasis. AIS structurally and functionally destroys the neurovascular units, leading to cerebral edema and hemorrhage. 15 This destruction occurs at an early stage of ischemia, and BBB rupture may be the initiating factor rather than the consequence of parenchymal cell damage. 43 Subsequent studies based on vascular recanalization have shown that therapeutic hypothermia can decrease the expression of matrix metalloproteinases, a group of protease responsible for the degradation of tight junction proteins.44,45 A recent study further suggests that hypothermia attenuates the increase in aquaporins levels and reverses the breakdown in tight junction protein and basal laminar protein, clearly indicating a beneficial effect of hypothermia on the integrity of the BBB. 46 In addition, hypothermia provides protection that lasts from hyper-acute to chronic phase of stroke. 17
Recently, some pre-clinical studies have further investigated the key parameters of hypothermic therapy. Lyden and colleagues find that shorter therapeutic hypothermia durations may be superior to longer in both cell culture and rodent stroke models, although longer cooling durations may overcome the moderate delays to cooling initiation. Furthermore, both 33 and 35°C hypothermia effectively preserve all cell types (neuron, astrocyte, and endothelia cells). 16 Our group shows that a relatively fast hypothermia will exert a long-term neural benefit on white matter preservation.17,18 Therefore, evidence from the latest pre-clinical findings suggests that, a fast (starting time), mild (depth), and not too long (duration) therapeutic hypothermia will provide long-term neuroprotective effects.
Therapeutic hypothermia for AIS
Recombinant tissue plasminogen activator has been approved to treat AIS for approximately 25 years. 47 Besides, the efficacy and safety of mechanical thrombectomy in AIS patients with large vessel occlusion have also been confirmed by several large clinical trials. 3 Nevertheless, the latest data suggest that stroke still accounts for almost 5% of all disability-adjusted life years as well as 10% of all deaths worldwide.48,49 Numerous studies have proved the efficacy of therapeutic hypothermia on AIS in the laboratory settings, but how to successfully translate these exciting discoveries from basic research into clinical treatment for AIS patients is still a big challenge. What is the great difference between bench and bedside? Why does therapeutic hypothermia not benefit AIS patients? How to conduct a rapid and simple hypothermic therapy in the era of reperfusion? These questions have to be answered for translational purposes.
As shown in Table 1, we summarize the completed and ongoing clinical studies on therapeutic hypothermia in AIS patients. After in-depth analysis of these studies, we try to elaborate the recent achievements and pitfalls of therapeutic hypothermia in the field of AIS. First, due to the technical limitations, patients enrolled in early clinical studies of hypothermia were unable to achieve vascular recanalization. However, with the emerging concept of vascular recanalization, recent clinical studies have placed more emphasis on opening the occluded arteries. Second, most studies have a significant delay in the onset of hypothermia, which may miss the most optimal time window to protect the penumbra, and can hardly be rescued by extending the hypothermic duration or lowering the temperature. Third, previous studies tend to use surface cooling or endovascular cooling as the method of therapeutic hypothermia, making it difficult to avoid the various side effects that follow. However, in recent years, more precisely targeted hypothermic methods have emerged, which reduces the risk of side effects. Taken together the above analysis, we postulate that the successful clinical translation of hypothermic therapy in AIS requires good control of the following three aspects: (1) recanalization states, (2) key parameters for hypothermia, and (3) optimized hypothermic method.
Clinical studies on therapeutic hypothermia in acute ischemic stroke patients.
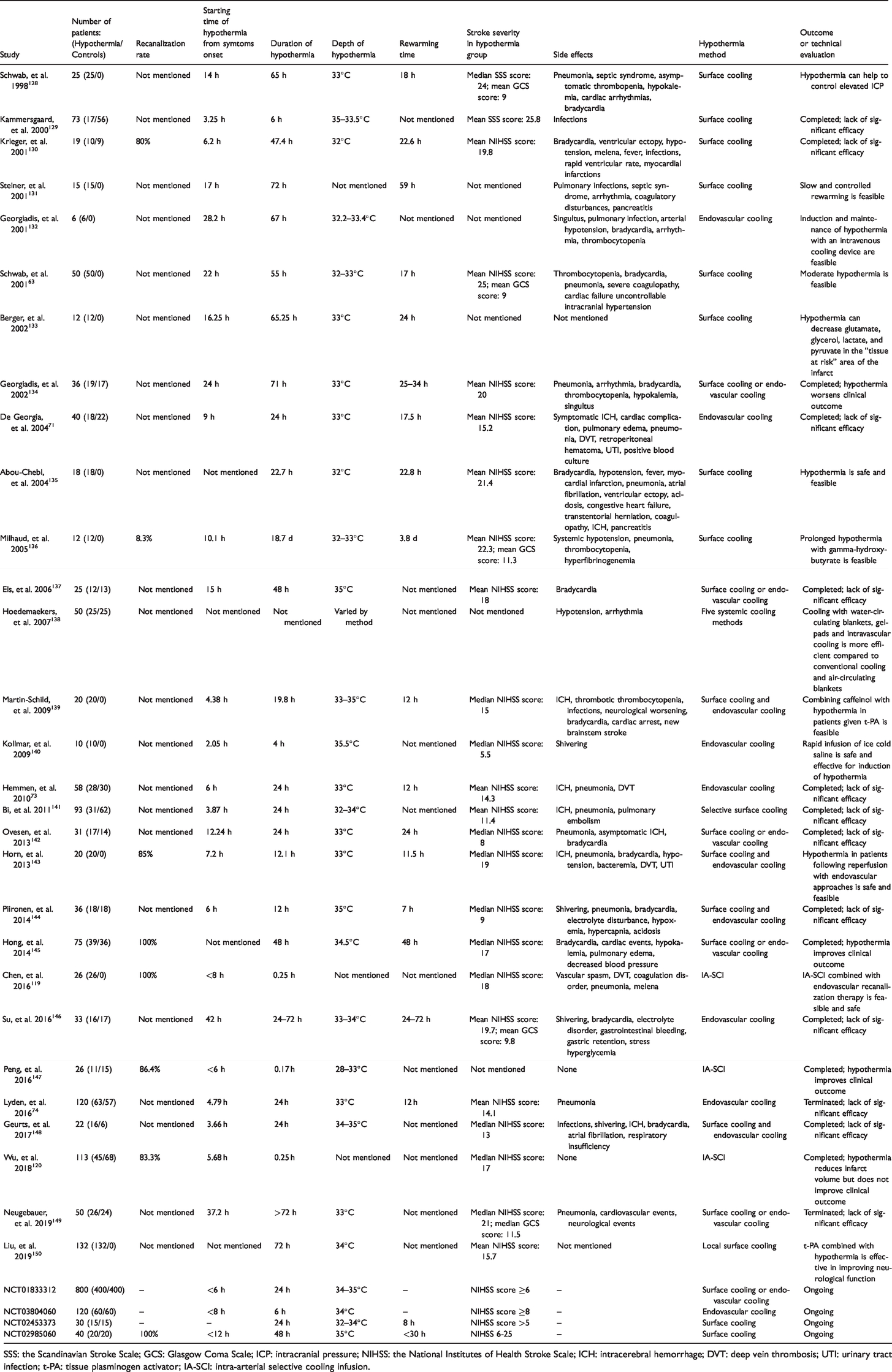
SSS: the Scandinavian Stroke Scale; GCS: Glasgow Coma Scale; ICP: intracranial pressure; NIHSS: the National Institutes of Health Stroke Scale; ICH: intracerebral hemorrhage; DVT: deep vein thrombosis; UTI: urinary tract infection; t-PA: tissue plasminogen activator; IA-SCI: intra-arterial selective cooling infusion.
Hypothermia based on recanalization
The most predominant mismatch between bench and bedside is that, transient middle cerebral occlusion models are used in pre-clinical studies but AIS patients are composed of different recanalization states in clinical studies. To investigate the effects of vascular recanalization on neuroprotection of hypothermia, we conducted a systematic literature search and analyzed all the pre-clinical studies regarding hypothermia for cerebral ischemia. Literature published in English between 1 January 1990 and 1 May 2019 in PubMed and Web of Science databases were assessed. The search strategies entered into PubMed database were: ((((((hypothermia OR cooling OR lower temperature)) AND (brain OR cerebral OR intracranial OR cranial OR intracerebral)) AND (ischemic OR ischemia OR infarct OR infarction)) NOT (hemorrhage OR hemorrhages OR intracranial hemorrhage OR intracranial hemorrhages OR cerebral hemorrhage OR cerebral hemorrhages OR bleeding)) AND (“1990/01/01”[Date - Publication]: “2019/05/01”[Date - Publication])) AND English[Language]. The search terms for the Web of Science database were: (TS = (brain OR cerebral OR intracranial OR cranial OR intracerebral) AND TS = (ischemic OR ischemia OR infarct OR infarction) AND TS = (hypothermia OR cooling OR lower temperature)) NOT TS = (hemorrhage OR hemorrhages OR intracranial hemorrhage OR intracranial hemorrhages OR cerebral hemorrhage OR cerebral hemorrhages OR bleeding) AND LANGUAGE: (English) AND DOCUMENT TYPES: (Article). These search strategies identified a total of 3540 articles in PubMed and 3946 articles in Web of Science. We further removed 1898 identical publications from the two databases and screened the remaining 5588 studies. Review articles or meta-analyses, clinical studies, in vitro studies, studies regarding hypoxic ischemic encephalopathy or global cerebral ischemia were excluded. Finally, we selected 198 articles for in-depth analysis. In these 198 studies, 152 studies used a vascular recanalization model, of which 143 studies (94.1%) showed that therapeutic hypothermia was effective in the treatment of AIS. However, other 46 studies used a vascular occlusion model without recanalization, of which only 32 studies (69.6%) showed that hypothermia was effective. The difference was statistically significant (P < 0.05) according to the chi-square test. Figure 2 shows the selection of literature and the distribution of publication time.
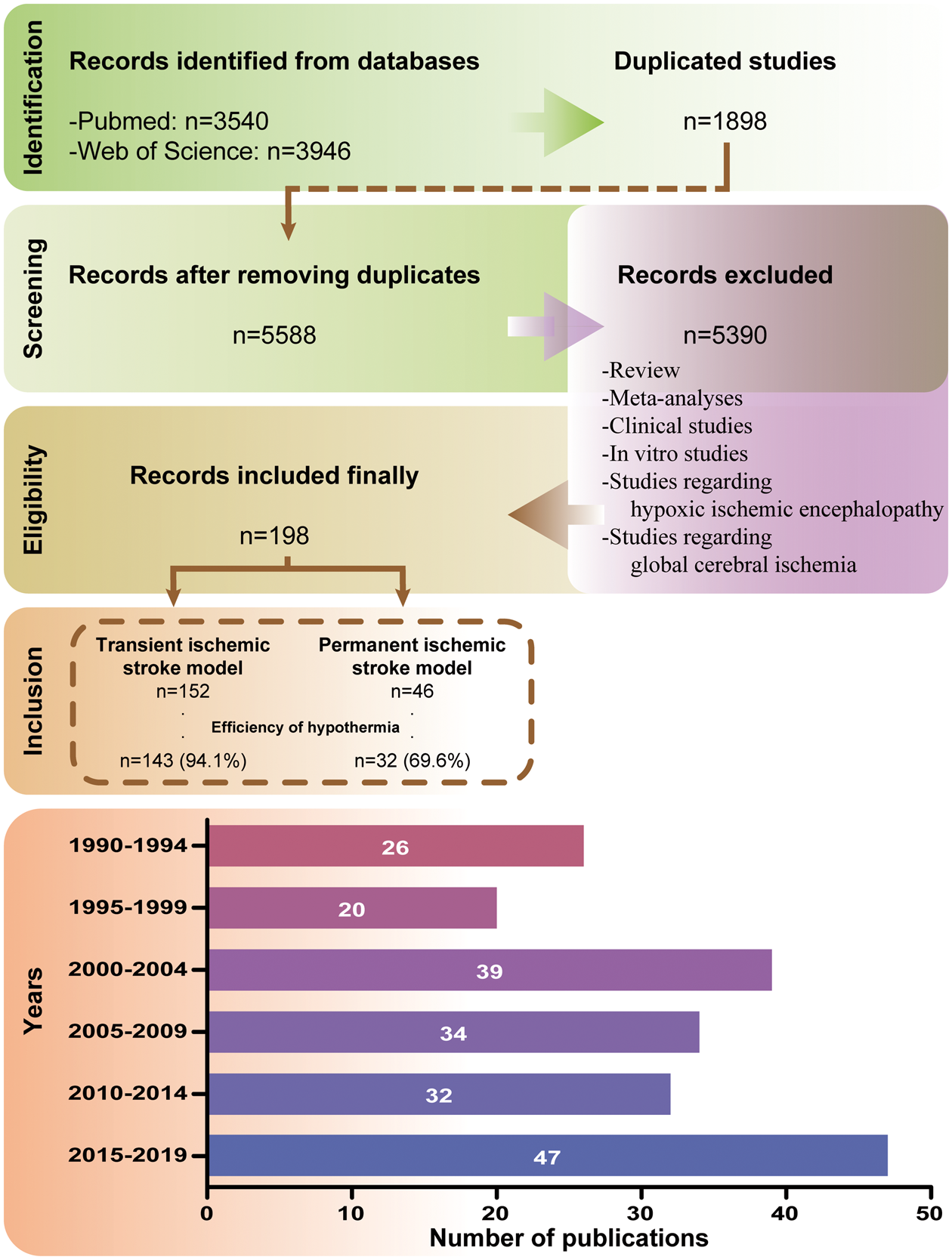
Study data. A total of 5588 pre-clinical studies on PubMed and Web of Science were screened. After excluding the studies that did not meet the requirements, we conducted an in-depth analysis of 198 studies. Since 1990, the overall number of studies has been increasing.
The above statistical results according to in-depth analysis indicate that hypothermia on top of recanalization is more likely to exhibit neuroprotective effects than that without recanalization in pre-clinical studies. In fact, recanalization has always been the first priority in AIS treatment. It can significantly prevent the progression of infarction. 50 A meta-analysis showed that recanalization was associated with a 343% increased possibility of attaining good functional outcome and a 76% decreased risk of death compared to those patients without recanalization. 51 However, because the recanalization rate of intravenous thrombolysis is low and mechanical thrombectomy has just been put into clinical application in recent years, it was not easy to achieve recanalization in the past. In reality, a lot of participants in past clinical trials related to neuroprotection did not experience recanalization. It was speculated that in the Stroke-Acute Ischemic NXY Treatment II trial, nearly 80% of subjects received study agent without recanalization. 52 Lack of recanalization may be responsible for the failure of clinical translation in previous neuroprotective studies. With the increasing maturity of vascular recanalization therapy, we believe that the clinical translation of hypothermic neuroprotection has also ushered in an unprecedented opportunity.
Key parameters of hypothermic therapy
Starting time of hypothermia
Another mismatch between pre-clinical and clinical studies is the starting time of hypothermia. Considering the possibility of further progression of infarct and the fact that ischemic penumbra brain tissue becomes more and more limited over time, therapeutic hypothermia should be initiated as early as possible in theory. This view was also supported by the findings from experimental models that hypothermia at or during the onset of ischemia maximized the neuroprotective effects. 53 However, time delay exists between symptoms onset and arrival at the hospital. In addition, after patient arrival, it is not feasible to implement hypothermia before the definite diagnosis is made. What is more, different cooling rates of different hypothermia methods could vary the time required to reach the target temperature. Therefore, practically, reaching the target brain temperature as fast as possible would be our purpose as soon as the AIS diagnosis is made.
Duration of hypothermia
The duration of hypothermia is also directly related to the ultimate neuroprotective effects. Although the optimal duration of hypothermia is not known, given that the cascade of damaging events after stroke can last for hours or even days, 54 we should enable the hypothermia to cover these harmful processes for better neuroprotection. Findings from experimental studies have suggested that longer duration of hypothermia might produce better neuroprotective effects.53,55 However, we should be alert that prolonged hypothermic therapy also increases the risk of side effects such as infections, especially in humans, which may weaken or even offset the neuroprotective effects from hypothermia. A balance should be carefully titrated to maximize the efficacy while minimize the side effects. According to the successful experience of hypothermic therapy in patients with cardiac arrest, it is appropriate to maintain the duration of hypothermia for 12–24 h.9,10
Depth of hypothermia
The depth of hypothermia is another key parameter in hypothermic therapy. The effects between mild hypothermia and moderate hypothermia were compared in an early study in rats. 56 Opposite from most people’s subjective perception, animals treated with mild hypothermia showed better tolerance and recovery. 56 In concert, a subsequent study also found that, in the range of 32–37°C, temperature of 34°C provided the best neuroprotective effects on infarct size, edema, and functional prognosis. 57 On the contrary, a meta-analysis based on a large number of pre-clinical studies found a significant relationship between the depth of hypothermia and the reduction in infarct volume, which means that the lower the hypothermic depth, the more pronounced the effect. 58 But this conclusion is not supported by another recent meta-analysis which did not find a superiority of a certain hypothermic depth over others. 59
As the pioneers applying hypothermic therapy into clinical applications, cardiologists have also carried out a thorough discussion on this topic. The targeted temperature management (TTM) trial was designed to investigate the most optimal target temperature which leads to the best outcome in patients with cardiac arrest. 60 Although the results of TTM trial did not find any difference between 33°C and 36°C on prognosis, there is no doubt that both degrees of hypothermia are beneficial. 60 Furthermore, a more recent trial, as well as two pioneer trials, has demonstrated the positive effects of 33°C on cardiac arrest patients, regardless of whether the cardiac arrest was in a shockable rhythm or non-shockable rhythm.9,10,61 Viewed from another perspective, in light of the complexity of clinical application, moderate to severe hypothermia treatment will undoubtedly cause discomfort in conscious patients. In this case, mechanical ventilation and sedation are always needed. These approaches will further increase the risk of pneumonia or other side effects in turn. Based on the above considerations combined with evidence from cardiology, mild hypothermia with a temperature set at 33–34°C may be appropriate.
Rewarming speed
As the final stage of hypothermic therapy, rewarming also determines the outcome of patients. The rewarming speed is the most critical parameter in this stage. At the beginning of hypothermia, we can apply chilled saline infusion to quickly reach the target temperature. However, we cannot induce rapid rewarming by the opposite approach, i.e. infusing high temperature saline, because this may damage the red blood cells or coagulation elements. 62 Furthermore, rapid rewarming may induce a rebound increase in intracranial pressure, leading to brain herniation and even death, which has been observed in a clinical study on massive hemispheric infarction. 63 In the latest international multicenter prospective randomized clinical trial, Prophylactic Hypothermia Trial to Lessen Traumatic Brain Injury (POLAR), which was designed to determine the neuroprotective effects of hypothermia on TBI patients, the rewarming stage was performed under the intracranial pressure monitoring. 64 For patients with intracranial pressure below 20 mmHg, the rewarming speed was set at 0.25°C per hour to minimize the risk of intracranial pressure rebound. 64 Other drawbacks of rapid rewarming, including hyperkalemia induced by redistribution of potassium, have also been found in TBI patients and may lead to malignant arrhythmias. 65 Therefore, it is critical to maintain a slow rewarming speed and closely monitor patients during rewarming. A 12-hour slow rewarming may be appropriate in clinical practice.
Stroke severity
The selection of the suitable enrolled patients determines whether a clinical study will ultimately yield positive result. Stroke severity is among the inclusion criteria in many clinical studies, which also predicts the prognosis of patients. An observational study indicated that, up to 82.9% of minor stroke patients (median National Institutes of Health Stroke Scale score of 3) were able to achieve self-care status at discharge, even if they did not receive intravenous thrombolysis. 66 This suggests that milder cases can recover well even without treatment; in other words, effective treatment is also difficult to further improve the prognosis of these patients. Therefore, we suppose that hypothermic therapy can only demonstrate its neuroprotective effects when used in moderate to severe cases.
Side effects
The mismatch also exists in side effects between experimental models and clinical patients. When hypothermia shows its powerful neuroprotective effects in pre-clinical studies, it often brings many side effects in clinical studies (Figure 3), which may directly determine whether the AIS patients can ultimately benefit from hypothermia.
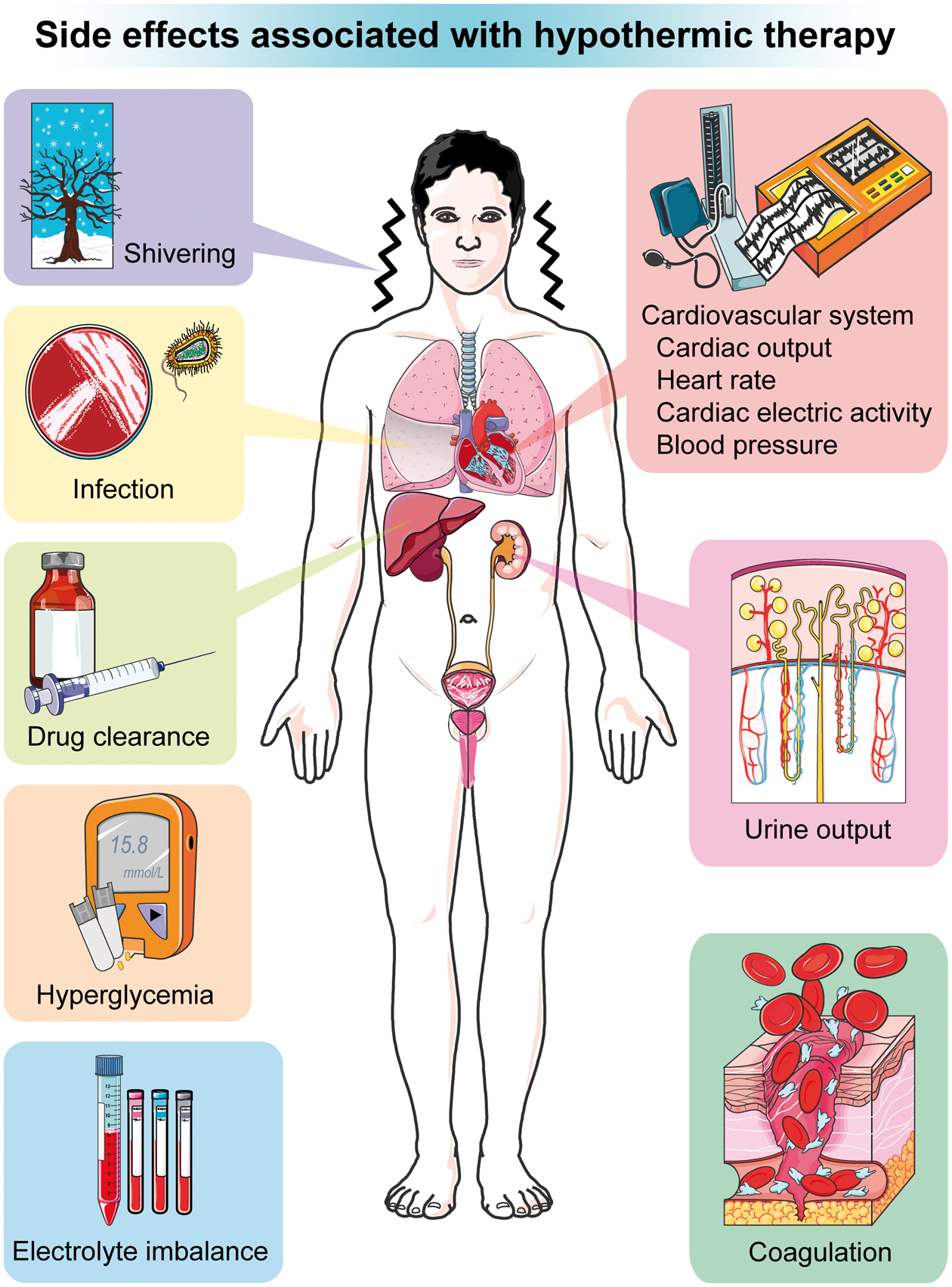
Side effects associated with hypothermic therapy. Side effects associated with hypothermic therapy include shivering, infection, effects on drug clearance, hyperglycemia, electrolyte imbalance, effects on cardiovascular system, changes in urine output, and effects on coagulation.
Shivering is a common side effect in the treatment of hypothermia, which is actually a physiological reaction of human body to cold. When the body temperature is detected to drop by more than 0.2°C, the muscles begin to oscillate rapidly, generating heat against the cold, thus offsetting the effort to reach the target temperature. 67 This physiological reaction also increases metabolic rate, oxygen consumption, heart rate, and respiratory burden, which is obviously unfavorable for patients with brain injuries. 19 Although we can eliminate shivering by sedation combined with mechanical ventilation, this approach per se brings side effects and makes frequent neurologic examinations undoable due to altered mental state, thus is somewhat excessive. 68 Some medications can also put down shivering by lowering the temperature threshold to initiate shivering, without the need for sedation. Meperidine and buspirone are two typical drugs used to suppress shivering. Meperidine is an opiate analgesic. Intravenous infusion of low dose meperidine can rapidly suppress shivering with little risk. 69 Buspirone is an oral anxiolytic that produce a synergistic effect when combined with meperidine. The combination of these two drugs can reduce the shivering threshold of volunteers to 33.4°C while causing little sedation or respiratory adverse effects. 70 It has now become a standard method for shivering control in most clinical trials.71–74 Other drugs with anti-shivering effects include magnesium, clonidine, ketanserin, tramadol, etc. 19
Infection is a major complication after stroke. AIS patients have a higher risk of pneumonia, which may be associated with some unique characteristics such as impaired airway reflexes, pharyngeal muscle relaxation, and dysphagia after stroke. 75 However, hypothermic therapy is thought to further increase the risk of infection, possibly due to immunosuppression. Every coin has two sides, and the anti-inflammatory effects that protect the central nervous system might be detrimental to the body as a whole. In addition, insulin resistance and hyperglycemia induced by hypothermia may also contribute to the elevated risk of infection. 19 Some clinical studies have also confirmed the fact that patients in the hypothermia group have a higher rate of infection. 67 According to a randomized multicenter trial of hypothermia and intravenous thrombolysis in AIS patients, although there was no impact on overall mortality, the incidence of pneumonia as high as 50% in the hypothermia group still raised doubts about the safety of hypothermic therapy. 73 Later study modified the hypothermia protocol in the hope of reducing the incidence of pneumonia, but failed to do so. 74
The effect of hypothermia on coagulation is another safety concern, because to some extent, we are most concerned about the hemorrhagic transformation in AIS patients. Although experimental reports have shown that hypothermia can decrease platelet aggregation and prolong coagulation time, 76 serious bleeding events associated with hypothermia were not observed in clinical practice.71–74 Another concern is whether hypothermia impacts the efficacy of intravenous thrombolysis, since the thrombolytic activity of tissue plasminogen activator is temperature dependent in vitro. 77 It was speculated that mild hypothermia reduced thrombolytic activity by 2–4%. 77 However, practically, the effect of tissue plasminogen activator can hardly overlap with hypothermia, owing to its short half-life in circulation and the significant delay in reaching target temperature in therapeutic hypothermia. As a result, such concerns may be unnecessary.
Hypothermic therapy also has effects on the cardiovascular system, including cardiac output, heart rate, cardiac electric activity, and blood pressure. 19 These effects appear to be related to the depth of hypothermia and do not require active intervention, as these effects tend to dissipate spontaneously when body temperature returns to normal. 78 Similarly, there is no evidence that the effects of hypothermia on the cardiovascular system will lead to poor outcomes in patients.
Other side effects caused by hypothermic therapy, which are often discussed, include changes in urine output, electrolyte imbalance, hyperglycemia, and effects on drug clearance. The reason for the changes in urine output during hypothermia is that hypothermia stimulates vasoconstriction and increases venous return, thereby promoting the release of atrial natriuretic peptide and lowering the level of antidiuretic hormone. 67 Changes in urine output will inevitably lead to changes in electrolyte levels such as sodium, potassium, calcium, magnesium, phosphate, etc., which may induce cardiac arrhythmias. 79 The cause of hyperglycemia is related to insulin resistance and the reduction of insulin secretion induced by hypothermia. 19 Considering that hyperglycemia may adversely affect outcome in critically ill patients, this situation should be taken seriously. Therapeutic hypothermia can also hinder the drug clearance, and the underlying mechanisms may be related to the hyperthermia-mediated reduction of hepatic enzymes activity, hepatic blood perfusion and bile secretion, alteration of drug distribution volume, and disturbance of renal tubular function. 19
Overall, there are many side effects associated with hypothermic therapy. Although most of them may not actually affect the overall prognosis of the patient, we still cannot take them lightly. Patients who are treated with hypothermia should preferably be arranged to stay in the intensive care unit, in order to closely monitor the condition and take timely remedies. On the other hand, we should try to avoid the side effects of hypothermia as well as minimize their threat when they happen. Developing new hypothermia strategies might be a good option.
Novel hypothermic therapies
Traditional hypothermic therapies generally refer to endovascular cooling and surface cooling. However, these methods are often criticized for their complex steps or unsatisfactory cooling effect. In recent years, several novel hypothermic therapies including pharmacologic cooling and selective brain cooling have emerged, serving as promising alternatives to traditional ones.
Pharmacologic cooling
Pharmacologic cooling is attractive because of its ease of use. There are currently eight classes of hypothermia-inducing drugs available for cooling, including cannabinoid, opioid receptor agonists, transient receptor potential vanilloid 1 agonists, neurotensin analogs, thyroxine derivatives, dopamine receptor activators, hypothermia-inducing gases, and adenosine derivatives. 67 The mechanism of drug-induced hypothermia is mainly related to its effect on the thermoregulation center in the hypothalamus. 20 Acting directly on the thermoregulation center can also reduce shivering and vasoconstriction, thus seemingly minimizing the side effects. However, we need to know that, unlike physical cooling, pharmacologic cooling has not only the cooling effect, but also the pharmacological effects, which may further affect cardiac electric activity, blood glucose, respiration, and consciousness, and may be detrimental to AIS patients. 67 Therefore, thorough and detailed safety assessment is necessary for pharmacologic cooling before clinical application.
Selective brain cooling
Actually, most of the side effects associated with hypothermia mentioned above are related to systemic hypothermia. However, for patients with central nervous system injury, the key to exerting the neuroprotective effects of hypothermia is to cool the brain rather than the whole body. To some extent, the side effects of systemic hypothermia have hindered the clinical translation of hypothermia in the field of stroke. Therefore, our group put forward a novel hypothermic idea “a relatively cool brain and normal body”. After decades of development, a variety of selective brain cooling methods have emerged. 80 The cooling target of these methods is limited to the brain, thus minimizing the side effects of systemic hypothermia. Selective brain cooling can be subdivided into non-invasive cooling and invasive cooling. 80 The former mainly includes surface cooling and nasopharyngeal cooling and, as expected, is less effective in cooling the brain parenchyma. Conversely, invasive cooling allows for easier heat exchange in the brain, thus showing a promising prospect. In this section, we introduce several methods of invasive selective hypothermia (Figure 4), including epidural cooling, subdural cooling, subarachnoid cooling, retrograde jugular venous cooling as well as IA-SCI, and expound the process of clinical translation of IA-SCI.
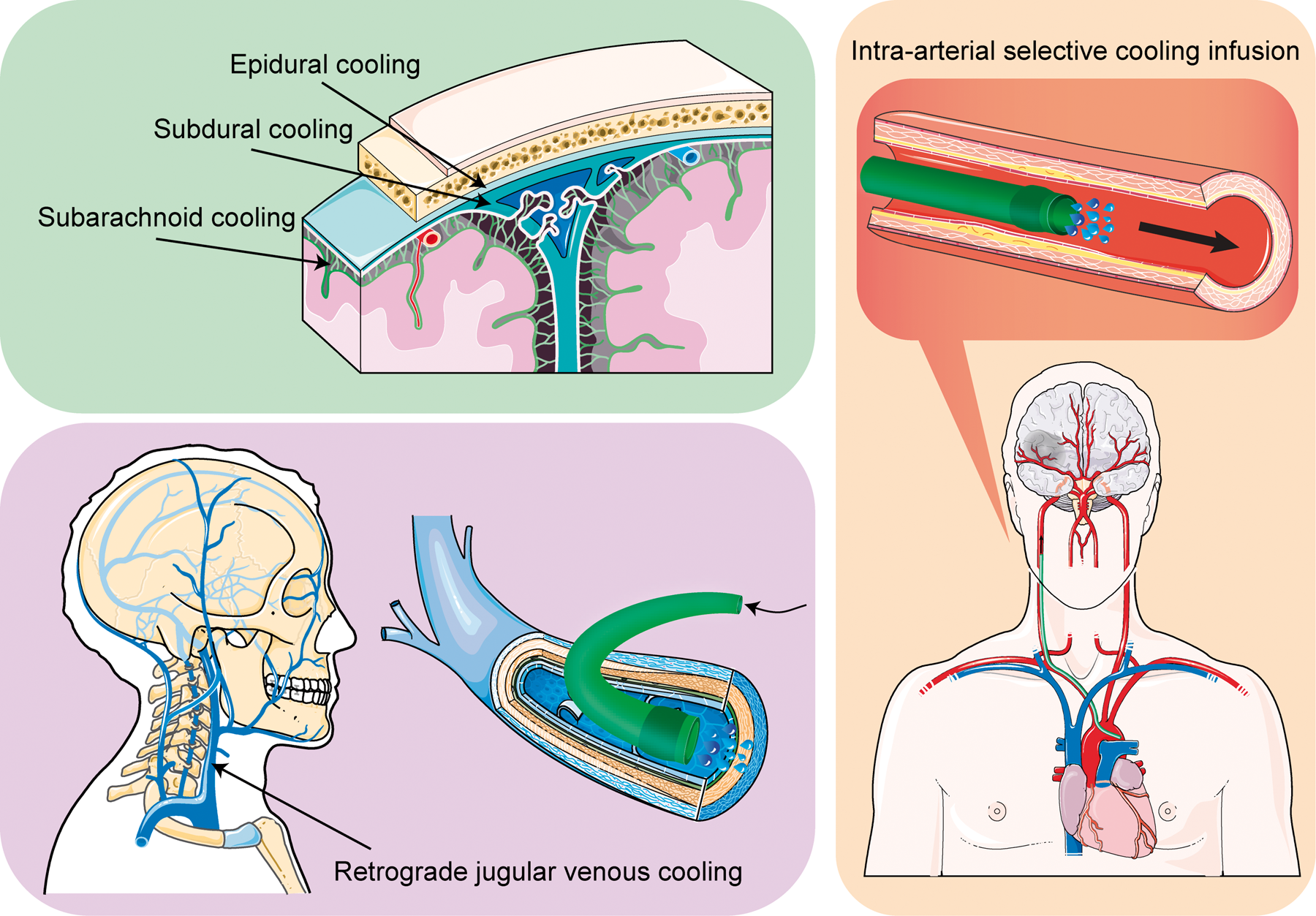
Methods of invasive selective brain cooling. Invasive selective brain cooling includes epidural cooling, subdural cooling, subarachnoid cooling, retrograde jugular venous cooling, and intra-arterial selective cooling infusion.
Epidural cooling
Epidural cooling is achieved by first exposing the dura mater by drilling holes, then irrigating the hypothermic fluid continuously inward. 81 A study using a porcine model showed that by perfusing 4°C saline at a rate of 720 ml per hour, the local brain surface temperature can be lowered to 17.3–21.6°C in 1 min and the deep brain temperature can be lowered to 27.2–29.7°C in 5 min without causing any changes in rectal temperature. 81 Further study based on a model of permanent middle cerebral artery occlusion demonstrated histologically that epidural cooling significantly reduced the infarct volume after the onset of ischemia. 82 Another cooling device called ChillerPad, which is applied to the dura, has also shown a powerful cooling effect in non-human primate TBI model. 83 By comparing the efficacy of epidural cooling with systemic hypothermia or exploring the neuroprotective effects of the combination of the two, the value of epidural cooling was proved.84,85 However, additional operations may be required when applied in AIS patients.
Subdural cooling
An early study compared the cooling effect of subdural cooling and epidural cooling, and found that subdural cooling could reduce the temperature of cerebrospinal fluid to a lower level, confirming the effectiveness of this cooling approach. 86 Another study using a cat model of vascular recanalization also evaluated the efficacy of subdural cooling, and found that this cooling method could quickly decrease the brain surface temperature to 33°C without changing the rectal temperature. In addition, it also improved the amplitudes of somatosensory evoked potential and diminished the water content in the gray matter. 87 Similar to epidural cooling, additional surgical procedures may limit its clinical use.
Subarachnoid cooling
There are few studies regarding subarachnoid cooling. Many years ago, 23 patients with intractable epilepsy received subarachnoid catheter irrigation of cold saline. The result showed that the local brain surface temperature could be reduced to at least 27°C through this cooling method, which confirmed its feasibility and efficacy. 88
Retrograde jugular venous cooling
Early studies on retrograde jugular venous cooling were based on rodent models of heatstroke. It was found that direct retrograde hypothermic perfusion via the jugular vein could significantly reduce brain temperature and neuronal damage score, maintain cerebral blood flow, and prolong survival time, without significantly affecting body temperature in the meantime.89,90 A basic research using TBI model also found that retrograde jugular venous cooling can alleviate motor deficits, brain nitrostative, and oxidative damage. 91 Additionally, retrograde jugular venous cooling has yielded positive results in large animal models. This cooling method can also rapidly induce decreased brain temperature in swine.92,93 As for stroke, a recent study compared the efficacy of retrograde jugular venous cooling with that of intra-carotid cooling infusion in rodents. Retrograde hypothermic perfusion via jugular vein exerted a similar hypothermia and neuroprotection against ischemic stroke compared with intra-carotid cooling infusion, which was characterized by rapid induction of brain temperature decline, significant reduction of infarct volume, neurological deficits and brain edema, protection of BBB and anti-apoptosis. 94 However, this cooling method is contradicted to the normal physiological status, so its safety needs to be verified prior to application in patients.
IA-SCI
Accounting 2.5% of the total body weight, human brain receives 15% of cardiac output, which means that robust relatively high temperature blood from the systemic circulation continues to warm the brain, thus hindering the effects of most selective brain cooling therapies. 95 However, as a novel hypothermic therapy, IA-SCI utilizes the characteristic of high blood flow to the brain to directly perfuse the hypothermic fluid into the cerebral arteries for rapid selective cooling, 95 which can be said to turn the disadvantage into advantage. Theoretical models showed that IA-SCI achieved a local hypothermia of brain tissue 18 to 42 times faster than the surface cooling, and 10 to 20 times faster than the systemic transvenous cooling.96,97 Moreover, in such an era of vascular recanalization, this accurate cooling method can be well integrated with mechanical thrombectomy, plus it also avoids the side effects caused by systemic hypothermia, therefore, the clinical application prospects are promising.
IA-SCI in rodent animal models of stroke
In 2002, Ding and colleagues first proposed the concept of prereperfusion saline infusion into the ischemic territory.98,99 They used a novel intraluminal hollow filament to induce a rat model of middle cerebral artery occlusion and perfused isotonic saline at body temperature or room temperature into the ischemic area through the filament before reperfusion. The results showed that this therapy significantly increased cerebral blood flow up to 48 h after reperfusion, reduced infarct volume, as well as improved short- and long-term neurological prognosis.98,99 Subsequent studies indicated that the mechanisms of this neuroprotective therapy was related to the reduction of inflammatory cytokines, adhesion molecules, brain edema, and protection of BBB.100,101
On the basis of this model, studies on IA-SCI began. In 2004, Ding and colleagues found that local infusion of cold saline into ischemic territory could induce regional brain cooling and neuroprotection.102–104 It was reported that the temperature in cerebral cortex and striatum could be rapidly reduced to 33.4 and 33.9°C, respectively, by perfusing 6 ml of saline at 20°C into the hollow filament for 10 min before reperfusion.102,104 This highly effective hypothermic therapy dramatically reduced infarct volume by up to 90%, greatly improved long-term motor behavior as well as extended the therapeutic time window of reperfusion from 2 to 2.5 h in ischemic rats.102,103,105 In 2012, Ji and colleagues studied postreperfused IA-SCI.106,107 By lowering the brain temperature through IA-SCI, 106 they investigated the effect of the starting time of hypothermia on neuroprotection. They reported that the neuroprotective effects declined with the prolongation of the time from reperfusion to the start of IA-SCI. 107
IA-SCI can also be used in combination with other neuroprotective agents to provide more powerful neuroprotective effects. In the AIS rats, compared with the IA-SCI of cold saline alone, IA-SCI of either magnesium sulfate 108 or human albumin 109 can further reduce the infarct volume and improve the neurological outcome, reflecting the positive synergy of IA-SCI with other neuroprotective agents.
IA-SCI in large animal models of stroke
Given the great differences between rodents and human species, it would be inappropriate to apply these positive results from rodent studies directly to clinical practice. 110 In order to exclude the effect of species differences on experimental results, Stroke Therapy Academic Industry Roundtable (STAIR) recommended that neuroprotective methods should be thoroughly evaluated in large animal models before being used clinically.111,112 As a result, IA-SCI research in large animal models has begun.
The safety, feasibility and efficacy of IA-SCI were validated in healthy rhesus monkey by infusion of 100 ml of ice-cold (0°C) lactated Ringer's solution into the proximal middle cerebral artery within 20 min via a microcatheter. 113 Mild hypothermia in brain tissue was induced within 10 min, and the lowest cerebral temperature could be reduced to 33.9°C. During the procedure, the vital signs of experimental animals were stable, and no adverse reaction such as brain edema and vasospasm occurred. 113 Similar results were obtained in a study using canines, showing that the local brain temperature could be reduced to 33.6°C by infusing Ringer’s solution precooled to approximately 6.5°C at a rate of 3 ml/kg/min into the right common carotid artery for 30 min through an angiographic catheter. 114 However, due to the large volume of total fluid infused, the rectal temperature, hemoglobin, hematocrit, and cardiac function showed different degrees of change. 114 Another study explored the application of IA-SCI in sheep models. 115 IA-SCI with a new balloon cooling catheter system introduced into a common carotid artery could rapidly induce mild hypothermia in ipsilateral cerebral hemisphere. This device, if combined with mechanical thrombectomy, may benefit stroke patients with large vessel occlusion. 115
IA-SCI was also proved effective in large animal AIS models.116,117 In 2016, Mattingly and colleagues developed a model of middle cerebral artery occlusion in swine with an aneurysm clip. After 3 h of occlusion, the clip was removed to achieve reperfusion. IA-SCI was induced with a dual lumen balloon occlusion catheter placed in the ipsilateral common carotid artery during reperfusion. This method lowered the cerebral hemisphere temperature to 30°C within 25 min, significantly reducing infarct volume on magnetic resonance imaging. 116 Recently, Caroff and colleagues investigated the cooling effect of IA-SCI using a novel insulated catheter in an ischemia-reperfusion canine model. 117 They found that continuous infusion of hypothermic saline (4.5°C) for 25 minutes at a rate of 22 ml per minute lowered the ipsilateral brain temperature to 31–32°C, and subsequent imaging examinations revealed that this method reduced the final infarct volume down to less than one tenth. 117 The results of IA-SCI in large animals seem equally gratifying. Other studies in large animal stroke models are still ongoing.
IA-SCI in AIS patients
Based on numerous encouraging results from pre-clinical studies, IA-SCI trials in humans have begun. The first study describing the safety and feasibility of IA-SCI in humans appeared in 2010. 118 Eighteen patients with previous diagnosis of vascular malformation received IA-SCI tentatively during the follow-up cerebral angiography. A 10-min infusion of hypothermic saline (4–17°C) at a rate of 33 ml/min into one side of the internal carotid artery could reduce the temperature of jugular venous bulb by 0.84°C without causing serious side effects, opening up a new potential avenue for neuroprotection in AIS patients. 118
Subsequently, the application of IA-SCI in AIS patients was launched. Our group conducted a pilot study of IA-SCI in 26 AIS patients with large vessel occlusion within 8 h after symptom onset. 119 This is a milestone because it put the concepts of prereperfusion and postreperfusion cold saline infusion in rodents into practice in AIS patients for the first time. The ischemic brain tissue can be cooled by at least 2°C by infusing 50 ml of cold isotonic saline (4°C) into the ischemic area via microcatheter at a rate of 10 ml/min prior to thrombectomy, followed by 300 ml of the cold saline for 10 min via the guiding catheter after recanalization. No obvious complications occurred, which confirmed the feasibility and safety of mechanical thrombectomy combined with IA-SCI in AIS patients. 119 Seeing hope in this pilot study, we continued to conduct a prospective cohort study to explore the neuroprotective effects of IA-SCI on AIS patients receiving thrombectomy. 120 Compared to those patients who received thrombectomy alone, thrombectomy plus IA-SCI significantly reduced the infarct volume, and also promoted the proportion of functional independence at 90 days numerically. 120 A similar study (UMIN000018255) and another larger randomized clinical trial (NCT03163459) are underway.
The increasing number of basic and clinical studies of hypothermic therapy in the field of AIS indicates that people are full of confidence in the neuroprotective effects of hypothermia. We believe that, as an effective means of neuroprotection, the days of hypothermic therapy for the treatment of AIS patients will come in the near future.
Perspectives
Selective brain cooling is increasingly considered as a potentially optimized modality to offer benefits for AIS patients, and IA-SCI is believed to be the most promising method of selective brain cooling owning to widely used endovascular therapy in recent years. However, there is still a dilemma concerning the total amount of fluid infused for brain cooling. It is difficult to achieve the target temperature quickly and maintain the duration of hypothermia with a relatively small liquid load, while excessive infusion will increase the burden of the heart and reduce the hematocrit. 117 In theory, it might be possible to cool the autologous arterial blood before selectively infuse the precooled blood into the target areas. This closed-circuit hypothermic system ensures long-term maintenance of hypothermia without increasing the liquid load. Currently, there are some pre-clinical studies investigating the efficacy of this hypothermic system in rodent and primate stroke models.121,122 Most excitingly, in addition to lowering the brain temperature, auto-circulating cold arterial blood also significantly reduced the infarct volume,121,122 reflecting its good clinical translation prospects.
We should pay much attention to significant effects of gender, age, and multiple comorbidities on the efficacy of translational studies. Specifically, animal models in pre-clinical studies are mostly young males, which is quite different from the reality that stroke occurs more frequently in elderly men and women in clinical practice. Gender differences are easily overlooked. Previous clinical study showed that although the stroke presentation and management were similar, women were more often institutionalized and had worse functional outcomes at six months after AIS. 123 In addition, gender differences in brain injury were also found in pre-clinical studies, partially owning to different levels of sex hormones. 124 Age mismatch exists between pre-clinical and clinical studies. Age per se and age associated comorbidities have been found to affect experimental results in both experimental and clinical studies.125,126 Thus, it is preferred to select multiple models to truly simulate the complex clinical situations.
Moreover, we should use both structural and functional outcomes to avoid the mismatch between pre-clinical and clinical studies. Infarct volume is generally used as a prognostic indicator in bench side, while the modified Rankin score at three months is widely used in clinical studies. A larger infarct volume does not equal to a severe neurological deficit. A long-term neurologic evaluation is necessary to improve the credibility and transferability of a study. Therefore, the clinical translation of hypothermic neuroprotection requires the coordination of pre-clinical studies and clinical studies in all aspects.
Another issue worth noting is publication bias, which is an indisputable fact that editors prefer to publish positive results. 125 This may also leave many people with the impression that pre-clinical studies have found numerous neuroprotective methods. To improve the translation of neuroprotectant efficacy including hypothermic therapy from bench to bedside, pre-clinical randomized controlled multicenter trial (pRCT) may be a new option. Pre-clinical studies conducted jointly by multiple laboratories will greatly enhance the reliability of experimental results and thus reduce the bias. 127
Conclusion
The treatment of AIS, a major cause of human death and disability, has always been concerned. Based on the plentiful encouraging results from recent pre-clinical and clinical studies, therapeutic hypothermia, the long-standing and powerful neuroprotective method, has again gained great attention due to its multiple targets and long-term benefits. Although there is still a lack of high-level evidence to support the neuroprotective effects of hypothermia in AIS patients, in the background of continuous progress of medical technology (such as recanalization therapy), hypothermic therapy has also ushered in an unprecedented development opportunity. More in-depth and meticulous research work is needed to accelerate the clinical translation of hypothermic therapy.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key R&D Program of China (grant nos 2017YFC1308401; 2016YFC1301502); Chang Jiang Scholars Program (#T2014251) from the Chinese Ministry of Education; National Natural Science Foundation of China (grant nos 81701287; 81871022; 81601006; 81620108011) and Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201706).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
