Abstract
Hypothermia is a promising therapeutic option for stroke patients and an established neuroprotective treatment for global cerebral ischemia after cardiac arrest. While whole body cooling is a feasible approach in intubated and sedated patients, its application in awake stroke patients is limited by severe side effects: Strong shivering rewarms the body and potentially worsens ischemic conditions because of increased O2 consumption. Drugs used for shivering control frequently cause sedation that increases the risk of aspiration and pneumonia. Selective brain cooling by intraarterial cold infusions (IACIs) has been proposed as an alternative strategy for patients suffering from acute ischemic stroke. Preclinical studies and early clinical experience indicate that IACI induce a highly selective brain temperature decrease within minutes and reach targeted hypothermia 10 to 30 times faster than conventional cooling methods. At the same time, body core temperature remains largely unaffected, thus systemic side effects are potentially diminished. This review critically discusses the limitations and side effects of current cooling techniques for neuroprotection from ischemic brain damage and summarizes the available evidence regarding advantages and potential risks of IACI.
Keywords
INTRODUCTION
Accounting for 9% of deaths worldwide, stroke is a leading cause of mortality in both developed and developing countries. 1 Furthermore, ˜25% of stroke survivors remain permanently disabled. 2 These problems are expected to further aggravate during the next decades, as global trends in aging continue. Despite considerable efforts, intravenous thrombolysis with recombinant tissue plasminogen activator within the first 4.5 hours from symptom onset remains the only proven acute therapy for ischemic stroke. 3 Because of a narrow therapeutic window and strict eligibility criteria only a small minority of patients does receive this treatment. 4
There has been a long quest to identify alternative treatment strategies for acute stroke patients. As many as 1026 different drugs have been tested for neuroprotective efficacy in acute stroke between 1957 and 2003. 5 While clinical trials of pharmacological neuroprotective strategies in stroke have failed to show clear benefit, hypothermia has prevailed as a promising therapeutic option for stroke patients. Supported by prospective randomized trials, hypothermia is an established neuroprotective treatment for global cerebral ischemia after cardiac arrest (CA) and for hypoxic-ischemic encephalopathy in newborns6–8 and is recommended in current clinical guidelines for these indications.9,10 A number of experimental studies have collected compelling evidence, which suggests that hypothermia does not only induce neuroprotection after CA, i.e., global cerebral ischemia, but also in ischemic stroke. 11 In a meta-analysis, the use of hypothermia in animal models of ischemic stroke has been shown to reduce infarct volumes on average by 44%. 12
Mechanisms of Hypothermia-Induced Neuroprotection
Some of the neuroprotective effects of hypothermia can be attributed to a reduction in oxygen demand: A decrease in brain temperature by 1°C lowers cerebral oxygen consumption by ˜5%, thus increasing tolerance to ischemic conditions. 13 In addition, hypothermia modulates a number of intracellular and extracellular reactions that control the development of ischemia/reperfusion injury. These include pathways of apoptosis and necrosis induction, tissue inflammation and blood–brain barrier disruption.14,15
Most potent neuroprotection are achieved if hypothermia is started before or soon after the onset of ischemia and maintained in the reperfusion period. 16 If hypothermia is induced late in the ischemic period or after reperfusion, neuroprotective effects are considerably diminished or even lost. 17 This narrow window of opportunity emphasizes the importance of rapid temperature reduction for the successful clinical application of hypothermia in stroke patients.
A number of studies have tried to determine the optimal brain temperature for neuroprotection in stroke. Experience from surgical procedures, where body core temperature is commonly reduced to 10°C to 28°C if full circulatory arrest is required, suggests that lower body core temperatures increase the neuroprotective effects of hypothermia.18,19 Concordantly, several animal experiments found lower temperatures associated with increased neuroprotection in severe ischemic brain injury.17,20,21 Conflicting results have been reported in models of mild focal brain ischemia: Here maximum neuroprotection was found at body core temperatures of 33°C or 34°C.22,23 More aggressive body core temperature reductions did not further reduce infarct volumes, but were associated with more hemodynamic and respiratory instability during surgery and delayed recovery after anesthesia. 23 Temperatures below 30°C were not tested in these latter trials.
Experiences with Whole Body Cooling in Stroke Patients
In clinical stroke, surface and endovascular cooling have been used for successful whole body hypothermia induction and maintenance: Surface cooling methods include cold rubs, ice packs, cooling blankets, or cooling pads that noninvasively reduce body core temperature. Endovascular cooling methods reduce body temperature invasively using intravenously placed cooling catheters or intravenous cold infusions.
The results of several case series and small clinical trials (n = 18 to 59)24–26 outline some fundamental limitations associated with these techniques: Surface cooling methods are easy to use and permit early treatment induction, but require at least 2 hours to reach target temperatures.26–29 Considerably shorter induction times have been reported for endovascular cooling devices (64 to 126 minutes).24,26,30,31 However, in patients receiving recombinant tissue plasminogen activator the requirement of invasive cooling catheter placement frequently caused delays in treatment initiation because of concerns about bleeding complications. 31
Intravenous infusion of large volumes of cold fluids has been successfully used to increase the speed of whole body cooling in CA survivors.32,33 Similar results have been reported in a limited number of ischemic stroke patients.26,29,34,35 Two currently active large multicenter trials that aim to evaluate the benefits of whole body cooling in ischemic stroke, the EuroHYP-1 (ref. 36) and the ICTuS 2/3 study, 37 include a combination of cold infusions and surface or endovascular cooling in their treatment protocol.
Adverse Side Effects of Whole Body Cooling
Whole body cooling is associated with a number of adverse side effects that can involve almost all organ systems and lead to changes in cardiovascular function, shivering, immunosuppression, coagulation impairments, electrolyte imbalances, and acid/base disorders. 38 Severe temperature reductions (<28°C to 30°C) are associated with potentially lethal side effects (e.g., malignant arrhythmias) that prohibit the use of deep hypothermia without extracorporeal life support. However, these complications hardly occur at higher temperatures (>30°C). 38 In the temperature range of mild (34°C to 36°C) to moderate (32°C to 34°C) hypothermia, which has been targeted for neuroprotection in ischemic stroke patients, shivering and immunosuppression are most significant. 31
Shivering is part of the physiological thermoregulatory response 39 and directly and indirectly diminishes the neuroprotective effects of hypothermia: Increased muscle activity rewarms the body, thus counteracting cooling efforts and prolonging the time- to-target temperature. In addition, shivering might aggravate ischemic conditions because of increased metabolic activity and oxygen consumption. 40 While shivering can easily be treated in intubated and sedated patients, most stroke patients remain awake and require pharmacological treatment to counteract shivering. A combination of meperidine and buspirone has been widely used in clinical trials, with mixed results in regard to shivering control.24,30,31,34
In the ICTuS-L study, target temperatures were missed in 4 of 28 patients because of uncontrollable shivering despite the use of very high doses of meperidine (14.5 ± 6.9 mg/kg).30,31 Furthermore, concerns have been raised about meperidine-induced sedation that might favor aspiration. 31 Aspiration is a major concern in awake stroke patients, who often have dysphagia. 41 If aspiration occurs, acute stroke patients are generally prone to infections, likely because of stroke-induced immunosuppression. 42 Whole body hypothermia further increases this risk as it reduces inflammatory reactions and tampers immune responses to infections. 38 The ICTuS-L study reported that 50% of cooled patients developed pneumonia, while only 10% in the control group were affected. 31
FROM WHOLE BODY COOLING TO SELECTIVE BRAIN HYPOTHERMIA
The protective effects of hypothermia during and after ischemia are not limited to the brain and have been shown in a number of other organ systems.43–45 This suggests that whole body cooling should be applied in settings of pan-ischemia like CA. However, hypothermia-induced neuroprotection depends on lowered brain temperature and does not require whole body cooling. 20 Therefore, selective brain cooling might be an attractive alternative to whole body hypothermia in cases of isolated brain ischemia like ischemic stroke. Compared with whole body cooling, selective brain cooling targets a significantly smaller tissue volume; hence higher rates of temperature reduction seem possible. In addition only minor body core temperature reductions are expected during selective brain cooling, which could help to minimize systemic side effects. The extent to which systemic side effects can be avoided remains uncertain. 46 A number of selective brain cooling methods have been investigated, including cooling helmets, nasopharyngeal cooling, local cooling coils, cerebrospinal fluid exchange, and intraarterial cooling.
Cooling helmets seem to exhibit limited impact on brain temperature. Passive cooling helmets, which need to be precooled before application on the skin of the patient, lead to no 47 or very modest reductions in deep brain temperature (–0.36°). 48 Active cooling helmets, which are continuously perfused with cold liquids, seem to have stronger effects on superficial brain temperature (−1.84°C). 49 However, theoretical analyses suggest that the effects on superficial brain layers are probably not achieved in deep brain regions.50–52
Conflicting results have been reported for nasopharyngeal cooling methods that attempt to use the dense vasculature in the nasal cavities and the pharynx and their close local proximity to the brain to induce selective brain hypothermia. Ventilation of the nasopharynx with cold air has failed to produce clinically significant brain temperature reductions.47,53,54 Cold saline perfused intranasal balloon catheters were estimated to reduce brain temperature by −1.7°C over 60 minutes in healthy volunteers based on magnetic resonance spectroscopic imaging data. 55 These results could not be replicated in a trial that performed direct measurement of brain temperatures in brain-injured patients treated with a similar device. In this trial, which aimed for normothermia in hyperthermic patients, brain temperatures decreased by an average of only 0.7°C over 72 hours. 56 Nasopharyngeal delivery of an evaporative coolant with the RhinoChill device reduced brain temperature by 1.2°C to 1.4°C within 60 minutes.57,58 However, it has been noticed that the use of this device might be linked to potentially dangerous increases in blood pressure even in deeply sedated stroke patients. 58
Other methods for selective brain cooling have been investigated in animal trials and small case series. Local cooling coils placed under the temporal muscle have been shown to selectively decrease brain temperature in rats and decreased infarct volumes by 42% in a model of transient middle cerebral artery occlusion.59,60 Moomiaie et al 61 successfully tested intraventricular cooling catheters in sheep and reported a mean brain temperature of 34.5°C that was established within less than 15 minutes after initiation of cooling. Bell et al 62 reported the feasibility of exchanging cerebrospinal fluid with an artificial oxygenated nutrient solution at room temperature in four stroke patients, but unfortunately brain temperature measurements were not performed. Even though some of these results are compelling, the high level of invasiveness of these approaches may limit future clinical application.
INTRAARTERIAL COLD INFUSIONS
Even though the brain only represents 2% of the total body weight in humans, it accounts for 20% to 25% of the resting metabolism. It is therefore hardly surprising that the brain is supplied with 15% of the cardiac output. The high blood flow through the brain effectively counteracts most selective brain cooling methods, as warm blood from the body core continuously rewarms the brain tissue. Rather different from other approaches, intraarterial cold infusions (IACIs) attempt to use the high cerebral blood flow to induce brain temperature reductions. Even though IACI might be deemed too invasive in some stroke patients, endovascular recanalization is frequently attempted in patients suffering from proximal vessel occlusions. 63 During these angiographic procedures, cold fluids could easily be infused through the endovascular catheter.
The idea of IACI as an emergency treatment in stroke patients is relatively new and current knowledge of effects and safety is only based on rodent experiments and computer simulations. However, many years of clinical and experimental experience with IACI during surgical procedures have shown the high effectiveness of IACI for the prevention of ischemic injury. In addition, IACI have already been successfully tested in large animals for hypothermia induction after CA. Clinical feasibility of IACI has also been shown in awake volunteers without acute brain damage. The results of these studies will be discussed in the following section.
Intraarterial cold infusion for the Prevention of Ischemic Injury During Total Circulatory Arrest
During surgical procedures that require total circulatory arrest (TCA), deep whole body hypothermia is routinely supplemented by selective perfusion of the brain with cold blood to prevent ischemic injury: While whole body hypothermia decreases metabolic demand, selective brain perfusion additionally ensures an adequate supply of the brain with oxygen and nutrients. 18 To minimize the side effects associated with deep whole body hypothermia, some case series investigated if selective cerebral perfusion can also be used for differential brain cooling during these procedures.64–66 In all reports, patients were precooled to a body core temperature of 28°C before TCA. Immediately after TCA induction, Bachet and Guilmet 64 used selective cerebral perfusion with cold blood (10°C to 12°C) to decrease brain temperature below body core temperature. In two other publications, separate perfusion of the brain and the viscera allowed to stabilize the brain temperature at 25°C 66 or 28°C, 65 while body core temperature was increased to 32°C 66 or 34°C to 36°C. 65 All case series reported good functional outcome after surgery without any increase in neurological complications.
The requirement for an established cardiopulmonary bypass presents a major drawback of these techniques. To avoid this limitation, early experiments conducted by Wolfson and colleagues67–74 used intracarotid infusion of cold crystalloids to induce profound brain hypothermia during TCA. In normothermic dogs, the infusion of 10 mL/kg body weight/min of ice-cold saline (0°C) for 6 minutes into the right common carotid artery plus local head cooling with ice packs rapidly reduced brain temperature in the ipsilateral hemisphere below 20°C (contralateral hemisphere <25°C). Twenty minutes of TCA did not result in any neurological symptoms in these animals. 70 Similar results were reported in baboons: Within 9 minutes ipsilateral and contralateral brain temperatures decreased to 16°C and 28°C, respectively. No neurological sequelae were found after of 30 minutes of TCA. 72
Finally, the group reported a total of five clinical cases of terminally ill patients who received IACI during neurosurgical procedures requiring TCA.74,75 They claim that in precooled patients (30°C body core temperature) the infusion of 10 mL/kg body weight/min of ice-cold Ringer's solution for 5 minutes into the common carotid artery allowed them to safely maintain circulatory arrest for up to 60 minutes. 75
Intraarterial cold infusion for the Treatment of Ischemic Injury after Cardiac Arrest
While the aforementioned experiments concentrated on the prevention of ischemic damage during surgery, the other investigations put their focus on potential applications of IACI as an emergency treatment after CA. An early report about experiments that tested IACI in dogs as an adjunct treatment during cardiopulmonary resuscitation was published more than 30 years ago. In this experiment, cold fluids infused into the common carotid artery reduced mortality and improved functional outcome after experimental CA. 76
Infusion of cold fluids into the proximal aorta has been tested in dogs and pigs as a more extensive method to protect both the brain and the heart during CA.77–85 Briefly, the thoracic aorta was occluded using a catheter-mounted balloon, and cold saline (2°C to 4°C) was infused into the proximal part of the vessel. The infusate distributed among the thoracic branches of the aorta, including the brain supplying and coronary arteries. Most experiments used extremely large infusion volumes of 100 mL/kg body weight or more.78–81,84 Although these experiments required the use of a cardiopulmonary bypass for resuscitation and sometimes venting of the right atrium to avoid volume overload, the intervention was highly effective in preventing ischemic brain damage: No neurologic deficits in dogs after up to 90 minutes of CA. 78 Aortic flushing with more modest fluid volumes was tested in combination with standard cardiopulmonary resuscitation in another experiment, which refrained from using a cardiopulmonary bypass. Infusion of a fluid volume of 30 mL/kg body weight (similar to the volumes that have been used for intravenous cold infusions) successfully reduced brain temperature from 38.3°C to 33°C within less than 5 minutes after 15 minutes of CA. Neuroprotective effects of this treatment remained elusive because of high mortality in treatment and control groups. 85
According to a conference report, rapid infusion of 500 mL (4°C to 8°C) cold Ringer's solution into the carotid artery has already been used in humans to reduce tympanic temperature to 35°C within 2 to 3 minutes after successful resuscitation. 86 Unfortunately, details on the experimental protocol or patient outcome have never been published.
INTRAARTERIAL INFUSIONS IN ANIMAL MODELS OF STROKE
Intraarterial infusions have been used for two main objectives in animal models of stroke: (1) High flow rates and relatively high infusion temperatures were used to remove vasoactive and inflammatory substances that accumulate in the ischemic territory during ischemia and contribute to the development of reperfusion damage (prereperfusion flushing).87–90 (2) Infusion of similar volumes at lower flow rates and with low infusion temperatures was used to induce selective brain hypothermia (IACIs).91–98 Experimental setups and results of all animal experiments that have investigated intraarterial infusions in ischemic stroke are summarized in Table 1.
Effects of IACI in animal stroke models
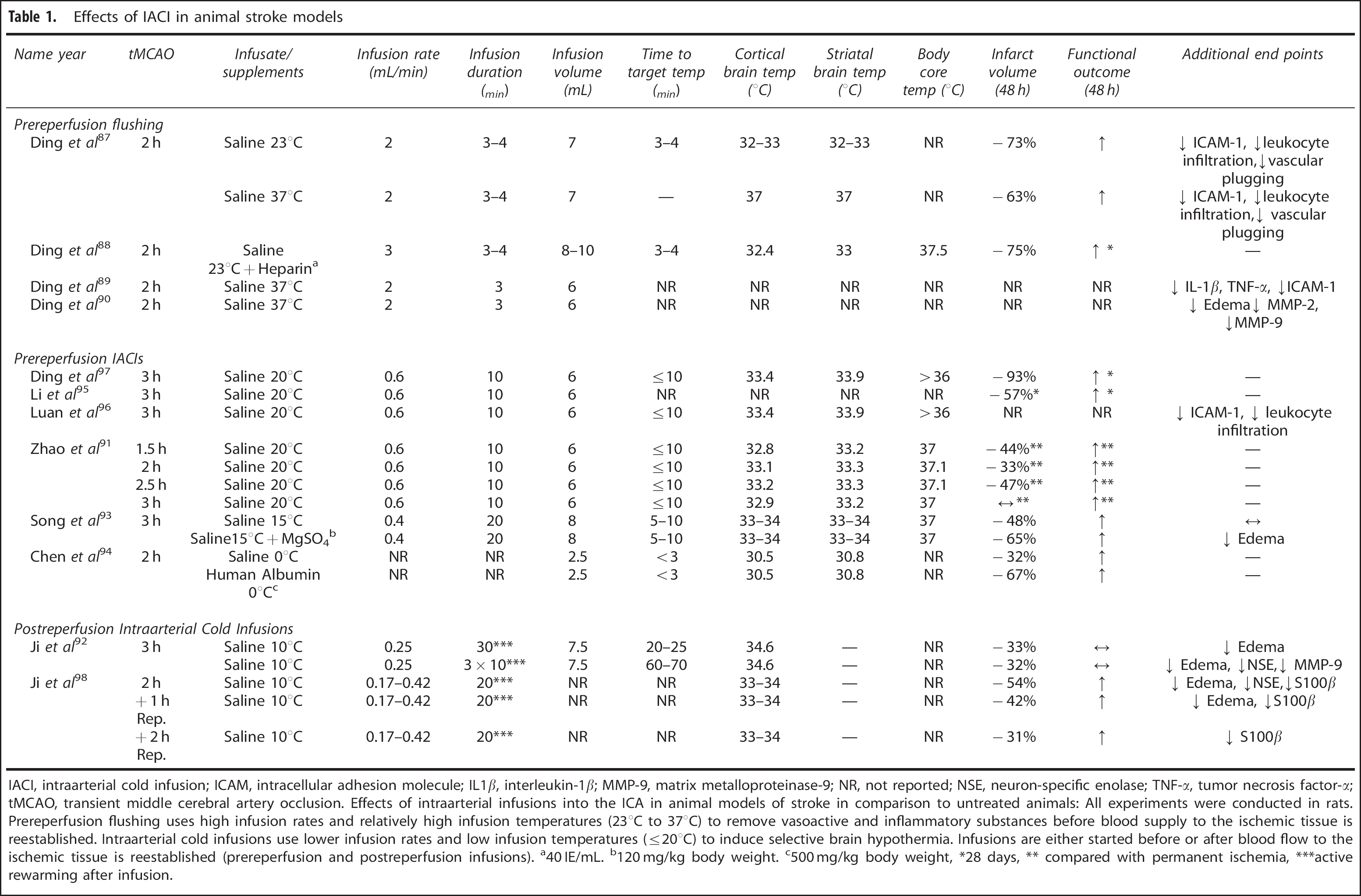
IACI, intraarterial cold infusion; ICAM, intracellular adhesion molecule; IL1β, interleukin-1β; MMP-9, matrix metalloproteinase-9; NR, not reported; NSE, neuron-specific enolase; TNF-α, tumor necrosis factor-α; tMCAO, transient middle cerebral artery occlusion. Effects of intraarterial infusions into the ICA in animal models of stroke in comparison to untreated animals: All experiments were conducted in rats. Prereperfusion flushing uses high infusion rates and relatively high infusion temperatures (23°C to 37°C) to remove vasoactive and inflammatory substances before blood supply to the ischemic tissue is reestablished. Intraarterial cold infusions use lower infusion rates and low infusion temperatures (≤20°C) to induce selective brain hypothermia. Infusions are either started before or after blood flow to the ischemic tissue is reestablished (prereperfusion and postreperfusion infusions).
40 IE/mL.
120 mg/kg body weight.
500 mg/kg body weight,
28 days,
compared with permanent ischemia,
active rewarming after infusion.
(1) PreReperfusion Flushing in Animal Models of Stroke
The first report on prereperfusion flushing was published by Ding et al 87 : Occlusion of the middle cerebral artery for 2 hours was performed in rats using a hollow filament. This filament was used to infuse saline into the ischemic territory before blood flow was reestablished. Prereperfusion flushing with 7 mL saline at a rate of 2 mL/min reduced infarct volumes and improved functional outcome after 48 hours 87 and 28 days. 88 Treatment benefits about infarct volume and functional outcome were not significantly different after the infusion of saline at body temperature (37°C) or room temperature (24°C). 87 This has been attributed to the fact that the short duration of the infusion (3 to 4 minutes) led to a substantial, yet very short decrease in brain temperature: Cortical temperature decreased from 37.5°C to 32.4°C during the infusion, but returned to normothermic levels (>36°C) only 2 minutes later. 88
Reduced inflammation and improved microcirculation were indicated by a decrease in leukocyte infiltration and microvascular plugging. 87 Further analyses also showed lowered expression of inflammatory cytokines (tumor necrosis factor-α, interleukin-1β) and adhesion molecules (intracellular adhesion molecule-1). 89 Reduced brain edema and matrix metalloproteinases-2 and -9 expression indicate protective effects on vascular permeability and the blood-brain barrier. 90
(2) IACI in Animal Models of Stroke
Two different approaches have been taken to apply IACI in animal models of stroke. The majority of experiments chose prereperfusion infusions and used the same hollow filament technique that has been used for prereperfusion flushing. Different from the flushing experiments, colder infusions (0°C to 20°C) were applied at lower flow rates (0.6 mL/min) and over longer time periods (10 min).91,95–97 Prereperfusion cold infusion of 20°C saline easily established moderate hypothermia (32°C to 34°C) in the ipsilateral hemisphere within 5 to 10 minutes in several experiments.91,95–97 A reduced infusion temperature of 0°C resulted in a decrease in cortical and striatal temperatures below 31°C within less than 3 minutes. 94 Prereperfusion IACI caused significant reductions in infarct volumes ranging from 30% (ref. 94) to 90%. 97 Improvements in behavioral performance could be shown after 48 hours91,93,94 and up to 28 days after ischemia.95,97
A postreperfusion approach was taken in two studies by Ji et al:92,98 In these experiments a catheter was introduced into the ICA after blood flow to the ischemic territory had been reestablished. In a first study, a volume of 7.5 mL 10°C saline was infused either continuously over 30 minutes (0.25 mL/min) or in three equal parts that were each infused over 10 minutes followed by a 20 minute break (3 × 2.5 mL/10 min + 20 min break). 92 In a second study, a brain temperature-guided infusion of 10°C cold saline for 20 minutes at a variable flow rate (0.16 to 0.42 mL/min) was used to induce and maintain moderate brain hypothermia. 98 Compared with the very fast induction speeds that have been concordantly reported for prereperfusion IACI (<10 min), postreperfusion infusions took longer to establish brain hypothermia: Infusion of 10°C saline at 0.25 mL/min required more than 20 minutes to reduce brain temperature below 35°C. 92 This might be because during postreperfusion infusions cold saline inevitably mixes with blood at body temperature as it enters the vasculature. The resulting perfusate is considerably warmer than the initial infusion and therefore exhibits smaller cooling effects. In addition, the infusion rate in this study was ˜50% lower than the rates used in prereperfusion IACI experiments.
Infarct volumes were reduced in all postreperfusion IACI experiments.92,98 This effect seems to be most pronounced if the infusion is started immediately after reperfusion, but benefits could be shown for delayed infusions up to 2 hours after blood flow had been reestablished. 98 Conflicting results have been reported regarding beneficial functional outcome, which were significant in one study 98 and insignificant in the other. 92 This could be explained by the relatively slow induction speed reported in the latter trial 92 or the use of rapid rewarming in both trials, which was used to normalize brain temperature as soon as the infusion had stopped.
Different from the temperature-independent effects of perre-perfusion flushing, IACI-induced neuroprotection was linked to brain temperature reduction: In several prereperfusion and postreperfusion IACI experiments, a comparison of cold (0°C to 20°C) and normothermic infusions (37°) concordantly showed cold infusions associated with stronger neuroprotective effects.92–94,96–98
Two studies investigated combinations of IACI and neuroprotective substances. Song et al
93
supplemented 15°C cold saline with magnesium sulfate, which acts as a N-methyl-
Few studies focused on the mechanisms behind IACI-induced neuroprotection. Prereperfusion cold infusion has shown to reduce inflammation in the ischemic territory indicated by a decrease in intracellular adhesion molecule-1 expression and leukocyte infiltration. 96 Amelioration of brain edema could be shown after postreperfusion cold infusion.92,98 An interrupted postreperfusion infusion regimen also significantly reduced matrix metalloproteinase-9 levels in the serum, a sign of improved blood-brain barrier stability. 92
COMPUTER SIMULATIONS OF IACI IN STROKE
Starting with Slotboom et al in 2004, several theoretical models have attempted to predict the effects of IACI on brain temperature in human stroke.100–105 Results of these computer simulations are summarized in Table 2.
Effects of IACI in computer simulations and healthy volunteers
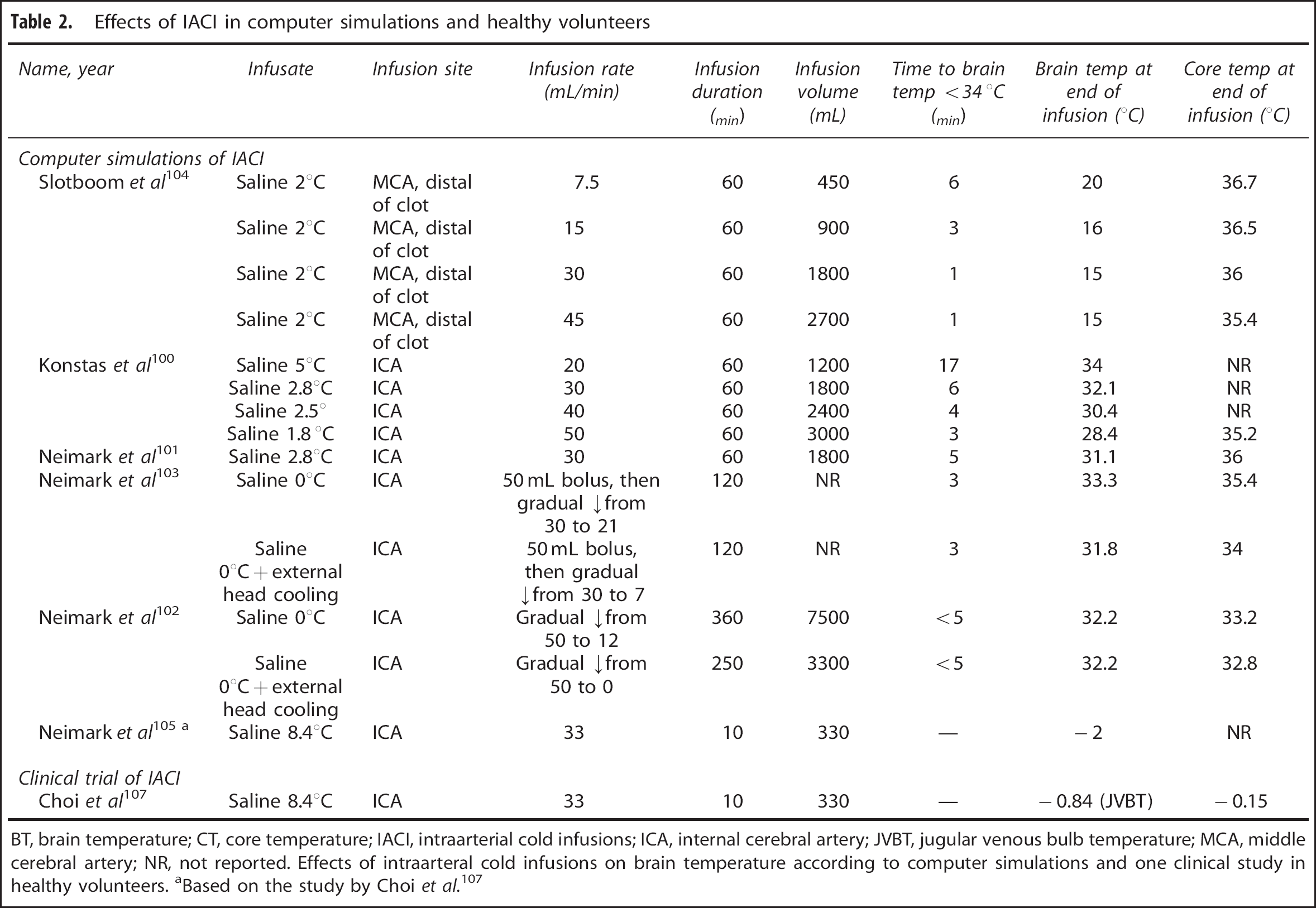
BT, brain temperature; CT, core temperature; IACI, intraarterial cold infusions; ICA, internal cerebral artery; JVBT, jugular venous bulb temperature; MCA, middle cerebral artery; NR, not reported. Effects of intraarteral cold infusions on brain temperature according to computer simulations and one clinical study in healthy volunteers.
Based on the study by Choi et al. 107
Slotboom et al104,106 proposed the use of prereperfusion IACI during local recanalization. They argued that in their experience endovascular catheters frequently pass through the occluding clot before recanalization is achieved, thus theoretically allowing the infusion of cold solutions into the ischemic territory distal of the clot. They estimated that cooling of an ischemic brain tissue of 300g below 34°C could be established within less than 6 minutes if 15°C saline is infused at a rate of 15 mL/min.104,106
Konstas et al 100 presented a sophisticated computer simulation of a brain suffering ischemic stroke, consisting of an ischemic core surrounded by a penumbra. According to their calculations, a small fluid volume (150 mL) infused at 30 mL/min should suffice to decrease brain temperature in the penumbra to 34°C within 5 minutes. 100 Continuous IACI for 60 minutes (total infusion volume = 1.8 L) would reduce the temperature in the ischemic territory to 32°C. 100 Even stronger reductions (<29°C) could be possible if the infusion rate is increased to 50 mL/min (total infusion volume = 3 L). 100 Both infusion rates are predicted to have only relatively minor influence on body core temperature, which would be reduced by ˜1°C (30 mL/min) (ref. 103) or 1.8°C (50 mL/min). 100 For comparison, intravenous cold infusion of 2.2 L saline (4°C) reduced body temperature by 1.6°C within 52 minutes in stroke patients. 34 It seems plausible that equal volumes of intravenous cold infusion and IACI would have similar effects on body core temperature. However, intraarterial fluid application likely increases local cooling effects in the ipsilateral hemisphere by a factor of 4 to 5 compared with intravenous infusions. Furthermore, calculations by Slotboom et al104,106 and Konstas et al100,104 concordantly predict that IACI could help to reach mild brain hypothermia 18 to 30 times faster than established surface cooling techniques and 10 to 20 times faster than invasive intravenous cooling catheters.
One limitation of all reviewed simulations is the assumption that cold saline passes through a thermally insulated catheter before injection into the internal carotid artery.100,104 This differs from current clinical practice, where noninsulated catheters are used during endovascular recanalization. The heat conduction properties of the catheter are critical for the cooling effects of IACI, as the infused fluid is exposed to heat transfer from the surrounding blood as it passes through the endovascular catheter from the groin to the neck region before entering the ICA. If standard endovascular catheters are used, effects on brain temperature will likely not be as pronounced as predicted by these simulations and local hypothermia will not be established as rapidly.
CLINICAL FEASIBILITY AND SAFETY OF IACI IN AWAKE PATIENTS
Based on the promising results of animal experiments and computer simulations, one clinical trial has attempted to investigate the feasibility and safety of IACI for selective brain cooling in awake patients. The patient population consisted of 18 patients undergoing routine diagnostic follow-up angiographies after treatment for vascular malformations. At the end of the procedure, a total of 330 mL cold saline (4°C to 17°C) was infused over 10 minutes (33 mL/min). Different from the aforementioned computer simulations, a standard noninsulated catheter was used for the infusion. Direct measurement of brain temperature was deemed inappropriate in the participants because its invasiveness and jugular venous bulb temperature was chosen as a surrogate. Cold saline infusion caused a gradual reduction in jugular venous bulb temperature over the infusion period and resulted in a maximum decrease of 0.84°C after 10 minutes. Significant changes in vital parameters or neurological symptoms were not observed. Mild shivering without discomfort or pain was reported in 3 out of 18 patients. 107
Measurement of jugular venous bulb temperature presents a major limitation to this experiment, as it strongly correlates with body core temperature but often differs from brain temperature. 108 The authors acknowledged this limitation and presented a simulation based on the theoretical model by Konstas and group 105 that was adjusted to predict the actual brain temperature based on the observed patient parameters. These calculations suggest a temperature reduction in ipsilateral brain temperature of 1.9°C to 2.1°C within 10 minutes. This is remarkable, as fairly modest infusion volumes (330 mL) and standard noninsulated neurointerventional catheters were used.
LIMITATIONS OF THE CURRENT EXPERIMENTAL EVIDENCE
Current experimental evidence of IACI in stroke is only based on rodent experiments and computer simulations. Animal models have been widely criticized for their poor translatability to clinical practice. 109 If hypothermia treatment is tested in rodent models, the much smaller body volume and different volume to surface ratio might lead to overestimations regarding induction speed. 12 In case of selective brain cooling, the thin skull and small lissencephalic brain of rodents make incorrect predictions about thermal changes even more likely. Despite these possible sources of bias, results of the aforementioned clinical and experimental studies that have tested IACI during TCA and in healthy volunteers suggest high cooling efficacy also in large animals and humans.64–75,107
The majority of animal stroke experiments used selective infusion of cold fluids into the ischemic territory before blood supply was reestablished, i.e., prereperfusion IACI. Although this approach maximizes cooling effects as mixing of the cold infusate with warm blood is avoided, it conflicts with early reperfusion. Early reperfusion is the most important factor for good functional outcome in ischemic stroke patients and should not be delayed. 110 Postreperfusion infusions might be a more appropriate approach as they do not require the cold infusion to be finished before reperfusion can be established. Unfortunately, this approach has only been investigated in two animal stroke trials.92,98 Furthermore, it should be noted that IACI have never been compared with whole body cooling, the current gold standard of hypothermia treatment.
Current knowledge about potential risks of IACI is also limited. Even though the good tolerability in a few of healthy subjects in a pilot trial 107 is reassuring, doubts remain about the safety of IACI in stroke patients.
POSSIBLE ADVERSE SIDE EFFECTS OF INTRAARTERIAL COLD INFUSION
One obvious concern is the risk of complications arising from the endovascular procedure. Most importantly, thromboembolism could be triggered by injury to the arterial endothelium or thrombogenic properties of the endovascular equipment. 111 Generally, cerebral angiographies carry a relatively low risk of thromboembolic events that result in permanent neurological impairment (0.1% to 0.5%). 112 Nonneurological complications other than groin hematomas (6.9%) are also very infrequent (0.3%). 113 Furthermore, IACI would most likely be used in patients who are routinely treated with endovascular recanalization. While in these patients IACI could easily be performed using the same endovascular catheters during a single procedure, the total procedure duration would inevitably be prolonged. Long procedure durations have been identified as a risk factor for neurological complications during cerebral angiographies.113,114 However, intraarterial endovascular catheters lodged in the ICA have been used for continuous local infusion over 2 to 5 days without occurrence of thrombosis in patients with intact coagulation. 115 In a patient receiving full-dose heparin, it was even possible to let the catheter remain in the vertebral artery for full 12 days. 116
Another critical variable is the increased duration of anesthesia caused by the prolonged endovascular procedure. However, complications such as aspiration occur more frequently during induction of anesthesia than during the maintenance phase. 117 In clinical trials investigating whole body hypothermia after endovascular procedures, stroke patients remained intubated and sedated for up to 96 hours with low complication rates.24,118 It should be noted that general anesthesia might not even be necessary during IACI, as conscious sedation has been found to be safe during endovascular treatment for acute ischemic stroke. 119
Another concern could be the induction of vasoconstriction or even vasospasm if cold fluids are infused into brain supplying vessels. In their pilot trial, Choi et al 107 did not find any signs of vasospasm on Doppler sonography or angiographic control. Furthermore, none of the other clinical case reports or animal experiments cited in this review reported any signs of vasospasm.
None of the reviewed studies have raised concerns about increased hemorrhagic transformation after IACI. Nevertheless, clotting disorders are a known adverse side effect during hypothermia and seem to depend on local blood temperature.15,120 This warrants caution, especially in case of severe brain temperature reductions.
Given the limited clinical experience with selective brain cooling in awake patients, it remains difficult to estimate to what extend selective brain cooling would lead to systemic side effects that have been reported during whole body hypothermia. For example, the role of central and peripheral thermosensory neurons in the hypothalamus and the skin for the development of thermoregulatory responses is still under debate.121,122 It is also unsure how hypothermic temperatures in the brain would influence the systemic immune response. 46
The administration of large fluid volumes during IACI bears the risk of systemic volume overload. Although 2.2 L of intravenous cold infusion has been safely administered in awake stroke patients, 34 the most recent trial that tested intravenous cold infusion after CA reported an increased incidence of pulmonary edema. 123
Systemic and local hemodilution is another concern. In simulations presented by the group of Neimark and colleagues, 103 saline infused at a rate of 30 mL/min for 60 minutes decreased systemic hematocrit from 42% to 37.5%. At the same time, hemodilution in the perfused territory reduced local hematocrit to 31%. 103 At an increased rate of 50 mL/min for 60 minutes, systemic and local hematocrit levels would decrease to 33.5% and 26.5%, respectively. 100 However, even hematocrit levels as low as 20% should be sufficient for oxygen delivery. 124
SUMMARY AND CONCLUSION
So far, most studies on hypothermia treatment in acute stroke applied whole body cooling. Whole body hypothermia can be easily induced in small animal stroke models, but rapid induction in humans has proven difficult because of the larger body volume. In addition, systemic side effects such as severe shivering are frequent and often cannot be sufficiently controlled in awake stroke patients. 31 Selective brain cooling targets both of these limitations, and could allow faster temperature reductions with minimized systemic side effects.
Promising experimental results have been reported for IACI that use the dense vasculature and high blood flow of the brain to induce rapid and profound brain temperature reductions. If IACI are used in rodent stroke models, moderate brain hypothermia can be established within few minutes and significant reductions in infarct size and improved functional outcome have been observed.91,95–98 In humans, theoretical calculations suggest that infusions at a rate of 30 mL/min could be sufficient for rapid brain hypothermia induction.100,104 A clinical feasibility trial in patients without acute brain damage has shown that IACI can effectively reduce brain temperature in humans. Even though most participants were awake during the intervention, only mild systemic side effects were reported. 107 Despite these promising results, open questions remain about the clinical translatability of these findings. Furthermore, knowledge about potential side effects of IACI in patients with acute brain injury is currently lacking.
Footnotes
SP receives speaker's fees from CR. Bard, BeneChill, EMCOOLs, and ZOLL Medical; project funding from ZOLL Medical and EMCOOLs; and receives supplies and equipment from BeneChill, Covidien, EMCOOLs, HVM Medical, Raumedic, and ZOLL Medical.
