Abstract
Cerebral edema (CE) is a major component of early brain injury from aneurysmal subarachnoid hemorrhage (SAH). Despite the utility of blood-based protein markers, biomarkers targeting cerebral edema have yet to be developed. We performed a targeted proteomic search analyzing 141 proteins in plasma samples taken from 80 adult patients within 48 hours after aneurysmal rupture to identify plasma biomarkers of cerebral edema. The protein expression profiles were analyzed for associations with a higher level of the subarachnoid hemorrhage early brain edema score measured in admission computed tomography scans. Five differentially expressed proteins—brorin, ectodysplasin A2 receptor, layilin, scavenger receptor class A member 5, and activin A receptor like type 1 (ACVRL1)—were identified. To confirm our findings, in a separate cohort of 75 adult patients with SAH, these biomarker candidates was analyzed using a different confirmatory analytic method—enzyme-linked immunosorbent assay—based on the plasma samples collected within 48 hours after aneurysmal rupture. ACVRL1 levels consistently showed a decrease expression levels as the severity of edema increased. The association between CE and ACVRL1 was significant before and after adjusting for patient factors.
Keywords
Introduction
Annually there are 500,000 cases of aneurysmal subarachnoid hemorrhage (aSAH) globally with one third of patients suffer from life-long complications, including cognitive dysfunction and impaired activities of daily life. 1 The failure of trials focused on the treatment of vasospasm, which was thought to be the major contributor to delayed cerebral ischemia (DCI) and, has warranted a shift to the identification of other pathophysiologic pathways for future therapeutic approaches. 2 Early brain injury (EBI), a complex collection of pathophysiological processes occurring within 72 hours after the index event, has been identified as a key process which connects the initial event to the delayed and long-term complications.3,4 Preclinical studies have identified the crucial components of EBI and potential targeted therapeutics, but clinical studies are still in their premature stages.
Cerebral edema (CE) is one of the major process of EBI. 5 Hemoglobin products’ direct toxicity and a sudden increase in the intracranial pressure due to extravasated blood cause cell death and disrupt the blood-brain barrier. The resultant cytotoxic and vasogenic edema perpetuate and aggravate the secondary injury from aSAH by arresting cerebral perfusion and inducing a proinflammatory response.6,7 Recent progress in clinical studies on CE has identified clinical biomarkers. The subarachnoid hemorrhage early brain edema score (SEBES) is a non-invasive, graded scale with high interrater reliability developed from computed tomography (CT) images obtained within 24 hours from the index event. 8 Its promise as a valuable biomarker is supported by its consistent associations with patient outcomes from SAH, including delayed ischemia, the need for decompressive craniectomy, cortical infarctions, and functional outcomes. 9
Despite the development of clinical biomarkers there is a need to identify blood-based protein biomarkers to supplement radiographic biomarkers. Blood biomarkers have the additional advantage of having the potential of being involved in the underlying pathophysiological mechanisms and being targeted for therapeutic intervention. They are more amenable to follow-up without the logistic burden of magnetic resonance imaging or side effects from repeated CT images or the invasiveness of cerebrospinal fluid biomarkers. Among the candidates for blood biomarkers, proteins are most widely chosen because they are the universal drivers of cellular processes and the mediators of intercellular communication. Additional advantages include established familiarity and cost-efficiency compared to other biomarkers, including metabolites and transcriptomes. Despite their importance, the current literature lacks blood protein biomarkers specifically targeting cerebral edema in EBI. The present study utilized a proteomics approach to discover serum protein biomarkers of cerebral edema in aSAH patients.
Material and methods
Study population
The present study is a retrospective analysis of a prospective observational cohort. Patients with acute aSAH admitted to the neuroscience intensive care unit at a single tertiary referral center were surveyed for enrollment. Inclusion criteria for the enrollment included: (1) age >18 years, (2) diagnoses of spontaneous aneurysmal SAH by computed tomography (CT) within 24 hours of ictus, and (3) the modified Fisher scale (mFS) > 2. 10 The last inclusion criterion was imposed to focus on the subjects with severe SAH who have higher chance of developing CE. 8 Exclusion criteria were: (1) non-aneurysmal SAH, such as aSAH due to trauma, arteriovenous malformation, or mycotic aneurysms and (2) having comorbidities that could significantly alter the baseline status, including autoimmune diseases, history of malignancy, and current pregnancy. The current study is based on two independent cohorts, and the cohorts were used for the discovery and confirmation of potential candidate proteins, respectively. Each cohort consisted of 80 consecutive patients recruited between 2016 and 2019, and 75 patients recruited between 2017 and 2021. No subject was enrolled into both cohorts, and cohort designation was randomized.
Sample collection and analysis
Plasma was collected from the patients in each cohort within 48 hours of aneurysmal rupture. Blood samples were collected in EDTA-treated tubes and processed within one hour of collection. The tubes were inverted gently to ensure that they are mixed properly. The tubes were then centrifuged at 1460 g for 10 minutes at 4°C to separate plasma from the cellular components. Following centrifugation, the upper plasma layer was carefully transferred to a new sterile 15 ml tube, avoiding disturbance of the buffy coat. Then, the 15 mL conical tube was centrifuged at 3260 g for 10 minutes at 4°C. The plasma was removed and aliquoted at 500 ul per tube. Samples were then stored at −80°C for long-term storage. In the first cohort, 184 proteins were analyzed using a proximity extension assay by a commercially available Olink Target 96 Neuro Exploratory panel and Olink Target 96 Neurology panel. Detailed descriptions of the experimental procedures and analysis techniques were reported in the previous studies.11–13 Briefly, matched pairs of antibodies targeting specific proteins and carrying unique oligonucleotide tags were bound to the respective proteins in the sample, if present. Matched pairs recognize different epitopes of a target protein, which increases the specificity of the results. Upon binding, the oligonucleotide tags hybridize with each other, forming a polymerase chain reaction (PCR) reporter sequence. Then, high-throughput real-time quantitative PCR is performed, measuring the quantity of the hybridized tag, representing the quantity of each target protein in the sample. Three classes of internal controls are added to detect aberrancies in each experimental step—binding of the tags, extension using PCR, and quantification—with additional control for inter-experimental comparison. Differences in the threshold cycle values are log-transformed with the base of 2 to produce normalized protein expression (NPX) values.
In the second cohort, the enzyme-linked immunosorbent assay (ELISA) kits for LAYN, ACVRL1, VWC2, SCARA5, EDA2R were used to measure the plasma level of the respective protein (catalog No.: RAB0706, RAB1257, RAB1292, RAB1678, and RAB0652, Millipore-Sigma). Experiments were conducted while the experimenters were blinded to clinical information and followed the protocols provided by the manufacturers.
Outcomes and clinical information
Upon each subject’s enrollment, data were prospectively collected including both demographic and clinical data, such as age, sex, ethnicity, and clinical severity determined by the Hunt-Hess Scale (HH) grade. 14 All brain CT scans on admission were graded for mFS, the modified Graeb score, and cerebral edema using SEBES by two independent raters who were blinded to clinical data.10,15 SEBES was defined on a scale from 0 to 4 points in relation to sulci effacement plus or minus the disruption of the grey-white differentiation at two predetermined levels in each hemisphere as previously described. 8 When there was a discrepancy, the CT scan was graded by a third rater. All raters are board-certified neurologists. SEBES was dichotomized into SEBES of 0–2 and 3–4 to reflect mild and severe edema, respectively.
Statistical and bioinformatics analysis
After obtaining the raw values for protein expression, post-processing methods differed based on the platform. For the first cohort, protein expression levels from the Olink platforms were expressed in NPX. NPX values were relative measures of expression already normalized and log-transformed with the base of two. For each protein, NPX values for all the subjects were compared against the limits of detection (LOD), and the proteins with more than 20% of their NPX values lower than LOD were excluded from the analysis to minimize artificial associations, resulting in 141 proteins being analyzed. For the second cohort, the protein expression levels were given in pg/ml or ng/ml, depending on the protein. These expression levels were log-transformed with a base of two for normalization.
Descriptive analysis of the subjects used Wilcoxon rank-sum tests and analysis of variance for continuous variables and Fisher’s exact tests and Chi-square tests for categorical variables. Bivariate comparison of protein expression levels used Wilcoxon rank-sum tests, and, for comparing more than two groups of subjects, Kruskal-Wallis tests were used.
During the discovery stage, significant proteins were filtered based on their fold changes (FC) and the p-values of the difference in the expression levels between the two groups being compared. The p-values were adjusted for multiple testing with the Benjamini-Hochberg method. 16 The false discovery rate of 0.1 was used as the threshold to screen for differentially expressed proteins. Among the differentially expressed proteins, five proteins— LAYN, ACVRL1, VWC2, SCARA5, and EDA2R —were chosen as the candidates based on their adjusted p-values, absolute values of fold changes, and pathophysiological plausibility based on their known functions. The proteins unrelated to inflammatory processes were preferred because our previous study had identified inflammatory biomarkers associated with delayed complications from SAH. 17
During the confirmatory stage, the protein expression levels were compared between SAH patients who presented with severe edema and those with mild edema, using Wilcoxon rank-sum tests. The proteins showing differential expression between the two groups were examined for possible associations between their expression levels and patient factors that could falsely represent or confound the relationship between the proteins and severe edema. Logistic regression models with the protein expression level dichotomized based on the median as a dependent variable and each patient factor, such as age, sex, histories of hypertension and diabetes mellitus, and the HH grade as the only independent variable, were fitted and examined for significance. To identify potential relationships between the dichotomized levels of protein expression and the odds of severe edema, two logistic regression models were used. First, a univariable logistic regression model was used without any covariates. Second, a multivariable model including the patient factors potentially associated with the level of proteins and the severity of edema as covariates was used to examine whether the observed association persists after adjustment. The covariates included a patient’s age, gender, and histories of hypertension and diabetes mellitus. Every statistical test used during the confirmatory stage was two-tailed, and the threshold of 0.05 was used to define statistical significance for the p-values. All analyses were performed with R 4.3.1 (R core team, 2023).
Standard protocol approvals, registrations, and patient consents
The study was approved by the institutional review board at the University of Texas McGovern School of Medicine (HSC-MS-12–0637). Written informed consent was obtained from each patient or surrogate. The study was conducted following STROBE guidelines. All methods were performed in accordance with the Declaration of Helsinki.
Results
Patient characteristics
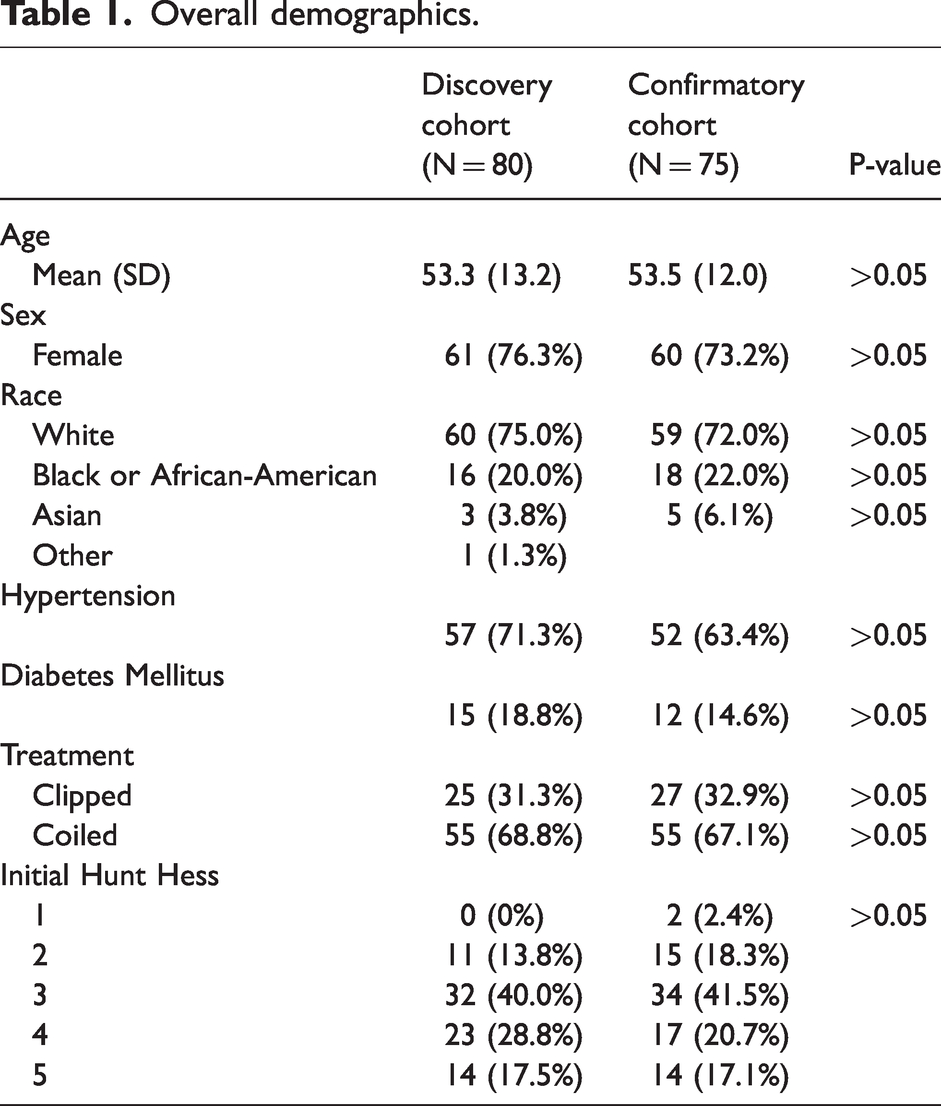
For the discovery and confirmatory cohorts, 80 and 75 patients were enrolled after the inclusion and exclusion criteria were applied. The cohorts were not significantly different with respect to demographic factors. The median ages were 53 [Interquartile range (IQR): 45–63] and 54 (IQR: 46–63) (p-value > 0.05), and 76% and 73% were females in each cohort (p-value > 0.05). The clinical severity of SAH and comorbid conditions were also not significantly different among the cohorts. 86% and 79% of the subjects had poor grade SAH, defined as the HH grade of 3 or more (p-value >0.05). 71% and 63% of the subjects in each cohort had history of hypertension, and 19% and 15% had history of diabetes mellitus (p-value >0.05) (Table 1).
Overall demographics.
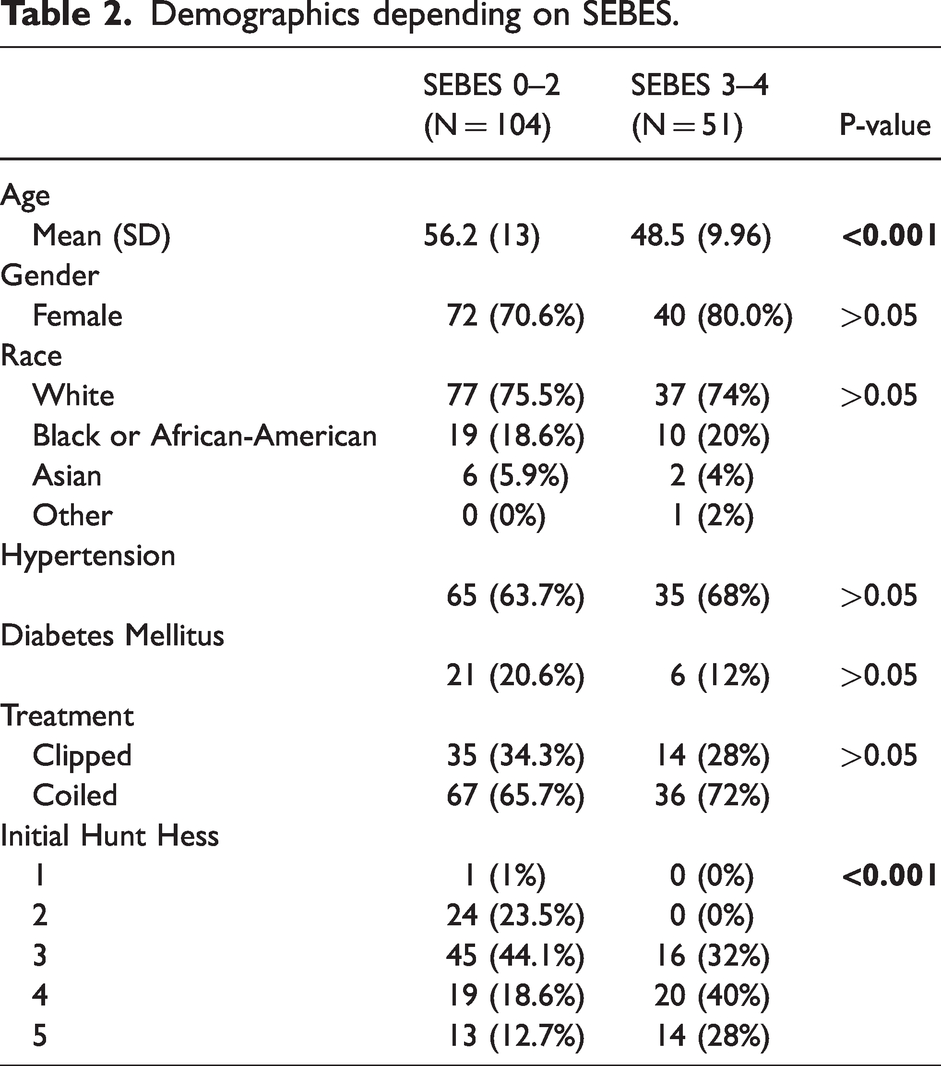
When the entire study population was divided into subjects with severe edema and those with mild edema, two important differences were noticed. The severe edema subpopulation was younger than the mild edema subpopulation, with the median age was 47 (IQR: 40–54) as compared to 59 (IQR: 47–65) (p-value < 0.001). The association between HH grade and SEBES was consistent with previous findings. 8 All the subjects in the severe edema subpopulation presented with poor grade SAH, while 73% of the subjects with mild edema presented with poor grade SAH (p-value < 0.05). Besides the differences in age and clinical severity, the two subpopulations did not show significant differences in other patient factors, including the proportion of female subjects, race distributions, histories of hypertension and diabetes mellitus, and treatment modalities (p-value > 0.05) (Table 2).
Demographics depending on SEBES.
Protein biomarkers of cerebral edema in the discovery cohort
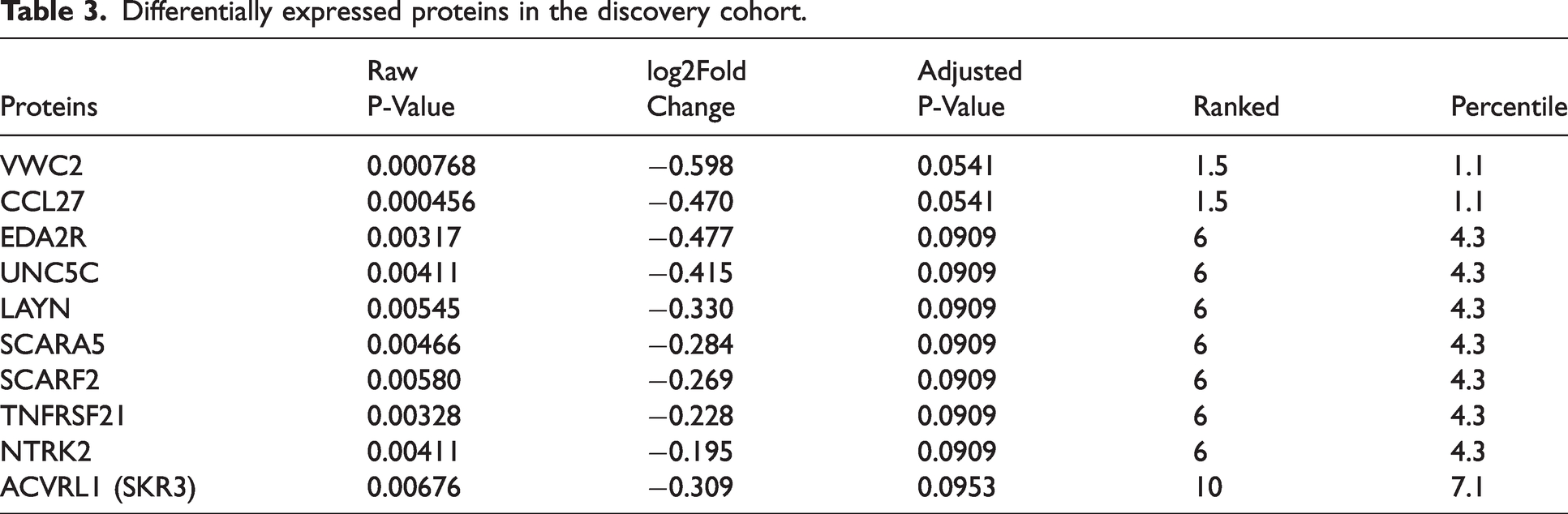
For the first cohort, out of 141 proteins analyzed, only 10 proteins—Brorin (VWC2), CC motif chemokine ligand 27 (CCL27), ectodysplasin A2 Receptor (EDA2R), unc-5 netrin receptor C (UNC5C), layilin (LAYN), scavenger receptor class A member 5 (SCARA5), scavenger receptor class F member 2 (SCARF2), TNF receptor superfamily member 21 (TNFRSF21), neurotrophic receptor tyrosine kinase 2 (NTRK2), and activin A receptor like type 1 (ACVRL1, ALK1, SKR3)—showed false discovery rates that are lower than the preset threshold 0.1 and were defined as differentially expressed proteins (Table 3). All the differentially expressed proteins showed decreased expression in patients with severe edema, with a mean fold change of 0.78. None of the analyzed proteins showed fold changes less than 0.5 or greater than 2.0 when comparing patients with severe edema to those with mild edema (Figure 1).
Differentially expressed proteins in the discovery cohort.
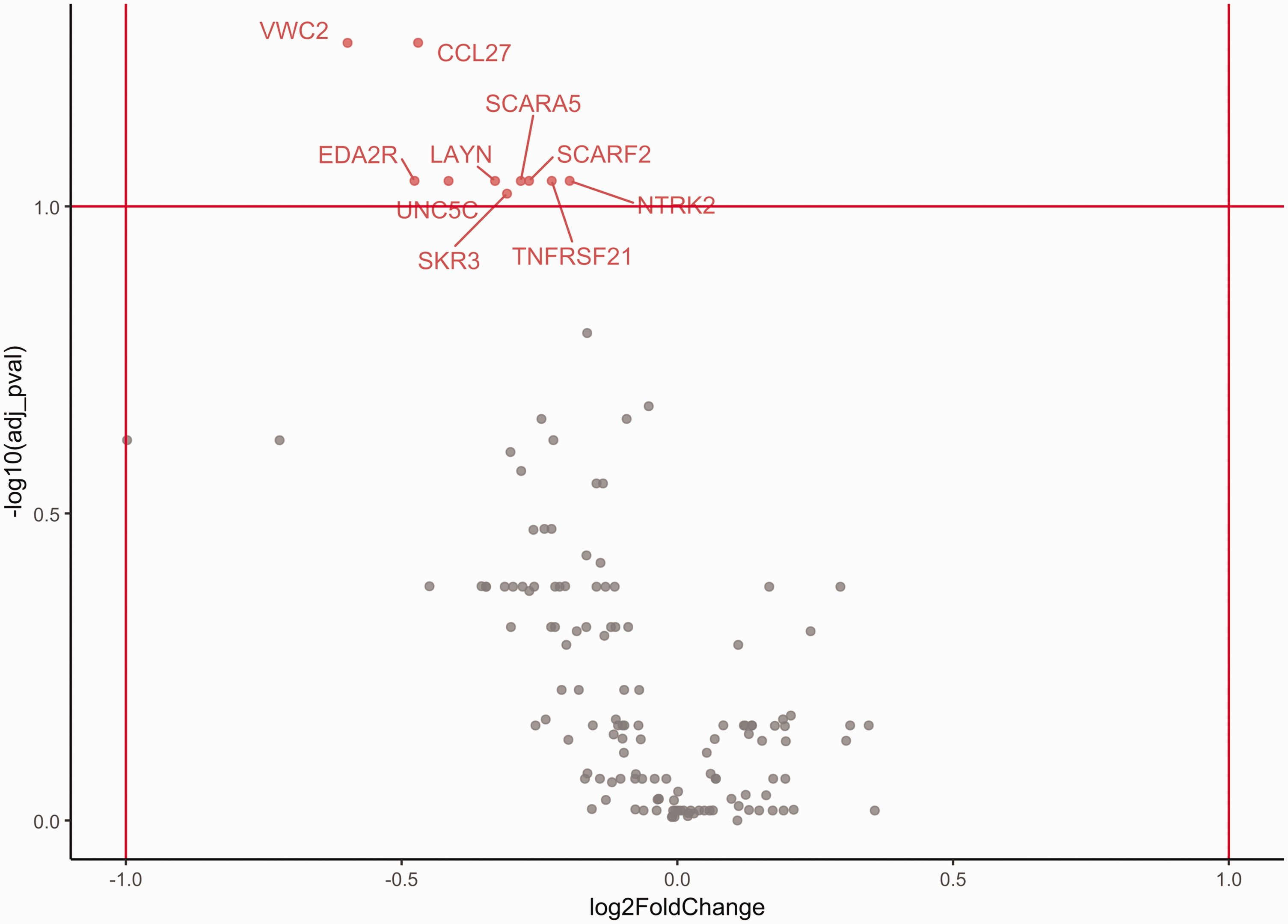
Differentially expressed proteins in the discovery cohort. A blood-based targeted proteomic search revealed 10 proteins to be associated with severe edema in subarachnoid patients.
Confirmation with ELISA
Plasma levels of VWC2, EDA2R, and SCARA5 did not show statistically significant difference between the patients with severe edema and those without severe edema, (p-value > 0.05) (Figure 2). The mean expression levels for the three proteins were 200 pg/ml (SD: 498), 78.3 pg/ml (SD: 112), and 0.197 ng/ml (SD: 0.0379), respectively, in the patients with mild edema, and 158 pg/ml (SD: 460), 79.3 pg/ml (SD: 150), and 0.189 ng/ml (SD: 0.0274), respectively, in the patients with severe edema. LAYN showed a trend toward a differential expression with the mean expression level in mild edema of 97.7 ng/ml (SD: 100), which was higher than 88.1 (SD: 109) in severe edema, but the difference did not achieve statistical significance (p-value = 0.11) (Figure 2). ACVRL1 was the only protein that showed a significantly different expression level between severe edema and mild edema. The expression level was higher in mild edema with a mean of 0.247 pg/ml (SD: 0.323) than 0.133 (SD: 0.242) in severe edema, (p < 0.01) (Figure 2). Both LAYN and ACVRL1 showed a trend where patients without severe edema showed higher protein expression levels than those with severe edema.
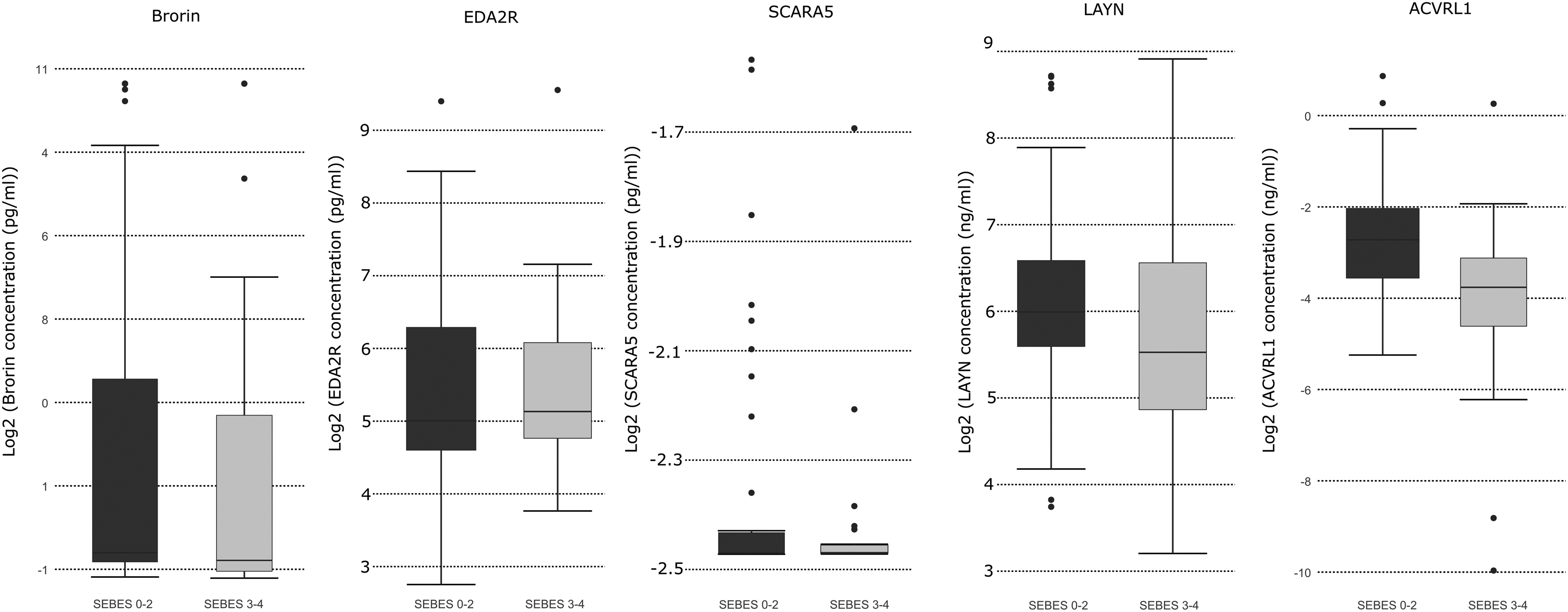
Expression levels of the candidate proteins based on the confirmatory cohort. Among the candidate biomarker proteins, only LAYN and ACVRL1 show identifiable differences between subjects with and without severe edema.
To gain more insights about the relationships between the proteins and cerebral edema, their expression levels were analyzed for differences among individual SEBES categories rather than between dichotomized SEBES categories. VWC2, EDA2R, and SCARA5 did not show noticeable trends or significant differences among different SEBES categories. LAYN and ACVRL1 showed statistically significant differences among different SEBES categories (p-value < 0.05). Also, they showed similar patterns throughout different categories of SEBES. The subjects with milder edema (SEBES = 1 or 2) showed the highest expression levels, which were higher than the subjects with no edema (SEBES = 0), and the subjects with more severe edema (SEBES = 3 or 4) had the lowest expression levels (Supplemental Figure 1).
Associations with severe cerebral edema
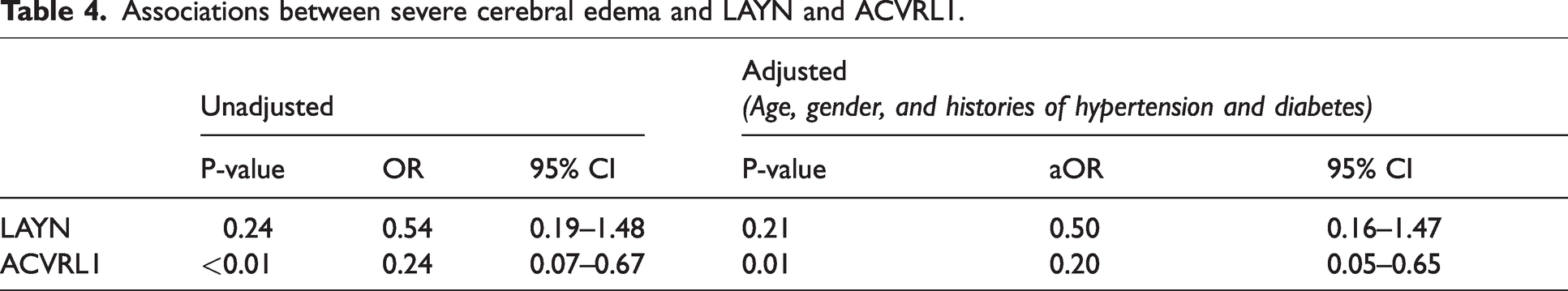
The two candidate biomarker proteins that showed perceivable differences between cerebral edema of different severities—LAYN and ACVRL1—were analyzed for their association with patient factors. Both proteins showed associations with age and history of hypertension, and the associated patient factors were included in the following models (Supplementary table 1). The associations among the HH grade, severe edema, and the two candidates were examined to prove the independent association between severe edema and the candidate biomarkers. While the HH grade was a significant predictor of severe edema—both in univariate and multivariate analysis—the two proteins’ levels were not associated with the HH grade (Supplementary table 2, 3, and 4).
Among the two candidates, only ACVRL1 retained its significance. A lower expression of ACVRL1 was associated with a higher odds of severe edema. An unadjusted analysis showed that a subject with a higher level of ACVRL1 was associated with 0.24-fold lower odds of severe cerebral edema (95% CI: 0.07–0.67, p-value < 0.01) (Table 4). The observed increase in the odds of severe edema in the subjects with lower expression levels persisted even after adjusting for age and history of hypertension. A higher level of ACVRL1 was associated with 0.2-fold lower odds of severe edema (95% CI: 0.05–0.65, p-value < 0.05) (Table 4). The expression levels of LAYN did not show significant associations with the odds of severe edema with or without adjustment for the prespecified covariates (Table 4).
Associations between severe cerebral edema and LAYN and ACVRL1.
Discussion
Our main finding is that ACVRL1 levels are associated with CE after SAH. Lower expression of ACVRL1 in plasma was associated with increased odds of severe cerebral edema. There are several strengths of our study. We utilized two independent, temporally separated cohorts to identify and confirm potential plasma biofluid markers of severe cerebral edema in patients presented with acute, severe aSAH. In addition, we used two separate independent protein detection methods. In the discovery cohort, 141 proteins were analyzed for their candidacy, and 9 proteins met the screening criteria. The confirmatory stage proceeded with 5 proteins, and only LAYN and ACVRL1 showed a discernibly different expression pattern between subjects with different degrees of cerebral edema. For both proteins, subjects with severe edema tended to present with lower expression levels, while age and history of hypertension were associated with their expression levels. The direct influence of their reduced expression on the odds of severe edema was analyzed, and ACVRL1 was the only protein to show a significant association.
Activin a receptor like type 1
ACVRL1 is an important regulator of angiogenesis. ACVRL1 is a membrane protein reported to form a complex with BMPR2, serve as a receptor for TGF-b, BMP9, or BMP10, and function as a major regulator of the TGF-b pathway. The TGF-b pathway is one of the key pathways regulating angiogenesis. The vascular endothelial growth factor (VEGF), Notch, and TGF-β pathways interact intricately to determine the morphogenic fate of endothelial cells.18–20 In preclinical studies, ACVRL1’s deletion has been shown to cause arteriovenous malformation characterized by hypervascularization and incomplete pericytic coverage of capillaries. 21 In humans, mutations in ACVRL1 are known to cause hereditary hemorrhagic telangiectasia, brain arteriovenous malformations, and its loss-of-function mutations have been reported to manifest as a distinct phenotype of pulmonary arterial hypertension (PAH), with younger presentation and poorer prognosis.22,23
aSAH triggers global hypoxia-driven angiogenesis within the neurovascular unit, a compensatory process that can paradoxically increases the risk of cerebral edema. Tissue hypoxia is an important driver of angiogenesis. Transient global ischemia induced by aSAH elicits a strong tissue hypoxia signal that leads to angiogenesis, evidenced by elevation of hypoxia-inducible factor (HIF) and VEGF in animal and human subjects following SAH.24,25 The beneficial effects of HIF upregulation on reducing hypoxic tissue damage are well-known in myocardial infarction and ischemic stroke.26–28 The evidence extends to aSAH. Higher levels of HIF and VEGF were associated with reduced hippocampal apoptosis and better functional outcomes in animal studies. 29 However, excessive angiogenesis can lead to exacerbation of cerebral edema. Blockage of HIF has indeed been shown to reduce brain water content and improve the blood-brain barrier (BBB) dysfunction in preclinical studies. 24 Yet, as alluded, complete blockage of angiogenesis might worsen tissue ischemia, neurovascular unit dysfunction, and neuronal damage. This dilemma is further exacerbated during the EBI phase, where microcirculatory failure, cellular energy depletion, and neuroinflammation further complicate the neurovascular response. 30
Elucidating the exact pathophysiologic mechanism explaining the identified negative association between the severity of cerebral edema and plasma level of ACVRL1 will require further experimental studies. Reduced plasma ACVRL1 levels may reflect either a physiological response aimed at limiting angiogenesis in the setting of edema, or a pathological consequence of impaired angiogenesis resulting in tissue ischemia, energy failure, and secondary edema. The versatility of ACVRL1 suggests the need to consider the possibility of a new cellular subtype specific to injured neurovascular units in the setting of aSAH. Evidence suggests that the role played by endothelial ACVRL1 can vary depending on microenvironmental contexts. In normoxic conditions, loss of ACVRL1 has been shown to affect arterial endothelial cells, while venous endothelial cells were primarily affected in hyperoxic conditions.21,31 ACVRL1 has also been linked to the lymphatic transition of endothelial cells, highlighting the need to investigate its role beyond the conventional pro- or anti-angiogenic framework. 32
Considering the time point at which ACVRL1 was measured, ACVRL1 may be a marker specific to the initial response of neurovascular units to compensate for ischemia following aneurysmal rupture. A previous study comparing patients whose global cerebral edema, detected in admission CT scans and those whose edema was detected in follow-up CT scans that were taken, on average, 6 days post-ictus showed a different set of predictors were associated with edema at each time point. Early edema was associated with the severity of the initial clinical presentation, including loss of consciousness at ictus and the HH grade. Delayed edema, on the other hand, was associated with the size of the aneurysm, vasopressor use, and IVH scores, in addition to loss of consciousness at ictus. Multiple preclinical studies suggest that early edema primarily arises because of early global ischemia owing to sharply increasing intracranial pressure, while the mechanisms underlying delayed edema are more complex, consisting of a combination of disrupted autoregulation of the blood-brain barrier, neuroinflammation, and toxicity of blood products. 33 The present study focused on CE found on admission CT scans taken within 24 hours post-ictus, which corresponds to the time point for early edema. Given that early CE is hypothesized to result from an initial ischemic insult, ACVRL1 may serve as a marker of the endothelial response to acute global hypoxia.
Layilin
Although not statistically significant, LAYN showed a trend towards lower levels of LAYN being associated with severe CE. LAYN is a transmembrane receptor that is highly specific to hyaluronan (HA), a major constituent of the extracellular matrix covering the apical surface of endothelial cells. 34 Released into the plasma in response to the endothelial damage, HA's association with the severity of inflicted injury has been well-recognized in ischemic and hemorrhagic stroke.35,36 HA-mediated signaling pathway is important in maintaining BBB integrity. When an in vitro model of the BBB was treated with HA, the expression levels of claudin-5, occludin, and zona occludens-1 were significantly decreased, with evidence of impaired physical and electrochemical integrity of the BBB. 37 Given the importance of HA in the pathogenesis of BBB dysfunction and the apparent specificity of LAYN to HA, further investigation into LAYN’s role in CE following aSAH may be beneficial.
Limitations
This is one of the few aSAH studies in which a biomarker was discovered using an unbiased proteomic approach and validated in a separate cohort using a separate detection method. However, the present study has important limitations in its study design. We only examined a single time point, soon after hospitalization. However, the delay between aneurysmal rupture, hospitalization and the sample collection was not accounted for. This limits the value of the identified proteins as biomarkers since the association with outcomes can be confounded by the chemical kinetic properties of the proteins. Serial analysis of the samples captured at multiple time points could provide more robust view of the associations. Our findings of decreased Layilin and ACVRL1 in severe CE may be from decreased production, increased uptake, or increased elimination in this group compared to those with mild CE. Also, since we took an unbiased, bottom-up approach in identifying biomarker proteins, the cellular sources of the proteins need to be investigated to examine the biological plausibility. Potential sources of the proteins include arterial endothelial cells and dendritic cells for ACVRL1 and epithelial cells and neuronal cells for LAYN.38–42 Further translational studies are required to address these issues. Additionally, the study focused on 141 proteins based on commonly associated proteins in neurological pathology, but a larger untargeted proteomics approach might yield a different set of biomarker candidates. It should also be noted that the clinical variables related to CT scans were evaluated only by neurologists, not independent neuroradiologists, which could have introduced a bias. However, this study put efforts to increase reliability by having two different neurologists evaluate the images separately while blinded to the patient information and have another neurologist confirm the finding if the two neurologists’ findings disagree.
This study’s findings are not free from the risk of false positivity due to the limited number of subjects. To mitigate the potential false positivity, this study focused on a rigorously specified population of subjects compared to other studies. The fact that only subjects with severe aSAH were included guarantees better homogeneity among subjects in terms of their probabilities of CE. In the initial process of filtering out proteins, we further lowered the possibility of type I errors by removing any proteins with more than 20% of their expression values lower than the detection limit. Moving onto the confirmatory stage, its findings are free from the concern for multiplicity testing since only five selected proteins were examined. It is important to note that the discovery and confirmatory stages consist of patient pools that are temporally separated from each other. Identical expression patterns appearing in two temporally isolated patient pools using two different analytic methods—proximity extension assay and ELISA—corroborate the authenticity of our findings. For ACVRL1, its association persisted even after controlling for age, a factor that has been reported to show a strong negative correlation with the severity of cerebral edema after aSAH, further substantiating its value as a biomarker of less severe cerebral edema. 43 On the contrary LAYN was not statistically significantly different between groups, however, it did show a similar trend in both cohorts. As both cohorts were relatively small cohorts with high variability in protein expression levels, it is possible LAYN will be shown to be significantly different in future larger cohorts. Only after a complete chemical kinetic identification of the discovered proteins through repeated experiments and validation through a larger prospective observational cohort, the identified proteins’ true potential as a biomarker can be justified. Considering the observed plasma levels of ACVRL1, we expect a larger cohort consisting of 220 SAH subjects with and without severe edema with a 1:1 allocation ratio will achieve a statistical power of 0.8 with a significance level of 0.05.
Conclusion
CE is a major consequence of aSAH that influences patient outcomes. Plasma biomarkers, as opposed to clinical and radiographic biomarkers, can identify potential therapeutic agents. The present study utilized a targeted proteomic approach to identify candidate proteins and confirmed their candidacy using a different analytic method on a separate patient cohort. ACVRL1 showed statistically significant correlations with cerebral edema. A validating study based on a larger external cohort and further investigation into their pathophysiological roles is needed.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251361250 - Supplemental material for Targeted proteomic analysis identifies ACVRL1 as a marker of cerebral edema in aneurysmal subarachnoid hemorrhage
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251361250 for Targeted proteomic analysis identifies ACVRL1 as a marker of cerebral edema in aneurysmal subarachnoid hemorrhage by Bosco Seong Kyu Yang, Lena O’Keefe, Sarah Hinds, Hua Chen, Athziry Paz, Jude Savarraj, Han-Gil Jeong, Moon-ku Han, Aaron Gusdon, Xuefang Sophie Ren, Spiros Blackburn and Huimahn Alex Choi in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Huimahn Alex Choi received support from the National Institute of Neurological Disorders and Stroke under award (1R61NS119640 and 1R01NS131469). Xuefang Sophie Ren received support from the National Institutes of Health (1R01NS117606 and 1R01AG073659) and new faculty start-up funds from the University of Texas Health Science Center at Houston. Aaron Gusdon received support from the National Institute of Neurological Disorders and Stroke under award (K23NS121628).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
B. Y. and H. C.: conceptualization, methodology, formal analysis, investigation, writing – original draft, writing – review & editing, visualization.
L. O., S. H., H. C., A. P., and J. S.: methodology, validation, formal analysis, data curation, visualization
H. J., M. H., A. G., X. R., and S.B.: validation, writing – review & editing, supervision.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
