Abstract
Early brain injury (EBI) is considered an important cause of morbidity and mortality after aneurysmal subarachnoid hemorrhage (aSAH). As a factor in EBI, microcirculatory dysfunction has become a focus of interest, but whether microcirculatory dysfunction is more important than angiographic vasospasm (aVS) remains unclear. Using data from 128 cases, we measured the time to peak (TTP) in several regions of interest on digital subtraction angiography. The intracerebral circulation time (iCCT) was obtained between the TTP in the ultra-early phase (the baseline iCCT) and in the subacute phase and/or at delayed cerebral ischemia (DCI) onset (the follow-up iCCT). In addition, the difference in the iCCT was calculated by subtracting the baseline iCCT from the follow-up iCCT. Univariate analysis showed that DCI was significantly increased in those patients with a prolonged baseline iCCT, prolonged follow-up iCCT, increased differences in the iCCT, and with severe aVS. Poor outcome was significantly increased in patients with prolonged follow-up iCCT and increased differences in the iCCT. Multivariate analysis revealed that increased differences in the iCCT were a significant risk factor that increased DCI and poor outcome. The results suggest that the increasing microcirculatory dysfunction over time, not aVS, causes DCI and poor outcome after aneurysmal aSAH.
Keywords
Introduction
Aneurysmal subarachnoid hemorrhage (SAH) comprises 3–4% of all strokes and it remains associated with severe mortality higher than 25%.1–3 Previously, it had been hypothesized that the main cause of delayed cerebral ischemia (DCI) and poor outcome was angiographic vasospasm (aVS) of the major cerebral arteries. However, the results of the CONSCIOUS-1 study, which was a randomized, blinded clinical trial using an endothelin antagonist, clazosentan, led us to doubt that aVS is the only factor causing DCI and poor outcome. 4 As a result, early brain injury (EBI), which consists of neuronal apoptosis, impaired autoregulation, brain edema, cortical spreading depression (CSD) and microcirculatory dysfunction, has become a focus of interest.5–12
Before the CONSCIOUS-1 study, there had already been several experimental and clinical studies showing that microcirculatory dysfunction due to microthrombosis and/or microvasospasm could contribute to DCI after SAH.13–15 After the CONSCIOUS-1 trial, investigations of microcirculatory dysfunction after SAH increased, and its role in DCI and outcome appears to be conclusive.10,13–16 However, whether microcirculatory dysfunction or aVS plays the more important role in DCI and outcome has not been concluded clinically, because it is difficult to establish a clear clinical index for evaluating microcirculatory dysfunction. Some studies have indicated that cerebral circulation time (CCT) can reflect the status of microcirculatory impairment.17–19 Several studies using computed tomography perfusion have examined the relationship between DCI and indicators related to the CCT, such as the mean transit time (MTT) and the time to peak (TTP), but they did not reach a definite conclusion about their relationship to DCI, and a consensus among studies could therefore not be reached.20–32 Digital subtraction angiography (DSA) is recognized as the most reliable method for the evaluation of CCT and aVS. While two studies investigated the correlation between CCT measured by DSA exclusively during the ultra-early phase within 24 hours after the ictus, and no consistent result emerged of the relationship between CCT and DCI.20,22,32–35 Importantly, they did not examine CCT during the subacute phase.22,32
In order to clarify the correlation between microcirculatory dysfunction and DCI, it is necessary to evaluate the CCT together with aVS by means of DSA during both an ultra-early and a subacute phase. The purpose of the present study was to clarify the role of microcirculatory dysfunction in DCI after aSAH by measuring CCT and aVS in patients enrolled in two prospective, double-blinded, randomized placebo-controlled trials (RCT). These were performed in four of our neurosurgical institutions and they tested the efficacies of cilostazol and statin.36,37 We defined the “ultra-early phase” as within 24 hours after the SAH onset, and all patients in these two RCTs underwent DSA twice, in the preoperative, ultra-early phase and in the subacute phase between 7 to 11 days.32,38,39
Methods
Study population
Participants who used cilostazol or pitavastatin in two multicenter RCTs from 2012 to 2016 were enrolled.36,37 The RCTs included 256 patients with ruptured cerebral aneurysms, and 128 respective control cases served for the analysis in the present study. Both RCTs were conducted according to the Declaration of Helsinki and the ethical guidelines on clinical studies in four neurosurgical institutions in Aomori prefecture, Japan. The protocol for these studies was assessed and approved by the Committee of Medical Ethics of the Hirosaki University Graduate School of Medicine. Both studies were registered with the University Hospital Medical Information Network Clinical Trials Registry (UMIN, No. 000014402 and No. 000015977). All participants or their legal representatives obtained written, informed consent before treatment.
Eligibility for the study was based on fulfillment of the inclusion criteria of adult patients aged between 20 and 80 years who were admitted within 24 hours after the onset of aneurysmal SAH and treated by aneurysmal neck clipping or coil embolization within 48 hours after the ictus. The other inclusion criteria were a diffuse (long axis > 20 mm) or localized (long axis <20 mm), thick (short axis > 4 mm) SAH clot on a CT scan done within 24 hours after of the ictus, and Hunt and Hess (H & H) grades I–IV as evaluated before clipping or coil embolization. The exclusion criteria were the current use of drugs that could affect intracerebral circulation time such as antiplatelet, anticoagulant agents, endothelin antagonist, clazosentan, and any kind of statin. The other exclusion criteria were preexisting cerebral damage from past stroke or traumatic brain injury confirmed on CT scan, postoperative neurological deficits due to a clipping or coiling procedure, major neurological deficits and severe H&H grading due to concomitant or acute hydrocephalus and/or intracerebral hematoma induced by aneurysmal rupture, pre-existing severe hepatic, renal, pulmonary or cardiac disease, and pregnancy.
Standard of care
After aneurysmal clipping or coil embolization, the cases were randomly assigned to the placebo or drug treatment group. In the control group, the administration of placebo orally twice per day was started within 72 hours after the ictus and continued for 14 days. The circulating blood volume was kept within normovolemia, and all participants were treated three times per day with intravenous administration of 30 mg of fasudil hydrochloride, which has a vasodilatory effect due to Rho-kinase inhibition and is recommended under the Japanese stroke guidelines for the management of aneurysmal SAH. 40 On the other hand, none of the participants received oral or intravenous administration of nimodipine for prevention of DCI.
Rescue therapy was initiated when DCI was diagnosed as described below. The standardized procedures included induced hypertension and endovascular infusion of fasudil, followed by balloon angioplasty if adequate vasodilation was not achieved by vasodilator infusion. Acute hydrocephalus during the subacute phase was treated with external ventricular drainage or a sustained lumbar drain.
Clinical assessment
A baseline DSA was determined on admission within 24 hours after the ictus and a follow-up DSA was performed again 9 ± 2 days after the SAH ictus and/or within four hours after the onset of DCI in order to assess aVS and iCCT. DCI was defined as clinical deterioration of more than two points on the Glasgow Coma Scale (GCS), the development of new, focal neurological signs, or both, when the cause was felt to be ischemia attributable to vasospasm after other possible causes of worsening had been excluded. 41 All cerebral angiography was performed by flat panel DSA (AXIOM-Artiszee®, Siemens Health Care). Using standard angiographic methods (transfemoral route) for all patients, image acquisition was performed via a 4 Fr catheter with the tip positioned at the C3–4 level. The 2D DSA series were acquired at a rate of 16 frames per second, as is routine in our department. For image acquisition, 6 ml of contrast material (Iohexol 300, GE Healthcare, Japan) were injected in all series by mechanical injection at a flow rate of 8 ml/s for two seconds. The diameters of the proximal cerebral arteries, that is the bilateral C1 segments of the internal carotid artery, M1 segments of the middle cerebral artery, and A1 segments of the anterior cerebral artery, were measured, and the percent reduction in the arterial diameter was calculated by comparing the baseline DSA to the follow-up DSA. The severity of cerebral vasospasm was categorized as none or mild: 0–25% decrease; moderate: 25–50% decrease; and severe: over 50% decrease in arterial diameter on follow-up images compared to the baseline DSA. 42
DSA images were saved to a separate DVD in the Digital Imaging and Communications in Medicine (DICOM) format. Representative, consecutive DICOM images from DSA were loaded to a personal computer and we analyzed the time-density curve to measure time to peak (TTP) and intracerebral circulation time (iCCT) with post-processing image analysis software (U11437 ver. 1313. Hamamatsu Photonics Co., Ltd.) The regions of interest (ROIs) consisting of four pixels were placed at the vertical intracavernous portion of the internal carotid artery (ROIC5), at the cortical segment of the rolandic artery (ROIM4), and at the rolandic vein (ROIVR). The time-density curve was analyzed at the selected ROIs with regard to the image intensity divided by the run-time of the DSA in order to measure the TTP (Figure 1). TTP at ROIM4 (TTPM4) and TTP at ROIVR (TTPVR) were defined as the time from TTP at the carotid siphon (TTPC5) to the maximum level of the contrast density at each ROI. We then defined the iCCT as the difference in time between TTPM4 and TTPVR. We defined the baseline iCCT as that measured in the ultra-early phase after the ictus, and the follow-up iCCT was measured in the subacute phase on 7–11 days after the ictus. In addition, the difference in the iCCT was obtained by subtracting the baseline iCCT value from the follow-up iCCT value for each individual patient. 39
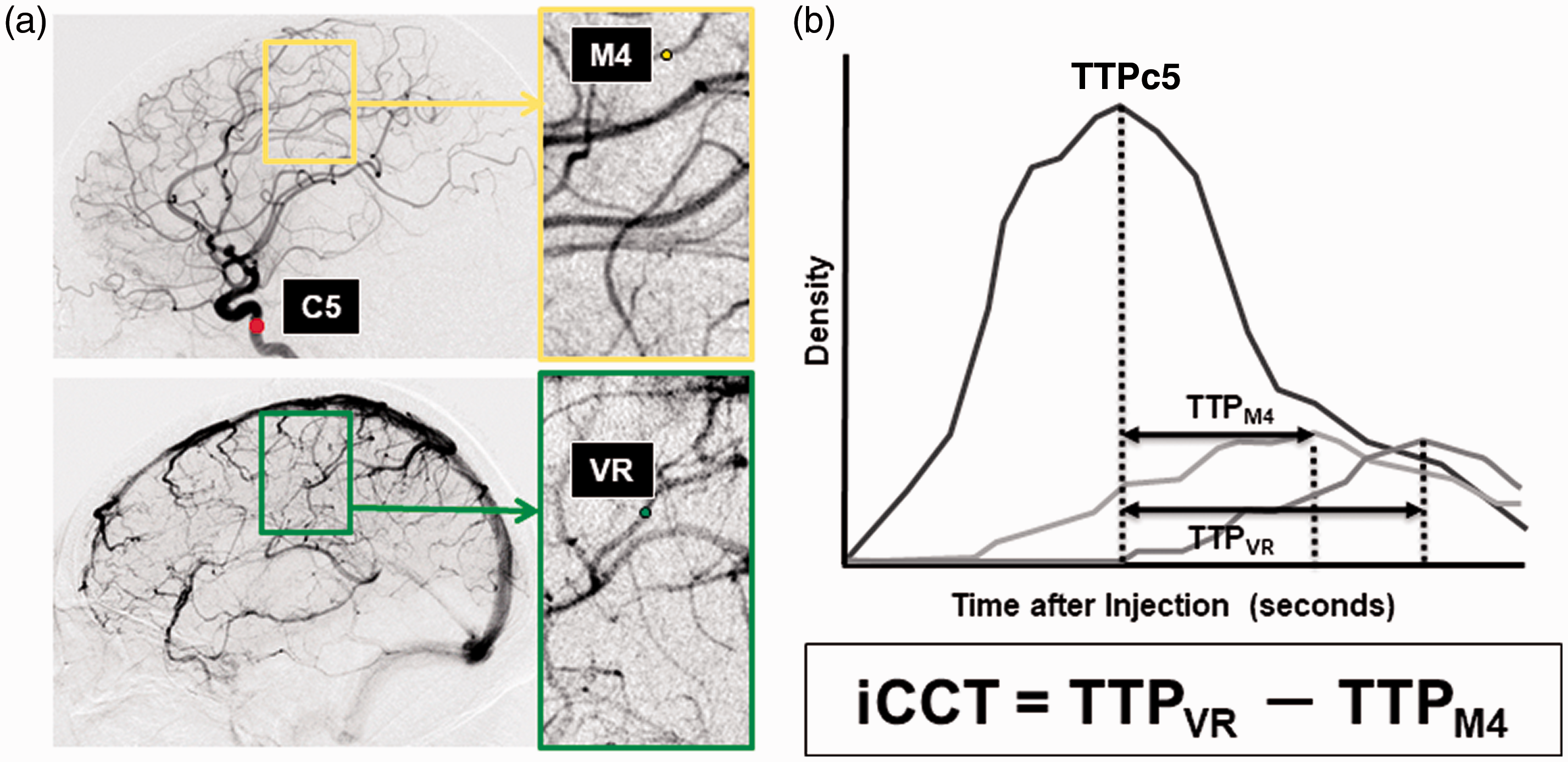
Measurement of iCCT.
Neurological examination and CT scan were performed on admission within 24 hours of SAH onset and 6–12 hours after the aneurysm treatment procedure. A follow-up examination was performed 7 ± 2 days, 14 ± 2 days, 1 and 3 months after the ictus, and whenever neurological worsening occurred. Clinical outcome was assessed by the Glasgow Outcome Scale (GOS). Poor outcome was predefined as severe disability (SD), vegetative state (VS) and death (D), and favorable outcome was predefined as good recovery (GR) and moderate disability (MD). This was assessed by two independent, blinded site investigators.
Based on these parameters, we compared aVS, the baseline iCCT, the follow-up iCCT, and the differences in the iCCT with the clinical characteristics of DCI and poor outcome. The factors affecting aVS and iCCT were also investigated.
Statistical analysis
Intergroup differences were analyzed using the Student's t-test, the Mann–Whitney U test, Pearson chi-square test, or the Fisher exact probability test. A two-sided probability value <0.05 was considered significant. To identify independent factors affecting DCI and outcome, multivariate analyses were performed by including possible confounding factors using a logistic regression model, and a receiver operating characteristics (ROC) curve analysis of iCCT was performed to assess a threshold value that allows identification of patients at risk for DCI. Sensitivity and specificity were calculated for this threshold value, as was the area under the curve (AUC) for quantifying diagnostic accuracy. The confidence interval was set at 95%. Multivariate analyses were performed by including possible confounding factors with a p < 0.2.36,43 All analyses used the statistics program JMP Pro ver. 13.
Results
A total of 128 patients were enrolled in this study. The baseline characteristics of age, sex, hypertension, smoking, IVH, ICH, aneurysmal clipping, H&H grade, severity of the SAH clot, and aneurysm location were comparable between patients with and without DCI and patients with and without unfavorable outcome (Table 1). All thirty of the participants with DCI underwent induced hypertension and received endovascular infusion of fasudil, and eight were followed by balloon angioplasty. Patients with unfavorable outcome had a significantly more severe H&H grade as compared to patients with favorable outcome (p < 0.01). The patients with DCI had significantly more severe aVS as compared to patients without DCI (p = 0.04); however, the rate of severe aVS did not differ between patients with favorable and with unfavorable outcome.
Demographics including intracerebral circulation time and incidence of endpoints for patients.
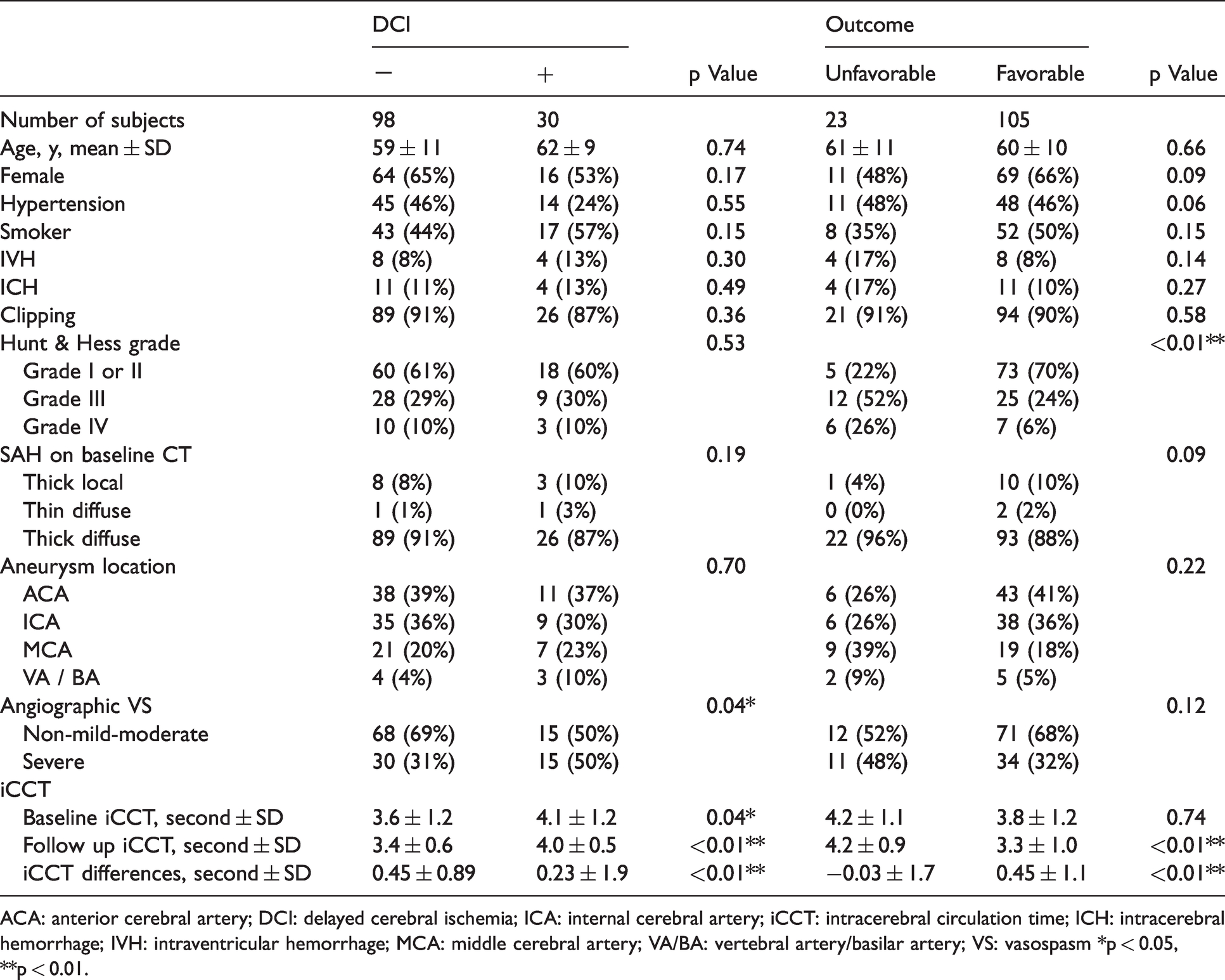
ACA: anterior cerebral artery; DCI: delayed cerebral ischemia; ICA: internal cerebral artery; iCCT: intracerebral circulation time; ICH: intracerebral hemorrhage; IVH: intraventricular hemorrhage; MCA: middle cerebral artery; VA/BA: vertebral artery/basilar artery; VS: vasospasm *p < 0.05, **p < 0.01.
The baseline and follow up DSA were performed 9.7 ± 12.5 hours and 8.5 ± 2.3 days after the SAH onset, respectively. The mean value of the baseline iCCT showed significant prolongation in the patients with DCI as compared to patients without DCI (p = 0.04), but showed no significant difference between patients with favorable and with unfavorable outcome. The follow-up iCCT showed significant prolongation in the patients with DCI (p < 0.01) and in the patients with unfavorable outcome (p < 0.01) as compared to patients without DCI and patients with favorable outcome. There was a significant increase in the differences in the iCCT in the patients with DCI (p < 0.01) and in the patients with unfavorable outcome (p < 0.01).
Assessment of the correlation of iCCT and aVS with DCI and outcome
Based on the above results, the factors that affect DCI and outcome were examined by multivariate analysis.
DCI
Multivariate logistic regression analysis revealed that the follow-up iCCT (OR 5.93, 95% CI 1.42–10.8; p = 0.024) and the differences in the iCCT (OR 8.84, 95% CI 1.69 –37.2; p = 0.015) were significant factors affecting the development of DCI, but factors such as the baseline iCCT, age, IVH, H & H grade, SAH hematoma on CT, clipping, and severe aVS did not attain statistical significance (Table 2).
Multivariate analysis of factors affecting endpoints.
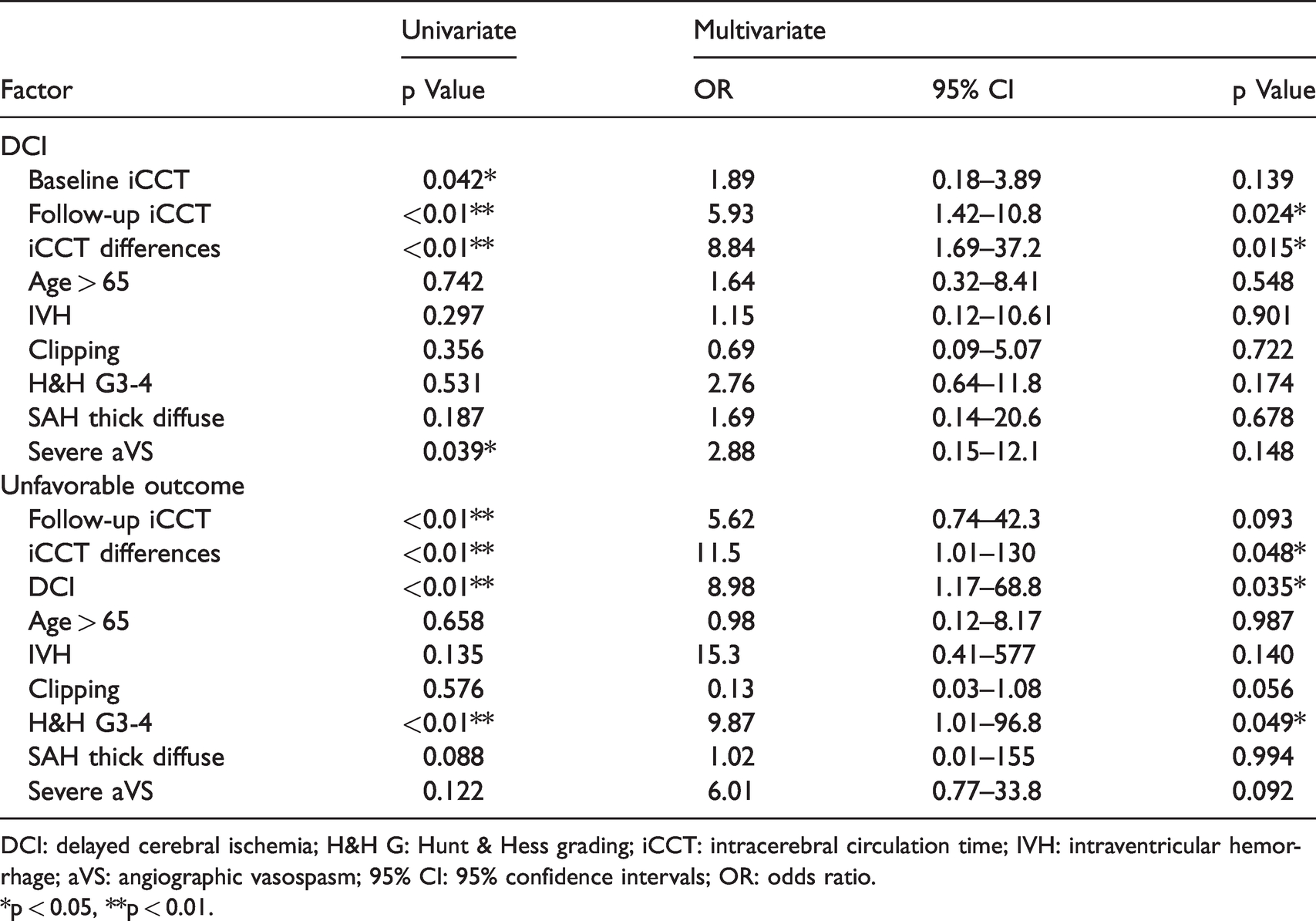
DCI: delayed cerebral ischemia; H&H G: Hunt & Hess grading; iCCT: intracerebral circulation time; IVH: intraventricular hemorrhage; aVS: angiographic vasospasm; 95% CI: 95% confidence intervals; OR: odds ratio.
*p < 0.05, **p < 0.01.
Outcome
Multivariate logistic regression analysis revealed that differences in the iCCT (OR 11.5, 95% CI 1.01–130; p = 0.048), DCI (OR 8.98, 95% CI 1.17–68.8; p = 0.035) and H & H grade (OR 9.87, 95% CI 1.01–96.8; p = 0.049) were significant risk factors for unfavorable outcome (Table 2).
Analysis of the factors affecting an increase of iCCT differences
In order to investigate the factors affecting differences in iCCT, the diagnostic threshold value for the follow-up iCCT and differences in iCCT for the development of DCI were evaluated. Sensitivity and specificity were then calculated. According to the threshold value, we divided the patients into two groups, and multivariate analyses were performed.
Threshold values for the iCCT differences
The ROC curve analysis revealed 4.02 seconds for the follow-up iCCT (73% sensitivity, 76% specificity, AUC 0.73), and 0.38 seconds for differences in the iCCT (89% sensitivity, 88% specificity, AUC 0.90) as possible threshold values that caused unfavorable outcome. The baseline characteristics for the patients as divided into two groups are shown in Table 3. Significant differences between patients with and without an increase in differences in the iCCT were shown in hypertension (p = 0.037), H&H grade (p = 0.041) and severe aVS (p = 0.017).
Demographics and vasospasm-related data for iCCT differences.
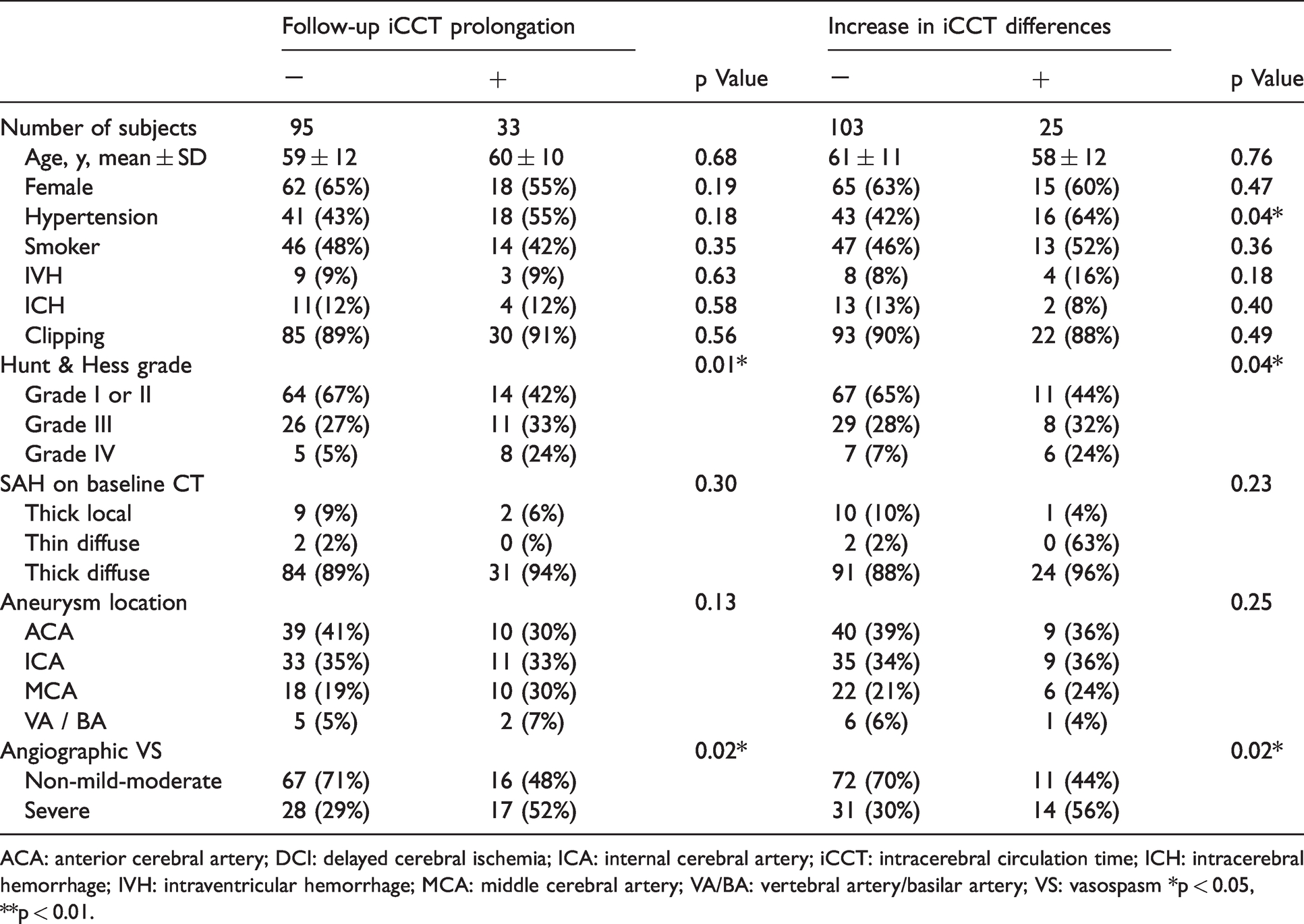
ACA: anterior cerebral artery; DCI: delayed cerebral ischemia; ICA: internal cerebral artery; iCCT: intracerebral circulation time; ICH: intracerebral hemorrhage; IVH: intraventricular hemorrhage; MCA: middle cerebral artery; VA/BA: vertebral artery/basilar artery; VS: vasospasm *p < 0.05, **p < 0.01.
Multivariate analysis
Severe aVS was a significant risk factor in the patients with a follow-up iCCT prolongation (OR 3.48, 95% CI 1.05–11.5; p = 0.04), but other confounding factors such as sex, hypertension, H&H grade, and aneurysm location did not attain a significant statistical difference (Table 4). None of the cofounding factors such as hypertension, H&H grade, IVH, and severe aVS factor demonstrated a significant difference in the patients with an increase in differences in the iCCT (Table 4).
Multivariate analysis of affecting factors of increase in the iCCT differences.
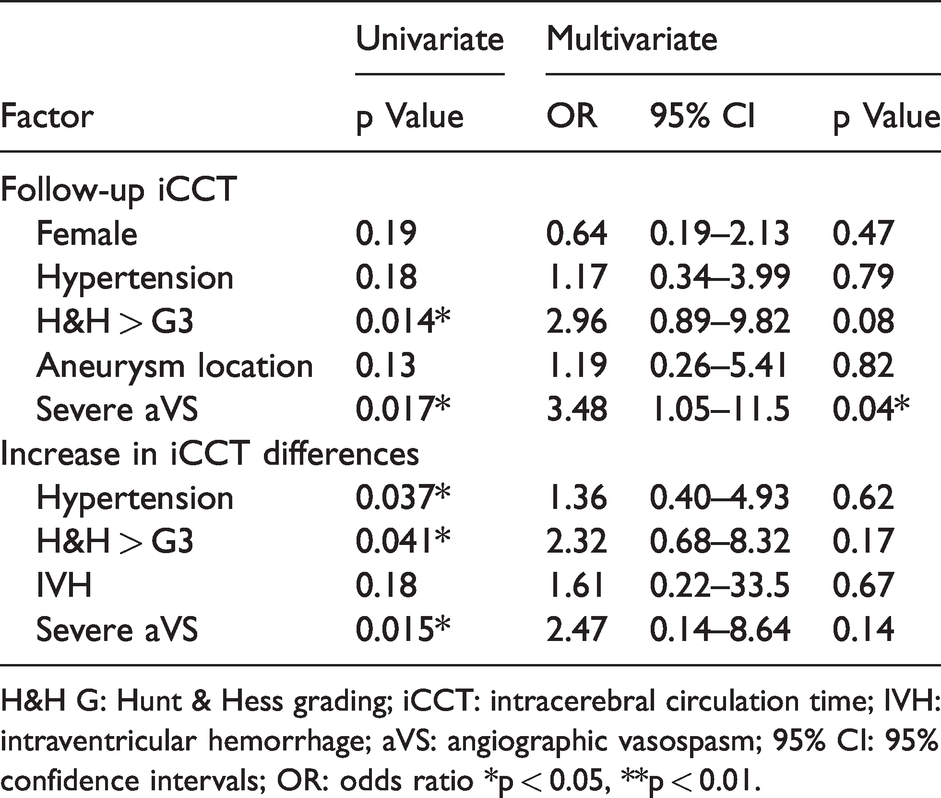
H&H G: Hunt & Hess grading; iCCT: intracerebral circulation time; IVH: intraventricular hemorrhage; aVS: angiographic vasospasm; 95% CI: 95% confidence intervals; OR: odds ratio *p < 0.05, **p < 0.01.
Discussion
After the CONSCOUS-1 trial, a meta-analysis using 14 double-blinded RCTs that investigated the effects of various agents on aVS, DCI and clinical outcome in SAH patients also demonstrated that pharmaceutical treatments significantly decrease the occurrence of aVS, but not that of poor outcome. Another meta-analysis of six RCTs studying the effects of various statins showed the same result as the above analysis.44,45
The results of the present study also suggest that aVS is not important enough to contribute to a reduction in the incidence of DCI and poor outcome. Thus, many recent studies have shown that EBI, which consists of neuronal apoptosis, impaired autoregulation, brain edema, cortical spreading depression (CSD), and microcirculatory dysfunction primarily affects DCI instead of aVS.6,9–11,15,17,46 The present study was performed to clarify the role of microcirculatory dysfunction on DCI and outcome by analyzing CCT, which is considered an indicator of microcirculatory dysfunction.17,47–50
Two studies have examined the relationship between CCT and DCI by using DSA that was performed only in the ultra-early phase after the ictus.22,32 However, the results differed between the two studies with regard to any correlation between increased CCT and DCI. In one study, the prolongation of peripheral CCT was significantly correlated with the occurrence of DCI. 32 The other study measured whole brain CCT and showed no significant difference between the CCT and the development of DCI. 22 Neither of these studies tested the correlation between aVS and CCT. Therefore, a correlation between microcirculatory dysfunction in the ultra-early phase and DCI is suggested but is not definitive. This determination corresponds to the results of the baseline iCCT of the present study, in which a significant correlation of the baseline iCCT with DCI was obtained only when univariate analysis was applied. This result suggests that although there is a possibility of microcirculatory disturbance in the ultra-early phase, severe H & H grade may be more meaningful for outcome in actual practice than iCCT prolongation, as shown in Table 2.
On the other hand, there are no studies on CCT during the subacute phase using DSA. In the present study, the follow-up iCCT measured in the subacute phase was correlated with DCI more than was the baseline iCCT measured during the ultra-early phase. A correlation of the follow-up iCCT with poor outcome, however, was not obtained. These uncertain correlations between microcirculatory dysfunction and DCI and poor outcome not only in the ultra-early phase but also in the subacute phase may be due to the fact that the physiological variation in the CCT of healthy individuals was reported to range between 4.67 s ± 1.08 s and 5.11 s ± 1.00 s.51,52 This range could therefore influence a one-time evaluation of CCT by masking abnormal changes.
Therefore, chronological differences in microcirculatory impairment from the ultra-early phase to the subacute phase were also investigated in the present study. The results showed that an increase in differences in the iCCT could be a significant risk factor for DCI and for poor outcome, which was demonstrated not only by univariate analysis but also by multivariate analysis. These results suggest that the differences in impaired microcirculation themselves, as they are aggravated from the ultra-early phase onward to the subacute phase, are important in DCI onset and poor outcome.
DCI and poor outcome may be affected not only by microcirculatory dysfunction during the ultra-early phase, but also by persistent microcirculatory dysfunction occurring as a part of EBI and continuing until the subacute phase with gradual aggravation. Therefore, differences in the iCCT during the clinical course can serve as an index for predicting DCI onset and poor outcome.
In terms of the mechanisms for microcirculatory impairments, previous clinical and experimental studies revealed that microthrombosis and microvasospasm occurred during both the ultra-early and the subacute phase.13,15,16,41,53–58 Microthrombosis can be induced by systemic hypercoagulability, increased platelet-activation, endothelial cell damage, and microcirculatory stasis.13–16,53–63 Microthrombosis can be induced by systemic hypercoagulability, increased platelet-activation, endothelial cell damage, and microcirculatory stasis.13–16,46,53–58,64 Microvasospasm can also be induced by vasoconstrictors derived from activated platelets or SAH clots, vasodilatory dysfunction due to endothelial impairment, and changed vascular reactivity to vasodilatory and/or vasocontractile stimuli.15,16,55,59–63 Regarding the relationship between microthrombosis and microvasospasm, the current study in vivo revealed that microvasospasm gradually progresses toward the subacute phase, and induces microthrombosis.16,65 An analysis of the factors affecting the prolongation of the iCCT in the present study revealed that aVS was the only factor influencing the follow-up iCCT. Endothelial damage to the major cerebral arteries induced by aVS might activate circulating platelets, thereby resulting in microcirculatory disturbance.
To treat microthrombosis, antiplatelet agents such as aspirin, ozagrel sodium, dipyridamole, and ticlopidine have been tried; however, a systematic review showed some improvement in DCI and outcome could be obtained by single agent administration, but without any unequivocally significant difference. 66 On the other hand, a recent systematic review in which the majority of cases were treated by coil embolization also showed a significant reduction in poor outcome.67,68 Although a recent retrospective study showed that double antiplatelet therapy might be effective, a large-scale RCT will be required to verify its significance.69,70 As another treatment method for microthrombosis, there have been reports showing the effectiveness of ADAMTS13, which has an antiplatelet effect by inhibiting VWF-dependent platelet aggregation. This, however, is still in the experimental stage and has not been clinically applied. 71 Additionally, as a treatment for microthrombosis with anticoagulants, low-dose intravenous heparin infusion has been reported and is associated with a decreased risk of DCI. 72 Based on the above, the efficacy of antiplatelet or anticoagulants agents seems promising, and further investigations on the proper types of agents and appropriate combination of agents as polypharmacy is necessary in order to obtain a more definite clinical effect.
At the same time, there is currently no clinically implemented treatment for microvasospasm. Although the effectiveness of NO inhalation therapy for microvessel spasm has been experimentally proven, its clinical application has not been verified.19,73,74 Several RCTs have shown the efficacy of cilostazol, one of the antiplatelet drugs.36,75 In addition to exerting antiplatelet action by inhibiting PDE3, cilostazol also results in vascular smooth muscle dilatation, and in vitro studies demonstrated that its vasodilatory action is particularly strong in microarteries. 73 Therefore, the efficacy of cilostazol for DCI and poor outcome may be partially attributed to its ameliorative action on microvasospasm. 76 In the future, it will be necessary to elucidate the mechanism of microvasospasm in more detail and to develop a therapeutic method.
As mentioned above, it is necessary to continue to explore treatment methods for microcirculatory disturbance, but methods related to the evaluation of microcirculatory disturbance need to be optimized. In this study, we performed post hoc analysis using DSA-based iCCT to elucidate the pathogenesis of microcirculatory disturbance after aSAH. However, frequent DSA is difficult to perform in clinical practice, and the analysis of the iCCT is not easy because it requires complicated image processing. Currently, new techniques are available as alternatives to DSA. These include arterial spin labelling (ASL) perfusion magnetic resonance imaging (MRI) sequences and/or time-resolved near-infrared spectroscopy, which offer longitudinal, non-invasive measurement of cerebral blood flow and microvascular, dynamic cerebral autoregulation.77–79 These methods can be used for frequent examinations or continuous monitoring, and are expected to be non-invasive methods for the analysis of microcirculatory disturbance in patients with aSAH.
Conclusion
An increase in differences in the iCCT showed a significant correlation with the occurrence of DCI and poor outcome, independent of aVS in aSAH patients. This suggests that greater microcirculatory dysfunction from the ultra-early phase to the subacute phase can be a causative factor for DCI development and poor outcome.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
We thank Mark Inglin (University of Basel) for his editorial assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
MN, NM, NS, and HO were involved in study design and data interpretation. MN, NM, and HO were involved in the data analysis. All authors critically revised the report, commented on drafts of the manuscript, and approved the final report.
