Abstract
Cortical spreading depolarization (SD) imposes a massive increase in energy demand and therefore evolves as a target for treatment following acute brain injuries. Anesthetics are empirically used to reduce energy metabolism in critical brain conditions, yet their effect on metabolism during SD remains largely unknown. We investigated oxidative metabolism during SD in brain slices from Wistar rats. Extracellular potassium ([K+]o), local field potential and partial tissue oxygen pressure (ptiO2) were measured simultaneously. The cerebral metabolic rate of oxygen (CMRO2) was calculated using a reaction-diffusion model. By that, we tested the effect of clinically relevant concentrations of isoflurane on CMRO2 during SD and modeled tissue oxygenation for different capillary pO2 values. During SD, CMRO2 increased 2.7-fold, resulting in transient hypoxia in the slice core. Isoflurane decreased CMRO2, reduced peak [K+]o, and prolonged [K+]o clearance, which indicates reduced synaptic transmission and sodium-potassium ATPase inhibition. Modeling tissue oxygenation during SD illustrates the need for increased capillary pO2 levels to prevent hypoxia. In the absence thereof, isoflurane could improve tissue oxygenation by lowering CMRO2. Therefore, isoflurane is a promising candidate for pre-clinical studies on neuronal survival in conditions involving SD.
Keywords
Introduction
Cortical spreading depolarization (SD) was first described by Aristides Leão more than a half-century ago. 1 Meanwhile, many experimental and clinical studies have detected SD in the course of critical brain disease, e.g. ischemic stroke, subarachnoid hemorrhage, spontaneous intracerebral hemorrhage, or traumatic brain injury (TBI). 2 Importantly, SD has been associated with tissue deterioration, secondary brain injury, and reduced outcomes in patients. 3 The duration of SD-induced spreading depression of cortical activity was the strongest predictor of delayed cerebral infarction in a recent clinical trial on subarachnoid hemorrhage. 4 SD, therefore, evolves as a candidate biomarker for monitoring and treatment after brain insults.
SD is characterized by an extensive translocation of ions between the intracellular and extracellular space, which practically occurs together with membrane depolarization. Neuronal depolarization occurs slightly before astroglial depolarization. 5 While Na+, Ca2+, and Cl− ions enter neurons, K+ ions exit them. The K+ exit seems to occur slightly before the Na+, Ca+, and Cl− entry in time.6,7 Overall, ion changes during SD are the largest that can occur in living neural tissue. 8 As a consequence of neuronal depolarization, voltage-gated Na+ channels are inactivated, which prevents neuronal firing and causes the spreading depression of neuronal activity.9 –11 However, the activity depression usually lasts significantly longer than the SD, suggesting that it is maintained by other mechanisms than the depolarization block, such as intracellular zinc or Ca2+ and/or extracellular adenosine accumulation.12 –14 Morphologically, SD is associated with dendritic and somatic swelling, which is termed cytotoxic edema. 15 To terminate SD, ion gradients have to be restored by increased activity of the sodium-potassium-ATPase (Na+/K+-ATPase). 16 This, in addition to the need for increased tissue clearance of accumulated metabolites, is assumed to be one of the reasons why hyperemia occurs in otherwise healthy tissue during the depolarization phase in mammalian species, such as rats, pigs, cats, and humans, somewhat outlasting tissue repolarization and then followed by mild oligemia. 2 However, despite hyperemia, tissue hypoxia may occur during SD even in otherwise intact tissues, because the increased cerebral metabolic rate of oxygen (CMRO2), which results from increased ATP demand, may outweigh oxygen delivery by increased regional cerebral blood flow (CBF). 17 Only a few studies investigated oxygen consumption indicative of energy demand during SD. Those consistently showed elevations of CMRO2 in rats.17 –19 In addition, brain ATP levels were shown to be reduced following KCl-induced SD in non-injured cortex in rats, 20 indicating that ATP consumption exceeds production. Following brain insults, SD can aggravate brain damage. 3 The situation becomes even more dangerous for the tissue when the neurovascular response to SD becomes inverse and instead of initial vasodilation and spreading hyperemia, severe vasoconstriction and spreading ischemia co-occur with simultaneously increased CMRO2.21 –25 In the clinic, various conditions, such as subarachnoid hemorrhage, the penumbra in ischemic cerebral infarction, and TBI show a very similar continuum as in other mammals and especially in rats, ranging from brief hyperemic/hypoxic to prolonged ischemic/severe hypoxic responses to SD.26 –29 In addition to normalizing the inverse hemodynamic response to SD in at-risk tissues, the reduction of SD-associated metabolic stress with a lower elevation of CMRO2 and corresponding lower hypoxia provides another conceivable and testable therapeutic option to reduce SD-induced secondary brain injury.
As anesthetics are known to reduce brain metabolism,30 –33 they may reduce SD-associated damage that results from a mismatch of energy supply and demand. In addition, anesthetics may modulate or even prevent SD as shown for ketamine and isoflurane in animals and ketamine in patients.34 –39 Empirically, deep anesthesia is used for neuroprotection in severe brain diseases, such as status epilepticus, TBI, stroke, and intracranial hypertension.40,41 However, the level of evidence for these indications of anesthesia is uncertain. 42 Of note, deep anesthesia has been associated with poor outcomes after surgical treatment and neurocritical care,43 –45 which implies that patients eligible to receive anesthesia for potential neuroprotection must be carefully selected. To choose the anesthetic with optimal protective properties and low neurotoxicity, it is of great interest to understand their effects on neuronal metabolism including the underlying molecular mechanisms. Anesthetics influence energy demand in the brain by suppressing neuronal activity,33,46 generating changes in regional CBF,33,47 and direct inhibition of mitochondrial enzymes.46,48 In particular, the gas anesthetic isoflurane has been shown to reduce cerebral metabolism by inhibiting synaptic transmission and network activity, e.g. by inhibiting N-methyl-D-aspartate receptors at the glycine site.33,49 In addition, transmitter release may be reduced due to direct inhibition of complex I of the respiratory chain in presynaptic terminals. 48 Moreover, isoflurane was shown to impair the Na+/K+-ATPase, 50 the major ATP consumer in the brain. 51 Anesthetics that limit the great energy demand during SD and preserve metabolism may subsequently reduce hypoxia in acutely brain-injured patients.
In this study, we tested the effects of isoflurane on oxygen consumption during SD in neocortical slices of Wistar rats. We took advantage of the acute brain slice preparation to study neuronal energy demand under a constant supply of nutrients and oxygen. To quantify SD-associated metabolic demand and its modulation by isoflurane, we integrated recordings of the local field potential (LFP), extracellular potassium ([K+]o), and partial tissue oxygen pressure (ptiO2) during SD with in silico calculation of CMRO2. Using a tissue model, we extrapolated changes in CMRO2 during SD to oxygen availability in vivo for a range of previously reported capillary pO2 values.
Material and methods
Animals
This study was conducted in nine male and five female Wistar rats (Janvier Labs, weight: 250 g, age: ∼8 weeks) and complies with the ARRIVE 2.0 and the Charité Animal Welfare Guidelines. The experimental protocols were approved by the State Office of Health and Social Affairs of Berlin (T-CH0039/21). Before experiments, the animals had at least seven days for acclimation and were housed in groups of two with access to food ad libitum and a 12-h light/dark cycle.
Slice preparation and maintenance
Animals were anesthetized using isoflurane/N2O (1.5%/70%, respectively) and decapitated. The brain was gently removed and coronal slices from the frontal cortex (thickness: 400 μm) were prepared with a Leica VT 1200 S vibratome (Wetzlar, Germany). Slices were immediately transferred to an interface chamber, where they were supplied with humidified carbogen (95% O2, 5% CO2, 1L/min, temperature ∼36 ± 0.5 °C) from the top and carbogenated artificial cerebrospinal fluid (aCSF) from the bottom at a flow of 2 mL/min. 52 The aCSF contained (in mM): 129 NaCl, 26 NaHCO3, 10 glucose, 3 KCl, 1.25 NaH2PO4, 1.6 CaCl2, and 1.8 MgCl2 (osmolarity 295–305 mosmol/L, pH 7.35–7.45, temperature ∼36 ± 0.5 °C). Experiments started two hours after slicing.
Electrophysiological and ptiO2 recordings and SD induction
Simultaneous LFP, [K+]o, and tissue ptiO2 measurements were performed in layer 2 using double-barrel ion-sensitive microelectrodes and Clark-type oxygen sensors (10 µm tip; Unisense, Aarhus, Denmark) as reported previously. 53 Baseline CMRO2 was calculated based on ptiO2 measurements in vertical steps of 20 μm beginning at the slice surface until reaching the minimum of ptiO2 (i.e. core) as described previously. 54 Stepwise measurements are time-consuming and therefore do not allow dynamic measurements during SD. To overcome this limitation, following baseline measurements, three oxygen probes were inserted at different depths (40 µm, 100 µm, and the measured core; Figure 1) in the same region. Ion-sensitive microelectrodes were manufactured using Potassium Ionophore I (Fluka, Buchs, Switzerland) as previously described. 55 Recorded potentials were converted to [K+]o in mM using Nernst’s equation and assuming baseline [K+]o of 3 mM. Oxygen electrodes were polarized overnight and two-point calibrated before experiments. SDs were induced by local application of 3 M KCl at >200 μm from the recording sites with a glass micropipette before, during, and after the application of isoflurane. Data concerning LFP and [K+]o recorded in five animals are published by Reiffurth et al., 2023. 50 To exclude significant changes on SD properties and energy demand due to repeated SD induction with KCl, we performed control experiments without pharmacological treatment with isoflurane. For this purpose, four consecutive SDs were induced with repeated local application of 3 M KCl every 10–12 minutes (see Supplementary Fig. 1).
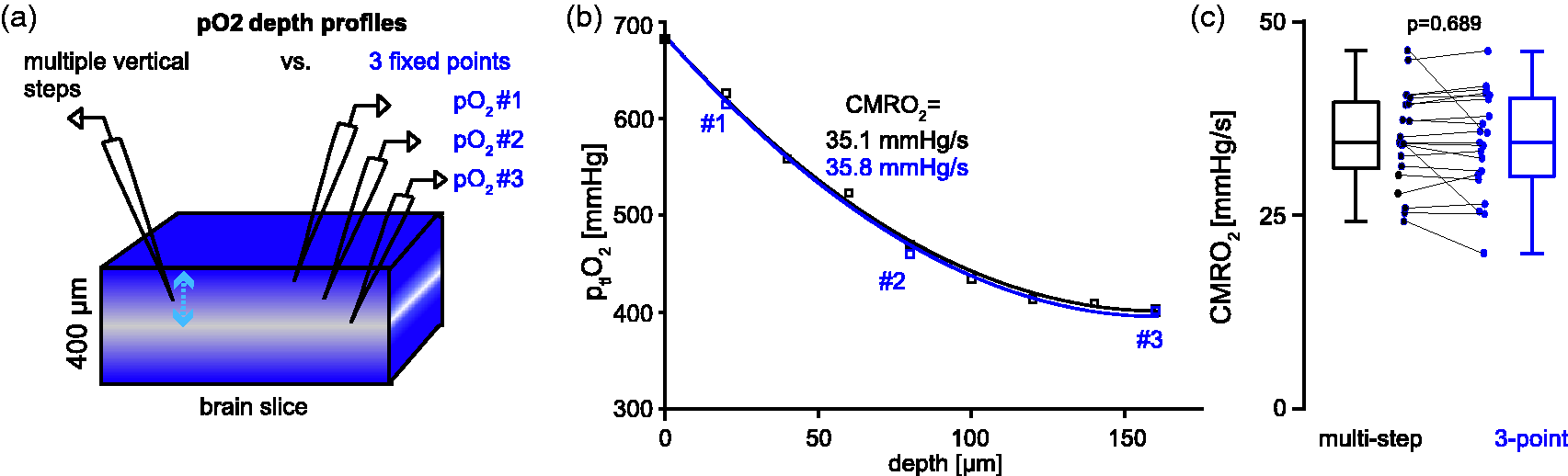
Three-point partial tissue oxygen pressure (ptiO2) recordings and cerebral metabolic rate of oxygen (CMRO2) calculations. a) Exemplary setup for measuring ptiO2 depth profiles using either one Clark-Style electrode for multiple consecutive vertical steps (left) from the slice surface to the core (blue arrow) in steps of 20 µm (left) or three electrodes at fixed positions from the surface to the core (right). Three fixed electrodes were used to increase temporal resolution, which allowed dynamic measurements during spreading depolarization (SD) (cf. Figure 2(b)). b) Exemplary baseline (pre-SD) ptiO2 depth profiles comparing multiple-vertical step recordings (black) with recordings by three fixed electrodes (blue). c) Bar graph summarizing baseline CMRO2 calculations based on multi-step and three-point ptiO2 recordings (n = 24 slices, ten animals, p = 0.69, Wilcoxon signed-rank test). Note the similarity of depth profiles (B) and subsequent CMRO2 calculations (C) for multi-step and three-point recordings.
Isoflurane application
Isoflurane was applied to an interface recording chamber with carbogen using a calibrated isoflurane vaporizer (Dräger, Germany) connected between the gas supply and the recordings chamber (constant gas flow rate of 1.5 l/min). The concentration of isoflurane was titrated to 1 and 3% vol. and controlled using a Vamos® mobile isoflurane monitor connected to a gas supply (Dräger, Germany). The recording temperature was maintained at ∼36°C. Given a water/gas partition coefficient of 0.5424 at 37 °C, the application of 1% and 3% correspond to 0.24 mM and 0.72 mM isoflurane in the aCSF, respectively. 56
Data acquisition and analysis
Analog signals were digitalized with Power CED1401 and Spike2 software (Cambridge Electronic Design, Cambridge, UK). Changes in ion concentrations were calculated using a modified Nernst equation as a mathematical expression for the generation of virtual channels on Spike2. Analyses and statistics were performed using Spike2, Excel (Microsoft, Seattle, USA), MATLAB (MathWorks Inc., Natick, MA, USA), and Origin (Version 6, Microcal Software, Northampton, USA).
Calculation of CMRO2
CMRO2 was calculated from ptiO2 depth profiles as previously described. 52 In short, we applied a reaction-diffusion model consisting of diffusive O2-transport and O2-consumption within the slice. Slices were divided into layers with an equal thickness of 1 μm. The diffusive oxygen distribution between the layers is described by Fick’s Law with a diffusion constant of 1.6 × 103 μm2/s. The oxygen consumption rate within each layer is given by Michaelis-Menten kinetics (Km-value: 3 mmHg). The CMRO2 was assumed to be homogeneous throughout the slice and is treated as an adjustable parameter to match the experimental data. For the boundary conditions, the ptiO2 concentration at the slice surface was fixed to the supply value, while at the ptiO2 minimum, the diffusive oxygen transport was put to zero.
Modeling perivascular oxygen diffusion
To simulate in vivo oxygen availability based on the calculated CMRO2 values during SD, we used a two-dimensional tissue model.57,58 The tissue that needs to be supplied is modeled as a cylinder constituted by a central capillary, which provides oxygen to the surrounding metabolically active neuronal tissue. Such a model was first described by A. Krogh. 59 The radius of the Krogh cylinder is assumed to be in the range of 10–35 μm corresponding to reported radiuses of perivascular diffusion cylinders including the repeatedly reported average intercapillary distance of 40 μm (i.e., two Krogh cylinders with a radius of 20 μm).60,61 Oxygen diffuses from the vessel into the surrounding tissue where it is consumed. Oxygen diffusion is modeled by a compartmental discretization subdividing the cylinder in a vessel compartment and concentric tissue compartments around the vessel with a thickness of 1 µm. It is assumed that no oxygen leaves the cylinder, which is equivalent to the assumption that the outflow of oxygen from the represented region is equal to the inflow from neighboring regions.
Statistical analysis
This was an exploratory study. We chose sample sizes that are common in the field and based on our own experience. Slices were subject to standardized wash-in protocols after recording SDs in standard conditions. This experimental design precluded the need to allocate slices to separate groups randomly and for anonymizing. No data were excluded.
Data were not normally distributed and are reported as median (25th, 75th percentile). Statistical inference was based on the Wilcoxon signed-rank test. P-values were adjusted for multiple comparisons by Bonferroni post hoc correction. Drug effects of 1% and 3% isoflurane were each compared with control but not with each other. Changes were stipulated to be significant for p-values <0.05.
Results
Three-point recordings allow measurements of ptiO2 depth profiles and CMRO2 with a high temporal resolution
Previously, calculations of CMRO2 relied on ptiO2 depth profiles acquired by vertical movement of one Clark-style oxygen-electrode in constant steps through acute brain slices (Figure 1(a) and (b)).30,46,52,54 This method can be used to capture steady-state CMRO2, but lacks the temporal resolution necessary for capturing the transient paroxysmal changes in ptiO2 during SD, which reach a minimum seconds after onset (cf. Figure 2(b)). To overcome this limitation, we established multielectrode recordings of ptiO2-depth profiles using three stationary electrodes inserted at fixed vertical positions in the slice (Figure 1(a)). We verified the accuracy of stationary three-electrode recordings with multi-step single electrode measurements (>7 vertical positions) at baseline before SD induction. As shown in Figure 1(b), depth profiles and CMRO2s were similar for single-electrode multi-step and stationary multi-electrode measurements (n = 21 slices, ten animals, p = 0.689, Wilcoxon signed-rank test), respectively (Figure 1(c)).
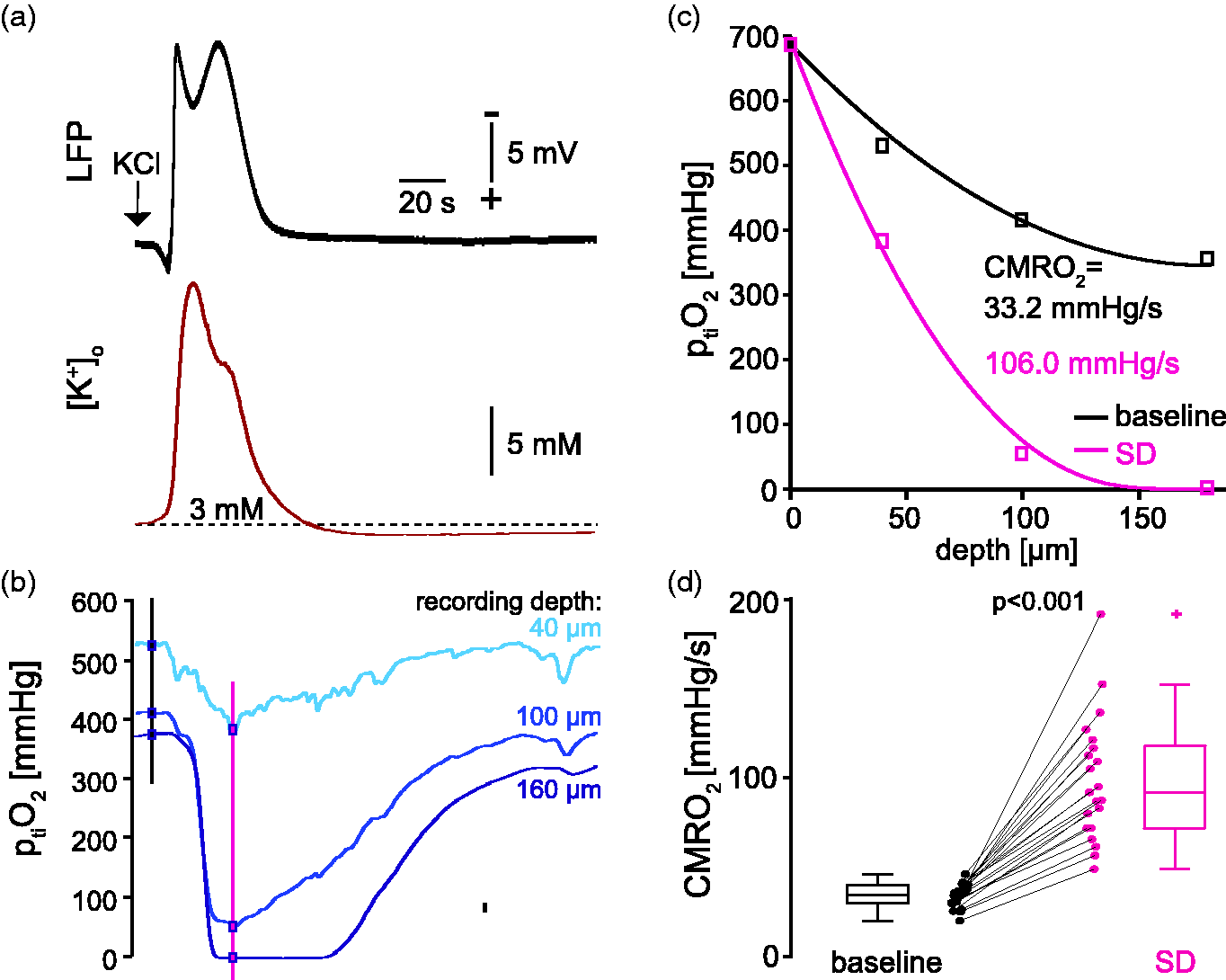
Exemplary time-dependent ptiO2 traces for multi-electrode recording during SD. a) Direct current (DC) local field potential (LFP) (top) and [K+]o recording (bottom) of one SD induced by a KCl droplet. b) Parallel recording of ptiO2 at a depth of 40 (top), 100 (middle), and 160 µm (bottom) corresponding to the SD in (A). Vertical lines indicate time points of baseline and SD-associated depth profiles (black and magenta, respectively) used to calculate CMRO2 (cf. C). Note that the DC potential and the [K+]o in A) return to baseline within ∼1 min while the ptiO2 is still reduced. c) Depth profiles taken before (black) and during SD (magenta) from the example shown in (B) and each corresponding CMRO2. d) Summary of CMRO2 values before (baseline) and during SD (n = 21 slices, ten rats, p < 0.0001, Wilcoxon signed-rank test).
SD causes a 2.7-fold increase in CMRO2
Next, we used the multi-electrode measurements to record rapid changes in the ptiO2 depth profiles and the related CMRO2 during SD. Focal application of KCl reliably initiated SD, which showed the characteristic negative deflection of the direct current (DC) potential and the increase in [K+]o (Figure 2(a)). Exemplary traces for the three stationary oxygen electrodes demonstrate reduced ptiO2 in all depths. Of note, the return of ptiO2 to baseline levels after SD outlasted the recovery of the DC potential and of [K+]o. In the slice core, ptiO2 dropped below the hypoxia threshold of ∼8 mmHg at which oxidative metabolism breaks down (Figure 2(b)). 62 Under a constant supply of oxygen, a drop of ptiO2 indicates increased oxygen consumption. Indeed, CMRO2 increased from 33.2 at baseline to 106.0 mmHg/s during SD in this example (Figure 2(c)) and ∼2.7-fold from 34.4 (30.8, 40.1) mmHg/s to 92.0 (72.0, 117.9) mmHg/s in summary (n = 21 slices, 10 animals, p < 0.0001, Wilcoxon signed-rank test, Figure 2(d)).
Isoflurane reduces CMRO2, peak [K+]o, and prolongs SD and [K+]o decay during SD
We then investigated the influence of isoflurane on oxidative metabolism and [K+]o dynamics during SD (Figure 3). We used concentrations clinically known to induce light and deep anesthesia, i.e., 1% and 3%. To avoid slice-dependent variability, a first SD was induced during perfusion of aCSF and served as a slice internal reference for subsequent SDs during the application of isoflurane and washout (Figure 3(a)). Neither 1% nor 3% isoflurane prevented the induction of SD and also had no significant effect on the SD-associated changes of the DC amplitude (Figure 3(a) and (d)). However, the duration of the SD-associated negative DC shift was prolonged from 43 sec (38, 55) to 60 sec (56, 83) and 107 sec (97, 134) for 1% and 3% isoflurane, respectively (Figure 3(e), n = 13 slices from nine rats, p = 0.0034 and p = 0.0015, respectively, Wilcoxon signed-rank test & Bonferroni correction). Furthermore, the example in Figure 3(a) shows a dose-dependent reduction of SD-associated [K+]o peaks, a prolongation of the [K+]o-decay, and an increase of minimal ptiO2 levels above the hypoxia threshold. Corresponding depth profiles of ptiO2 and associated CMRO2 showed that isoflurane reversibly reduced oxygen consumption before and during SD (Figure 3(b)). We also calculated the CMRO2 continuously for 5 min following SD onset, which shows that 3% isoflurane reduced CMRO2 during the initial phase of SD, characterized by the negative shift of the DC potential and the rise in [K+]o, and also during the phase where [K+]o returns to baseline levels (Figure 3(c)). The cumulative CMRO2 that exceeded baseline CMRO2 was approximately halved by 3% isoflurane during these 5 min. Before SD, CMRO2 was reduced from 38.9 mmHg/s (30.2, 41.0) during perfusion of aCSF to 34.7 mmHg/s (26.1, 36.8) following wash-in of 3% (but not 1%) isoflurane (p = 0.001, n = 12 slices, eight rats, Wilcoxon signed-rank test & Bonferroni correction). During SD, isoflurane significantly reduced the increase in CMRO2 (ΔCMRO2) from 51.8 (40.8, 86.6) mmHg/s during perfusion of aCSF to 39.4 (26.3, 68.3) and 18.4 (10.0, 22.7) mmHg/s at 1% and 3% isoflurane, respectively (p = 0.003 and p = 0.002, n = 12 slices, eight rats, Wilcoxon signed-rank test & Bonferroni correction, Figure 3(i)). In addition, isoflurane reduced the proportion of slices affected by core hypoxia from 58% (7 out of 12 slices) to 33% (4 out of 12 slices) and 17% (2 out of 12 slices) at 1% and 3% isoflurane, respectively. The peak change in [K+]o (Δ[K+]o) during SD was reduced from 22.6 mM (17.8, 34.8) to 16.5 mM (13.0, 26.1) at 3% isoflurane (p = 0.001, n = 13 slices, nine rats, Wilcoxon signed-rank test & Bonferroni correction, Figure 3(f)), whereas 1% isoflurane had no significant effect. In addition to Δ[K+]o, we analyzed the [K+]o decay time to 50% (T150 [K+]o) and 10% (T250 [K+]o) of the peak Δ[K+]o during SD. 3% isoflurane increased both T150 and T250 from 20.0 sec (16.2, 35.3) to 38.3 sec (29.1, 82.9) and from 65.0 sec (47.7, 95.3) to 235.7 sec (139.5, 277.5), respectively (Figure 3(f) and (g), p = 0.01 and p = 0.002, respectively, n = 13 slices, nine rats, Wilcoxon signed-rank test & Bonferroni correction), whereas 1% isoflurane did not significantly alter [K+]o decay times. Despite some slice- and sex-dependent heterogeneity, the effects of isoflurane were similarly observed in most slices and in males and females (Figure 3(d) to (i), Supplementary Table 1). Importantly, the effects of isoflurane on CMRO2, DC shift duration, Δ[K+]o, T150 [K+]o, and T250 [K+]o were transient as washout of isoflurane partly restored all parameters (Figure 2(a) to (c), (e) to (h)). Furthermore, in control experiments in the absence of isoflurane, all measured parameters were stable during four consecutive SDs (n = 8 slices, three rats, Supplementary Fig. 1). In summary, isoflurane significantly prolonged SD duration and [K+]o recovery, whereas Δ[K+]o and CMRO2 were reduced.
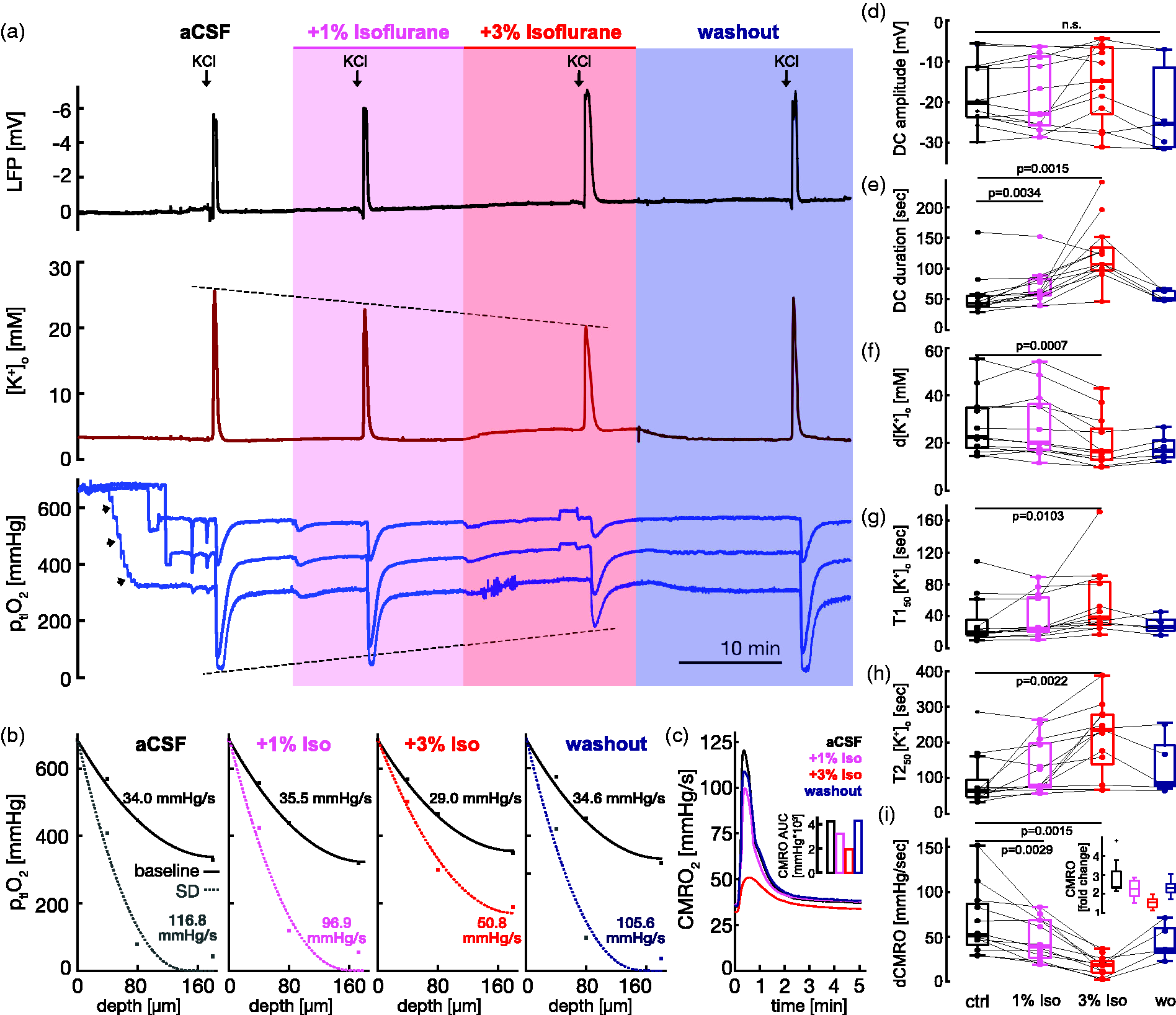
Effect of isoflurane on LFP, [K+]o, ptiO2, and CMRO2 during SD. a) Exemplary recordings of four KCl-induced SDs during perfusion of aCSF with the addition of 1% and 3% isoflurane, and during washout of isoflurane by aCSF. Arrows labeled with ‘KCl’ indicate the time point of KCl application by a glass microelectrode. Recorded were (from top to bottom) LFP, [K+]o, and ptiO2 at a depth of 40, 80, and 180 µm. Arrowheads in ptiO2 recording point to measurements of multi-step depth profiles at the beginning of the experiment. This electrode remains in the slice core thereafter. Note the successive lowering of peak [K+]o and the increase in ptiO2 during SDs at 1% and 3% isoflurane. b) Depth profiles of ptiO2 before and during SD for the four conditions shown in (A), i.e. aCSF, aCSF + 1% isoflurane, aCSF + 3% isoflurane, and washout with aCSF. CMRO2s were calculated and displayed in the corresponding depth profiles. c) Dynamic display of CMRO2 for 5 min following SD onset corresponding to the example shown in A. Note the reduction of CMRO2 by 3% isoflurane at all time points relative to the onset of SD. The inset shows the cumulative CMRO2 calculated as the area under the curve (AUC) for the first 5 min following SD onset. Summary boxplots of d) SD-associated DC amplitudes, e) DC shift duration, f) Δ[K+]o, g) T150 [K+]o, h) T250 [K+]o, and i) CMRO2. The inset in I shows the change in CMRO2 during SD relative to the baseline before each SD.
Isoflurane improves tissue oxygenation
Given the 2.7-fold increase of CMRO2 during SD in vitro, we investigated how this would affect tissue oxygenation in a tissue model. Based on pO2 in the range of 20–55 mmHg, which has been recorded in capillaries of anesthetized and awake rodents,60,63,64 we simulated minimal ptiO2 within perivascular Krogh cylinders, i.e. at the edge of the cylinder, with radiuses in the range of 10 to 35 µm (see Figure 4(a)).58,60,61 For a capillary pO2 as low as 20 mmHg, we found oxygen supply >8 mmHg for Krogh cylinders with radiuses of up to 27 µm under baseline CMRO2 (see the top panel in Figure 4(a)). This extends beyond the midline of the reported average cortical intercapillary distance of ∼40 µm58,60,61 and thus shows sufficient oxygen supply to most brain tissue. However, the increased CMRO2 during SD induced a severe drop in ptiO2 thereby locally shifting the hypoxia boundary (ptiO2<8 mmHg, black line in Figure 4(a)) into the reported size range of Krogh cylinders. 60 This highlights the need for an increase in capillary pO2 during SD to prevent hypoxia in some, although certainly not all brain tissue. To evaluate the effect of isoflurane, we repeated the simulation using the CMRO2s determined for 1% and 3% isoflurane. 1% isoflurane increased the radius of Krogh cylinders expected to receive >8 mmHg O2 during but not before SD, whereas 3% improved both baseline and SD-associated tissue oxygenation (Figure 4(b) and (c)).
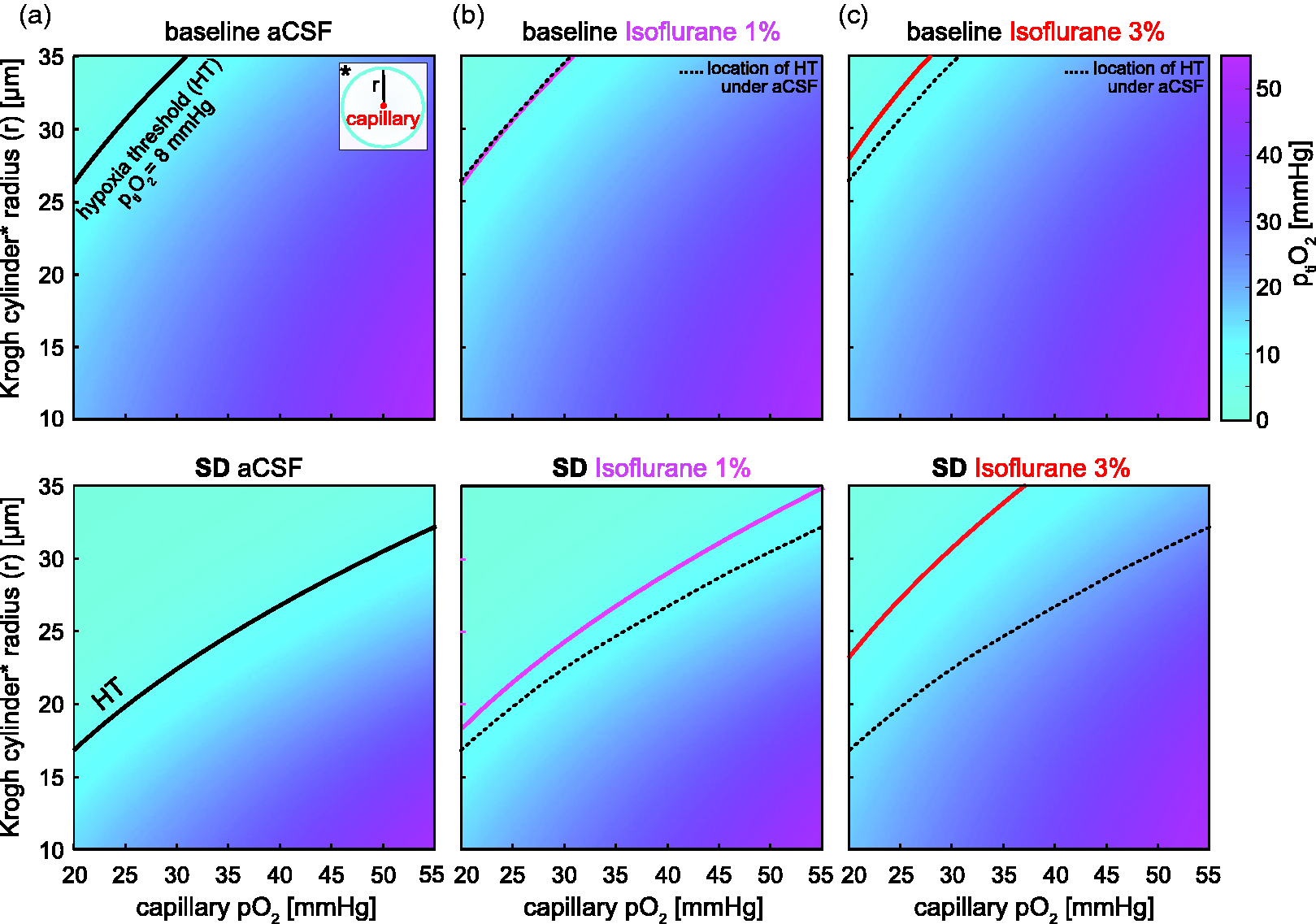
Modeling of oxygen diffusion based on CMRO2 during SD. a) Modeling of minimal ptiO2 levels depending on the radius (r) of Krogh cylinders (y-axis and inset) and the capillary pO2 (x-axis). The top and bottom panels show ptiO2 for the average CMRO2 measured before and during an SD, respectively, under the perfusion of aCSF. The black line indicates capillary pO2-dependent radiuses of Krogh cylinders where the hypoxia threshold (HT, ∼8 mmHg) would be reached. Note the considerable shift to smaller Krogh cylinders during SD. b) Panels equivalent to A for baseline (top) and SD-associated CMRO2 (bottom) under 1% isoflurane. Note that isoflurane increased the range of Krogh cylinders above HT during SD but not for baseline CMRO2. c) Panels equivalent to A for baseline (top) and SD-associated CMRO2 (bottom) under 3% isoflurane. Note that isoflurane increased the range of Krogh cylinders above HT before and during SD. The dotted black line (b,c) indicates the location of the HT in isoflurane-free aCSF as shown in a.
Discussion
Tissue oxygenation reflects the balance of oxygen supply and CMRO2. Monitoring tissue oxygenation with parenchymal probes has become a standard procedure after subarachnoid hemorrhage and TBI, i.e. conditions during which SD has been recorded.3,26,27,65,66 However, CMRO2 during SD has been studied only sparsely, although it is known that the associated energy demand increases dramatically. 3 We, therefore, performed quantitative measurements of CMRO2 during SD in acute brain slices. In vivo, SD was shown to cause temporary hypoxia despite vasodilation in some cases. 17 Therefore, lowering CMRO2 could improve tissue oxygenation and prevent hypoxia. Importantly, we have previously shown that isoflurane lowers cerebral metabolism, while neurovascular coupling to burst activity and mitochondrial respiration were preserved 33 . This made isoflurane a promising candidate for testing its lowering effect on CMRO2 during SD.
CMRO2 increases 2.7-fold during SD
We investigated CMRO2 during SD in acute brain slices, which allows a constant supply of oxygen. Therefore, changes in ptiO2 depth profiles directly reveal changes in CMRO2. Using novel three-point recordings, we were able to continuously monitor ptiO2 depth profiles and thereby provide calculations of peak CMRO2 during SD in vitro. We found that CMRO2 increased ∼2.7 fold during SD. Quantitatively, this increase in CMRO2 was higher than in anesthetized rats (1.5–1.7-fold change) in vivo.17 –19 CMRO2 calculations in vivo were derived from CBF measurements combined with ptiO2 recordings17,19 or combined with arterial-venous pO2 differences. 18 The quantitative difference between these in vivo and our in vitro measurements, may have several reasons: (a) Supply of surplus oxygen via carbogen in vitro may allow greater CMRO2 than in vivo where provided oxygen may be fully consumed at lower CMRO2. (b) Neuronal activity before SD may be lower in vitro, which would augment the relative change in CMRO2 during SD in our experiments; (c) The absence of confounding effects of CBF may allow more accurate assessment of CMRO2 in vitro; and (d) due to the high temporal resolution of our recordings, we may have captured greater peak CMRO2.
Compared to our other in vitro studies, the SD-related increase in CMRO2 (2.7-fold) was significantly higher than during other energy-demanding types of network activity, such as gamma oscillations or seizure-like events, which induced a 1.3-fold and 1.4-fold increase in CMRO2, respectively.30,54 This underlines the great metabolic demand induced by SD.
Isoflurane decreases CMRO2 during SD: possible mechanisms in light of [K+]o dynamics
Isoflurane was shown to reduce CMRO2 in animals at concentrations that induced burst suppression,33,67,68 and in 12 patients suffering from subarachnoid hemorrhage. 69 We show that isoflurane similarly reduced CMRO2 during SD by approximately 5% and 35% at concentrations of 1% and 3%, respectively (Figure 3(g), inset). These concentrations of isoflurane were chosen because they clinically correspond to light (1%) and deep (3%) anesthesia.
Isoflurane has multiple molecular targets that could reduce CMRO2 during SD, and [K+]o recordings provide mechanistic hints. Isoflurane lowered peak [K+]o thereby limiting the demand for oxidative metabolism to restore the transmembrane K+ gradient after SD. The mechanisms underlying the initial neuronal release of K+ have not been resolved. Subsequently, it is not known how isoflurane lowers peak [K+]o. Reduced synaptic transmission under isoflurane may explain lower peak [K+]o values in the early phase of SD, when typically [K+]o and the extracellular concentration of glutamate rise rapidly (see Figures 2 and 3 and Menyhart et al., 2022). 70 Application of glutamate has been shown to elevate [K+]o. 71 In parallel with the initial glutamate release and increase in [K+]o, the frequency of excitatory postsynaptic potentials increases during SD, 72 although inactivation of voltage-gated Na+ channels causes depression of activity at the network level. Synaptic transmission could be reduced by isoflurane by inhibition of NMDA receptors, 49 increased GABAergic input, 73 opening of 2-pore-domain potassium channels, 74 and impaired presynaptic Ca2+ influx. 75 Furthermore, inhibition of mitochondrial complex I in presynaptic terminals by isoflurane has been shown to impair transmitter release. 48 Together, these effects could reduce initial synaptic transmission during SD and thereby lower activity-dependent energy demand.
In contrast to the lowering of peak [K+]o, prolonged [K+]o clearance by isoflurane cannot be explained by synaptic effects. However, this might result from reduced Na+/K+-ATPase activity. 76 Conversely, the reduction in peak [K+]o during SD was shown to be insensitive to pharmacological inhibition of the Na+/K+-ATPase 76 and therefore cannot be explained by its reduced activity. Of note, we recently found a dose-dependent impairment of the Na+/K+-ATPase containing α2/3 subunits in the cortex of rat brains by isoflurane. 50 Similar to [K+]o clearance, the return of [Na+]o to baseline levels following SD was prolonged in those experiments, which is in line with the inhibition of the Na+/K+-ATPase. 50 Since the Na+/K+-ATPase is the greatest energy consumer in the brain, 51 a decrease in its activity lowers oxygen consumption. Still, inhibition of the Na+/K+-ATPase may cause adverse events. The prolonged reduction of [Na+]o following SD in the presence of isoflurane may have secondary effects on Ca2+ homeostasis, i.e. it may prolong the cellular Ca2+ overload, which could promote death signaling. 77 One of the main transporters for the efflux of intracellular Ca2+ is the Na+/Ca2+ exchanger (NCX), which exchanges three extracellular Na+ ions for one intracellular Ca2+ ion. 78 Elevated [Na+]i, as indicated by reduced [Na+]o, inhibits the Ca2+ exit mode of the NCX. Therefore, prolonged elevation of [Na+]i, as during Na+/K+-ATPase inhibition by isoflurane, may extend the intracellular Ca2+ surge during SD. On the other hand, ATP has been shown to increase the affinity of the NCX for intracellular Ca2+ and extracellular Na+ by enabling the phosphorylation of the transporter. 78 Isoflurane may thus facilitate Ca2+ efflux via the NCX if it indeed prevents ATP shortages. Similarly, isoflurane may increase Ca2+-ATPase activity, although this is thought to contribute less to Ca2+ efflux than NCX due to a lower turnover rate. 79 Isoflurane may also lower the SD-induced Ca2+ load itself because the dendritic Ca2+ influx during SD was shown to depend on NMDA receptor activation, 72 which isoflurane inhibits. 49 In addition, experiments on mouse brain slices suggest that the magnitude and mechanisms of the neuronal Ca2+ increase differ for SDs induced by hypoxia or by high [K+]o under normoxia. 80 Therefore, the effect of isoflurane on neuronal Ca2+ dynamics may also depend on the trigger of SD. In summary, while we provide evidence that isoflurane lowered CMRO2 during SD by reducing synaptic transmission as well as Na+/K+-ATPase activity, the neuronal outcome has yet to be investigated.
Several studies on cellular and functional outcomes following experimental ischemia suggest the protective effects of isoflurane. Isoflurane was shown to be neuroprotective in postnatal day 10 pups that underwent unilateral carotid ligation. 81 Isoflurane also reduced cell damage in acute rat brain slices that underwent a period of oxygen-glucose deprivation. 82 Following middle cerebral artery occlusion, isoflurane reduced infarct volume and improved neurological outcome 24 hours 82 and four weeks after stroke.83,84 The suggested mechanisms that underlie neuroprotection include the sphingosine-1-phosphate/phosphatidylinositol-3-kinase/Akt pathway, activation of nuclear factor-κB, production of interleukin-1β, and increased expression of B-cell lymphoma-2 (Bcl-2) protein, all of which putatively result from hypoxia. Therefore, the reduction of secondary hypoxia during SD or prevention of SD by isoflurane, as shown for KCl-induced SD in non-ischemic tissue, 39 may be a missing link to neuroprotection in these studies. In this context, it is of interest that isoflurane was shown to increase CBF in anesthetized rabbits and humans,85,86 presumably due to vasodilation, which in turn may improve tissue oxygenation and thus add to the putative protective effects secondary to reduced CMRO2. Of note, we did not demonstrate complete reversibility of isoflurane effects in all experiments. This could limit applicability, e.g. due to side effects of prolonged Na+/K+-ATPase inhibition (see previous paragraph).
Translational relevance of reduced CMRO2 during SD
We next asked to what extent a reduction of CMRO2 during SD could improve tissue oxygenation. The tissue model predicted that SD will lead to a transient episode of hypoxia in some brain tissue in the absence of increased oxygen supply, e.g. by a physiological hyperemic blood flow response, which is typical in otherwise healthy tissue 2 . In conjunction with the experimental data, the tissue model generates and supports the hypothesis that it is feasible to lower CMRO2 during SD to a degree that would prevent critically low ptiO2 levels within the range of reported intercapillary distances.60,61 Therefore, the application of isoflurane could be beneficial in conditions when resting state CBF is maintained but the capacity to increase the supply of oxygen and energy-rich substrates by neurovascular coupling is impaired. This could affect pathological conditions such as subarachnoid hemorrhage, TBI, or the ischemic penumbra.26 –29,87
In conclusion, clinically relevant concentrations of isoflurane could improve tissue oxygenation after SD by lowering the demand for oxidative metabolism, especially when the neurovascular coupling is impaired. Of note, the inhibitory effects of isoflurane on Na+/K+-ATPase activity could weaken or outweigh the protective effects of improved tissue oxygenation. Previously demonstrated neuroprotective effects of isoflurane following ischemia are a promising observation. Based on our data, we propose further studies focusing on metabolic effects during SD.
Study limitations
For the calculation of CMRO2, we assumed the oxygen demand and the effective affinity of the respiratory chain enzymes to oxygen to be constant throughout individual slices. This is certainly an abstraction. Local variations in oxygen consumption are likely present due to the spatial arrangements of different cell parts, such as dendrites, soma, and axons due to different cell types, such as neurons, astrocytes, or microglia, and due to different local activity states. The inhomogeneous distribution of metabolic activity might also result from inhomogeneities in substrate supply (not only of oxygen but also of glucose and lactate). Furthermore, the slicing procedure induces tissue damage and disrupts the neuronal network thereby influencing local metabolic activity. However, although we are not able to dissect the sources of metabolic inhomogeneity, the assumption of homogenous oxygen consumption overall fits the measured oxygen depth profiles.
In addition, due to fundamental experimental differences between brain slice and intravital recordings (e.g. absence of CBF, constant surplus oxygen supply, distortion of neuronal networks), peak CMRO2 values during SD found in vitro may not reflect in vivo oxygen consumption accurately.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231222306 - Supplemental material for Isoflurane lowers the cerebral metabolic rate of oxygen and prevents hypoxia during cortical spreading depolarization in vitro: An integrative experimental and modeling study
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231222306 for Isoflurane lowers the cerebral metabolic rate of oxygen and prevents hypoxia during cortical spreading depolarization in vitro: An integrative experimental and modeling study by Karl Schoknecht, Mathilde Maechler, Iwona Wallach, Jens P Dreier, Agustin Liotta and Nikolaus Berndt in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X231222306 - Supplemental material for Isoflurane lowers the cerebral metabolic rate of oxygen and prevents hypoxia during cortical spreading depolarization in vitro: An integrative experimental and modeling study
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X231222306 for Isoflurane lowers the cerebral metabolic rate of oxygen and prevents hypoxia during cortical spreading depolarization in vitro: An integrative experimental and modeling study by Karl Schoknecht, Mathilde Maechler, Iwona Wallach, Jens P Dreier, Agustin Liotta and Nikolaus Berndt in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutsche Forschungsgemeinschaft (DFG) grant no. 408355133 (A.L., N.B.). J.P.D. was supported by the DFG (DR 323/10-2) and the Bundesministerium für Bildung und Forschung (BMBF) under the frame of ERA-NET NEURON (EBio2, 0101EW2004).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
K.S., J.P.D., A.L., and N.B. conceptualized the study; M.M. and A.L. conducted the experiments; K.S., M.M., I.W., A.L., and N.B. analyzed the data; K.S., J.P.D., A.L., and N.B. wrote the manuscript. M.M. and I.W. revised the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
