Abstract
Whole-brain mapping of drug effects are needed to understand the neural underpinnings of drug-related behaviors. Amphetamine administration is associated with robust increases in striatal dopamine (DA) release. Dopaminergic terminals are, however, present across several associative brain regions, which may contribute to behavioral effects of amphetamine. Yet the assessment of DA release has been restricted to a few brain regions of interest. The present work employed positron emission tomography (PET) with [11C]raclopride to investigate regional and temporal characteristics of amphetamine-induced DA release across twenty sessions in adult female Sprague Dawley rats. Amphetamine was injected intravenously (2 mg/kg) to cause displacement of [11C]raclopride binding from DA D2-like receptors, assessed using temporally sensitive pharmacokinetic PET model (lp-ntPET). We show amphetamine-induced [11C]raclopride displacement in the basal ganglia, and no changes following saline injections. Peak occupancy was highest in nucleus accumbens, followed by caudate-putamen and globus pallidus. Importantly, significant amphetamine-induced displacement was also observed in several extrastriatal regions, and specifically in thalamus, insula, orbitofrontal cortex, and secondary somatosensory area. For these, peak occupancy occurred later and was lower as compared to the striatum. Collectively, these findings demonstrate distinct amphetamine-induced DA responses across the brain, and that [11C]raclopride-PET can be employed to detect such spatiotemporal differences.
Introduction
The addictive and neurodegenerative properties of amphetamine are clearly established. 1 Still, studies monitoring whole-brain amphetamine-induced dopamine (DA) activity, and their temporal trajectories, are still scarce. Such knowledge is important, as it offers a more complete understanding for the cerebral underpinnings of drug-induced behavior. One approach that allows for continuous assessment of whole-brain DA activity is positron emission tomography (PET) and a radioligand that can be displaced upon elevation of endogenous DA levels. Amenability of [11C]raclopride – a moderate-affinity D2-like DA receptor (D2DR) antagonist – for detection of amphetamine induced DA release in the striatum is well established. However, the applicability of [11C]raclopride for quantification of extrastriatal displacement from D2DRs remains unclear. The overarching aim of the present work was to map the spatiotemporal profiles of amphetamine-derived DA release across the brain with PET and [11C]raclopride in Sprague Dawley rats. While doing so, we learn in which regions of the rat brain [11C]raclopride displacement can be reliably observed.
The popularity of [11C]raclopride relates to its beneficial properties such as good test-retest reliability,2,3 and sensitivity to measure competitive binding with endogenous DA.4,5 Being a moderate-affinity ligand, the signal-to-noise ratio for [11C]Raclopride has historically been deemed too low for reliable extrastriatal assessments,6 –8 and therefore, it has mainly been used for striatal D2DR quantifications. Instead, high-affinity D2DR radioligands, with improved cortical signal-to-noise ratio, are preferred for extrastriatal measurements. Notably, high-affinity ligands have limitations, as these are not optimal for assessment of striatal D2DR assessments and endogenous DA release.9,10 It is therefore appropriate to reassess the applicability of a moderate-affinity ligand, such as [11C]raclopride, for whole-brain coverage in the rat. In fact, a growing literature now provide compelling evidence that test-retest statistics of [11C]raclopride are good-to-excellent for striatal as well as for extrastriatal measurements,2,11 and that extrastriatal [11C]raclopride-derived D2DR levels are coherent with D2DR levels estimated with high-affinity D2-ligands.3,11 Furthermore, robust associations between striatal and extrastriatal DRD2 measured using [11C]raclopride have been observed in cross-sectional settings, 11 but also in terms of 5-year change. 12 Furthermore, associations between extrastriatal [11C]raclopride binding potential (BPND) has been linked with parameters including age 13 and cognitive function.14 –16 Together, these studies suggest that extrastriatal [11C]raclopride binding measure functionally relevant and repeatable D2DR binding, rather than noise.
Past studies have unequivocally shown an amphetamine-induced reduction in specific [11C]raclopride binding in the striatum, and attributed this to increased endogenous DA release.
17
Amphetamine elevates extracellular DA levels through intracellular synaptic vesicle depletion and promotion of reverse transport through monoamine transporters such as the DA transporter (DAT),1,18 which is primarily expressed in the striatum. Despite low cortical DAT concentrations,
Taken together, [11C]raclopride-PET may constitute a suitable approach for identification and quantification of DA release across the rat brain. The present study simultaneously investigated striatal and extrastriatal amphetamine-induced DA-release using an [11C]raclopride displacement approach. For this purpose, we employed automatic whole-brain segmentation and a temporally sensitive pharmacokinetic model (linear parametric neurotransmitter PET; lp-ntPET),24,25 to depict a temporally variant responses across the brain.26,27 Our primary aim was to characterize the spatiotemporal patterns of amphetamine-induced DA release across the rat brain, and while doing so, assess the validity of in-scan [11C]raclopride technique in extrastriatal regions. We predicted a robust amphetamine-induced [11C]raclopride signal change in the striatum, possibly with a ventro-dorsal striatal gradient in the magnitude of DA release, 28 and absence of change following saline injections. Furthermore, we hypothesized that [11C]raclopride displacement would be observed in selected extrastriatal regions, in a patterns similar to regions previously associated with drug addiction and where D2DRs are relatively abundant (e.g. prefrontal cortex and medial temporal lobe). 29
Materials and methods
Animals and study design
Animal experiments were performed in accordance with the EU directive 2010/63/EU and Swedish legislation on the protection of animals used for scientific purposes and approved by the animal review board at the court of appeal of Northern Norrland in Umeå (approval number A33-2019). Experiments are reported in compliance with the ARRIVE guidelines (Animal Research: Reporting In Vivo Experiments, www.nc3rs.org.uk/arrive-guidelines). All efforts were made to minimize animal suffering, and animals were housed under 12:12 h light-dark conditions and had access to food and water
PET imaging
[11C]raclopride was synthesized on-site at Umeå University Hospital, and imaging data was acquired on a small animal PET/CT scanner (Mediso nanoScan PET/CT, Hungary). The specific activity was determined for each batch, which was used to calculate the mass (in µg) of non-labelled raclopride for each injection. The rat tail vein was cannulated, and the cannula was flushed with heparinized saline before injecting the radiotracer. The scan procedure started with a 50 kV helical CT scan covering the head and upper body, followed by a 60-minute list-mode PET acquisition commencing at injection of 12.8 ± 5.3 MBq (non-labelled raclopride mass 0.015 ± 0.016 µg, range = 0.001 – 0.06 µg) [11C]raclopride. List mode data was binned to 12 × 10 s, 6 × 20 s, 6 × 40 s, 6 × 80 s, 6 × 160s, 7 × 240 s frames. This was reconstructed to dynamic PET-images of voxel-size 0.4 mm using the built-in TeraTomo 3D iterative reconstruction; employing 4 iterations, 4 subsets, and correction for attenuation, scatter, and random events. At 25 min after the start of the PET scan, rats were injected intravenously with 2 mg/kg amphetamine (d-Amphetamine sulfate) or an equivalent volume of saline (0.9% sodium chloride).
Magnetic resonance imaging
When PET scans were completed, MRI imaging was performed in each animal for the purpose of anatomical co-registration of PET images. MRI was carried out in a 9.4 T Bruker BioSpec 94/20 scanner equipped with a BG12S-HP gradient set, utilizing a 40 mm quadrupolar volume coil from Bruker and running Paravision 6 software (Bruker, Ettlingen, Germany). A T2-RARE 3D sequence was acquired (TR/TE = 1800/40 ms) with a matrix = 160/128/64, repetitions = 2, field-of-view = 32/25.6/12.8 cm.
Image analyses
Dynamic PET-images were co-registered with the anatomical MRI using a sum image of the first eight minutes of the PET scan to determine transformation parameters. Automated registration was achieved using mutual information algorithm implemented in statistical parametric mapping software (SPM12, Wellcome Institute, London, UK). Next, non-linear image registration implemented in SPM12 was used to find a mapping between individual anatomical MR-images and a Sprague Dawley rat brain template. 30 Subsequently, dynamic PET-images were spatially aligned and resliced to match the atlas coordinates using the same mappings. Voxel size of the atlas was resampled to 0.1 mm × 0.1 mm × 0.1 mm in favor of reasonable data matrix size relative to the inherent spatial resolution of PET imaging data. Time-activity course (TAC) data were extracted from all anatomical regions (n = 222) defined in Waxholm space atlas of the Sprague Dawley rat brain version 4 (https://www.nitrc.org/projects/whs-sd-atlas).
PET pharmacokinetic analysis
Administration of amphetamine was expected to lead to displacement of [11C]raclopride from its D2DR binding sites according to the competition principle.
17
The primary variable of interest from the PET pharmacokinetic analysis was D2DR occupancy time-course data following in-scan amphetamine injections. To that end, lp-ntPET was used to estimate dynamic binding potential (BPND) relative to non-displaceable binding in the cerebellum from ROI-wise TACs.25
–27,31 In brief, grey matter voxels of the cerebellum was used as a reference region, as it is largely devoid of specific D2DR binding. A library of 180 gamma functions was generated
32
with varying onset and peak times as well as growth and decay rates. This library was used as basis functions, and fitted as an additional variable in the multilinear reference tissue model.
33
The onset time for ligand displacement was allowed to vary between 25 and 29 minutes in the model fitting. Models accounting for no displacement (base model) and 180 different shapes of displacement were assessed using Akaike information criteria (AIC), and the best fitting function was interpreted to reflect the most likely trajectory of [11C]raclopride displacement following the amphetamine/saline injections. The basis function approach permits analysis of inter-rat and inter-regional differences in [11C]raclopride displacement, which is appropriate given the unknown dynamics of DA release. Model fits were conducted for each animal and ROI independently, to generate a data set of regional dynamic BPND curves for subsequent statistical analysis. In the next step, the dynamic BPND estimate curves were transformed to dynamic D2DR occupancy profiles:
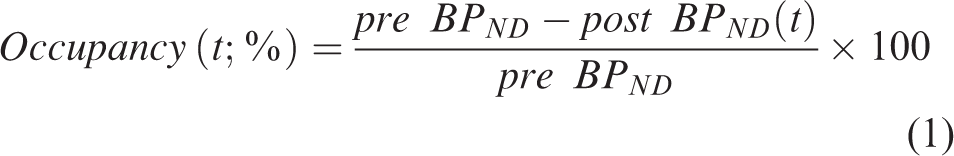
Statistical analysis
The main aims of the statistical analyses were: 1) to evaluate interregional differences in [11C]raclopride BPND across the brain (Figure 1), 2) to map regions showing statistically significant differences in [11C]raclopride occupancy profiles following amphetamine, as compared to saline, and 3) to characterize amphetamine-induced temporal trajectories of [11C]raclopride displacement. Significance level was set to
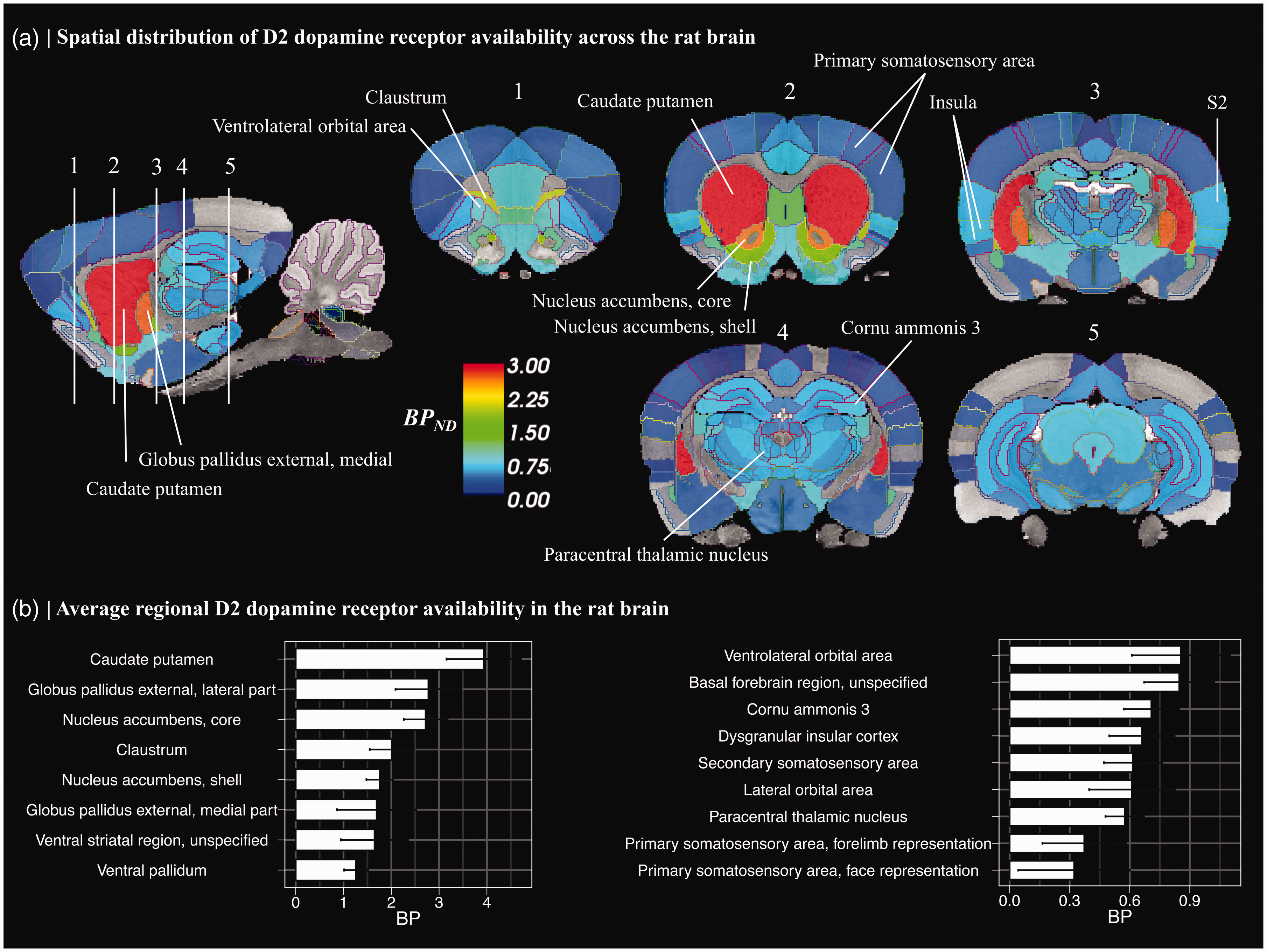
(a) Spatial distribution of [11C]raclopride binding potential (BPND). Average of control animals’ (n = 6 scans) ROI-wise BPND values were overlayed on standard Sprague Dawley rat T2* MR-image. Lines depict region-of-interest borders of the anatomical atlas. BPND was calculated in regions of gray matter, with cerebellum serving as a reference region. Regions where BPND > 0.1 are shown. S2 = secondary somatosensory area and (b) Regional average (±95%CI) for [11C]raclopride BPND values in selected brain regions.
Interregional differences in BPND levels (aim 1) were investigated in the control group, exclusively (n = 6). A likely difference in the mean BPNDs of two regions was inferred when non-overlapping mean ± 95% confidence intervals (CI) between the two regions was observed. For aim 2, hierarchical general additive models (HGAM) were set up to assess for a group (amphetamine/saline) difference in non-linear occupancy profiles by having group membership as a random effect.
34
Package mgcv (version 1.8) implemented in R (R Foundation for Statistical Computing, Vienna, Austria) was used for HGAM analysis.
35
Specifically, for each region, temporal occupancy profiles in equation (1) were entered into GAM to allow modeling of group-average non-linear occupancy profiles. Base model (model 1) made no distinction between animals receiving amphetamine or saline. Improvement in model fit by inclusion of the random effect of group (amphetamine/saline, model 2) was interpreted as an indication of group difference in occupancy. Models were compared in terms of AIC and using chi-square test for unbiased calculation of 
Results
Interregional differences in [11C]raclopride binding potential in the rat brain
Spatial distribution and rank order of select regional [11C]raclopride BPND estimates are presented in Figure 1. As expected, highest bindings were observed in the basal ganglia, including caudate-putamen, ventral striatum, and globus pallidus (Figure 1(a) and (b)). Binding in the thalamus, hippocampal complex, and cortical regions was approximately five-fold lower as compared to the striatum (Figure 1(b)). Interregional differences in mean BPND were observed in extrastriatal regions, with highest cortical binding in the ventral orbital cortices and basal forebrain, followed by insular cortex, secondary somatosensory area, hippocampal complex and thalamus, and markedly lower binding in the primary somatosensory areas (Figure 1(b)). Binding in the primary visual cortex, a region analogous to the primate occipital cortex did not exceed 0.1 (Figure 1(a)) and did not statistically differ from zero (t-test, p = 0.062), thus, our results are in accordance with past studies showing negligible [11C]raclopride binding in the occipital cortex. 36
Interregional differences of amphetamine-induced [11C]raclopride displacement
Next, pharmacokinetic modeling of [11C]raclopride signal using lp-ntPET was conducted to estimate D2DR occupancy profiles following amphetamine and saline injections. As expected, amphetamine injections were associated with positive occupancy profiles, and no consistent changes in occupancy following saline injections (Figure 2). Amphetamine was associated with distinct [11C]raclopride responses compared to saline in several striatal as well as extrastriatal regions (Table 1); amounting to 49 ROIs, or 20% of all examined ROIs. Largest AIC differences – reflecting saline/amphetamine group difference in HGAM – were found in the basal ganglia and parts of thalamus, followed by insula, orbitofrontal cortex and secondary somatosensory area (Table 1, Figure 3). The whole-brain map of AIC differences (Figure 3) highlights pronounced interregional variation in the effects of amphetamine relative to saline.
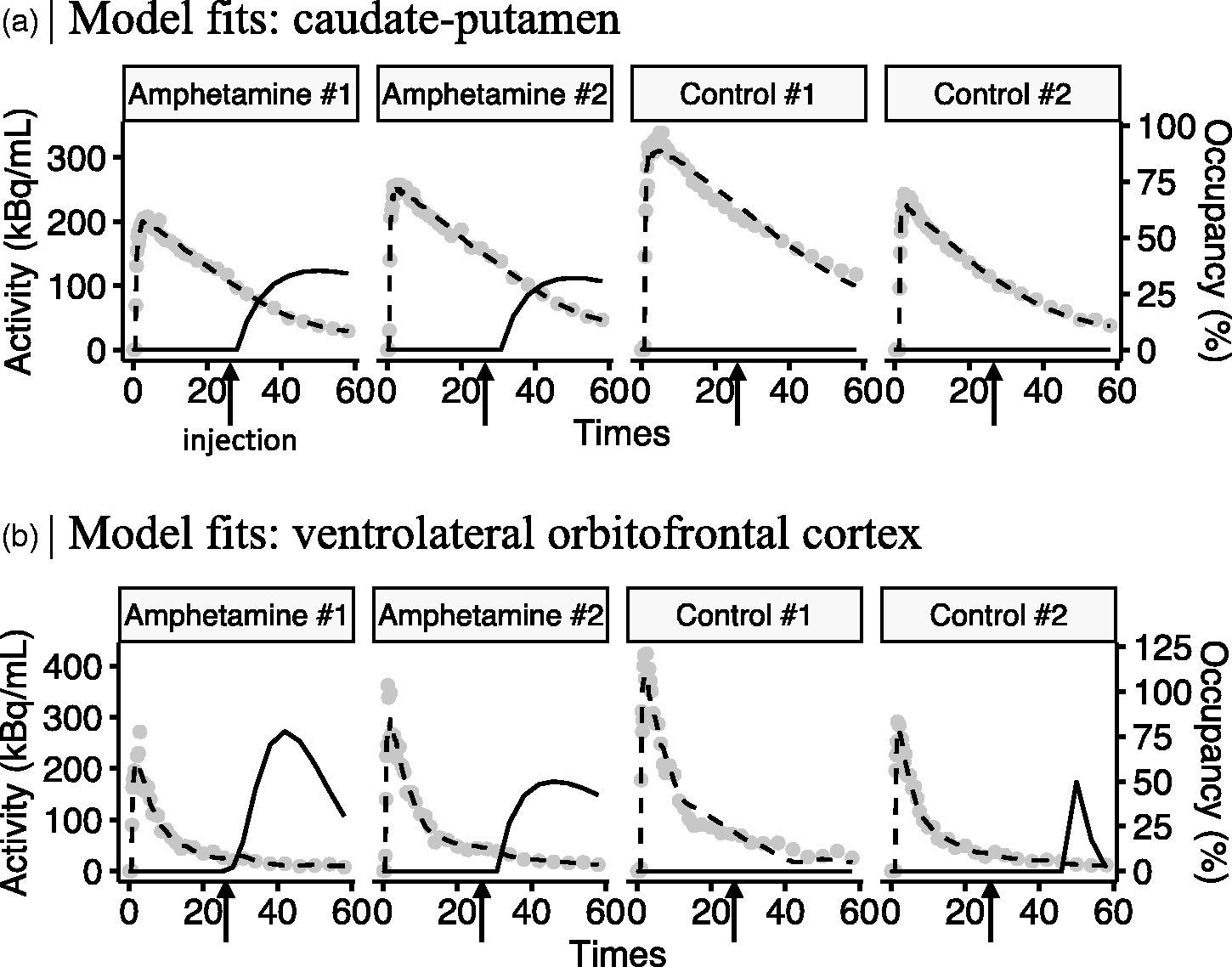
Representative pharmacokinetic model fits and corresponding dopamine occupancy profiles for caudate-putamen (a) and ventrolateral orbitofrontal cortex and (b) in 2 animals receiving amphetamine, and 2 animals injected with saline (control). Gray dots represent average radioactivity in the caudate-putamen, and dashed lines the lp-ntPET model fits. Occupancy (%) curves were calculated relative to a temporally stationary [11C]raclopride BPND estimate during first 25 minutes of the scan (solid black lines). Amphetamine or saline injections were given at 25 minutes after [11C]raclopride. Hence, occupancy (%) reflects how much specifically bound [11C]raclopride displacement was induced by each injection.
Summary of model-comparison statistics for identification of regions where the temporal profile of specific [11C]raclopride binding was altered differently after amphetamine as compared to saline injections.
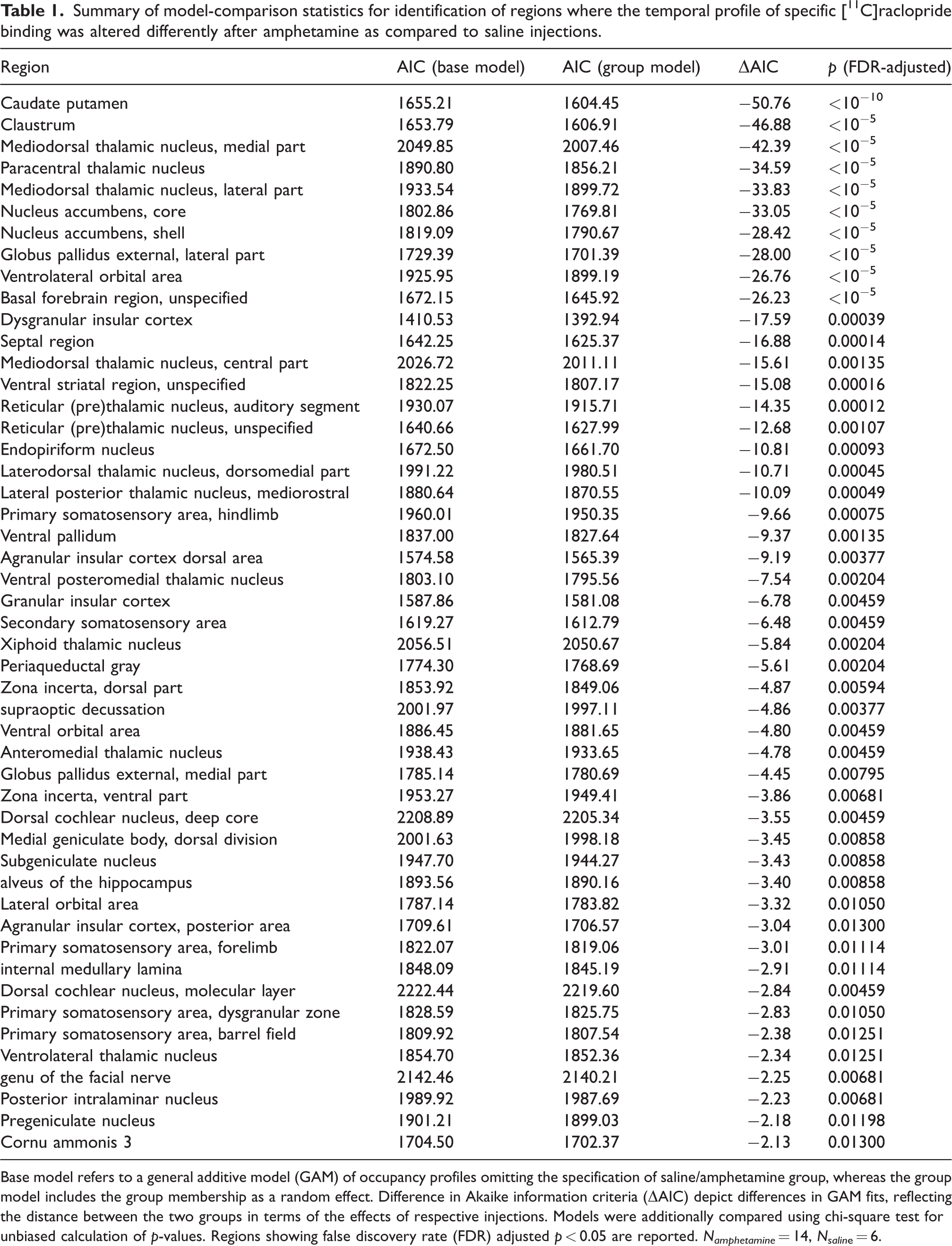
Base model refers to a general additive model (GAM) of occupancy profiles omitting the specification of saline/amphetamine group, whereas the group model includes the group membership as a random effect. Difference in Akaike information criteria (ΔAIC) depict differences in GAM fits, reflecting the distance between the two groups in terms of the effects of respective injections. Models were additionally compared using chi-square test for unbiased calculation of
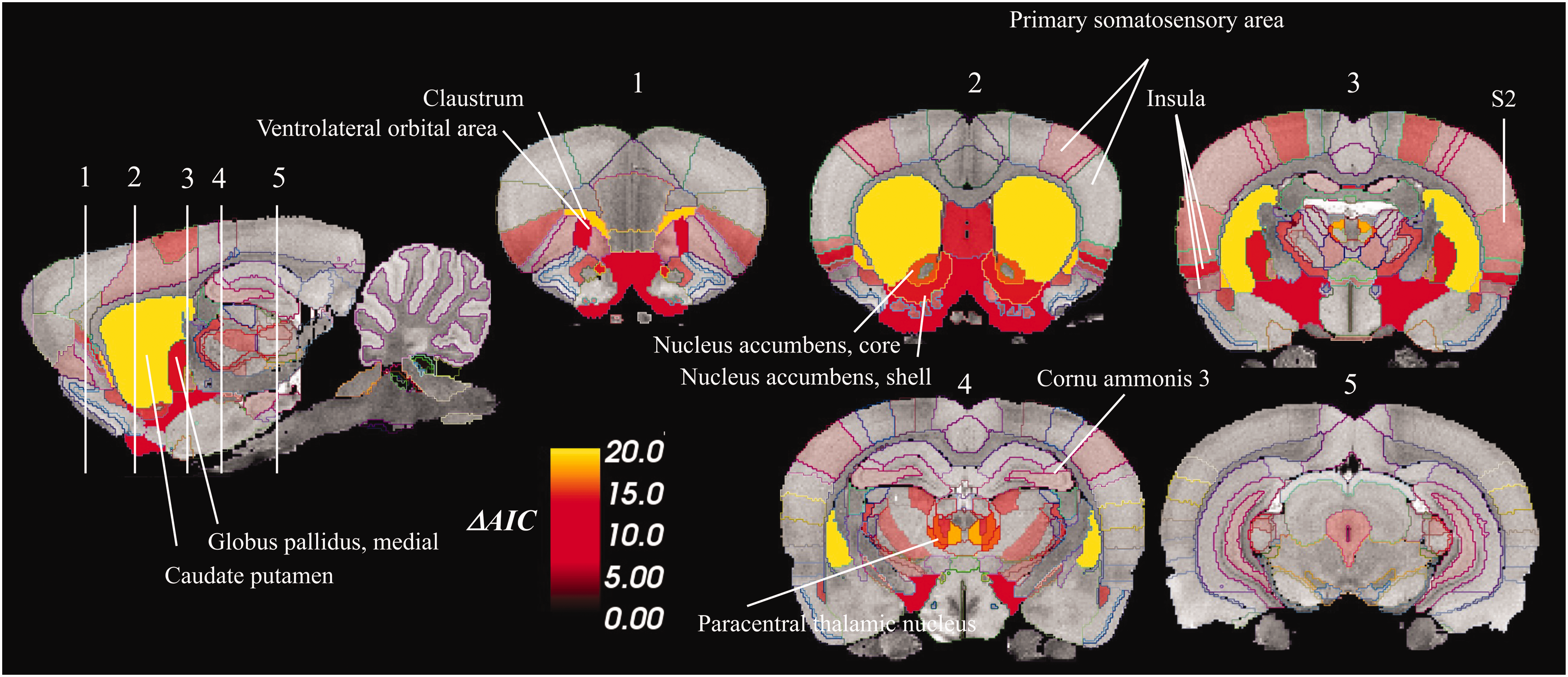
Spatial distribution of model comparison statistics (ΔAIC) for identification of regions where saline- versus amphetamine-induced [11C]raclopride displacement were significantly different. Difference in Akaike information criteria (ΔAIC) depict differences in GAM fits, reflecting the distance between the two groups in terms of the effects of respective injections. Models were additionally compared using chi-square test for unbiased calculation of
Temporal profiles of amphetamine-induced [11C]raclopride displacement
Regions showing the most clear change in occupancy following amphetamine administration (parts of basal ganglia and thalamus, followed by insula, orbitofrontal cortex and secondary somatosensory area (Table 1; Figure 3), were further analyzed in terms of temporal DA occupancy profiles.
First, we focused on amphetamine-induced occupancy time-courses in subregions of the basal ganglia (caudate-putamen, nucleus accumbens, and globus pallidus), i.e. the main targets of dopaminergic innervation. The shapes of occupancy time-courses were not markedly different across these regions (Figure 4(a)), suggesting a fairly similar temporal trajectory of amphetamine across these nuclei. However, the peak occupancies varied from ∼45% in the nucleus accumbens, to ∼40% in the caudate-putamen, and ∼35% in the globus pallidus. Further data-driven analysis using the GAM-derivatives revealed significant fluctuations in the occupancy rate (Figure 4(b)). Peak rate of occupancy increase was detected at 9 minutes after amphetamine injection (Figure 4(b); first derivative), while peak acceleration was seen at approximately 5 minutes after the injection (Figure 4(b); second derivative). Peak receptor occupancy occurred at approximately 22 minutes after the injection (Figure 4(b); prediction), and significant decline in occupancy was observed at approximately 26 minutes (Figure 4(b); first derivative shows significant negative trend).
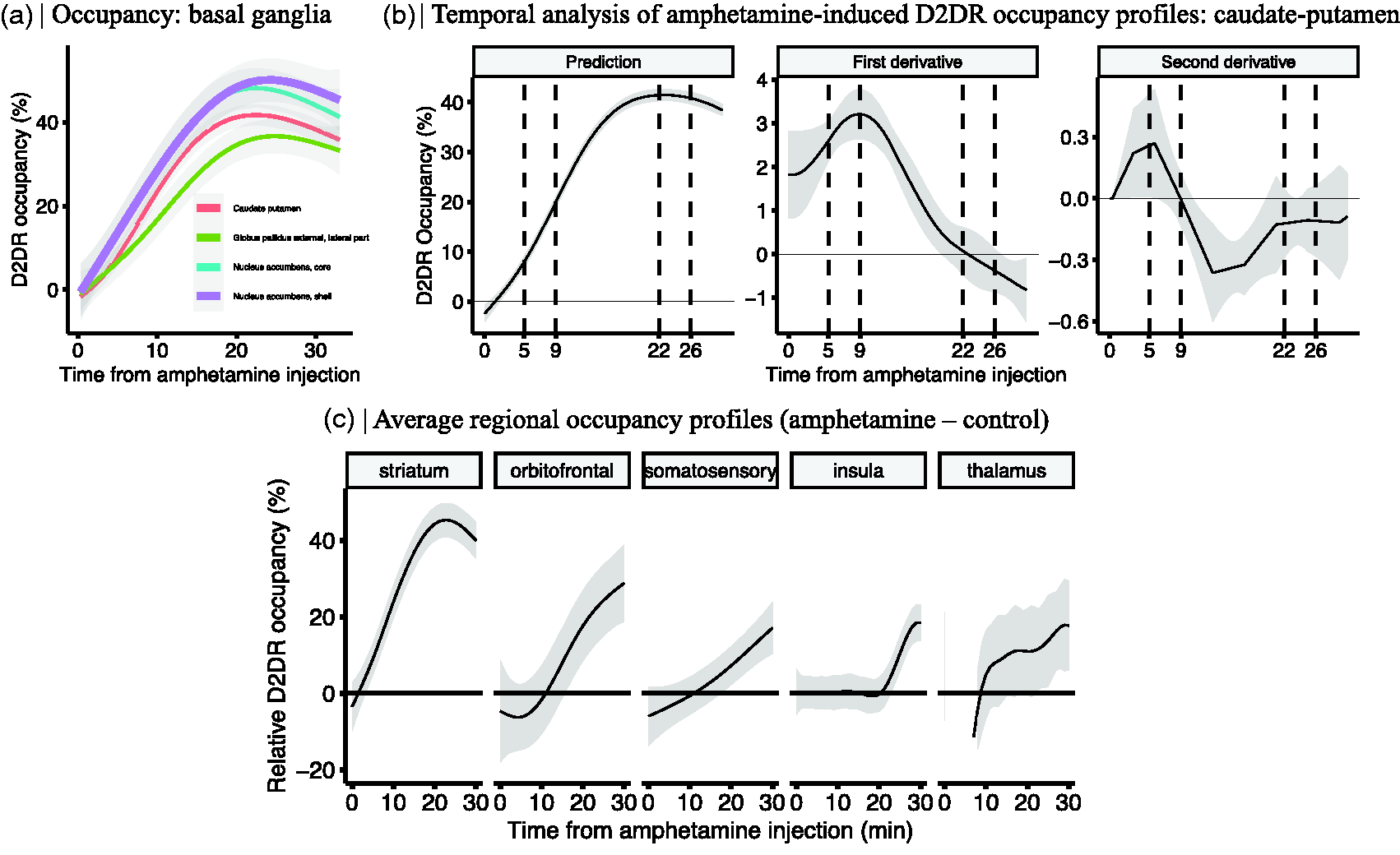
(a) Regional differences in the occupancy profiles of the basal ganglia. (b) Derivatives of the predicted occupancy time profiles of the caudate-putamen and (c) Striatal and extrastriatal amphetamine-induced D2R occupancy (%) time profiles (n = 14) relative to a control group (n = 6). Shaded area represents 95% CI.
Next, we considered extrastriatal regions showing amphetamine-induced change in occupancy and compared these with the striatal occupancy profiles. In order to mitigate the effects of statistical noise and to summarize the results across the brain, the estimates of occupancy (Table 1) were pooled across ROIs to form anatomical clusters. Specifically, five anatomical regions were considered, constituting the striatum (caudate-putamen, nucleus accumbens), orbitofrontal cortex, somatosensory cortex, insula and thalamus, each consisting of three to twelve subregions. Shape differences in the regional occupancy profiles were estimated by extracting the predicted occupancy profile of the control group (6 scans) from that of the amphetamine group (14 scans; Figure 4(c)). The relative occupancy time profiles revealed marked interregional differences in the timing as well as magnitude of amphetamine-induced [11C]raclopride displacement. Notably, the peak magnitude of occupancy was higher and increase in occupancy was more rapid in the striatum, in comparison to extrastriatal regions (Figure 4(c)). While the narrowest 95% CI was found for the striatum, select extrastriatal regions also exhibited reliable amphetamine-induced time-resolved occupancies relative to the control condition.
Discussion
The present work shows interregional differences in amphetamine-induced DA release, as assessed using PET and [11C]raclopride. In line with previous findings in non-human primates, 28 there was a ventro-dorsal gradient of DA release in the striatum, with significantly higher D2DR occupancy in the nucleus accumbens as compared to caudate-putamen. Furthermore, timing of peak striatal D2DR occupancy was remarkably similar to previously reported amphetamine-induced DA concentration peak using microdialysis. 1 Apart from the striatum, DA release was also found in several extrastriatal regions, including thalamus, insula, orbitofrontal cortex and secondary somatosensory area. Previously, D2DR mRNA quantifications indeed show that D2DR are expressed across the cortex in rats, and particularly in prefrontal regions.37 –39 In agreement, we show higher [11C]raclopride binding in cortical regions, as compared to the reference region (where D2DR expression is close to zero). In concordance with the present work, prior human studies have found DA release following amphetamine in the orbitofrontal cortex and insula.40,41 In terms of temporal profiles for occupancy changes, striatum and globus pallidus were characterized by earlier onset and higher peak magnitude of occupancy, as compared to extrastriatal regions. This is expected given prior findings of large differences in striatal and cortical DA release following a comparable dose of d-amphetamine, peaking at about 1500% versus 300-600% above baseline, respectively.19,20 Together these findings suggest reliable detection of amphetamine-induced DA release using [11C]raclopride in striatal as well as in several extrastriatal regions. Nevertheless, given the low extrastriatal occupancies relative to methodological noise (Figure 4(c)), it is advisable to proceed with caution in experimental settings in which a group difference is desired outside the striatum. The present proof-of-concept study exploited a maximum contrast between groups (amphetamine/saline), whereas more modest differences would likely require larger groups and/or regularization of methodological noise to detect significant effects.
Drug addiction is associated with altered function of the orbitofrontal cortex. 42 Volumetric changes for orbitofrontal cortex, insula, striatum, and medial temporal lobe follow chronic use of cocaine and amphetamine. 43 Furthermore, activation of insula and striatum during reward-based tasks are associated with liking of amphetamine. 44 Here, we show that DA activity is altered in these regions following amphetamine intake, with the exception for medial temporal lobe. The absence of occupancy change in medial temporal lobe was partly surprising given significant amphetamine-induced DA level elevations in past microdialysis work. 19 It may be the case that DA release is lower in medial temporal lobe, as compared to prefrontal regions, and thus displacement is not detectable. Pum and colleagues indeed reported an elevation of approximately 300% above baseline at 40 minutes in parts of the medial temporal lobe, 19 while twice as high (∼600%) DA release was observed in the medial prefrontal cortex, not far from the ventrolateral orbital cluster observed here. The combination of low signal-to-noise ratio of [11C]raclopride with the well-established ceiling effects to [11C]raclopride displacement by endogenous DA17,45 may have prohibited the detection of putative DA release in some extrastriatal regions. Specifically, based on investigations including rodents, it has been postulated that approximately 50% of D2DRs that bind [11C]raclopride are in low-affinity state towards endogenous DA, and therefore, may not contribute to the pool of receptors under active competition.17,45 Here, this ceiling effect likely contributed to the striatal D2DR occupancy data plateauing at approximately 50%, despite up to 1500% increase in DA concentration following a similar dose of amphetamine as employed here has been reported. 1 Furthermore, it has been pointed out that while specific [11C]raclopride binding is detectable outside of the striatum, cerebellum may not be optimal as reference region for low-density D2DR regions in occupancy studies. 46 This since the D2DR levels are low, but not completely absent, in the cerebellum. 8
[11C]raclopride has been used as a radioligand for over 30 years. When introduced, it was deemed suitable for striatal quantifications only, due to its low signal-to-noise ratio. 8 A growing amount of evidence now suggests that, although extrastriatal D2DR levels are low, they are detectable with [11C]raclopride. 46 To exemplify, test-retest reliability is good-to-excellent not only for striatal regions, but also for example for thalamus, and frontal and temporal cortices.2,3 Furthermore, extrastriatal D2DR levels determined with [11C]raclopride map well with high-affinity DRD2 ligand [18F]fallypride.3,11 Here, evidence of amphetamine-induced extrastriatal [11C]raclopride displacement was found for some extrastriatal regions, which is not surprising given variability in extrastriatal DAT concentration across brain regions. 47 In regions with low DAT concentration, such as the frontal cortex, noradrenaline transporters may have a significant role in DA clearance. 48 The striatal and cortical D2DR occupancy trajectories differed not merely by a clear level difference, but also in terms of the rate of accumulation. This finding is particularly well-aligned with past non-human primate research, showing a striking difference in magnitude and timing between striatal and cortical amphetamine-induced DA release as assessed using microdialysis. 20 The present finding thus support previous observations of greater magnitude and more rapid displacement of [11C]raclopride in the striatum, which is due to higher concentration of DA nerve terminals and, therefore, more capacity for amphetamine-induced reverse-transport of DA21. That said, future studies should investigate occupancy profiles beyond thirty minutes from amphetamine administration, in order to capture the peak in extrastriatal occupancy. In contrast to prior studies, whole-brain coverage and temporal characterization of amphetamine-induced DA release was achieved by regional, time-sensitive analysis of [11C]raclopride signal.
Effects related to limited spatial resolution of PET scanning may have affected the outcome of the present analysis. Specifically, the remarkably high [11C]raclopride BPND in the striatum relative to the background renders the signal in the neighboring regions susceptible to the so-called spillover effect. 49 Therefore, it is conceivable that spillover effect may have contributed to the high BPND estimates in the secondary somatosensory area and insula, but it is harder to reconcile why other close-by regions such as primary motor area did not show high BPND; as the spillover effect should be fairly similar across regions close to striatum. 49 Moreover, the majority of past PET studies have used a difference between static measures of pre- and post-amphetamine BPND. Here, instead, amphetamine was administered during PET scanning and the dynamics of [11C]raclopride displacement were assessed with the help of temporally sensitive PET pharmacokinetic modeling (lp-ntPET).24 –27 The dynamic approach is advantageous to static measures, given that static measures depend on the time of measurement (relative to amphetamine administration), and that amphetamine-induced DA release exhibit markedly different temporal dynamics across the brain.19,20 Therefore, the current study presents an approach that might be crucial for simultaneous detection of amphetamine-induced [11C]raclopride displacement in the striatum and extrastriatal regions. Notably, the present study presents cortical [11C]raclopride binding potential values that are approximately 15% of those in the striatum, as compared to 6%–8% reported in past high-resolution human studies.2,7 This potential species difference in the strength of cortical [11C]raclopride signal should be considered when translating the present findings to human imaging.
In conclusion, the present findings suggest feasibility of single-scan [11C]raclopride competition study for whole-brain mapping of amphetamine-induced DA release in the rat brain. Translation of a similar technique to human imaging studies appears possible, given a growing body of evidence that not only striatal but also extrastriatal displacement of [11C]raclopride follows from pharmacological as well as non-pharmacological stimulation of DA release.7,21 –23
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the Umeå University Medical Faculty (Insamlingsstiftelsen; to NK and AV); Hjärnfonden (NK) and Vetenskapsrådet (NK).
Acknowledgements
We acknowledge the Small Animal Research Imaging Facility (SARIF) at Umeå University for providing assistance in using the 9.4 T MRI and PET/CT. We also acknowledge the Strategic Research Program in Diabetes at Umeå University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conception of the study and formulation of research questions (JJ, SAB, AV, NK)
