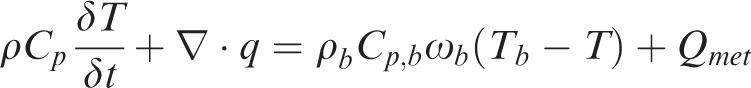Abstract
In ischemic stroke, selectively cooling the ischemic penumbra might lead to neuroprotection while avoiding systemic complications. Because penumbral tissue has reduced cerebral blood flow and in vivo brain temperature measurement remains challenging, the effect of different methods of therapeutic hypothermia on penumbral temperature are unknown. We used the COMSOL Multiphysics® software to model a range of cases of therapeutic hypothermia in ischemic stroke. Four ischemic stroke models were developed with ischemic core and/or penumbra volumes between 33–300 mL. Four experiments were performed on each model, including no cooling, and intraarterial, intravenous, and active conductive head cooling. The steady-state temperature of the non-ischemic brain, ischemic penumbra, and ischemic core without cooling was 37.3 °C, 37.5–37.8 °C, and 38.9–39.4 °C respectively. Intraarterial, intravenous and active conductive head cooling reduced non-ischemic brain temperature by 4.3 °C, 2.1 °C, and 0.7–0.8 °C respectively. Intraarterial, intravenous and head cooling reduced the temperature of the ischemic penumbra by 3.9–4.3 °C, 1.9–2.1 °C, and 1.2–3.4 °C respectively. Active conductive head cooling was the only method to selectively reduce penumbral temperature. Clinical studies that measure brain temperature in ischemic stroke patients undergoing therapeutic hypothermia are required to validate these hypothesis-generating findings.
Introduction
Clinical trials are yet to demonstrate any benefit of therapeutic hypothermia in patients with acute ischemic stroke. 1 , 2 There has been re-emerging interest in strategies that aim to protect the ischemic penumbra in patients undergoing endovascular thrombectomy (EVT) in the prehospital setting, and during interhospital transfer, and the procedure itself.3 –6 Methods for inducing hypothermia in stroke patients include body surface cooling, intravenous cooling, intraarterial cooling and active conductive head cooling, where the scalp is cooled externally with a cooling cap. 6 These methods of inducing hypothermia are likely to have differential effects on the ischemic core, ischemic penumbra, and non-ischemic brain given that ischemic brain has reduced cerebral blood flow compared to the surrounding non-ischemic brain. Most clinical trials have focused on inducing systemic hypothermia through body surface or intravenous cooling, both of which are practically challenging and can lead to significant complications such as pneumonia. 1 , 2 In patients undergoing EVT, intraarterial cooling has been shown to be safe in single-center non-randomized studies, but this technique is limited in that it cannot be delivered in the prehospital setting, during interhospital transfer, or following EVT. 4 In theory, selectively cooling the ischemic penumbra (selective penumbral cooling) may lead to neuroprotection while avoiding the systemic complications that accompany body or intravenous cooling. 4 Active conductive head cooling has shown promise at reducing brain temperature without causing systemic hypothermia and could be applied in the prehospital setting. 7 However, active conductive head cooling may only lead to modest brain cooling and there remains skepticism that it would be of therapeutic benefit. 8 Measurement of in vivo brain temperature, particularly in ischemic stroke, remains a significant challenge. 9 We therefore aimed to develop thermal models of a range of ischemic stroke patients in order to test the effects of intraarterial, intravenous and active conductive head cooling on ischemic and non-ischemic brain temperature in clinically meaningful timeframes.
Methods
We used the COMSOL® software,
10
to perform three-dimensional thermal modeling. COMSOL is a multiphysics tool that can analyze complex three-dimensional thermal models. Three-dimensional solid models for various stroke geometries were created using SolidWorks®. The solid models were imported into COMSOL to perform the thermal analysis using COMSOL’s Bioheat Transfer Interface. The interface was configured to solve for the bioheat equation using Penne’s Approximation,
11
which accounts for heat from blood perfusion and cellular metabolism using the classical heat equation:
ρ is the density of the tissue (kg/m3)
Cp is the specific heat capacity at constant pressure of the tissue (J/(kg·K))
T is the absolute temperature of the tissue (K)
q is the heat flux by conduction in the tissue (W/m2)
ρb is the blood density (kg/m3)
Cp, b is the blood specific heat capacity at constant pressure (J/(kg·K))
ωb is the blood perfusion rate calculate based on volumetric blood flow per unit brain volume (1/s)
Tb is the arterial blood temperature (K)
Qmet is the metabolic heat source (W/m3)
Four different ischemic stroke models were developed based on the geometries of one half of the human head. Each model was split into distinct regions, including the skull, non-ischemic brain, ischemic penumbra, and ischemic core. Cerebral blood flow was assumed to be 50 mL/100 g/min in the non-ischemic brain, 20 mL/100 g/min in the ischemic penumbra, and 0 mL/100 g/min in the ischemic core.
12
,
13
The remaining model properties are included in the Supplemental Materials.
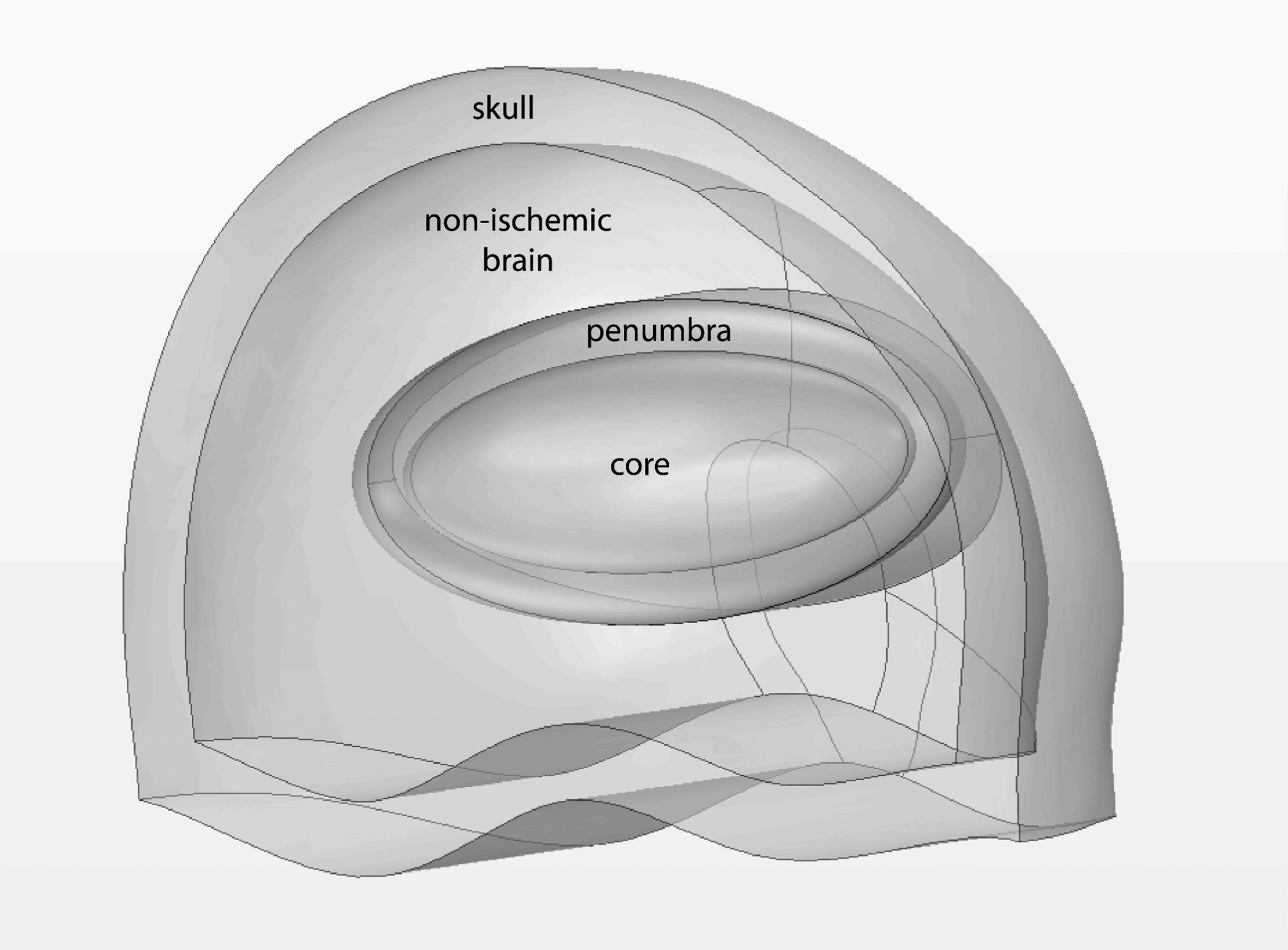
Model 1 represented an occlusion of the proximal middle cerebral artery with ischemia affecting the entire vascular territory, estimated to be on average 300 mL.
14
Arbitrarily, ⅓ of the territory (100 mL) was assumed to be ischemic core, and ⅔ assumed to be ischemic penumbra (200 mL), in order to represent a so-called “large core” (≥70 mL) ischemic stroke, as defined by the Extending the Time for Thrombolysis in Emergency Neurological Deficits — Intra-Arterial (EXTEND-IA) and Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke (DEFUSE 3) trials (Figure 1).
15
,
16
3 D representation of the proximal middle cerebral artery occlusion (100 mL core, 200 mL penumbra) solid model. Models 2 and 3 represented more “standard” occlusions of the middle cerebral artery, with 100 mL of ischemic brain tissue, similar to that reported in EXTEND-IA and DEFUSE 3.
15
,
16
Model 2 had a large core (70 mL) and small penumbra (30 mL), whereas model 3 had no core (0 mL) and a large penumbra (100 mL). Model 4 represented a distal middle cerebral artery occlusion, with a volume of ischemic brain tissue arbitrarily ⅓ of the volume of model 2 and 3 (0 mL core, 33 mL penumbra), but also similar to the ischemic volumes (34 mL) reported in patients undergoing EVT for distal medium vessel occlusions.
17
To simplify the simulations, some physical assumptions were made. The boundaries between anatomical regions were assumed to be infinitesimally small, and the thermal conductivity across the boundary was assumed to be infinite. Rather than modeling each individual layer separately, scalp, bone and cerebrospinal fluid layers were collapsed into a homogenous “skull” layer. The dura was neglected since its thickness, at 270 micrometers, is negligible compared to the other layers. 18 To determine an aggregate thickness and mean thermal conductivity, the values shown in the Supplemental Materials were used.
A total “skull” thickness of 13 mm was modeled, being the sum of the scalp, bone and cerebrospinal fluid thicknesses. The thermal conductivity for the skull was set to the weighted average of the thermal conductivities of its constituent layers, being 0.40 W/m/K. The density of the skull layer was set to 1908 kg/m3 and the COMSOL Material Library specific heat capacity of bone was used. 19 The cerebrospinal fluid was assumed to start at normal body temperature and remain stationary during the modeling. The initial temperature of all regions was set to normal arterial blood temperature (37 °C). The exterior of the scalp was treated as a boundary condition with the temperature fixed to a configurable value; that is, the head cooling thermal dynamics and/or interaction with the atmosphere, in the case of no cooling, was not simulated.
Four experiments were performed on the four models. The arterial blood temperature or the scalp boundary temperature were varied to simulate different methods of therapeutic hypothermia. Experiment 1 tested the non-cooled control. Carotid temperature was set to 37 °C in order to represent a normal body temperature. 20 Scalp temperature was set to 33 °C based on the mean forehead temperature of 1000 apparently healthy subjects. 21 Experiment 2 tested intraarterial cooling, based on Wu et al.’s prior work, with carotid temperature set to 32.7 °C, which is based on 30 mL/min (∼12%) of the carotid blood flow being at 4 °C. 4 Experiment 3 tested intravenous cooling based on the Intravascular Cooling in the Treatment of Stroke 2 (ICTuS-2) and Cooling Plus Best Medical Treatment Versus Best Medical Treatment Alone for Acute Ischaemic Stroke (EuroHYP-1) trials, 1 , 2 with carotid temperature set to 35 °C. Experiment 4 tested active conductive head cooling based on Tolerability and efficacy of non-invasive head COOLing in normal and HypopErfused brain: A magnetic resonance stuDy (COOLHEAD-1) with scalp temperature set to 5 °C. 7 The time-varying temperature within the 3 D model was solved using COMSOL and the mean temperatures of each of the regions was computed at each time-step. A 2 D section view of the temperature distribution across the brain at the end of the simulation was also generated.
The modeling techniques were partially validated using experimental data from intraarterial cooling experiments in rhesus monkeys reported by Wang et al. 22 The goal of the validation was to build confidence in the general modeling methodology using the reference case above. Wang et al.’s experiments were chosen because they provided the closest analog to the cases modeled for this work and, of the potential reference cases reviewed, required the fewest model assumptions. Wang et al. cooled the right hemisphere of rhesus monkey brains using intraarterial cooling with 100 mL iced Ringer’s lactate solution (0 to 4 °C) infused into the M1 segment of the middle cerebral artery over 20 minutes (5 mL/min). This is similar, but not identifical to Experiment 2 above, where coolant was injected into the internal carotid artery rather than the middle cerebral artery. Probes inserted into the striatum and parietal cortex were used to monitor the time-varying brain temperature during the experiment. The Wang et al. intraarterial cooling experiment was modeled using the same modeling approach as Experiments 1–4. All modeling techniques and parameters were kept constant except for necessary modifications to account for the primates’ anatomy and physiology and the differences in the specific experimental process; these modifications are described below. Solid Model 1 above was used as a base for this model; however, the overall volume was scaled down to reflect a rhesus monkey brain volume of approximately 100 mL. 23 All brain territories used a normal rhesus monkey perfusion rate of 50 mL/100 g/minute. 24 , 25 The difference in coolant fluid injection location (middle cerebral artery in Wang et al. versus internal carotid artery in the current study) necessitated a different approach for the modeling of the perfused blood temperature. For the human model, the perfusion temperature was set to a constant value since coolant is injected into the internal carotid artery, which is the major supply of the cerebral hemisphere. Furthermore, since we are mostly interested in the steady-state temperature distribution for cooling method comparisons, it was not necessary to estimate the transient perfusion temperature – just the steady-state temperature which should be primarily influenced by the internal carotid artery temperature at steady-state.
However, for the rhesus monkey, coolant was injected into the middle cerebral artery which supplies only a part of the hemisphere, thus a time-varying perfusion temperature, following an exponential decay curve, was used to account for mixing from other vascular territories during the experiment. Core body temperature was set based on experimental measurements in Wang et al., starting at 37.5 °C and reducing to 37.1 °C, over the duration of cooling. As for the human model, a composite scalp-bone-cerebrospinal fluid thickness was modeled with a fixed boundary temperature on the scalp. This approach does not directly account for the extra scalp muscle mass present in rhesus monkeys compared with humans. To compensate for the additional insulative properties, the scalp boundary temperature was fixed to 35 °C, 2 °C higher than a human. The exact value of this boundary condition should not significantly affect the results since the primary cooling method is via cooled blood perfusion.
Results
Table 1 shows the steady-state temperatures of the non-ischemic brain, the ischemic penumbra and the ischemic core, for each of the models and cooling methods. Without cooling, the steady-state temperature of the non-ischemic brain, ischemic penumbra, and ischemic core was 37.3 °C, 37.5–37.8 °C, and 38.9–39.4 °C respectively (Figure 2). Intraarterial, intravenous and active conductive head cooling reduced non-ischemic brain temperature by 4.3 °C, 2.1 °C, and 0.7–0.8 °C, respectively, to 33.0 °C, 35.2 °C, and 36.5–36.6 °C, respectively. Intraarterial, intravenous and active conductive head cooling reduced ischemic penumbra temperature by 3.9–4.3 °C, 1.9–2.1 °C, and 1.2–3.4 °C respectively, to 33.3–33.6 °C, 35.4–35.8 °C, and 34.2–36.4 °C, respectively (Figure 3). After cessation of intraarterial cooling, the penumbral temperature rapidly approached the non-cooled temperature (Figure 3). The non-ischemic brain temperature dropped below penumbral temperature with all cooling methods except for active conductive head cooling (Figures 4 and 5). In the non-cooled controls, penumbral temperature was 0.2–0.5 °C higher than non-ischemic brain, whereas penumbral temperature was 0.2–2.4 °C lower than non-ischemic brain with active conductive head cooling.
Steady-state temperature of non-cooled control, and intraarterial, intravenous and active conductive head cooling.
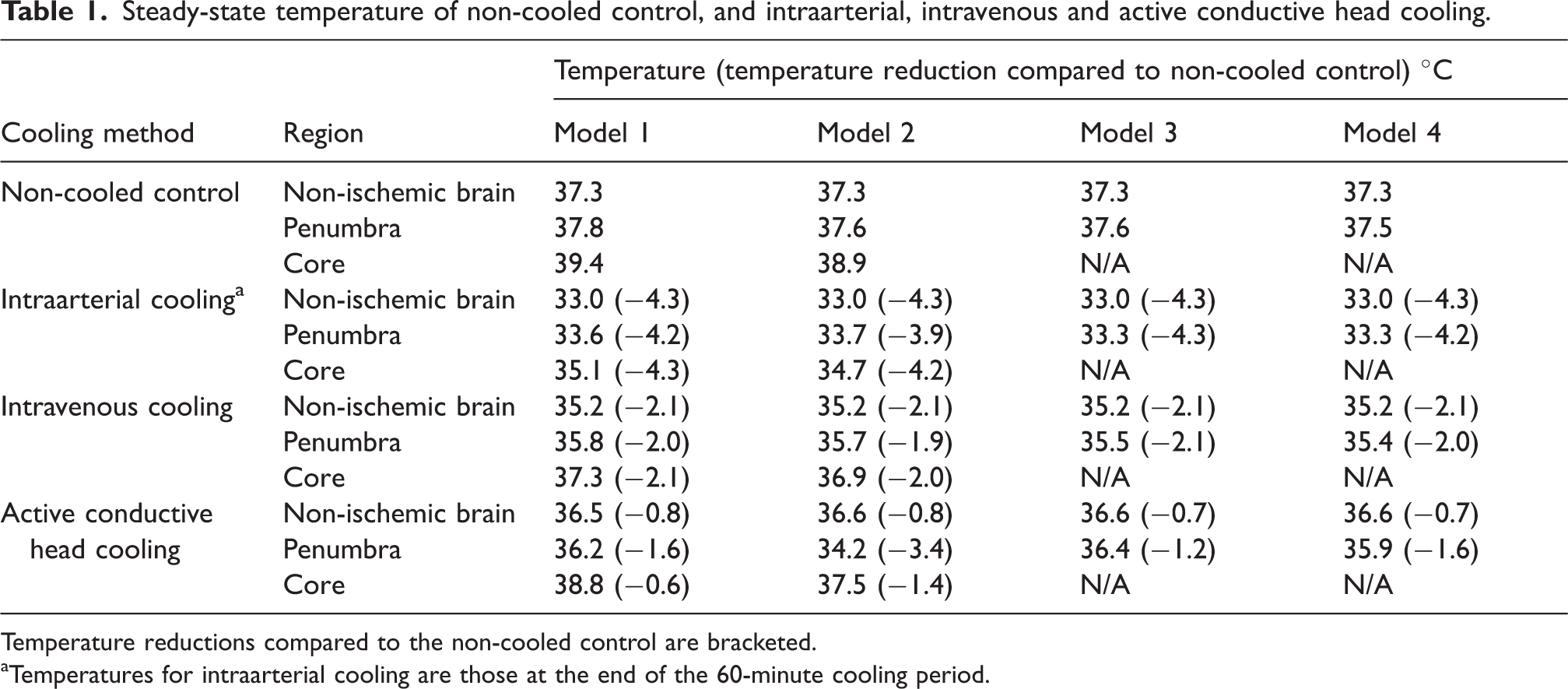
Temperature reductions compared to the non-cooled control are bracketed.
Temperatures for intraarterial cooling are those at the end of the 60-minute cooling period.
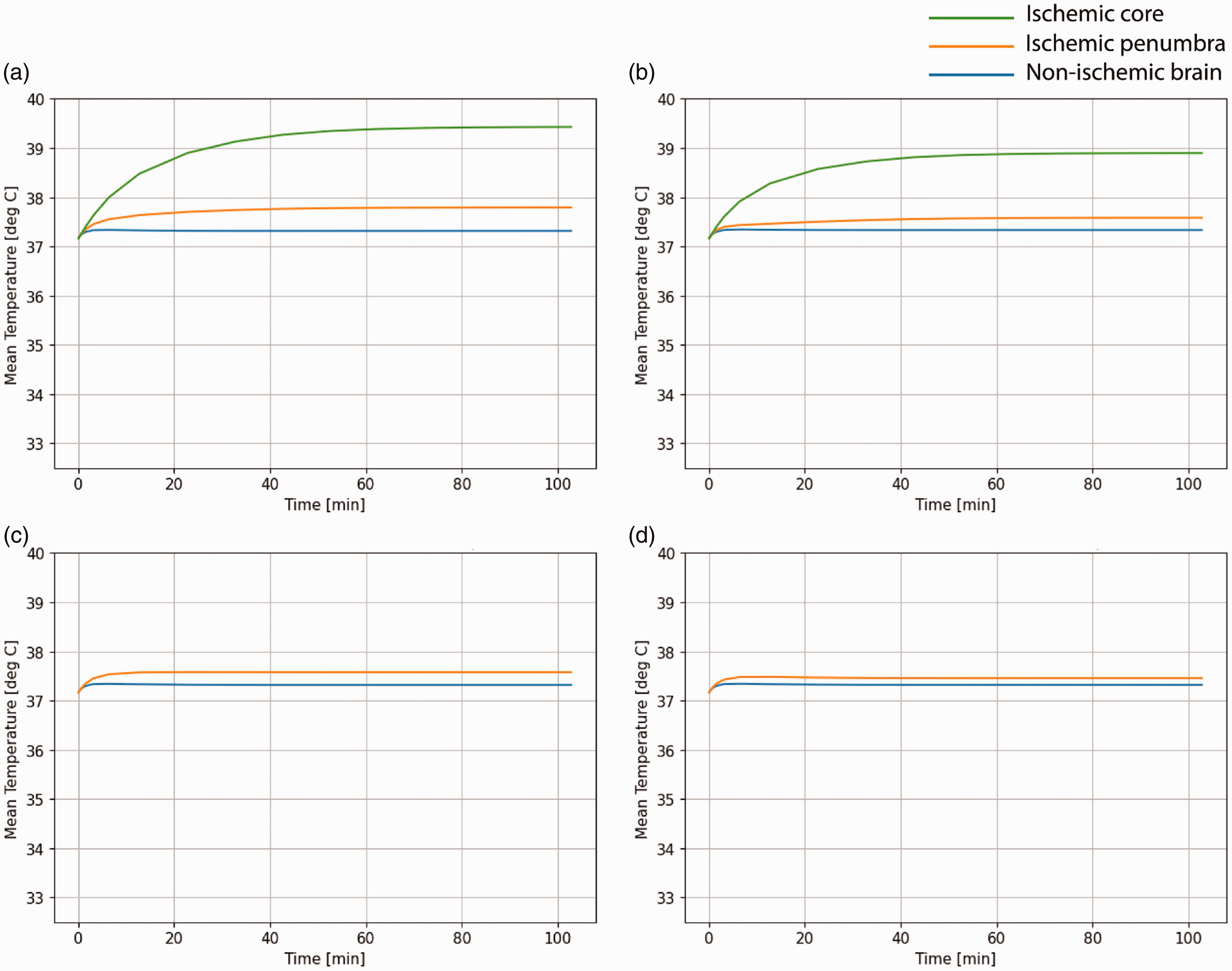
Non-cooled brain temperature in ischemic stroke over time. a = Model 1 (100 mL core, 200 mL penumbra), b = Model 2 (70 mL core, 30 mL penumbra), c = Model 3 (0 mL core, 70 mL penumbra), d = Model 4 (0 mL core, 33 mL penumbra).
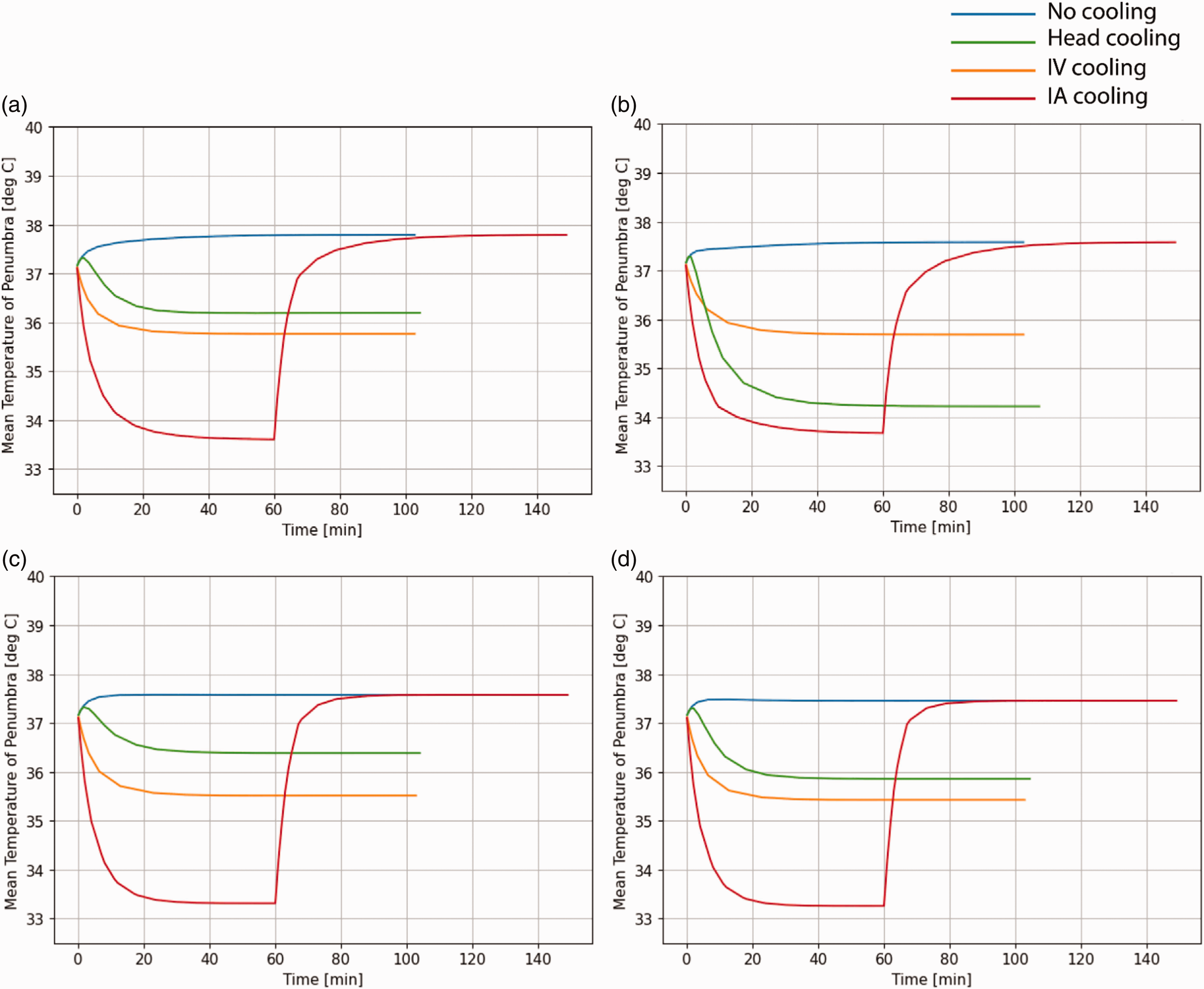
Comparison of methods of therapeutic hypothermia in ischemic stroke. a = Model 1 (100 mL core, 200 mL penumbra), b = Model 2 (70 mL core, 30 mL penumbra), c = Model 3 (0 mL core, 70 mL penumbra), d = Model 4 (0 mL core, 33 mL penumbra).
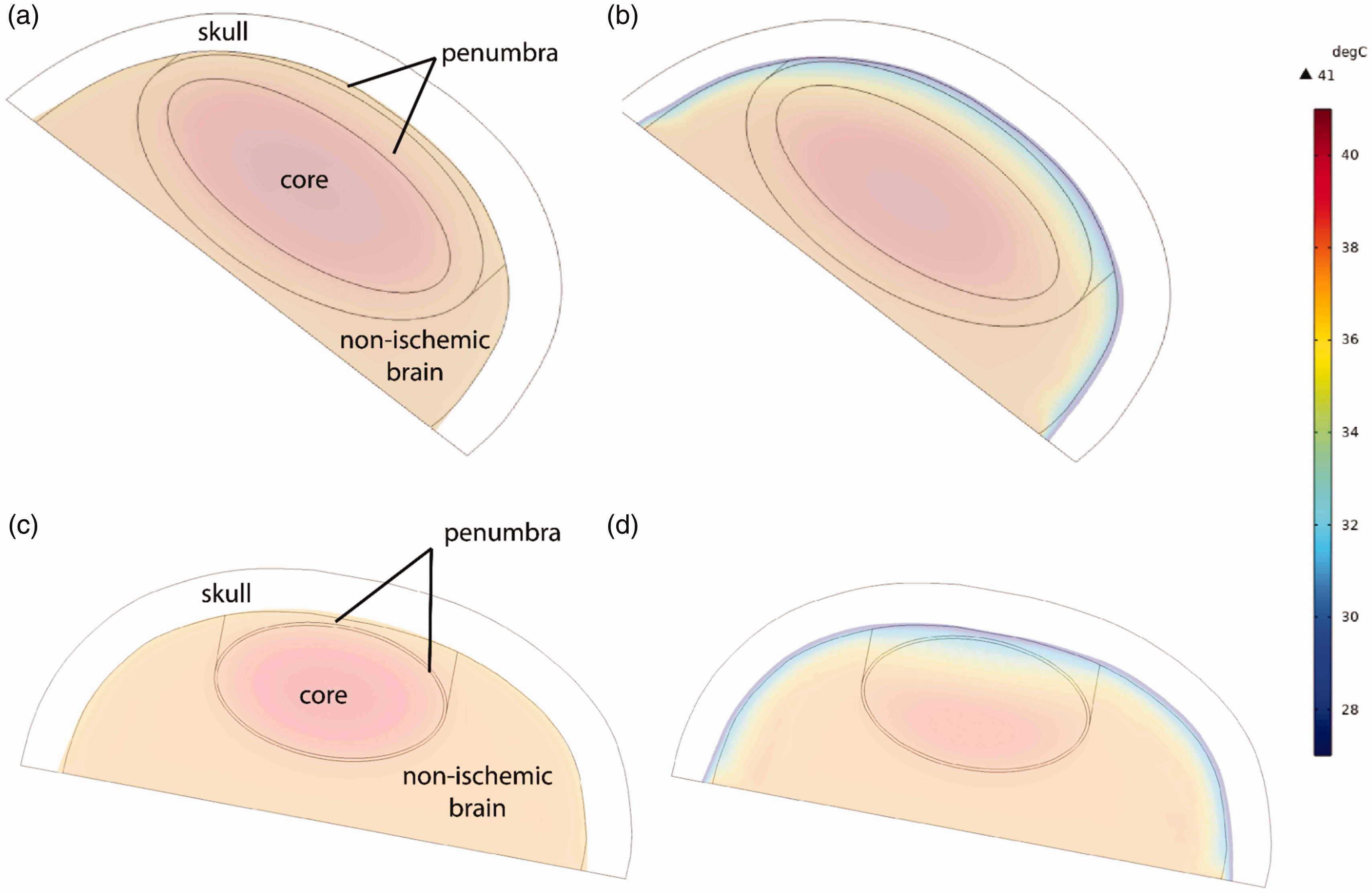
Section views of 3 D thermal models. Model 1 (100 mL core, 200 mL penumbra) without cooling (a) and with active conductive head cooling (b). Model 2 (70 mL core, 30 mL penumbra) without cooling (c) and with active conductive head cooling (d).
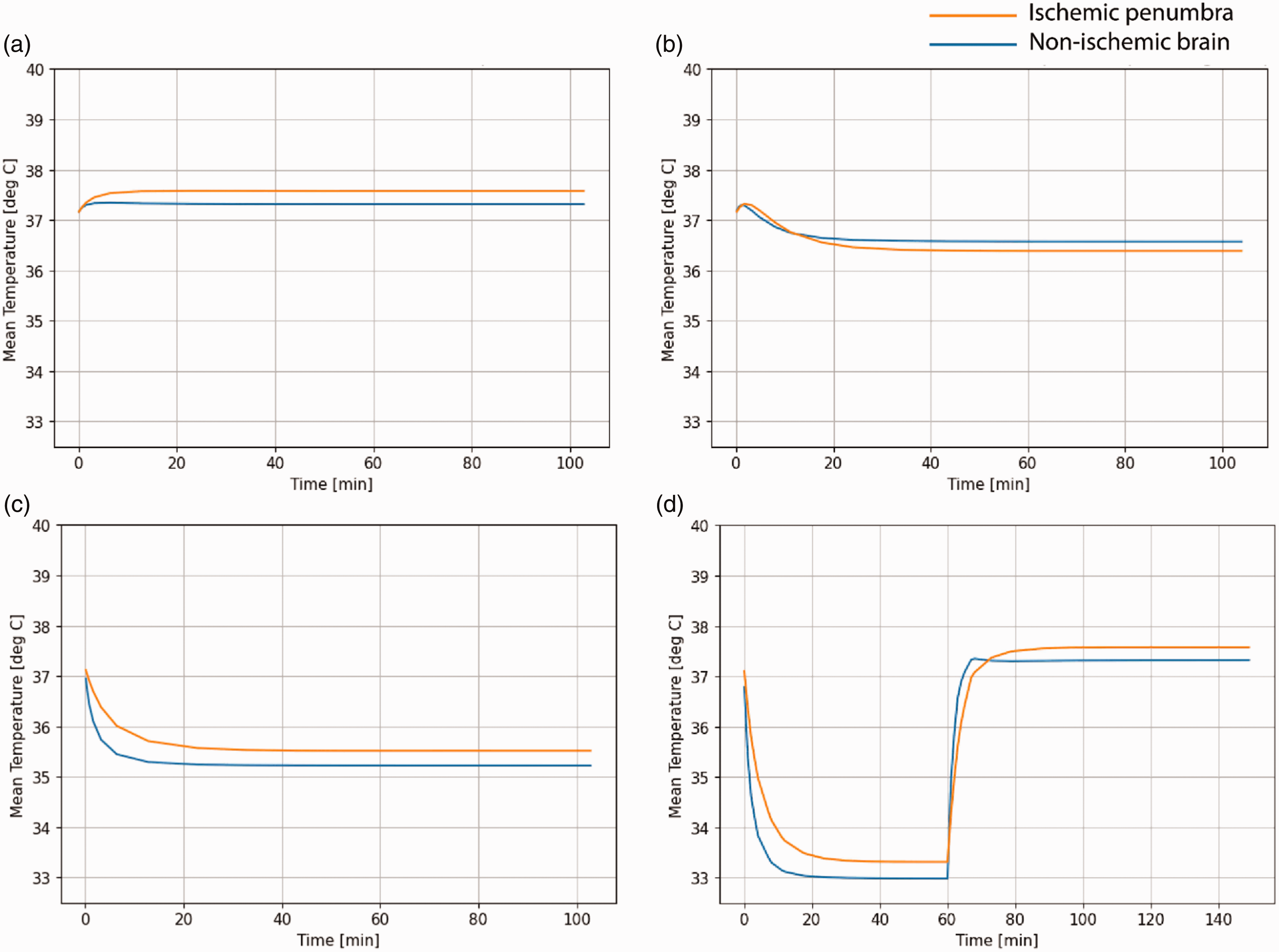
Effect of different therapeutic hypothermia methods on the temperature of ischemic penumbra (100 mL) and non-ischemic brain. a = No cooling, b = Active conductive head cooling, c = Intravenous Cooling, d = Intraarterial cooling for 60 minutes.
The modeling validation results based on Wang et al. are shown in Figure 6. Our numerical model closely reproduced the time-varying behavior of rhesus monkey brain temperature during intraarterial cooling as reported by Wang et al. The margins of error shown in Figure 6 represent the declared measurement error of the temperature probes reported by Wang et al. The differences between the modeled and experimental brain temperature are within the margins of measurement error.
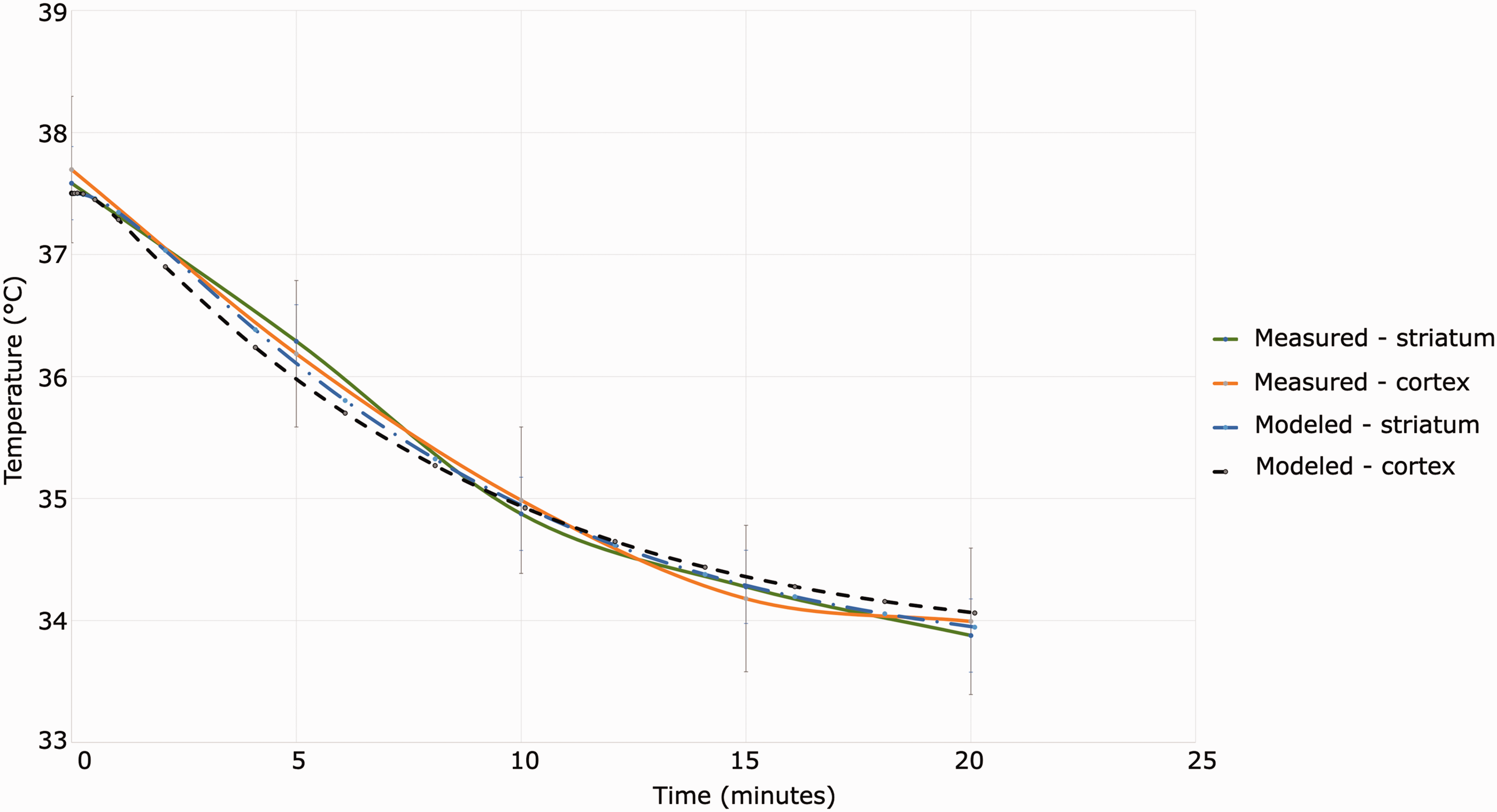
Validation of modeling techniques. As-modeled and as-measured time-varying cortical and striatal temperature in rhesus monkey during intraarterial cooling.
Discussion
This hypothesis-generating study of four thermal models designed to simulate a range of patients who present with ischemic stroke has demonstrated that there are potential advantages and disadvantages with different methods of therapeutic hypothermia. Previous clinical studies have found that active conductive head cooling reduced whole brain temperature by 0.8–0.9 °C when measured using invasive or magnetic resonance spectroscopic temperature measurements, 7 , 26 and this is comparable to the current study’s estimate of 0.7–0.8 °C. Furthermore, the temperature dynamics observed in the non-cooled control experiments is consistent with those observed in acute ischemic stroke patients who underwent magnetic resonance spectroscopy. 27 That is, ischemic brain increases in temperature, with higher temperatures in larger lesions (i.e., model 1 > model 2 = model 3 > model 4), and lesions with less blood flow (i.e., model 2 > model 3). This suggests that, although the current study needs further validation in patients, the temperature changes seen in the current study’s experiments are valid hypothesis-generating findings.
Although there are multiple potential mechanisms for increased temperature in ischemic brain, the mechanisms in the current model are limited to (1) metabolic heat, and (2) impaired heat exchange from reduced cerebral blood flow. With cooling, the temperature of the non-ischemic brain, ischemic penumbra and ischemic core variably change over time. This reaffirms the need to utilize methods such as magnetic resonance spectroscopy to measure regional brain temperature in studies involving therapeutic hypothermia in ischemic stroke. 9 The ischemic stroke population being studied will have implications for the potential efficacy of a particular method of therapeutic hypothermia. For example, proximal and distal vessel occlusions may respond differently to cooling, due to variations in lesion size and location; interhospital transfer patients may receive greater benefit from early therapeutic hypothermia when compared to direct presenters.
Intraarterial cooling was the most effective method of cooling the ischemic penumbra, reaching 33.3–33.7 °C, but resulted in rapid rewarming after cessation of cooling, and overshoot into the hyperthermic range in the ischemic penumbra. These findings have implications for trials of intraarterial cooling as rapid rewarming may exacerbate ischemic and post-ischemic inflammatory brain injury.28 –32 Intraarterial cooling has the disadvantage of only being administered during the EVT procedure. So although intraarterial cooling may be effective at inducing penumbral hypothermia, adjunctive methods to induce penumbral cooling in the minutes or hours leading up to EVT, and to slow rewarming after cessation of intraarterial cooling are required. 6
Intravenous cooling resulted in penumbral temperatures of 35.4–35.8 °C. Previous studies have shown that intravenous cooling may be effective at inducing and maintaining modest hypothermia but is practically difficult to administer. Intravenous cooling is also an indirect method of cooling the penumbra as it requires the induction of systemic hypothermia to remove heat from the non-ischemic and ischemic brain tissue, and this can lead to significant side effects. However, systemic hypothermia may offer additional benefits over selective brain/penumbral cooling because post-stroke brain inflammation has been linked to systemic inflammation and multiorgan injury. 33 , 34 ICTuS-2 administered 2 liters of 4 °C normal saline followed by 24 hours of cooling via a femoral intravenous cooling catheter and 12 hours of rewarming. 1 EuroHYP-1 used a 20 mL/kg intravenous infusion of 4 °C normal saline over 30–60 minutes followed by surface or intravenous cooling for 24 hours and passive rewarming. 2 ICTuS-2 and EuroHYP-1 were both stopped early due to slow recruitment and cessation of funding. Pneumonia was more frequent in the hypothermia groups in both studies, and all-cause mortality was more frequent in the hypothermia group in ICTuS-2.
Trials of inducing systemic hypothermia following cardiac arrest to prevent hypoxic-ischemic brain damage have reported mixed results. Early trials found that hypothermia at 33 °C increased survival and improved neurological recovery when compared to normothermia, 35 , 36 and this was supported by a single recent trial. 37 However, two other recent trials comparing hypothermia at 33 °C with either 36 °C or normothermia (with early treatment of fever) did not demonstrate improved neurological outcomes. 38 , 39 In contrast, therapeutic hypothermia is routinely used after neonatal hypoxic-ischemic insults. A meta-analysis of 28 randomized controlled trials comprising 1832 birth asphyxiated infants treated with cooling caps or blankets, and 1760 controls, found a pooled relative risk of mortality of 0.74 in treated infants (95% CI, 0.67–0.80). 40 Given differences in pathophysiology, there are limitations in extrapolating these findings to focal (and temporary) brain ischemia.
Active conductive head cooling resulted in penumbral temperatures of 34.2–36.4 °C, and was the only method that achieved selective penumbral cooling. That is, active conductive head cooling resulted in penumbral temperature dropping below the non-ischemic brain. This occurred despite the tendency for penumbral temperature to increase above non-ischemic brain temperature without cooling. This was most pronounced in model 2, where there was a large ischemic core (70 mL), and the ischemic penumbra (30 mL) was disproportionately closer to the skull. Active conductive head cooling relies on heat transfer from the brain, through the skull, and into the circulating coolant. Because ischemic penumbra has less (warm) cerebral blood flow to replenish the heat lost to the surroundings (i.e., the circulating coolant), ischemic penumbra closer to the skull (e.g., cortex) may be most amenable to cooling with active conductive head cooling. A single published case report found that a cooling cap established an interhemispheric gradient of −8°C in a patient with reduced cerebral blood flow from internal carotid artery occlusion. 41 We, and others have found that active conductive head cooling (1) results in a modest brain-body temperature gradient in non-ischemic brain, (2) does not lead to significant systemic hypothermia, and (3) is well-tolerated by awake participants for up to 120 minutes. 7 , 26 , 42
The current study has limitations. The scalp, bone and cerebrospinal fluid layers were collapsed into a single “skull” layer that assumed the density and specific heat capacity of bone. It is unclear whether this reflects the density and specific heat capacity of the combination of scalp, bone and cerebrospinal fluid. However, according to Fourier’s law, a difference is unlikely to significantly affect the results of the modeling, as the temperature gradient across the skull layer is most likely to be driven by its thermal conductivity. 43 Density and specific heat capacity affect transient temperature dynamics rather than steady-state temperature distribution. The impact of cerebrospinal fluid flow was not taken into account in the modeling as it was assumed that cerebrospinal fluid did not contribute to metabolic heat given that it is replaced every 7.5 hours, 44 and the cerebrospinal fluid volume (150 mL) comprises a small percentage of the overall experiment volume. 45 For these, and other reasons, the solid models may be overly simplistic compared to the human head to draw meaningful conclusions about the absolute but not the relative temperature changes over time with and without cooling.
In the intraarterial cooling experiments we overestimated the duration that cooling could safely take place, assuming a 30 mL/min infusion of 4 °C normal saline over 60 minutes. Clinical trials have used a 10 mL/min infusion of 4 °C normal saline over 5 minutes before recanalisation, followed by a 30 mL/min infusion of 4 °C normal saline over 10 minutes after recanalisation. 4 Therefore, the current study may have overestimated the efficacy of intraarterial cooling. In the active conductive head cooling experiments, we assumed that the boundary condition of the scalp was 5 °C. In reality, scalp temperature is likely to vary significantly depending on the thermal insulation (e.g., hair, air) between the cooling cap and scalp, and the temperature of the cooling cap. 46 Further experimental evidence will be required to determine the effect of active conductive head cooling on scalp temperature. However, the assumed 5 °C scalp boundary condition resulted in comparable reductions in non-ischemic brain temperature to COOLHEAD-1 (−0.9 °C after 80 minutes of cooling). 7 Finally, other variables encountered in real-world clinical situations are not taken into account in the model, for example, alterations in cerebral blood flow due to general anesthesia, which may be used during EVT. 47
The model validation based on middle cerebral artery cooling in rhesus monkeys showed good agreement with in-vivo measurements. However, since the data provided by Wang et al., did not fully constrain the model, some assumptions, as described in the methodology, were necessary. Nevertheless, such a result provides confidence that the modeling techniques can provide valuable comparisons between Experiments 1–4 since good agreement between the model and the experimental data was achieved using reasonable assumptions where measured values were not available. Furthermore, in the comparative analysis of Experiments 1–4, all parameters are both known and consistent between the experiments, thus ensuring comparability of the results, which is the most important aspect of this study. It is also important to note that most of the uncertainties in this validation case relate to the transient, rather than steady-state behavior. Since only the steady-state values are compared in our human models, uncertainties in transient behavior do not directly impact the validity of our results.
In summary, this study demonstrated that different methods of therapeutic hypothermia have potential advantages and disadvantages when applied to ischemic stroke. Intraarterial cooling appears to be the most effective method of reducing penumbral temperature, but cannot be initiated early and may require adjunctive cooling methods in the prehospital and postprocedural settings to improve efficacy and avoid harm. Intravenous cooling may be effective at inducing and maintaining modest penumbral cooling, but previous studies have shown that it is practically difficult to administer and results in significant side effects. Active conductive head cooling appears to result in modest (selective) penumbral cooling and is unlikely to cause systemic hypothermia. Furthermore, it is non-invasive, can be applied in the prehospital setting, during interhospital transfer, and during and after EVT. Clinical studies that measure brain temperature in ischemic stroke patients undergoing therapeutic hypothermia are required to validate these hypothesis-generating findings.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231203025 - Supplemental material for Penumbral cooling in ischemic stroke with intraarterial, intravenous or active conductive head cooling: A thermal modeling study
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231203025 for Penumbral cooling in ischemic stroke with intraarterial, intravenous or active conductive head cooling: A thermal modeling study by William K Diprose, Avinash Rao, Kaustubha Ghate, Zoe Dyer, Doug Campbell, Mohammed Almekhlafi and P Alan Barber in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
W.K.D. conceived the study. W.K.D. and A.R. designed the study. K.G. performed background research. A.R. performed thermal modeling. W.K.D. drafted the manuscript. Z.D., D.C., M.A., and P.A.B. contributed to the intellectual content of the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.