Abstract
Active conductive head cooling is a simple and non-invasive intervention that may slow infarct growth in ischemic stroke. We investigated the effect of active conductive head cooling on brain temperature using whole brain echo-planar spectroscopic imaging. A cooling cap (WElkins Temperature Regulation System, 2nd Gen) was used to administer cooling for 80 minutes to healthy volunteers and chronic stroke patients. Whole brain echo-planar spectroscopic imaging scans were obtained before and after cooling. Brain temperature was estimated using the Metabolite Imaging and Data Analysis System software package, which allows voxel-level temperature calculations using the chemical shift difference between metabolite (N-acetylaspartate, creatine, choline) and water resonances. Eleven participants (six healthy volunteers, five post-stroke) underwent 80 ± 5 minutes of cooling. The average temperature of the coolant was 1.3 ± 0.5°C below zero. Significant reductions in brain temperature (ΔT = –0.9 ± 0.7°C, P = 0.002), and to a lesser extent, rectal temperature (ΔT = –0.3 ± 0.1°C, P = 0.03) were observed. Exploratory analysis showed that the occipital lobes had the greatest reduction in temperature (ΔT = –1.5 ± 1.2°C, P = 0.002). Regions of infarction had similar temperature reductions to the contralateral normal brain. Future research could investigate the feasibility of head cooling as a potential neuroprotective strategy in patients being considered for acute stroke therapies.
Introduction
Therapeutic hypothermia is an effective neuroprotective strategy in animal studies of ischemic stroke.1,2 However, clinical trials have not yet demonstrated any benefit of therapeutic hypothermia in patients with ischemic stroke.3,4 Inducing hypothermia with surface or endovascular cooling is practically challenging and leads to complications associated with systemic hypothermia.3 –8 Animal and human observational studies have suggested that hypothermia in the early ischemic phase, prior to successful reperfusion may be most beneficial.1,9 Following the establishment of endovascular thrombectomy as the standard of care for large vessel occlusion stroke, there has been re-emerging interest in neuroprotective strategies to protect the ischemic penumbra in the prehospital setting, interhospital transfer, and during the endovascular thrombectomy procedure.10,11
Active conductive head cooling is easily applied in the prehospital setting and has shown promise at reducing brain temperature without causing systemic hypothermia. 12 Earlier studies have been performed in sedated patients with intracranial pressure monitors in-situ, facilitating tolerability and invasive measurement of brain temperature.12 –14 COOLHEAD-1 (Tolerability and efficacy of non-invasive head COOLing in normal and HypopErfused brain: A magnetic resonance stuDy) aimed to test the tolerability, and hemodynamic and cooling effects, of active conductive head cooling within timeframes required for interhospital transfer for acute reperfusion therapies. Whole brain echo-planar spectroscopic imaging (EPSI) was used to evaluate whether brain and infarct cooling was achievable, 15 and is a non-invasive method that could, in principle, be applied in future studies evaluating therapeutic hypothermia in stroke patients. 16
Materials and methods
Study population and ethics approval
The New Zealand Northern B Health and Disability Ethics Committee (20/NTB/31) and Auckland District Health Board Research Review Committee (A + 8793) approved this study. The National Ethical Standards for Health and Disability Research and Quality Improvement governed the conduct of this study. All available data are included in this manuscript. The study was prospectively registered with the Australian New Zealand Clinical Trials Registry (ACTRN12620000751976). Healthy volunteers and patients six months after ischemic stroke were invited to participate in this study. Acute and chronic stroke are both characterised by hypoperfused brain that might influence the efficacy of head cooling. Chronic stroke patients were more practical to study than acute stroke patients in this proof-of-concept study. Inclusion criteria required participants to be aged 18 years or older, and able to provide informed consent. Exclusion criteria included a history of uncontrolled hypertension or cardiovascular disease, hematologic dyscrasias that affect thrombosis (cryoglobulinemia, sickle cell disease, serum cold agglutinins), vasospastic disorders (Raynaud's or thromboangiitis obliterans), skin lesions not allowing secure application of the cooling cap, and contraindications to having an MRI scan.
Head cooling
The WElkins Temperature Regulation System, 2nd Gen (TRS-2) was used to actively cool the brain. The TRS-2 consists of a cooling unit which circulates coolant through umbilical tubing into a cooling cap. The cooling cap contacts the participants’ scalp and posterolateral neck. The delivered temperature ranges between –5°C and 15°C. In order to maintain a safe stand-off distance between the system’s ferrous components (e.g. metal housings) and the MRI, the TRS-2 was modified by WElkins to have an extra-long umbilical cord (10 meters), fed through the waveguide so that the cooling unit could be situated outside the magnet room.
Six healthy volunteers (four males and two females) first attended a study visit to specifically assess tolerability and hemodynamic effects of the cooling cap. The volunteers had a mean ± SD age of 37.2 ± 14.8 years, mean ± SD height of 176 ± 8 cm, and mean ± SD weight of 70.3 ± 9.4 kg. The volunteers were placed in Fowler’s position and the cooling cap was applied. Baseline numerical pain rating scale score, heart rate, non-invasive blood pressure, and axillary temperature measurements were recorded. The TRS-2 was then set to a target temperature of –5°C for two hours of cooling. Numerical pain rating scale scores, heart rate, blood pressure, axillary temperature, and shivering were assessed at 10-minute intervals throughout the cooling period.
At a separate study visit, all participants (healthy volunteers and chronic stroke patients) had the cooling cap fitted and underwent baseline imaging, including a magnetization-prepared rapid gradient-echo (MPRAGE) and whole brain EPSI scans. The chronic stroke patients had an additional arterial spin labelling scan in order to quantify the cerebral blood flow within the region of infarction. The healthy volunteers had a rectal temperature probe inserted 10 cm from the anal verge to measure the core body temperature throughout the cooling period. A TeslaM3 MRI-compatible monitor captured core body temperature via the rectal probe at ten-minute intervals. The EPSI sequence was then repeated following 80 minutes of cooling, with the TRS-2 set to a target temperature of –5°C. 11 participants (eight males and three females; five with chronic stroke) underwent 80 ± 5 minutes of active conductive head cooling. The 11 participants had a mean ± SD age of 47.3 ± 16.7 years, mean ± SD height of 178 ± 11 cm, and mean ± SD weight of 78.4 ± 17.0 kg. The five chronic stroke patients had a mean ± SD age of 59.4 ± 9.3 years, mean ± SD height of 181 ± 15 cm, and mean ± SD weight of 88.0 ± 20.0 kg. The mean ± SD volume of middle cerebral artery territory infarction was 148.3 ± 67.1 cm3.
Image acquisition during head cooling
Imaging was performed using a MAGNETOM Skyra 3 T MRI scanner (Siemens Healthcare, Erlangen, Germany), using a 20-channel head and neck coil. A T1-weighted MPRAGE sequence with repetition time (TR)/echo time (TE)/inversion time (TI) = 2200 ms/2.45 ms/900 ms, flip angle = 8°, voxel size =1 × 1 ×1 mm collected in the same orientation as the EPSI sequence with whole-brain coverage, was acquired for anatomical reference and to aid image registration. Magnetic resonance spectroscopy imaging (MRSI) data were collected for the purpose of brain temperature mapping. A whole brain EPSI prototype sequence, 17 was used with the following parameters: TR1 = 1,500 ms (for metabolite data), TR2 = 511 ms (for water reference data); TE = 17.6 ms; lipid inversion nulling with TI = 198 ms; spin-echo excitation with selection of a 120 mm oblique transverse slab covering most of the cerebrum; flip angle = 71°; field of view = 280 × 280 × 120 mm; matrix = 50 × 50 × 8, yielding an acquired voxel resolution = 5.6 × 5.6 ×15 mm, interpolated to 5.6 × 5.6 × 3.8 mm. The total scan time for each EPSI scan was 8 min 1 s. Prior to data acquisition, shimming consisted of a 3D automatic shim, followed by an interactive shim where required to achieve a full width at half maximum ≤30 Hz. Whole brain arterial spin labelling images were acquired with currently recommended parameters, 18 namely a 3D gradient and spin echo readout and pseudo-continuous labelling, background suppression, labelling duration = 1800 ms and single post-labelling delay = 2000 ms. Acquired voxel size was 3 × 3 × 4 mm, field of view = 194 × 194 × 168 mm, phase partial Fourier = 6/8, segments = 6, receiver bandwidth =2694 Hz/Px, TR/TE = 5000/14.84 ms, with each control-label pair repeated eight times, yielding a scan time of 8 min 5 s. An equilibrium magnetization (M0) scan was collected separately, with acquisition parameters matched to the arterial spin labelling scan except only one measurement collected, with no labelling and TR = 4 s, scan duration = 28 s.
Image processing
The Metabolite Imaging and Data Analysis System (MIDAS) software was used to perform image reconstruction and spectral analysis. 19 Quality map settings appropriate for measuring brain temperature were used. 20 Firstly, voxels outside of the brain were excluded based on the tissue segmentation result. Secondly, voxels were excluded if they had a fitted metabolite linewidth outside of the 2 to 12 Hz range. Thirdly, voxels were excluded with the spectral outlier test, where voxels with spectra exceeding 2.5 times the standard deviation of all valid voxels in the image were excluded. The spectral outlier test was only performed on voxels with a parts per million range from 3.6 to 1.7 and did not include major peak regions. In the chronic stroke patients, infarct and non-infarct segmentations were created using 3D Slicer. 21 The MPRAGE scans were exported from MIDAS, saved in Analyze format and imported into 3D Slicer. The infarct segmentations were manually annotated (by WKD), and the non-infarct segmentations were created by mirroring the infarct segmentation onto the contralateral normal brain. The segmentations were then exported as label maps, saved in Analyze format and imported as regions of interest (ROI) for processing with MIDAS using the Mapper tool. Temperature for each ROI was calculated using the MIDAS Project Review and Analysis and Map Integrated Spectrum modules. Project Review and Analysis was used to map the Atlas into subject space. Map Integrated Spectrum was then used to estimate the average temperature in each ROI, including for the whole brain, the Atlas ROIs (frontal, parietal, temporal, occipital, and cerebellar regions) and the infarct and non-infarct ROIs. Temperature was calculated using the metabolites N-acetylaspartate (TNAA), creatine (TCre), choline (TCho) and the amplitude-weighted average (TAve) of all three. TAve was used for the primary analysis. 22 The Bayesian Inference for Arterial Spin Labelling MRI toolbox was used to motion correct the repeat arterial spin labelling data, using the M0 image as a reference, and then average them to generate a single perfusion-weighted image in native space. 23
Statistical analysis
Statistical analysis was performed with Graph Pad Prism version 8.2 (California, USA) and IBM SPSS version 26 (New York, USA). The designated outcome measure for determining sample size was the mean difference between baseline and post-cooling brain temperature. Power calculations conducted with NCSS PASS 12 showed that a minimum of six participants were required to detect a difference of 1°C between baseline and post-cooling temperatures, at 80% power (β = 0.2) and a two-sided statistical significance level of 5% (α = 0.05), with the estimated standard deviation of normal values of 0.6°C. 24 Baseline and post-cooling temperatures were compared using the paired t-test. Repeated measures one-way analysis of variance was conducted to analyze continuous variables during cooling, following confirmation of normal distribution by Shapiro-Wilk testing (P > 0.05). In cases where sphericity was violated (Mauchly’s test P < 0.05), a Greenhouse-Geisser or Huynh-Feldt correction was applied according to whether ε < 0.75. Post-hoc analysis was conducted using multiplicity-adjusted Dunnett’s test to determine the time points in which parameters were significantly different from baseline. Analysis of ordinal data was performed using the Friedman test, with post-hoc analysis conducted using the multiplicity-adjusted non-parametric Dunn’s test. Categorical data were compared using the chi-squared or Fisher’s exact tests. All tests were two-tailed, and P < 0.05 was considered significant. Data are presented as mean ± SD, median (interquartile range), number of participants (% of participants) unless otherwise stated.
Results
Tolerability and hemodynamic effects
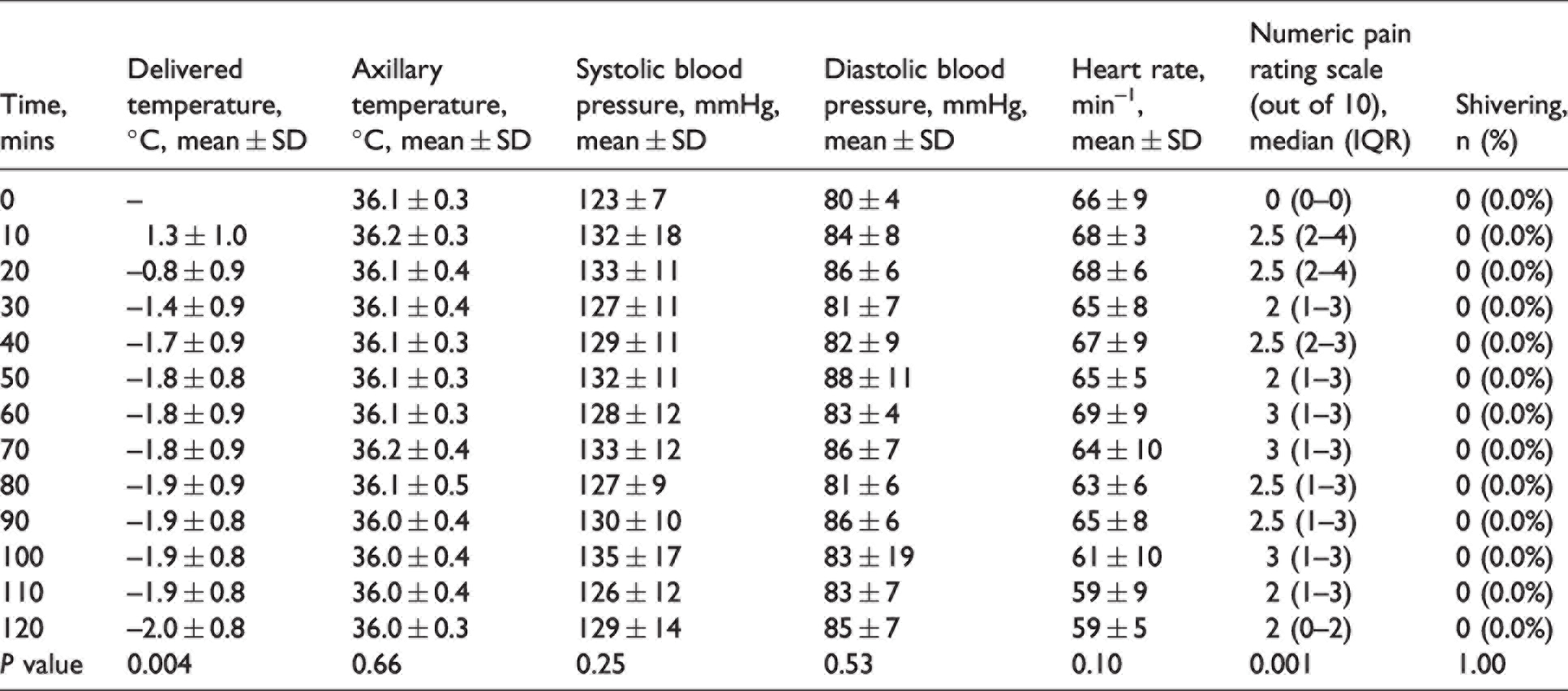
Clinical measurements during cooling are presented in Table 1. By the end of cooling, an absolute temperature delivered by the cooling cap of −2.0 ± 0.8°C was achieved. Axillary temperature, heart rate, and blood pressure did not change significantly during cooling (all P ≥ 0.10). Numerical pain rating scales were greater than baseline at 10, 20 and 100 minutes (all P ≤ 0.03). Shivering was not observed in any participant during cooling.
Clinical measurements of healthy volunteers during active conductive head cooling.
Brain cooling
After quality mapping, the 22 EPSI scans (11 pre- and 11 post-cooling scans) included in the analysis had a mean of 48 ± 8% of the total voxels included in the temperature calculations. The mean pre-cooling rectal temperature in the healthy volunteers was 37.6 ± 0.2°C, while the mean pre-cooling brain temperature in the same participants was TCho = 37.5 ± 0.3°C, TAve =37.2 ± 0.3°C, TNAA = 37.2 ± 0.3°C, and TCr =37.0 ± 0.3°C. The coolant reached an average absolute temperature of –1.3 ± 0.5°C at the end of the 80-minute cooling period. Rectal and brain temperatures (TAve) before and after cooling are summarized in Figure 1. Significant reductions in both rectal temperature (ΔT = –0.3 ± 0.1°C, P = 0.03) and whole brain temperature (ΔTAve = –0.9 ± 0.7°C, P = 0.002) were observed following cooling.
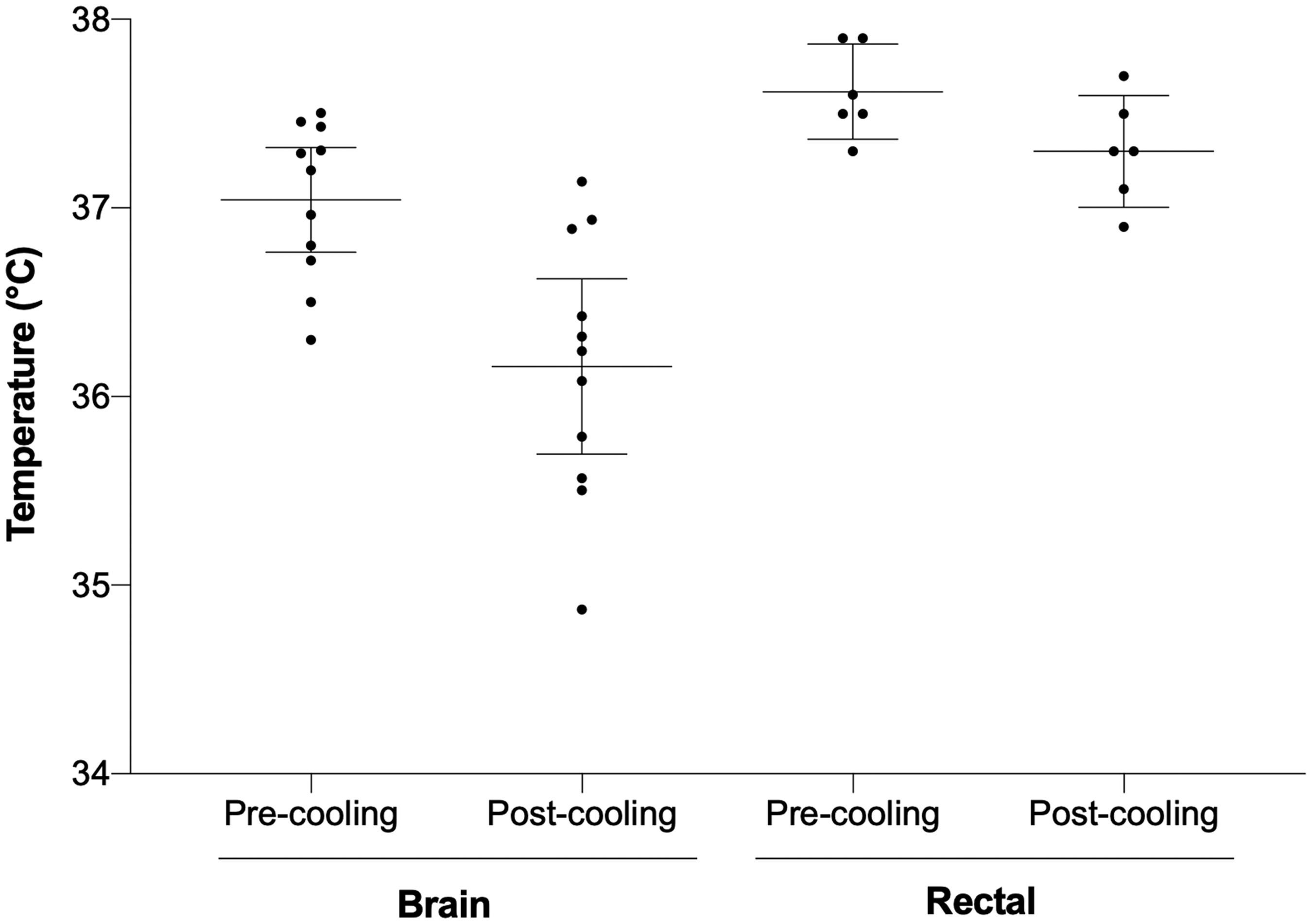
Rectal and brain temperatures before and after 80 minutes of active conductive head cooling. The horizontal lines represent the mean temperature at a given time, and the error bars represent the 95% confidence intervals.
Exploratory analysis of regional brain temperatures after cooling are summarized in Table 2. The occipital lobes had the greatest reduction in temperature (ΔT = −1.5 ± 1.2°C, P = 0.002). In regions of infarction, there was an average perfusion-weighted signal reduction of 36.3 ± 25.6% when compared to the contralateral normal brain. The average temperature reduction in regions of infarction was ΔT = −0.6 ± 0.4°C, P = 0.04, compared to the contralateral equivalent normal brain of ΔT = –0.6 ± 0.2°C, P = 0.002.
Changes in brain temperature by regions of interest.
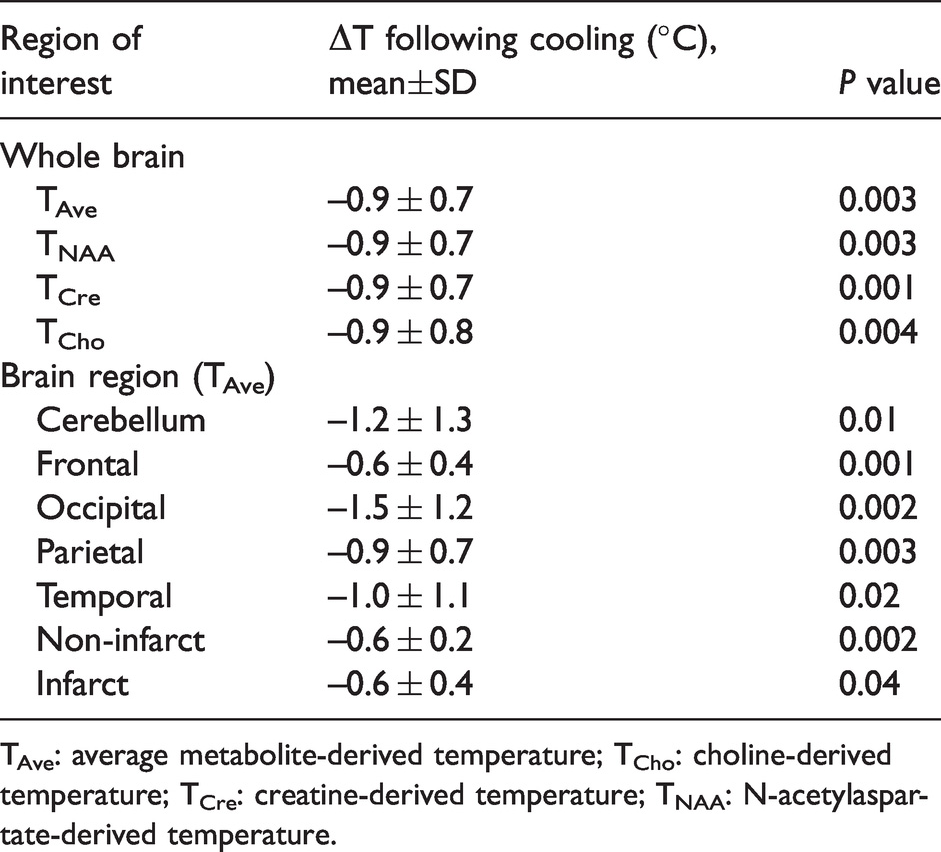
TAve: average metabolite-derived temperature; TCho: choline-derived temperature; TCre: creatine-derived temperature; TNAA: N-acetylaspartate-derived temperature.
Discussion
This study demonstrated that selective brain cooling can be achieved with active conductive head cooling. Using whole brain EPSI, mean brain temperature reduced by 0.9°C within 80 minutes of the cooling cap being applied. Over the same duration, core body temperature reduced by 0.3°C, suggesting that a brain-body temperature gradient can be established. Conductive head cooling was well-tolerated in all participants, and there was no significant change in blood pressure. These results suggest that conductive head cooling could be investigated further in the clinical setting, for example, in stroke patients transferred from primary to comprehensive stroke centers for endovascular thrombectomy. 9
Conductive head cooling was well-tolerated for up to 120 minutes, with only mild local discomfort reported by participants. Earlier studies have also demonstrated favorable tolerability and safety signals in healthy volunteers, stroke, traumatic brain injury, cardiac arrest, and epilepsy.12,13,16,25 –27 However, one major potential adverse consequence of conductive head cooling is induced hypertension, which could prove problematic if it were administered to ischemic stroke patients receiving intravenous alteplase. 28 One study showed that conductive head cooling caused increased systolic blood pressure in patients with severe ischemic stroke, although there was no increased risk of intracerebral hemorrhage. 25 In contrast, the largest randomized study of conductive head cooling in traumatic brain injury patients did not find any differences in hemodynamic parameters, including systolic blood pressure. 14 Blood pressure was measured to assess if a cold pressor response was apparent. In the current study, blood pressure increased by 7.0 mm Hg at the onset of cooling, but this change was not statistically significant.
The magnitude of brain cooling that we observed (0.9°C within 80 minutes) is consistent with earlier studies that measured brain temperature invasively via intracranial pressure monitors.12,14 In patients with severe stroke or traumatic brain injury admitted to the intensive care unit, an earlier version of the TRS-2 (CoolSystem Discrete Cerebral Hypothermia System) achieved a reduction of 1.84°C (range 0.9–2.4°C) in brain temperature (0.8 cm below the cortical surface) when measured at one hour. 12 It took 3.4 hours to achieve brain temperatures of 34°C. 12 In this earlier study, participants’ heads were shaved in order to improve contact between the scalp and cooling cap as mammalian hair has insulating properties, 29 and may attenuate the effect of conductive head cooling. In another study using the same CoolSystem Discrete Cerebral Hypothermia System in traumatic brain injury patients, approximately 0.8°C of brain cooling was achieved at one hour. 14 This is similar to the magnitude of cooling demonstrated in the current study, possibly because neither study shaved participants’ heads.
Non-invasive measurement of brain temperature is challenging. However, MRSI is an established method to estimate brain temperature that correlates well with direct measurements.30,31 The change in the frequency of the temperature-sensitive water peak is compared to one or more metabolite peaks (e.g NAA, Cr, Cho) that are temperature-insensitive. 15 MRSI has previously been used during other forms of head cooling, with reductions in brain temperature demonstrated with forced convective cooling (0.45 ± 0.23°C), 32 intranasal cooling (1.7 ± 0.8°C), 33 and consumption of an ice slurry (0.4°C). 34 These studies have performed MRSI-based temperature measurements at a single brain location, or a limited spatial region.
More recently, EPSI has been developed, allowing whole brain temperature maps to be collected in clinically acceptable scan times. 15 An advantage of this method is that temperature analyses can be performed in virtually any brain region. This is of particular interest for therapeutic hypothermia studies in ischemic stroke, as the ischemic penumbra may be cooled at a different rate to normal brain tissue due to differences in cerebral blood flow. 35 For example, in a patient with reduced cerebral blood flow from internal carotid artery occlusion, conductive head cooling established an interhemispheric gradient of −8°C during 12 hours of cooling. 16 The current study is the first to use whole brain EPSI to demonstrate that conductive head cooling causes brain cooling, including the frontal, parietal, temporal, and occipital lobes, the cerebellum, and regions of (hypoperfused) cerebral infarction.
Following the establishment of endovascular thrombectomy as the standard of care for large vessel occlusion stroke, there has been renewed interest in neuroprotective strategies that could slow infarction prior to reperfusion.10,11 Therapeutic hypothermia is routinely used after cardiac arrest and neonatal hypoxia-ischemia, and confers significant neuroprotection.36,37 We have previously shown that higher median pre-endovascular thrombectomy temperature (per 1°C increase) was an independent predictor of reduced functional independence (odds ratio, 0.66 [95% confidence interval, 0.46–0.94]; P = 0.02), higher modified Rankin Scale scores (common odds ratio, 1.42 [95% confidence interval, 1.08–1.85]; P = 0.01), and increased mortality (odds ratio, 1.65 [95% confidence interval, 1.02–2.69]; P = 0.04). 9 However clinical trials, largely in the pre-reperfusion era, have failed to demonstrated any benefit of therapeutic hypothermia in ischemic stroke.3,4 The most commonly used methods of inducing hypothermia are surface or intravenous cooling, but these are practically challenging in the emergency setting, leading to poor recruitment in stroke trials. 4 The complications associated with inducing systemic hypothermia, such as pneumonia, have also tempered interest.3 –8
Selective hypothermia, where only the head and neck are cooled, might avoid these issues by establishing a temperature gradient between the brain and the body. 12 While intra-carotid cooling may be more effective at reducing brain temperature than conductive head cooling, 38 it cannot be administered in the minutes or hours from the onset of stroke symptoms, during initial assessment in the emergency department, or during interhospital transfer to thrombectomy-capable stroke centers. It is currently unknown whether the modest reductions in brain temperature (e.g. 0.5–2°C) expected with conductive head cooling prior to reperfusion could lead to improved clinical outcomes. Finally, because conductive head cooling relies on heat transfer from the brain, through the skull, and into the circulating coolant, a penumbra-body temperature gradient might be more likely to occur in large vessel occlusion stroke in particular.12,16 This is because normal (warm) cerebral blood flow will not be present to replenish the heat lost to the surroundings (i.e. the circulating coolant). 35
This study has limitations. The number of participants was small, and the use of EPSI to estimate brain temperature is associated with measurement error. 15 The mean pre-cooling brain temperature was lower than rectal temperature, although the 95% confidence intervals overlapped. The sample size did not allow for investigation of the impact of anthropomorphic features, such as height, body weight, head circumference, or hair characteristics on cooling. It is likely that at least some of these factors will impact the effectiveness of conductive heat transfer. In future studies, methods of wetting the hair may improve the heat transfer between the brain, scalp, hair and cooling cap. The modified extra-long tubing meant that the target coolant temperature (–5°C) could not be reached, and it is therefore possible the standard TRS-2 would cause greater selective brain cooling.
In summary, this study demonstrated that selective brain cooling can be achieved with active conductive head cooling, and it is well-tolerated in awake participants. Conductive head cooling could be investigated further in the clinical setting, for example, in stroke patients transferred from primary to comprehensive stroke centers for endovascular thrombectomy.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded with grants from the Auckland District Health Board Myrtle G Martin Neurological Research Fund (2020221) and the Julius Brendel Trust.
Acknowledgements
The authors would like to acknowledge Siemens Healthcare for the provision of the EPSI prototype sequence and Fraunhofer Institute for Digital Medicine MEVIS, for provision of the 3D pCASL sequence. We would also like to thank the imaging staff of the Centre for Advanced Magnetic Resonance Imaging for MRI scanning.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
W.K.D. conceived the study. W.K.D., D.C., C.A.M., and P.A.B. designed the study. W.K.D., C.A.M., and J.C.L. collected in vivo imaging data. M.T.M.W., J.P.D, S.S., C.A.M., and J.C.L. conducted imaging and statistical analyses. W.K.D. drafted the manuscript. All other authors contributed to the intellectual content of the manuscript.
