Abstract
Hypothermia reduces cell death and promotes recovery in models of cerebral ischemia, intracerebral hemorrhage and trauma. Clinical studies report significant benefit for treating cardiac arrest and studies are investigating hypothermia for stroke and related conditions. Both local (head) and generalized hypothermia have been used. However, selective brain cooling has fewer side effects than systemic cooling. In this study, we developed a method to induce local (hemispheric) brain hypothermia in rats. The method involves using a small metal coil implanted between the Temporalis muscle and adjacent skull. This coil is then cooled by flushing it with cold water. In our first experiment, we tested whether this method induces focal brain hypothermia in anesthetized rats. Brain temperature was assessed in the ipsilateral cortex and striatum, and contralateral striatum, while body temperature was kept normothermic. Focal, ipsilateral cooling was successfully produced, while the other locations remained normothermic. In the second experiment, we implanted the coil, and brain and body temperature telemetry probes. The coil was connected via overhead swivel to a cold-water source. Brain hypothermia was produced for 24 h, while body temperature remained normothermic. A third experiment measured brain and body temperature along with heart rate and blood pressure. Brain cooling was produced for 24 h without significant alterations in pressure, heart rate or body temperature. In summary, our simple method allows for focal brain hypothermia to be safely induced in anesthetized or conscious rats, and is, therefore, ideally suited to stroke and trauma studies.
Introduction
Hypothermia has been studied extensively as a treatment for stroke. Early studies established that cooling during global or focal cerebral ischemia greatly diminishes cell death and promotes functional recovery (for comprehensive reviews, see Ginsberg et al, 1992; Maher and Hachinski, 1993; Thornhill and Corbett, 2001; Wagner and Zuccarello, 2005). Although initially controversial (Dietrich et al, 1993), delayed postischemic hypothermia also reduces cell death and improves functional recovery (Carroll and Beek, 1992; Colbourne and Corbett, 1995; Colbourne et al., 2000; Maier et al, 1998; Yanamoto et al, 1996); therefore, hypothermia has great potential as a clinical therapy. Indeed, recent studies show that systemic hypothermia improves survival and recovery in out-of-hospital cardiac arrest victims (Bernard et al, 2002; The Hypothermia After Cardiac Arrest Study Group, 2002). Overall, it appears that hypothermia is the gold standard of neuroprotection for ischemic cell death.
Systemic hypothermia, which is most commonly used, has several side effects that limit its use and perhaps its efficacy. For instance, systemic hypothermia causes shivering, increases the risk of infection and results in cardiovascular complications (Schubert, 1995; Schwab et al, 2001). Such side effects may be avoided through the use of local brain cooling, an old idea that is currently being advocated by many investigators (Wagner and Zuccarello, 2005).
A variety of methods have been used to induce hypothermia in rodents, which are the most commonly used species to model stroke. These techniques fall into several categories, including: (1) focal versus systemic cooling; (2) anesthetized versus nonanesthetized cooling; (3) invasive versus noninvasive; and (4) short-term (e.g., < 12 h) versus prolonged cooling. Besides having clinical relevance, one must consider the practical limitations with these methods (e.g., feasibility, safety, cost). Systemic cooling can be easily produced in anesthetized or conscious animals. For instance, animals can be placed on a cooling blanket or in a cold room. Hypothermia can also be induced pharmacologically (Babcock et al, 1993; Dowden et al, 1999), although maintaining sufficient prolonged cooling while avoiding side effects has not yet been achieved. Local brain cooling can be produced in anesthetized animals with a cooling blanket (Nurse and Corbett, 1994) or coil (Taniguchi et al, 2005) wrapped around the head while body temperature is maintained. Intravascular cooling of the ischemic territory has also been used successfully (Ding et al, 2004). Studies have established that protracted cooling provides superior protection in global (Colbourne and Corbett, 1994) and likely focal ischemia resulting from middle cerebral artery occlusion (MCAO) (Colbourne et al, 2000; Maier et al, 1998; Yanamoto et al, 1996). Long-term cooling (e.g., 24 h) cannot be easily performed in anesthetized rodents due to excessive anesthetic-induced mortality, which is likely due to anesthetic overdose under hypothermic conditions coupled with the inability to take sufficient blood samples (e.g., blood gases) to effectively regulate physiologic variables. Therefore, studies investigating prolonged cooling have used exposure techniques (e.g., fan and water spray) in conscious, mobile rodents (Colbourne et al, 1996).
In this study, we developed a method to selectively lower brain temperature (Tbrain) in otherwise normothermic rats. The method involves placing a metal coil next to the skull. The coil is attached to a cold-water source either directly, as used in anesthetized rats, or through an overhead swivel in conscious rats. The latter allows for considerable animal mobility. Three experiments were conducted. First, we tested whether focal hypothermia could be quickly produced in anesthetized rats. Thus, we measured Tbrain in three locations along with rectal temperature (Trec). The second study used Tbrain and core temperature (Tcore) telemetry probes to test whether prolonged (24 h) local brain cooling was feasible in conscious rats. The third experiment used telemetry probes to assess body and brain temperature along with mean arterial blood pressure (MABP) and heart rate (HR). We report that local cooling can be easily and safely produced in anesthetized or conscious rodents. Accordingly, our method provides a simple and inexpensive way to induce focal brain hypothermia. This technique is, therefore, ideally suited for stroke (i.e., focal ischemia, intracerebral hemorrhage (ICH)) and traumatic brain injury studies.
Materials and methods
Animals
In total, eight male Sprague–Dawley rats (Ellerslie, Edmonton, Alberta, Canada) were used. Rats weighed between 350 and 500 g and were approximately 4 to 6 months old at the time of cooling. Rats were housed individually with free access to food and water. All surgical procedures were performed aseptically and all were in accordance with the Canadian Council on Animal Care guidelines. This study was approved by the Biological Sciences Animal Policy and Welfare Committee at the University of Alberta.
Cooling Coil
A 3 cm length of hypodermic tubing (20 G) was bent into a spiral pattern measuring 8 mm in diameter and 2 mm in thickness (Figure 1). This was achieved by bending a 20-G needle around an immobilized post. This coil was then connected to a 0.76-mm internal diameter silastic tubing (Dow Corning Corp., Midland, MI, USA), through which water could be perfused. The sterilized coil was then surgically placed under the Temporalis muscle on the right side of the skull (see below).

Cooling coil (mm scale bar below) with attached tubing through which cold water is flushed. The coil is placed underneath the Temporalis muscle, with the flattest face adjacent to the skull. In this case, the coil was constructed out of a 20-G needle. Care must be taken to avoid kinking the tube when bending it.
Experiment 1: Focal Brain Cooling in Anesthetized Rats
Rats (N =3) were anesthetized with isoflurane (4% induction; 1.5% to 2% maintenance in 70% N2O and 30% O2) and placed in a stereotaxic frame. A rectal temperature probe estimated body temperature (Trec) that was servo-regulated by a heating pad placed under the animal. The scalp was shaved and treated with Betadine, and an incision was made along the midline of the scalp. The Temporalis muscle was then gently separated from the underlying skull, thereby forming a pocket on the right side. The muscle was not cut. In this space, we placed the cooling coil that was attached via a silastic tubing to a cold-water reservoir placed ∼2 m above the animal (i.e., gravity perfusion). A segment of the tubing close to the rat was placed in an ice bath to cool the water entering the coil, thereby facilitating brain cooling. Flow was initially off. Two burr holes were then made in the skull. One was situated contralateral to the coil at 3 mm left of and 1 mm anterior to Bregma. A thermocouple probe (HYP1-30-1/2-T-G-60-SMP-M, Omega, Stamford, CT, USA) was stereo-taxically lowered 5 mm below the skull surface through this burr hole so that temperature in the contralateral striatum was measured (TCS). The second burr hole was made over the ipsilateral striatum (3 mm right of Bregma, 1 mm anterior). Two thermocouple probes were inserted into this hole—one lowered directly down 5 mm into the ipsilateral striatum (TIS) and the other inserted at 27°C (from vertical) to a distance of 5.59 mm, which placed it in the ipsilateral cortex (TIC) underlying the cooling coil.
After baseline temperature measurements, local brain cooling was achieved when cooled water was allowed to flow through the coil (∼ 180 mL/h) for 60 mins and then stopped. Temperature was recorded every 5 mins during cooling and for 10 mins of re-warming. After cooling, animals were quickly euthanized and thermocouple placement was confirmed by histology (fresh-frozen; sectioned at 40 μm).
Experiment 2: Focal Brain Cooling in Conscious Rats
Brain and Core Probe Implantation: Rats (N =3) were anesthetized with sodium pentobarbital (65 mg/kg intraperitoneally; Somnotol; MTC Pharmaceuticals, Cambridge, ON, Canada). After shaving and treatment with Betadine, a 2 cm incision was made in the abdominal wall. Sterilized telemetry probes (model TA10TA-F40; Transoma Medical, St Paul, MN, USA) that measured the core temperature (Tcore) were then inserted and the wound closed as described previously (DeBow and Colbourne, 2003). Animals were then placed in a stereotaxic frame as described above. First, a small burr hole was made 2 mm lateral and 1 mm anterior to Bregma on the right side. Three surrounding burr holes were also made, into which small metal screws (Model MX-080-2; Small Parts, Miami Lakes, FL, USA) were placed. Second, a 5-mm-long guide cannula (20 G) was placed into the center hole, but not into the brain, and secured with dental cement. Third, we placed the cooling coil between the right Temporalis muscle and the skull. Fourth, we secured an ∼ 2.5 cm length of a plastic cylinder (5 mL syringe barrel) surrounding and centered on the guide cannula. This was secured with dental cement as described and illustrated previously (Colbourne et al, 1996). The head cap assembly subsequently permitted the safe insertion of a Tbrain telemetry probe thermocouple shaft through the guide cannula. The bottom of the 5 mL syringe barrel also held the cooling coil in place, which was further secured by dental cement. Fifth, the wound was closed and the silastic tubing attached to the cooling coil was fed through a flexible wire sheath (model CIH95; 30 cm long, Instech Laboratories, Plymouth Meeting, PA, USA). This end of the sheath was taped securely to the 5 mL syringe barrel to avoid movement of and stress on the cooling coil. The other end of the metal sheath and the silicon tubing was then attached to a swivel (model 375/D/22, Instech Laboratories) and counterbalance arm (CM375BS, Instech Laboratories) mounted to the top of the rat's cage. Rats then recovered from Somnotol anesthesia in their home cages, which rested on RPC-1 receivers (Transoma Medical), while Tcore was recorded every 30 secs via telemetry (A.R.T. v. 2.2; Transoma Medical). Somnotol was used instead of isoflurane as animals remain immobile for hours, allowing time for the dental cement to harden sufficiently.
After a monitoring period of 2 days, the rats were briefly anesthetized (∼ 5 mins of isoflurane) and a telemetry probe (model VM-FH-BP, Mini-Mitter Co. Inc, Sun River, OR, USA) was inserted to measure the temperature of the ipsilateral (to coil) striatum (TIS) at a depth of 4 mm from the skull surface. It was not possible to measure more laterally, because of the need to place a protective cylinder around the brain probe and to have this assembly securely attached to the skull. Owing to signal interference and size restrictions, it was not possible to measure with multiple brain probes. Likewise, it was not possible to simultaneously measure Tcore and TIS via telemetry. Accordingly, TIS was measured for a baseline period of 1 day, while the core probe was turned off (activation/deactivation via magnetic switch). However, it was possible to measure Tcore accurately when the brain probe was on (see below), as the ART software treated the brain probe's signal as noise and excluded it to analyze only the Tcore signal. We have previously used this method to near-simultaneously (within 30 secs) measure Tcore and Tbrain with telemetry (DeBow and Colbourne, 2003).
Focal Brain Cooling in Awake Rats: At 2 days after coil implantation, water was allowed to flow from a reservoir (∼ 2 m above animal) through the coil as described above. Cooling continued for 24 h. The rate of flow was ∼ 100 mL/h. This was lower than that before, owing to the flow restriction imposed by the 22 G swivel, which was needed to allow animal mobility. Striatal temperature (TIS) was continually monitored (every 30 secs) during cooling and Tcore was intermittently sampled (every 4 h) by briefly activating the implanted core probe. Rats remained free to move about throughout these procedures. Water flow was turned off at the end a 24-h period, but TIS was monitored for an additional 24 h. Rats were euthanized a day after cooling with an overdose of somnotol and transcardially perfused with 0.9% saline and then 10% neutral buffered formalin. Coronal sections (40 μm) were taken with a cryostat every 400 μm starting at +1.7 mm to bregma and extending back to −4.8 mm to bregma. Sections were stained with hematoxylin and eosin and examined by an experienced investigator for signs of tissue injury (FC).
Experiment 3: Blood Pressure and Heart Rate Measurements During Cooling
In Experiment 3, rats (N =2) were first operated on to implant TML2 C50-PXT telemetry probe (Transoma Medical) under isoflurane anesthesia (∼ 45 mins duration). In addition to measuring Tcore, this probe has two leads, which are tunneled under the skin, to measure HR, and a catheter, which was inserted into the descending aorta, to measure MABP. The catheter does not obstruct blood flow. After 1 week, rats were operated upon again (Somnotol anesthesia) to implant the guide cannula and cooling coil as described for Experiment 2. However, in this case, the TA10TA-F40 probe was not implanted because the C50-PXT probes measure Tcore. After a 1-day recovery period, the brain probe was inserted under brief isoflurane anesthesia and the C50-PXT probe was turned off to allow Tbrain recordings. Brain temperature was recorded for 24 h, during which time the C50-PXT probe was turned on and cooling was initiated. Brain temperature was measured every 4 h by intermittently turning off the C50-PXT probe for < 1 min. In this study a larger bore swivel was used (375/D/20, Instech Laboratories), which allowed a flow rate of ∼ 150 mL/h. The temperature of the water was ∼ 11°C at the entry point into the coil.
Data Analysis
All data are presented as mean ± standard deviation (s.d.). Using SPSS (v. 12) software, data were analyzed using analysis of variance. A P-value of < 0.05 was considered statistically significant.
Results
Experiment 1
Focal hypothermia was quickly (within minutes) produced in the ipsilateral hemisphere during the cooling protocol (Figure 2). The average TIS during cooling (31.3°C ± 0.6°C) was 5°C below baseline (36.3°C ± 0.3°C), which was statistically significant (P < 0.01). The average TIC during cooling (26.7°C ± 0.8°C) was more than 9°C cooler than baseline (36.2°C ± 0.6°C; P < 0.01). While the TCS was statistically (P < 0.05) lower during cooling than baseline, the difference amounted to only 0.6°C (36.1°C ± 0. 3°C versus 36.7°C ± 0.2°C) and thus TCS was well within the normothermic range. The average Trec during the focal cooling did not significantly change (37.7°C ± 0.3°C versus 36.9°C ± 0.3°C; P > 0.05). Thermocouple probe placement was found to be accurate in all animals (data not shown).
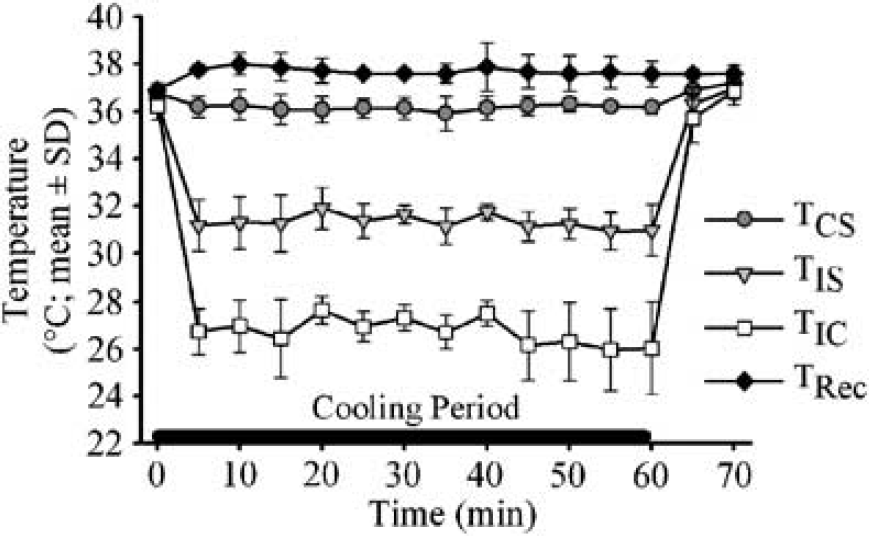
Temperature (contralateral striatum—TCS; ipsilateral striatum—TIS; ipsilateral cortex–TIC; rectum—Trec) before, during and after induction of cooling (black bar on x-axis) in anesthetized rats. Ipsilateral cooling was significant, whereas Trec and TCS remained normothermic.
Experiment 2
Figure 3 shows the average Tcore and Tbrain of rats during baseline and the 24-h cooling period. During cooling, the Tcore was only intermittently recorded, owing to technical limitations with telemetry probes as noted in Materials and methods. Cooling was induced within a few minutes and the 24-h average TIS during cooling (34.7°C ± 0.2°C) was significantly lower than baseline (37.1°C ± 0.1°C; P < 0.01). The Tcore during cooling (37.4°C ± 0.2°C) was not significantly different from baseline (37.5°C ± 0.2°C; P > 0.05).
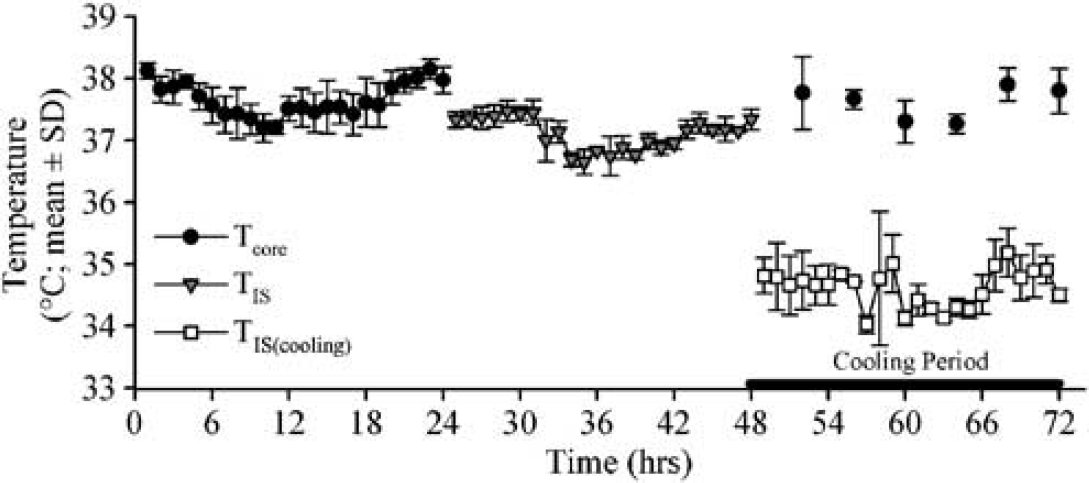
Body and brain temperature during baseline readings (0 to 48 h) and during cooling (black bar from 48 to 72 h). The ipsilateral striatal temperature (TIS) was significantly reduced during cooling, whereas body temperature (Tcore) remained normothermic.
Examination of several coronal sections for these animals revealed no signs of pathology (e.g., acidophilic neurons) aside from the direct injury caused by the brain probe insertion (data not shown).
Rats tolerated the cooling setup well (e.g., no loss of weight). Some rats initially scratched at the head cap assembly, but this stopped within a few hours, which was by the time they had completely aroused from Somnotol. Some rats also initially scratched during cooling; however, this behavior quickly ceased.
Experiment 3
Physiologic variables (i.e., HR, MABP, Tcore, TIS) during baseline and cooling periods for Experiment 3 are shown in Figure 4. The Tcore (Figure 4A) did not significantly change during the 24-h focal cooling (37.6°C ± 0.5°C versus baseline value of 37.7°C ± 0.1°C; P > 0.05). The TIS, however, was significantly cooler (33.4°C ± 0.6°C during cooling versus baseline average of 37.2°C ± 0.4°C; P < 0.05). Cooling did not significantly affect MABP (Figure 4B; 114.571.4 mm Hg versus baseline average of 117.7 ± 5.5 mm Hg; P > 0.05). Similarly, HR was not significantly altered during cooling (Figure 4C; 347.7 ± 34.8beats/min versus baseline of 361.57 40.6 beats/min; P > 0.05). The activity and health of rats were monitored closely and found to be comparable to the rats in Experiment 2.
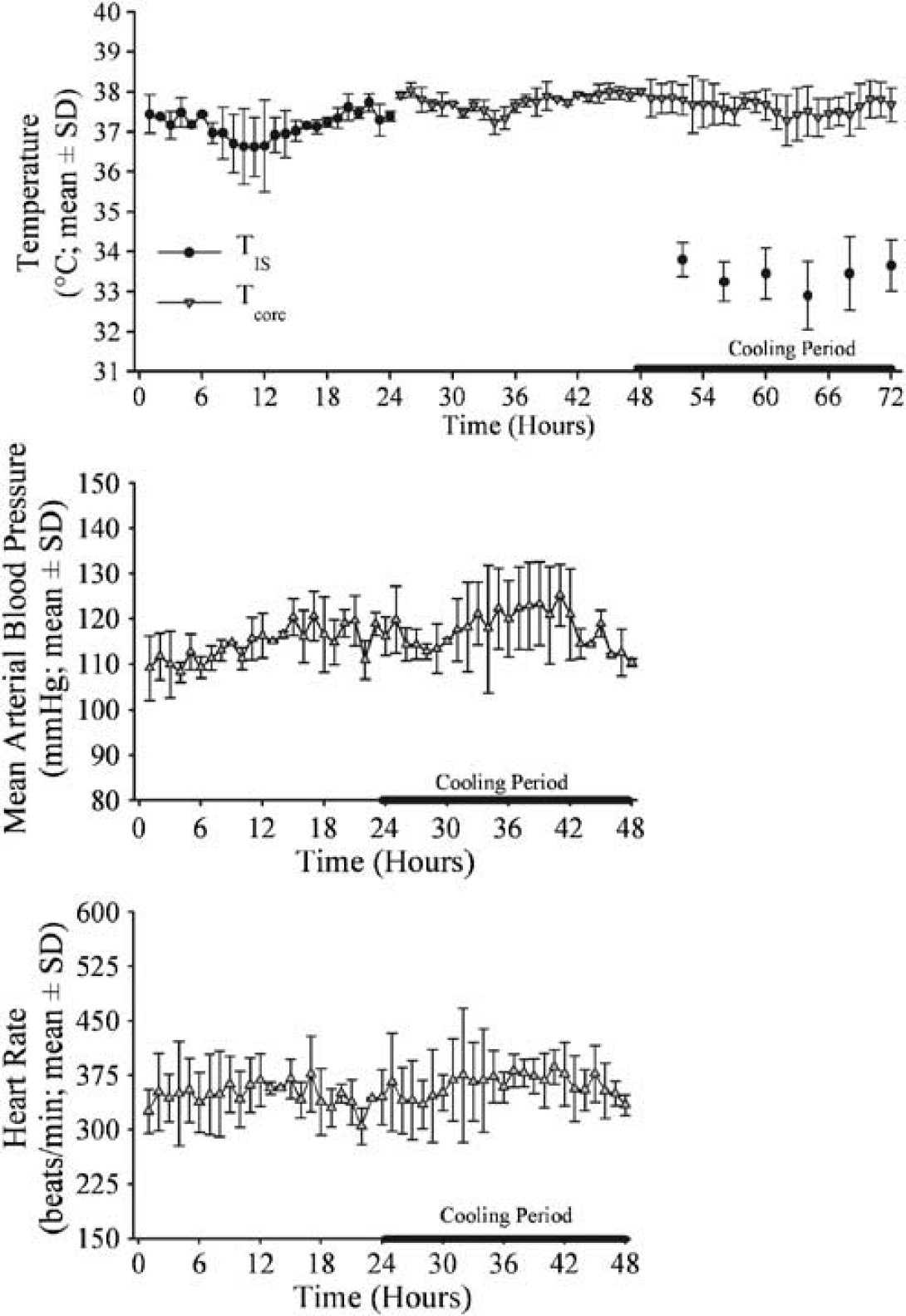
Temperature (ipsilateral striatum—TIS; body temperature—Tcore; top graph), MABP (middle graph) and HR (bottom graph) during baseline and cooling (black bar) periods. Significant local hypothermia occurred in otherwise normothermic rats. HR and MABP did not substantially change during cooling.
A comparison of the average TIS during cooling revealed that significantly (P < 0.05) greater cooling was achieved in Experiment 3 (20 G swivel; B150 mL/h flow rate) than Experiment 2 (22 G swivel; ∼ 100 mL/h flow rate). Therefore, a higher flow rate caused greater cooling. The temperature reduction was even greater in the first experiment, owing to the lack of a swivel (e.g., minimizing heat transfer) and/or a higher flow rate (∼ 180 mL/h), which was due to the lack of a swivel. We did not use the swivel, as the rats were anesthetized in Experiment 1.
Discussion
Herein we describe a simple, safe and effective method to induce local brain hypothermia in anesthetized or awake rats. The technique involves surgically placing a small, metal, coiled tube adjacent to the skull and subsequently flushing the coil with cold water. Our first experiment showed that local cooling of one hemisphere is possible without cooling either the contralateral hemisphere (TCS) or the body (Trec) when the animal is anesthetized. Second, we showed that prolonged mild hypothermia is easily produced in conscious rats and that local brain cooling for 1 day did not affect Tcore. Third, we showed that local brain cooling did not change HR or MABP. Furthermore, local cooling also did cause brain cell death, which is in line with earlier observations that mild hypothermia does not cause structural alterations in hippocampal neurons (Colbourne et al, 1999). Given that therapeutic levels of local brain hypothermia are safely achieved, our cooling method is ideally suited to studies of focal brain injury such as occurs with MCAO, ICH and traumatic brain injury.
Our method has several advantages over current techniques to induce systemic hypothermia. First, our method allows for a greater reduction in Tbrain than that safely achieved with whole-body cooling, where complications can occur at any hypothermic temperature, but especially those below 32°C (Schubert, 1995). Mild systemic cooling (e.g., 33°C) with water spray and fans significantly alters MABP and HR in rats (Colbourne et al, 2003; MacLellan et al, 2004) and increases loss of body weight (Colbourne and Corbett, 1995). Our local cooling method avoided these complications. Importantly, side effects may impact efficacy, as found recently in an ICH model in rats (MacLellan et al, 2004). Accordingly, we predict that, at least in some situations, local cooling would be superior to systemic hypothermia, given an equivalent brain target temperature. Second, our present method achieved a stable level of cooling (versus baseline temperature) without the need for any adjustment; although it is recognized that this may not always be the case and variability may be underestimated because we used small group sizes. Conversely, induced hypothermia through use of water spray and fans requires constant servo-regulation, thus necessitating expensive telemetry recording equipment and a sophisticated control system (Colbourne et al, 1996; DeBow and Colbourne, 2003). Focal cooling, however, could be done without invasive and expensive temperature recordings once an ideal cold-water flow rate has been established. In this case, investigators are strongly encouraged to determine the ideal cooling parameters for their particular setup, as it would vary depending on water temperature, flow rate and coil design among other factors. We recommend the 20 G swivel (versus 22 G) for producing hypothermia in conscious animals, owing to the greater cooling with gravity-induced water flow, which could be replaced and augmented with forced perfusion. The amount of cooling would also depend on the metabolic activity of underlying tissue (e.g., greater cooling during ischemia) as well as generalized temperature responses to the insult. For instance, many (Abraham et al, 2002; Reglodi et al, 2000; Zhao et al, 1994), but not all (Corbett et al, 2000), MCAO studies report that spontaneous hyperthermia occurs. Accordingly, this would make it more difficult to achieve a target temperature with our local cooling method. Nonetheless, this problem can be circumvented by increased cold-water perfusion. Using a servo-controlled cold-water flow system analogous to our computerized systemic-cooling apparatus (Colbourne et al, 1996) would offer additional flexibility, but also increase system complexity and cost.
Our technique also offers advantages over current local cooling methods such as using a cooling blanket (Nurse and Corbett, 1994) or coil wrapped around the head of anesthetized animals (Taniguchi et al, 2005). First, our method does not result in cooling of the entire head, which means that less or no systemic cooling will occur. Second, other local cooling methods require anesthesia, which potentially confounds the use of hypothermia and limits the duration of cooling obtainable. Cooling under anesthesia also increases costs and severely limits the number of animals that can be cooled concurrently. With our system, many animals can be simultaneously cooled at a low cost. Third, our method is minimally invasive, as it does not require either vessel catheterization or intracranial placement of a cooling device as is required with some other methods. Fourth, local cooling can be done in mobile, conscious rodents, allowing for simple behaviors (e.g., activity levels, spontaneous rotation, paw usage) and physiology to be easily monitored throughout cooling. Fifth, various depths and durations of hypothermia can be easily induced by simply changing the cold-water temperature or flow rate. Indeed, this was shown in comparing Experiments 2 and 3. A significantly greater decrease in TIS occurred in Experiment 3, owing to using a larger bore swivel, which allowed for a greater flow rate.
The described method is also limited in its scope. The coil is best suited to single-hemisphere injuries such as MCAO, ICH or focal traumatic brain injury. A gradient of cooling also exists with cortical structures undergoing cooling to a greater extent than subcortical structures. Thus, in MCAO models (e.g., intraluminal suture occlusion), one may protect the cortex better than the striatum, in part owing to temperature gradients. Other local cooling methods, such as a cooling blanket wrapped around the head, likely also have this limitation. Therefore, temperature gradients must be considered in such studies and should be determined in each situation (e.g., model, species). Our method is not entirely suitable for global ischemia studies, as only one hemisphere would be protected; although this might be considered an advantage if an internal control was desired (i.e., a normothermic hemisphere). Given the temperature gradient shown in Experiment 1, it is likely that wide structures, such as the hippocampus, might experience a gradient in protection. However, global brain injuries could potentially be treated if two coils were implanted—one on each side to provide whole-brain cooling, perhaps with smaller temperature gradients. However, cooling the entire head in this manner would likely increase the risk of developing systemic hypothermia and associated complications. Thus, further study with this method is needed if it is to be used to treat global ischemia. Likewise, focal cooling to moderate or severe hypothermia levels (e.g., 20°C) would likely increase the risk of systemic hypothermia. Such deep levels may only be feasible if either external heating was provided to the rest of the body or if a combination of local deep hypothermia and mild systemic cooling was acceptable, as done recently in neonatal pigs (Tooley et al, 2003). Nonetheless, we have shown that prolonged mild hypothermia, in the therapeutic range for global (Colbourne and Corbett, 1995; Hickey et al, 2000) and focal ischemia (Colbourne et al, 2000; Maier et al, 1998; Yanamoto et al, 1996) and ICH (MacLellan et al, 2004), can be safely achieved without systemic problems. Another limitation is that we did not assess this technique in either the gerbil or mouse, which are common stroke models. We expect that similar results can be achieved with a smaller coil suitable for these species, but further study is needed to test efficacy, safety and practicality. It should be noted that we are not advocating this method for larger animals (e.g., pigs), including humans. Owing to thicker skulls and larger brains, it is highly unlikely that the current method would be sufficiently effective nor would it be better than devices placed under the skull. Finally, we did not vary either the rate of cooling or rewarming. Our method induces rapid hypothermia, which is followed by rapid rewarming on cessation of water flow, which was gravity powered. If slower rates are desired, the water flow rate could be slowed; however, this would have to be actively monitored and regulated (e.g., infusion pump, flow regulator).
In summary, the described method offers investigators a way to study focal brain hypothermia that is effectual, simple, cost-effective and safe in rats. Importantly, this method allows for more flexibility than is available with current methods of cooling. This will permit researchers to better identify the ideal hypothermia treatment (e.g., depth and duration of cooling) as well as uncover its mechanisms of action. Our method also suits the growing interest in using local cooling in the clinic (Wagner and Zuccarello, 2005). We hope that this simple method will encourage more investigators to study therapeutic (mild and prolonged) hypothermia, which, at present, is arguably the best neuroprotectant.
Footnotes
Acknowledgements
The authors thank Isaac Lank and Sobia Kamal for technical assistance.
