Abstract
Ca2+/calmodulin-dependent protein kinase II alpha (CaMKIIα) is a major contributor to physiological and pathological glutamate-mediated Ca2+ signals, and its involvement in various critical cellular pathways demands specific pharmacological strategies. We recently presented γ-hydroxybutyrate (GHB) ligands as the first small molecules selectively targeting and stabilizing the CaMKIIα hub domain. Here, we report that the cyclic GHB analogue 3-hydroxycyclopent-1-enecarboxylic acid (HOCPCA), improves sensorimotor function after experimental stroke in mice when administered at a clinically relevant time and in combination with alteplase. Further, we observed improved hippocampal neuronal activity and working memory after stroke. On the biochemical level, we observed that hub modulation by HOCPCA results in differential effects on distinct CaMKII pools, ultimately alleviating aberrant CaMKII signalling after cerebral ischemia. As such, HOCPCA normalised cytosolic Thr286 autophosphorylation after ischemia in mice and downregulated ischemia-specific expression of a constitutively active CaMKII kinase proteolytic fragment. Previous studies suggest holoenzyme stabilisation as a potential mechanism, yet a causal link to in vivo findings requires further studies. Similarly, HOCPCA’s effects on dampening inflammatory changes require further investigation as an underlying protective mechanism. HOCPCA’s selectivity and absence of effects on physiological CaMKII signalling highlight pharmacological modulation of the CaMKIIα hub domain as an attractive neuroprotective strategy.
Introduction
Even though ischemic stroke is one of the leading causes of death and disability, 1 treatment options are limited. Thrombolysis using recombinant human tissue-type plasminogen activator (tPA, alteplase) or clot retrieval using mechanical thrombectomy have been shown to be effective in alleviating ischemic injury. However, only a small number of patients meet the criteria for treatment.2,3 Despite a great need for novel therapies targeting ischemic cell death mechanisms and enhancing functional outcome, so far, no cytoprotective therapy has successfully reached the market (reviewed in 4 ). Yet, positive results from a recent phase III trial (ESCAPE-NA1) bring new hope to the field, showing the feasibility of translating cytoprotective strategies to the clinic. 5
The enzyme calcium/calmodulin-dependent kinase II alpha (CaMKIIα) is a major regulator of both physiological and pathological Ca2+ signals downstream of glutamate receptor activation, and has recently gained increasing attention as an attractive target for novel neuroprotective therapies.6–10 CaMKII has four subtypes, whereof CaMKIIα and CaMKIIβ are highly expressed in cerebral neurons forming large homo- and/or heteromeric structures made up of 12 to 14 subunits. Each subunit consists of a kinase, regulatory domain, linker, as well as a hub association domain. 11 The enzyme is most known for its role in synaptic plasticity, where fine-tuned regulation by Ca2+ signals, multiple autophosphorylation sites as well as subcellular targeting enables a dual role of CaMKII in promoting both long-term potentiation and long-term depression (LTP and LTD).12,13 As such, elevated intracellular Ca2+ leads to stimulated activity mediated by Ca2+/CaM binding to the regulatory segment. Subsequent inter-subunit autophosphorylation at the central residue Thr286 (Thr287 in CaMKIIβ) infers Ca2+-independent autonomous activity, which is associated with translocation of the holoenzyme to the postsynaptic density (PSD) region in excitatory synapses, where it co-localises with N-methyl-D-aspartate (NMDA) and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors.14,15 Conversely, autophosphorylation of Thr305/306 (Thr306/307 in CaMKIIβ) can block Ca2+/CaM re-binding and inhibit further Ca2+-stimulated activity (reviewed in16,17).
During ischemia, intracellular Ca2+ overload leads to CaMKII dysregulation, as evidenced by an increase in autophosphorylation levels, long-lasting autonomous activity, and augmented PSD association.8,12,18,19 Furthermore, we recently reported the ischemia-specific cleavage of autophosphorylated CaMKII by calpain, releasing a constitutively active and stable CaMKII fragment (ΔCaMKII), which possibly contributes to enhanced substrate phosphorylation after ischemia. 19 However, the individual contribution of these ischemic effects on CaMKII to cell death is unclear, and conflicting reports exist about CaMKII’s involvement in cell survival and cell death (reviewed in 20 ). Remarkably, autonomous activity has been suggested as the main contributor to ischemic cell death based on strong neuroprotective effects with the peptide inhibitor, tatCN21.6,7 Yet, increased vulnerability to focal ischemia after conventional CaMKIIα knockout also indicates a role of CaMKII in cell survival.8,21 Due to CaMKII’s major role in cell signalling in both physiological and pathological conditions, there is a need to understand how CaMKII promotes cell death or survival to guide future pharmacological treatments.
We recently reported that pharmacological modulation of CaMKIIα by γ-hydroxybutyrate (GHB) and the closely related small molecule, 3-hydroxycyclopent-1-enecarboxylic acid (HOCPCA), is neuroprotective when administered 30 min – 12 h after a photothrombotic stroke (PTS) in mice.8,22 GHB itself is a metabolite of the main inhibitory neurotransmitter γ-aminobutyric acid (GABA), and its neuroprotective properties have been described by several groups, however the mechanism has remained elusive.23–25 Whereas GHB also displays a low affinity for GABAB receptors, possibly contributing to its neuroprotective actions via hypothermia, 26 its brain-penetrant analogue HOCPCA is endowed with pronounced selectivity and distinct affinity for the alpha subtype of CaMKII only, which is evident by a complete absence of binding of radiolabelled HOCPCA to brain tissue from Camk2a−/− mice.8,27 Furthermore, we have previously performed screening of HOCPCA against 45 different neurotargets and found a 100-fold selectivity for the CaMKIIα binding site. 28 This generally correlates well with its hydrophilic nature and preference for the highly solvated hub domain cavity. 29 In contrast to ‘classical’ CaMKII inhibitors, 30 HOCPCA binds to a deep cavity of CaMKIIα’s hub domain. 8 Its binding has no direct effect on substrate phosphorylation or Thr286 autophosphorylation, but instead leads to substantial stabilisation of the oligomeric state of the hub domain when tested in a thermal shift assay. 8 Changes in CaMKII oligomerisation upon HOCPCA binding are believed to alter holoenzyme functionality, yet the molecular mechanism and how this results in neuroprotection is unclear. Nonetheless, pharmacological modulation of the CaMKIIα hub domain presents an interesting therapeutic avenue, particularly because HOCPCA did not affect physiological CaMKIIα signalling, such as LTP. 8
HOCPCA’s unique properties led us to characterise its neuroprotective potential further. Here, we present HOCPCA’s in vivo neuroprotective effect in two clinically relevant experimental stroke models in mice: the permanent model of distal middle cerebral artery occlusion (dMCAO) and the thromboembolic stroke model. To investigate treatment effects on CaMKIIα biochemistry early after ischemia, we assessed CaMKII expression, autophosphorylation levels, as well as the presence of the cleavage product ΔCaMKII in membrane and cytosolic fractions after dMCAO. Moreover, we investigated the effects of HOCPCA on hippocampal function post-dMCAO.
Materials and methods
Materials
The sodium salt of HOCPCA (3-hydroxycyclopent-1-enecarboxylic acid) was synthesized as previously described. 28 Mouse α-thrombin (#90792 MIIA) was obtained from Stago BNL (the Netherlands) and tPA (Actilyse®) from Boehringer Ingelheim (Germany). A detailed description of the antibodies used is provided in the supplementary material.
Animals
All animal experiments were approved by and conducted in accordance with the Danish Animal Health Care Committee (J.nr. 2019-15-0201-01620), French ethical laws (Decree 2013-118), European Communities Council guidelines (2010/63/EU), and the University of Otago Animal Ethics Committee. Studies are reported after the ARRIVE guidelines (Animal Research: Reporting In Vivo Experiments). Mice were allocated to treatment groups at random. All experiments were conducted and analysed with experimenters blinded to treatment groups. Mice were excluded if the stroke surgery was unsuccessful and no infarct or a haemorrhagic transformation was observed. Studies were performed in three different laboratories and the respective experimental details (e.g., strain, sex, weight, age, vendor, total number of mice, dead and excluded animals) are provided in Suppl. Table 1 for overview.
dMCAO
Permanent occlusion of the distal middle cerebral artery (MCA) was performed by electrocoagulation as previously described with small deviations in anaesthesia and post-operational care between different laboratories (see Suppl. Table 1).31–33 Sham surgeries were performed in the exact same way, except that the surgery was performed without MCA electrocoagulation.
Thromboembolic stroke
Thromboembolic stroke surgery was performed as previously described 34 by injection of 1 µl of murine α-thrombin (1.0 IU) with a custom-made glass micropipette into the lumen of the MCA to induce in situ formation of a clot. Cessation of cerebral blood flow was monitored by laser Doppler flowmetry using a fiber-optic probe (Oxford Optronix, Abington, UK). Sham surgeries were performed the exact same way, but mice were not injected with α-thrombin.
Compound treatment
Mice were treated with an intraperitoneal (i.p.) injection of 175 mg/kg HOCPCA diluted in dH2O (10 mg/ml stock solution) or saline control either 30 min or 3 h post-stroke. tPA was administered intravenous (10 mg/kg, 10% bolus/90% infusion over 40 min) or the same volume of 0.9% saline starting from 20 min after α-thrombin injection via a catheter inserted into the tail vein.
Infarct size assessment
Mice surviving 3-days post-dMCAO were euthanized by cervical dislocation, brains were extracted and snap-frozen in gaseous CO2. Mice surviving 14-days post-dMCAO were anaesthetised, transcardially perfused with 4% paraformaldehyde, and brains were extracted. For both experiments, brains were processed into 30 µm-thick coronal sections, which were processed histologically using cresyl violet staining to quantify infarct volume as previously described.8,33,35 After thromboembolic stroke, T2-weighted images were obtained by magnetic resonance imaging on a Pharmascan 7 T (Bruker, Germany) for infarct size determination as previously described.36,37 T2*-weighted images were obtained to examine if a haemorrhagic event occurred.
Behavioural assessment
Mice were tested for sensorimotor and cognitive performance as described previously in the grip strength,36,38 bilateral tactile stimulation,39,40 vibrissae-paw,40,41 novel object recognition (NOR) 42 and y-maze 43 test. A brief description of each test is provided in the supplementary methods.
Electrophysiological experiments
Mice were anaesthetised two-weeks post-dMCAO with isoflurane, decapitated, and brains were rapidly removed. Coronal slices (400 µm-thick) were cut with a vibratome (Campden Instruments model #MA752) in ice-cold artificial cerebrospinal fluid (aCSF (in mM): NaCl 124, KCl 5, NaH2PO4 1.25, NaHCO3 26, MgSO4 1.3, CaCl2 2, glucose 10; pH 7.4; gassed with Carbogen: 95% O2/5% CO2). Electrophysiological assessment was carried out as previously described8,44, as reported in the supplementary material.
Subcellular fractionation and Western blot
Mice were euthanized by cervical dislocation. Brains were isolated and snap-frozen on crushed dry ice. For dissection of peri-infarct cortical tissue, 1 mm-thick coronal sections were cut in the chamber of a CM1860 cryostat at −20°C, and peri-infarct tissue was collected from the top quadrant of the left hemisphere using a tissue punch. The subcellular fractionation protocol was adapted from previous reports. 45 In brief, tissue homogenization was achieved on ice using a tissue douncer (15 strokes) in 1 ml homogenization buffer containing 0.32 M sucrose, 1 mM NaHCO3, 1mM MgCl2, 10 mM HEPES, pH 7.4, as well as 1.5% cOmpleteTM protease inhibitor cocktail (Roche Diagnostics), 1% phosphatase inhibitor cocktail 2 (P5726, Sigma-Aldrich) and 1% phosphatase inhibitor cocktail 3 (P0044, Sigma-Aldrich). Homogenates were centrifuged at 94 × g for 5 min at 4°C in a table centrifuge (5424 R, Eppendorf, Germany). The supernatant was transferred to a new tube and again centrifuged at 4°C for 5 min at 184 × g. After this removal of cell debris, the supernatant was centrifuged at 21,130 × g for 15 min at 4°C, resulting in the cytosolic-enriched supernatant (S2) and membrane-enriched pellet (P2). The pellet was resuspended in 300 µl 1 mM HEPES buffer, and the supernatant was saved. Protein concentration was determined with the BCA protein assay kit (Pierce). Western blot analysis of subcellular fractions and whole-cell homogenate of peri-infarct cortical tissue was performed as previously described. 8
Statistics
Statistical analysis was performed using GraphPad Prism (v.9). All data were tested for normal distribution with the Kolmogorov-Smirnov test. Details of specific statistical tests are displayed within the figure legends. Statistical significance was accepted at p < 0.05.
Results
HOCPCA is neuroprotective after dMCAO
We have previously shown that a single dose of HOCPCA is neuroprotective when given 30 min and 3–12 h after PTS, with effects on both infarct size and motor function. 8 As ischemic stroke is a clinically heterogeneous disease, it is recommended to study potential neuroprotectants in multiple models covering different disease aspects.46,47 Here, we tested HOCPCA after dMCAO, which shows a more pronounced ischemic penumbra compared to PTS.48–50 Hence, we initially treated mice intraperitoneally with 175 mg/kg HOCPCA or saline control early after dMCAO (30 min). Similar to Bach et al., neuroprotective outcomes were evaluated 3 days post-stroke by cresyl violet staining and grip strength test (summarized in Figure 1(a)). 38 HOCPCA treatment significantly reduced infarct volume by 26% compared to saline-treated mice (16.6 ± 5.9 mm3 for saline versus 12.3 ± 6.2 mm3 for HOCPCA, p = 0.0485; Figure 1(b)) and improved acute deficits in forelimb grip strength. Saline-treated mice displayed a significant asymmetry between the strength of the left and right front paw after dMCAO (p = 0.0129; Figure 1(c)). This asymmetry was alleviated by HOCPCA treatment (Figure 1(c)). Intrigued by the neuroprotective effects of HOCPCA achieved with treatment 3–12 hours after PTS, 8 we next tested if HOCPCA is also neuroprotective when given at a time when infarct development is known to have significantly progressed, i.e., 3 h post-dMCAO. 50 Whereas such late treatment with HOCPCA did not influence infarct volume (19.5 ± 6.9 mm3 for saline versus 20.4 ± 7.8 mm3 for HOCPCA; Figure 1(d)), the effect on alleviating grip strength asymmetry persisted. As such, dMCAO introduced grip strength asymmetry in saline-treated mice (p = 0.0035), which was alleviated by HOCPCA treatment (Figure 1(e)).
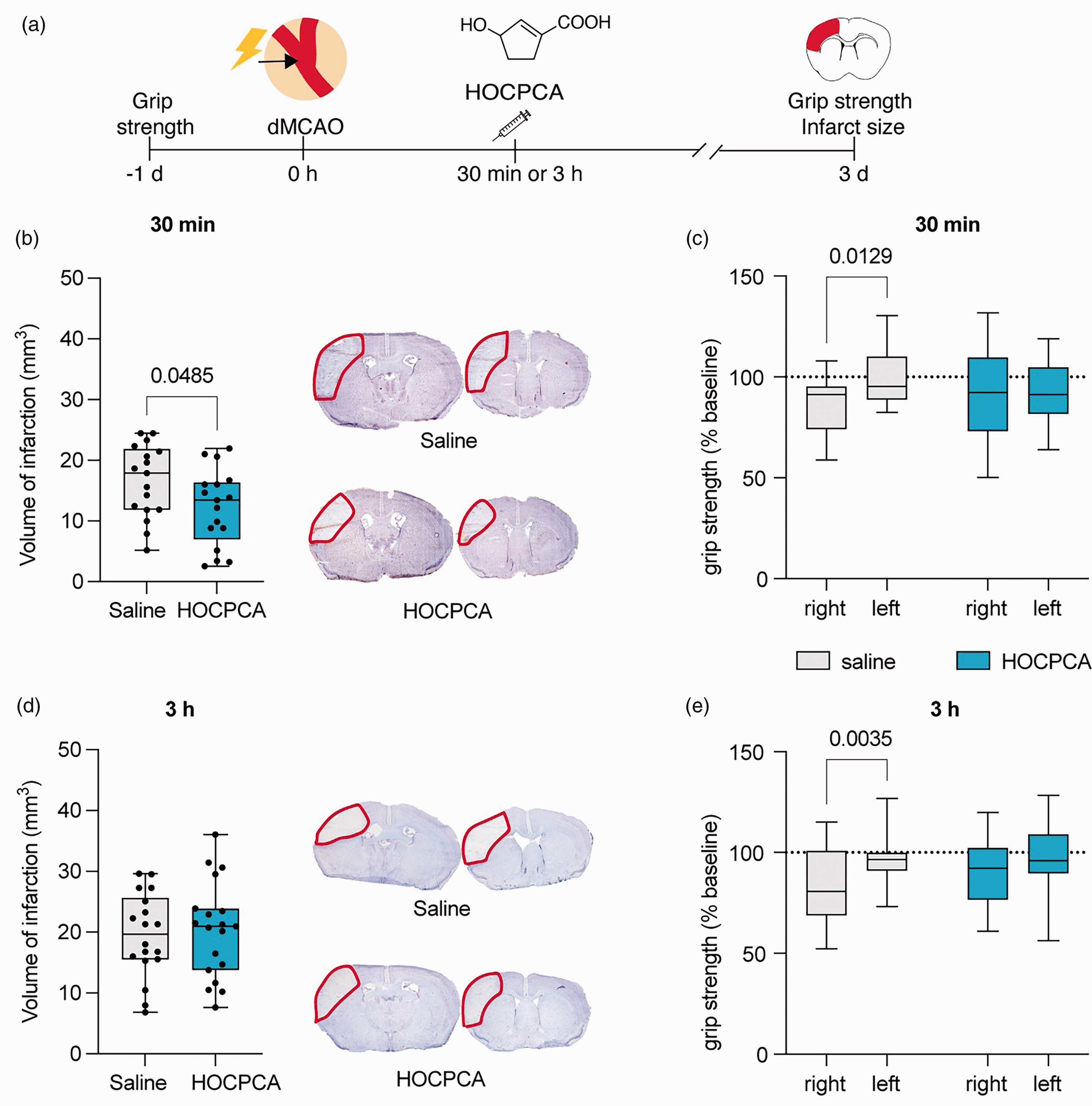
Effect of HOCPCA on infarct size and motor function after dMCAO. (a) Schematic illustration of experimental timeline. (b–e) Adult male mice were treated at 30 min (b–c, n = 17/group) or 3 h (d–e, n = 18–19/group) after dMCAO with 175 mg/kg HOCPCA or saline control (intraperitoneal). Infarct volumes were assessed 3 days post-stroke (Left), and representative cresyl violet stained tissue sections are shown (Right) (b, d; two-tailed Student’s t test) and (c, e) Grip strength asymmetry was evaluated 3 days post-stroke and is shown as % of baseline grip strength for the right (contralateral) and left (ipsilateral) front paw (c, e; two-way repeated measures ANOVA (front paw, treatment), post hoc Šídák’s test) (Box plot (boxes, 25–75%; whiskers, minimum and maximum; lines, median).
HOCPCA reverses pThr286 levels in the cytosolic fraction of peri-infarct cortex after stroke
Prompted by HOCPCA’s neuroprotective effects after dMCAO, we next investigated the potential mechanism of action in vivo. Given HOCPCA’s well-characterised binding to the CaMKIIα hub domain, 8 an allosteric effect was envisaged. However, little is known about HOCPCA’s biochemical effects on CaMKIIα signalling. After an ischemic insult, CaMKIIα is rapidly activated, resulting in increased autophosphorylation and translocation to the PSD. 51 We thus initially evaluated HOCPCA’s effect on Thr286 autophosphorylation after stroke. As we saw a robust neuroprotective effect of HOCPCA in this model 30 min post-dMCAO (Figure 1(a)), we chose this time point for treatment and harvested peri-infarct tissue 2 h post-stroke (Figure 2(a)), which is reportedly a suitable time point to detect CaMKII biochemical alterations after MCAO.18,51 Surprisingly, we did not observe any effect of stroke or HOCPCA treatment on Thr286 autophosphorylation in homogenates from the peri-infarct cortex (Suppl. Figure 1).
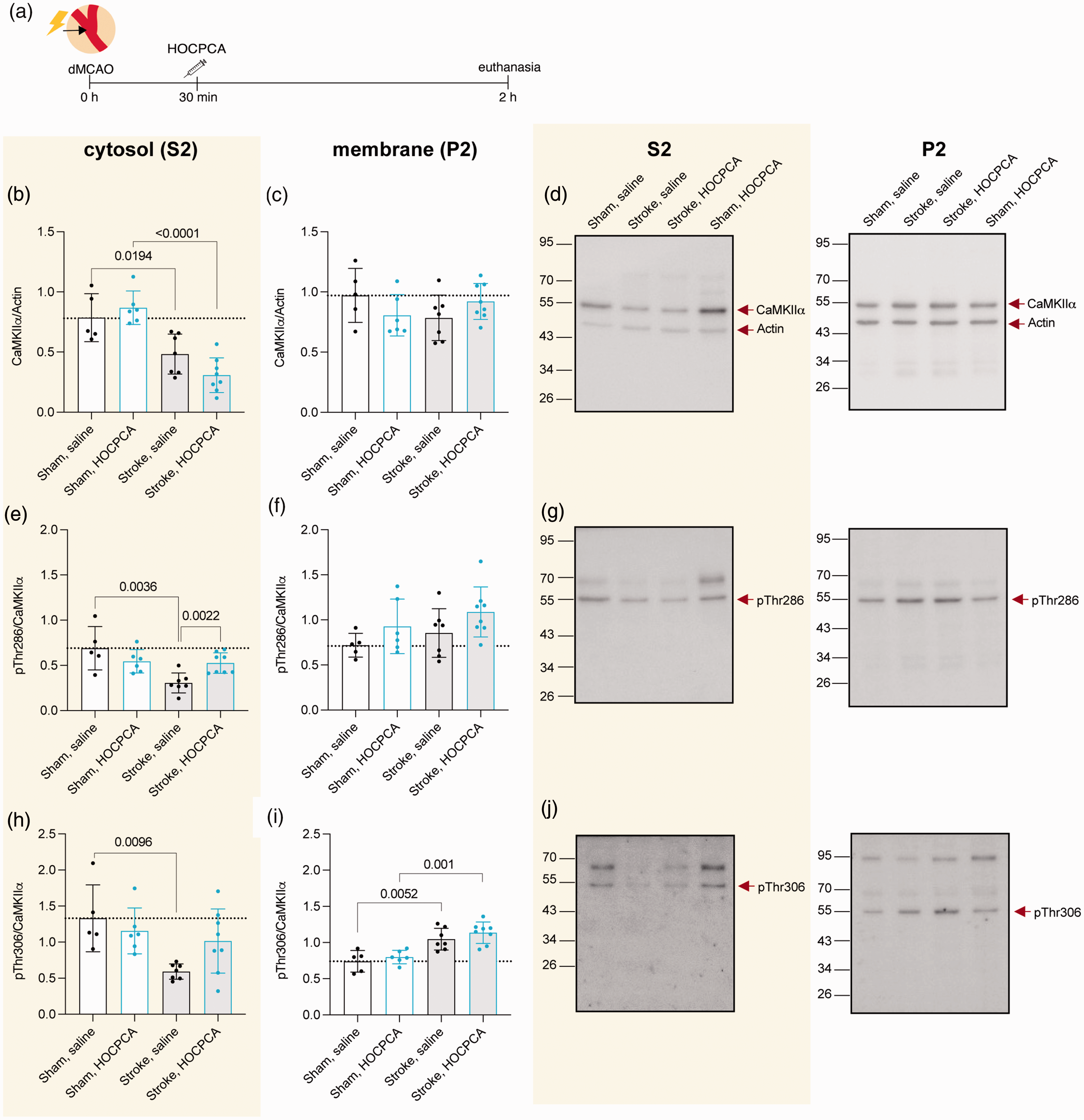
Effect of HOCPCA on CaMKIIα expression and autophosphorylation in subcellular fractions after dMCAO. (a) Schematic illustration of experimental timeline. Adult male mice were subjected to sham (n = 5–6/group) or dMCAO-surgery (n = 7–8/group), treated with 175 mg/kg HOCPCA or saline control, and brains were harvested 2 h post-stroke. (b-j) Membrane (P2) and cytosolic (S2) fractions of homogenates from peri-infarct tissue were probed for total CaMKIIα (b–d), pThr286 (e–g), or pThr306 (h–j) and normalized to Actin or total CaMKIIα expression and (d, g, j) Representative Western blots. (Note: Western blots were performed blinded, and the order of treatment groups differs in bar diagrams for most logical interpretation; Full blots are shown, and three technical repetitions were performed; One-way ANOVA, post hoc Tukey’s test, only significance for the following comparisons is presented in the graphs for simplification: sham, saline versus stroke, saline; sham, HOCPCA versus stroke, HOCPCA, and stroke, saline versus stroke, HOCPCA).
After ischemia-induced translocation to the PSD, CaMKIIα interacts with the NMDA receptor subunit GluN2B. 52 Because HOCPCA reduces this colocalisation in vitro, 8 we investigated the effect on total CaMKIIα expression in both membrane (P2) and cytosolic (S2) subcellular fractions after dMCAO. Consistent with previous reports, 18 we observed that total CaMKIIα expression significantly decreased in the cytosolic S2 fraction after dMCAO compared to sham (p = 0.0194). However, we did not observe any effect of HOCPCA on total CaMKIIα expression in sham or stroke-injured tissue (Figure 2(b)). As CaMKIIα is differently autophosphorylated depending on its subcellular location, 6 we also evaluated its autophosphorylation levels in these subcellular fractions. We observed a decrease in pThr286 after dMCAO in the cytosolic S2 fraction (p = 0.0036). Interestingly, HOCPCA treatment significantly restored Thr286 phosphorylation to sham levels (66% increase, p = 0.002; Figure 2(e)). A similar, albeit smaller non-significant decrease was seen for pThr306 autophosphorylation (p = 0.1329; Figure 2(h)).
No stroke or treatment effect was observed on total CaMKIIα as well as pThr286 expression in the membrane P2 fraction (Figure 2(c) and (f)). However, we observed increased pThr306 levels after dMCAO in the membrane P2 fraction, both in saline- and HOCPCA-treated mice (Figure 2(i)).
HOCPCA does not affect pThr287-CaMKIIβ in the cytosolic fraction after dMCAO
As CaMKIIα assembles with CaMKIIβ into mixed holoenzymes with a 3:1 ratio within forebrain regions, 53 we also evaluated whether HOCPCA binding to CaMKIIα could affect CaMKIIβ activity after dMCAO. Like CaMKIIα, total CaMKIIβ expression was significantly decreased in the cytosolic fraction after dMCAO, and no effect of HOCPCA treatment was observed (Figure 3(a)). However, in contrast to CaMKIIα, the corresponding Thr287 autophosphorylation of CaMKIIβ was neither affected by dMCAO nor HOCPCA treatment (Figure 3(d)). This underlines the alpha subtype selectivity of HOCPCA, also in vivo. Moreover, whereas we did not observe effects of ischemia or treatment on total and pThr286 expression of CaMKIIα in the membrane P2 fraction, dMCAO resulted in an increase in total CaMKIIβ and decrease in pThr287 expression within the P2 fraction (Figure 3(b) and (e)) in the HOCPCA treated samples only.
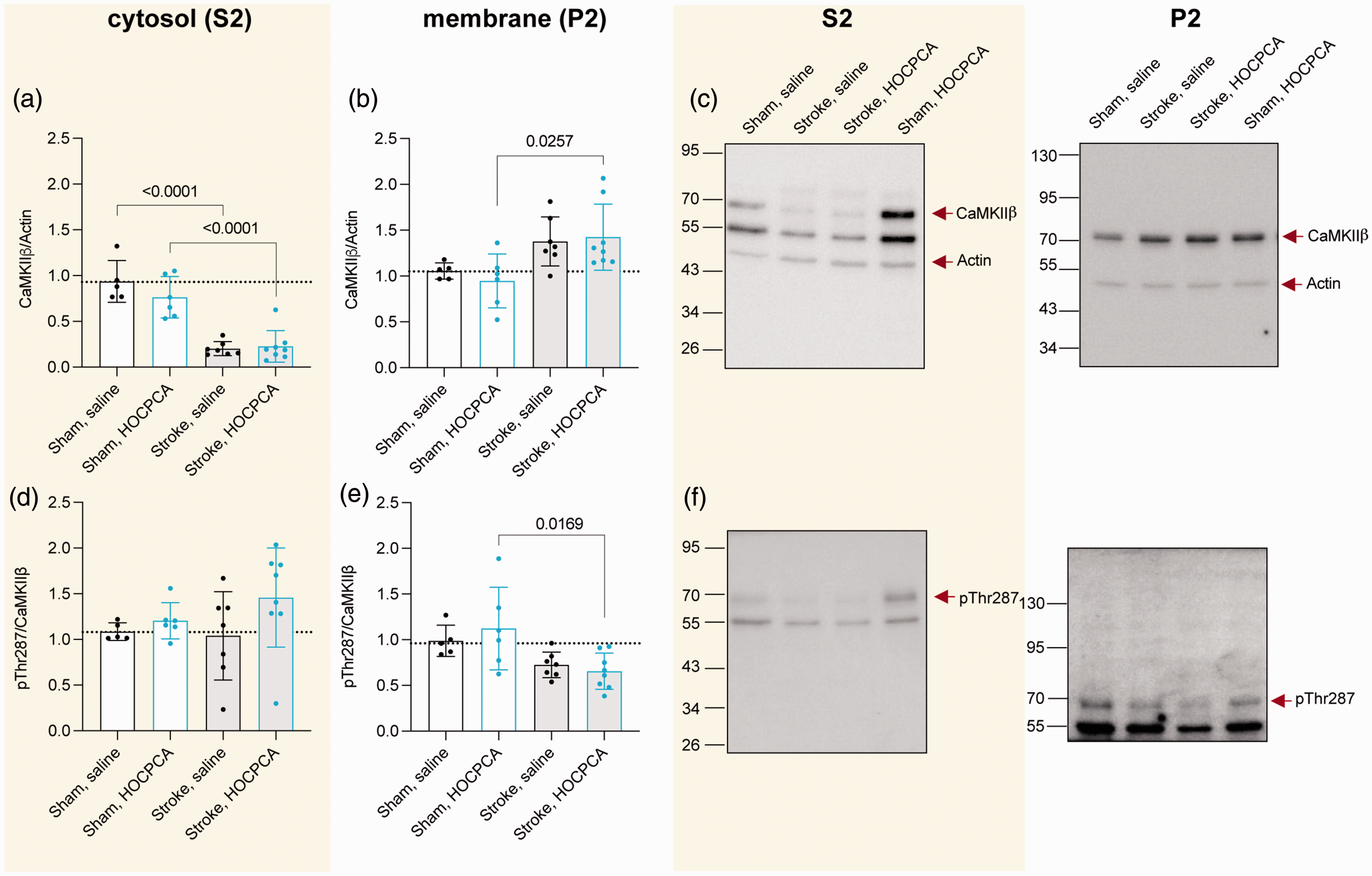
Effect of HOCPCA on CaMKIIβ expression and autophosphorylation in subcellular fractions after dMCAO. (a–f) Membrane (P2) and cytosolic (S2) fractions of homogenates from peri-infarct tissue from sham (n = 5–6/group) or dMCAO-operated mice (n = 7–8/group) were probed for total CaMKIIβ (a–c) and pThr287 (d–f). Expression was normalized to Actin or total CaMKIIβ expression, respectively and (c, f) Representative Western blots. (Note: Western blots were performed blinded, and the order of treatment groups differs in bar diagrams for most logical interpretation; Full blots are shown, and three technical repetitions were performed; One-way ANOVA, post hoc Tukey’s test, only significance for the following comparisons is presented in the graphs for simplification: sham, saline versus stroke, saline; sham, HOCPCA versus stroke, HOCPCA, and stroke, saline versus stroke, HOCPCA). Experimental details are the same as in Figure 2.
HOCPCA downregulates an ischemia-specific cleavage fragment of CaMKII in the membrane fraction
We have previously reported the ischemia-induced cleavage of both Thr286 autophosphorylated CaMKIIα as well as Thr287 autophosphorylated CaMKIIβ by calpain after PTS. 19 Specifically, we observed a 31 kDa-fragment of CaMKII (referred to as ΔCaMKII) after stroke, which corresponds to the kinase domain devoid of its regulatory, linker, and hub domain, rendering the kinase constitutively active. We suggest that this contributes to ischemia-induced CaMKII dysregulation (Figure 4(a)), 19 and that HOCPCA influences CaMKII cleavage after ischemia via a unique molecular interaction with the hub. To investigate this, we initially investigated whether ΔCaMKII was present in the subcellular dMCAO samples. To this end, we probed with a CaMKIIpan antibody, which detects an epitope within the highly conserved kinase domain. Hence, the antibody does not discriminate between CaMKII subtypes, and the ΔCaMKII might be derived from either CaMKIIα or CaMKIIβ. As in the PTS model, 19 we saw a dramatic increase in ΔCaMKII expression in the membrane P2 fraction after dMCAO (230% increase compared to sham; p = 0.006; Figure 4(b)). Remarkably, in the HOCPCA-treated samples this was significantly reduced (48% decrease compared to dMCAO; p = 0.0067), thus indicating a treatment effect on the ischemia-specific expression of ΔCaMKII. We were unable to detect ΔCaMKII in the cytosolic S2 fraction, possibly due to methodological limitations. Similar to pThr286, no effect of stroke or treatment on ΔCaMKII expression was detected in unfractionated samples (Suppl. Figure 1E, F).
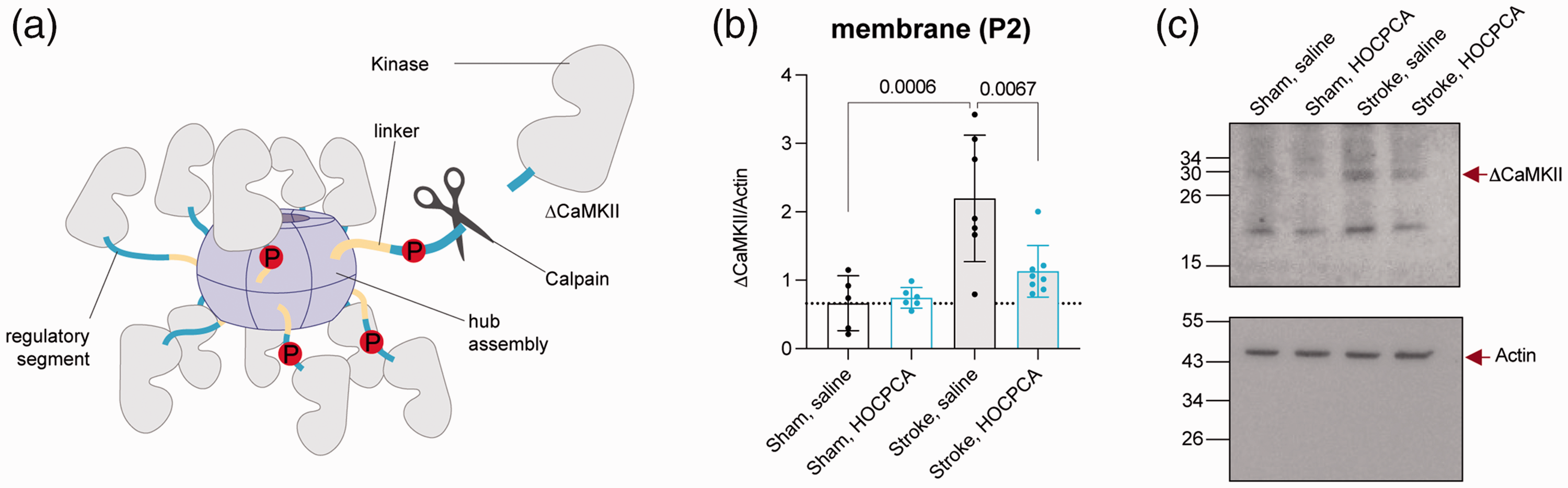
Effect of HOCPCA on CaMKII cleavage after dMCAO. (a) Graphical illustration of CaMKII cleavage after ischemia. (b) A 31 kDa N-terminal fragment of CaMKII (ΔCaMKII) was detected by CaMKIIpan antibody in the P2 membrane fraction after dMCAO (n = 7–8/group) or sham surgery (n = 5–6/group), and expression was normalized to Actin (see Figures 2(c) and 3(b) for total CaMKIIα and CaMKIIβ expression) and (c) Representative Western blots. (Western blots were performed blinded; Full blots are shown, and three technical repetitions were performed; One-way ANOVA, post hoc Tukey’s test, only significance for the following comparisons is presented in the graphs for simplification: sham, saline versus stroke, saline; sham, HOCPCA versus stroke, HOCPCA, and stroke, saline versus stroke, HOCPCA). Experimental details are the same as in Figure 1.
HOCPCA improves motor function after thromboembolic stroke
To extend the clinical translatability, we further assessed HOCPCA’s neuroprotective effect in a mouse model with a reperfusion component, in this case a thromboembolic stroke. Here, in situ injection of thrombin leads to MCAO by the local formation of a fibrin-rich clot, thus enabling the study of neuroprotective agents in combination with tPA-induced reperfusion. 34 The model is uniquely characterised by gradual reperfusion hours after thrombin injection mediated by spontaneous clot dissolution by the endogenous fibrinolytic system,54,55 and thus resembles human pathology more closely compared to classical mechanical transient MCAO where blood flow is reversed promptly and with a much larger lesion. 50
To our knowledge, ischemia-induced CaMKIIα dysregulation has never been addressed in this model. Therefore, we initially examined pThr286 levels as a measure of CaMKIIα dysregulation. A time point of 30 min post-stroke was selected as this represents the desired time for HOCPCA treatment (Figure 5(a)). Compared to sham, we found that pThr286 levels were significantly increased after stroke while total CaMKIIα expression remained unchanged (Figure 5(b)), confirming pathological CaMKIIα activation also in this model. Subsequently, we tested HOCPCA’s neuroprotective effect on infarct size and motor function. To this end, mice received either a single dose of 175 mg/kg HOCPCA at 30 min, intravenous administration of 10 mg/kg tPA starting 20 min post-thrombin injection, saline control, or a combination of HOCPCA and tPA treatment (Figure 5(a)). Infarct volumes were evaluated at 1-day post-stroke using T2-weighted imaging. While tPA treatment significantly reduced infarct size by 39% (23.7 ± 7.2 mm3 for saline versus 14.4 ± 5.8 mm3 for tPA; p = 0.0035), which is similar to previous reports, 37 neither HOCPCA treatment alone nor a combination of tPA and HOCPCA treatment affected infarct size (23.3 ± 7.7 mm3 for HOCPCA and 17.6 ± 7.2 mm3 for HOCPCA + tPA; Figure 5(c)). By contrast, we could detect neuroprotective effects of all treatment regimens in alleviating grip strength asymmetry after thromboembolic stroke. Specifically, HOCPCA, tPA as well as HOCPCA + tPA treatment relieved stroke-induced forelimb asymmetry in grip strength when assessed 3 days post-stroke (Figure 5(d)). Only saline-treated mice showed forelimb grip strength asymmetry (p < 0.0001 on day 1 and p = 0.008 on day 3), similar to previous reports in this model. 36 Moreover, the protective effect of HOCPCA treatment on grip strength was already seen on day 1 post-stroke, whereas this was not the case for tPA (p = 0.0044) or HOCPCA + tPA (p = 0.0056) groups (Figure 5(d)). Finally, to further characterize HOCPCA’s neuroprotective effect, we evaluated if early HOCPCA treatment affects the gene expression of selected inflammatory markers. To this end, we evaluated mRNA expression of the cytokine TNFα and the microglia/macrophage activation markers Iba1 and CD68 in the ischemic cortex 3 days after both thromboembolic stroke and dMCAO. HOCPCA treatment was able to reduce mRNA expression of inflammatory markers compared to saline in both models. Similar effects were seen for tPA and tPA + HOCPCA treatment after thromboembolic stroke (Suppl. Figure 2).
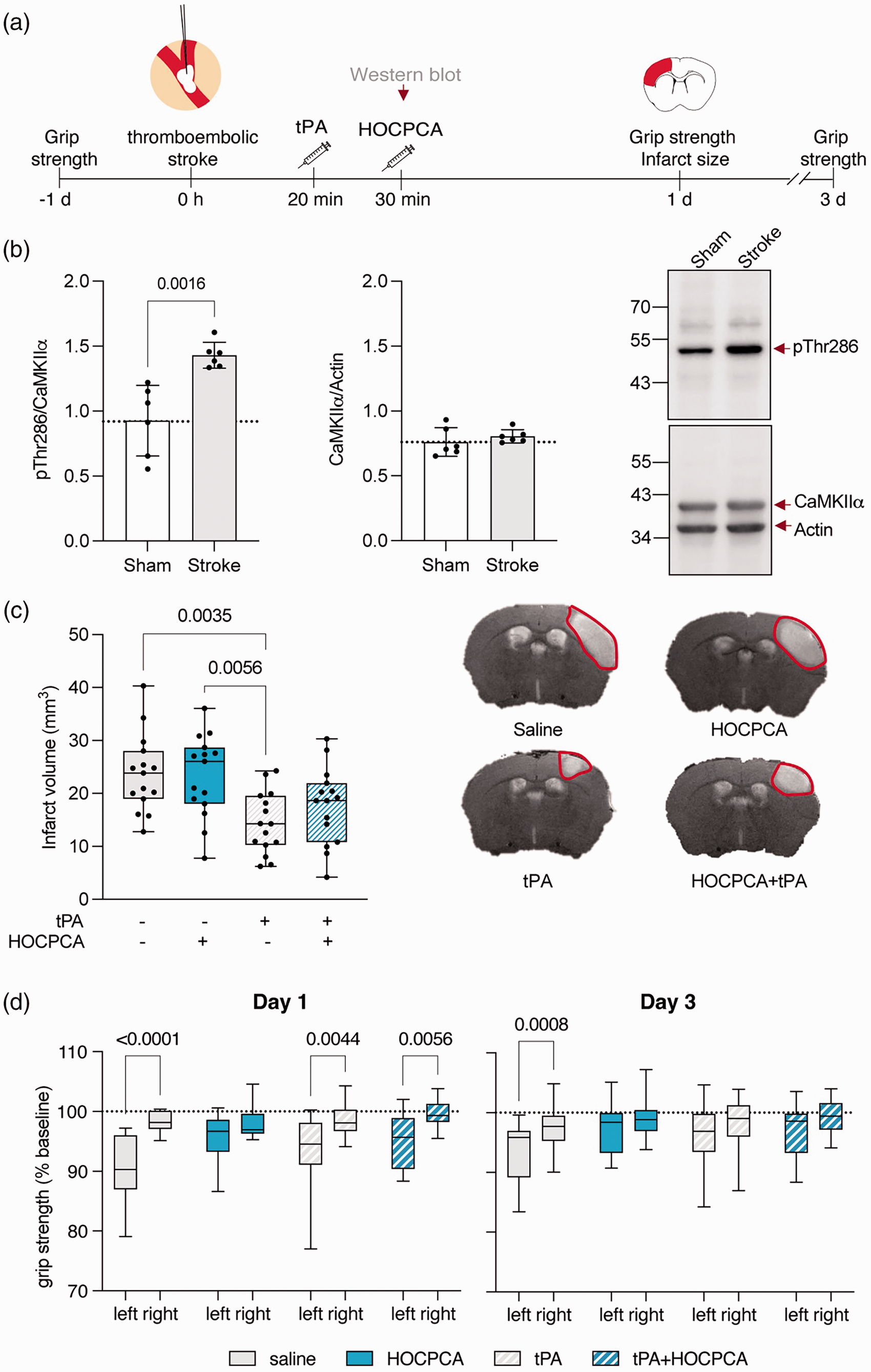
Validation of HOCPCA’s neuroprotective effect using a thromboembolic stroke model with reperfusion component. (a) Schematic illustration of experimental timeline. (b) CaMKIIα pThr286 autophosphorylation and total CaMKIIα expression 30 min post-thromboembolic stroke compared to sham (n = 6/group). Expression was normalized to total CaMKIIα or Actin, respectively. Representative Western blots are shown to the right. (c, d) Adult male mice were treated either with saline, tPA (intravenous, 10 mg/kg, 10% bolus/90% infusion over 40 minutes starting from 20 min after occlusion), HOCPCA (intraperitoneal, at 30 min post-stroke) or a combination of tPA and HOCPCA (n = 15/group). (c) Infarct size was assessed by T2-weighted imaging 1-day post-stroke (One-way ANONA, post hoc Tukey’s test). (Right) Representative by T2-weighted images. (d) Grip strength asymmetry was evaluated (left) at 1 day and (right) 3 days post-stroke and is shown as % of baseline grip strength for the left (contralateral) and right (ipsilateral) front paw (two-way repeated measures ANOVA (front paw, treatment), post hoc Šídák’s test) (Box plot (boxes, 25–75%; whiskers, minimum and maximum; lines, median)).
HOCPCA-mediated improvements in sensorimotor function post-dMCAO are sustained
To evaluate the functional effects of HOCPCA, we investigated whether a single dose of HOCPCA (175 mg/kg) at 3 h can also improve sensorimotor function one and twoweeks post-dMCAO (Figure 6(a)). Quantification of brain tissue loss 14 days post-dMCAO showed no difference between the saline-treated (9.58 ± 2.78 mm3) and HOCPCA-treated mice (8.43 ± 2.31 mm3, p = 0.6607; Figure 6(b) and (c)). To assess forepaw impairments and asymmetries in somatosensory function, we used the bilateral tactile stimulation test. A small adhesive patch was stuck to the plantar surface of each forepaw and the time taken to contact with their mouth was recorded (Figure 6(d)). Sham animals rapidly contacted and removed both stimuli. Stroke mice showed impairment in sensing the adhesive patch one-week post-dMCAO (p < 0.001 compared to sham controls), however no difference between stroke and sham animals was observed by two-weeks post-stroke in this task (Figure 6(d)). Treatment with HOCPCA facilitated a significant improvement in the time to sense the patch as shown by a decrease in time to touch one-week post-dMCAO (p < 0.01) compared to the saline-treated control dMCAO group (Figure 6(d)).
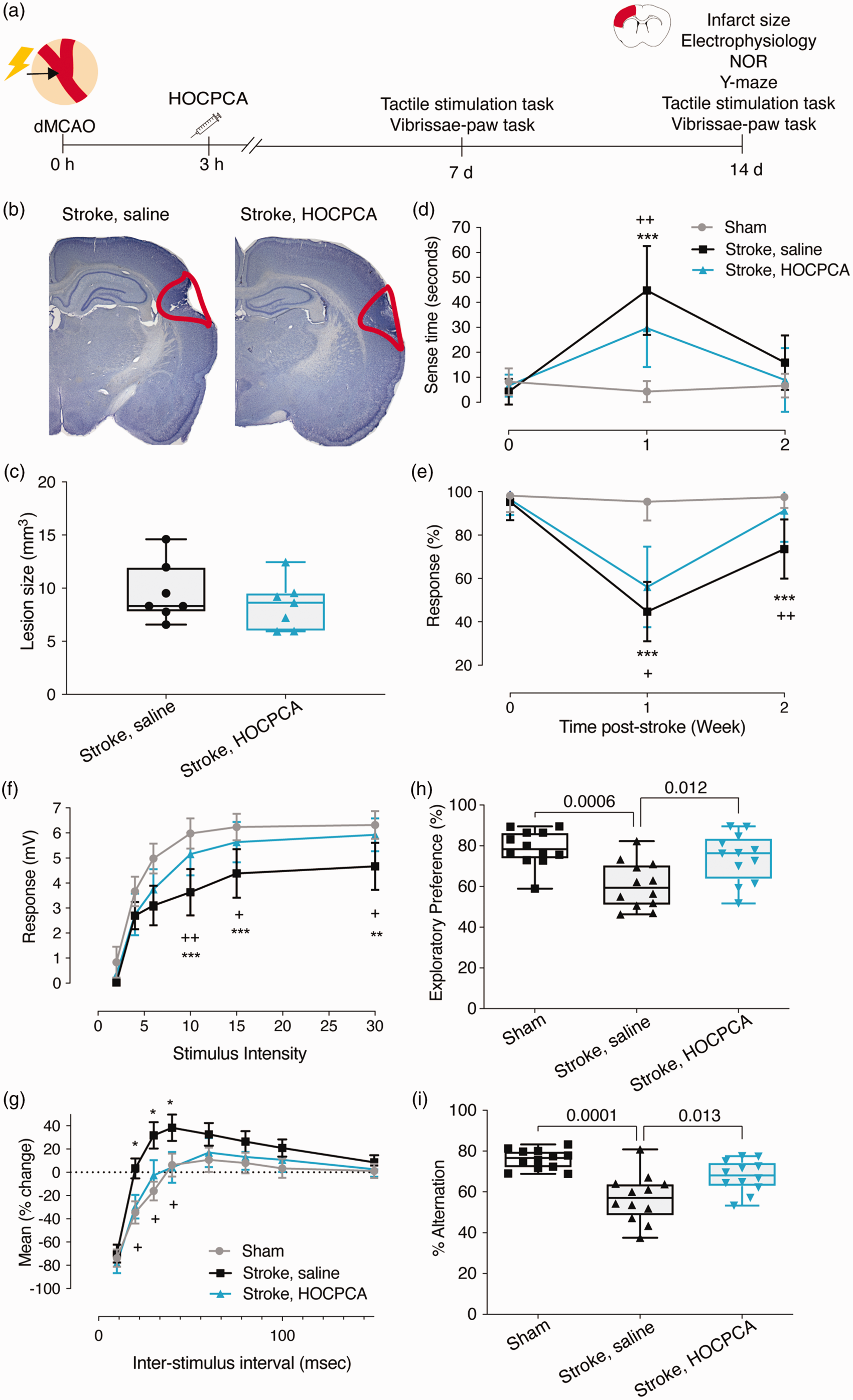
HOCPCA improves sensorimotor and cognitive function after dMCAO stroke. (a) Schematic illustration of experimental timeline. Adult male mice were treated 3 h after dMCAO with 175 mg/kg HOCPCA or saline control (intraperitoneal). (b, c) Brain tissue loss was assessed 14-days post-stroke (n = 7/group). (b) Representative cresyl violet stained tissue sections, and (c) quantification of brain tissue loss. (d, e) Assessment of sensorimotor function was performed on (d) the tactile stimulation task and (e) the vibrissae-paw task both 7- and 14-days post-stroke (n = 12; two-way ANOVA (time, treatment), post hoc Tukey’s test). (f, g) Evaluation of hippocampal neuronal activity. Input/output functions from region CA1 from sham controls (grey) or mice subjected to dMCAO-surgery, treated with saline vehicle (black) or HOCPCA (blue) when assessed 14-days post-stroke. (f) Population spike amplitudes (mV) versus stimulus intensity at 14-days post-stroke. (g) Field EPSP slopes (mV/ms) versus stimulus intensity at 14-days post-stroke (n = 5/group; two-way ANOVA (stimulus strength, treatment), post hoc Tukey’s test) and (h, i) Assessment of cognitive function on the (h) NOR and (i) y-maze tests 14-days post-stroke (n = 12/group; one-way ANOVA, post hoc Dunnett’s test). (Data is expressed as mean ± SD, except the cognitive tests and infarct size, which are expressed as box plots (boxes, 25–75%; whiskers, minimum and maximum; lines, median). *p < 0.05, **p < 0.01, ***p < 0.001 = Stroke, saline versus sham; +p < 0.05, ++p < 0.01, +++p < 0.001 = Stroke, HOCPCA versus Stroke, saline).
We further investigated dMCAO-induced impairments in sensorimotor function on the vibrissae test, which addresses the animal’s forelimb reflex following whisker stimulation. Sham control animals showed no impairment on the vibrissae test (Figure 6(e)). Stroke mice showed impairment in response to vibrissae stimulation one-week post-dMCAO (p < 0.001 compared to sham controls). Although stroke animals show rapid (spontaneous) recovery, significant impairment was still observed two-weeks post-dMCAO (p < 0.001 compared to sham controls; Figure 6(e)). Treatment with HOCPCA showed a significant improvement in response to vibrissae stimulation one- and two-weeks post-dMCAO (p < 0.05 and p < 0.01 respectively) compared to saline-treated stroke controls (Figure 6(e)).
HOCPCA improves hippocampal neuronal activity after dMCAO stroke
Previous studies have reported altered hippocampal function following dMCAO. 56 Interestingly, the impairments in hippocampal cognitive function reported by Doyle and colleagues appear to occur at a delay and are linked to increased inflammation. Further, longitudinal MRI assessments have also revealed changes in hippocampal volume that correlate to cognitive impairments in humans. 57 To substantiate this in our model, we initially verified an increase in inflammatory response two weeks after dMCAO by showing that the expression of the microglia marker IBA1 is significantly increased in the CA1 region of the hippocampus compared to sham controls (p = 0.003, Suppl. Figure 3). Treatment with HOCPCA at 3 h post-dMCAO decreased the expression of IBA1 compared to saline-treated mice (p = 0.050, Suppl. Figure 3), whereas HOCPCA treatment did not affect reactive astrogliosis assessed by GFAP labelling (data not shown). Next, we aimed to assess if hippocampal neuronal excitability was altered two-weeks following dMCAO and if treatment with HOCPCA at 3 h post-dMCAO could influence these potential alterations (Figure 6(f) and (g)). Assessment of hippocampal function after dMCAO in saline-treated animals revealed impaired neuronal excitability two-week post-dMCAO across a range of stimulus intensities (maximal population spike amplitude: 4.67 ± 0.95 mV) compared to sham controls (6.32 ± 0.55 mV, p < 0.001; Figure 6(f)). Treatment with HOCPCA resulted in a near complete functional restoration in evoked population spike responses differing after dMCAO only slightly from sham controls (HOCPCA: 5.93 ± 0.66 mV, p < 0.001; Figure 6(f)).
Moreover, local circuit inhibition in CA1 was assessed by paired-pulse analysis after stimulation of Schaffer collateral-commissural fibers. In sham control animals, slices exhibited strong inhibition (∼70%) at the shortest inter-stimulus intervals (ISIs) with a return to baseline population spike amplitudes by ∼40 ms ISIs and little paired-pulse facilitation observed thereafter (Figure 6(g)). Assessment of local circuit inhibition (paired-pulse facilitation) two-weeks post-dMCAO revealed a significant treatment effect across treatment groups (Figure 6(g)). As such, in the saline-treated dMCAO group, a robust increase in paired-pulse facilitation was observed between ISIs 10–60 ms compared to sham controls. Treatment with HOCPCA exhibited enhanced inhibition (i.e., reduced population spike amplitudes) across the full range of ISIs relative to saline-treated dMCAO animals, with significances observed between ISIs at 20–40 ms (Figure 6(g)).
HOCPCA improves memory performance following dMCAO stroke
Having shown deficits in hippocampal excitability and circuit inhibition, we next assessed if cognitive performance was also impaired at two-weeks post-dMCAO. First, we used the NOR task measuring recognition memory, which takes advantage of the innate preference of mice for exploring a novel object rather than a familiar one. Saline-treated stroke mice (60.71 ± 11.45%, p = 0.0006) spent significantly less time investigating the novel object after dMCAO, relative to sham controls (79.05 ± 8.77%; Figure 6(h)). Assessment of HOCPCA-treated mice after dMCAO revealed that mice spent more time interacting with the novel object (74.18 ± 11.9%, p = 0.012) compared to the saline-treated dMCAO control group. No difference between sham and HOCPCA-treated dMCAO mice was observed (p = 0.517; Figure 6(h)).
To further assess cognitive function, we also tested mice on the alternating y-maze test to evaluate spatial memory. This test relies on the innate preference of mice to explore a novel environment and measures their ability to remember which arm of the y-maze was not previously explored. Similar to the NOR test, we observed that saline-treated mice after dMCAO investigated the novel arm less often (57.08 ± 11.68%; p = 0.0001) in comparison to sham controls (77.99 ± 4.78%; Figure 6(i)). Assessment of HOCPCA-treated stroke mice (67.67 ± 7.72%, p = 0.013) revealed that mice investigated the novel arm more often compared to the saline-treated dMCAO group. No difference between sham and HOCPCA-treated dMCAO mice was observed (p = 0.058).
Discussion
CaMKIIα is a major mediator of physiological and pathological Ca2+ signals regulating numerous downstream pathways with possible roles in cell death or survival (reviewed in 20 ). Moreover, ischemia leads to CaMKII dysregulation in various ways. For instance, subsets of CaMKII with increased autonomous activity, elevated levels of multiple autophosphorylation sites, PSD translocation as well as calpain-mediated cleavage into a stable constitutively active kinase fragment have been described.6,18,58 Yet, the exact interplay and which mechanism constitutes the best pharmaceutical target is still to be fully elucidated and warrants much needed further studies.
HOCPCA treatment is neuroprotective and reverses ischemia-induced CaMKII dysregulation
In this study, we establish pharmacological modulation of the CaMKIIα hub domain by GHB ligands as an attractive strategy to reduce CaMKII dysregulation after stroke in mice. First, we show that the GHB analogue, HOCPCA, improves motor function in two clinically relevant murine stroke models and is able to dampen inflammatory changes. Second, we present unforeseen and differential effects of HOCPCA treatment on distinct subsets of ischemia-dysregulated CaMKII pools. And thirdly, we show improved sensorimotor and cognitive function linked to altered hippocampal neuronal activity secondary to dMCAO. Yet, elucidating the underlying molecular mechanisms mediated by ligand binding to the hub domain requires further studies.
HOCPCA reversed an ischemia-induced decrease in cytosolic Thr286 autophosphorylation. Ischemia-induced translocation of the cytosolic CaMKIIα has been previously reported to coincide with a further decrease in cytosolic pThr286. 6 Yet previous studies focused on the effects of CaMKII dysregulation at the PSD, where increased pThr286 as well as autonomous activity were shown to be associated with death promoting CaMKII effects. 6 Collectively, this might indicate that the previously described dual role of CaMKIIα during ischemia is differentiated by pThr286 levels in distinct subcellular locations, i.e., membrane-anchored pThr286 CaMKIIα might promote cell death, whereas normalization of pThr286 levels within the cytosolic compartment might instead lead to neuroprotection.
Moreover, we show that HOCPCA reduces ΔCaMKII expression levels in the membrane fraction, which constitutes an ischemia-specific stable fragment of CaMKII. 19 Strikingly, we recently documented that calpain mediates interdomain cleavage of Thr286 autophosphorylated CaMKIIα right before the regulatory segment, thus releasing a kinase domain fragment (ΔCaMKII) that is by nature constitutively active. 19 In fact, ΔCaMKII has been widely used as a tool to study the impact of CaMKII activity,59–62 yet, its natural occurrence after dMCAO has never been reported and highlights it as a potential pathospecific target in excitotoxicity. Interestingly, we detected ΔCaMKII only in the membrane fraction and not in the cytosol, supporting a membrane-associated death-promoting role for CaMKIIα. Of note, the binding site for GluN2B subunit of the NMDA receptor is mapped to the kinase domain of CaMKIIα, 14 thus suggesting that membrane anchoring of ΔCaMKII is still feasible. Mechanistically, calpain-mediated cleavage of pThr286-CaMKIIα might act as a downstream neurotoxic effect of the sustained ischemia-induced increase in pThr286.6,51 As such, unregulated kinase activity of ΔCaMKII is likely contributing to excessive substrate phosphorylation after ischemia, resulting in ischemic cell death. Most interestingly, HOCPCA treatment reduced the expression of ΔCaMKII after stroke, suggesting an ischemia-specific compound effect that might be involved in the reversal of CaMKII dysregulation. Of note, even though calpain inhibition has been shown to be neuroprotective, 63 the exact role of ΔCaMKII in neuronal cell death as well as the mechanism of HOCPCA-mediated reduction in ΔCaMKII remains to be determined. Still, it offers an attractive new way of salvaging tissue damage after stroke.
HOCPCA acts selectively and specifically under ischemic conditions
HOCPCA is part of a new class of CaMKIIα ligands (GHB analogues) with unparalleled selectivity for the hub domain of the alpha subtype. While ‘classical’ CaMKII inhibitors such as KN93 and CN-peptides, not only show off-target effects, they also inhibit all four CaMKII subtypes.64,65 This increases the risk for unwanted side effects as CaMKII subtypes are expressed throughout all tissues. In fact, CaMKIIδ plays a major role in cardiomyocyte signalling, however, emerging evidence is also showing a role for CaMKIIδ in regulating astrocyte and neuronal cross-communication and cell survival after ischemia.66–68 Moreover, the use of CaMKII inhibitors might be hampered due to possible effects on learning and memory. As such, tatCN21 interfered with learning ability in mice by inhibiting LTP induction. 69 In contrast, HOCPCA did not affect LTP, and most intriguingly, we did not observe effects of HOCPCA on CaMKIIα during naïve nonpathological conditions. 8 Accordingly, HOCPCA was only neuroprotective when applied after a glutamate insult in primary cortical neurons, thus indicating the need for CaMKII dysregulation for effective hub modulation. 8 Interestingly, we did however see a reversal in the ischemia-induced suppression in hippocampal neuronal activity and improved working memory following dMCAO. Most likely, this reversal in neuronal activity occurred via the suppression of tissue inflammatory markers that are known to impair neuronal activity and both sensorimotor and cognition functions. 56 In addition, HOCPCA selectively normalized cytosolic pThr286-CaMKIIα after stroke, while no treatment effect was seen in the membrane fraction nor in naïve mice or on pThr287-CaMKIIβ, supporting its selectivity and pathospecific action in vivo. The observed effect on pThr286 is in contrast to our previous finding that HOCPCA did not affect pThr286 in basal or Ca2+-stimulated cortical primary neurons. 8 Yet, a potential effect of HOCPCA on pThr286 after Ca2+-stimulation was likely masked by studying whole cell lysates instead of subcellular fractions, similarly we did not observe any effect of HOCPCA on pThr286 in whole-cell homogenates in this study.
Mechanistic thoughts behind HOCPCAs effects in peri-infarct subcellular compartments
At this point, it is unclear how pharmacological modulation of the hub domain results in the normalization of cytosolic pThr286 levels, yet an allosteric effect via the hub domain is envisaged. In fact, recent reports highlight the emerging functional importance of the hub domain. For instance, a role in allosteric modulation of kinase activity 70 as well as in activation-triggered destabilization of holoenzymes and subsequent dimer release and subunit exchange have been suggested.71–73 Interestingly, we previously showed that HOCPCA is able to stabilize CaMKIIα hub oligomerization in vitro in a thermal shift assay. 8 Although a causal mechanistic link is absent, it is tempting to speculate that the ischemia-induced restorative effect of HOCPCA on pThr286 levels within the cytosolic fraction could be mediated by increased holoenzyme stability possibly associated with a reduction in activation-triggered subunit release. Specifically, as pThr286 occurs in trans between neighbouring activated subunits, 74 stabilization of the oligomeric state of the holoenzyme could be apparent in increased Thr286 autophosphorylation. Yet, other mechanisms could also be involved, such as differentially regulated phosphatase activity. 75
Moreover, we did not observe increased pThr286 or total CaMKIIα expression, nor a treatment effect of HOCPCA in the P2 membrane fraction 2 h after dMCAO in this study. This is in contrast to our previous finding that HOCPCA inhibited glutamate-induced CaMKIIα-GluN2B colocalization in primary hippocampal neurons 8 and to several groups reporting sustained ischemia-induced CaMKII translocation and autophosphorylation at the PSD.6,18,51 Yet, the P2 fractions generated in this study include the enrichment of total cell membranes. PSD or synaptic/extrasynaptic fractions might have been better suited for the accurate detection of CaMKIIα translocation. Nonetheless, the discrepancy could also be explained by inherent differences in tissue and/or injury-specific CaMKIIα responses. As such, dysregulated CaMKII translocation and autophosphorylation are well characterized in hippocampal tissue after global ischemia6,76–80 and cortical tissue after transient MCAO.18,51,81 CaMKIIα’s subcellular location after dMCAO might involve differential kinetics compared to previous studies, including injury with a reperfusion component. This is supported by data from Skelding et al., who studied the timing of pThr286 after transient MCAO before and after reperfusion. 82 Skelding and colleagues observed an initial increase in pThr286, which was downregulated at 90 min after MCAO, and then subsequently after reperfusion, a second Ca2+ wave triggered another increase in pTh286, indicating a pattern of phosphorylation and dephosphorylation depending on intracellular Ca2+ levels. 82 Intriguingly, the same study did not detect an effect of ischemia on pThr305/306 in homogenates, whereas our results indicated an ischemia-induced increase in pThr305/306 in the P2 membrane fraction. As pThr305/306 is located within the CaM binding element, this could indicate dissociation of Ca2+/CaM from CaMKIIα, which may further support the hypothesis that 2 h after dMCAO the initial Ca2+ wave is ceased. Nonetheless, CaMKIIα dysregulation might still be sustained at this time point, and yet manifested in a way other than pThr286 activation. This is supported by the fact that HOCPCA treatment 3 h post-dMCAO could still improve functional outcome.
HOCPCA is neuroprotective in multiple in vivo stroke models
Until this point, we have observed neuroprotective effects of HOCPCA in three different animal models of stroke performed in separate laboratories (this study and Leurs et al.). 8 Not surprisingly, the therapeutic window and effect size was considerably different between models, which might be explained by their different aetiology.49,83 Of note, HOCPCA was able to improve motor function after thromboembolic stroke in combination with tPA, suggesting no negative interaction of co-administration as observed in the ESCAPE-NA1 trial.4,5
Nonetheless, HOCPCA treatment 30 min post-stroke was less effective after thromboembolic stroke compared to dMCAO. Interestingly, similar to human pathology, this model is characterized by spontaneous reperfusion.54,55,84 As shown by Skelding et al., 82 reperfusion could lead to a second Ca2+ wave and CaMKIIα dysregulation, possibly further aggravating the injury. As HOCPCA is endowed with a short half-life of 20 min, 27 its effect might have been dampened by secondary injury or CaMKII activation caused by reperfusion. Consequently, it would be interesting to see if a second dose after reperfusion could lead to effects on infarct size in that model. Nonetheless, the reduced effect of HOCPCA in the thromboembolic model compared to dMCAO could also be caused by differences in anaesthesia type and length (inhalation versus general anaesthesia). As such, the anaesthetics might differently regulate the blood pressure, the cerebral blood flow or exert neuroprotective properties which in itself could affect the neuroprotective outcome. 49 Finally, HOCPCA’s ability to dampen inflammatory changes substantiates its effects on functional outcome after stroke, yet the underlying mechanisms require further investigation. These findings are of significant interest, as secondary degeneration has been observed in brain regions, including the hippocampus, that are distinct from but connected to the lesion site in humans, 57 and therefore targeting the CaMKII hub domain could offer hope for treating humans in the future.
Conclusion
In this study, we present pharmacological modulation of the CaMKIIα hub domain by HOCPCA as a strong therapeutic candidate for ischemic stroke by improving functional outcome in multiple stroke models via a novel mechanism-of-action. HOCPCA treatment leads to differential effects on distinct subcellular pools of CaMKIIα, thereby alleviating ischemia-induced CaMKII dysregulation, i.e. normalizing pThr286 levels in the cytosol and downregulation of ΔCaMKII in the membrane fraction. Even though previous studies suggest holoenzyme stabilization as a potential underlying mechanism, further studies are needed to establish a causal link and to decipher downstream functional consequences. Yet, HOCPCA’s high degree of selectivity, pathospecific mechanism as well as the neuroprotective effect in multiple in vivo mouse models shows that the brain-penetrable small-molecule shows great potential. Further understanding of both temporal and mechanistic CaMKII dysregulation after different ischemic insults will direct future pharmacological treatment regimes.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231167920 - Supplemental material for The GHB analogue HOCPCA improves deficits in cognition and sensorimotor function after MCAO via CaMKIIα
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231167920 for The GHB analogue HOCPCA improves deficits in cognition and sensorimotor function after MCAO via CaMKIIα by Nane Griem-Krey, Anders B Klein, Bettina H Clausen, Mathias RJ Namini, Pernille V Nielsen, Mozammel Bhuiyan, Raghavendra Y Nagaraja, T Michael De Silva, Christopher G Sobey, Heung-Chin Cheng, Cyrille Orset, Denis Vivien, Kate L Lambertsen, Andrew N Clarkson and Petrine Wellendorph in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X231167920 - Supplemental material for The GHB analogue HOCPCA improves deficits in cognition and sensorimotor function after MCAO via CaMKIIα
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X231167920 for The GHB analogue HOCPCA improves deficits in cognition and sensorimotor function after MCAO via CaMKIIα by Nane Griem-Krey, Anders B Klein, Bettina H Clausen, Mathias RJ Namini, Pernille V Nielsen, Mozammel Bhuiyan, Raghavendra Y Nagaraja, T Michael De Silva, Christopher G Sobey, Heung-Chin Cheng, Cyrille Orset, Denis Vivien, Kate L Lambertsen, Andrew N Clarkson and Petrine Wellendorph in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Lundbeck Foundation (R277-2018-260) and the Novo Nordisk Foundation (NNF14CC0001). The Emil Herborg Grant and the Julie von Müllen Foundation supported the work with travel grants.
Acknowledgements
We thank Bente Frølund for providing HOCPCA. A special thanks to Florent Lebrun for his help with the thromboembolic stroke surgery. We thank Diana Wichmann Graff, Monica Santiveri-Saez and Carina Bachmann for their help with stroke experiments.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The University of Copenhagen and Otago Innovation Ltd. have licensed patent rights (EP 3 746 064) for the use of HOCPCA in acute brain injury to Ceremedy Ltd., of which P.W. is a co-founder.
Authors’ contributions
NGK, ABK, BHC, CO, DV, ANC, KLL and PW designed the studies. NGK, ABK, BHC, PVN and KLL performed and analysed the 3-day survival dMCAO in vivo studies, and BHC performed grip strength test. NGK and CO performed and analysed thromboembolic stroke and grip strength studies. RYN, TMDS, CGS, MB and ANC performed and analysed the 2-week survival dMCAO in vivo and ex vivo studies. NGK and MRJN performed and analysed subcellular fractions, Western blot and qPCR studies. HCC conceptualised and assisted with CaMKII cleavage studies. The manuscript was written by NGK and revised by PW. All authors reviewed, edited, and approved the final version of the manuscript.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
