Abstract
Febrile status epilepticus (FSE) is an important risk factor for temporal lobe epilepsy and early identification of those at high risk for epilepsy is vital. In a rat model of FSE, we identified an acute (2 hrs) novel MRI signal where reduced T2 relaxation values in the basolateral amygdala (BLA) predicted epilepsy in adulthood; this T2 signal remains incompletely understood and we hypothesized that it may be influenced by vascular topology. Experimental FSE induced in rat pups reduced blood vessel density of the cortical vasculature in a lateralized manner at 2 hrs post FSE. Middle cerebral artery (MCA) exhibited abnormal topology in FSE pups but not in controls. In the BLA, significant vessel junction reductions and decreased vessel diameter were observed, together with a strong trend for reduced vessel length. Perfusion weighted MRI (PWI) was acutely increased cerebral blood flow (CBF) in cortex, amygdala and hippocampus of FSE pups that correlated to decreased T2 relaxation values compared to controls. This is consistent with increased levels of deoxyhemoglobin associated with increased metabolic demand. In summary, FSE acutely modifies vascular topological and CBF in cortex and BLA that may underlie acute MRI signal changes that predict progression to future epilepsy.
Keywords
Introduction
Febrile seizures are the most common types of seizure in human infants and young children. 1 Although most febrile seizures are generally short and have no long-term adverse effects, seizures lasting more than 30 minutes are categorized as febrile status epilepticus (FSE) and are important risk factors for developing temporal lobe epilepsy (TLE) in adulthood.2,3 FSE is a significant risk factor for epilepsy later in life, 4 and many adults with TLE report a history of prior prolonged febrile seizures during childhood.5,6 A rodent model of FSE has been established and is being used to characterize the consequences of experimental FSE. 7 This paradigm uses immature rats at a brain development age equivalent to that of human infants and young children and induces FSE by hyperthermia that involves endogenous fever mediators.8,9 Like some reports in children, 30–40% of rats develop TLE in adulthood. 9
A predictive marker for epileptogenesis that could be applied in the clinic is critically needed to dichotomize those who are at risk for future epilepsy. In a step towards such a biomarker, we recently identified a novel MRI signal (reduced T2/T2* signal) in the basolateral amygdala (BLA) and hippocampus 2 hours following resolution of FSE. This MRI signal, which was confined to one side (lateralized) was observed in ∼40% of rat pups, 10 and was highly predictive of which rats developed epilepsy in adulthood.10,11 The observed reduction in T2 relaxation times was a result of paramagnetic susceptibility effects, that correlated strongly with increased deoxygenated hemoglobin. This MRI signal, denoting locally augmented deoxygenated hemoglobin might derive from increased brain tissue oxygen utilization or reduced blood supply acutely after FSE. The latter may be a result of abnormalities in blood vessels, including their vasoconstriction. Because the MRI signal was no longer present at 24–48 hours, it was considered unlikely that persistent tissue or blood vessel injury was a contributing cause. Cerebral blood flow (CBF) measures have not been performed acutely after FSE in animal models nor in human studies but studies at later time points report divergent findings.12,13 Clearly, additional studies are necessary to investigate how cerebral perfusion is acutely altered after FSE.
The motivation of the current study was to identify putative mechanisms contributing to the acute reduced T2/T2* signal immediately post FSE. 10 Given that one mechanism for the altered T2/T2* signaling could be a mismatch between metabolic demand and vascular delivery, we set out to examine the vascular topology and angioarchitecture immediately after the cessation of SE in postnatal day (P)10 rat pups. We utilized our vessel painting approach 14 that directly labels the lumen of arteries and veins and thereby facilitates whole brain examination of the vascular network. In addition, we acutely examined CBF in FSE rats to assay the functional consequences of an altered vascular topology. We hypothesized that the vascular lumen would be reduced in vulnerable regions such as the BLA. Our experiments identified region-specific alterations in vascular topology and lateralization within 2 hrs after FSE cessation, which may contribute to altered cerebral perfusion and potentially set the stage for future transition to epilepsy.
Materials and methods
Animals
All animal procedures were approved by the University of California Irvine animal care committee and performed according to NIH and ARRIVE guidelines. Sprague Dawley dams (Envigo, Livermore, California) were maintained under controlled temperatures and light-dark cycles. FSE pups and their littermate normothermic controls were generated as detailed below. For technical reasons, several cohorts were used, and each cohort consisted of rat pups interleaved from different litters to minimize batch/litter effects. Within 90 minutes following FSE cessation, rat pups were removed from the dam, and all were sacrificed at the 2-hour time point.
Experimental febrile status epilepticus induction
Experimental febrile status epilepticus (FSE) was induced as previously described7,11 and pups were randomly assigned to control and FSE groups. Briefly, Sprague Dawley male and female rat pups at P10 or 11 were placed in pairs into a 3.0 litter glass container lined with absorbent paper. All pups were healthy and weighted 19.4–24.6 grams prior to starting FSE. Hyperthermia was induced by a continuous stream of warm air (38.5–40.5°C for 60 minutes) to induce behavioral seizures. Core temperatures were measured at baseline, seizure onset, and every 2 minutes during hyperthermia. Following FSE, rats cooled on a chilled metal plate until their core temperature was reduced to the normal range of age. Pups were allowed to recover on a euthermic pad for 15 minutes before returning to their home cage. All pups subjected to hyperthermia experienced FSE and there were no FSE-related deaths. For normothermic controls, pups were removed from the cage for 60 minutes (to control for potential stress) and their core temperatures kept within normal range of age (34–36°C). The vascular topology study comprised of total of n = 18 normothermic and n = 14 FSE pups. A separate cohort of FSE and control pups (n = 4/group) were generated for perfusion weighted imaging assessments (see details below).
Vessel painting
Vessel painting (VP) was performed as previously described. 14 Briefly, rat pups were anesthetized with an intraperitoneal injection of ketamine (90 mg/kg) and xylazine (10 mg/kg). The chest cavity was opened and Dil solution (0.45 mg/ml in PBS with 4% dextrose) was manually injected into the left ventricle. Rat pups were then perfused with 10 ml of phosphate buffered saline (PBS) and 20 ml of 4% paraformaldehyde (PFA) at a flow rate of 7.8 ml/min using a peristaltic pump After fixation, brains were extracted from the cranium and the dura mater was carefully removed. Brains were post-fixed in 4% PFA for 24 hours and stored in PBS until microscopic imaging. In the vascular topology study 11/14 FSE and 12/18 control pups had successful unilateral or bilateral staining of the hemispheres.
Wide-field fluorescent and confocal microscopy acquisition
Acquisition of wide-field cortical (axial) images were obtained by positioning the brain between two Superfrost Plus microscope slides (Fisher Scientific, Pittsburg, PA). A BZ-X810 Keyence microscope (Keyence Corp Osaka, Japan) was used to acquire wide-field cortical images. High resolution images of the MCA and its branching vessels from the right hemisphere were acquired with a Zeiss LSM 700 NLO laser scanning confocal microscope (Jena, Germany).
(Additional acquisition and analysis methods are further detailed in Supplementary Materials).
Classical vessel analysis
Quantitative analysis was performed as previously described. 14 Briefly, in Fiji software, regions of interest (ROIs) were drawn around the right and left hemispheres on the axial cortical images. On coronal images, ROIs were drawn on Neurotrace images (see Immunohistochemistry below) and then overlayed onto the processed fluorescent images of the vessel painting for the BLA and dorsal hippocampus (see Supplementary Materials for details). Analysis results included vessel density (percentage of area covered by vessels), junctional branch points (number of branches/mm2), average vessel length (average length of vessel segments), and total vessel length (total length of vessel segments) (Figure 1). Due to the unilateral or asymmetric nature of FSE-related MRI changes in children 15,16 and rodents,9,17 we also compared the vessel features in the hemisphere with the lower values. For comparison of right vs. left hemisphere and high vs. low vascular features, only pups that had bilateral uniform staining were used. From cortical images, the hemisphere with the lower vessel density value was designated as low whereas the opposite side was designated as high. Branch points, average vessel length, and total vessel length were then dichotomized for each brain using the vessel density designations. Data from the coronal images were obtained using the low side designation derived from the axial cortical images.
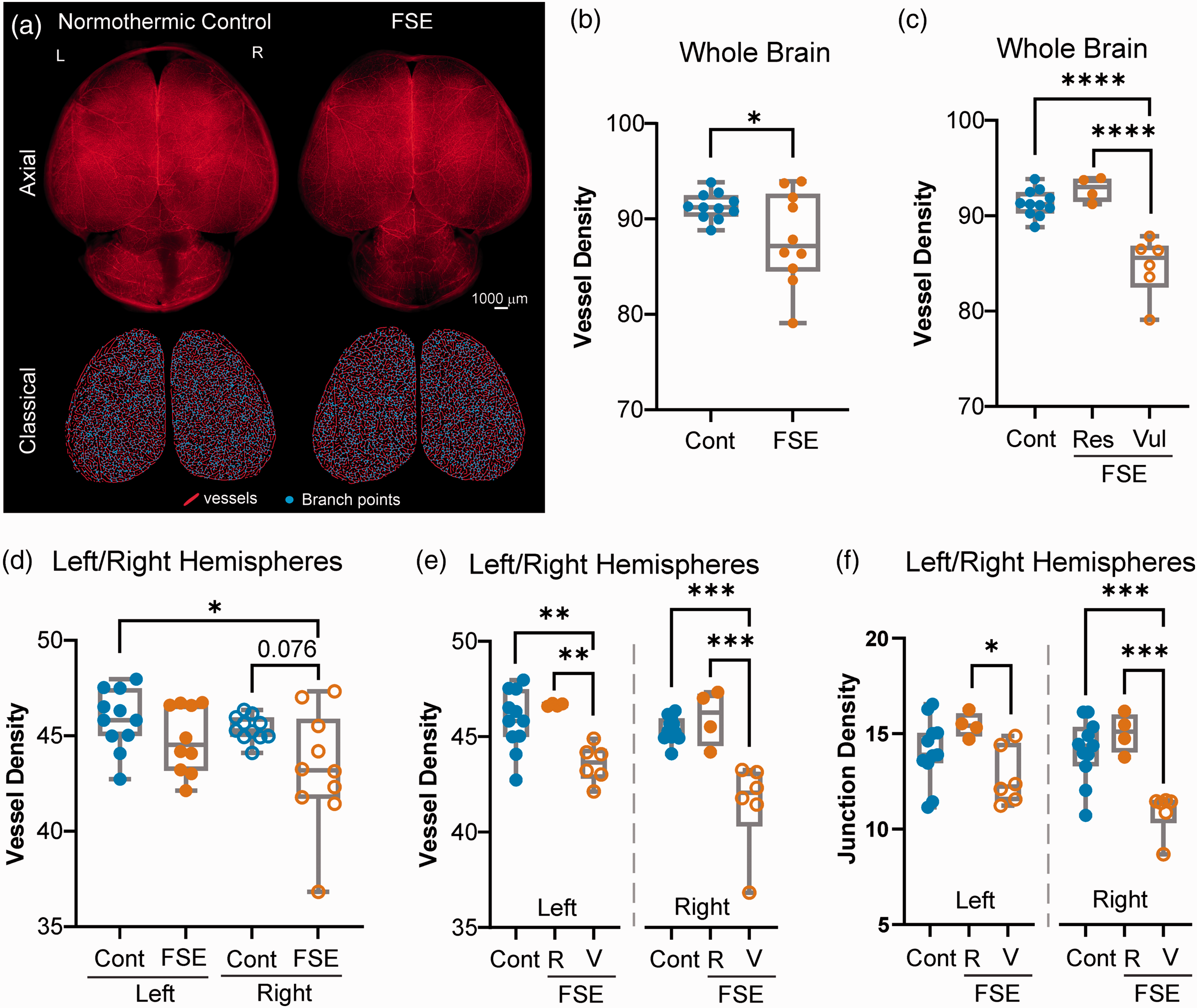
Whole brain and hemispheric vessel features. (a) Vessel painting of normothermic control and FSE rat pups were undertaken at 2 hrs after the onset of status epilepticus. Uniform vascular labeling occurred in both groups as shown in axial images (top panel). Vascular features were extracted from whole brain and right/left hemispheres for vessel length (red lines) and branch points (junctions, blue dots). (b) Whole brain axial cortical vessel density (% of hemisphere area) was significantly different between groups (t-test p = 0.039). However, the FSE group appeared to have two distinct groups. We have previously reported on individual vulnerability following FSE. (c) Segregating the FSE pups into either low or high vessel density exhibited significant differences (one-way ANOVA, ****p < 0.0001) between the vulnerable and resistant vessel density group and controls (Tukey’s post-hoc, p < 0.0001) and the high vessel density group (****p < 0.0001). (d) When right/left hemispheres were individually examined for vessel density, trending reductions (p = 0.076) were observed in the right hemisphere. (e) Significant differences in vessel density were found between resilient (R) and vulnerable (V) FSE rat pups, with the right side having larger decrements (**p < 0.001, ***p < 0.0001). (f) Vascular junction density followed similar patterns as vessel density, where the right hemisphere in vulnerable FSE rat pups appeared to have significant reductions compared to controls and FSE resilient pups (*p < 0.05, ***p < 0.0001). Individual data points represent measurements from individual rat pups.
Fractal analyses
Fractal analyses for vascular complexity on the right and/or left somatosensory cortex on the confocal images were performed using the Fraclac Fiji plugin. 18 Local fractal dimensions (LFD) were obtained using the box counting method where LFD (complexity) versus frequency (vessel number) are displayed.
Vessel diameter analysis
To examine vessel diameters, extracted ROIs were processed in Fiji to create tubular structures (Tubeness filter, sigma value: 10), converted to 8 bit, and thresholded (cortex images, 15 to 255; coronal images, 17 to 255). Images were then cropped to minimize black space. The Fiji plugin DiameterJ was used to obtain the mean vessel diameter and frequency histograms. 19 We opted to use the histogram average as the mean vessel diameter as the data fell within a Gaussian distribution and the skewness and kurtosis values were between −1 and 1. The resultant histogram displays vessel diameter (µm) vs. frequency (vessel pixels along the fiber length).
Neurotrace staining for BLA and hippocampal localization
After vessel painting analyses coronal slices were incubated with Neurotrace (NeuroTrace
Perfusion-weighted magnetic resonance imaging (PWI)
We generated a cohort of FSE and control pups (FSE n = 4, controls n = 4) that underwent contrast-based PWI at 2–4hrs post-FSE. For PWI, pups were anesthetized (2% isoflurane in air) and a cannula was inserted and glued intraperitoneally as tail veins were impacted during FSE induction. Syringes containing gadolinium (Multihance™ gadobenate dimeglumine 529 mg/ml, Bracco Diagnostics Princeton NJ) at a dose of 10 mmol/kg were attached. PWI data were obtained on a 9.4T (Bruker Avance) imager with a 30 cm bore and images were acquired using a 72 mm diameter RF coil for multi-echo T2-weighted imaging and a flow compensated gradient echo (GEFC) sequence for PWI (4 slices 1 mm thick encompassing the dorsal hippocampus and amygdala), with gadolinium being injected at 90 sec after sequence onset. Pre- and post-T1 3D sequences were also acquired with a total imaging time per rat pup being less than 1 hr (see supplementary materials for additional details).
Perfusion and T2 MRI analysis
Multi-echo T2-weighted scans were processed using Jim software (V9.1, Xinapse Systems Ltd, UK), as previously reported.10,11 PWI data were also processed using Jim software, using the perfusion module. Arterial input function (AIF) was obtained using the auto AIF routine. CBF parametric maps were then generated for region of interest (ROI) analyses upon which the dorsal hippocampus, the cortical region above the hippocampus and the amygdala were delineated. An ROI in the lateral masseter muscle external to the brain was used for CBF normalization (denoted as CBFnorm). The identical ROIs were copied to the T2 images to extract relaxation values. CBFnorm were lateralized (as the vessel data) based on low/high CBFnorm values and all data were summarized in MS Excel.
Statistics
All vascular topology measurements and analysis were performed by two investigators and without knowledge of the groups. All analyses were performed using GraphPad Prism 6.0 or 8.0 (GraphPad, San Diego, CA). Outliers were identified if they were below or above 1.5 times the interquartile range. Statistical tests are noted as performed in the text. Box and whisker plots are shown with mean with the box bounding the 25th and 75th percentiles while the error bars are from the minimum to maximum values. In bar graphs and fractal histograms all error bars are presented as standard deviation of mean (SD). Perfusion and T2-weighted MRI data were analyzed using a 2-way matched ANOVA with Sidak post-hoc testing. Statistical significance was noted at *p < 0.05, **p < 0.01, or ***p < 0.001.
Results
Global features of cortical vessels
We first examined the vascular features of the entire cerebral cortex (Figure 1). Vessel density of the entire bi-hemispheric cortices in FSE rat pups were significantly different from control pups (t-test p = 0.039) (Figure 1(b)). No significant differences in branch points (p = 0.128), average vessel length (p = 0.467) and total vessel length (p = 0.543) were observed between controls and FSE pups when combined hemispheres were examined. The distribution of vascular density in both control and FSE pups was normal (Kolmogorov-Smirnov testing) but the distribution of values in FSE pups suggested two distinct clusters (Figure 1(b)). When we separated those individuals with apparently higher vessel densities (resistant) from those with decreased vascular densities (vulnerable), we observed a highly significant difference between both the control and the resistant FSE groups compared to the vulnerable group (one-way ANOVA, p < 0.0001) (Figure 1(c), Supplemental Fig. 1). These results were expected, because the MRI signal, which might be a result of these vessel differences, was also found only in a subgroup of FSE rats. 11
In both rodent models10,11 and humans,15,16 MRI changes after FSE are uniformly lateralized. Therefore, we re-analyzed the cortical vasculature separating the right and left hemispheres (Figure 1(d) to (f)). When vessel density in FSE pups was compared to controls we found a strong trend for decreased vessel density within the right hemisphere (one-way ANOVA, p = 0.076) (Figure 1(d)). Specifically, FSE pups were separated into the resistant and vulnerable groups and a significant decrease (two-way ANOVA, p = 0.025) in vessel density was found between the control and resistant FSE pups compared to the vulnerable FSE pups, where the vulnerable FSE group was significantly different from control and high FSE pups (Tukey’s post-hoc, p < 0.0001) (Figure 1(e)). A significant difference in junction density in the right hemisphere was detected between control and resistant FSE pups when compared to vulnerable FSE pups (two-way ANOVA, p = 0.04, Tukey’s post-hoc, p < 0.0001). In the left hemisphere, differences were only observed between resistant and vulnerable FSE pups (p < 0.05) but not compared to controls (Figure 1(f)). Significant differences between the FSE and control groups were also found for average vessel length (one-way ANOVA, p = 0.005; vulnerable vs control p = 0.017, vulnerable vs resistant p = 0.006).
Other vascular features such as average vessel length were also significantly different between hemispheres with control and resistant FSE pups having larger vessel segments on average compared to vulnerable FSE pups (Tukey’s post-hoc, left hemisphere p < 0.05, right hemisphere p < 0.05). Total vessel length was not altered in any of the groups. Lacunarity (space between vessels) was significantly increased in the right hemisphere of vulnerable FSE compared to control and resistant FSE pups (Tukey’s post-hoc p < 0.05) but no statistically significant differences were found on the left side. In sum, FSE elicits decrements in cortical vascular features (density, junctions, average length) compared to controls. Dichotomizing the FSE into resilient and vulnerable groups clearly showed that a subset (vulnerable) of FSE pups have dramatic vascular abnormalities at 2 hrs after FSE onset compared to resistant FSE pups that exhibit vascular topology similar to controls.
Hemispheric differences in cortical vessels
FSE leads to MRI signal changes that are lateralized or often confined to a single hemisphere. In addition, in the rodent model of FSE, the right hemisphere appeared to be more involved. 17 We plotted right/left vascular features for each control and FSE pup for comparison (Figure 2(a)). Hemispheric differences were not observed (controls p = 0.977, FSE p = 0.153) in vessel density (Figure 2(a)). In FSE rats, there were significant differences between the two hemispheres, but they did not involve the right or left hemisphere consistently (t-test, p = 0.009) (Figure 2(a)). Importantly, in the more impacted hemisphere we observed highly significant decreases in vessel density in vulnerable FSE pups compared to FSE resistant and control groups (Figure 2(b)). Interestingly, the vulnerable FSE pups had reduced vessel density clustered entirely on the right hemisphere unlike the resistant FSE pups which were equally distributed.
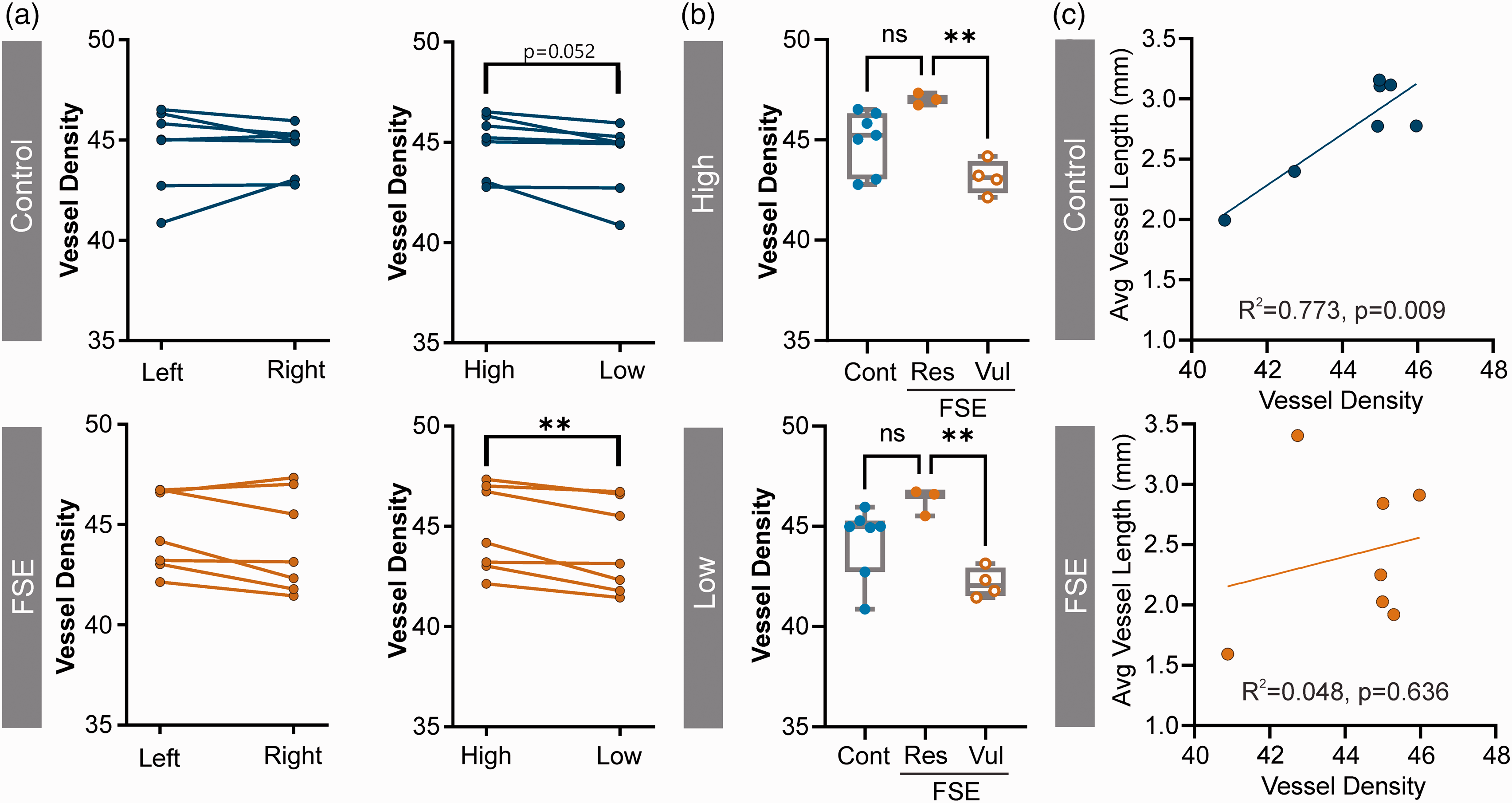
Lateralization in FSE. (a) Right and left hemispheres showed no significant differences in vessel density between controls and FSE pups (left panel). When vessel densities were dichotomized into high vs. low vessel density control pups approached significance (p = 0.052) but FSE pups were strongly significantly different (**p = 0.009) (right panel). (b) Further separation based on the vulnerable and resistant FSE pups further demonstrated highly significant reductions in vessel density in the vulnerable group compared to FSE (High **p = 0.004, Low **p = 0.007) and (c) Correlations between the low hemisphere vessel density and average vessel length were strongly correlated in control pups (R2 = 0.773, p = 0.009) but was disrupted in FSE pups (R2 = 0.048, p = 0.636). Individual data points represent measurements from individual rat pups.
Vascular junction density was not significantly different between right and left hemispheres. When right and left hemispheres were dichotomized into high junction density vs. low junction density as a percent change, both FSE pups and controls exhibited a significant decrease in junction density compared to the high hemisphere (6.96% ± 3.33, p = 0.004 control; 6.02% ± 2.81, p = 0.003 FSE). Within FSE pups, there was a significant difference in junction density between vulnerable and resistant pups (p = 0.012) but not compared to controls. Thus, junction density does not differentiate between FSE pups and controls.
The right hemisphere of every FSE pup had reduced total vessel length compared to the left side. Accordingly, total vessel length was significantly reduced in the right hemisphere of FSE pups (p = 0.010) compared to the left with no significant differences in controls. In contrast, average vessel length was not significantly different between hemispheres in control or FSE pups. When hemispheric average vessel length data were sorted into high vessel length vs. low vessel length values, there was a significant difference in controls (p = 0.023) and in FSE pups there was a more prominent decrease in average vessel length (p = 0.003).
In control pups there was a strong correlation between vessel density and average vessel length (R2 = 0.773, p = 0.009). Notably, this correlation was not found in FSE pups (R2 = 0.048, p = 0.636), as shown in Figure 2(c).
Middle cerebral artery characteristics
The MCA is one of the most prominent cortical vessels and supplies the amygdala, a primary site of MRI signal abnormalities (Figure 3(a)). We measured the diameter of the MCA branches (M2-4) in controls and FSE pups (Figure 3(a), Supplemental Figs. 2, 3) and no significant differences were found between groups (two-way ANOVA, p = 0.846). As expected, reductions in vessel diameter were found within both control and FSE pups (p < 0.0001) as we moved to increasing distal branches (Figure 3(b)). There were no significant MCA branch X Group effects (p = 0.748). Examination of FSE pups for resilient vs. vulnerable dichotomization found no significant differences after repeated measures ANOVA testing (p = 0.089). However, resilient FSE MCA vessel diameters tended to be larger than controls and vulnerable FSE vessels (Figure 3(c)). Vessel diameter frequency histograms did not identify overt global changes in vessel diameters when either right, left or lower vessel diameter histograms were examined (Figure 3(d)).
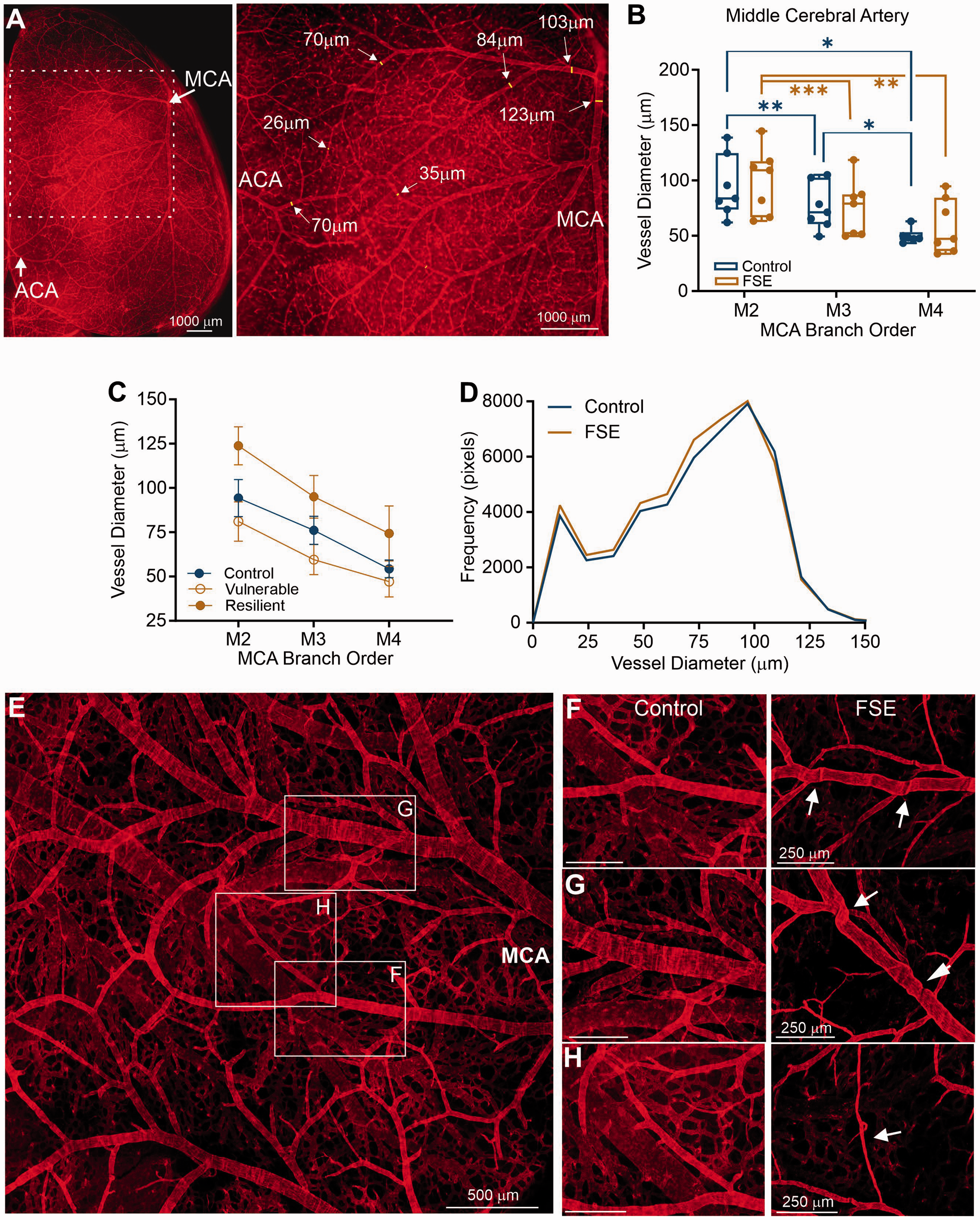
Vascular features of the middle cerebral artery (MCA). (a) The most prominent vascular structure on the axial surface of the brain is the MCA (image shown is from a Control rat pup; representative images FSE pup shown in Supplementary Figure 3). Diameter measurements were applied to confocal images from vessel painted brains. (ACA – anterior cerebral artery). (b) There were no significant differences between control and FSE pups in vessel diameters but both groups progressed to small diameters with increased branching (M2-4). (c) Resilient and vulnerable FSE groups were compared and while the resilient group had elevated diameters compared to controls and vulnerable FSE pups, these findings were not significant. (d) Frequency histograms of vessel diameter did not exhibit any overt differences when comparisons were made to lower hemispheric vessel diameters. (e) In control pups the MCA territory exhibited a robust vascular network with normal appearing vascular elements and (f–h). However, comparisons of vessels between control (left panel) and FSE (right panel) exhibited robust differences in FSE animals with vessel folding (f), tortuosity at junctions (g) and vessel thinning (h). Individual data points represent measurements from individual rat pups.
While quantitative MCA metrics were not overtly different between control and FSE pups, significant qualitative vascular abnormalities between the groups were visible. In control pups there was a substantial vascular network present within the MCA territory region that is comprised of a distribution of larger and small vessels (Figure 3(e)). Closer examination in the controls showed normal vascular diameters, branching and vascular density (Figure 3(f) to (h), left panels). The vascular features in controls were in stark contrast with the features from the MCA in FSE pups (Figure 3(f) to (h), right panels). Many FSE vessels in the MCA territory had apparent kinks (Figure 3(f), right), tortuosity at vascular bifurcations (Figure 3(g), right) and numerous thin vessel segments (Figure 3(h), right). Thus, while global features may be only moderately altered, examination of the vascular network at high magnification revealed dramatic differences in vessel angioarchitecture, and these resembled changes reported in several brain pathologies. 20
Vessel characteristics of the basolateral amygdala
The basolateral amygdala (BLA) appeared to be uniquely sensitive to FSE and MRI signal changes in this region predicting long-term development of epilepsy.10,11 A random subset of whole vessel painted brains (n = 4/group) were blocked at the level of the dorsal hippocampus which included the BLA (∼Bregma −2.40 mm) (Figure 4(a) and (b), Supplementary Fig. 4). First, we identified group (control vs FSE) differences in the total number of vessel junctions (mixed model, p = 0.025) (Figure 4(c)). Vascular characteristics within the right versus left BLA were not overtly different between control and FSE pups, including vessel density (one way ANOVA p = 0.30), average vessel length (p = 0.237) and junctional density (p = 0.255). We noted a trend for differences in vessel area p = 0.076 and total vessel length (p = 0.085). We then divided for each pup the high vs low hemisphere/BLA as above and identified a significant inter-hemispheric difference in the number of junctions in FSE but not control pups (post-hoc t-test, p = 0.047) (Figure 4(d)).
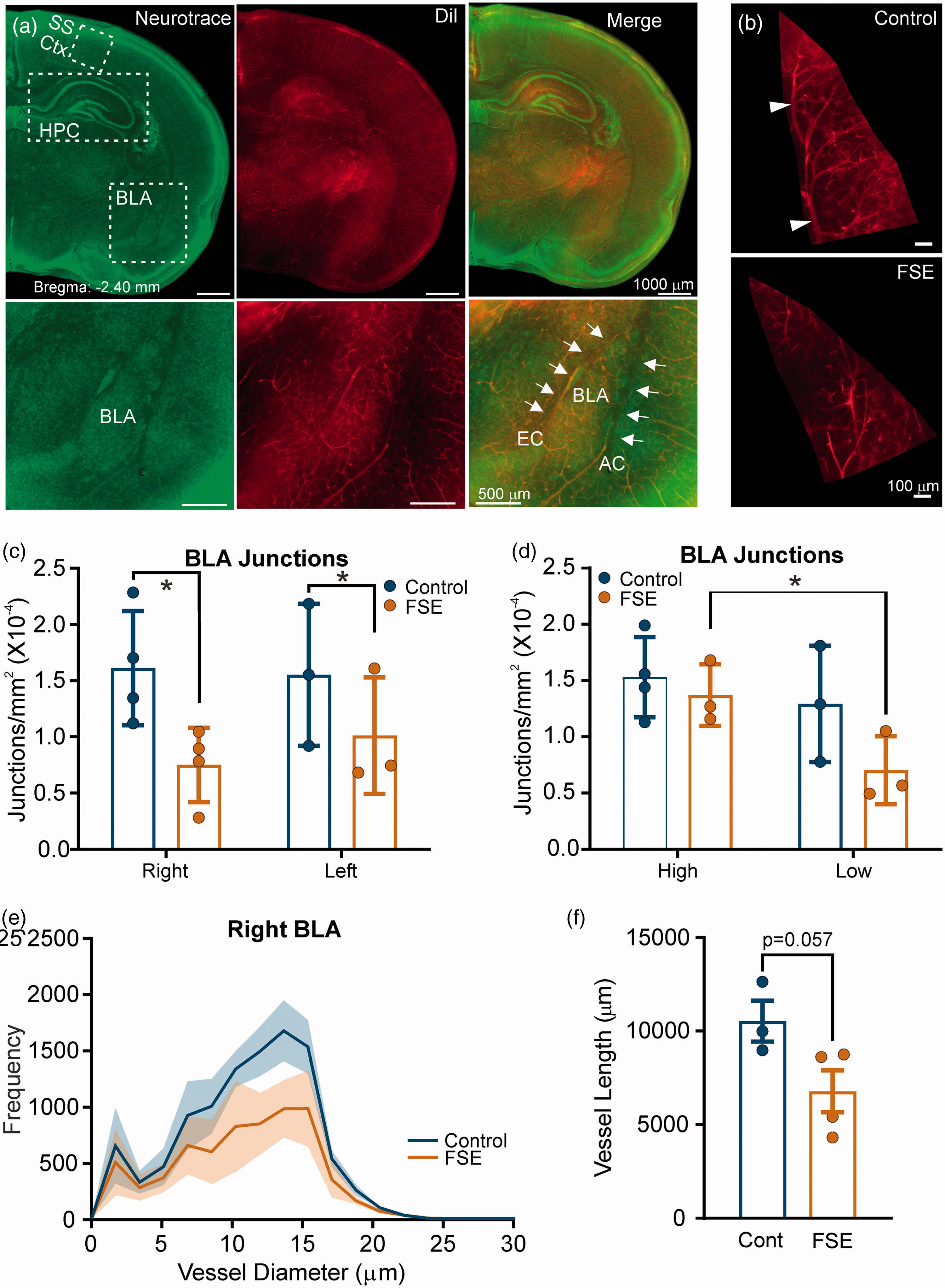
Basolateral amygdala vessel features. (a) Representative coronal slices at the level of the dorsal hippocampus for somatosensory cortex (SS Ctx), hippocampus (HPC) and basolateral (BLA) vessel analyses in a control subject (Supplementary Figure 4 illustrates a representative FSE rat pup). Left panel are sections stained for Neurotrace to assist in region localization. The middle panel is the vessel painted image from the same section and the right panel is the merged images. The bottom row of images was taken at higher magnification to show localization of the BLA. (b) Representative vessel painted images from the right BLA of a control (top) and FSE (bottom). As can be seen vascular density is reduced. (c) Vessel junctions in the right and left BLA were significantly different between groups (Control vs FSE; mixed model, *p = 0.025) but not different between right and left. (d) Junctions were then dichotomized to high and low with no significant differences between controls (mixed model, p = 0.126), but in FSE pups the low side was significantly reduced compared to the high side (post-hoc t-test, p = 0.047). (e) Vessel diameters within the BLA were also quantified with a 55.5% decrease in the area under the curve (t-test, p = 0.067) but only 17.7% on the left BLA (data not shown).(f) Trending significant reductions in total vessel length were observed in the right (but not left) BLA (t-test, p = 0.057). Individual data points represent measurements from individual rat pups.
Diameters of vessels in the BLA were robustly (55.5%) decreased in 10–15μm sized vessels (Figure 4(e)). In the right BLA, area under the curve analysis found a trend for decreased vessel diameters between control and FSE curves (t-test, p = 0.067) (Figure 4(e)) with no significant differences in curve skewness or kurtosis. There was a trend for a decrease in the frequency of vessels sized 11–20 μm (Mann-Whitney, p = 0.057) in the right BLA. There were no significant differences in vessel diameters <11 or >20 μm. In contrast with the right hemisphere, there was a non-significant 17.7% decrease in vessel diameters in the left BLA based on area under the curve.
Fractal analysis for vessel complexity did not find significant differences between control and FSE pups, although there were reduced numbers and complexity in FSE pups compared to controls (data not shown), This trend was more prominent in the right compared to the left BLA.
Vessel characteristics of the hippocampus
Hippocampus is also a site of altered MRI signal after FSE,10,17 and is highly involved in MRI signal changes in the human.16,21 Here, a trend for an interaction between groups (control vs. FSE) and right vs left hippocampus (mixed effects, p = 0.054) was noted. Significant interactions were observed for average vessel length (mixed effects, p = 0.006) and junction density (mixed effects, p = 0.032) between control and FSE pups (Figure 5(a)). Of note, vessel length was increased in left FSE hippocampus compared to control pups (post-hoc testing p = 0.027), and junction density trended higher (post-hoc test p = 0.079) (Supplemental Fig. 5).
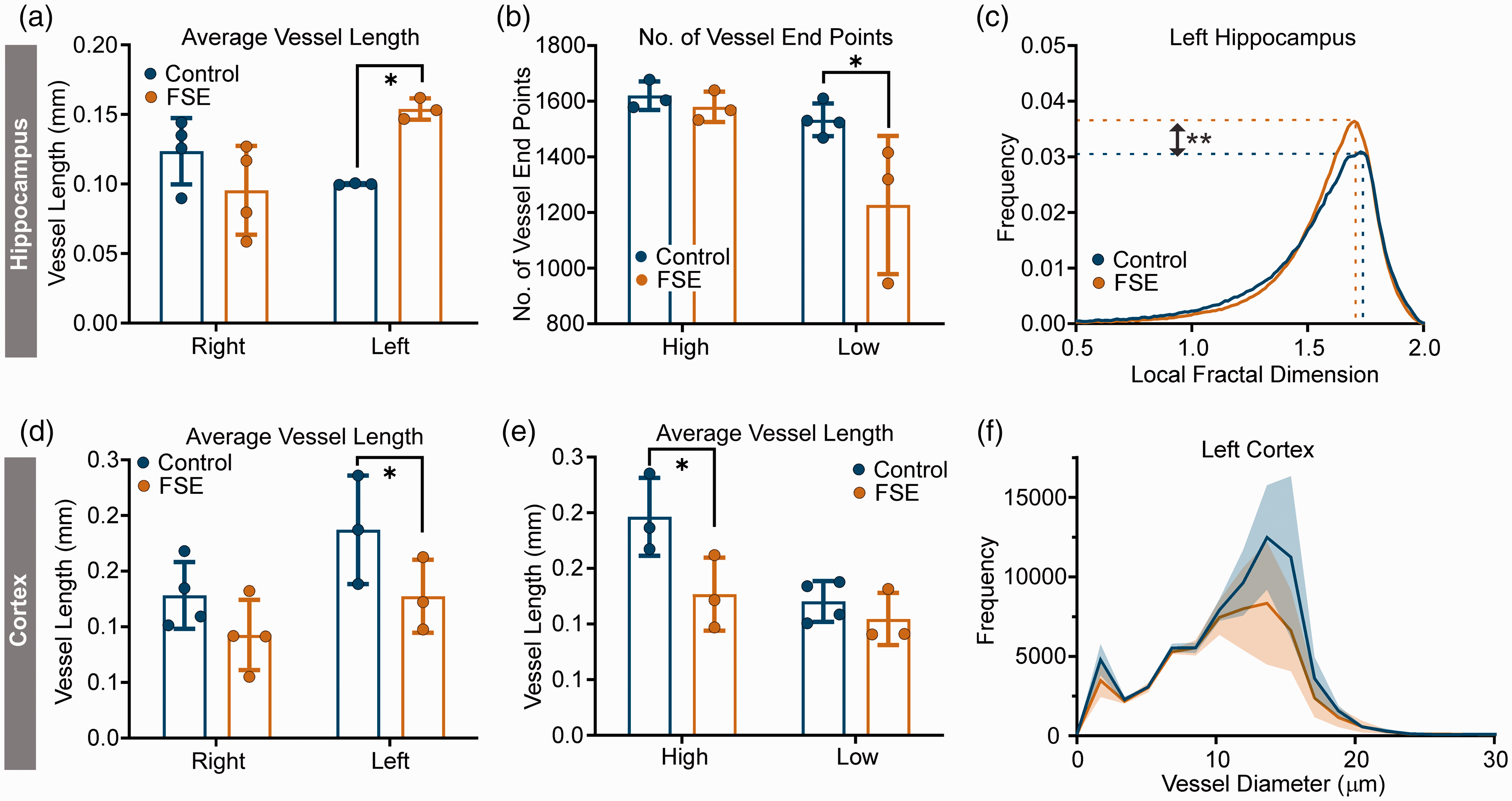
Vascular features of the hippocampus and cortex. (a) Average vessel length was increased in FSE pups compared to controls in the left hippocampus (mixed effects post-hoc, p = 0.027). (b) The number of vessel end points were significantly reduced in FSE pups compared to controls when dichotomized into high/low groups (mixed effects post-hoc, p = 0.023). (c) In fractal analysis the left hippocampus of FSE pups displayed increased peak frequency (t-test, p = 0.007) consistent with increased vessel numbers (see a). (d) In the cortex, average vessel length was significantly different in the left cortex between control and FSE pups (*p = 0.032). (e) Average vessel length was then grouped into high/low designations and there was a significant reduction in the high FSE group compared to controls (mixed effects, *p = 0.012). There were no differences between FSE high and low lateralization in the cortex. (f) Vessel diameter histogram for the left cortex illustrates reductions in vessel diameter in the FSE pups compared to controls; this reduction was not significant. Data shown as mean ± SD. Individual data points represent measurements from individual rat pups.
As in the BLA, we compared high vs. low hemispheric vessel features. Vessel density was significantly lateralized (mixed effects, p = 0.0002), with both controls and FSE pups having similar high/low values (data not shown). Similar inter-hemispheric differences in junction density (mixed effects, p = 0.0003), vessel area (mixed effects, p = 0.015), total vessel length (mixed effects, p = 0.023) and average vessel length (mixed effects, p = 0.002) were observed. Only the number of vessel end points (terminations) was significantly reduced in FSE compared to controls (mixed effects, p = 0.018; post-hoc p = 0.023) (Figure 5(b)). There were no significant changes in vessel diameters in either the right or left hippocampus of FSE pups compared to controls.
Fractal analysis was performed to buttress the classical neuroanatomical approach (Figure 5(c)). Maximum frequency was elevated in the left hemisphere (t-test, p = 0.007) but not the right hemisphere (t-test, p = 0.707) of FSE compared with controls. The maximum frequency is considered a measure of vessel number. 14 Similarly in the left hemisphere, skewness and kurtosis were significantly different from controls (t-tests, p = 0.007, p = 0.015, respectively) with a trend for changes in the right hemisphere (skewness p = 0.065, kurtosis p = 0.071). These fractal changes within the hippocampus are in accord with and confirm the increased classical vascular features.
Somatosensory cortex
The somatosensory cortical region immediately above the hippocampus was also assessed for vascular modifications. There were no significant changes in vessel area, total number of junctions, vessel junction density, total vessel length between control and FSE pups nor between right and left hemispheres. However, there was a significant difference in average vessel length between right and left hemispheres (mixed effects, p = 0.034) and between FSE and control pups (mixed effects, p = 0.032) and no significant interaction between group and hemisphere (mixed effects, p = 0.544) (Figure 5(d)).
When hemispheres were dichotomized into high and low vessel features, total number of junctions (mixed effects, p = 0.005), junction density (mixed effects, p = 0.001) and average vessel length (mixed effects, p = 0.012) were significantly different between groups and high/low designations. In all significant parameters the high designation was significantly different between the groups, where vascular features were significantly lower in FSE pups than controls (Figure 5(e)). The number of vessel end points was higher in FSE compared to control pups, but this was not significant (mixed effects, p = 0.177). The increased average vessel lengths potentially suggests that there may be increased vessel fragmentation or reduced branching in the cortex as other vascular parameters were not significantly different between controls and FSE pups.
Vessel diameter histograms showed reductions in vessel diameters with distance from vessel origin in both the left and right cortex but were not significantly different between controls and FSE pups (Figure 5(f)). Similarly, no other vessel diameter parameters were significantly altered. Accordingly, fractal analyses found no significant differences in any measure within the somatosensory cortex.
Perfusion-weighted imaging (PWI)
PWI experiments uncovered several important features in support of our hypothesis that altered vascular topology explains epilepsy predictive T2 changes where increased deoxygenated blood coincides with increased CBF. We observed that CBFnorm was elevated throughout the brain in FSE pups relative to controls in the cortex (Figure 6(a)), hippocampus (Supplemental Fig. 6 A) and was largest in the amygdala (Figure 6(d)). CBFnorm data were then lateralized into low (L) and high (H) CBFnorm groups. Significant elevations in CBFnorm between control vs FSE pups (two-way matched ANOVA, cortex p = 0.0002, amygdala p = 0.0007) (Figure 6(b) and (e)) with Sidak post-hoc testing reporting a trending (p = 0.077) increased CBFnorm unilaterally in the High (H) amygdala of FSE groups compared to control High (H) (Figure 6(b) and (e)).
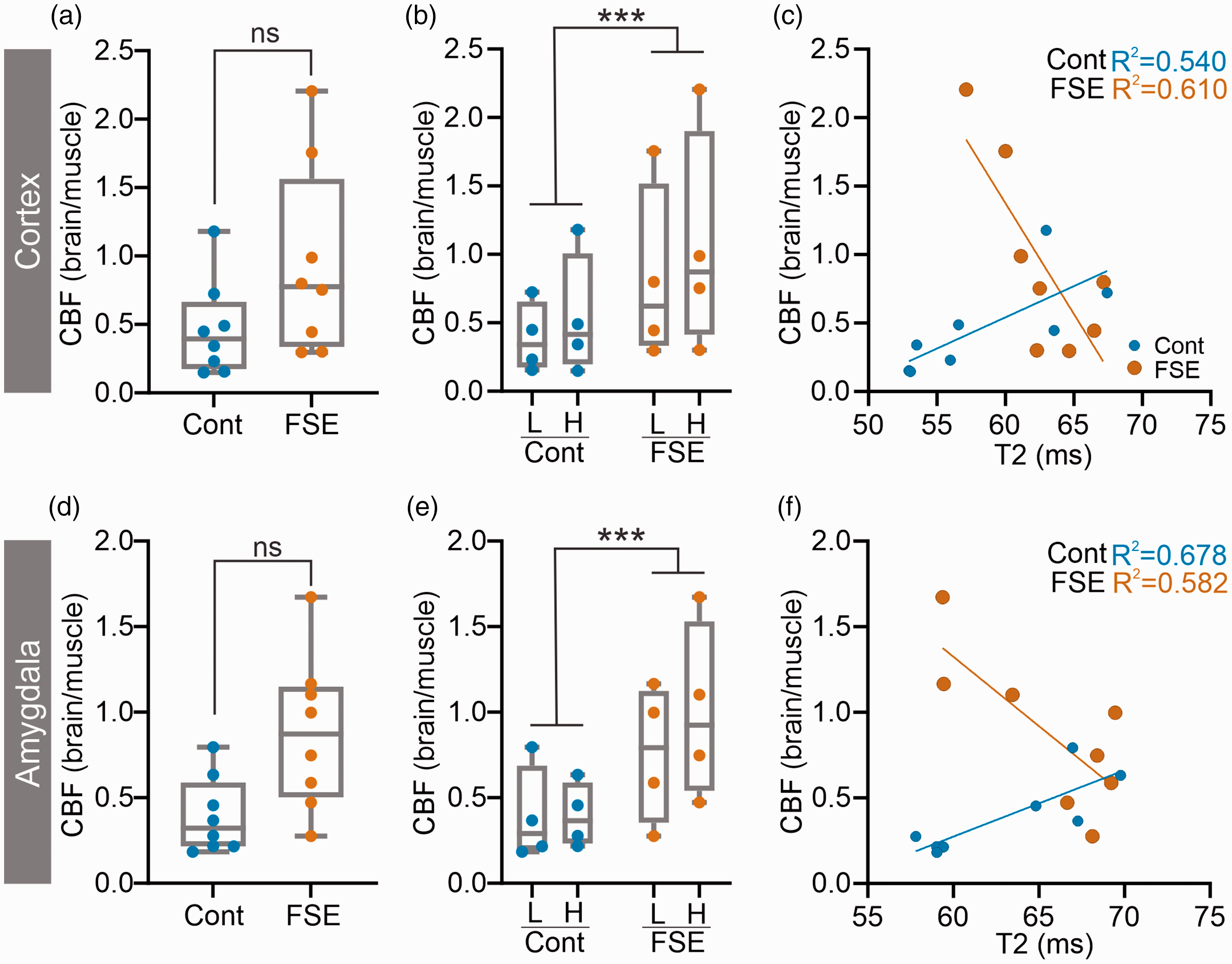
Elevated CBF after FSE. Increased CBFnorm was observed in the cortex (a–c) and the amygdala (d–f) of FSE pups. In the cortex (b), FSE pups had elevated CBFnorm when both low (L) and high (H) T2 values were used to dichotomize asymmetry. Similarly, in the amygdala (e) CBFnorm was elevated with trending differences observed on post-hoc testing between control H and FSE H regions (p = 0.07). Correlations between regional tissue CBFnorm and T2 values illustrated an opposing correlation in Control compared to FSE pups (c, f). In FSE pups reduced T2 values were associated with increased CBFnorm. Individual data points represent measurements from individual rat pups.
T2 relaxation (ms) measures from the identical regions were elevated in FSE pups relative to controls but were not significantly different from each other (data not shown). However, T2 values were significantly different between control and FSE pups in cortex (two-way matched ANOVA, p = 0.003), amygdala (p = 0.001) and hippocampus (p = 0.011). No significant differences were found between L and H within any region.
As we previously hypothesized a relationship between T2 values and cerebral metabolism, 10 we undertook regional correlations between T2 and CBFnorm. As shown in Figure 6(c), control pups had positive correlation between increasing T2 values and increasing CBFnorm in the cortex. In contrast, FSE pups exhibited the exact opposite correlation with low T2 values relative to high CBFnorm in the cortex (Figure 6(c)). This was also found in the amygdala (Figure 6(f)) and hippocampus (Supplementary Fig 6 C). Correlation slope values for control and FSE pups illustrated the large negative correlations in FSE pups for hippocampus, amygdala and cortex compared to positive correlation slopes in the control rat pups (Supplementary Fig. 7 A). Intriguingly, we also observed increased thinning and tortuosity in cortical vessels (Supplementary Fig. 7B) that may contribute to increased CBF, but this awaits further investigation. Thus, acutely after FSE, elevations in CBFnorm correlated strongly with reduced T2 relaxation, consistent with increased deoxyhemoglobin and oxygen utilization as we previously reported after acute FSE. 10
Discussion
These studies report on novel and acute changes in vascular topology after experimental FSE. Notably, these changes provide a potential mechanism for the acute MRI signals observed in a subset of rats after experimental FSE, changes that predict the future development of epilepsy.
Using a hyperthermic model of FSE, we report acute changes in the vascular topology. The major findings in FSE pups are: 1) global reductions in cortical vascular density wherein a subset demonstrated highly significant reductions (FSE vulnerable). 2) Global changes were lateralized, i.e. one hemisphere was more affected than the other. 3) The MCA territory vessel topology exhibited tortuosity, thinning (string-like) and abnormal kinks at the vessel bifurcations. 4) The BLA exhibited reductions in junctional density related to reduced vascular length. 5) The hippocampus had lateralized increases that contrasted with decreased average vessel length in the somatosensory cortex. In summary, the novel predominant finding was that there were reductions in vascular elements (density, junctions etc) acutely after FSE. These acute modifications may have implications in the development of subsequent epilepsy.
Altered vascular topology has been reported both acutely and long-term in adult models of status epilepticus. 22 To our knowledge there are no hyperacute (hours post SE) studies examining the vascular morphology nor studies examining the vascular consequences of FSE. Thus, Ndode-Ekane in a pilocarpine model of status epilepticus (SE) found reduced hippocampal vessel density, vessel length accompanied by increased blood-brain barrier (BBB) leak at 2 days post SE using immunohistochemical staining. 23 Other models of SE, including chemical induction of SE in adult rodents resulted in vessel degeneration23,24 and leakage of the BBB,23,25–27 which are primarily evident in the hippocampus, amygdala, thalamus, and parietal cortex.23,25,27 Fabene and colleagues found that within 2 hrs after adult rat pilocarpine-induced SE onset, cortical vessels exhibited flattened morphology but had increased vascular diameters (∼20%). 28 Interestingly, in deeper cortical layers vascular diameter was decreased by 46% and related to decreased cerebral blood volumes. The authors also noted cell death in the upper cortical layers but not in the lower layers. Using a similar model of pilocarpine-induced SE but at P9 and 21, Marcon and colleagues mapped vessel density at 7 days post SE. 29 Using immunohistochemistry (FITC-albumin, laminin) they found no change in hippocampal, amygdala nor cortical vessel density in P9 rat pups compared to controls. In contrast, at 7 days post SE in P21 rats there was a 30% increase in hippocampal vessel density but only in a subset (62%) of SE rats; the other SE rats were similar to controls. Similar elevations in vessel density were found in cortex (38%) and in amygdala (30%). 29 In combination with other evidence the authors suggest that these increases in vessel density 7 days after SE are the result of ongoing angiogenesis. Similar angiogenic mechanisms were proposed by Ndode-Ekane and colleagues after pilocarpine induced SE in adult rats using immunohistochemical methods. 23 At 2 days post SE there was decreased vessel density and length within the hippocampus (CA1/CA3) that only marginally improved over the 60-day experimental period. However, there was a robust increase in BrdU-labeled endothelial cells at 2, 4 and 14 days after SE, that correlated with increasing vessel length and suggested an angiogenic process. 23 In brain trauma we have demonstrated a strong neo-vasculogenesis mechanism that appears to be independent of vascular endothelial growth factor (VEGF). 30 The role of VEGF in epilepsy has recently been reviewed. 31 Our own data demonstrate that cortical vessel density (Figure 1) is already significantly decreased within 2 hrs after FSE, with similar reductions in the BLA (Figure 4) and increases in the hippocampus (Figure 5). While it is difficult to compare across models and time points, SE results in perturbations of vessel topology.
It is important to recognize that in the developing brain (P10) there is a considerable angiogenesis and refinement of the vasculature 32–34 with the potential for vascular leak. However, in our assessments of the vasculature in cortex, hippocampus and the amygdala we did not observe any overt leakage of DiI, suggesting a relatively preserved BBB at 2 hrs after FSE, in line with our previous studies. 35 The vessel painting method can identify BBB leaks as we have demonstrated in other neonatal brain injury models. 36 In our model the cortical disruptions in vessel density and decreases in vessel junctions (branching) did not lead to overt disruption of the BBB. 35 Thus, changes in vascular angioarchitecture may contribute to ongoing and future pathophysiology of FSE.
A second novel aspect of the altered vascular network was the apparent lateralization within the cortex of the vascular decrements. There were no significant differences between right/left hemispheres vessel density in controls (0.02%) or FSE (1.67%) pups. In our previous MRI studies, we observed that there was lateralization of the T2 biomarker signal.10,11 We used a similar strategy in our vascular assessments (high vs. low values) to lateralize these findings. When lateralization was performed, FSE pups had a 2% reduction in vessel density (high vs. low) that was significant whilst controls had a 1.6% change that was trending. While no studies exist on vascular lateralization there is some support for lateralization of blood flow in seizures, with increases during ictal discharges and decreased blood flow after seizure cessation that was asymmetric between hemispheres. 37 We recently reported similar asymmetric differences in MRI T2 signal and CBF in a preliminary study of FSE patients that underwent MRI evaluations within 6 hrs of SE onset. 38 CBF has been also shown to lateralize with pathology in newborns with focal seizures. 39 Thus, while there is scant literature on vascular lateralization, future studies should examine if alterations are asymmetric and as such may have the potential for diagnostic interpretation.
In human sclerotic hippocampi there was a 60–70% reduction in the number of microvessels that were also accompanied by a wide variety of vascular abnormalities. 40 The vasculature of the sclerotic CA1 had a rough appearance with spine-like protrusions and abnormal tubular structures that were associated with severe neuronal loss and gliosis. In two mouse models of epilepsy, microvascular vasospasms were increased relative to control mice with increased mural cell numbers and vessel constrictions. 41 The authors went on to demonstrate that capillary vasospasms increased 400% within 80 sec prior to seizure onset, suggesting that vascular constriction may lead or certainly contribute to seizure onset. These vessel spasms then lead to local hypoxia with subsequent cell death. 41 In our model of FSE there is no overt neuropathology, but within the MCA territory we observed tortuous vessels, increased vascular “kinking” or abnormal folding of the vessels adjacent to vessel branches (Figure 3(g) and (h)). While the cellular basis for these abnormalities is uncertain, it is tempting to speculate that capillary pericytes that are associated with vessel branches may contribute to the altered morphology. 42 Similar modified pericyte structures in tissues excised from human temporal lobe epilepsy patients were associated with tortuous and abnormal vessels, frequently at vessel junctions. 43 Thin or string-like vessels, like those we observed in the cortex of FSE pups (Figure 3(h)), have been associated with a variety of disease states.20,44 These abnormal structures have not been rigorously studied in SE and specifically in FSE but likely represent ongoing vascular pathology that may impact long-term patient outcomes.
An interesting aspect of our study is that vessel painting allows brain-wide vessel staining. Thus, in addition to cortical assessments, we can probe specific regions of interest, such as the hippocampus, BLA and somatosensory cortex above the hippocampus (Figures 4 and 5). We found generalized decreases in vascular features in the BLA and cortex but increases in the hippocampus. Increased vascular densities were found in the cortex and amygdala, days and weeks after adult rat pilocarpine and kainate-induced SE.29,45 Within the hippocampus the overwhelming evidence is for increased microvascular density days to weeks after induced adult SE (see Table 1 in Marcon 22 for a concise summary). In our study we found increased vessel length in the hippocampus of FSE pups at 2 hrs after the cessation of SE that was mirrored by our fractal analyses (Figure 5(c)). Functionally, cerebral blood flow and volume were not altered in the hippocampus at 2 days post pilocarpine SE but was increased at 14 days when vessel density increased. 23 In human temporal lobe epilepsy, bilateral decreases in CBF were found in unilateral epilepsy which was unrelated to neuronal loss. 46 Given the paucity of studies on regional vessel density, the use of different models and time points, it is difficult to craft a picture of how the vasculature responds to SE. Our own data provide a glimpse into the acute vascular response to FSE but clearly additional studies are warranted.
We also undertook measurement of local tissue CBF to assess if the altered vascular topology impacts local blood flow. We observed elevated CBF within the cortex, amygdala and hippocampus (Figure 6) acutely after FSE. Intriguingly, a case report study with three FSE patients found elevated CBF at the site of the epileptic foci. 12 A recent larger study during the sub-acute phase (3 days post-FSE) found that there were no significant differences in CBF within hippocampus. 13 An interesting aspect of our current study is the strong negative correlation between increased CBF and decreased T2 relaxation values (Figure 6(c)). These correlations imply a link between increased deoxyhemoglobin (increased oxygen consumption leads to decreased T2) and increased CBF, similar to our previously reported conjecture. 10 Nehlig and colleagues, using pentylenetetrazol induced SE in P10 rats found an acute 2 fold increase in CBF, that was less prominent with increasing age. 47 A follow-up study demonstrated that P21 pups during SE did not have blood flow/metabolism mismatch while adult SE rats did. 48 The authors posited that the lack of CBF-metabolic mismatch in young rats prevented frank neuronal degeneration, unlike cell loss observed in the adult. Our findings suggest that CBF increases in vulnerable regions, such as the amygdala, in concert with local increased metabolic demand (decreased T2).
Several limitations of the current study are noted. Firstly, temporal monitoring of vascular topology at later chronic and latent epileptogenic epochs would provide insights into the dynamic nature of the vascular response to FSE. A second limitation is that vascular mechanisms were not directly evaluated. As noted, the goal of the present study was to identify the acute (2–8 hr) vascular changes in vessel structure after FSE. Herein we identified those modifications but unanswered are if these topological changes are transient, permanent, or potentially recover to an altered physiological state as a consequence of FSE. We will explore these concepts in detail in future studies. The present study could not exclude the effects of potentially collapsed vessels and might lead to reduced vessel density (via vessel painting) (see Supplementary Fig 6B). Finally, reduced flow in some segments of the vasculature could potentially contribute to reduced vessel DiI staining, although the increased CBF appears to mitigate those effects (see Figure 6). Future studies will aim to identify the linkage between angioarchitectural features, blood flow and modifications to the associated cellular constituents (astrocytes, pericytes etc) within the same animals.
The current studies employed a preclinical model of FSE, in which an acute MRI signal presaged the development of TLE-like limbic epilepsy. Recently, the same signal has been observed in infants with FSE, 38 but it is not yet clear if the same signal predicts epilepsy in children with FSE. This possibility, now under study, 38 provides strong impetus for identifying the underpinnings of the predictive signal that associates with augmented deoxygenated hemoglobin. The abnormal vessel topology found here, including tortuosity and reduced diameters and vessel density, provide a plausible and important mechanism for the MRI observation, potentially leading to strategies for intervention.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221117625 - Supplemental material for Vascular topology and blood flow are acutely impacted by experimental febrile status epilepticus
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221117625 for Vascular topology and blood flow are acutely impacted by experimental febrile status epilepticus by Arjang Salehi, Sirus Salari, Amandine Jullienne, Jennifer Daglian, Kevin Chen, Tallie Z Baram and Andre Obenaus in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were supported by an NIH Training grant from the National Institute of Neurological Disorders and Stroke (T32 NS-45540) and with access to the Optical Biology Core Facility of the Developmental Biology Center supported by the Cancer Center Support Grant (CA-62203) and Center for Complex Biological Systems Support Grant (GM-076516) at the University of California Irvine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Concept and Design: AS, TZB, AO
Data Acquisition: AS, JD, KC, AJ
Data Analysis: AS, SS, AO, AJ
Data Interpretation: AS, TZB, AO
Manuscript Drafts and Revisions: AS, AJ, TZB, AO
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
