Abstract
Microthrombi formation in the brain following subarachnoid hemorrhage (SAH) has been recognized and suspected to contribute to cerebral ischemia. A recent study found that ultra-early cerebral micro-thrombosis occured four hours after experimental SAH. The number of thrombotic microvessels correlated with brain-blood barrier disruption and neuronal injury. If acute cerebral micro-thrombi also occur in humans, is it time to develop a therapy with systemic thrombolysis for SAH patients?
Subarachnoid hemorrhage (SAH) is a life-threatening cerebrovascular disease. Even with recent advanced SAH management, the death rate is still 10 to 24%, 1 and about half of survivors suffer from cognitive impairment. Many studies have focused on the later phase of thrombosis (days or weeks after the onset of SAH) and delayed cerebral ischemia. In contrast, early vascular changes and early brain damage after SAH (less than 24 hours after onset) are still relatively poorly understood. Recently, early vascular injury, especially the development of micro-thrombi in small arterioles, venules and capillaries, has drawn research focus, 2 and those micro-thrombi may be related to delayed neurological deficits. However, little is known about how and when micro-thrombosis occurs after SAH. Our recent study detected ultra-early micro-thrombosis (4 hours after the onset of SAH) by T2* gradient-echo sequence magnetic resonance imaging (MRI) in a mouse model of SAH, and this was confirmed by histology. 3 The T2* positive vessels might be venules. T2* imaging may be a way of tracking the time course of micro-thrombosis formation and resolution after SAH, including in human.
The mechanisms of the acute micro-thrombi formation may be multifactorial. Within 1 minute of SAH onset, intracranial pressure (ICP) can rise as high as mean arterial blood pressure and, although it then falls, it still remains above normal. 4 The dramatic increase in ICP results in transient cerebral circulation stasis or severely decreased cerebral blood flow in both arteries and veins. Arteriolar constriction and micro-thrombi have been observed 3 hours after SAH. 5 This hypo-perfused state leads to global ischemia followed by a hypoxic state and cerebral cell damage and dysfunction. Global ischemia disrupts the brain-blood-barrier by damaging endothelial cells. Blood in the brain also activates the coagulation cascade and thrombin formation. All these factors contribute to Virchow’s triad of stasis of blood flow, endothelial injury and hypercoagulability, and subsequently to acute micro-thrombosis.
The development of micro-thrombi over time warrants further research. To date, there is a lack of evidence on whether ultra-early micro-thrombi will experience self-resolution. Studies have found micro-thrombi from a few hours up to 96 hours after SAH induction with a peak at 48 hours. 5 The nature of histological studies makes it difficult to monitor changes in thrombi continually over time. Our current research indicates that MRI might be a way to monitor micro-thrombi changes and relate ultra-early thrombosis to late functional outcomes. Micro-thrombi were first found in a SAH patient autopsy studies, while most studies of microthrombi formation after SAH were done in rodent models. Future studies utilizing MRI in SAH will be more valuable if performed in large animals and eventually in human. In current clinical practice, SAH patients rarely undergo MRI in the acute phase, and such MRI research on human is limited. It would be intriguing to see if a similar phenomenon were found in SAH patients.
The presence of microthrombi suggests that they could be a therapeutic target (e.g., with anti-coagulant or anti-platelet agents or thrombolysis; Figure 1). Such an approach raises issues related to possible rebleeding and just when such agents might be started. There has been research trying to prevent SAH-induced microvascular thrombosis. Researchers found that intravenously administering recombinant human ADAMTS13, the vWF cleaving enzyme, reduced SAH induced micro-thrombi in mice. 6 It should be noted that plasma ADAMTS13 is decreased in SAH patients. 2 The role of antiplatelet agents has also been investigated after SAH, including the effects of dual antiplatelet therapy (aspirin and clopidogrel) in SAH patients. 7 Interestingly, the risks of clinical vasospasm and delayed ischemia were significantly lower in patients undergoing stent-assisted coiling with dual antiplatelet than those without, while the risk of hemorrhagic complications was similar. Though those studies did not directly look into early microthrombi formation, the results suggest that suppression of platelet activation might reduce microthrombi. The effect of anticoagulants, including heparin and low-molecular-weight fractionated heparin, has also been studied in SAH. A few human studies of the role of enoxaparin showed mixed effects for SAH patients with follow-up time from 3 months to 1 year. 2 However, recent randomized control trials showed safety and some benefits with continuous infusion of low-dose intravenous unfractionated heparin after craniotomy or surgical clipping. The investigations' endpoint included symptomatic vasospasm, CT infarction, modified Rankin scores at 6 months and, Montreal Cognitive Assessment test over 3 months. 8 An ongoing phase 2 clinical trial, ASTROH (NCT02501434), is further examining the effects of heparin in SAH patients and may provide more evidence to move it to phase 3 trial and eventually clinical practice. The current clinical examinations of heparin after SAH have started 12 hours after securing the aneurysm. 9 Our study reveals ultra-early micro-thrombosis after SAH and raises questions over when such therapy should be initiated. It also raises the idea of whether early systemic thrombolysis to remove microthrombi could be a new therapy for SAH patients after endovascular embolism treatment or clipping of the aneurysm provided it doesn’t promote rebleeding. An initial treatment regimen might be that used in ischemic stroke with a standard dose (0.9 mg/kg) or low dose (0.6 mg/kg) of alteplase. 10 Similarly, for rodent studies, a standard dose of tPA treatment of 10 mg/kg or a low dose tPA treatment of 6.67 mg/kg could be explored. We aim to find out how the standard and low doses of tPA work for the removal of micro-thrombi in vessels after SAH in our future studies.
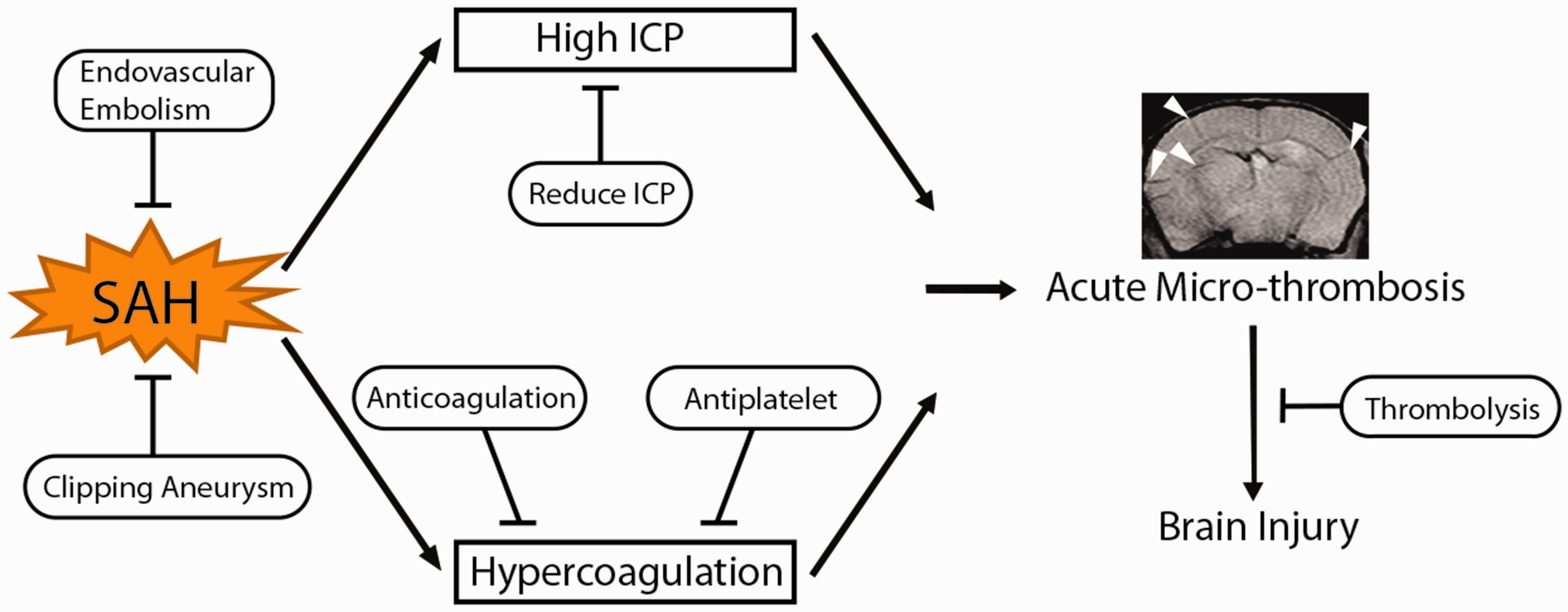
An overview of acute micro-thrombosis after subarachnoid hemorrhage. Arrowheads indicate T2* positive micro-thrombosis in a mouse model of subarachnoid hemorrhage. ICP: intracranial pressure.
In summary, micro-thrombosis after SAH occurs within a few hours of ictus in rodents. Future studies should address whether this phenomenon also occurs in humans. More detailed analyses of the micro-thrombus including formation and resolution should be performed. Systemic thrombolysis could be a new therapeutic strategy for SAH patients (Figure 1).
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: YH, RFK and GX are supported by grants NS-096917, NS-106746, NS-112394 and NS-116786 from the National Institutes of Health (NIH).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
