Abstract
Ischemic strokes are highly prevalent in the elderly population and are a leading cause of mortality and morbidity worldwide. The risk of ischemic stroke increases in advanced age, corresponding with a noted decrease in circulating insulin growth factor-1 (IGF-1). IGF-1 is a known neuroprotectant involved in embryonic development, neurogenesis, neurotransmission, cognition, and lifespan. Clinically, several studies have shown that reduced levels of IGF-1 correlate with increased mortality rate, poorer functional outcomes, and increased morbidities following an ischemic stroke. In animal models of ischemia, administering exogenous IGF-1 using various routes of administration (intranasal, intravenous, subcutaneous, or topical) at various time points prior to and following insult attenuates neurological damage and accompanying behavioral changes caused by ischemia. However, there are some contrasting findings in select clinical and preclinical studies. This review discusses the role of IGF-1 as a determinant factor of ischemic stroke outcomes, both within the clinical settings and preclinical animal models. Furthermore, the review provides insight on the role of IGF-1 in mechanisms and cellular processes that contribute to stroke damage.
Introduction
Strokes are the fifth leading cause of death in the United States and a major cause of disability in the elderly. 1 Ischemic strokes are the most common form as they account for 87% of all stroke incidents. 1 Efforts to reduce the time between the onset of ischemia and the reperfusion of blood flow (with pharmacological and/or surgical interventions) are key for favorable outcomes in stroke patients. Importantly, there are also endogenous molecular signals that contribute to favorable outcomes with ischemic stroke, suggesting avenues for novel therapeutic interventions in the time surrounding the incident. Specifically, there have been several lines of evidence showing that the levels of insulin-like growth factor 1 (IGF-1) in the time surrounding insult positively correlate with favorable outcomes. IGF-1 has also been implicated in many pathologies associated with vasculature as well as neuronal health.2–4 However, within stroke research, conflicting findings have also been published showing no association of IGF-1 levels with physical or behavioral outcomes. The aim of this review to evaluate the role of IGF-1 in clinical and preclinical studies and discuss how IGF-1 contributes to the viability and function of key cell types in the brain following ischemic conditions.
Insulin-like growth factor-1
IGF-1, also known as Somatomedin C, is a 70 amino acid polypeptide primarily produced and secreted by hepatocytes into the bloodstream, where it exerts effects throughout the system (Figure 1). Expression and release is tightly regulated by the somatrophic system. Pulsatile release of growth hormone from the anterior pituitary gland activates growth hormone receptor signaling on hepatocytes thereby increasing downstream IGF-1 transcription and translation. Nearly 75% of IGF-1 in circulation is produced and released by these GH-responsive hepatocytes.5,6 Circulating IGF-1 is able to cross the blood brain barrier to regulate a number of cells within the brain. 7 When labeled versions of IGF-1 are injected via intravenous (iv) or subcutaneous (sc) routes, significant uptake is noted within 20 minutes of administration.8,9 Unfortunately, the half-life of exogenous IGF-1 is quite limited due rapid, extensive proteolytic degradation in serum.8,9 Hepatic-derived, circulating IGF-1 is bound by insulin-like growth factor binding proteins, like IGFBP-3, which modulate bioavailability, increase the half-life, and prevent non-specific binding to other receptors (as reviewed by 10 ) (Figure 1).
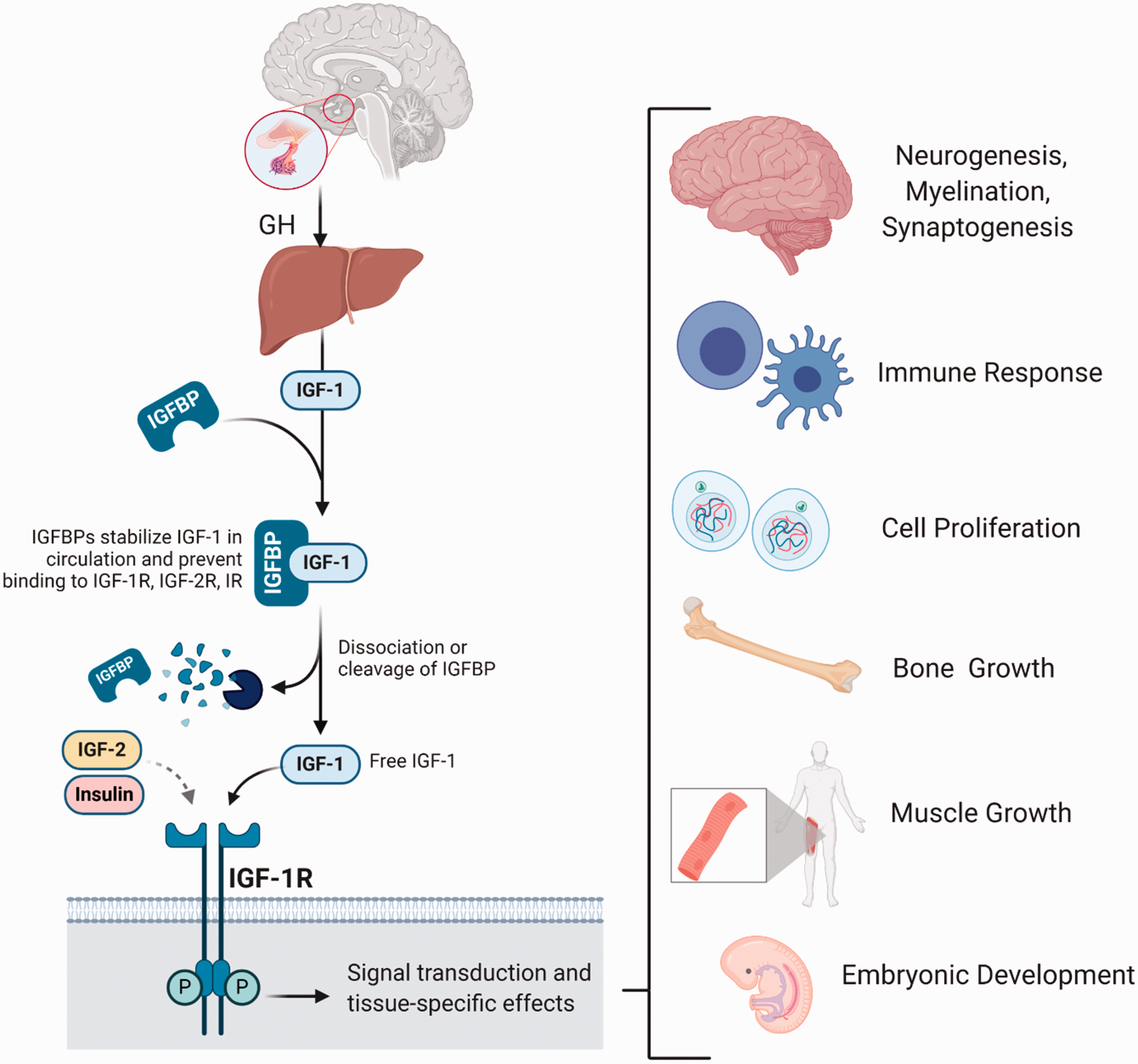
Regulation of IGF-1 mediated effects throughout the body. Left) IGF-1 signaling is tightly regulated by growth hormone control of hepatocytic IGF-1 production and release, IGFBP binding and dissociation in circulation, and its selective receptor, IGF-1R. Right) Known effects of IGF-1 in tissues throughout the body.
Several tissues, including the gonads, muscles, and brain, also express and release IGF-1 locally where it acts in both a paracrine and autocrine manner. Within the brain, IGF-1 is synthesized by both neurons and supporting glial cells. Regardless of the tissue or origin, IGF-1 predominantly acts as an agonist of the insulin-like growth factor-1 receptor (IGF-1R), which is found in all major cell types throughout the body, including the various cells within the brain.11–13 IGF-1R can also be activated by IGF-2 and insulin, as these ligands are structurally homologous to IGF-1. 14 Similarly, IGF-1 shows weak affinity to the insulin receptor (IR) as well as affinity towards hybrid receptors of IGF-1R/IR. Despite crosstalk between these receptors and ligands, the functional effects attributed to each signal is varied. IGF-2 mainly functions during gestation and embryonic growth within the muscle and vasculature, and is not tightly-controlled by growth hormone secretion.15–17 Insulin secretion and effects are modulated by growth hormone throughout the lifespan, although the function of insulin is largely associated with metabolism and nutrient sensing. 18 IGF-1 has many pleiotropic functions and is expressed throughout the post-natal period and in adulthood. The importance of IGF-1 and activation of IGF-1R is has been documented in proper embryonic development, myelination, synapse formation, development of the nervous system, neurogenesis, and neurotransmission 19–23 (Figure 1). We will discuss the importance of IGF-1 in a wide array of cellular roles that are essential in maintaining brain homeostasis following stroke (later in the review). Similar to other growth factors, IGF-1 is also a highly potent antiapoptotic factor for eukaryotic cells and is known to be a neuroprotectant in inflammatory and excitotoxic conditions, which will be discussed further. Overall, through its numerous pro-survival actions in the brain, IGF-1 has the capacity to reduce tissue damage induced by ischemia and reperfusion.
Clinical connections between IGF-1 and ischemic stroke
Effects of IGF-1 on risk, mortality, and survival rate of ischemic stroke
There are several risk factors for ischemic stroke that include, but are not limited to, hypertension, diabetes, smoking, atrial fibrillation, and transient ischemic attack. 24 Aging is also considered a major risk factor, as the incidence of stroke and stroke-associated mortality and morbidity significantly increase in advanced age. 1 Notably, IGF-1 levels decline with age, and several studies, including a study from the large Framingham cohort, have showed an inverse correlation between baseline circulating IGF-1 levels and stroke incidence (Table 1).25–29 When circulating levels of IGF-1 fall within the lowest quartile or quintile within these studies, the risk of ischemic stroke was increased over 2 fold.25,26 Not all studies have observed a significant inverse relationship, as Kaplan et al suggests that IGF-1 levels in circulation are not associated with the incidence of stroke. 30 The reasons for this discrepancy are not understood; however, it should be noted that the Framingham cohort was a prospective study in which patients were enrolled prior to stroke and blood samples for IGF-1 quantification were taken at the time of enrollment. 25 Many of the other studies quantify the levels of IGF-1 in the days following ischemic insult, which may be more phenotypic of a response to the insult rather than a read-out of risk. Interestingly, reduced IGF-1 levels are also associated with increased risk of hypertension, diabetes, and other cardiovascular diseases.31–35 This indicates that the age-related reductions in IGF-1 correspond with increases in other major stroke risk factors.
Clinical relevance of IGF-1 on stroke outcome.
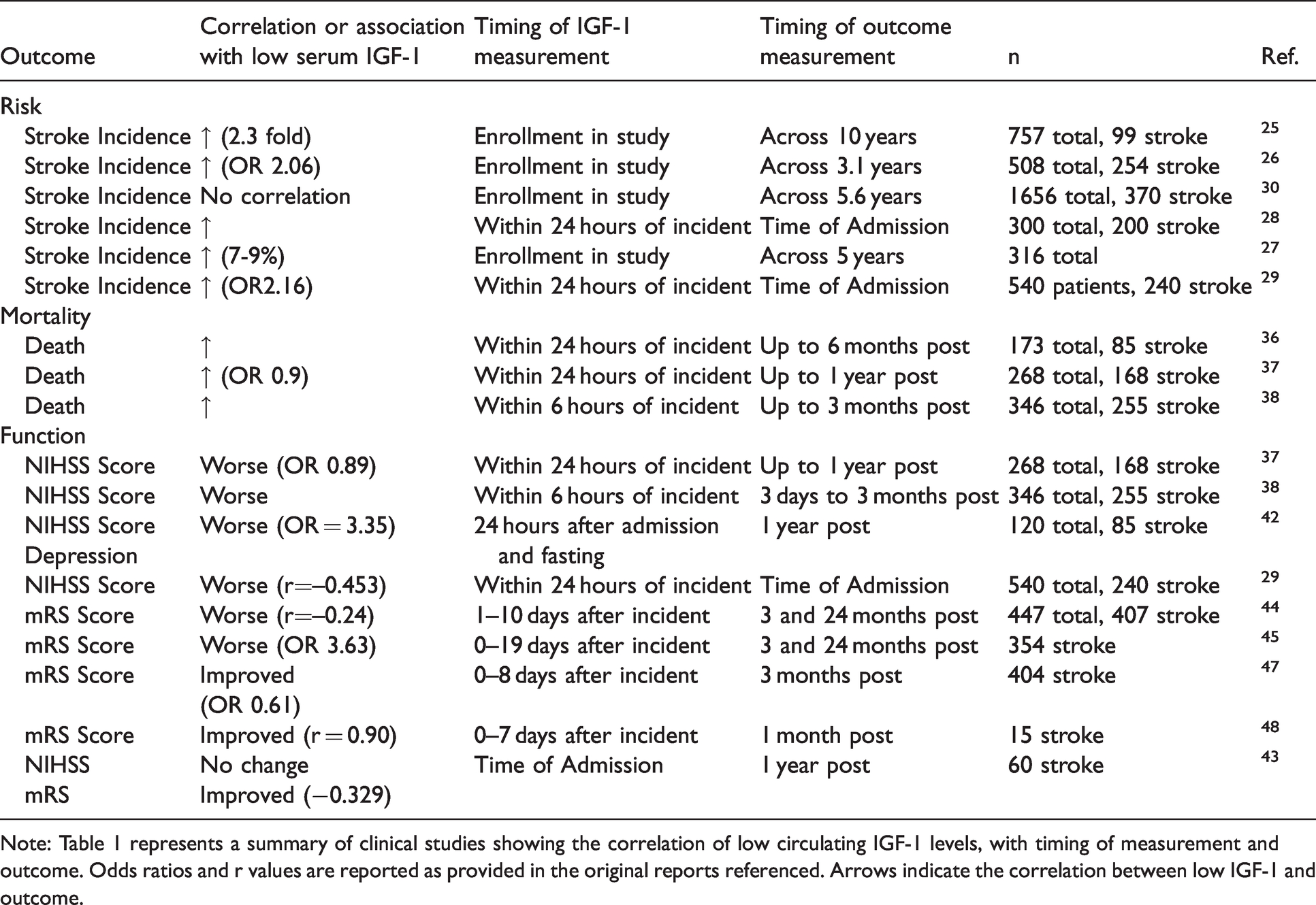
Note: Table 1 represents a summary of clinical studies showing the correlation of low circulating IGF-1 levels, with timing of measurement and outcome. Odds ratios and r values are reported as provided in the original reports referenced. Arrows indicate the correlation between low IGF-1 and outcome.
In addition to impacting the risk of stroke, IGF-1 levels can influence mortality rate, the extent of damage, and recovery of stroke patients. Mortality rate following stroke is affected by a variety of intrinsic and extrinsic conditions, such as age, ethnicity, comorbidities, the latency to medical intervention, and the length/extent of oxygen deprivation in the tissue. Several studies have shown that individuals with higher circulating IGF-1 levels at the time of hospital admission have reduced immediate mortality, while patients with lower levels of IGF-1 have a concomitant increase in mortality.36–38 This finding extends past immediate mortality, as survival rate continues to be increased 90 days following the incident when IGF-1 levels are higher than 75 ng/mL. 38 Later time points were not assessed in this study, however the difference in cumulative survival at 90 days remained quite pronounced. 38 A recent study indicated that within chronic ischemic stroke survivors, the levels of IGF-1 in circulation 3 months following insult were not associated with changes in mortality 7 years later, nor were they associated with increased risk of recurrent stroke. 39 Thus, while IGF-1 levels prior to, or at the time of ischemic insult may influence mortality, the levels present within circulation several months following insult may not.
Effects of IGF-1 on functional outcomes
The levels of IGF-1 within the serum are also inversely associated with neurological deficits following stroke. Neurological deficits are correlated with infarct size and are commonly measured using The National Institutes of Health Stroke Scale (NIHSS) and Modified Rankin Scale (mRS) scores. The NIHSS is used to determine stroke severity based upon a scoring of the level of consciousness, eye movements, integrity of visual fields, facial movements, limb muscle strength, coordination, language, speech, neglect, and sensation. 40 The higher the NIHSS score, the more severe the stroke. The mRS scale classifies patients by a combination of favorable or unfavorable outcomes after a stroke based on the ability to perform daily activities. 41 Increased mRS scores indicate increased disability caused by a stroke. Multiple in vivo and clinical studies agree that lower levels of circulating IGF-1 upon hospital admission correlate with an increase in the NIHSS score in the months following insult.29,37,42,43 Acute stroke survivors (<6 months post insult) with favorable outcomes on the mRS scale also exhibited significantly higher levels of serum IGF-1 when compared to individuals classified with unfavorable outcomes. 37
In order to better understand the relationship between circulating IGF-1 and post-stroke recovery, Åberg and collaborators have followed a cohort of chronic ischemic stroke survivors in the years following insult. A reduction in circulating IGF-1 levels is noted within the months following stroke, as well as overtime as the patients age. 44 Three months following stroke, higher IGF-1 levels were beneficial as they correlated with favorable mRS scores. 44 Follow-up studies at 2 and 7 years indicate a much subtler change in outcome.39,45 Interestingly, the beneficial effects of physical exercise on cognition in chronic stroke patients are also accompanied by higher circulating IGF-1, suggesting that effective interventions are also marked by increased IGF-1 signaling. 46
On the other hand, Armbrust et al observed more favorable mRS scores in acute stroke patients when IGF-1 levels following insult were low 8 days after insult. 47 Similarly, a progressive decline in circulating IGF-1 during the first week after a stroke has been shown to be a predictor of favorable outcomes, including shorter length of hospital stay and greater independence at 1 month. 48 The differences seen in these contradictory studies has yet to be explained, and it should be noted that the second study consisted of a low sample size (n = 15). It is possible that the timing of IGF-1 surges and depletions is critical for neurological outcomes. This will require further examination in future studies.
Influences of IGF-1 on Post-Stroke morbidities
IGF-1 levels are not only associated with the direct deficits and outcomes of ischemic stroke, but also the onset of subsequent disorders and morbidities commonly linked with stroke (Figure 2). Evidence provided by Zhang et al indicates that levels of IGF-1 in humans are negatively correlated with the development of post-stroke depression. 42 These are consistent with studies which show that IGF-1 levels are predictors for and/or can protect against other forms of depression and anxiety in rodents, as assessed by forced swim and open field activity.49–51 Interestingly, patients suffering from post-stroke depression have higher levels of high sensitivity C reactive protein (hs-CRP)- an indicator of inflammation. 42 The levels of hs-CRP are inversely correlated with IGF-1, as they are higher in patients with lower serum IGF-1 levels. 37 Moreover, within this same chronic stroke patient population, lower IGF-1 is associated with higher NIHSS scores, which ties the inflammatory phenotype, IGF-1, and neurological deficits. 37 Several other studies have highlighted the inverse relationship between IGF-1 and hs-CRP and other inflammatory mediators associated with major depressive disorders. 52
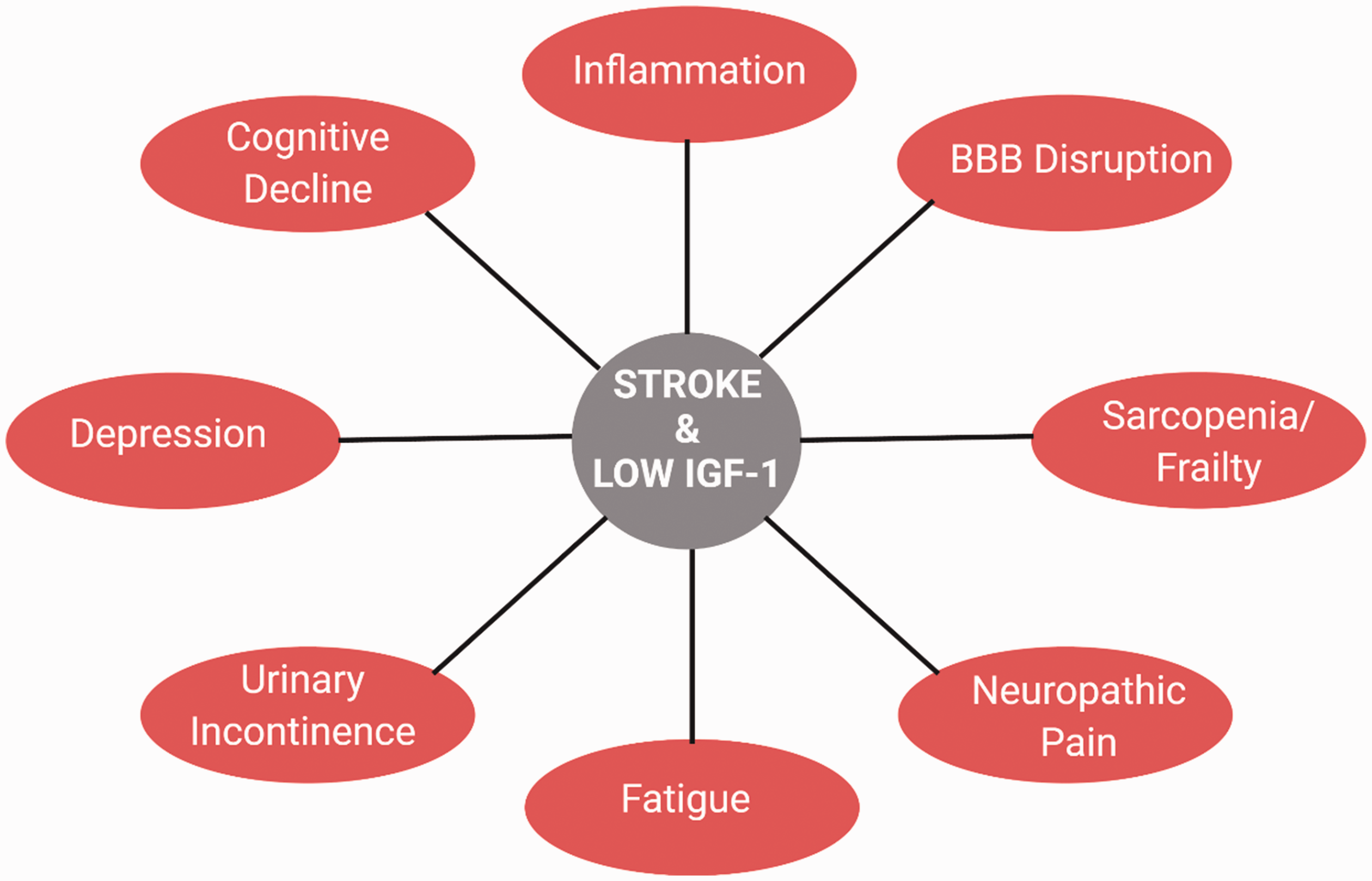
Overlapping pathophysiologies and behavioral deficits associated with ischemic stroke and low levels of circulating IGF-1.
Additional neurological disorders associated with stroke are also influenced by IGF-1. Chronic fatigue and sleep dysfunction are commonly observed in patients following stroke 53 Studies in humans and animal models have shown that IGF-1 promotes a healthy sleep cycle, and is inversely associated with chronic fatigue (as reviewed by Chennaoui et al. 54 ) Moreover, IGF-1 reduces neuropathic pain across several preclinical models,55–57 suggesting IGF-1 may be advantageous for reducing post-stroke pain as well. This association has yet to be tested clinically or pre-clinically.
IGF-1 also promotes and maintains muscle structure and function in advanced age, which could be beneficial to patients following stroke. 58 In this context, a cross-sectional study in chronic stroke patients indicated that low levels of circulating of IGF-1 are associated with muscle atrophy and weakness. 59 This study also had a low sample size (n = 14) and was limited to a single time point for analysis of both IGF-1 and muscle function. Thus, further studies examining the association between IGF-1 and hemiparesis or sarcopenia following stroke are warranted. Preclinical models suggest IGF-1 can promote both skeletal muscle and smooth function, thereby reducing risk of incontinence, salivary dysfunction, dysphagia, and other muscle-related dysfunctions.60–62 Given the substantial literature showing sex-specific effects of IGF-1 on sarcopenia in aged rodents, it will also be important to explore whether IGF-1 affects muscle function following stroke in a sex- or gender-specific manner in humans and animal models.
Many facets of vascular function are also supported by IGF-1. IGF-1 increases blood flow through nitric oxide-mediated vasodilation and promotes survival and function of the blood brain barrier and the cerebrovasculature.63–68 Acute administration of exogenous IGF-1 in healthy adults increases blood flow which could impact reperfusion and cerebral blood flow following ischemic stroke.69,70 Animal studies have shown that IGF-1 infusion causes an acute reduction in mean arterial pressure.71,72 IGF-1 also reduces endothelial dysfunction by altering plaque stability, inhibiting endothelial cell apoptosis, and inducing anti-inflammatory markers.67,73–75 Thus, the health and function of the cerebrovasculature is dependent on IGF-1 signaling.
Altogether, current data suggest that IGF-1 may influence multiple aspects of health and function following ischemic stroke. Low levels of circulating IGF-1 increase the risk of stroke, reduce survival rate, increase behavioral deficits due to the ischemic tissue damage, and promote microenvironments known to accompany and/or precipitate a number of post-stroke morbidities.
Clinical assessments of IGFBP-3
As mentioned above, IGF-1 is regulated by a number of IGFBPs. In fact, IGF-1 in circulation is usually bound tightly by IGFBPs with an equal or higher affinity than the IGF-1R. 76 This binding increases the half-life of IGF-1 and modulates its ability to activate IGF-1R or the insulin receptor (IR). 77 The most predominant form of IGFBP in circulation is IGFBP-3, which is regulated by growth hormone and produced in the liver similar to IGF-1. 78 Therefore, as GH increases the production of IGF-1, IGFBP-3 is also produced and secreted to be able to bind to IGF-1. 78
Due to the key role of IGFBP-3 regulating the availability of IGF-1, the levels of IGFBP-3 and the ratio of IGF-1 to IGBFP-3 are often assessed in clinical studies of ischemic stroke. Unfortunately, the role that IGFBPs, and specifically IGFBP3, play in stroke remains unclear. The Cardiovascular Health Study did not find an association between the levels of IGFBP3 and the incidence of ischemic stroke. 30 However, studies show that those with a higher IGF-1/IGFBP-3 ratio within 24 hours of admission experience better outcomes three to six months later. 36 On the other hand, multiple studies also show that a decrease of the IGF-1/IGFBP-3 ratio from admission until the first week post-stroke is associated with favorable outcomes as indicated by a lower mRS score one month following stroke.47,48 Similarly, low IGFBP-3 in the first week following stroke was predictive of unfavorable functional outcomes and lesion size up to a year after stroke. 79 Thus, there are many contradictory studies on the influence of IGFBP-3 on stroke risk, damage, and recovery. The question remains whether IGFBP-3 exerts functions independent of IGF-1 that might influence stroke outcomes.
There are noteworthy connections between the function of IGFBPs and use of tissue plasminogen activator (tPA). Administration of IV tPA in patients with stroke increases the bioavailability IGF-1 by inducing a limited plasmin-mediated proteolysis of IGFBP-3.80,81 Specifically, tPA activates fibrin-bound plasminogen. Plasmin then binds to IGFBP-3 and leads to its partial cleavage which is enough to decrease IGF-1 affinity for IGFBP-3. 82 This presumably increases free IGF-1 levels especially in the site of injury available to exert its neuroprotective actions. In fact, pharmacological inhibitors that displace IGFBPs from IGFs have been shown to have neuroprotective effects and reduce infarct volume in animal models.83,84 Importantly, measured IGFBP-3 levels are not affected by tPA treatment as with a limited proteolysis, the protein still maintains its ternary conformation. Additionally, IGFBP-5 has been proposed to enhance the activity of tPA. 85 It is unclear in the previous clinical studies examining IGF-1/IGFBP-3 ratios whether the use of tPA or other acute ischemic stroke therapies prior to serum sampling was controlled for, as this may present a confound in the levels of free IGF-1 able to act on IGF-1R.
Preclinical conclusions regarding IGF-1 and ischemic stroke
Role of IGF-1 on infarct size in preclinical models
Many preclinical studies have tried to determine whether IGF-1 plays a correlative or causative role in the extent of stroke damage and neurological deficits (Table 2). In rodents, these models often induce ischemia through middle cerebral artery occlusion (MCAO), hypoxic ischemia (HI), or administration of the vasoconstricting peptide, endothelin-1 (Et-1). Assessment of stroke-induced damage in IGF-1 knockout models versus exogenous administration of IGF-1 have yielded conflicting results as it relates to the potential protective role of IGF-1. A study by Endres et al examining life-long deficiencies in liver-derived (circulating) IGF-1 deficient mice showed a reduced lesion size following MCAO. 86 Yan et al found that IGF-1 deficient rats show no significant difference in infarct size after with Et-1-induced ischemia, although it was trending towards improvement similar to the study with IGF-1 deficient mice. 87 Considering that these life-long deficiencies in IGF-1 impact development of a healthy brain and vasculature throughout the system, further studies utilizing genetic tools and/or pharmacological manipulations after development are critical. Unlike the life-long IGF-1 knockout models, most studies using exogenous IGF-1 administration after ischemia show a protective effect. In a neonatal rat model subjected to HI and immediately given a 50 µg intracerebroventricular (i.c.v.) injection of IGF-1 followed by an additional two daily injections, infarct zone was attenuated. 88 A 30-minute pre-MCAO treatment (i.c.v.) of 5 mg/kg of IGF-1 in male rats also reduced infarct volume. 89 Overall, this evidence suggests that damage caused by ischemic infarctions can be reduced with an acute treatment with IGF-1 in various doses and routes of administration in rodents.
Summary of preclinical studies that observed attenuated infarct size with IGF-1 treatment.
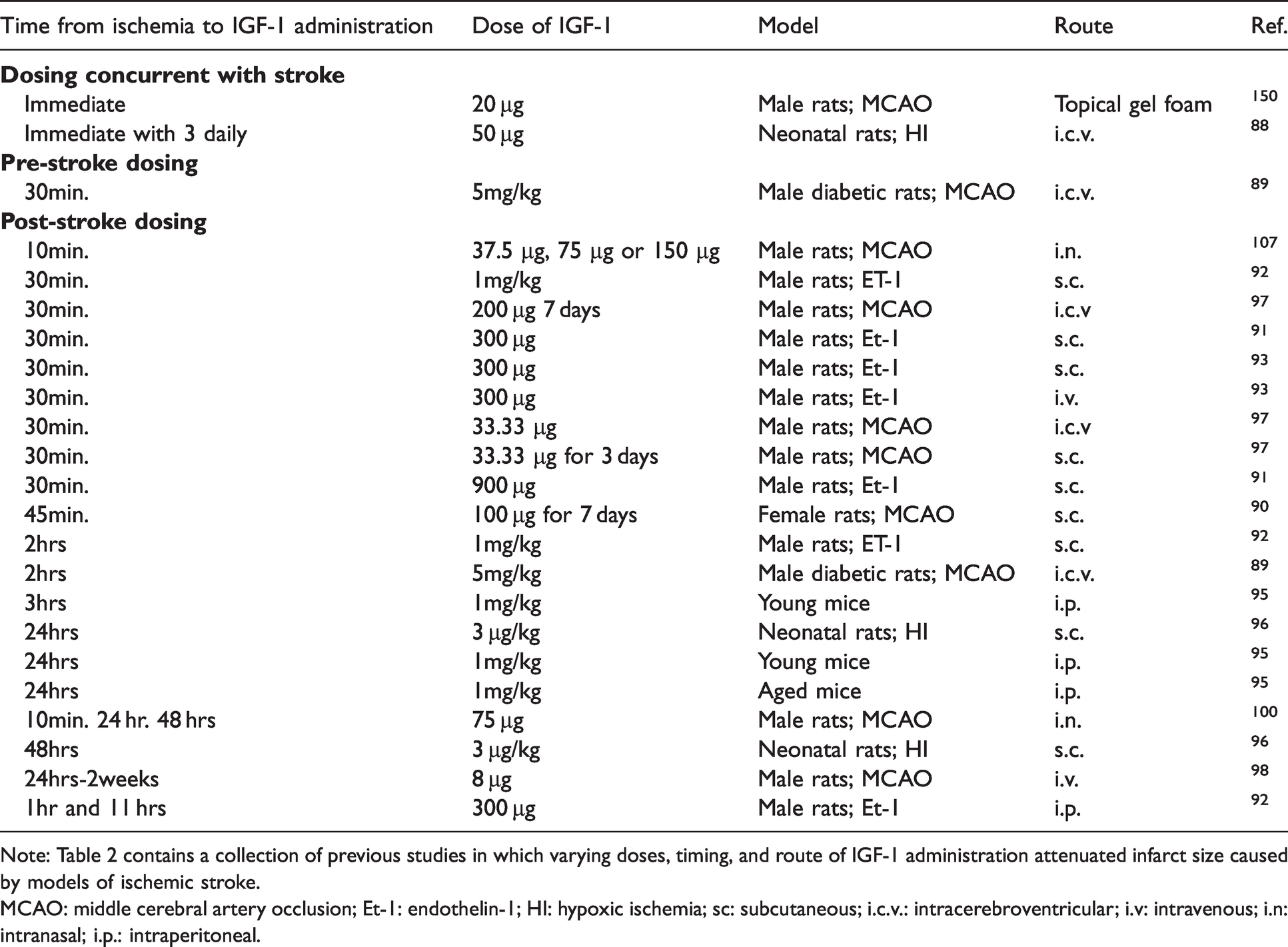
Note: Table 2 contains a collection of previous studies in which varying doses, timing, and route of IGF-1 administration attenuated infarct size caused by models of ischemic stroke.
MCAO: middle cerebral artery occlusion; Et-1: endothelin-1; HI: hypoxic ischemia; sc: subcutaneous; i.c.v.: intracerebroventricular; i.v: intravenous; i.n: intranasal; i.p.: intraperitoneal.
The precise time window for providing the best possible treatment for stroke is not fully understood; however, physicians and scientists agree that the earliest a person receives treatment decreases the odds of permanent and severe damage. Given the difficulties in predicting the occurrence of stroke, post-stroke intervention studies are more clinically relevant for developing effective treatment options. Whether there is an exact time point or route of administration in which IGF-1 would be more effective in reducing stroke damage is not yet evident. I.c.v. administration of IGF-1 beginning at 45minutes after MCAO reduced infarct volume and associated neuroinflammation. 90 Many studies have focused on the 30-minutes treatment time point and have used less invasive techniques for IGF-1 delivery such as subcutaneous injections or intranasal administration. Several studies in rodents have demonstrated the protective effect of IGF-1 in reducing infarct size when administered subcutaneously or intravenously at 30-minutes to 2 hours post-ischemia (Table 2).91–94 Another recent study highlighted post-stroke efficacy using a PEGylated version of IGF-1, which improves stability and other pharmacokinetic parameters of metabolically labile exogenous IGF-1. 95 Significant reductions in infarct volume were noted in young mice when administered 3 hours or even 1 day after MCAO. 95
Repeated administration of exogenous IGF-1 in the hours and days following a stroke has also been shown to be beneficial in reducing infarct damage. In 7-day-old rat pups subjected to HI, 3 µg/kg IGF-1 administered subcutaneously at 24 and 48 hours after insult protected immature rat brains from HI injury. 96 Similarly, administration of 5 mg/kg IGF-1 i.c.v. 24 and 48 hours after MCAO significantly reduced infarct to the same extent as a 2-hours post-MCAO treatment. 89 Daily treatments (3 or 7 days) of IGF-1, as well as every other day treatments (across 14 days) using a variety of routes of administration (sc, i.c.v., iv) also significantly minimized MCAO infarct zone.90,97,98 Whether or not the initial dose which was administered 30–45minutes following the insult is responsible for the change infarct is not discussed, but IGF-1 administration resulted in an attenuation of infarct size. This raises two additional questions. First, do multiple doses of IGF-1 post-stroke increase the likelihood of reducing infarct and neurological damages? Second, in what timeframe following a stroke does the initial dose of IGF-1 need to be administered in order to be beneficial? Addressing these questions will provide a better understanding of the potential use of IGF-1 in stroke. In this regard, it is interesting to note the study by Endres et al in which chronic supplementation of IGF-1 for 24 days before MCAO led to a significant increase in the lesion areas. 86 This result does not align with the predictions from the clinical studies showing that increased levels of circulating IGF-1 reduce the risk of ischemic stroke and improve functional outcomes.25,26,37 This surprising effect draws attention to the need to better understand the roles and regulation of IGF-1 levels in the brain during healthy aging versus an acute insult response.
Among the possible routes of administration of IGF-1, the subcutaneous, i.c.v., and topical gel foam forms present with the difficulty of not knowing the exact amount of IGF-1 that crosses the blood brain barrier (BBB) and reaches the damaged site. Intranasal administration is less invasive and may be a more effective and less invasive technique given the pharmacokinetic benefits of direct access to the brain through the nasal cavities. 99 Indeed, intranasal administration of IGF-1 has shown efficacy in reducing stroke damage when given 10 minutes after MCAO in rats. 100 Further studies are needed to determine the therapeutic time window using this route of administration. Nonetheless, regardless of the route of administration, numerous studies agree that IGF-1 is a beneficial treatment in several animal models of stroke.
Increased IGF-1 signaling has also been noted in ischemic brain tissue in the hours following stroke, 101 particularly in the brains of rodents protected by other effective therapeutic interventions. For example, exogenous GH, which significantly increases local IGF-1 production, reduces neuronal, oligodendritic, and vascular damage following photothrombotic stroke.102,103 Similarly, the protective effects of physical exercise on damage within the peri-infarct area are associated with robust increases in IGF-1 signaling. 104 Moreover, a recent study highlighted that a novel IGF-1R agonist, GLP-1(9-36), reduced infarct volume following MCAO. 105 The correlations between increased IGF-1 signaling and neuroprotection highlight the potential of IGF-1 as a prognostic marker of effective stroke therapies.
IGF-1 improves behavioral deficits caused by stroke in vivo
In addition to examining structural changes in the infarcted as brain described above, a variety of behavioral assays have been conducted to assess the efficacy of IGF-1 in promoting favorable functional outcomes. It is interesting to note that many of the preclinical studies where changes in infarct size are reported do not concurrently report changes in behavior. Rather separate studies of behavioral outcomes are commonly reported. This may be due to the timing of outcome measurements and/or a concern of behavioral analyses may influence tissue damage (physical activity, stress, etc). Nevertheless, there has been several lines of studies have shown that IGF-1 administration prior to or following ischemia can improve sensorimotor deficits and spatial and learning phenotypes
Summary of preclinical studies that observed improved post-stroke behaviors with IGF-1 treatment.
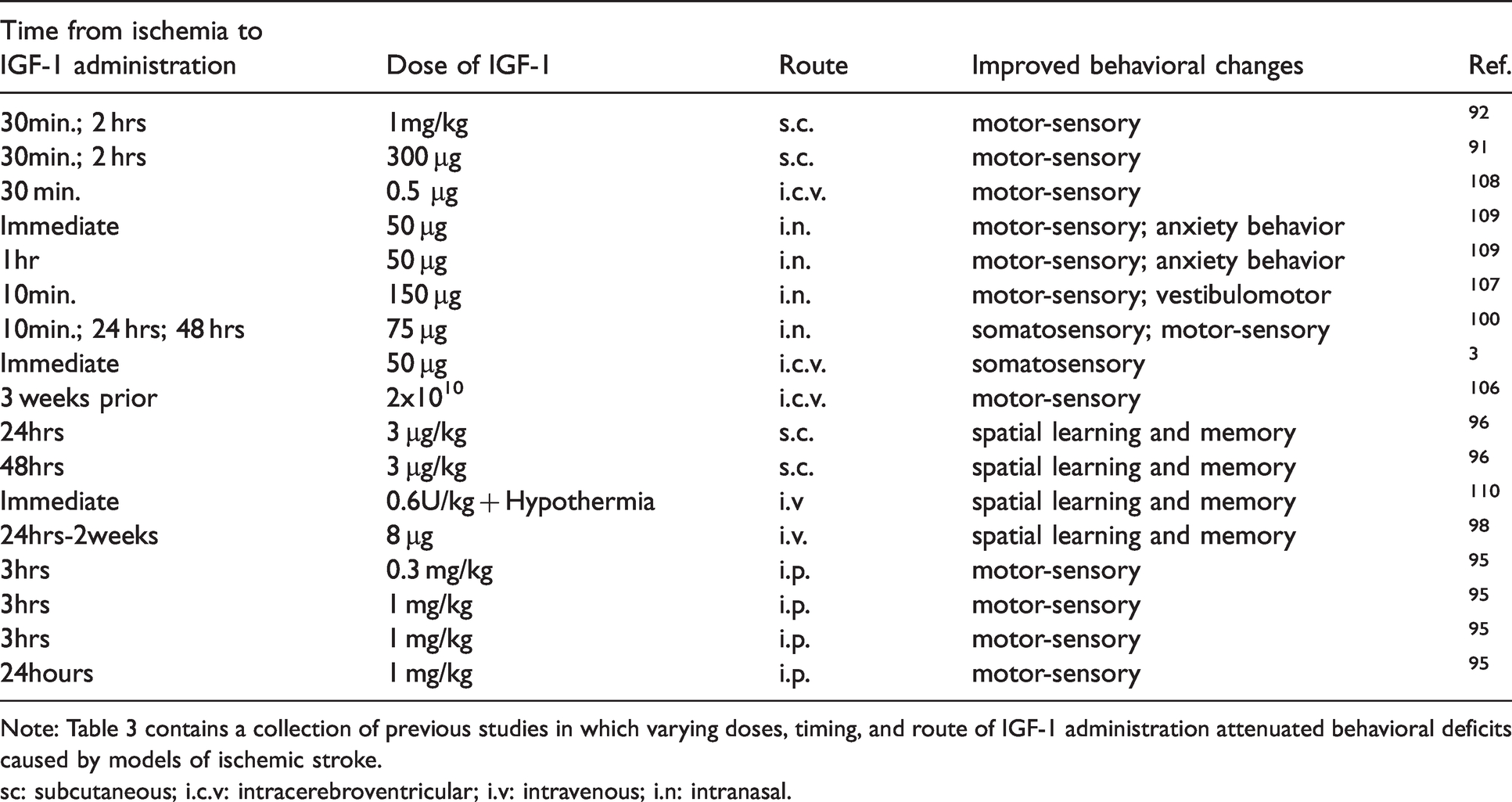
Note: Table 3 contains a collection of previous studies in which varying doses, timing, and route of IGF-1 administration attenuated behavioral deficits caused by models of ischemic stroke.
sc: subcutaneous; i.c.v: intracerebroventricular; i.v: intravenous; i.n: intranasal.
Post-ischemia learning and memory is also enhanced by IGF-1 administration. Immediate IGF-1 treatment at the time of global brain ischemia in rats reduced the latency to escape the radial arm water maze (spatial learning and memory) and improved neurological outcome indexes. 110 This improvement was further enhanced with concomitant hypothermia. 110 Repeated IGF-1 injections beginning 24 hours post stroke resulted in improved cognitive function in the Morris Water Maze. 98 Similarly, 7-day old rats pups treated with 3 µg/kg IGF-1 subcutaneously given 24 and 48 hours post HI showed an improvement in long-term memory and behavior in the Morris Water Maze. 96 These benefits may be age-dependent, as post-ischemia treatment with IGF-1 in the Et-1 rat model improves sensorimotor function in adult rats but not aged rats. 92 Similarly, pegylated-IGF-1 significantly improved motor function in young mice when administered 3 hours or 1 day following MCAO; however, the timing of this improvement was restricted to only the 3-hour post stroke window in aged mice. 95 Together, these data indicate that exogenous IGF-1 is beneficial to both sensorimotor and cognitive performance following ischemic stroke. These benefits are likely mediated by the reductions in brain tissue damage observed with exogenous IGF-1 treatment (reported above).
The extent of protection with IGF-1 treatment may depend on the effector cell type. Interestingly, genetic knockouts of IGF-1 in neurons in the cerebral cortex, hippocampus, and striatum of mice reduces anxiety in open field test, preservers spatial and short-term memory, but does not alter results in Y-maze, O-maze. 111 Whether neuronal IGF-1 in this circumstance is acting on a paracrine or autocrine level is not understood, as neurons and the nearby glial cells as well as cerebrovasculature all express IGF-1R. Understanding the role of each cell type following a stroke is essential to unravel the mechanism by which IGF-1 can protect against neurological and tissue damage following ischemia.
Cell-specific effects of IGF-1 within the ischemic brain
IGF-1 provides protection to neurons
Following ischemia, neurons are highly sensitive to the reduced oxygen availability, hypoglycemia, and the effects of reperfusion as well as secondary insults such as increased inflammation, excess extracellular glutamate, alterations in pH, and generation of ROS. Together, a combination of these primary and secondary insults lead to reduced neuronal survival and viability. The extent of neuronal death is exacerbated by the secretion of the excitatory neurotransmitter glutamate by actively dying cells, a process commonly referred to as excitotoxicity. 112 Primary cultures of pure neurons are highly susceptible to excitotoxic death. IGF-1 is protective against NMDA induced excitotoxicity. 113 Additionally, IGF-1 can protect neurons from inflammatory and hypoglycemic damage.11,114,115 Within the brain, exogenous IGF-1 administration protects neurons in the hippocampus and septum from hypoglycemic damage. 115 In addition to the numerous studies examining the benefits of exogenous IGF-1 administration following ischemia, IGF-1 gene transfer 24 hours post-ischemia promotes neuronal proliferation and enhances neurogenesis. 116 Thus, IGF-1 may not only reduce the extent of excitotoxic death, it may promote the development and maturation of new neurons and glial cells within the damaged tissue.
In inflamed and neurotoxic environments, administering IGF-1 provides protection to neurons in the presence or absence of glial cells, suggesting direct protective actions of IGF-1 on neurons.11,114,117 However, IGF-1 may also support neuronal survival by acting on their surrounding glial cells. This is supported by evidence that IGF-1R blockade specifically on astrocytes co-cultured with neurons causes an attenuation of neuroprotection from oxidative stress. 118 In this scenario, the cell-specific protection may be dependent on the type of insult as IGF-1 does not protect neurons from oxidative stress when astrocytes are not present.118,119 Together, these data indicate that neuro-glial interactions can influence the neuroprotective effects of IGF-1 (Figure 3).
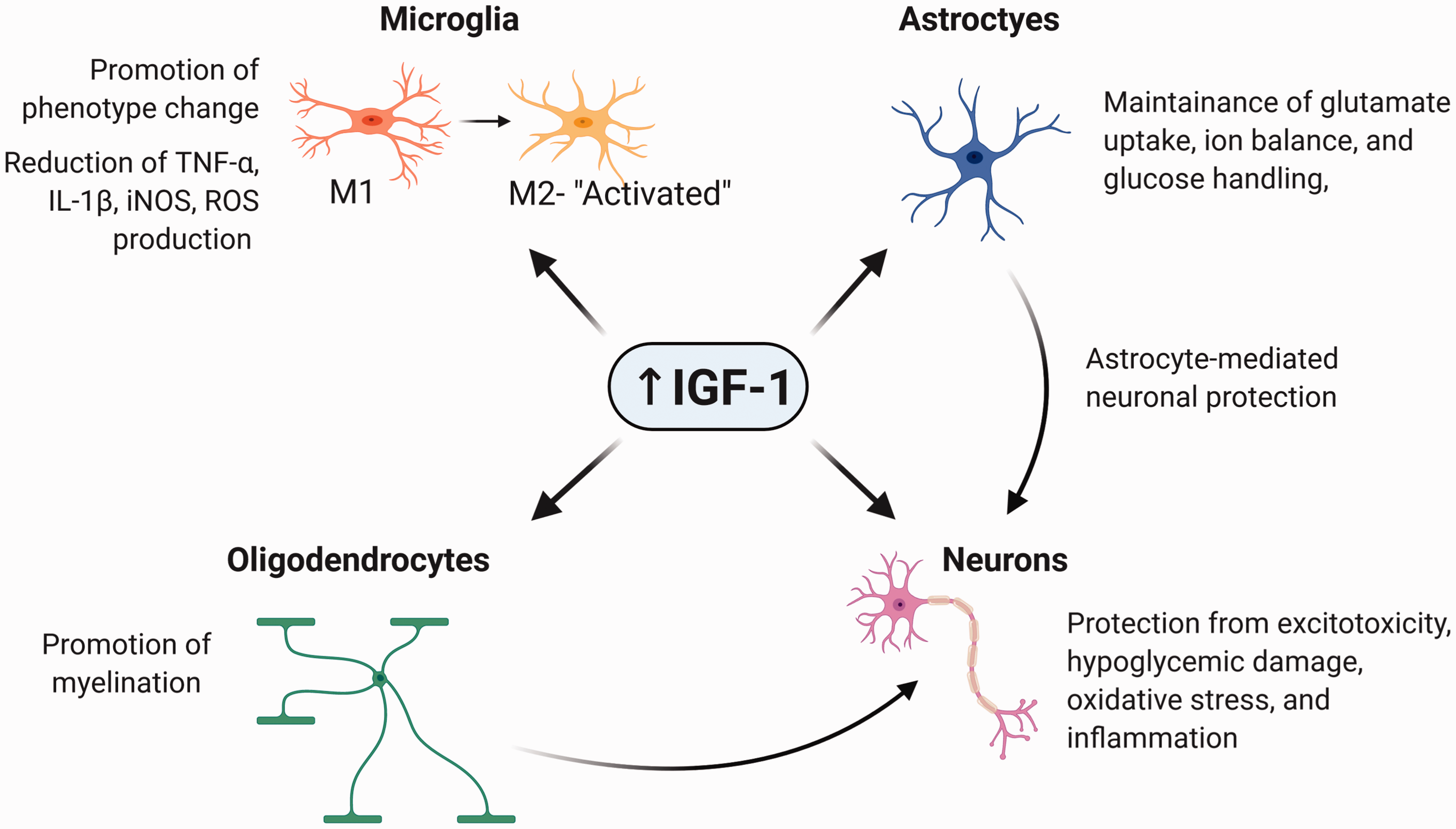
Key cellular functions within the brain known to be regulated by IGF-1.
Potential roles of IGF-1 in astrocytes following a stroke
Astrocytes are more resistant to many of the insults that accompany ischemia, including excitotoxicity, oxidative stress, and ion imbalances. In the ischemic core of in vivo stroke models, the rate of astrocyte survival is significantly higher than neurons.120,121 Whereas neurons in the ischemic-core die within minutes, astrocytes are shown to be preserved up to three hours following ischemia in a rat model. 122 Whether IGF-1 influences the survival of astrocytes in the ischemic brain is unclear, however IGF-1 has shown to increase/preserve astrocyte viability. 118 Astrocytes are however very resistant to insults associated with ischemia such as increase oxidative stress and disruption of ion homeostasis. In vitro studies of oxidative stress show that treating astrocytes with IGF-1 increases survival rate and reduces levels of reactive oxygen species which seems to be mediated by a decrease in inflammatory responses.118,123,124 Protection of IGF-1-treated astrocytes following oxidative stress requires FOXO activity, a transcription factor involved in the response of the brain to oxidative stress. 118 As mentioned, astrocytes serve as critical protectors of neurons from stroke-like insults as robust neuroprotection is apparent when neurons are cocultured with astrocytes versus to primary neuronal cultures without astrocytes present.125,126
When extracellular potassium and glutamate is elevated during cerebral ischemia, astrocytes become reactive and begin to buffer ions and excitotoxins from the environment. 127 Reductions in IGF-1 signaling is associated with impairments in several of these astrocytic protective effects. Prabhu et al showed that reductions in IGF-1 in astrocytes caused a decrease in glutamate uptake. 128 In fact, a significant increase in glutamate release from astrocytes was observed when IGF-1R was inhibited. 128 The loss of glutamate handling when IGF-1 signaling is reduced would increase the likelihood of an excitotoxic environment and ultimately promote tissue damage following stroke. Inhibition of IGF-1 also impairs glucose homeostasis in astrocytes, 129 which would negatively influence neurotransmission and metabolic activity of neurons given their dependence on astrocyte-derived glutamine and lactate. Whether IGF-1 signaling alters other critical functions of astrocytes following ischemic insult is not yet known. However, it is clear that treatment of astrocytes with IGF-1 provides protection from ischemia-related dysfunction, including neuroinflammation, oxidative stress, and disruption of glutamate homeostasis (Figure 3). Specifically targeting astrocytic IGF-1R for therapeutic treatments of ischemia could open novel options for ischemic patients. To date, more effort has been put on understanding the role of astrocyte-derived IGF-1 than astrocytic regulation by IGF-1, as overexpression of IGF-1 specifically in astrocytes confers neuroprotection in vivo.123,130 A better understanding of how IGF-1 regulates astrocytes will provide additional insight on potential therapeutic treatments using IGF-1 signaling in astrocytes.
Differential roles of IGF-1 in oligodendrocytes and microglia role in stroke
IGF-1 promotes phenotypic changes in microglia- the resident immune cells of the brain- thereby altering their activation state (Figure 3). Activated microglia increase the levels of reactive oxygen species (ROS) within brain tissue by secreting cytokines and pro-inflammatory mediators, which ultimately worsen tissue damage. Pronounced microglial activation is observed following ischemia and reperfusion. Administration of exogenous IGF-1 after ischemic or hemorrhagic stroke decreases in microglial activation and the accompanying inflammation and ROS.94,131 Similarly, administration of exogenous IGF-1 directly into the brain reduces the production and release of tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), inducible nitric oxide synthase (iNOS), and ROS within the brain is significantly reduced by IGF-1.51,132 These are hallmark stressors for neurons; thus, it is unsurprising that IGF-1 reduces tissue damage and behavioral impairments given it reduces microglial activation.
Microglia are also a source of local IGF-1 production within the brain. Significant increases in microglial and astrocytic IGF-1 is noted in the lesioned penumbra, as well as the neurogenic region of the subventricular zone following an ischemic insult in rats.133,134 This might represent a compensatory increase in autocrine and paracrine IGF-1 signaling by the glial cells in an attempt to decrease neuronal death and inflammation while promoting neurogenesis.133,135 Regardless of whether this concept holds true, it is clear that there is a cyclical relationship between IGF-1 and microglial function.
Far less is known about how IGF-1 regulates oligodendrocytes after ischemic stroke. During development, IGF-1 promotes myelination which in turn benefits oligodendrocytes ability to cover axons and thus increase neuronal conduction/transmission (Figure 3). Oligodendrocytes are more likely to be affected by homeostatic changes such as energy deprivation, oxidative stress, and hypoxia; therefore, ischemic conditions have a strong impact on oligodendrocytes functions.136–138 It has been shown that cerebral ischemia causes white matter dysfunction, demyelination, and axonal impairment in oligodendrocytes. 136 Therefore, it is possible that promotion of myelination by IGF-1 could protect oligodendrocyte function following ischemia. If this were true, we would again see reduced tissue damage, increased neuronal function, and increased sensorimotor/cognitive function. Further studies on the influence of IGF-1 on oligodendrocyte structure and function following ischemia are warranted.
Potential effects of IGF-1 on stem cell therapy in stroke
As noted, IGF-1 is a critical regulator of neurogenesis, cell proliferation, and cellular differentiation. Thus, recent studies have examined the relationship between IGF-1 and stem cell transplants for a variety of neurological disorders, including stroke. There are numerous reports of IGF-1 promoting neural stem cell development in vitro and in vivo.139,140 Interestingly, mesenchymal stem cells (MSCs), secrete several key growth factors, including IGF-1. 141 Preconditioning of MSCs with hypoxia and oxygen-glucose deprivation leads to significant increases in IGF-1 production and secretion, which is likely an attempt to support cell survival and development when in a stressed environment.142,143 In fact, the transplant of MSCs into ischemic tissue leads to a corresponding increase in IGF-1 levels within the infarcted region. Importantly, this increase in IGF-1 expression and secretion with MSC transplant is accompanied by significant improvements in behavioral function.144,145 The causal role of MSC-derived IGF-1 as a neuroprotector is supported by recent findings that overexpression of IGF-1 in MSCs transplanted into a rodent model of spinal cord injury showed a significant reduction in inflammation, oxidative stress, and neurological deficits.144,145 There are limited studies on IGF-1 as a modulator of stem cell therapies in humans, however, a recent study in a small cohort of chronic stroke patients disclosed that exogenous IGF-1 was used to prep neural stem cells for effective transplant. 146 Additional work in this area is needed to understand whether supplemental IGF-1 can support effective stem cell therapies for individuals in recovery from stroke.
Conclusions and future directions
IGF-1 is a critical protective signal during healthy brain development and after acute neuronal injury. Lower plasma levels of IGF-1 correlate with an increased stroke mortality rate in numerous clinical studies.36–38 In addition, lower IGF-1 levels also correlate with exacerbation of stroke damage and neurological deficits.29,37,42 Although IGF-1 levels can be correlated with essential aspects of assessing a patient’s outcome, it is not the only factor. Certainly, age, comorbidities, race, and living conditions contribute to stroke deficits, outcome, and survival. Nevertheless, it can be concluded that IGF-1 levels could be used as a biomarker in evaluating stroke outcome.
In ischemic stroke patients, more effective therapeutic interventions that can be administered in the hours and days following stroke are still needed. Currently, the most effective treatment for stroke patients is thrombolytic therapy with recombinant tissue plasminogen activator, but not all patients receive thrombolytic therapy due to the narrow time window of effectiveness.147,148 Since IGF-1 levels directly correlate with beneficial outcomes of stroke patients, this opens a possible new therapeutic target for ameliorating stroke deficits. IGF-1 given in a variety of routes (i.c.v., subcutaneously, or intranasally) pre- and post-ischemia attenuates infarct size.86,89,91,93,96,97 IGF-1 also reduces sensorimotor and learning and memory deficits caused by ischemia in a variety of stroke models, routes, and administration time points.100,106,108–110 Again, these benefits in post-stroke outcome are likely attributed to the diminished infarct size. Adding IGF-1 to existing treatments could prove beneficial for stroke patients and improve the outcome of traditional interventions. This concept is of particular interest for continued advances in treating ischemic stroke with stem cell therapies and/or mechanical thrombectomy. In fact, a recent study postulated that concomitant IGF-1 treatment with endovascular thrombectomy may abrogate the local tissue damage while enhancing neuroprotection in the infarcted area. 95 As discussed, there have been a number of studies that highlight the beneficial effects of IGF-1 on stem cell survival and development within preclinical models of ischemic stroke.131,146,149 Additional information related to the specific cellular and molecular mechanisms IGF-1 therapies could target, such as cell-specific reductions in inflammation or ROS generation, is now needed.
Although viral transduction and i.c.v. treatments of IGF-1 allow us to gain insight on the role of IGF-1 following ischemia in animal models, they are not likely to translate to clinical interventions. Less invasive approaches to treating ischemia with IGF-1 are more ideal for clinical populations. Subcutaneous and intranasal administration is far less invasive, however, the pharmacokinetic profiles using these routes of administration require extensive consideration. The amount of bioavailable IGF-1 needed within the core and peri-infarct zone is not clear, nor is the timing. Although many studies use 30-minutes to 2 hours as their therapeutic target window, the rationale for limiting this window is not clearly defined. Nevertheless, the in vivo time targets do correlate with a common amount of time it takes for an individual to receive medical treatment following a stroke.
A majority of in vivo IGF-1 administration studies in animal models thoroughly incorporate a combination of structural and behavioral assessments following post-ischemic interventions. However, the number of studies that do not incorporate sex as a biological variable is striking, especially given the known differences in stroke incidence and prevalence between males and females. Future studies should consider incorporating independent cohorts of male and female animals. Furthermore, predispositions such as diabetes and hypertension should also be included in ischemic rodent models to evaluate the potential role of IGF-1 in decreasing stroke damage in the presence of these common comorbidities. In addition, studies utilizing genetic approaches need to be conducted to evaluate the role of IGF-1 in specific cell types and their role in responding to ischemia. With improvements in health and technology allowing for increased human lifespan, the number of individuals expected to suffer from ischemic stroke will likely increase as our population ages. The potential use of IGF-1 as a new, supplemental therapy that targets specific processes and cell types that contribute to neurological damage is a compelling avenue to improve stroke outcome.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by NIH R15: AG059142. Images were created using Biorender.com.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
