Abstract
Ischemic stroke is a debilitating disease that causes significant brain injury. While restoration of blood flow is critical to salvage the ischemic brain, reperfusion can exacerbate damage by inducing generation of reactive oxygen species (ROS). Recent studies by our group found that non-invasive mitochondrial modulation with near-infrared (NIR) light limits ROS generation following global brain ischemia. NIR interacts with cytochrome c oxidase (COX) to transiently reduce COX activity, attenuate mitochondrial membrane potential hyperpolarization, and thus reduce ROS production. We evaluated a specific combination of COX-inhibitory NIR (750 nm and 950 nm) in a rat stroke model with longitudinal analysis of brain injury using magnetic resonance imaging. Treatment with NIR for 2 h resulted in a 21% reduction in brain injury at 24 h of reperfusion measured by diffusion-weighted imaging (DWI) and a 25% reduction in infarct volume measured by T2-weighted imaging (T2WI) at 7 and 14 days of reperfusion, respectively. Additionally, extended treatment reduced brain injury in the acute phase of brain injury, and 7 and 14 days of reperfusion, demonstrating a >50% reduction in infarction. Our data suggest that mitochondrial modulation with NIR attenuates ischemia–reperfusion injury and evokes a sustained reduction in infarct volume following ischemic stroke.
Introduction
Stroke is one of the leading causes of death and disability in the Western world and accounts for approximately 1 in 20 deaths in the United States. 1 Ischemic stroke occurs due to occlusion of an intracranial artery resulting in rapid and cytotoxic reductions in blood flow to the brain parenchyma. To date, the only approved treatment for ischemic stroke is rapid restoration of blood flow either pharmacologically 2 or surgically. 3 While patient outcomes have improved due to these interventions, a substantial amount of tissue damage occurs during the reperfusion phase. 4 As the ischemic brain is re-oxygenated, ROS are quickly generated, beginning at the early stages of reflow which further induces cell death by way of complex, interacting molecular pathways. While ROS have been implicated to play a key role, to date, pharmacological strategies targeting ROS have proven ineffective. 5 This incongruence is in all likelihood explained by the inability to deliver effective concentrations of compounds to sub-cellular targets at the critical stages of ROS generation during early reperfusion. 6 Interestingly, some promising results were obtained recently by pharmacologically targeting gene expression of antioxidant enzymes as a potential therapy for ischemic stroke. 7
Mitochondria are a source of post-ischemic ROS generation. 8 Hyper-activation of the mitochondrial electron transport chain (ETC) due to changes in regulatory modifications during ischemia promotes hyperpolarization of the mitochondrial membrane potential in the early minutes of reoxygenation and, thus, triggers an exponential increase in mitochondrial ROS production.9,10 A treatment that targets this mechanism during reperfusion could potentially provide a novel, non-pharmacologic strategy to limit ROS generation.
Evidence from our group 11 and others12,13 have identified novel photoreceptive properties of COX, the terminal enzyme in the mitochondrial ETC. The enzymatic activity of COX can be regulated in a wavelength-specific manner using NIR. Previous studies demonstrated that COX activity can be stimulated by applying NIR wavelengths of 670 nm and 808 nm, thus increasing mitochondrial respiration. 12 Indeed, this stimulatory effect was utilized to enhance mitochondrial function during permanent ischemic stroke in animal models 14 but failed to improve outcomes in a clinical stroke study. 15 Data from our laboratory suggest that the aforementioned strategy of increasing COX and thus mitochondrial activity during early reperfusion would exacerbate (rather than reduce) injury. 10 ETC complexes are rendered hyperactive due to cellular stress during ischemia exposure, resulting from changes in their regulatory modifications.16–19 This mitochondrial stress state drives post-ischemic ROS generation in the early phase of reperfusion.9,20
Importantly, our group has also found that COX activity can be reduced by applying specific inhibitory NIR wavelengths (750 nm, 950 nm and the combination). 11 Reduction of COX activity by inhibitory NIR leads to transient, reversible reductions in both mitochondrial respiration, and the mitochondrial membrane potential, and subsequent attenuation of superoxide production. In neuronal culture models, irradiation with inhibitory NIR wavelengths salvages neurons following oxygen glucose deprivation or glutamate exposure. 11 Moreover, we have in vivo evidence that inhibitory NIR, applied during reperfusion, is profoundly neuroprotective in the rat model of global brain ischemia. 11 However, the effect of inhibitory NIR wavelengths in the setting of acute ischemic stroke, which differs in the pathological evolution from global brain ischemia, has not been investigated.
Accordingly, the goal of the current study was to evaluate the efficacy of NIR treatment at limiting brain injury progression after ischemic stroke using the rat middle cerebral artery occlusion (MCAO) model. To increase translational relevance, experiments were conducted in the classic comorbid model displaying spontaneous hypertension. 21 We hypothesized that dual-wavelength inhibitory NIR, applied immediately following resolution of ischemia, would provide neuroprotection measured by multiple, clinically validated MRI modalities that are becoming the gold standard for identifying potentially salvageable brain tissue.22–24 We chose this window of treatment opportunity in order to target the initial ROS burst that occurs early during reperfusion. In a clinical setting, the onset of reperfusion would typically coincide with tPA administration or mechanical thrombectomy. We also hypothesized that NIR therapy would directly target salvageable tissue that succumbs to injury following reperfusion. Brain injury was quantified by delineating infarct volume (using DWI during early reperfusion and T2WI at late reperfusion) and, to increase rigor, incorporating the ‘area at risk’ of infarction (i.e., volume of brain rendered ischemic during MCAO, derived from perfusion weighted images-PWI) as a covariate in the analysis. Of note, this approach has, for decades, been considered compulsory in the analysis of ischemia–reperfusion injury and infarct size in heart. 25 The overarching goal of these studies was to establish whether NIR treatment limits infarct size expansion (reperfusion injury) in the early acute phase following stoke (24 h following restoration of blood flow), and whether this would persist as neuroprotection in late chronic phases of reperfusion (7 and 14 days after ischemia).
Materials and methods
Chemicals, reagents, and diodes
Chemicals and reagents were obtained from Sigma-Aldrich unless otherwise stated. Light emitting diodes (LEDs) were obtained from Roithner Lasertechnik. For animal experiments, LED array 60 chips were used (750 nm, LED750-66-60; 950 nm, LED950-66-60). Diodes were mounted on heat sinks (black aluminum, 47 × 20 for LED array 60 chips) together with a fan (Evercool) operated in reverse mode. Diodes were calibrated with an optical power meter (842-PE) and operated with an energy density of 200 mW/cm2.
Experimental design
Animal use protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at Wayne State University and all experiments were performed in accordance with the Guide for the care and use of Laboratory Animals and conform to the ARRIVE guidelines for reporting of In vivo experiments. Animals underwent 90 min of transient right MCAO as described below. One hour into ischemia PWI was assessed using arterial spin labeling (ASL). After 90 min of focal brain ischemia, reintroduction of blood flow was established, and treatment was initiated and maintained for 120 or 240 min (Protocols 1 and 2, respectively). Following treatment, animals were imaged with PWI and DWI. Twenty-four hours following reperfusion, DWI scans were obtained. On days 7 and 14, the animals were imaged with T2WI.26–28
MCAO/reperfusion
Focal brain ischemia was induced in male spontaneously hypertensive rats (SHR) 21 (223–336 g, Envigo) with the MCAO method as previously described. 29 SHR animals were utilized to introduce a clinically relevant comorbidity to the model as well as to reduce variation in infarct size. 20-Hydroxyeicosatetraenoic acid (20-HETE) has been shown to constrict cerebral arteries and is synthesized more abundantly in the vasculature of SHR animals. 30 This study reported that inhibiting 20-HETE reduced superoxide formation to the same levels as normotensive rats, suggesting that 20-HETE can generate oxidative stress and vascular dysfunction. Furthermore, male animals were utilized in this model to provide a reproducible infarct, omitting changes in estrogen levels in females as another variable. Animals were anesthetized with isoflurane (5% induction, 2% maintenance in a mix of 70% nitrous oxide, 30% oxygen). Surgery was conducted at the same time of day for each animal. Temperature was maintained at 37 ℃ using a homeothermic blanket (Harvard Apparatus). A neck incision was made, and the external carotid was ligated. A silicon coated tip monofilament suture (Doccol Corporation) was inserted in the external carotid and advanced into the internal carotid artery until it occluded the MCA. The filament was secured in place and the rats were placed in a temperature- and humidity-controlled chamber (Tecniplast). Following ischemia animals were re-anesthetized, the filament withdrawn and NIR treatment was immediately initiated. Animals were given subcutaneous buprenorphine (0.015 mg/kg) and recovered in a temperature/humidity-controlled environment. Weight loss exceeding 10% was addressed with enhanced nutritional support (DietGel) and/or subcutaneous administration of 5% dextrose in normal saline.
Administration of NIR treatment
Rats were randomly enrolled in NIR-treatment or untreated groups during MCAO, immediately prior to reperfusion. NIR was administered with combined COX-inhibitory wavelengths of 750 nm and 950 nm as described in our previous studies. 11 Briefly, LEDs (200 mW/cm2) were placed 1.5 cm from the shaved scalp immediately upon filament withdrawal and continued for 120 min (Protocol 1) or 240 min (Protocol 2). During treatment the rats were maintained at 37 ± 0.5 ℃ under isoflurane anesthesia (1.5%). Untreated animals underwent the same procedures (MCAO surgery, anesthesia dose and duration) but were not treated with NIR. Importantly, in optimizing this treatment regime, we have demonstrated that irradiation with the 750 nm and 950 nm at 200 mW/cm2 does not alter brain, rectal, or skin surface temperature in rats. 11
Imaging procedures and analysis
All MRI protocols were performed on a 7.0-Tesla, 20-cm bore superconducting magnet (ClinScan; Bruker, Karlsruhe, Germany) with a Siemens console. Animals were anesthetized with isoflurane. Pulsed ASL (PWI) images were acquired according to the following sequence parameters: Relaxation time (TR) of 3500 ms; and echo time (TE) of 16 ms; field-of-view (FoV) read 35.0 mm; FoV phase 81.3%; distance factor 25%; slice thickness 2.0 mm; 4 slices. DWI sequence was conducted according to the following parameters: TR 10000 ms; TE 50 ms; FoV read 32.0 mm; FoV phase 100.0%; distance factor 0%; slice thickness 0.5 mm; 32 slices. T2WI was acquired according to the following parameters: TR 3530 ms; TE 38 ms; FoV read 32 mm; FoV phase 100.0%; distance factor 0%; slice thickness 1.0 mm; 24 slices. Relative cerebral blood flow in the MCA territory during ischemia and reperfusion was calculated as previously described.31,32 Briefly, voxel intensity in ipsilateral and contralateral hemispheres was averaged across coronal slices of the MCA territory using ImageJ, giving the average relative cerebral blood flow (relCBF) voxel intensity value. The boundary between hypoperfused and physiologically perfused brain tissue was clearly identifiable on the relCBF. Then, using the kinetic model equations, 33 CBF was calculated for both hemispheres giving cerebral blood flow rates in mL/100 g/min. Brains were traced from PWI of each animal during ischemia and following treatment (or matched time-points in untreated controls).24,34 Infarct volumes were computed using the semi-automated segmentation tool in Analyze 11.0 (Biomedical Imaging Resource, Mayo Clinic). Seed points were set in the middle of the hyperintense MCA territory on a single slice and thresholds based on voxel intensity were set by the software until the entire injured region was outlined. The accuracy of the software generated outline was confirmed by a blinded investigator, and then the software applied the threshold parameters to all slices within a sequence. This process provided an automated and unbiased calculation of the area within each slice. All the slices in the brain were summated, and then multiplied by the area thickness to generate infarct volume in mm3.
Statistical analysis
Acquisition and analysis of all MRI images were conducted in a blinded manner, without knowledge of the treatment group. Group sizes were determined by power analysis (GPower3.1). Statistical analysis was performed using GraphPad Prism 7. Brain injury volume in the acute phase, chronic phase, and relative cerebral blood flow was compared using two-factor ANOVA (for group and time) with replication, with Neuman–Keuls multiple comparisons test for post hoc analysis with significance at p < 0.05. Slopes and intercepts of the DWI/PWI linear regression lines were compared using analysis of covariance (ANCOVA) with significance at p < 0.05.
Results
Protocol 1: NIR treatment for 2 h following MCAO
Measurements of relative cerebral blood flow during ischemia and reperfusion
During ischemia (prior to randomization), all animals showed reductions in blood flow below established relCBF thresholds in the ipsilateral hemisphere (Figure 1).
35
Following the 2-h NIR treatment window, restoration of blood flow to the ipsilateral hemisphere was confirmed and relCBF was statistically equivalent to the contralateral hemisphere. There was no significant difference in relCBF in within the ipsilateral hemispheres between control and NIR-treated animals either during ischemia or reperfusion.
Relative blood flow during ischemia and following reperfusion. (a) Experimental protocol for pulsed arterial spin labeling (PWI) imaging. (b) Representative PWI images during ischemia and from both groups following reperfusion indicate reduction of blood flow in the ipsilateral hemisphere during ischemia and restoration of blood flow during reperfusion. (c) Averaged regional cerebral blood flow in the ipsilateral MCA territory and contralateral hemisphere both during ischemia and following reperfusion (n = 7–8/group).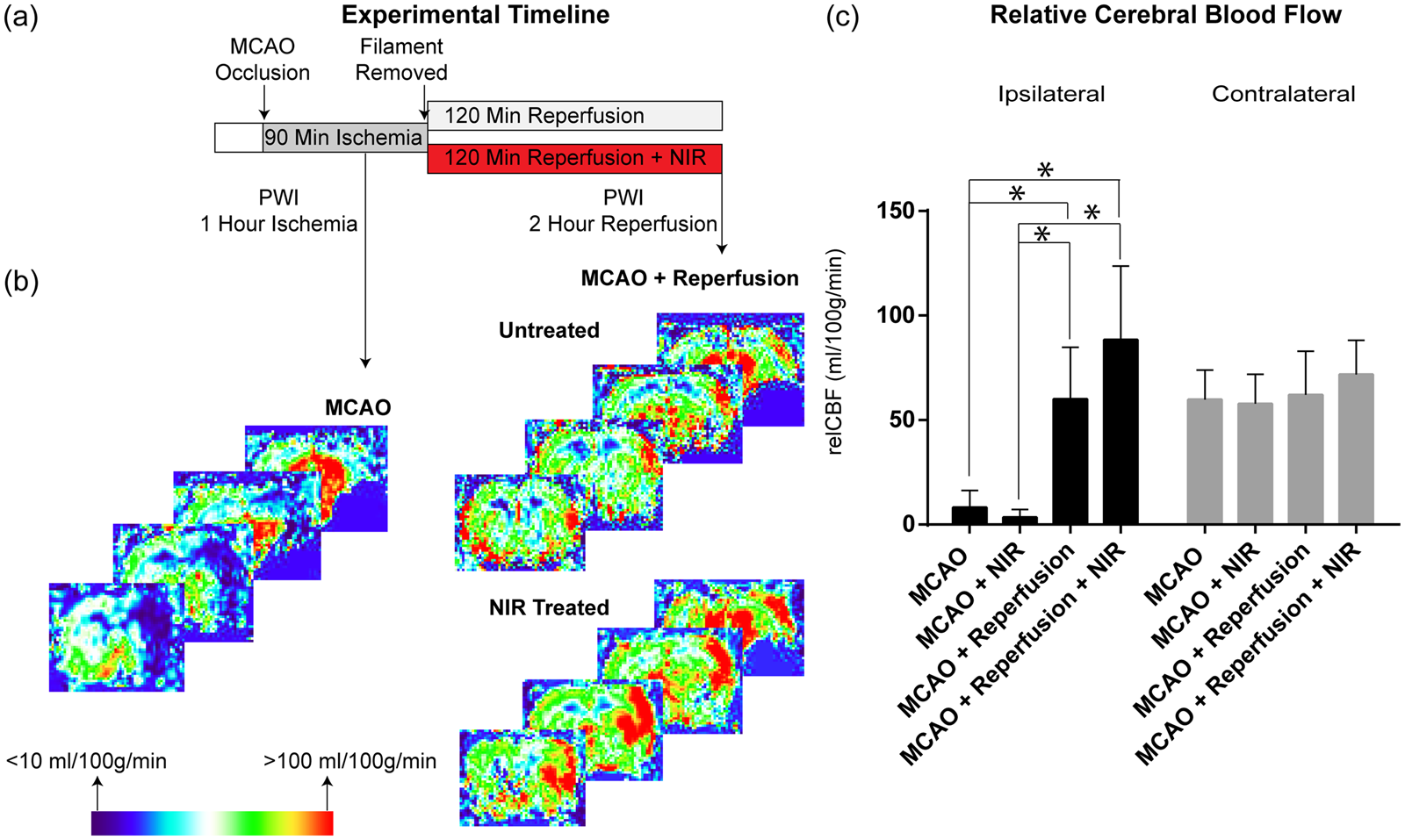
Brain injury in the acute phase of stroke
The effect of NIR on cerebral injury in the acute post-ischemia phase (i.e., less than 24 hours after reperfusion – Figure 2(a)) was assessed using DWI, considered to be the most effective imaging modality for quantification of early cytotoxic edema and prediction of final infarct size.22,27 Figure 2(b) shows that DWI hyperintensity volumes immediately following treatment (2 h of reperfusion) did not significantly differ between NIR-treated and control groups (210 vs. 256 mm3, respectively, p > 0.05). At 24 h following reperfusion, NIR-treated animals showed a significant reduction of 21% in DWI hyperintensity volume when compared with untreated controls: 317 vs. 399 mm3, p < 0.05 (Figure 2(b)). Repeated measures ANOVA revealed an overall significant increase in DWI-hyperintensity at 24 vs. 2 h of reperfusion and a significant difference between NIR-treated and untreated groups at 24 h of reperfusion. These data suggest that the infarct continues to expand between 2- and 24-h post-reperfusion and NIR-treatment limits this expansion.
Analysis of cerebral injury in the acute reperfusion phase. (a) Experimental design for measuring early phase outcomes. (b) Diffusion-weighted imaging (DWI) images were obtained and no significant difference in infarct volume was seen between NIR treated and control animals after 2 h of treatment. At 24 h, compared with untreated controls, NIR-treated MCAO rats showed significant 21% reduction of infarct volume. (c) At-risk area analyzed by PWI at 1-h ischemia (x-axis) and DWI hyperintensity volume (y-axis) 2 h and 24 h following reperfusion, indicating that the regression relationship between infarct volume and area at-risk for NIR-treated animals was below the relationship observed in controls.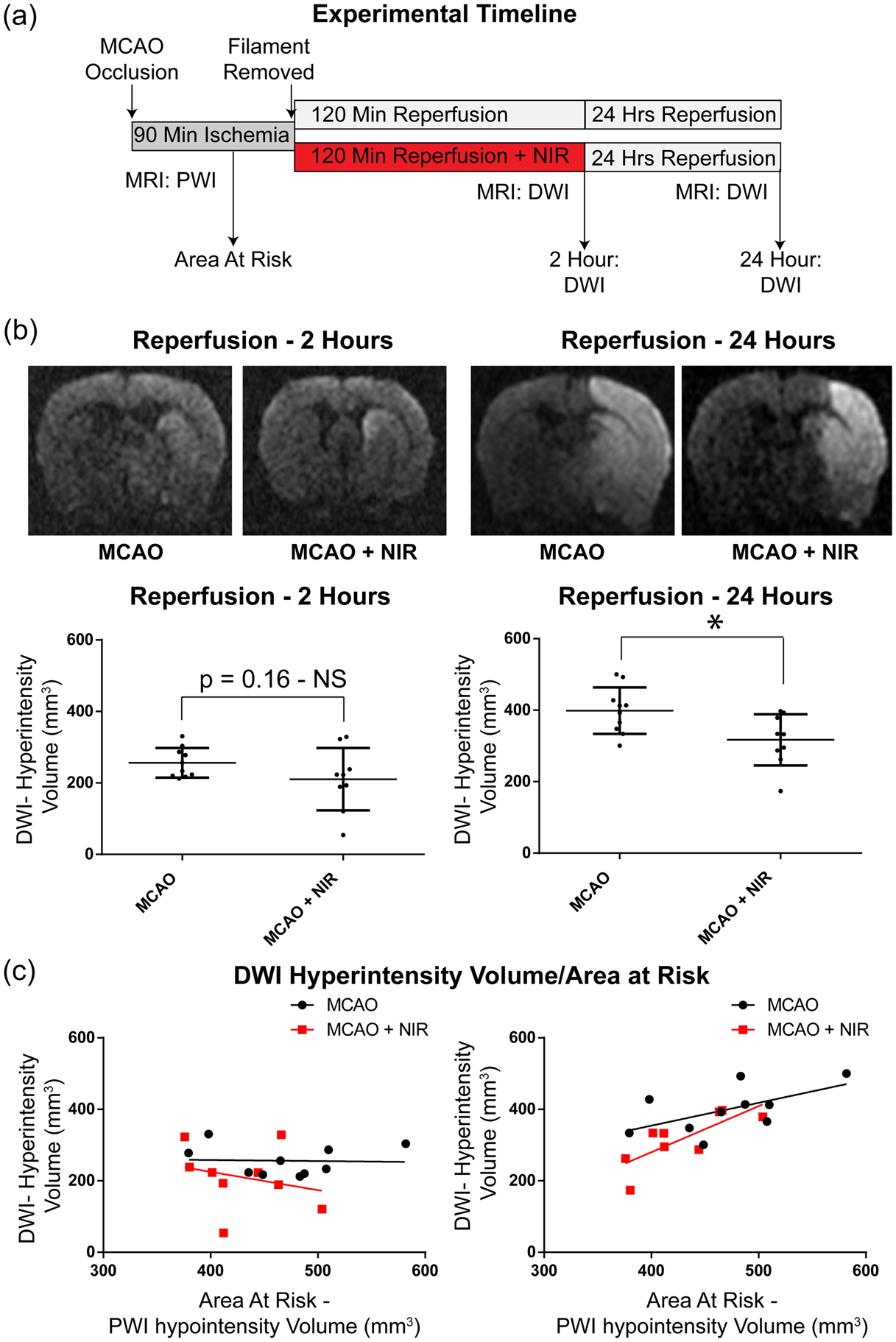
These concepts are supported by ANCOVA, incorporating ‘area at risk’ (volume of brain rendered ischemic during MCAO, delineated by the volume of flow deficit identified by PWI) as a covariate in the analysis of infarct size. Immediately following treatment at 2-h reperfusion, the volume of DWI hyperintensity occupied only ∼50% of the area at risk in both groups, and ANCOVA revealed no difference in the regression relationship between DWI hyperintensity volume and area at risk between cohorts (p > 0.05), suggesting no measurable treatment effect at this early timepoint. In contrast, after 24 h of reperfusion, the volume of at-risk brain that displayed DWI hyperintensity increased to 85% in the control cohort and 73% in the NIR-treated group, and the regression relationship between infarct volume and area at risk for NIR-treated group fell below the relationship observed in controls (*p < 0.05; Figure 2(c)).
Infarct volume in the chronic phase of stroke
Assessment of candidate therapies in rat models of stroke traditionally utilizes early infarct analysis at 24 h following MCAO.36,37 However, multiple studies have demonstrated early reductions in infarct volume are often not predictive of final brain injury at later stages of injury progression and thus early signs of efficacy may not indicate meaningful, lasting neuroprotection.38,39 Accordingly, to address this issue, animals were maintained for an additional two weeks and the therapeutic benefits of NIR were also evaluated with T2WI at 7 and 14 days following MCAO-reperfusion (Figure 3(a)). Seven days following MCAO, NIR-treated animals maintained a significant 25% reduction in infarct size when compared to untreated controls (271 vs. 363 mm3, *p < 0.05) that persisted at 14 days after MCAO (241 vs. 317 mm3, *p < 0.05, Figure 3(b)), with no significant change over time. That is: infarction had fully evolved by 7 days of reperfusion and neuroprotection persisted at the final time-point of 14 days post-stroke. The volume of hyperintensity (infarct), expressed as a % of the at-risk volume of brain, averaged 64% vs. 73% in the NIR-treated vs. control groups at 7 days, and 57% vs. 69%, respectively, at 14 days post-reperfusion. These conclusions were, again, supported by ANCOVA at both time-points, the regression relationship between infarct volume and risk region for NIR-treated rats fell below the relationship observed in controls (*p < 0.05; Figure 3(c)).
Cerebral infarct in chronic phase of post-stroke reperfusion injury. (a) Experimental design at 7- and 14-days following reperfusion. (b) representative T2-weighted imaging (T2WI)-images of MCAO vs. MCAO with NIR treatment. Compared with untreated controls, NIR-treated MCAO rats showed a significant 25% reduction in infarct volume. Significant reduction in infarct volume persisted at 14 days post stroke. (c) Regression relationship of PWI (area at risk) vs. infarct volume in MCAO vs. MCAO treated with NIR, showing that the regression relationship between infarct volume and risk region for NIR-treated animals was below the relationship observed in controls.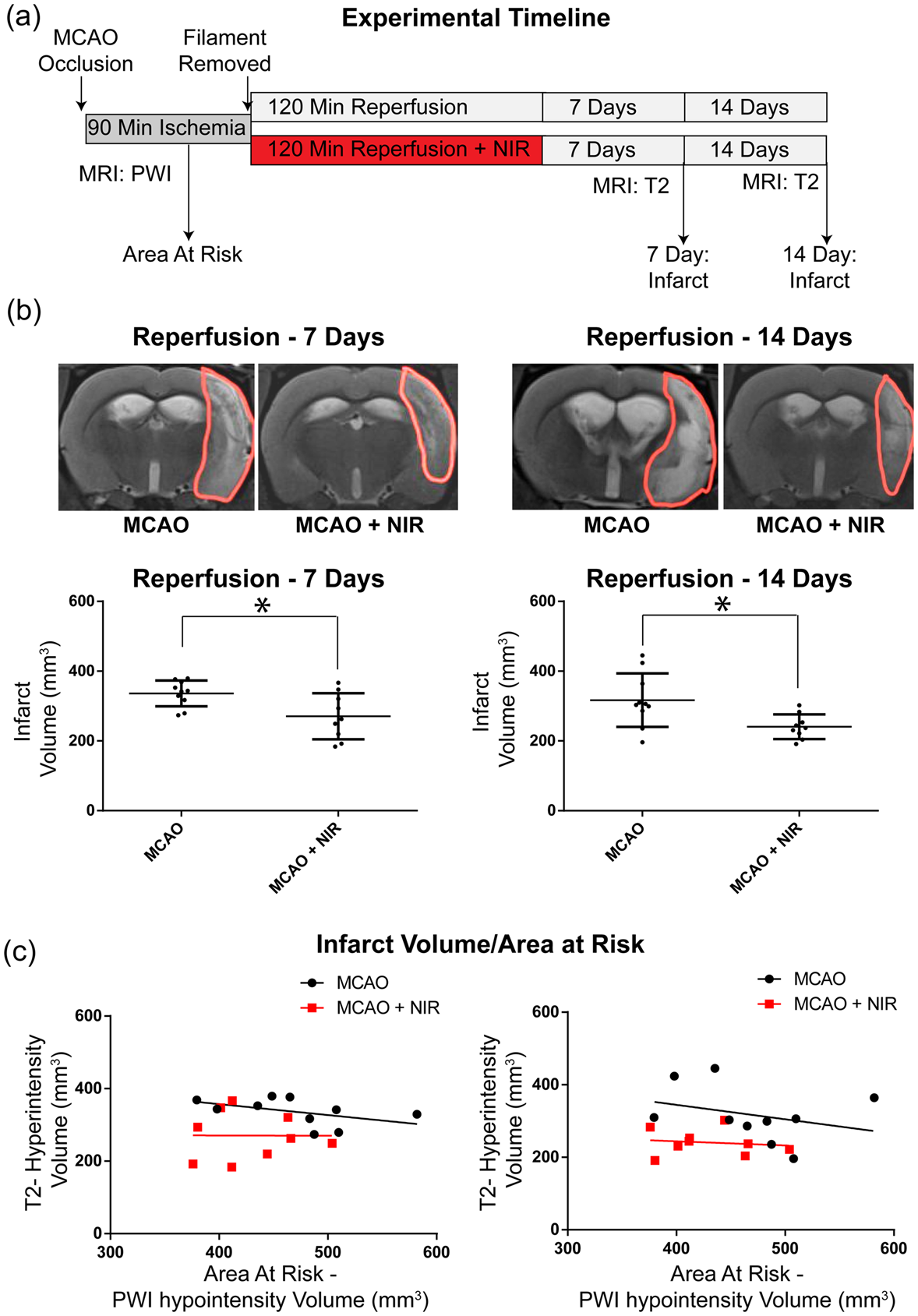
Protocol 2: Extended treatment: NIR therapy for 4 h following ischemia
Relative cerebral blood flow
During ischemia (before randomization), mean relCBF in the ischemic hemisphere was reduced to 10.94 mL/100 g/min, while the contralateral hemisphere maintained physiological flow rates of 50.78 mL/100 g/min. Following resolution of ischemia, blood flow significantly increased with no significant difference between animals receiving NIR treatment versus untreated controls during ischemia or reperfusion (Figure 4(b), *p < 0.05).
Relative cerebral blood flow during ischemia and following a 4-h NIR treatment. (a) Experimental design. (b) Representative PWI images during ischemia and from both groups following reperfusion indicate restoration of blood flow. (c) Averaged regional relative cerebral blood flow in the ipsilateral and contralateral MCA territories during ischemia and following 4 h of reperfusion (n = 6–7/group).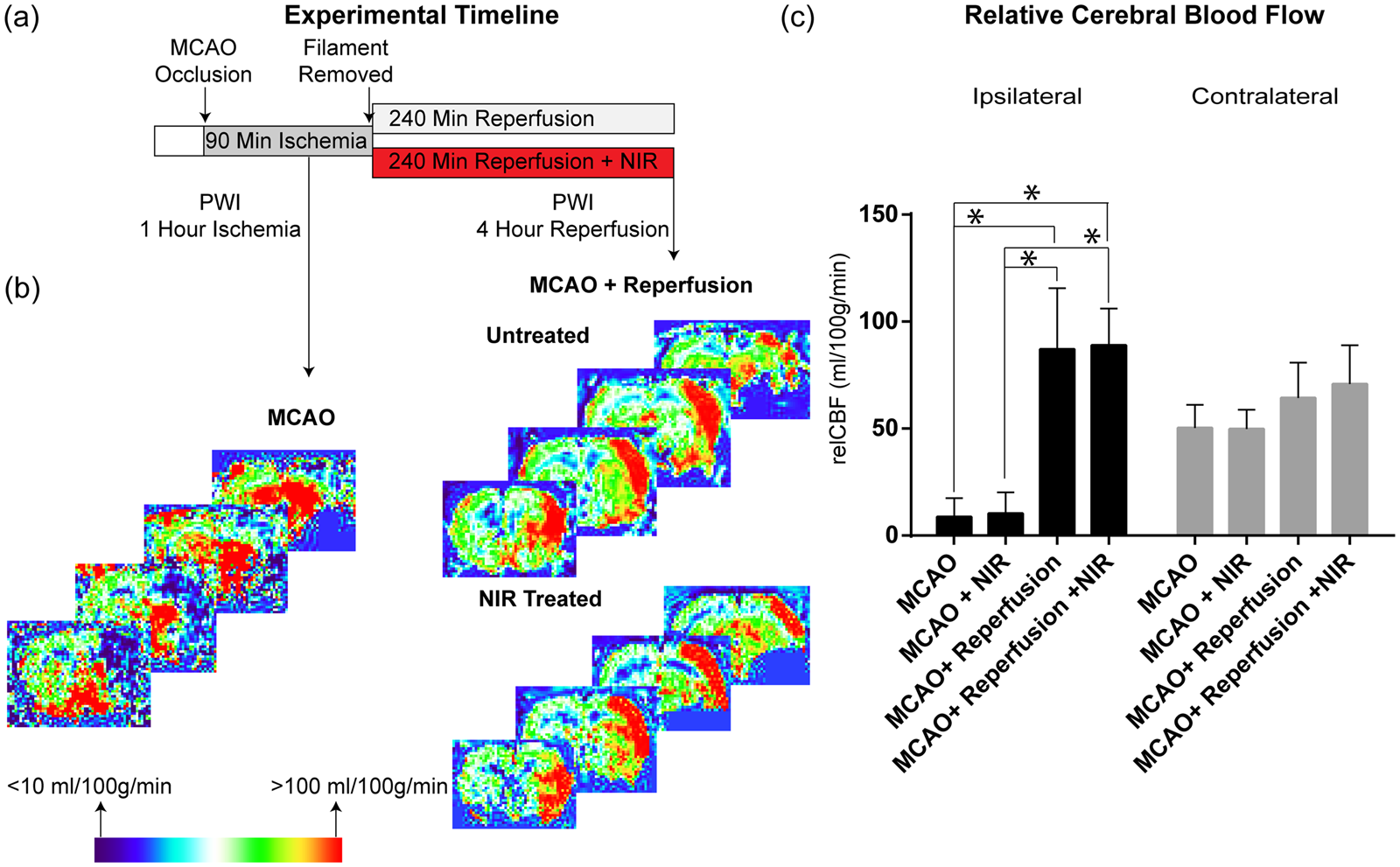
Brain injury in the acute phase of stroke
The efficacy of extended 4-h treatment was assessed using the analytical approach described in Protocol 1. Early indices of brain injury were quantified with DWI after the 4-h treatment period (4-h of reperfusion) and at 24 h after reperfusion (Figure 5(a)). In contrast to Protocol 1, there is a significant difference in infarct size between control and NIR-treated groups immediately at the end of treatment (228 vs. 138 mm3, Figure 5(b), p < 0.05), that was maintained at 24 h post-treatment despite the expected, temporal expansion of the infarcts (351 vs. 266 mm3, *p < 0.05). These data were supported by ANCOVA (Figure 5(c)) and, taken together, suggest that even in the early stage of brain damage (4 and 24 h of reperfusion) NIR therapy limited the progression of brain injury.
Infarct volumes in the acute phase of stroke with NIR for 4 h. (a) Experimental design. (b) Representative images of DWI 4 h following reperfusion in NIR treated vs. control, indicating a significant reduction in infarct size of the NIR-treated animals compared to the controls at the end of the 4-h treatment, which was maintained at 24 h post-treatment. (c) At-risk area during ischemia (PWI) vs. area of infarction (DWI) at 4 h following reperfusion.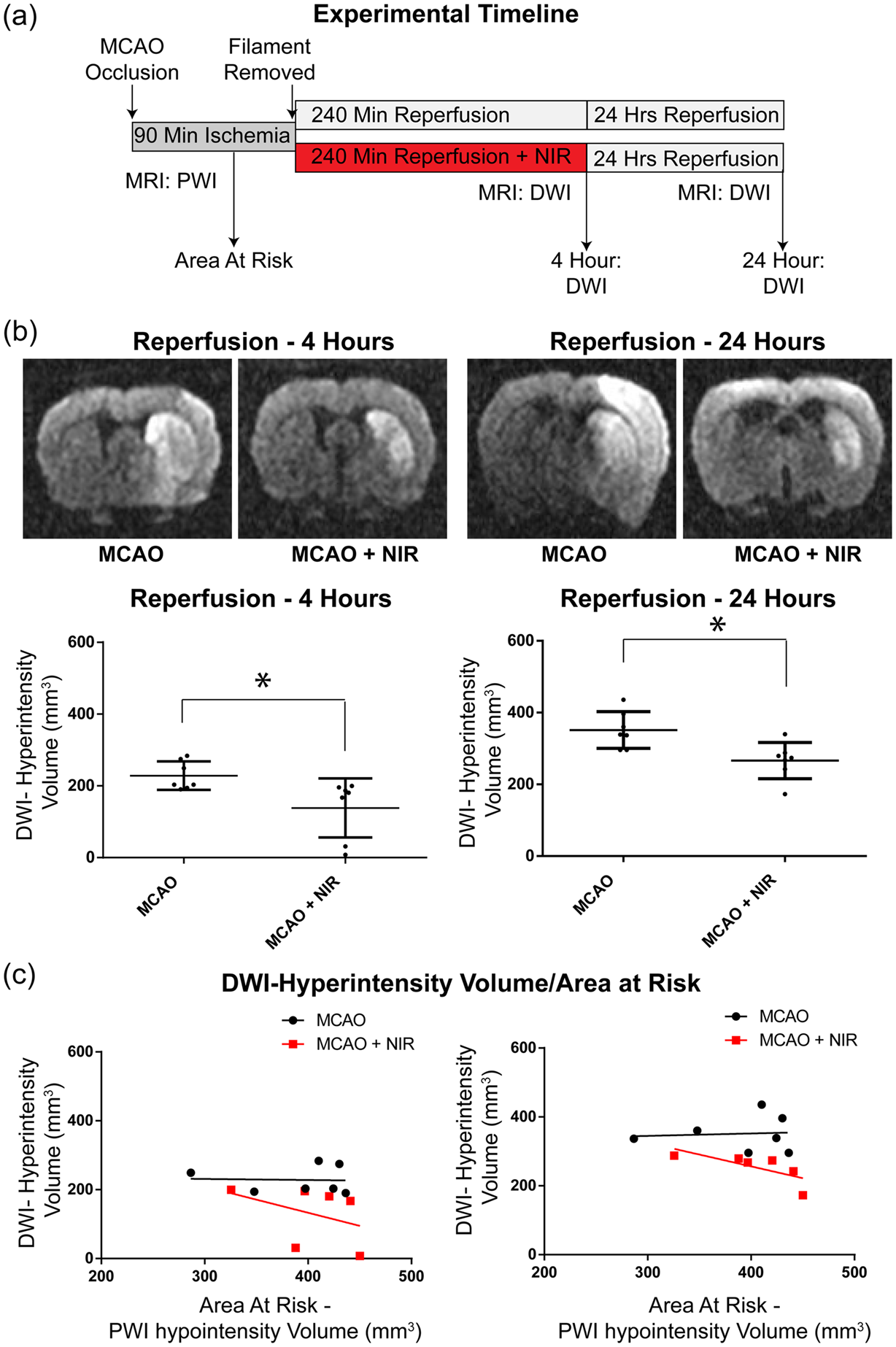
Infarct volume in the chronic phase of stroke
As in Protocol 1, we next sought to establish whether the 4-h NIR treatment paradigm evoked sustained neuroprotection in the chronic stage of reperfusion injury (Figure 6). Using T2WI, control and NIR-treated groups were imaged seven days following reperfusion (Figure 6(b)). Mean T2 hyperintensities were reduced by 52% at both 7- and 14-days post-reperfusion (Figure 6(b)), suggesting that infarction is fully evolved by 7 days of reperfusion and a positive treatment effect remains at the final time-point of 14 days post-reperfusion. The relationship between cerebral infarction measured with T2WI with tissue at-risk for infarction revealed a significant difference (ANCOVA) in regression line elevation between control and NIR-treated groups and ANOVA of the DWI/PWI ratio found a significant reduction in this ratio between control and NIR-treated groups at both 7 and 14 days of reperfusion (Figure 6(c)).
T2WI of infarct volumes during the chronic phase of stroke. (a) Experimental design. (b) Representative images of NIR treated rats showed a significant 52% reduction in infarct volume at 7- and 14-days following ischemia vs. untreated controls. (c) At-risk area vs. area of infarction at 7- and 14-days post reperfusion show significant reduction with NIR-treatment.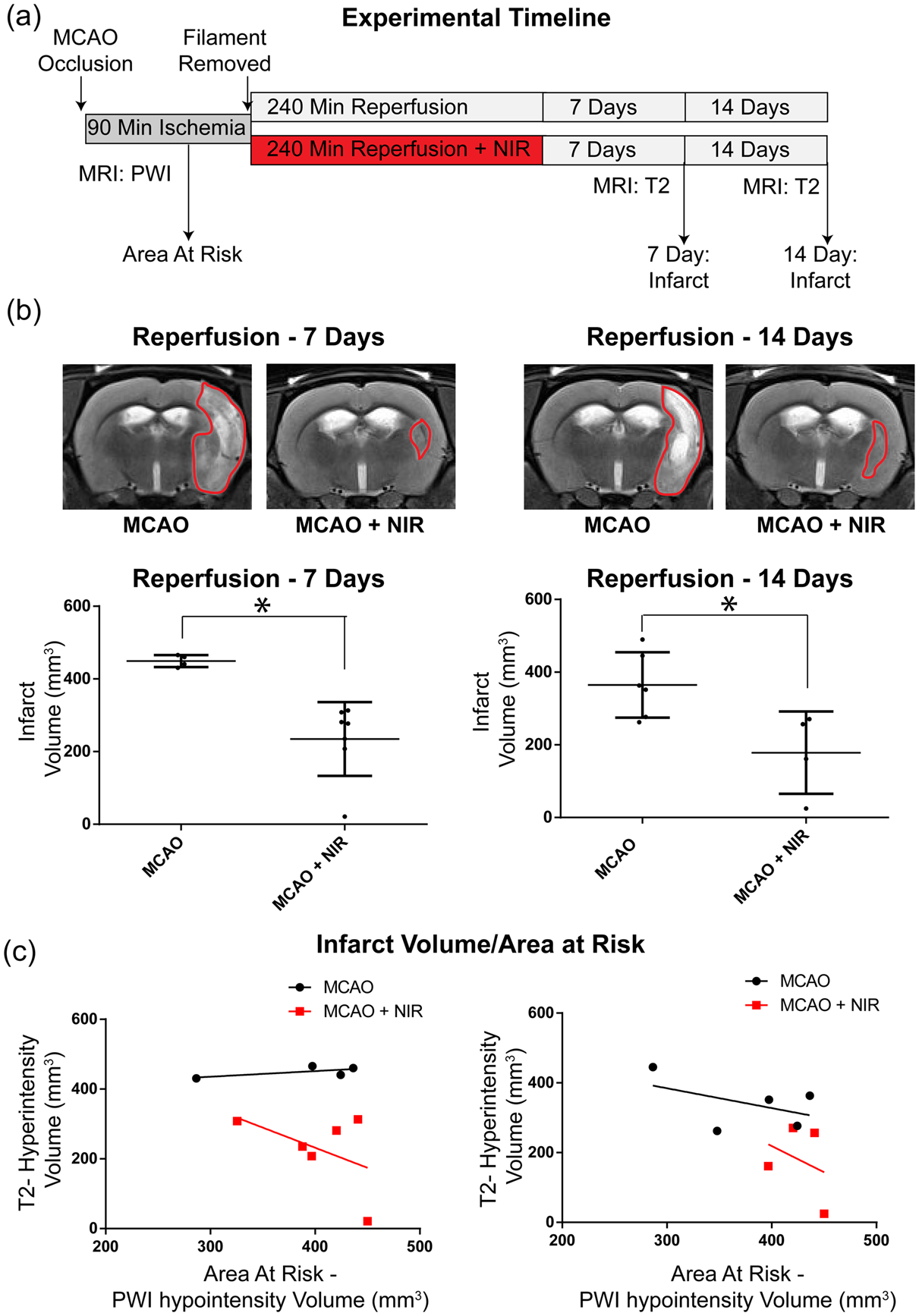
Discussion
Ischemic stroke accounts for about 9% of deaths globally and is the second leading cause of death following heart disease. Those who survive this debilitating disease often face a lifetime of neurological deficits and hence a drastic reduction in quality of life, including cognitive, emotional and functional impairments. The gold standard for stroke treatment is rapid restoration of blood flow. However, timely reperfusion is associated with a perpetuation of brain injury, due in part to the production of ROS during early reoxygenation. Oxidative stress is a common pathological mechanism in many disease states and arises from an imbalance between ROS production and ROS scavenging. Once produced, ROS damage cells either directly, or through diverse and complex cell signaling cascades. 40
In our previous studies, we published a theoretical framework10,41,42 and experimental data11,17,18 to support the concept that ROS production is exponentially related to mitochondrial membrane potential. Additional evidence for this concept can also be found in the role played by uncoupling proteins (UCPs) in rendering the brain more impervious to ROS-induced reperfusion injury by reducing membrane potential. 43 Membrane potential is subject to fluctuations in both substrate availability, and, in higher organisms, post-translational modifications such as changes in the phosphorylation state of ETC enzymes. 9 We argued that enzymatic dephosphorylation during ischemia leads to priming for hyperactivation which, upon reperfusion, results in bursts of ROS.10,19
In this study, we sought to evaluate the effectiveness of targeting ROS at their source: directly in the mitochondrial ETC during early reperfusion using NIR therapy. Building on our previous work, we reasoned that this therapy is noninvasive, can be applied immediately at the onset of reperfusion, and has been shown to have sufficient penetrating depth to target brain tissue during reperfusion. We hypothesized that irradiation of the brain with NIR immediately upon reperfusion would reduce infarct size both acutely and chronically following stroke.
Our data indicate that cerebral blood flow was significantly reduced below ischemic thresholds in the ipsilateral hemisphere and returns to normal levels following reperfusion with no treatment effect. Interestingly, infarct volumes were not different between NIR-treated and control animals immediately following 120-min NIR-treatment. However, animals treated for 240 min showed immediate, significant reductions in infarct volume when compared to untreated controls. Importantly, reductions in infarct volume were manifest in both Protocols 1 and 2 at 24-h following reperfusion.
While these data imply that the penumbra was being salvaged by NIR therapy, we needed to confirm this by establishing whether a significant mismatch existed between brain injury and perfusion deficit. When we plotted infarct volumes against area at risk, we observed, in both Protocol 1 and Protocol 2, that for all groups the relationship between area at risk and area of infarction is linear, i.e., as might be expected, infarct size is proportional to the area at risk. In addition, and most importantly, there was a significant downward shift in the regression relationship (with no difference in slope) for the NIR-treated cohorts versus controls. This finding allows us to conclude that over the full range of risk regions, the penumbra was salvaged with NIR treatment, resulting in significantly smaller infarct volumes.
We next asked whether the benefit of NIR could be extended into the chronic phase of stroke. Our data show that infarct volumes remained significantly smaller at 7- and 14-days post stroke with NIR in both Protocols – outcomes that were confirmed by ANCOVA. Interesting, in Protocol 2, the paradigm of prolonged NIR-treatment was associated with a ∼50% reduction in infarct volume, indicating that the extended treatment duration significantly improved neurological outcome. Additional targeting of late-stage ROS production caused by excitotoxicity, inhibition of inflammatory cell conversion through ROS signaling as well as a possible, longer-than-predicted window of mitochondrial stress and other, unknown mechanisms may potentially explain how the longer treatment duration had such profound reductions in infarct volume.
A limitation to this study is its applicability to all clinical stroke presentations. Not all stroke patients are candidates for thrombolytics or mechanical thrombectomy 44 and thus manifest a permanent focal ischemic pathology, in which ROS production differs from transient ischemia due to the lack of reperfusion. Another limitation is the lack of neurobehavioral data corroborating our imaging findings.
In conclusion, this is, to our knowledge, the first study to demonstrate that non-invasive partial inhibition of mitochondrial COX by dual-wavelength NIR treatment following stroke is accompanied by profound and sustained reductions in cerebral infarction. Furthermore, these reductions in infarct volume were measured using clinically relevant MRI modalities, which allowed assessment of stroke evolution over time to be monitored in the intact animal. Thus, based on our previous work demonstrating that NIR treatment limits mitochondrial ROS production when applied during reperfusion after global brain ischemia, we here demonstrate that such treatment reduces the progression of reperfusion injury following stroke. Furthermore, this work has shown that NIR can target more than the initial burst of ROS following reperfusion. Extending treatment duration had profound beneficial effects on infarct volume. As a result, this data has shifted the paradigm on the role that NIR can play at later stages of the reperfusion injury process. Future studies are needed to examine the effect of NIR on neuro-inflammation, and other potential long-duration targets of NIR in brain ischemia/reperfusion pathologies.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants R01NS091242 and R42NS105238, by the Office of the Assistant Secretary of Defense for Health Affairs through the Peer Reviewed Medical Research Program-Award No. W81XWH-16-1-0175, and the Wayne State University Cardiovascular Research Institute Pilot Grant Program. Opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the funding agencies including the Department of Defense or the National Institutes of Health.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: THS and MH are co-founders of Mitovation Inc. that develops infrared light therapy for ischemia/reperfusion injury applications. All other authors declare that they have no conflicts of interest with the contents of this article.
Authors' contributions
CS THS and MH conceived the study, and JW, CR, PM, and KP assisted with study design. CS, THS, JW, IL, and MM performed and/or analyzed the in vivo brain ischemia experiments. THS, CS, and MH wrote the manuscript, assisted by JW and KP.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
