Abstract
The generalization of perfusion-based, anterior circulation large vessel occlusion selection criteria to posterior circulation stroke is not straightforward due to physiologic delay, which we posit produces physiologic prolongation of the posterior circulation perfusion time-to-maximum (Tmax). To assess normative Tmax distributions, patients undergoing CTA/CTP for suspected ischemic stroke between 1/2018-3/2019 were retrospectively identified. Subjects with any cerebrovascular stenoses, or with follow-up MRI or final clinical diagnosis of stroke were excluded. Posterior circulation anatomic variations were identified. CTP were processed in RAPID and segmented in a custom pipeline permitting manually-enforced arterial input function (AIF) and perfusion estimations constrained to pre-specified vascular territories. Seventy-one subjects (mean 64 ± 19 years) met inclusion. Median Tmax was significantly greater in the cerebellar hemispheres (right: 3.0 s, left: 2.9 s) and PCA territories (right: 2.9 s; left: 3.3 s) than in the anterior circulation (right: 2.4 s; left: 2.3 s, p < 0.001). Fetal PCA disposition eliminated ipsilateral PCA Tmax delays (p = 0.012). Median territorial Tmax was significantly lower with basilar versus any anterior circulation AIF for all vascular territories (p < 0.001). Significant baseline delays in posterior circulation Tmax are observed even without steno-occlusive disease and vary with anatomic variation and AIF selection. The potential for overestimation of at-risk volumes in the posterior circulation merits caution in future trials.
Introduction
Perfusion-based patient selection in acute ischemic stroke (AIS) is supported by expert consensus, and guides late-time window therapy.1–6 Endovascular trials have thus far emphasized treatment in anterior circulation, predominately ICA and proximal MCA, large vessel occlusion (LVO). While anterior circulation LVO accounts for the majority of AIS, about one-fifth of AIS occur in the posterior circulation, 7 often presenting with non-specific symptoms and with high risks for adverse outcomes and early recurrent stroke.7–9
The extension of commonly used, threshold-based perfusion selection criteria from anterior circulation LVO trials, however, may not be straightforward due to: i. physiologically normal, relative hemodynamic delays in anatomic territories of the posterior circulation; ii. the propensity for anatomical variation in the posterior circulation (e.g. shared origins of vertebral and basilar artery branches, symmetric or asymmetric fetal disposition of the posterior cerebral arteries, etc.) 10 ; and iii. the tendency for asymmetric dominance of the vertebral arteries, most commonly on the left, which may interact with (or counteract) the preceding variables. 11 Any such factors may influence delay-sensitive hemodynamic parameters such as the time-to-maximum (Tmax) of the tissue residue function, commonly used for estimation of at-risk (i.e. penumbral) tissue volumes using dynamic bolus passage CT or MR perfusion.9,12–14 Importantly, despite the widespread adoption of delay-insensitive deconvolution algorithms, which mitigate cerebral blood flow (CBF) errors relating to arterial input function (AIF) selection, the inherent arrival time sensitivity of tissue Tmax would be anticipated to vary under the influence of AIF selection, for example between the anterior and posterior circulations.15–17
Whole-brain volumetric CTP techniques are now commonly available from most major vendors and are facilitated by extended detector geometry or multi-station shuttle techniques.18,19 Accordingly, perfusion imaging through the posterior cerebrovascular circulation is now achievable and could allow for investigation into perfusion-based selection criteria in posterior circulation LVO. A thorough understanding of the normative distribution of posterior circulation tracer kinetics is prerequisite to such applications yet remains unmet.
This study aims to establish the distribution of Tmax in the posterior circulation of patients with no macroscopic steno-occlusive disease by CT angiography (CTA), and with specific attention to common anatomic variations anticipated to influence macrovascular arrival times. The impact of varying AIF selection throughout the intracranial circulation upon posterior circulation Tmax was investigated, positing that generally later posterior circulation AIF may be more temporally suited to tracer kinetic features of posterior circulation territories, and hence may mitigate baseline Tmax delay. A retrospective analysis of consecutive patients with negative CTA and CTP obtained for clinically suspected AIS was undertaken, hypothesizing that slower posterior circulation flow produces inherent delays in Tmax, despite normal arteriography and blood flow, and with measurable effects relating to the degree of contribution from the anterior circulation such as from the posterior communicating artery. We anticipate that conventional Tmax thresholds (e.g. Tmax > 6s) prescribed for identification of at-risk tissue volumes in anterior circulation AIS may be susceptible to false positive imputation of critical hypoperfusion in the posterior circulation, and that normal anatomic variation and AIF selection may contribute significantly to Tmax elevation in this setting.1,2,20–22
Materials and methods
Patient selection
A New York University Langone Medical Center Institutional Review Board approved (s17-01842) retrospective clinical and radiologic informatics query was performed for identification of suspected AIS patients undergoing CTP of the head and concurrent CTA of the head and neck at our comprehensive stroke center between 1/2018 and 3/2019. The need for written, informed consent was waived due to the retrospective nature of the study. The research was conducted in accordance with the Helsinki Declaration of 1975 (and as revised in 1983). The electronic medical record was reviewed for patient clinical and demographic information, including age, sex, presenting symptoms, NIH stroke scale at presentation, additional imaging performed, and final hospital diagnosis, including follow up imaging (CT or MRI) confirming the absence of acute ischemic injury. Subjects were excluded if CTP or CTA could not be confidently interpreted due to technical limitations including: those arising from patient motion; bolus truncation or timing errors; metallic or other attenuation artifacts (including severe streak and beam hardening arising from the occiput or temporal bones which could contaminate tissue time-attenuation courses or basilar AIF selection as detailed below); interrupted/incomplete examinations; insufficient craniocaudal coverage for characterization of the entirety of the posterior circulation; significant underlying parenchymal pathology (e.g. tumors or encephalomalacia and ex vacuo changes such as from past infarctions); or significant structural anomalies such as developmental, surgical, or post-traumatic etiologies. Subjects were further excluded if CTA exhibited any visible cervical or intracranial steno-occlusive disease, including either calcific or non-calcific arteriopathy in the anterior or posterior circulation or any visible narrowing in the anterior or posterior circulation by NASCET criteria. 23 Subjects were similarly discarded if follow-up MRI or final clinical diagnosis documented any acute or chronic infarctions.
Imaging protocol
All CT examinations were performed on a Siemens SOMATOM Force whole body clinical CT scanner. Perfusion imaging was obtained with a volumetric whole brain protocol (kV 70, mAs 180, dose-length product 3078.2 mGy*cm) using standardized dosing of intravenous iodinated contrast (Iodixanol 320 mg/mL [GE Healthcare], 50 ml) power injected at 5 ml/s and followed with 50 ml normal saline flush at the same injection rate. CTP were acquired in 48×1.2 mm slices reconstructed to 10 mm slices in helical shuttle mode covering two stations (∼11.4 cm total craniocaudal coverage), and across 47 equally spaced temporal volumes at 1.5 s intervals following a 2 s delay (∼71 s total) ensuring fully captured dynamic bolus passage in accordance with recent recommendations.24,25 CTP images were auto-forwarded to a dedicated network node running a fully user- and CT vendor-independent analysis platform (RAPID, iSchemaView, Menlo Park, CA) for production of CTP parametric maps and lesion segmentation as described elsewhere including infarction core estimated at relative cerebral blood flow (rCBF) less than 30% normal and total hypoperfusion estimated at >6 s time-to-maximum of the deconvolved residue function. 20
CTA was acquired using standard dosing (70 ml Iodixanol 320 mg/mL) with bolus triggering using inline vendor tools based upon carotid enhancement.
CTP post-processing: AIF selection
CTP scans were processed using RAPID to generate Tmax perfusion maps. Automated AIF detection was first employed using an operator-independent algorithm to identify high-likelihood arterial voxels with optimal tracer kinetic features including earliest, greatest, and fastest upslope. The AIF was deconvolved from the tissue time course using a delay-independent deconvolution as previously elaborated.15,20 To assess the influence of varying AIF upon territorial Tmax, we subsequently manually enforced AIF placement throughout the anterior and posterior circulations, selecting pre-defined regions of interest exhibiting the earliest, fastest rising, and highest peaking time-attenuation features on the right and left MCA or carotid terminus, the ACA, and basilar artery. All manual AIF placement was performed by a neuroradiology fellow (post-graduate year 6) under the supervision of a neuroradiologist with subspecialty certification (11 years of experience in advanced stroke imaging). AIF were specifically selected through arterial segments of maximal cross-sectional area to minimize partial volume averaging with surrounding tissues and structures which could spuriously blunt AIF features. Vertebral artery and posterior cerebral artery AIF, as well as smaller basilar arterial penetrating branches, were not selected because consistent regions-of-interest (ROI) free of partial-volume averaging effects could not be ensured in the majority of subjects. When automated (default) AIF were selected by RAPID in the ICA or MCA, a contralateral anterior circulation ROI was also selected, mirroring the index ROI as closely as possible. For subjects in which the ACA or basilar artery were selected by the automated AIF detection module, bilateral ROI were additionally placed within mirrored distal ICA or proximal M1 segments, again optimizing AIF characteristics.
CTP post-processing: Segmentation
Tmax parametric outputs for all AIF, and in all subjects, were segmented into pre-defined vascular territories and subsequently the average values of Tmax in these regions were computed in the following steps: First, a CT-based anatomic template representing an average human head was elastically fitted to the individual subject CTP images using the initial phases of the CTP before contrast bolus arrival with rigid, affine, and non-rigid registration (Image Toolkit, KitWare, Clifton Park, NY; and elastix non-rigid registration library, University Medical Center Utrecht, Netherlands). Secondarily, the derived co-registration parameters and deformation field were applied to a vascular territory atlas (originating in the coordinate space of the anatomical template) to identify vascular regions in the subject dataset space (sub-segmented to the left and right sides of the brain, except for the brainstem). In the final anatomical region masks, voxels representing cerebrospinal fluid (CSF) or bone were removed based on thresholds applied to the subject data HU density, and this step was manually checked case by case for correctness. Lastly, the Tmax values in voxels of interest were analyzed and average Tmax values from the input Tmax images were computed in each vascular region independently. Anterior circulation territories (ACA and MCA) were computed and averaged as aggregated anatomic regions.
CTA evaluation
CT of the head and CTA of the head and neck were reviewed initially by a neuroradiology fellow (post-graduate year 6) who screened the formal interpretation provided in routine clinical practice for each patient to identify angiographically negative studies, and subsequently under the supervision of a neuroradiologist with subspecialty certification (11 years of experience in advanced stroke imaging). The cervical and intracranial circulation of each subject was assessed for the presence of anatomic variation including the following features: 1. Presence of fetal PCA disposition (i.e. posterior communicating artery larger then P1 posterior cerebral artery (PCA) segment); 2. Presence of a complexed/shared origin of the anterior inferior (AICA) and posterior inferior (PICA) cerebellar arteries or superior cerebellar arteries (SCA); 3. Vertebral artery dominance (unilateral larger vertebral artery) or co-dominance; 4. The presence of any other primitive carotid-basilar or carotid-vertebral anastomoses (i.e. persistent trigeminal arteries) which could further modulate posterior circulation arrival times.
Statistical analysis
Statistical analysis was performed using Stata version 12.1 (StataCorp, College Station, TX). Categorical variables are reported as proportions. Continuous variables were expressed as mean and standard deviation when normally distributed and median and interquartile range when not. Continuous variables that were not normally distributed were compared by the Mann–Whitney, Wilcoxon signed-rank test, or Kruskal–Wallis test, when appropriate. Tmax values from different anatomic and vascular territories were treated as dependent variables for within-subject comparisons. For each vascular territory of interest, a voxel-wise average Tmax was computed for each individual patient. These patient-level territorial averages were then compared between subjects. As these values were not normally distributed within the study population, non-parametric statistical methods were applied. A two-tailed p-value <0.05 was considered for statistical significance.
Results
Subject characteristics
Of the 262 CTP obtained for suspected AIS during the study period, 71 were included in the final analysis. A total of 191 subjects were excluded, including 78 subjects with CTP suggesting any acute infarction core (rCBF < 30%), 49 subjects with acute lacunar infarction on follow-up MRI undetected by CT, CTA, or CTP, 61 with any cervical or intracranial steno-occlusive disease on contemporaneous CTA, and 3 with CTP degraded by motion sufficient to preclude analysis based upon quality control criteria in RAPID. Subject characteristics are summarized in Table 1. Mean cohort age was 64 ± 19 years, while 55% of subjects were male and 45% were female. Median NIHSS at presentation was 4 (IQR 2-12), and the most common presenting symptom was weakness (51%), followed by unspecified encephalopathy (35%). Six subjects received tissue plasminogen activator (tPA) before imaging, although none showed any steno-occlusive lesions by CTA or evidence for AIS based on follow-up imaging or final clinical disposition. The most common diagnoses on discharge were toxic/metabolic encephalopathy (35%) and seizures (15%).
Clinical characteristics (n = 71).
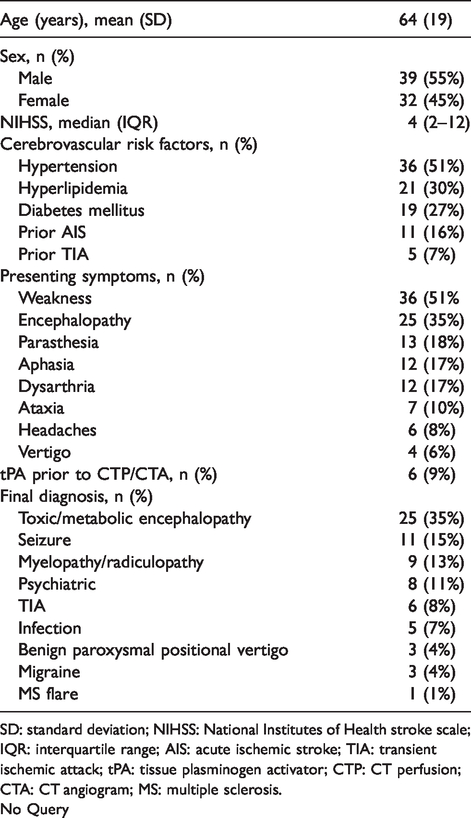
SD: standard deviation; NIHSS: National Institutes of Health stroke scale; IQR: interquartile range; AIS: acute ischemic stroke; TIA: transient ischemic attack; tPA: tissue plasminogen activator; CTP: CT perfusion; CTA: CT angiogram; MS: multiple sclerosis.
No Query
Tmax variation by vascular territory
When assessed based on vascular/anatomic territory and using automated AIF selection, average Tmax values were significantly delayed in the cerebellum and PCA territories compared with the anterior circulation territories (p < 0.001, Wilcoxon signed-rank test for the right and left cerebellum and right and left PCA compared with either the brainstem or right and left anterior circulation median territorial Tmax values, Figure 1). Specifically, the median of the voxel-wise territory average Tmax values measured 3.0 s (IQR 2.7 s–3.8s) and 2.9 s (IQR 2.6–3.7) in the right and left cerebellar hemispheres, respectively, and 2.9 s (IQR 2.6 s–2.9s) and 3.3 s (2.6 s–3.5s) in the right and left PCA territories, respectively, which were significantly higher than the median Tmax delays of 2.4 s (IQR 2.1–3.2) measured in the brainstem and 2.4 s (IQR 2.1 s–2.9s) and 2.3 s (IQR 2.0 s–2.8s) in the right and left anterior circulation (p < 0.001).
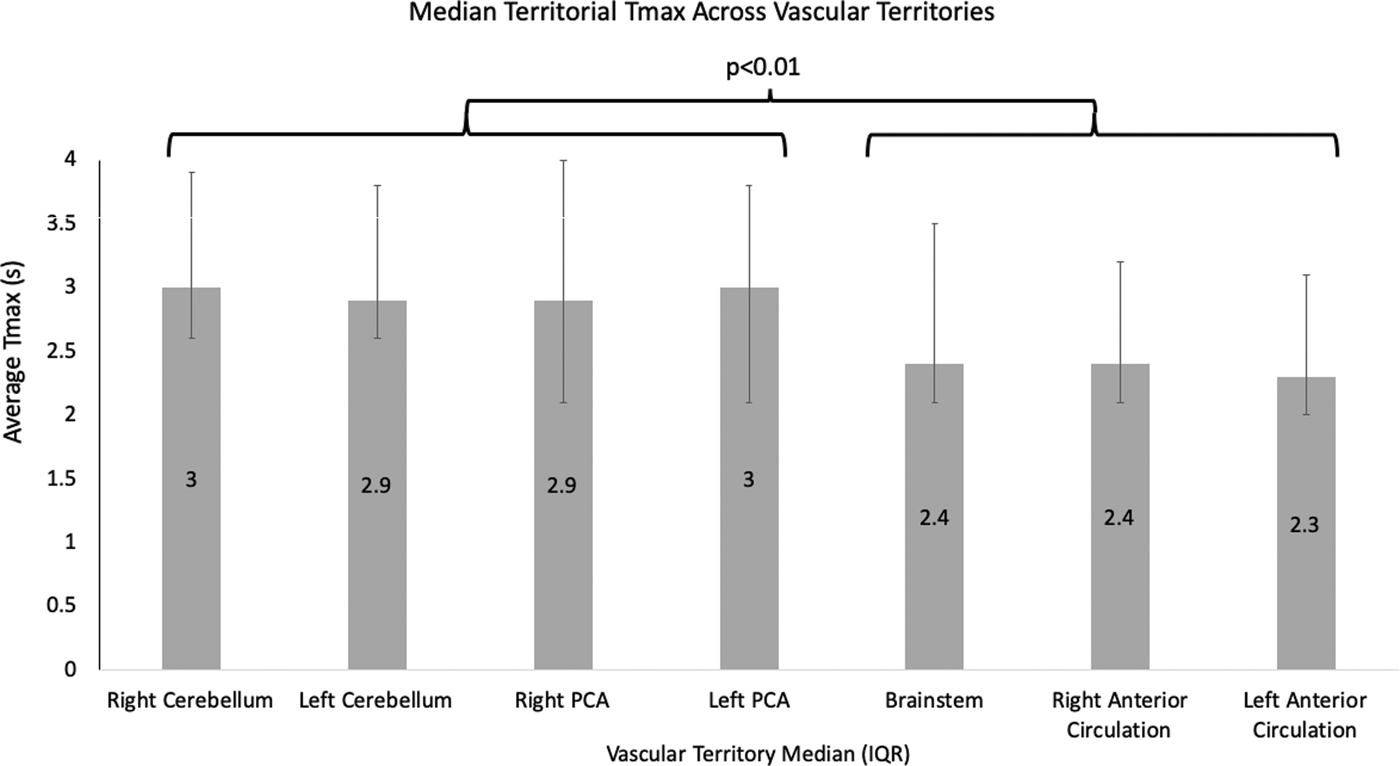
Comparison of median territorial Tmax values between pre-defined vascular territories. Median Tmax is significantly elevated in the right and left cerebellar hemispheres and right and left posterior cerebral artery (PCA) territories compared with the brainstem and right and left anterior circulation. Brainstem delays may more closely approximate anterior circulation delays relating to the shorter and more direct course of perforator vessel perfusion in the brain stem by comparison to the longer and/or more circuitous path of the AICA, PICA, SCA, or PCA territories.
Subjects with final diagnosis of seizure (n = 11) demonstrated higher Tmax values in the cerebellar hemispheres than patients without seizure diagnosis (p < 0.05), although without significant differences in the PCA territories and anterior circulation or brainstem (p > 0.05). For instance, the median Tmax in the right and left cerebellar hemispheres of subjects with seizures were 3.5 s (IQR 3.0 s–2.4s) and 3.6 s (2.8–4.5), respectively, and 2.9 s (IQR 2.6 s–3.8s) and 2.8 s (IQR 2.6 s–3.7s) in those without seizures, respectively (p = 0.015 and p = 0.034).
Variability of tmax by AIF placement
The effects of AIF placement between the anterior circulation vessels (ICA, MCA, or ACA) and basilar artery were tested, to assess the influence of varying AIF placement within subjects, positing that macrovascular delay sensitivity in Tmax would be potentiated in the posterior circulation territories when an anterior circulation AIF is selected, or conversely, that manually enforced posterior circulation AIF would mitigate the magnitude of macrovascular delay in those territories. Within the cohort, RAPID automatically selected AIF on the left MCA/ICA in 24 subjects, basilar in 21 subjects, right MCA/ICA in 16 subjects, and ACA in 10 subjects. The cerebellar hemispheres and brainstem vascular territories exhibited greater delays when an anterior circulation automated AIF was selected by comparison to an AIF in the posterior circulation (Figure 2). Greatest effects were observed in the right cerebellar territory, wherein median Tmax measured 3.2 s (IQR 2.8 s–3.9s) with AIF selection on the left MCA/carotid terminus and 3.5 s (IQR 2.9 s–4.1s) with AIF on the right MCA/carotid terminus, compared with 2.8 s (IQR 2.3 s–3.1s) with AIF on the basilar artery (p = 0.01 for Kruskal-Wallis tests, with post-hoc Mann-Whitney p < 0.05 for MCA vs basilar arteries). The general trend for greater Tmax values in all posterior circulation territories using anterior circulation failed to reach statistical significance in some territories (Figure 2).
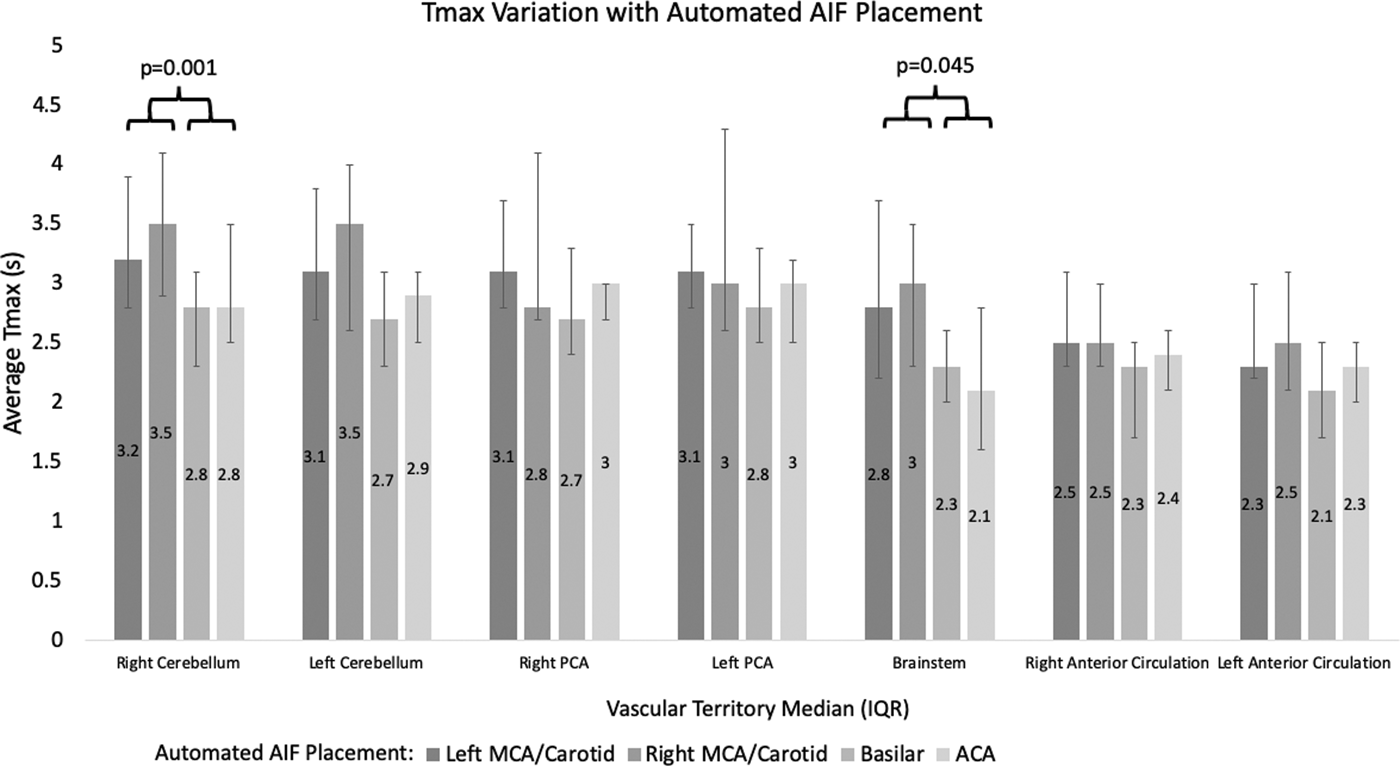
Median vascular territorial Tmax by location of automated arterial input function (AIF) placement demonstrating that Tmax is significantly higher in the right cerebellum and brainstem with AIF placement on the right and left MCA or carotid terminus (see Methods).
To determine how alternating AIF placement affects territorial Tmax within individual patients, AIF were placed on the right and left MCA or distal ICA, ACA, and basilar artery for each individual subject. We found that median territorial Tmax was significantly lower when AIF was placed on the basilar artery compared with either the right or left MCA, ICA, and ACA for all vascular territories (p < 0.001) (Figure 3). For instance, median territorial Tmax in the right cerebellum measured 3.3 s (IQR 2.4 s-3.9s), 3.8 s (IQR 2.8 s-4.1s), 3.3 s (IQR 2.5 s-3.7s), and 2.8 s (IQR 2.3 s-3.2s) when AIF were placed on the right and left MCA/ICA, ACA, and basilar arteries, respectively, with the basilar AIF placement significantly lower than the other AIF placements (p < 0.001, by the Wilcoxon signed-rank test). Tmax values for every vascular territory were found to be significantly higher when AIF was selected over the left MCA/ICA as compared with the right MCA/ICA (p < 0.05).
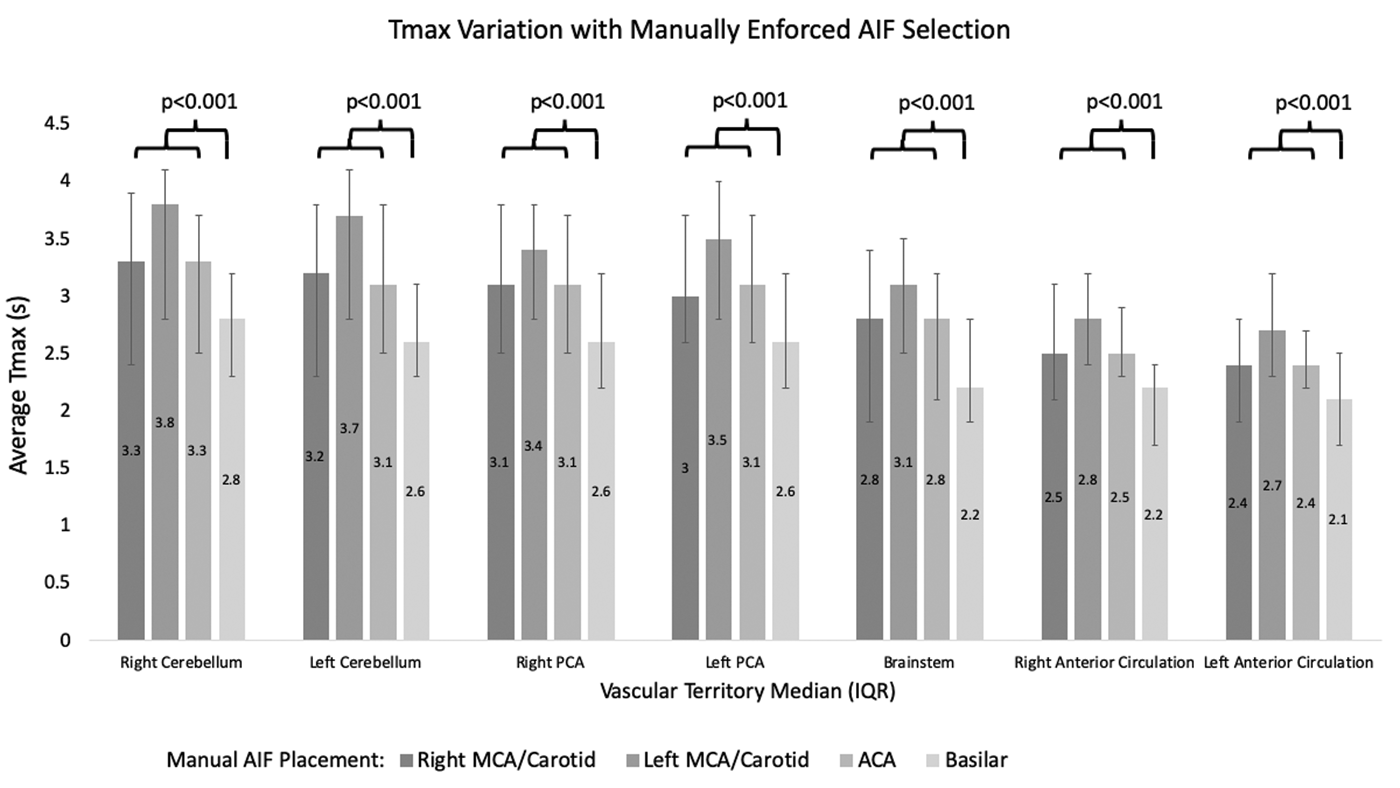
Median vascular territorial Tmax varying, manual arterial input function (AIF), demonstrating that Tmax is significantly lower in all vascular territories when AIF was placed over the basilar artery (see Methods).
Effect of anatomic variation on vascular territory tmax
Among the 71 subjects included in the analysis, 16 had fetal PCA origins, including 5 right-sided, 9 left-sided, and 2 bilateral. As expected, median territorial Tmax in the PCA territory was lower ipsilateral to the fetal PCA (right and left PCA territory median Tmax 3.4 s (IQR 2.9 s-4.1s) vs 3.6 s (IQR 3.4 s-5.2s) in subjects with a right fetal PCA, respectively, p = 0.057; and right and left PCA territory median Tmax of 2.8 s (IQR 2.7 s-3.0s) vs 2.4 s (IQR 2.3-3.0s) in subjects with a left fetal PCA, p = 0.012). There was no difference between left and right PCA territorial Tmax in subjects with bilateral fetal PCA (p = 0.317) in whom equivalent contributions from the anterior circulation were present to both PCA territories (Figures 4 and 5).
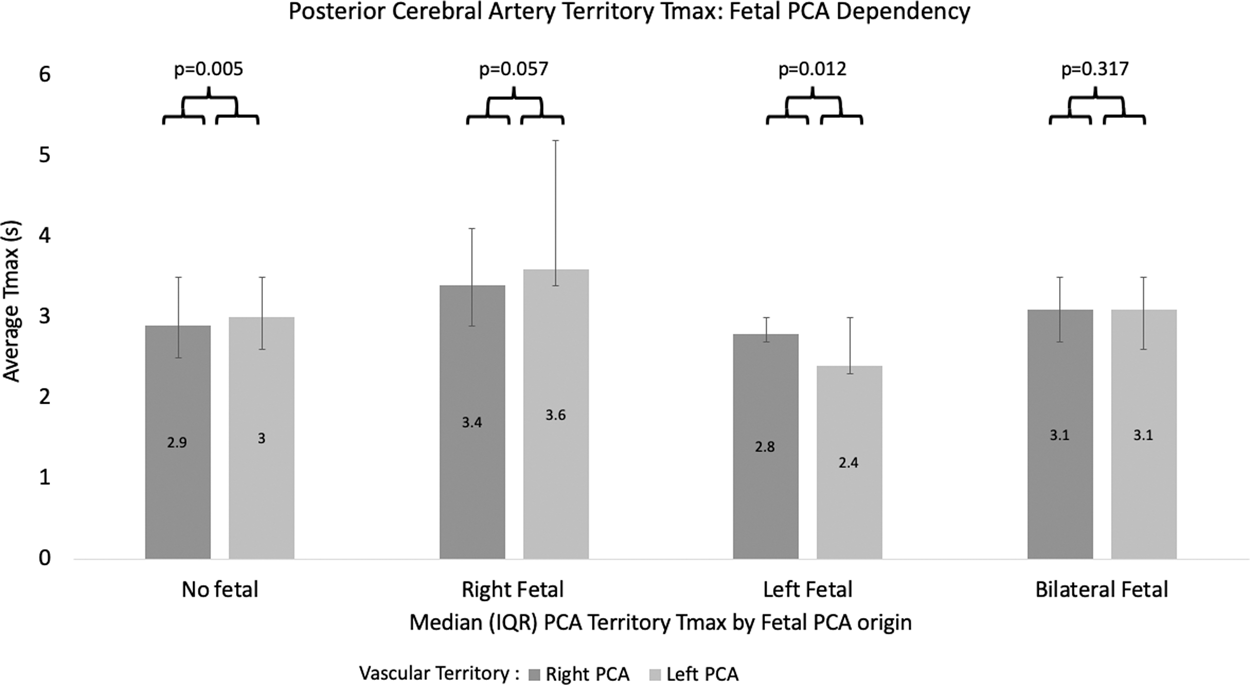
Median Tmax in the right and left PCA territory by the presence of fetal PCA origin. Tmax is elevated on the side contralateral to a unilateral fetal PCA origin, by comparison with similar PCA territory Tmax values in circumstances of bilaterally absent or present fetal PCA.
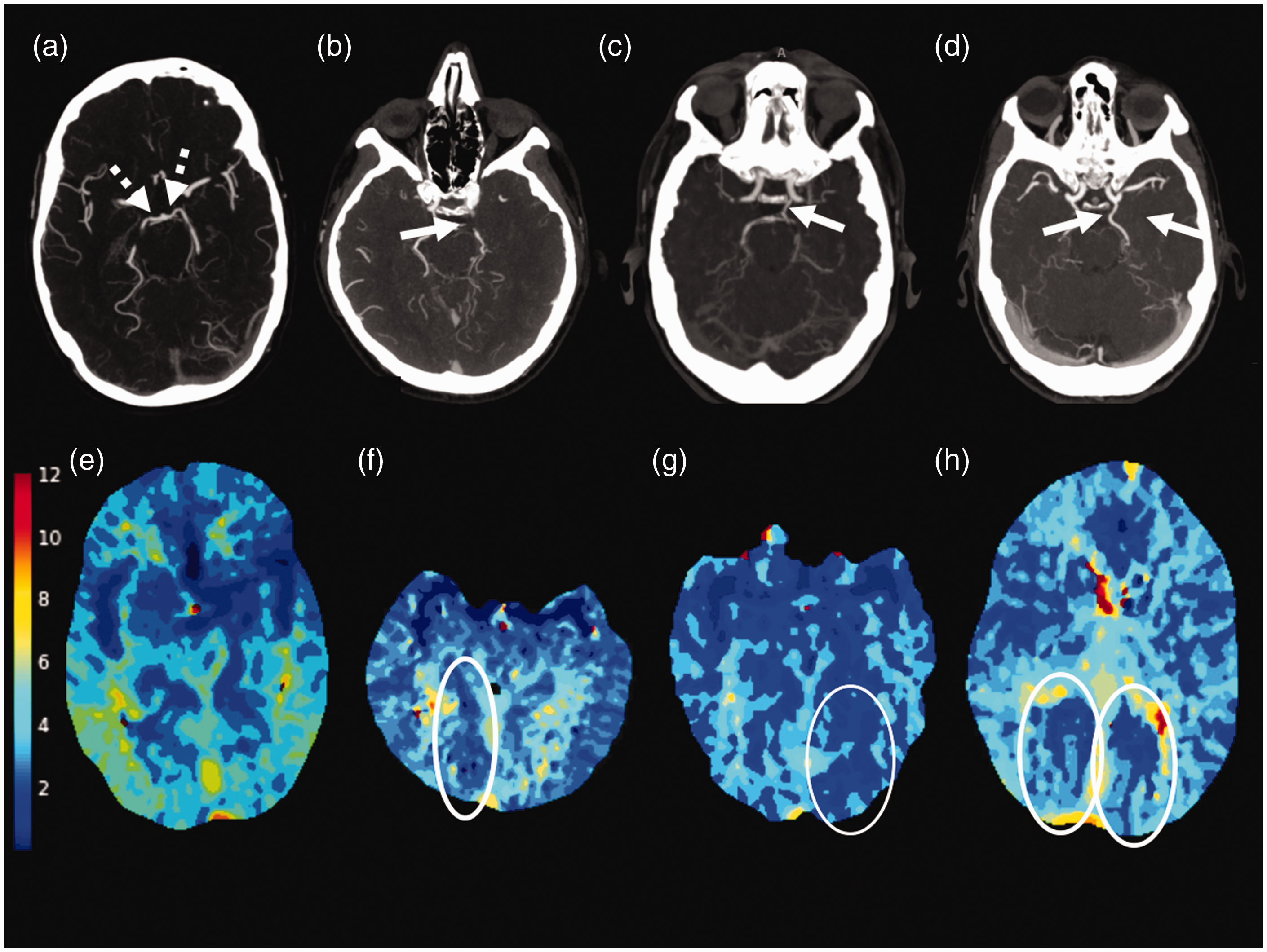
CTA and CT perfusion maps demonstrating differences in PCA territory Tmax in the presence and absence of fetal PCA. In a patient without fetal PCA, in whom the PCA-P1 segment is supplied by the basilar artery (a; dashed arrow), average right and left anterior circulation Tmax were lower (3.7 s and 3.3 s, respectively) than either PCA territory average Tmax (4.2 s and 4.5 s, respectively) (e). In a patient with a right fetal PCA (b; arrow), average right anterior circulation Tmax was similar to the right PCA territory (f; 2.6 and 2.9, respectively [right PCA territory circled]), while the left anterior circulation Tmax was lower than the left PCA territory Tmax (2.4 s and 3.6 s, respectively). In a patient with a left fetal PCA (c; arrow), average left anterior circulation Tmax was similar to the left PCA territory (G; 2.1 and 2.4, respectively [left PCA territory circled]) while the right anterior circulation Tmax was lower than the right PCA territory (2.3 s and 2.8 s, respectively). In a patient with bilateral fetal PCA (d; arrows), average right and left anterior circulation Tmax (3.2 s and 3.0 s) were similar to the PCA territory average Tmax (3.5 s and 3.5 s; circles in h).
Among all posterior circulation territories with ipsilaterally hypoplastic/absent posterior communicating arteries, statistically significantly longer PCA Tmax (p = 0.005) were observed by comparison to those PCA territories fed primarily by posterior communicating arteries. No significant differences were present between the left and right PCA territory Tmax in subjects with bilateral hypoplasia (median 2.9 s (IQR 2.5 s-3.5s) and 3.0 s (IQR 2.6 s-3.5s), respectively). Further, in the absence of ICA contributions to either PCA territory, such that the territories were fully dependent upon vertebrobasilar flow bilaterally, significantly higher Tmax values were demonstrated by comparison to the anterior circulation territories among those subjects (Figure 6, median absolute differences between PCA and anterior circulation territorial Tmax of 0.6 s (IQR 0.2 s-0.7s) on the right and 0.7 s (IQR 0.5 s-0.9s) on the left, p < 0.001 for both). With an ipsilateral fetal PCA, however, these differences significantly decrease in those PCA territories to 0.3 s (IQR 0.2 s-0.3s) and 0.4 s (IQR 0.3 s-0.5s) in the right and left hemispheres, respectively, with differences in the contralateral hemisphere remaining elevated at 0.6 s and 0.8 s, p = 0.049 and 0.004, respectively.
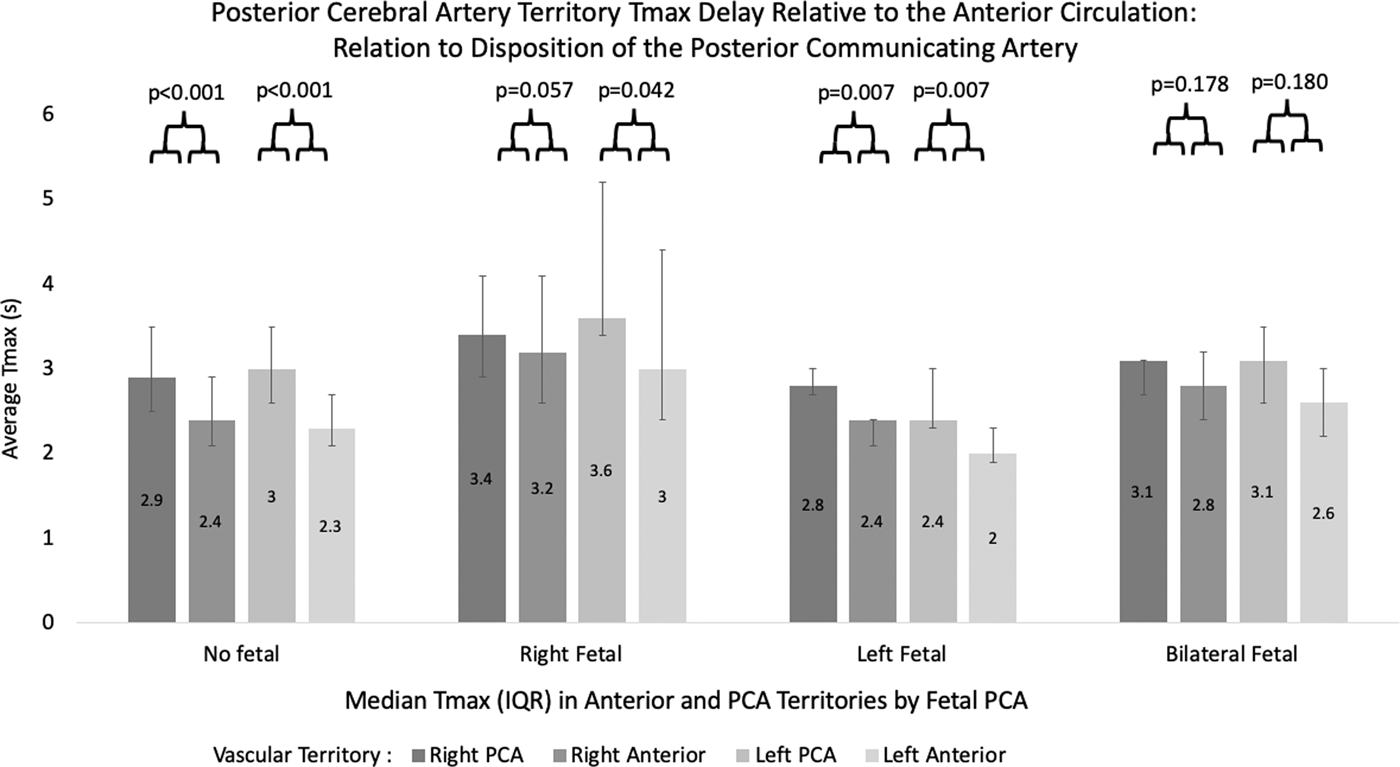
Differences in the median territorial Tmax between the anterior circulation and PCA territories, relative to disposition of the PCA. PCA territory Tmax is significantly delayed when compared with the ipsilateral anterior circulation in the absence of fetal PCA, while significant differences are absent between the two in subjects with fetal PCA subserving the ipsilateral PCA territory.
Significant differences between the anterior circulation and PCA territories and their interactions with disposition of the PCA persisted when AIF was enforced on either MCAs or ICAs and for both sides. For example, when AIF was enforced on the right MCA/ICA, the difference between the anterior circulation and PCA territories was significantly lower on the side ipsilateral to the fetal PCA as compared to the contralateral side which lacked fetal circulation (differences on the right of 0.3 s (IQR 0.2 s-0.4s) and 0.5 s (IQR 0.3 s-0.8s) in the presence and absence of a right fetal PCA, respectively (p = 0.026), and 0.3 s (IQR 0.3 s-0.4s) and 0.6 s (IQR 0.4 s-1.0s) on the left in the presence and absence of a left fetal PCA, respectively (p = 0.003). Similar results were observed with AIF enforced on the left MCA or ICA. No significant differences were noted between the right and left anterior circulation and PCA gradients in the presence or absence of fetal PCA whether AIF were placed on the right or left MCA or ICA (p > 0.05).
Nineteen subjects had a complexed/shared origin of the AICA/PICA, including six on the right, 11 on the left, and 2 bilaterally. Dominant vertebral artery circulation was identified in 32 subjects with 12 on the right, 20 on the left. The remaining 39 subjects had codominant vertebral arteries. There was no significant difference in cerebellar Tmax values with the presence or absence of an ipsilateral or contralateral AICA-PICA, or based on vertebral artery dominance (p > 0.05). There was no significant difference in brainstem Tmax based on vertebral artery dominance. Primitive carotid-basilar (e.g. persistent trigeminal arteries) were not identified in any study subjects.
Discussion
This study represents, to our knowledge, the first formal investigation into anatomic Tmax distributions in CTP, offering potentially critical insights into the future use of perfusion-based selection criteria in posterior circulation AIS. In this study we test the hypothesis that the intrinsic macrovascular delay sensitivity of the deconvolved tissue time-to-maximum would reveal baseline elevations throughout the physiologically slower and potentially more circuitous posterior circulation.15,20,26 Further, the greater tendency for posterior circulation anatomic variations that, to a variable extent and potentially asymmetrically, preserve primitive communications between the anterior and posterior circulation (such as fetal PCA) allowed for Tmax profiles to be examined with the inherent physiologic/anatomic contrast between territories. We found that amongst subjects without any macroscopic cerebrovascular steno-occlusive disease, posterior circulation Tmax values were significantly affected by anatomic variation and AIF placement, again despite the use of robust and delay-insensitive deconvolution algorithms for CBF estimation. While these effects appear unlikely, in isolation, to engender falsely positive imputation of critically hypoperfused (i.e. Tmax > 6s) tissue volumes in normal subjects, their potential to disproportionately elevate the delay in pathologic tissues and/or increase the overall estimated hypoperfusion volume during AIS merits consideration and may require dedicated calibration in the setting of even mild proximal stenoses. Further, a vulnerability to exaggerated posterior circulation Tmax may be expected in inherently low-flow areas such as the white matter, in particular when microangiopathic white matter disease might further compound noise related corruption and overestimation of the deconvolved Tmax. The tendency for significantly greater delays in the posterior circulation, even in the absence of any visible steno-occlusive lesions, interacts significantly with AIF selection between the anterior and posterior circulation, suggesting that attention to AIF selection is warranted, including potentially reformulated hypoperfusion thresholds when estimating posterior circulation penumbral volumes even with circular or Fourier deconvolution and other delay correction strategies.15,17,26–28 We additionally identified an interaction between seizure and delays in the posterior circulation Tmax, which were significantly higher in subjects with final seizure diagnosis. Inter-ictal cerebellar hypoperfusion has previously been described, likely related to spatially remote effects mediated by the corticopontocerebellar pathway.29,30 We did not find significant differences in the supratentorial brain in seizure patients, possibly because small regions under inter-ictal influences may be obscured when averaging the Tmax of the entire vascular territory, although, the overall small number of such subjects within our study sample limits broad generalization of this effect.
Due to a high rate of vascular anatomic variability, the posterior circulation is particularly susceptible to differences in baseline perfusion profiles. Blood flow velocities and flow volumes in the internal carotid arteries exceed those in the vertebral arteries, which has shown to result in slightly earlier bolus arrival on the side of a fetal PCA.10,31,32 Although older studies have shown the effects of bolus delay on perfusion parameters such as the mean transit time (MTT) and time-to-peak (TTP), both parameters have been supplanted in favor of the Tmax of a deconvolved tissue residue function, modeled as the delay in impulse response to a theoretically instant tracer bolus in the tissue bed. Tmax has emerged as a serendipitous nuisance parameter of tracer kinetics, owing to its high signal and generally uniform distribution in gray and white matter, producing high lesion-to-background contrast and facilitating voxel-wise thresholding. Together with its relative immunity to bolus truncation effects, Tmax has been suggested as the hypoperfusion variable of choice in multiple studies, including in the clinical trial setting and by expert consensus.1,2,5,21,22,25,33–38 By comparison to the use of TTP or MTT, the isolation of Tmax requires deconvolution of an automatically- or user-selected AIF with which to deconvolve the effects of delay and dispersion relating both to the profile of the injected bolus and the course of the bolus through the proximal circulation, either of which produce deviations of the observed tissue voxel from idealized tracer kinetic features. Past approaches, such as singular value decomposition, are now well-recognized for their vulnerability to delay-related errors, which can be addressed through timing-invariant deconvolutional approaches including those used in this study.15,16 Tmax, however, specifically leverages this delay in the estimation of hypoperfused volumes. Consequently, delay corrections and delay insensitive deconvolutions would be anticipated still to reflect such macrovascular delay as might occur through a longer, slower, or more circuitous vascular network, in particular when the chosen AIF arises from within a faster or more direct vessel as often occurs using automated AIF selection. While AIF deconvolution mitigates the delay and dispersion effects in CBF estimation, Tmax remains sensitive to macrovascular transit effects. Further, because local (i.e. within the tissue itself) AIF selection on a per-voxel basis is precluded by limits in spatial resolution and signal-to-noise in conventional protocols, a global AIF selection is commonly employed in CTP deconvolution. This effect becomes most apparent when examining the greater Tmax of the posterior circulation territories when an anterior circulation AIF is selected, or, importantly when asymmetries between PCA territories are present, such as when fetal circulation subserves only one hemisphere. Similar effects of fetal PCA circulation have been demonstrated using MRI perfusion techniques, with occipital lobe Tmax shortening strongly correlated with the degree of vascular asymmetry, as well as with arterial spin labeled perfusion.31,39 We propose that the strong interaction of posterior circulation Tmax with AIF selection poses yet another potential pitfall compelling circumspection before anterior circulation perfusion imaging guidelines are applied to posterior circulation stroke. AIF selection is shown only to modestly influence volumetric assessment of anterior circulation stroke, provided the use of delay insensitive deconvolution methodologies; however, the effect has primarily been interrogated for cerebral blood flow in past studies. Tmax, because of its inherent delay sensitivity, has performed well for anterior circulation estimations of at-risk volumes despite this vulnerability, which we find affected both hemispheres relatively equally under putatively physiologic conditions. The extension of this methodology to the posterior circulation, however, compels that the uniquely delayed/slower hemodynamic features of the vertebrobasilar system be considered closely before a macrovascular delay parameter is employed for use in this region. We would underscore that the differences, while significant, reflect small relative delays in absolute terms, and the impact of discretization, interpolation, and data sampling rates upon these Tmax differences warrants dedicated consideration. 27 Nevertheless, the effects of AIF placement and variant vascular anatomy require further consideration when designing CTP threshold-based treatment selection trials in the posterior circulation.
Several limitations to our study merit specific discussion, including those inherent to its retrospective nature. As the study objectives required identification of macroscopically normal subjects requiring CTA and CTP to establish putatively normal perfusion profiles, prospective or randomized enrollment of healthy subjects was not practicable. We acknowledge that occult microvascular disease, and potentially impaired cardiac output could affect arterial arrival times, however, the absence of any Tmax delays in the anterior circulation militate against significant global hemodynamic impairment in our population. The possibility for fully reversible and transient ischemia at presentation, such as from a transient ischemic attack (TIA), may also be difficult to entirely exclude. Nevertheless, past TIA trials have reported the combined sensitivity of arteriographic and perfusion imaging to be very high in their ability to identify patients with a significant risk for future ischemic injury, and, in combination with normal MRI and the final clinical determination of non-ischemic etiologies, we consider the likelihood of that scenario as biasing our results to be low. 40 Mild errors related to poor SNR in low flow areas such as microangiopathic white matter disease can be observed, and our study did not specifically address such variables; however, it has been our observation that such effects rarely produce significant Tmax delays in isolation and likely would not account for large, vascular territorial differences as described herein. Nevertheless, future studies should consider such structural and technical variables, as well CT attenuation spikes or suppression related to sporadic streak and beam hardening artifacts commonly occurring in the posterior fossa, which we aimed to exclude. While our data set may have been insufficient to identify more subtle statistical interactions, the study population was comparable to or exceeded other retrospective studies of similar design. Notwithstanding, a dedicated analysis with a larger and prospectively collected cohort of patients with steno-occlusive injury and early MRI or CT follow up is critical to better formulate paradigms of posterior circulation mismatch. Lastly, the vascular anatomic template registered to CTP in our study does not specifically delineate AICA, PICA, superior cerebellar or perforator territories, and a more nuanced description of the territory specific delays might identify effects related, for instance, to vertebral artery dominance patterns which failed to show differences in this study. Similarly, limits in CTP slice thickness and spatial resolution precluded more detailed investigation into posterior circulation AIF placement elsewhere besides that in the basilar artery. With the preceding in mind, we propose that the results reflect a reasonable estimation of true, normative distributions of the tissue time-to-maximum in the posterior circulation from which future paradigms of critical hypoperfusion in the setting of acute ischemia can be derived.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Adam Goldman-Yassen: Nothing to report
Matus Straka – Consulting fees, shareholder, iSchemaView, Inc., Menlo Park, CA, which developed RAPID automated CT perfusion software.
Michael Uhouse – Nothing to report
Seena Dehkharghani – Travel support (modest) from iSchemaView, Menlo Park, CA, which developed RAPID automated CT perfusion software.
Authors’ contributions
Conception and design of the study: AGY, MS, SD Acquisition and analysis of data: AGY, MS, MU, SD Drafting a significant portion of the manuscript or figures: AGY, MS, SD.
