Abstract
Crossed cerebellar diaschisis (CCD) is the phenomenon of hypoperfusion and hypometabolism of the contralateral cerebellar hemisphere caused by dysfunction of the related supratentorial region. Our aim was to analyze its influence on morphologic and functional outcome in acute ischemic stroke. Subjects with stroke caused by a large vessel occlusion of the anterior circulation were selected from an initial cohort of 1644 consecutive patients who underwent multiparametric CT including whole-brain CT perfusion. Two experienced readers evaluated the posterior fossa in terms of CCD absence (CCD−) or presence (CCD+). A total of 156 patients formed the study cohort with 102 patients (65.4%) categorized as CCD− and 54 (34.6%) as CCD+. In linear and logistic regression analyses, no significant association between CCD and final infarction volume (β = −0.440, p = 0.972), discharge mRS ≤ 2 (OR = 1.897, p = 0.320), or 90-day mRS ≤ 2 (OR = 0.531, p = 0.492) was detected. CCD+ patients had larger supratentorial cerebral blood flow deficits (median: 164 ml vs. 115 ml; p = 0.001) compared to CCD−patients. Regarding complications, CCD was associated with a higher rate of parenchymal hematomas (OR = 4.793, p = 0.035). In conclusion, CCD is frequently encountered in acute ischemic stroke caused by large vessel occlusion of the anterior circulation. CCD was associated with the occurrence of parenchymal hematoma in the ipsilateral cerebral infarction but did not prove to significantly influence patient outcome.
Introduction
Crossed cerebellar diaschisis (CCD) is the phenomenon of a decreased cerebellar perfusion and glucose metabolism secondary to a supratentorial malfunction of brain tissue in the contralateral hemisphere, first described by Baron et al.1–3 It can be detected through changes in electrical activity, cerebral blood flow, or cerebral metabolic rates for glucose and oxygen using methods such as electroencephalography, single-photon emission-computed tomography (SPECT), or positron emission tomography (PET).4,5 CCD has been reported in supratentorial tumors, 6 epilepsy, 7 encephalitis, 8 and cerebral infarction. 9 Animal studies suggest that CCD is explained by deactivation of cerebellar neurons caused by reduction of excitatory impulses via the corticopontocerebellar tract.5,10 In ischemic stroke, CCD has been observed during the acute, subacute, and chronic phase.11,12 Current scientific concepts attribute acute CCD to functional neuronal deafferentiation, whereas chronic CCD probably reflects a state of transneuronal degeneration. 13 In ischemic stroke due to large anterior circulation vessel occlusion, CCD has been shown to be present in about one-third of the affected patients in the acute phase.14,15
While the original definition of CCD was based on the idea that it is a transient condition, 16 several publications have reported CCD up to decades after the index stroke.17–20 Regarding the reversibility of CCD, however, several serial SPECT and PET studies demonstrated that some patients show complete reversal of CCD during follow-up.11,13,21–23 With respect to functional outcome, the majority of studies in stroke patients using SPECT or PET investigated subacute and chronic CCD, reflecting the fact that these imaging methods are not eligible to routinely assess perfusion in the acute stroke setting. Several studies with relatively small sample sizes report correlations with clinical severity scales for subacute11,13,24,25 and chronic CCD.26,27 Yet, the two largest studies on subacute CCD report no independent predictive value after statistical correction for the infarct hypoperfusion volume.28,29 For acute CCD, no correlations to clinical severity scales were established so far.11,25,26 However, current sample sizes are too small to draw general conclusions regarding its clinical impact.
Unlike SPECT and PET, computed tomography perfusion imaging is a method that reliably demonstrates areas of hypoperfusion in clinical routine stroke workup. 30 Advances in imaging technology meanwhile allow to cover the entire brain tissue at a reasonable radiation dose (whole-brain CT perfusion, WB-CTP). 31 While recent WB-CTP studies on stroke patients have shown the occurrence of CCD in the acute phase to be dependent on the location of the supratentorial perfusion deficit and the severity of supratentorial hypoperfusion, 14 the key question of the clinical impact of CCD in acute ischemic stroke remains unanswered.
Therefore, the aim of the present whole-brain CT perfusion study was to determine the influence of CCD occurrence in the acute phase on morphologic and functional outcome in patients with acute ischemic stroke due to large anterior circulation vessel occlusion.
Material and methods
Study design and population
The institutional review board of the LMU Munich (Ethikkommission der Medizinischen Fakultät der Ludwig-Maximilians-Universität München) approved this retrospective study according to the Helsinki Declaration of 1975 (and as revised in 2013) and waived requirement for informed consent. Our initial cohort consisted of 1644 consecutive patients who had undergone WB-CTP due to suspected stroke between April 2009 and June 2014.
Out of this cohort, we included all subjects with
occlusion of the internal carotid (ICA), carotid T and/or middle cerebral artery (MCA), perfusion deficit in the MCA territory on WB-CTP, and follow-up confirmed ischemic infarction.
We excluded patients with
any abnormality of the posterior vasculature on CT angiography (CTA), cerebellar infarction or other cerebellar pathology on initial or follow-up imaging, missing follow-up imaging, or incomplete coverage of the cerebellum or non-diagnostic quality of WB-CTP.
Out of the initial 1644 patients, 323 patients had an ICA, carotid T, or MCA occlusion. Out of these, 20 patients with missing follow-up imaging, 89 with abnormalities of the posterior circulation, 14 with pathologies of the cerebellum, 6 with acute cerebellar infarction on follow-up imaging and 38 with incomplete cerebellar coverage or non-diagnostic quality of WB-CTP were excluded. The remaining 156 patients formed the final study population.
CT acquisition, CT perfusion processing and follow-up imaging
All patients underwent a standardized multiparametric CT protocol consisting of non-enhanced CT (NECT), single-phase CT angiography (spCTA), and WB-CTP. The acquisition protocol has been described in detail before. 32 The source image processing was performed with SYNGO Volume Perfusion CT Neuro software using a semi-automated deconvolution algorithm (Auto Stroke MTT) on a dedicated workstation (Syngo MMWP, VA 21A; Siemens Healthcare, Erlangen, Germany). A series of 31 color-coded slices was generated for each of the hemodynamic parameters cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), time to drain (TTD), and time to peak (TTP). Follow-up imaging was performed with MRI in 56% and NECT in 44% of patients as previously described. 14 The median time from initial WB-CTP imaging to follow-up imaging was two days for MRI (range: 1–49) and one day for NECT (range: 1–16).
Image analysis
The assessment of presence of CCD was performed qualitatively by two independent readers (one neurologist with nine years and one radiologist with seven years of experience in CTP reading, respectively) blinded to all clinical data and the location of supratentorial infarctions by cropping all images to the posterior fossa. In case of disagreement, a consensus was reached in a separate session. Only perfusion anomalies in the cerebellar hemisphere contralateral to the supratentorial lesion were counted as CCD positive. Figure 1 shows representative examples of CCD− and CCD+ patients.
Examples of CCD− and CCD+ patients. Patient examples of acute ischemic stroke without and with signs of crossed cerebellar diaschisis (CCD). NECT, supratentorial CBF map, infratentorial CBF and MTT map, and follow-up DWI are depicted from for a CCD− (a) and a CCD+ patient (b). NECT: non-enhanced CT; CBF: cerebral blood flow; MTT; mean transit time; DWI: diffusion-weighted imaging.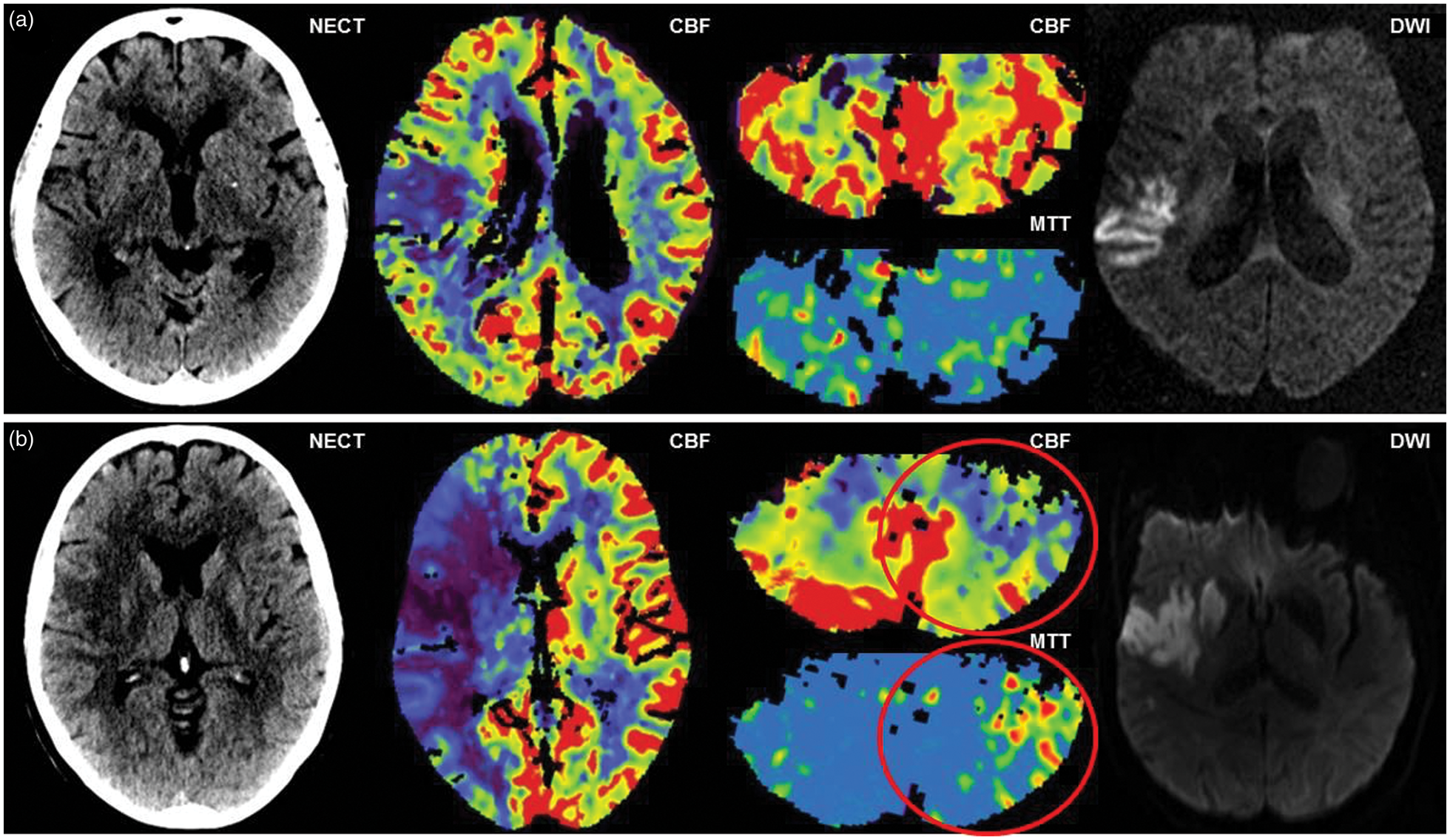
The extent of the supratentorial ischemic region was assessed on NECT using the semi-quantitative Alberta Stroke Program Early Computed Tomography Score (ASPECTS). 33 CBF and CBV deficit volume and final infarction volume were determined as previously described. 31 The primary morphologic outcome parameter was final infarction volume. As secondary morphologic outcome, we determined parameters that represent the morphologic course from ischemia to infarction using an approach comparable to previous studies.34–37 To quantify the change from the initial hemodynamic impairment to the final infarcted tissue, we used the following calculated parameters as surrogates: [final infarction volume/CBF deficit volume], [final infarction volume/(CBF deficit volume – CBV deficit volume)], and [CBF deficit volume – final infarction volume]. The rationale of these parameters is illustrated in supplementary Figure 1. All available follow-up imaging prior to discharge was assessed for subacute stroke complications. Hemorrhagic infarction and parenchymal hematoma were categorized as type I and II according to the European Cooperative Acute Stroke Study (ECASS) criteria. 38 Parenchymal hematoma type II development has a negative influence on functional outcome. 39 Intracranial hemorrhage (ICH) remote to the infarction area was classified as extraischemic. The presence of space-occupying edema was defined as previously described and assessed using follow-up NECT on day 3 ± 2. 40
Acute stroke therapy
Intravenous thrombolysis (IVT) was administered to eligible patients at a dose of 0.9 mg/kg bodyweight (maximum dose: 90 mg) in two parts: 10% of the total dose was administered as an IV bolus, immediately followed by an IV infusion over 60 min of the remaining dose diluted in 50 ml of sodium chloride 0.9%. IVT was started approximately 10–20 min after imaging evaluation, i.e. time from symptom onset plus 10–20 min. If IVT was given, preexisting antiplatelet or anticoagulation therapy was halted for 24 h.
Endovascular therapy (EVT) was performed as a mechanical stent retriever thrombectomy procedure either under general anesthesia or, whenever deemed appropriate by the interventional neuroradiologist and the anesthesiologist, under conscious sedation. All procedures were performed in a triaxial fashion using a distal access catheter and a microcatheter to deploy a stent retriever device. All used stent retrievers in this cohort were latest generation devices (Solitaire, ev3 Neurovascular, Irvine, CA, USA; Preset, phenox GmbH, Bochum, Germany; Trevo, Concentric Medical, Mountain View, CA, USA). After affirmation of recanalization, the catheter material was removed.
In general, therapy with aspirin (100 mg/day) was initiated immediately after admission to the stroke unit. Exclusion criteria were intracerebral hemorrhage and IVT administration. In case of IVT, aspirin therapy was suspended for at least 24 h and initiated only after repeated imaging to exclude IVT-related hemorrhage. In case of cardioembolic stroke, the decision to start anticoagulation was made individually depending on the size of infarction.
Functional outcome data
The functional outcome evaluation in this study was based on the National Institutes of Health Stroke Scale (NIHSS) score 41 determined on admission and the modified Rankin Scale (mRS) score 42 assessed on admission, on discharge and 90 days after the stroke event. Furthermore, the premorbid mRS score representing patient disability prior to the current stroke event was estimated by taking detailed medical history of the patient whenever possible. Patients were excluded from the functional outcome analysis in case of premorbid mRS > 1, missing clinical documentation, second stroke event, or death to other cause within 90 days. None of the included patients had a history of premorbid ischemic injury to the ipsilateral MCA territory. Detailed characteristics of excluded patients are provided in supplementary Table 1.
Statistical analysis
We performed all statistical analyses using SPSS Statistics 23 (IBM, Armonk/NY, USA). Normal distribution was evaluated using the Kolmogorov–Smirnov test. In case of non-normal distribution, we applied the Chi-squared test for categorical and the Mann–Whitney-U test for continuous variables to identify significant differences between patients classified as CCD+ and CCD−. Univariate linear regression analysis was used to test the association between predictors and continuous morphologic outcome variables. Logistic binary regression analysis was used between predictors and the categorical functional outcome variables. All metric and normally distributed variables are reported as mean ± standard deviation; non-normally distributed variables are presented as median (interquartile range). Categorical variables are presented as frequency and percentage. P values below 0.05 were considered to indicate statistical significance.
Results
Patient characteristics
Characteristics of CCD− and CCD+ acute ischemic stroke patients.
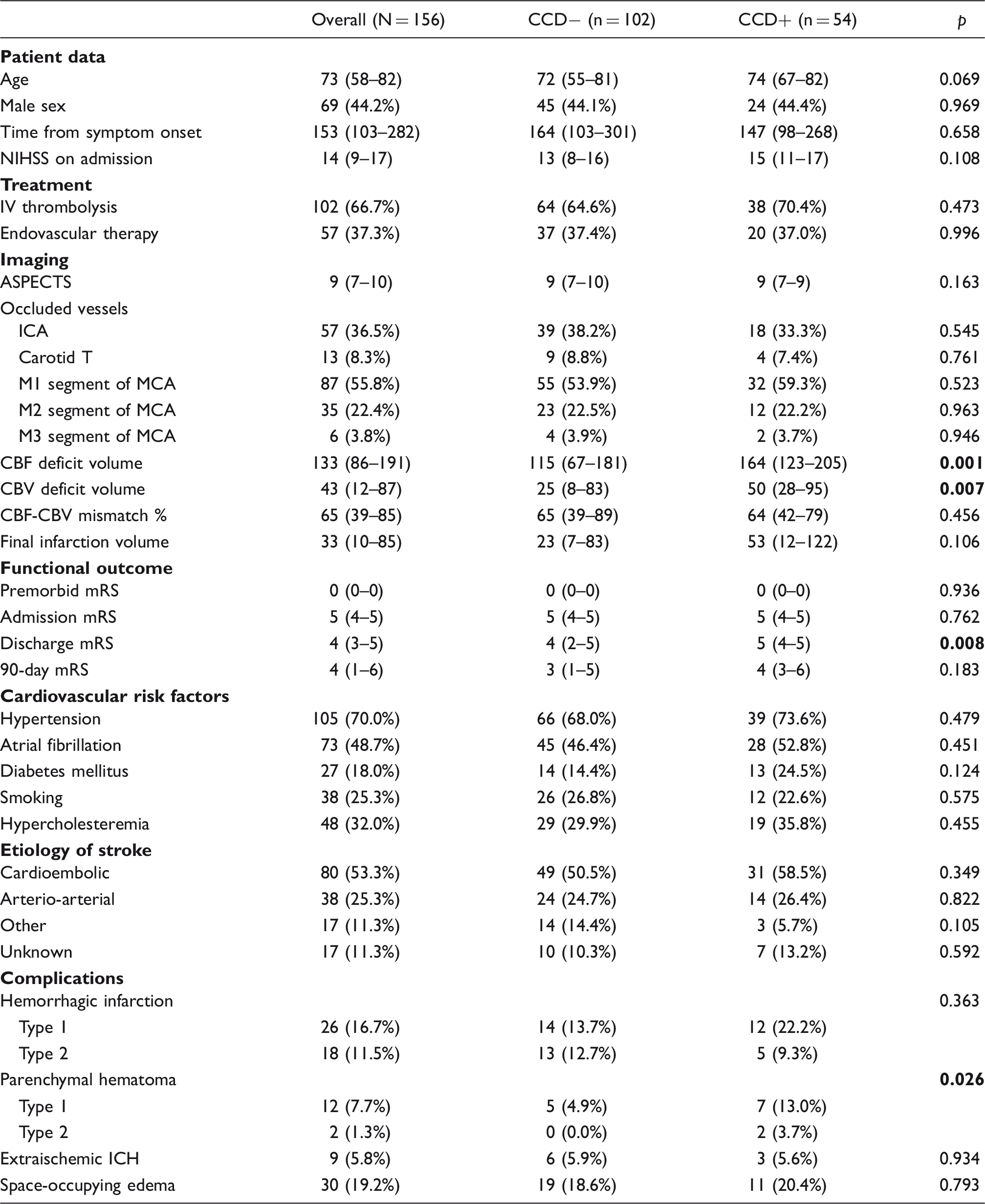
CCD: crossed cerebellar diaschisis; NIHSS: national institutes of health stroke scale; ASPECTS: Alberta stroke program early CT score; ICA: internal carotid artery; MCA: middle cerebral artery; CBF/CBV: cerebral blood flow/volume; mRS: modified Rankin Scale; ICH: intracranial hemorrhage.
Note: Values presented are count (percentage) for categorical and median (interquartile range) for ordinal or continuous variables. Proportion analysis tests for categorical variables were performed using the χ2 test. Nonparametric tests for non-normally distributed continuous variables were performed using the Mann–Whitney U test, and for ordinal variables using the independent samples median test. Time parameters are measured in minutes; volume parameters are measured in mL. Bold p values indicate statistical significance.
Association of CCD with morphologic outcome
Predictors of morphologic and functional outcome.
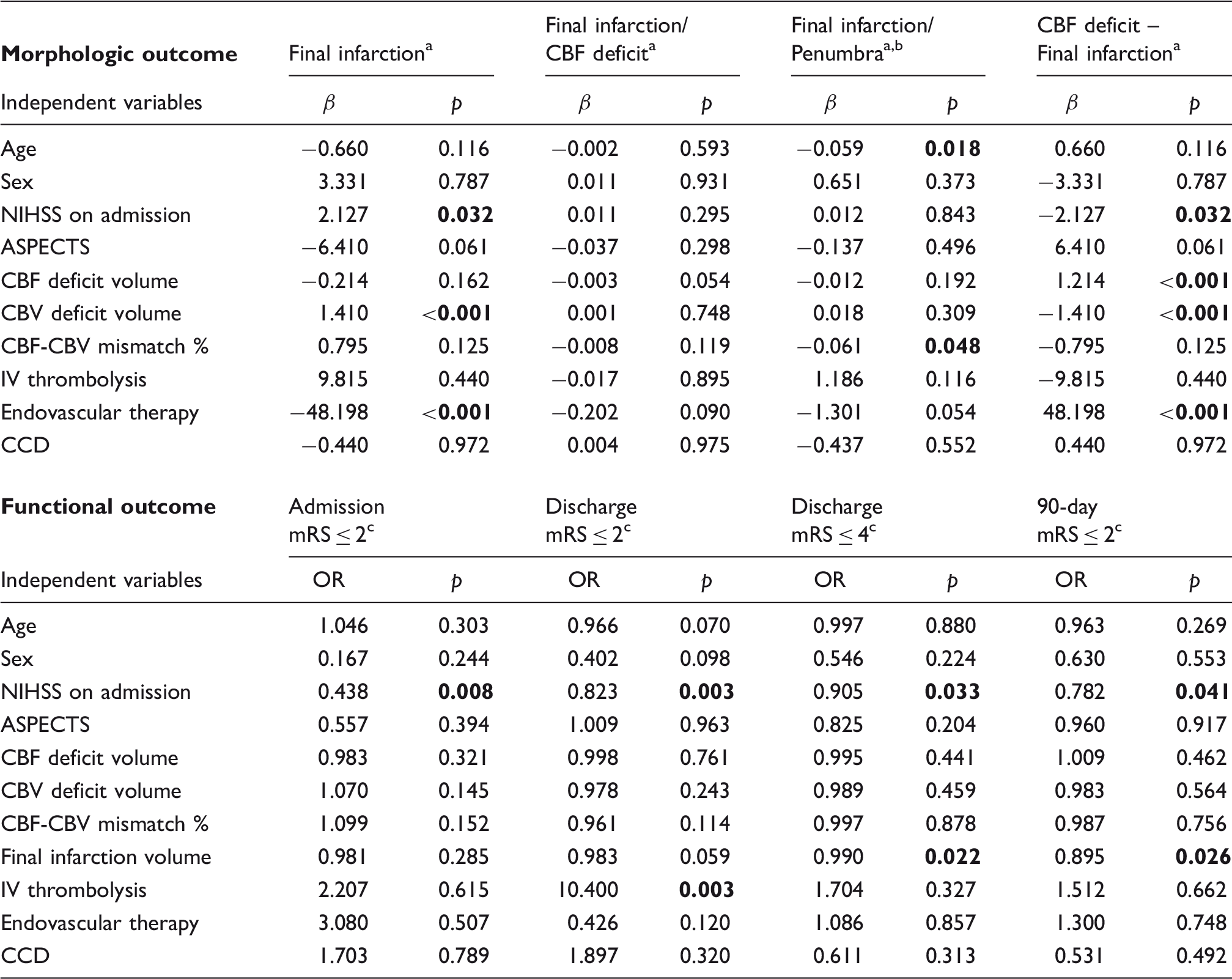
All parameters represent volumetric measures.
Penumbra is defined as CBF deficit volume – CBV deficit volume.
Available data: Admission mRS 131/131, Discharge mRS 131/131, 90-day mRS 72/131.
mRS: modified Rankin Scale; NIHSS: national institutes of health stroke scale; ASPECTS: Alberta stroke program early CT score; CBF / CBV: cerebral blood flow/volume; OR: odds ratio.
Note: A univariate linear regression analysis was performed for the indicated morphologic outcome parameters for the complete study population of 156 patients. A binary logistic regression analysis was performed for the indicated functional outcome parameters for the patient selection according to Figure 2. Bold p values indicate statistical significance.
Association of CCD with functional outcome
According to our exclusion criteria for the analysis of functional outcome, we excluded patients with premorbid mRS > 1 (N = 16), missing documentation (N = 5), second stroke events within 90 days (N = 2), and death to non-stroke-related causes within 90 days (N = 2). A flow chart of the patient selection is presented in Figure 2. Bar graphs representing admission, discharge, and 90-day mRS scales are shown in Figure 3.
Flow chart of patient selection for functional outcome analysis. Modified Rankin Scale scores of CCD− and CCD+ patients.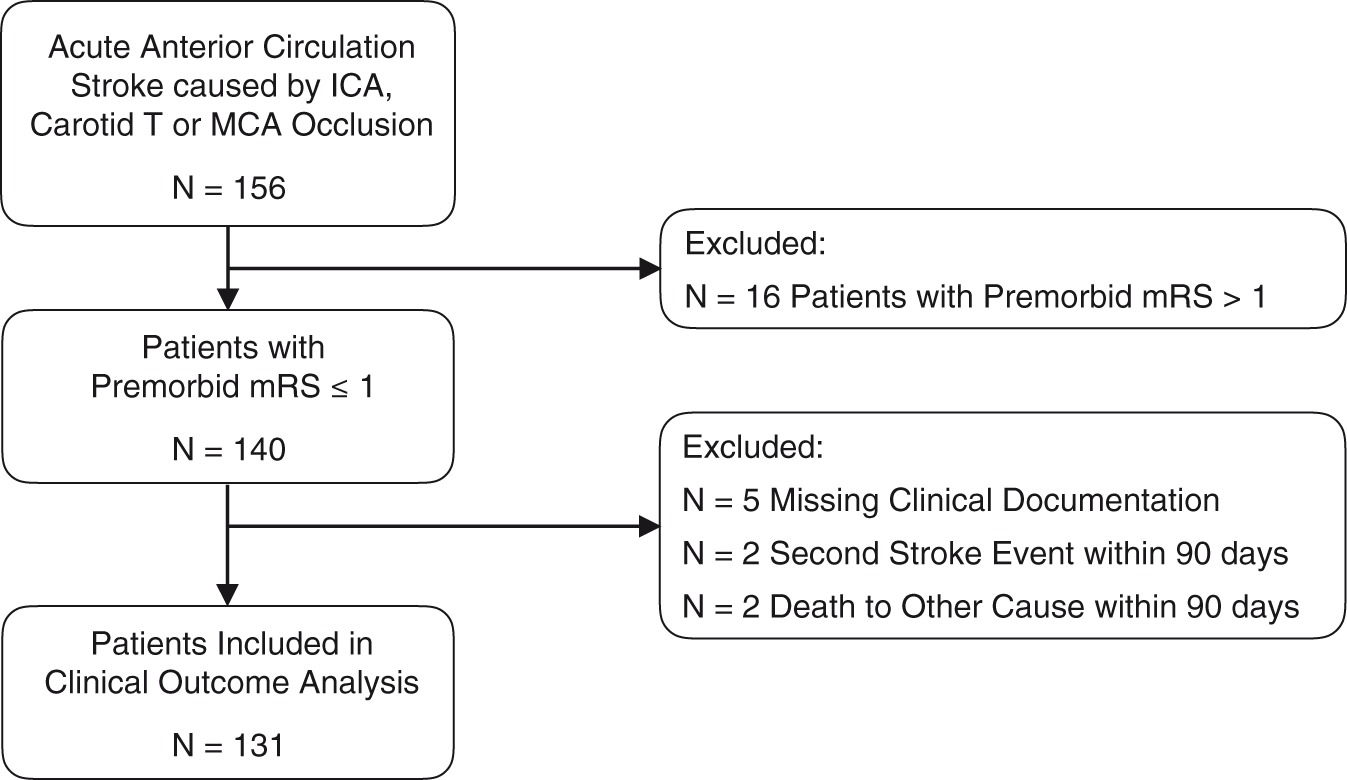
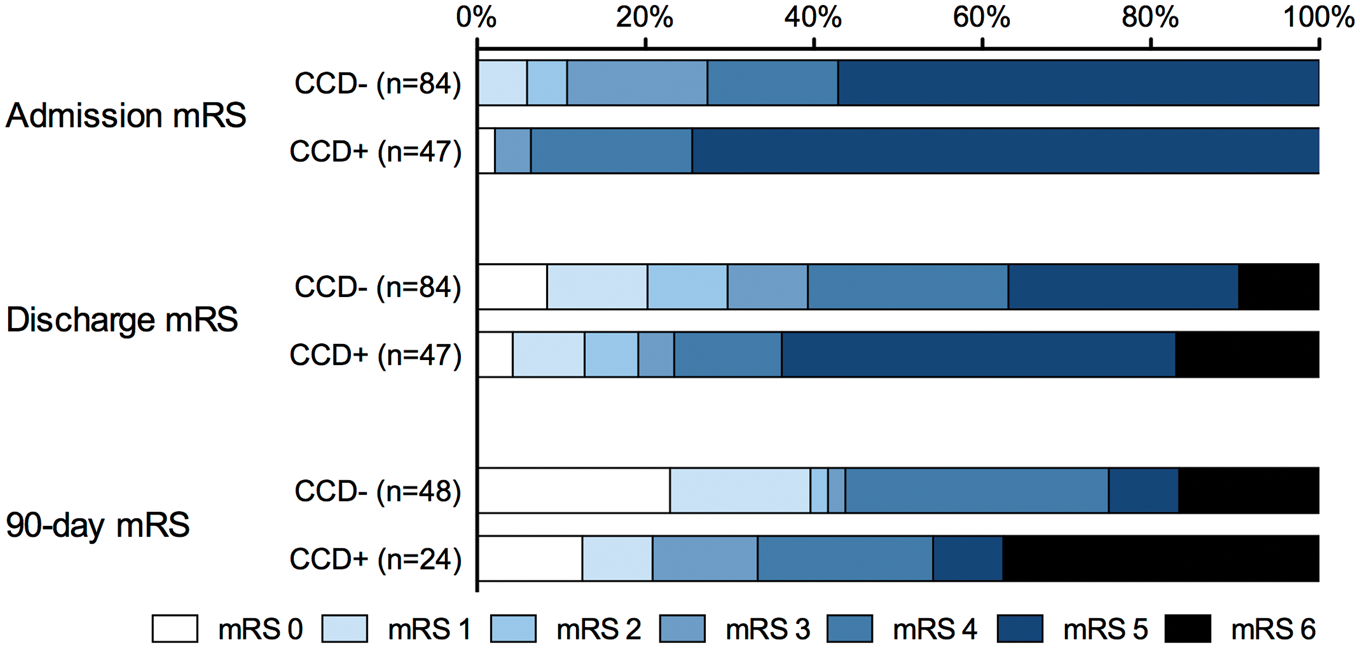
In the binary logistic regression analysis, no statistically significant associations between CCD and functional outcome parameters were evident (each with p > 0.05). Higher NIHSS scores on admission and larger final infarction volume had significant negative associations with favorable outcome parameters, and the administration of IV thrombolysis had a significant positive association with favorable discharge mRS. Results are presented in Table 2. Additional per-patient mRS data plots stratified for treatment groups and CCD status are provided in supplementary Figure 3.
Association of CCD with subacute stroke complications
Predictors of subacute stroke complications.
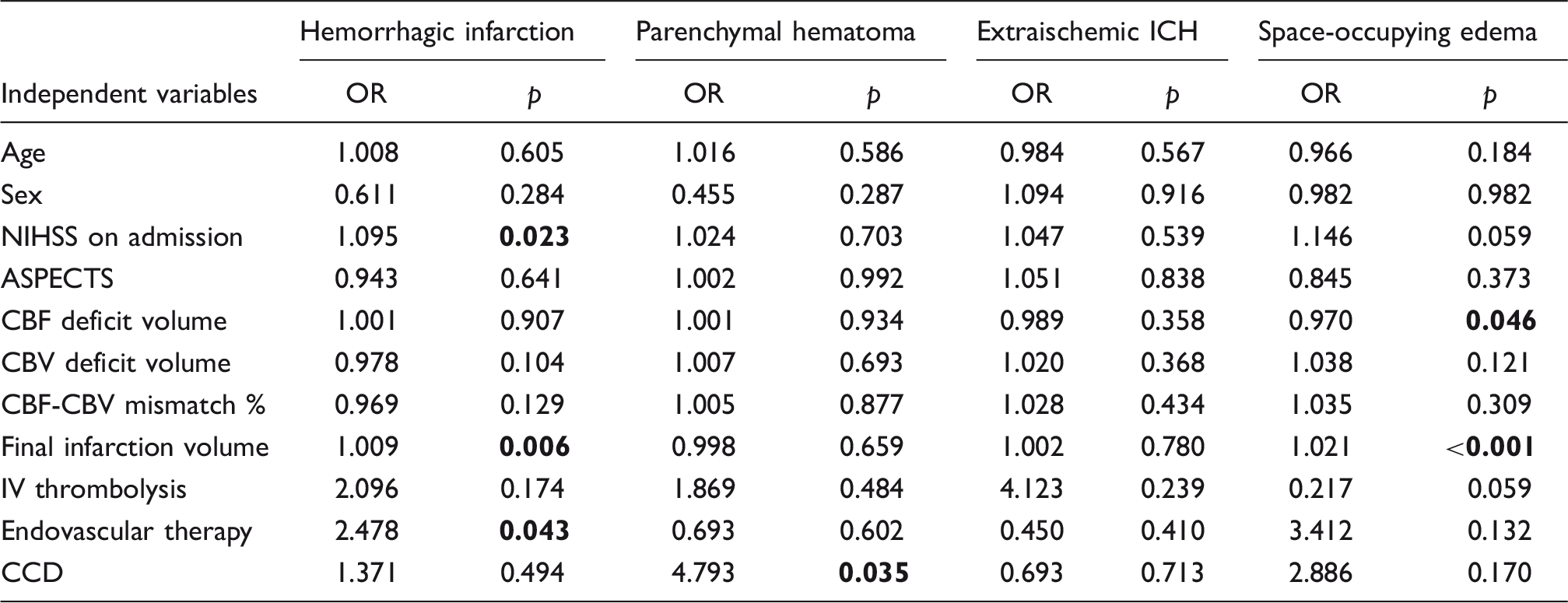
ICH: intracranial hemorrhage; NIHSS: national institutes of health stroke scale; ASPECTS: Alberta stroke program early CT score; CBF / CBV: cerebral blood flow / volume; OR: odds ratio.
Note: A binary logistic regression analysis was performed for the indicated complications for the patient selection according to Figure 2. Bold p values indicate statistical significance.
Discussion
In our study on patients with acute ischemic stroke due to an anterior circulation occlusion, the presence of CCD as assessed by WB-CTP in the acute phase showed no independent association with morphologic and functional patient outcome. Among the most common complications after acute ischemic stroke, the occurrence of parenchymal hematoma was associated with the presence of CCD.
CCD in the setting of ischemic stroke has mainly been studied in the subacute and chronic phase25,28,29. Although only few studies compared the phase-specific clinical implications of CCD, 25 the results suggest a negative impact of subacute and chronic CCD, but not acute CCD, on functional outcome.11,26 As possible associations between the presence of CCD with both functional outcome and complications in acute ischemic stroke are controversial,28,29 our study makes an important contribution to the significance of CCD as it is the first CT perfusion study on the clinical impact of acute CCD. Recent studies on CCD occurrence and perfusion characteristics suggest that CT perfusion is an appropriate technique to study the phenomenon of CCD.14,15
Building on this, we could show that the mere presence of CCD in the acute phase has no impact on functional outcome in ischemic stroke patients. The original notion of diaschisis being a reversible condition, either in the form of CCD or transhemispheric diaschisis, 43 is not contradicted by our observations as significant clinical improvement was observed in patients with acute CCD. Animal studies of transhemispheric diaschisis have proven reversibility of the acute flow alterations in the subacute phase of stroke. However, as WB-CTP is not established as a method of follow-up imaging due to reasons of radiation hygiene, our study cannot validate the reversibility of CCD in these clinically improved patients. Still, our results support previous reports suggesting that rather chronic than acute CCD is associated with irrevocable brain degeneration17,28 and impaired neurologic function. 25 The differences, however, might also be partially influenced due to other imaging techniques and inclusion criteria.
In contrast to functional patient outcome and final infarction volume, the occurrence of parenchymal hematoma was associated with the presence of CCD. As opposed to hemorrhagic infarction, parenchymal hematoma (in particular type II) has been shown to impair patient outcomes. 39 Besides IV thrombolysis, risk factors for parenchymal hematoma development are early ischemic changes, 44 prior medication with anticoagulants, 45 severe leukoaraiosis, 46 decreased cerebral blood flow, and increased blood-brain barrier permeability.47–52 However, we only observed a non-significant trend for IV thrombolysis treatment towards parenchymal hematoma development. This might be explained by the sample size, the inclusion criteria, and/or by clinical patient selection for IVT. From a pathophysiologic point of view, we could not establish an explanation linking CCD with parenchymal hematoma development. Neither size nor severity of the acute ischemic injury had an independent influence in our study population. Yet, after further clinical validation, CCD could potentially serve as a quickly identifiable prognostic parameter in routine clinical stroke work-up.
Similar to the impact of CCD on patient outcome measures, factors influencing occurrence and severity of CCD are controversial. In line with previous studies,9,11,28,29 we found a significant positive association between the size of the supratentorial perfusion deficit and CCD occurrence. It is important to note, however, that the final infarction volume was not associated with CCD. This result fits well with the missing association between CCD and functional outcome and with the proposed notion that acute CCD is a temporary and potentially reversible condition.
Our data must be interpreted in the context of the study design. As a first limitation, the most commonly used functional outcome measure, mRS at 90 days, was not available for all patients. Due to missing associations of CCD with other outcome measures that were fully available to us (discharge mRS, final infarction volume), we however expect that our results can be confirmed in larger prospective study cohorts. Second, the study was conducted in a retrospective fashion which does not allow a sample size estimation and power analysis. We would, however, like to point out that the present study includes the largest number of patients that were examined in the immediate stroke situation, underscoring the clinical relevance of the results. Finally, CCD presence was assessed qualitatively in a dichotomized fashion, reflecting current practice in the clinical routine. We cannot rule out that a quantitative approach would lead to different results.
In conclusion, our study suggests that the presence of CCD in the acute phase has no significant impact on functional outcome in patients with ischemic stroke. The association between acute CCD and occurrence of parenchymal hematoma, however, is a noteworthy finding that warrants further research.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conceived and designed the experiments: WGK, WHS, KMT. Performed the experiments: WGK, WHS, LVB. Analyzed the data: WGK, CH, MPF, FS, WHS, LVB. Wrote the manuscript: WGK, WHS, MPF, FS, FD, AEO, FGM, LVB, MFR, BEW, KMT.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
