Abstract
Selective therapeutic cooling is a promising technique for reducing final infarct volume and improving outcomes in ischemic stroke, especially as research regarding brain reperfusion continues to be explored. A recent study provided promising results on the safety and feasibility of selective therapeutic hypothermia via a closed-loop cooling catheter system for intra-carotid blood cooling in an ovine stroke model, but they failed to find efficacy of this method in this model. It is a major step forward from bench to bed side, but enhancing benefits of selective therapeutic cooling may need to take into account a more targeted induction of brain hypothermia and should mitigate potential side effects related to inducing hypothermia.
Compelling evidence supports the efficacy of hypothermia in experimental models of stroke. However, multiple clinical studies did not find benefits from hypothermia in stroke patients. Potential reasons include several mismatched variables that did not translate from bench to bedside. 1 Reperfusion can be achieved in most animal models with middle cerebral artery occlusion when filament is withdrawn, but the reperfusion could not be attained in many patients until the wide application of mechanical thrombectomy in 2015. Another mismatch is the time-delay in achieving the targeted temperature between models and patients. Therapeutic hypothermic (TH) is easily achieved in animal models before or after reperfusion without delay, but remains very difficult to achieve in patients.
Another promising approach for neuroprotection, which avoids the complications related to systemic therapeutic hypothermia, is selective therapeutic cooling. 2
A recent study by Cattaneo et al. proved the safety and feasibility of selective therapeutic cooling in an ovine stroke model, however, they did not find efficacy. 3 A closely-loop cooling catheter (CLCC) system for intra-carotid blood cooling was inserted into the right common carotid artery (CCA) in the hypothermia group. Selective therapeutic cooling was initiated 20 min before recanalization (which included ischemia for 3 h) and was maintained for 3 h. This protocol was feasible based on an intranasal temperature decrease of 2.06°C within the first 30 min of cooling and demonstrated to be safe with no histological and angiographical injuries in the carotid arteries related to inducing TH. However, they did not find therapeutic benefits of this protocol on infarct volume and neurological outcome. We want to pursue a discussion with the authors concerning how to enhance the benefits of selective TH and minimize side effects during the induction of selective TH.
Targeted (selective) hypothermia might provide enhanced efficacy and benefits. Since the CLCC system was only placed at the right CCA, a large amount of hypothermic blood traveled through the external carotid artery. 3 This could induce a rapid reduction of rectal temperature and brain (nasal) temperature at the same time. However, if CLCC system were placed at the internal carotid artery (ICA) or middle cerebral artery (MCA), as what had been done in rats, 2 it could induce a more targeted reduction of brain (nasal) temperature and a relatively normal rectal temperature. The efficacy of selective brain hypothermia might be related to both a rapid decrease of brain temperature and a notable difference between brain (nasal) and rectal temperature.
One disadvantage of the CLCC system was that the addition of the cooling sheath increased the diameter of the catheter and limited its further placement in the ICA or MCA (Figure 1). 3 Selective brain cooling can also be achieved through intra-arterial infusions (via the micro-catheter during mechanical thrombectomy). Both pre-reperfusion and post-reperfusion flushing and infusion with cold saline solutions decreased brain temperature, reduced ischemia/reperfusion injury, and improved functional outcomes in rat models of stroke (Figure 1). 4 This method had advantages of selective and rapid brain cooling (directly cooling MCA-supplying areas), and a combined use with mechanical thrombectomy. 5 This protocol also showed benefits in monkey stroke models and stroke patients. 6 , 7 However, the rate and duration of brain cooling was volume dependent, and a higher load of saline was potentially associated with side effects, such as abnormal hemodynamic parameters and serum electrolytes. Several studies found cold saline supplemented with magnesium sulfate or low-dose albumin had greater neuroprotective benefits than saline alone in rat models. 5 Focal perfusion of circulating cooled blood had been reported to reduce the infarction volume and improve neurological outcomes in a rat model of MCA occlusion. 8 The safety and feasibility of this method needs to be further tested in a higher order brain.
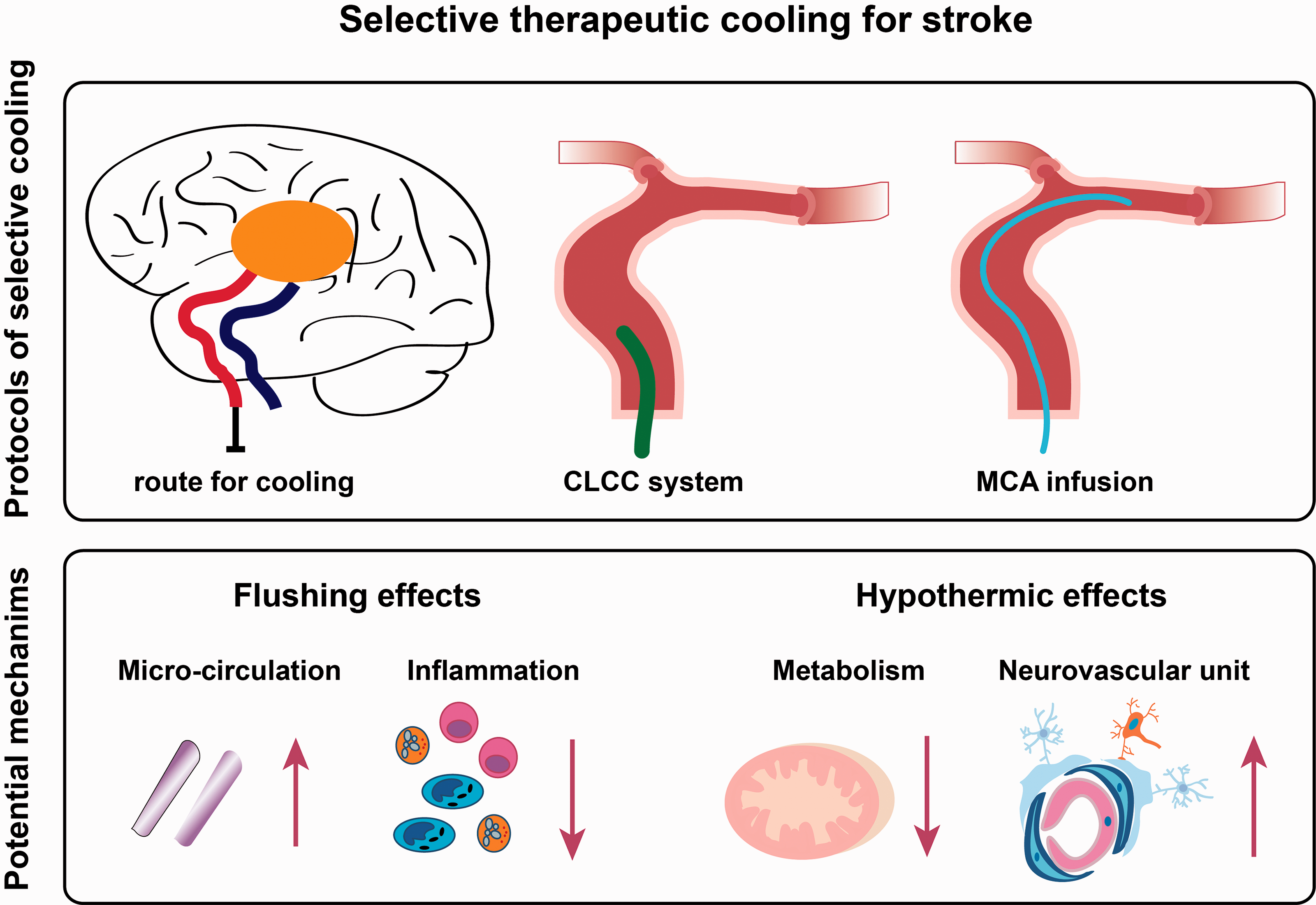
The schematic diagram exhibits cooling methods by a closed-loop cooling catheter (CLCC) system and middle cerebral artery (MCA) infusion. Potential mechanisms include local flushing and hypothermic benefits.
Minimizing the side effects related to selective brain cooling should also be explored. Vasospasm, a notable side effect during endovascular procedures, is a major concern and can significantly affect outcomes. In this study, both a higher mean flow velocity at the end of cooling and a severe hypo-perfusion based on CT perfusion in the hypothermia group indicated a long-term vasospasm, even after the withdrawal of the CLCC system. 3 Cerebral vasospasm had been previously reported in clamping the middle cerebral artery of the squirrel monkey during microscopic surgery. 9 Severe cerebral vasospasm and arterial ischemic stroke were reported in children receiving endovascular procedures. 10 Vasospasm was also observed when ischemic stroke was induced in rhesus monkeys. Some measures were taken to avoid or minimize cerebral vasospasm related with endovascular procedures, such as pre-setting an angle to the micro-catheter and giving Papaverine Hydrochloride to prevent vasospasm. 6 Additionally, local cooling can be easily performed using the same endovascular catheter during a single procedure, while the total procedure duration, including duration of anesthesia, would inevitably be prolonged. 5 Therefore, a brief selective brain cooling is preferred, although depth and duration of hypothermia should be further explored.
Recent clinical studies have demonstrated that 70%–80% of patients with large-vessel occlusion achieved successful reperfusion after receiving endovascular treatment. 1 These successes revive the opportunity to retest previously failed approaches, especially those that target multiple injury phases and neurovascular units, such as selective brain cooling. 7 The optimization of selective brain cooling protocols must be explored to reduce brain damages and potentially improve clinical outcomes in stroke patients.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (82071466, 82071312, 81871022, and 82171304).
Acknowledgments
The authors also thank Patrick Coan for language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
