Abstract
Calcitonin gene-related peptide (CGRP) can cause migraines, yet it is also a potent vasodilator that protects against hypertension. Given the emerging role of CGRP-targeted antibodies for migraine prevention, an important question is whether the protective actions of CGRP are mediated by vascular or neural CGRP receptors. To address this, we have characterized the cardiovascular phenotype of transgenic nestin/hRAMP1 mice that have selective elevation of a CGRP receptor subunit in the nervous system, human receptor activity-modifying protein 1 (hRAMP1). Nestin/hRAMP1 mice had relatively little hRAMP1 RNA in blood vessels and intravenous injection of CGRP caused a similar blood pressure decrease in transgenic and control mice. At baseline, nestin/hRAMP1 mice exhibited similar mean arterial pressure, heart rate, baroreflex sensitivity, and sympathetic vasomotor tone as control mice. We previously reported that expression of hRAMP1 in all tissues favorably improved autonomic regulation and attenuated hypertension induced by angiotensin II (Ang II). Similarly, in nestin/hRAMP1 mice, hypertension caused by Ang II or phenylephrine was greatly attenuated, and associated autonomic dysregulation and increased sympathetic vasomotor tone were diminished or abolished. We conclude that increased expression of neuronal CGRP receptors is sufficient to induce a protective change in cardiovascular autonomic regulation with implications for migraine therapy.
Keywords
Introduction
Migraine is a complex, multifactorial neurovascular disorder that has a significant impact on society with a life-time incidence of 43% in women and 18% in men.1,2 Despite this prevalence, the underlying mechanisms that contribute to migraine remain speculative. Migraine was long thought to be a vascular disease and is linked with increased risk of ischemic and hemorrhagic stroke,3–5 although the significance of any vascular connection has been a major topic of debate.6–9 However, actions on the vasculature could contribute to migraine independently of vasodilation per se, for example by release of agents, such as nitric oxide and inflammatory mediators that could sensitize peripheral nerves.10,11 Indeed, given that migraine is a neurovascular disorder, it is perhaps not surprising that the neuropeptide calcitonin-gene-related peptide (CGRP) is a key player in migraine pathogenesis. 12
CGRP is a multifunctional neuropeptide expressed throughout the nervous system and its receptors are widely distributed in the brain and periphery. 13 The canonical CGRP receptor is a multimeric G-protein-coupled receptor consisting of calcitonin receptor-like receptor (CLR) and receptor activity-modifying protein 1 (RAMP1). RAMP1 is required for trafficking of CLR to the cell surface and high-affinity binding of CGRP. 14 Recently, it has been recognized that RAMP1 also forms a second CGRP receptor by interacting with the calcitonin receptor (CTR). 15 Importantly for the transgenic mice described below, recombinant CGRP receptors containing combinations of mouse or human RAMP1 (hRAMP1) have similar CGRP binding activities. 16 Studies from our laboratory have demonstrated that increasing expression of RAMP1 in trigeminal ganglia and vascular smooth muscle enhances functional responses to CGRP, both in vitro and in vivo.16–19 Thus, RAMP1 is rate-limiting for CGRP receptor-mediated signaling.
Since CGRP is one of the most potent vasodilators known, 20 its antihypertensive actions are commonly attributed to a direct effect of CGRP on arterioles influencing vascular resistance. CGRP-containing nerve fibers heavily innervate the vasculature, 20 and CGRP and/or CGRP receptor expression is elevated in various models of hypertension, including animals and humans infused chronically with angiotensin II (Ang II).21–23 While pharmacological inhibition of CGRP receptors does not alter mean arterial pressure (MAP) of normotensive subjects, blocking the receptors exacerbates hypertension in a number of experimental models.22,24,25 These findings suggest that CGRP may operate via a negative-feedback protective mechanism against the development of hypertension. The recent finding that hypertension and vascular oxidative stress, hypertrophy and fibrosis are enhanced in αCGRP knockout mice infused with Ang II implicates vascular mechanisms in the protection. 23 However, there may be other mechanisms that contribute to the ability of CGRP to protect against hypertension. CGRP and its receptors are found in sensory neurons and in brain regions that can influence blood pressure through their effects on parasympathetic and sympathetic nerve activity.26–29 In support of a non-vascular role, our previous results with transgenic mice with increased expression of hRAMP1 in all tissues showed that these mice were not only protected against hypertension induced by Ang II infusion, but also exhibited increases in baroreflex sensitivity and heart rate (HR) variability and decreased sympathetic vasomotor tone compared to control littermates. 30 These results suggest a neural component to the protection against hypertension.
In this study, we have restricted overexpression of hRAMP1 to the nervous system with nestin-Cre dependent expression of hRAMP1. The nestin/hRAMP1 transgenic mice have previously been shown to display a CGRP-sensitized phenotype for two migraine-associated symptoms, photosensitivity and mechanical allodynia,31–33 as well as enhanced metabolism. 34 We now report that nestin/hRAMP1 mice display very similar protection against Ang II-induced hypertension as seen with the ubiquitous hRAMP1 mice. In addition, the mice were also protected against phenylephrine (PE)-induced hypertension. These data demonstrate that neural CGRP receptors are sufficient for attenuating hypertension in a mouse model and that actions of endogenous CGRP on the vasculature are not required. This suggests that cardiovascular protective actions of CGRP receptors may lie within the brain and hence may not be directly affected by migraine therapeutic interventions designed to target CGRP in the periphery.
Materials and methods
Animals
Expression experiments were performed on male and female mice, while cardiovascular studies were done only with male mice to allow comparison with previous studies. 30 All mice were 10–20 weeks old, 20–30 g body weight, and housed on a 12:12-h light:dark cycle, with normal mouse chow and access to water. Hemizygous transgenic mice (strain 28412/3) with GFP flanked by loxP sites followed by the hRAMP1 cDNA and generation of nestin/hRAMP1 double transgenic mice, including PCR genotyping, have been described. 18 All procedures were approved by the University of Iowa Institutional Animal Care and Use Committee and the U.S. Department of Veterans Affairs Office of the Chief Veterinary Medical Officer. They were conducted in accordance with standards set by the National Institutes of Health Office of Laboratory Animal Welfare and are reported in accordance with ARRIVE guidelines.
GFP fluorescence imaging
Whole tissues were placed in cold KREBS buffer with glucose. Imaging was done using a stereo fluorescence microscope (Leica MZFIII) with an attached CCD camera (QImaging Digital Camera v100). The images were captured under bright field and with a GFP filter (425/60 nm excitation).
Quantitative PCR
Total RNA was isolated from tissues using Ambion Trizol Reagent (Thermo Fischer Scientific). RNA integrity was determined by agarose gel electrophoresis and concentration and purity was determined using NanoDrop ND1000 spectrophotometer. Total RNA (50 ng) was reverse transcribed into cDNA using PrimeScript RT reagent Kit (Takara Bio USA, Inc., Mountain View, CA) per manufacturer’s instructions. Real-time quantitative PCR (Q-PCR) was used to measure human RAMP1 (hRAMP1) (NM_005855), mouse RAMP1 (mRAMP1) (NM_016894), and β-Actin (NM_007393) levels using previously validated primer sequences18,30 and SYBR Advantage qPCR Premix (Takara Bio USA, Inc.). Reactions contained 225 ng cDNA and 50 nM of each primer in an 18 µl reaction and performed in triplicate using ABI SDS 7900 HT thermocycler. Analysis was performed using ABI SDS v2.4 software. The mRAMP1 and hRAMP1 Ct values were converted to absolute copy numbers and normalized to 100,000 copies of β-actin using standard curves generated with 1:10 serial dilutions of pGEM-QmRAMP1, pbsCX1-LEL-hRAMP1, and pGEM-Qmbeta-actin plasmids in 10 ng/ml yeast tRNA.
Animal surgery and assessment of cardiovascular/autonomic phenotypes
A radiotelemetry probe (PC10, DSI) that enabled measurement of arterial blood pressure, HR and locomotor activity was inserted into the thoracic aorta through the left common carotid artery as described previously.30,35 The mice were anesthetized beforehand with ketamine (91 µg/g, IP) and xylazine (9.1 µg/g, IP) and allowed a week to recover. Each animal was housed in individual cages in the Animal Care Facility, and monitored daily for signs of ill-health. Thereafter, changes in MAP, HR, and locomotor activity were measured over a minimum of 24 h at a sampling rate of 500 Hz for 10 s once every 5 min using the DSI Acquisition software. In addition, blood pressure was continuously recorded at a high sampling frequency of 2000 Hz for 1–2 h, between 10:00 a.m. and 1:00 p.m., to collect data for assessment of spontaneous baroreflex sensitivity (sBRS), HR variability, sympathetic vasomotor tone, and baroreflex blood pressure buffering capacity (see below for methods/protocols).
After baseline measurements were made, osmotic mini-pumps (model 1002, Alzet) containing either Ang II (1000 ng/kg/min) or PE (4000 ng/kg/min) were implanted subcutaneously on the back of the mice anesthetized with either 2% isoflurane or ketamine/xylazine. 30 Mice were randomly assigned to untreated and Ang II or PE groups. The comparison between Ang II and untreated mice was independently replicated in additional groups of mice. Blood pressure, HR and locomotor activity data were collected throughout the 14-days of drug infusion at 500 Hz (10 s once every 5 min). In addition, blood pressure data was collected at 2000 Hz at the end of week 1 and/or week 2 for the repeat assessment of sBRS, HR variability, sympathetic vasomotor tone, and baroreflex blood pressure buffering capacity.
In some protocols, a sterilized catheter (polyurethane, 0.84 mm outer diameter, 0.36 mm inner diameter) was inserted into the surgically exposed right jugular vein under either 2% isoflurane or ketamine/xylazine anesthesia. The catheter was exteriorized at the back of the neck, the incision site was closed with suture (coated Vicryl 6-0), and the mouse was allowed to recover for one day before measuring MAP responses to intravenous (IV) bolus injections of either CGRP (1, 2, 3 ng) or PE (20, 40, 60 µg) in conscious mice. We measured acute depressor responses to IV injections of CGRP to provide functional evidence that RAMP1 expression was not increased in peripheral vasculature of nestin/hRAMP1 mice.
sBRS for control of HR was calculated from spontaneous fluctuations in systolic arterial pressure and cardiac interbeat interval (instantaneous HR) when the mice were active using the sequence technique.30,35 Sequences of four-or-more consecutive blood pressure pulses where systolic arterial pressure and pulse interval were positively correlated (r2 > 0.85) were detected using HemoLab software. sBRS was calculated as the average slope of the systolic pressure-pulse interval relationships (Δms/ΔmmHg).
HR variability was calculated in the time domain from beat-to-beat measurements of systolic arterial pressure and interbeat interval recorded for 3 min when the mice were relatively inactive using Hemolab and Batch Processor software. The standard deviation (SD) of pulse interval measurements provided a general measure of HR variability. The root mean square of successive differences (RMSSD) in pulse intervals provided a measure of rapid, parasympathetic-mediated modulation of HR.30,36
Sympathetic vasomotor tone was calculated as the decrease in MAP following intraperitoneal (IP) injection of the ganglionic blocker chlorisondamine (12 µg/g) when the mice were relatively inactive.
Baroreflex blood pressure buffering capacity was assessed by measuring acute increases in MAP in response to bolus IV injections of PE, both before and after ganglionic blockade with chlorisondamine. The baroreflex opposes increases in MAP. Thus, interruption of the efferent limb of the baroreflex by ganglionic blockade enhances the PE-induced increases in MAP in proportion to the strength of the baroreflex blood pressure buffering capacity.37,38 The jugular vein catheters were chronically implanted during the baseline period or on day 12–13 of Ang II or PE infusion, and responses to PE injections recorded one day later.
At the end of each experiment, mice were euthanized with sodium pentobarbital (150 µg/g, IP) followed by cervical dislocation and radio-telemeters were removed, cleaned and sterilized for future use.
Data analysis
The results are expressed as means ± SEM. Significant differences were defined at p < 0.05. The number of animals in each group was derived from power analysis to yield robust, reproducible and unbiased results. For cardiovascular measurements, to minimize the number of mice used, multiple measurements were made in the same mouse. Statistical evaluation was performed using unpaired t-test to compare between two groups and two-way repeated measures ANOVA with Fishers protected least significant difference (PLSD) post hoc test to determine effects of genotype, drugs and ganglionic blockade (StatView SAS Institute, Cary, NC). For Q-PCR measurements, evaluation of mRAMP and hRAMP expression between control littermates and nestin/hRAMP1 mice and between male and female mice was performed using an ordinary one-way ANOVA with Bonferroni multiple comparisons test. Evaluation of total mRAMP1 and hRAMP1 levels compared to endogenous mRAMP1 was performed by two-way ANOVA, with Bonferroni test. Data were analyzed using GraphPad Prism software.
Results
Neural, but not vascular, expression of hRAMP1 in nestin/hRAMP1 mice
The GFP-hRAMP1 mice were crossed with mice expressing Cre recombinase under control of the nestin promoter, which is primarily active in CNS and peripheral nervous system neural progenitor cells and some isolated cells in the heart and kidney.39–41 However, nestin has also been reported to be transiently expressed in developing arteries 42 and expression of nestin-Cre has been reported in the vasculature. 43 To address the possibility that the nestin promoter might be driving cardiovascular expression of the hRAMP1 transgene in nestin/hRAMP1 mice, we analyzed expression of the GFP reporter and hRAMP1 RNA in the heart and vessels of the nestin/hRAMP1 mice. Expression of hRAMP1 is dependent on Cre, which is needed to remove an upstream GFP stop cassette. 18 Hence, in the absence of Cre activity, there should still be GFP fluorescence.
The double transgenic nestin/hRAMP1 mice still expressed GFP in the aorta, indicating that the GFP stop sequence had not been removed by Cre recombinase (Figure 1(a)). Likewise, GFP was still detected in the heart. For comparison, GFP was not detected in the brain cortices and trigeminal ganglia of nestin/hRAMP1 mice. As controls, GFP was detectable in tissues from parental single transgenic GFP-hRAMP1 mice, but not from single transgenic nestin-Cre mice (Figure 1(a)). The levels of hRAMP1 and endogenous mRAMP1 gene expression were measured by Q-PCR (Figure 1(b)). Unfortunately, there are no available antibodies for quantitative measurement of RAMP1 protein levels. Consistent with the GFP expression, there was very little detectable hRAMP1 RNA in the aorta or heart. In addition, there was little or no detectable hRAMP1 in mesenteric arteries. While there was some hRAMP1 detected in cerebral arteries (basilar, middle cerebral, and posterior communicating arteries), we cannot rule out contamination from neural tissue during the dissection procedure. As a control, in agreement with previous studies,18,34 the combined total of hRAMP1 and mRAMP1 RNA in the brain of the nestin/hRAMP1 mice was about 2-fold greater than the endogenous mRAMP1 RNA level (Figure 1(b)). Likewise, hRAMP1 was also abundant in the trigeminal ganglia, although the ∼10-fold overexpression is greater than our previous report of ∼2-fold.
18
The difference is apparently due to higher levels of hRAMP1 RNA detected in this study, which may reflect drift in the colony over the past decade or differences in Q-PCR amplification protocols. In addition, while the levels of endogenous mRAMP1 RNA in the aorta in this study are relatively high, similar to control littermates in a previous study,
16
an earlier study reported much lower levels.
19
The reason for this discrepancy is not known, but could reflect dissection of primarily the aortic arch in the early study,
19
versus more distal regions in this study and Bohn et al.
16
In either case, the relative abundance of endogenous mRAMP1 RNA in resistance vessels (cerebral and mesenteric arteries) is consistent with their sensitivity to CGRP. There were no statistical differences in mRAMP1 RNA levels between sexes or between genotypes and no statistical difference in hRAMP1 levels between sexes of the double transgenic nestin/hRAMP1 mice. As in previous studies, we detected little or no hRAMP1 RNA in the liver (Figure 1(b)). Thus, among the tissues examined, the only statistically significant increased expression of RAMP1 (total mouse and human) in nestin/hRAMP1 mice compared to endogenous mRAMP1 were the brain and trigeminal ganglia (Figure 1(c)).
RAMP1 expression in nestin/hRAMP1 transgenic mice. (a) Schematic of the GFP-hRAMP1 expression vector. The loxP sites that flank the GFP reporter allow for Cre-mediated excision of GFP and associated stop sequences when the parental mice are crossed with mice expressing Cre recombinase. Following mating with nestin-Cre mice, GFP expression was observed in the aorta and heart, but not brain or trigeminal ganglia of nestin/hRAMP1 double transgenic (R+C+) mice, indicating nestin-driven Cre removal of the GFP stop sequence in the brain and ganglia, but not the aorta and heart. As controls, GFP was detected in both tissues of single transgenic mice containing only the parental GFP-hRAMP1 transgene (R+C−) and not in single transgenic mice containing only the nestin-Cre transgene (R−/C+). (b) Q-PCR measurement of RAMP1 levels in nestin/hRAMP1 (R+C+) mice and control littermates (both R+/C− and R−C+). The copies of mouse and human RAMP1 RNAs were calculated from standard curves and normalized to 100,000 copies of β-actin mRNA. (c) Total RAMP1 RNA (combined mouse (m) and human (h) RAMP1) in nestin/hRAMP1 mice is significantly increased only in neural tissues compared to the averaged mRAMP1 expression in control littermates and nestin/hRAMP1 tissues. Data are means and SEM from four R+C+ mice (except liver, n = 8; heart, n = 5), and from five control mice in each group, *p < 0.05, **p < 0.01.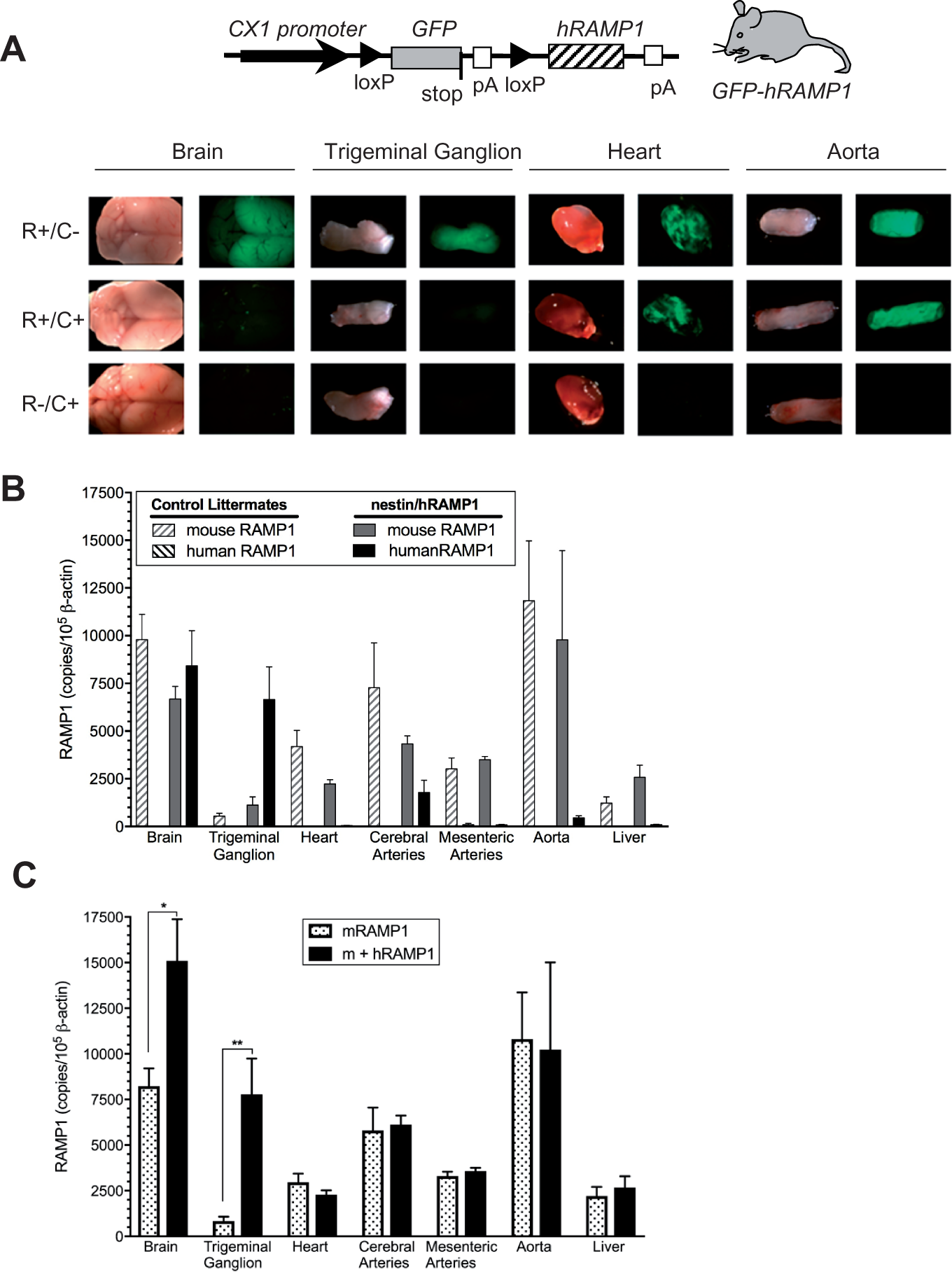
To provide functional evidence that RAMP1 expression was not increased in peripheral vasculature of nestin/hRAMP1 mice, we measured the blood pressure response to acute IV injections of CGRP. In a previous study, we demonstrated that CGRP-induced vascular relaxation was enhanced in mice with ubiquitous expression of hRAMP1 in all tissues including blood vessels.
19
In the present study, acute IV CGRP-induced decreases in MAP were slightly attenuated and not enhanced in nestin/hRAMP1 mice consistent with the lack of hRAMP1 RNA expression in blood vessels (Figure 2(a)). Acute IV CGRP-induced increases in HR were also slightly attenuated and not enhanced in nestin/hRAMP1 mice (Figure 2(b)). The reasons for the attenuation of the decreases in MAP and increases in HR are not clear. We speculate that the lesser decrease in MAP may reflect a greater baroreflex-mediated increase in sympathetic vasomotor tone that limited the fall in MAP.
Acute depressor response to CGRP is not enhanced in nestin/hRAMP1 mice. Shown are (a) decreases in mean arterial pressure (MAP) and (b) increases in heart rate (HR) in response to bolus IV injections of CGRP (1, 2, 3 ng) in control (n = 3) and nestin/hRAMP1 (n = 3) mice. These results are consistent with absence of transgene hRAMP1 expression in resistance arteries of nestin/hRAMP1 mice. *Significant decrease in MAP or increase in HR from baseline in response to CGRP (ANOVA, PLSD test, p < 0.05). †Significant decrease in magnitude of depressor response in nestin/hRAMP1 vs. control mice (ANOVA, PLSD test, p < 0.05). Data are means ± SEM.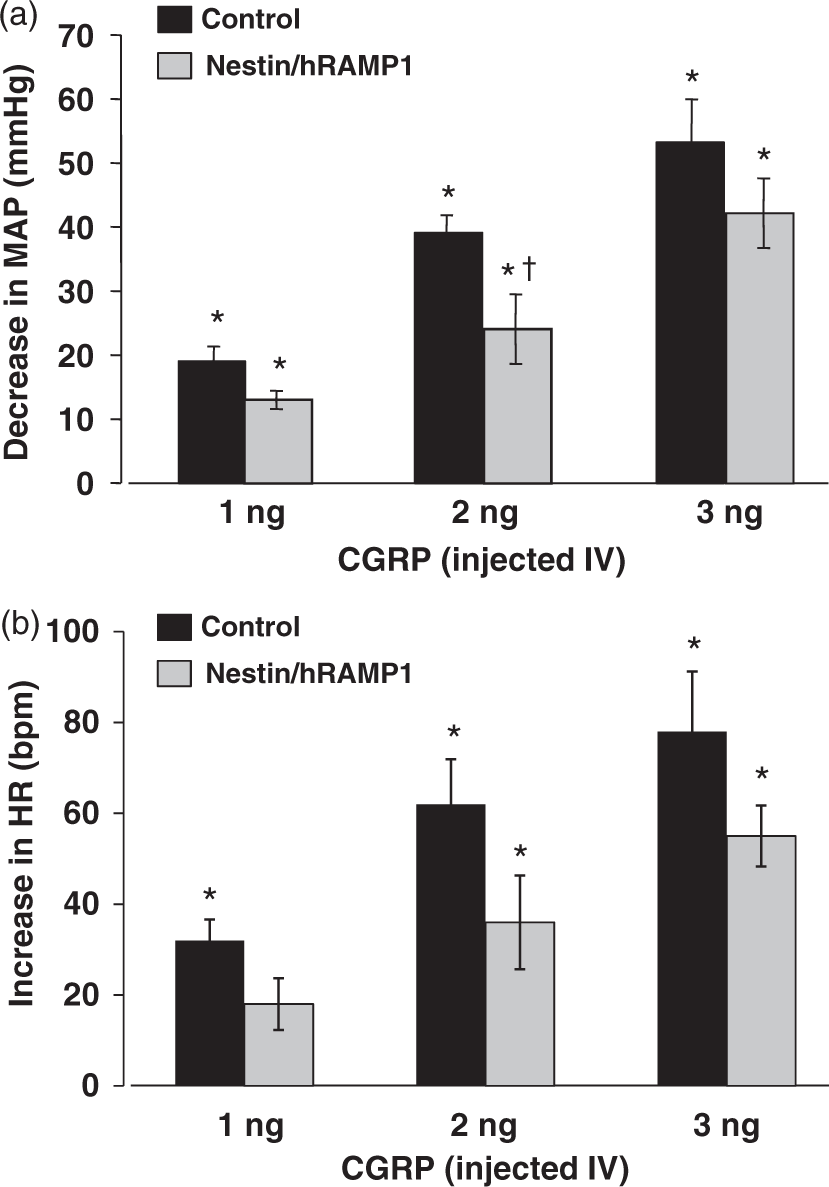
Cardiovascular/autonomic phenotypes in nestin/hRAMP1 versus control mice
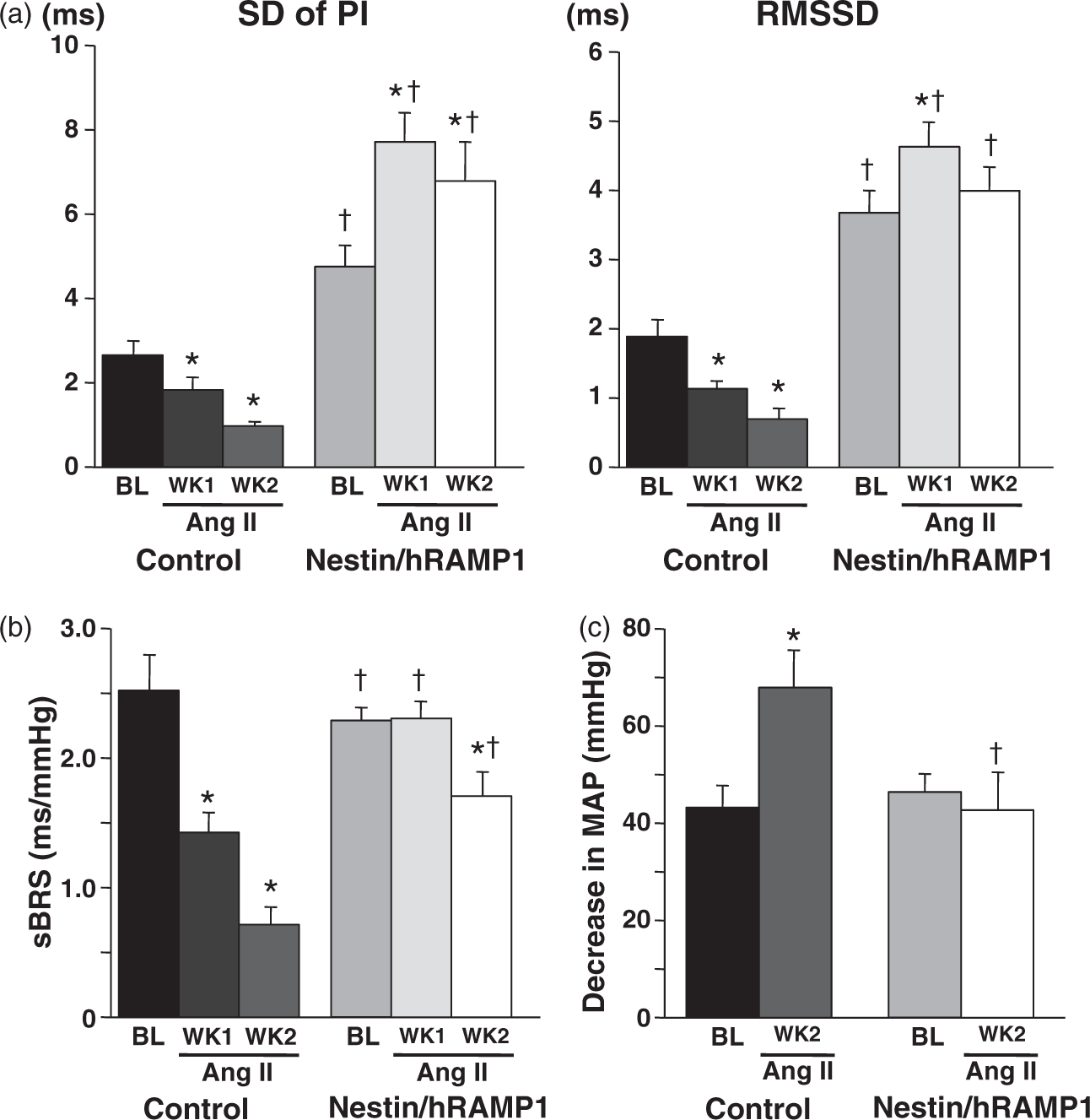
Twenty-four hour average values of MAP and HR did not differ in control and nestin/hRAMP1 mice, while locomotor activity was significantly lower in nestin/hRAMP1 mice (Figure 3). Although MAP and mean HR were not different in nestin/hRAMP1 mice and control littermates, HR variability (calculated as SD and RMSSD of pulse intervals) was higher in nestin/hRAMP1 mice (Figure 4(a)). While the SD of pulse intervals is influenced by both sympathetic and parasympathetic nerve activity, the RMSSD is dependent on parasympathetic modulation only.
36
sBRS and sympathetic vasomotor tone were not different in nestin/hRAMP1 versus control mice at baseline (Figure 4(b) and (c)).
Ang II-induced hypertension is strongly attenuated in nestin/hRAMP1 mice. Measurements (24 h avgs) of mean arterial pressure (MAP, top); heart rate (HR, middle); and locomotor activity (bottom) are shown for baseline (Day 0) and during continuous infusion of Ang II (1000 ng/kg/min, SC) for 14 days in control (n = 7) and nestin/hRAMP1 (n = 8) mice. The arrow indicates the beginning of Ang II infusion. *Significant increase from baseline in response to Ang II infusion (ANOVA, PLSD test, p < 0.05). †Significant difference in nestin/hRAMP1 vs. control mice during Ang II infusion (ANOVA, PLSD test, p < 0.05). Data are means ± SEM. Ang II-induced autonomic dysregulation in control mice is abrogated in nestin/hRAMP1 mice. Shown are measurements of: (a) HR variability quantified as SD of PI (standard deviation of pulse intervals) and RMSSD (reflecting parasympathetic modulation); (b) Spontaneous baroreflex sensitivity (sBRS) for control of HR. (c) Sympathetic vasomotor tone (decrease in MAP after ganglionic blockade). Measurements were obtained at baseline (BL) and after 7 days (WK1) and 14 days (WK2) of Ang II infusion in control (n = 7) and nestin/hRAMP1 (n = 8) mice. *Significant change from baseline in response to Ang II infusion (ANOVA, PLSD test, p < 0.05). †Significant difference between nestin/hRAMP1 vs. control mice (ANOVA, PLSD test, p < 0.05). Data are means ± SEM.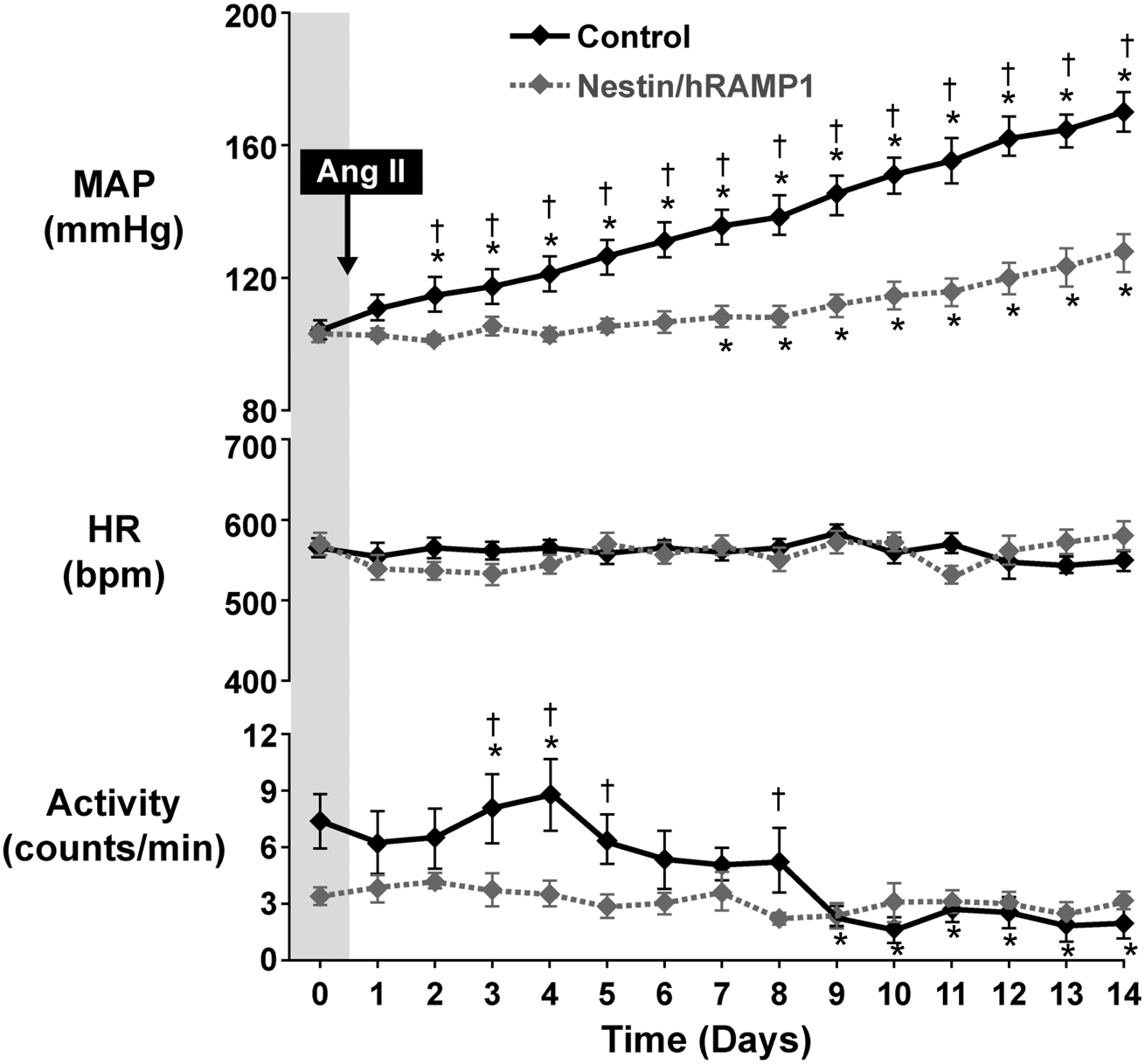
Baroreflex control of HR and blood pressure are differentially regulated. To evaluate baroreflex control of blood pressure, we measured acute increases in MAP in response to bolus IV injections of the vasoconstrictor PE, both before and after ganglionic blockade (Figure 5). The baroreflex opposes the PE-induced increases in MAP. Thus, interruption of the efferent limb of the baroreflex by ganglionic blockade enhances the acute PE-induced increases in MAP in proportion to the strength of the baroreflex blood pressure buffering capacity.37,38 Blood pressure buffering capacity was close to 2-fold greater in nestin/hRAMP1 mice versus controls (Figure 5(a)). The MAP just before injecting PE was not significantly different in untreated control and nestin/hRAMP1 mice, both before and after ganglionic blockade (Table 1), consistent with the similar MAP and sympathetic vasomotor tone measured independently on separate days (Figures 3 and 4(c)).
Nestin/hRAMP1 mice exhibit enhanced baroreflex blood pressure buffering capacity at baseline and after Ang II infusion. Enhancement of acute pressor responses to injections of phenylephrine (PE: 20, 40, 60 µg, IV) after ganglionic blockade (GB) reflects blood pressure buffering capacity. (a) Measurements obtained at baseline in control (n = 4) and nestin/hRAMP1 (n = 5) mice. (b) Measurements obtained after two weeks of Ang II infusion in control (n = 3) and nestin/hRAMP1 (n = 3) mice. GB was achieved by injection of chlorisondamine (12 µg/g, IP). Baroreflex buffering capacity (right panel) was calculated from enhancement of the pressor response to the 60 µg dose of PE. The absolute values of MAP measured just before the bolus injections of PE are provided in Table 1. *Significant difference after vs. before GB (ANOVA, PLSD test, p < 0.05). †Significant difference between nestin/hRAMP1 vs. control mice (unpaired t-test, p < 0.05). #Significant difference between Ang II infusion vs. baseline in control mice (unpaired t-test, p < 0.05). Data are means ± SEM. Mean arterial pressures (MAP) measured before bolus intravenous injections of phenylephrine, before and after ganglionic blockade, in untreated, Ang II-infused and PE-infused mice. PE: phenylephrine; Ang II: angiotensin II; GB: ganglionic blockade. Note: MAP (mean ± SEM) measured by radiotelemetry, units of measurement, mmHg. n= number of mice. MAP measured three times in each mouse and values averaged. After vs. before ganglionic blockade, p < 0.05. Ang II- or PE-infused mice vs. untreated mice, p < 0.05. Nestin/hRAMP1 mice vs. control mice, p < 0.05.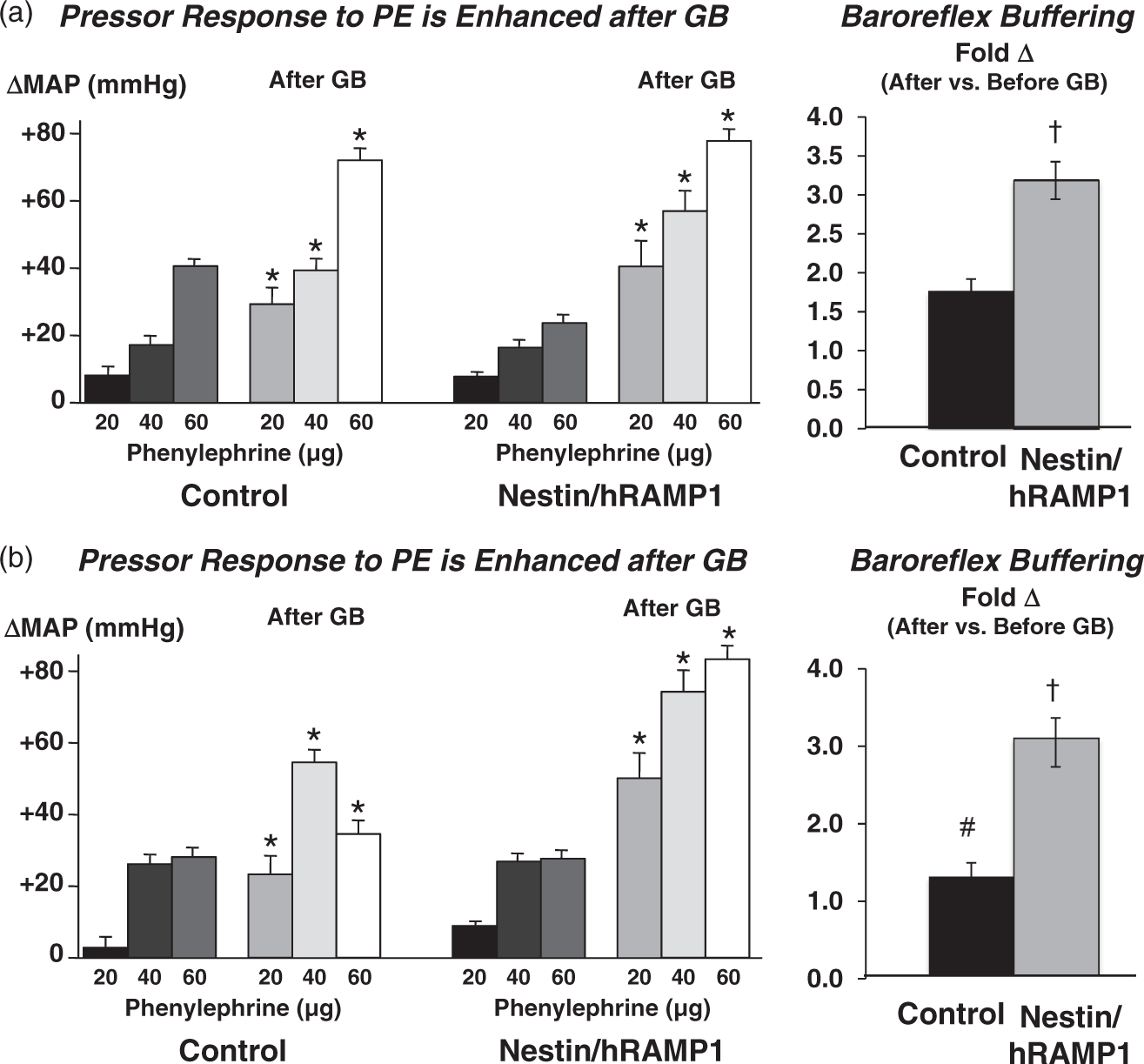

Nestin/hRAMP1 mice are protected from autonomic dysregulation and hypertension
In control mice, infusion of Ang II for two weeks increased MAP progressively, did not affect HR, and decreased locomotor activity (Figure 3). In contrast, the increased MAP during Ang II infusion was delayed and markedly attenuated in nestin/hRAMP1 mice (Figure 3). Ang II infusion did not affect HR in nestin/hRAMP1 mice, similar to control mice. Activity was significantly lower in nestin/hRAMP1 mice than in littermate controls at baseline, and remained at the same low level throughout the Ang II infusion (Figure 3).
Ang II infusion decreased HR variability, sBRS and baroreflex blood pressure buffering capacity, and increased sympathetic vasomotor tone in control mice (Figures 4 and 5). These deleterious effects of Ang II were nearly or totally abolished in nestin/hRAMP1 mice, with the decrease in HR variability being reversed to an increase (Figure 4). Baroreflex buffering of pressor responses to acutely injected PE remained much stronger in nestin/hRAMP1 mice than in control littermates after two weeks of Ang II infusion (Figure 5(b)).
To determine whether responses to Ang II infusion were secondary to hypertension or specific to Ang II, we infused nestin/hRAMP1 and control mice with the alpha adrenergic receptor agonist PE. While the initial rise in MAP was slower during PE infusion compared with Ang II, the severity of hypertension after two weeks of drug infusion was similar (MAP = ∼160–170 mmHg) (Figures 3 and 6(a), respectively). PE- and Ang II-induced decreases in sBRS were robust and essentially identical (Figures 4(b) and 6(b)). Sympathetic vasomotor tone increased significantly during both PE and Ang II infusions in control mice (Figures 4(c) and 6(c)). Nestin/hRAMP1 mice were markedly resistant to PE-induced autonomic dysregulation and hypertension (Figure 6). PE infusion did not significantly impair baroreflex blood pressure buffering capacity calculated using the 60 µg dose of PE in control mice (Figures 6(d) vs. 5(a), respectively), unlike Ang II (Figure 5(b)). MAP was significantly lower in nestin/hRAMP1 mice infused with either Ang II or PE than in control mice infused with either drug, not only before but also after ganglionic blockade (Table 1).
Phenylephrine-induced hypertension and autonomic dysregulation are strongly attenuated in nestin/hRAMP1 mice. (a) Mean arterial pressure (MAP) and heart rate (HR) measured at baseline (BL) and during PE infusion for two weeks (4000 ng/kg/min, SC). (b, c) Baroreflex control of HR (sBRS) and sympathetic vasomotor tone measured at BL and after two weeks of PE infusion. *Significant change from baseline in response to PE infusion (ANOVA, PLSD test, p < 0.05). †Significant difference between nestin/hRAMP1 vs. control mice (ANOVA, PLSD test, p < 0.05). Data are means ± SEM from four to eight mice in each group. (d) Baroreflex blood pressure buffering capacity calculated from the enhancement of the pressor response to the 60 µg dose of PE was measured after two weeks of PE infusion. *Significant enhancement of pressor response to PE after vs. before ganglionic blockade (GB) (ANOVA, PLSD test, p < 0.05). †Significant difference between nestin/hRAMP1 vs. control mice (ANOVA, PLSD test, p < 0.05). Data are means ± SEM from four to eight mice in each group.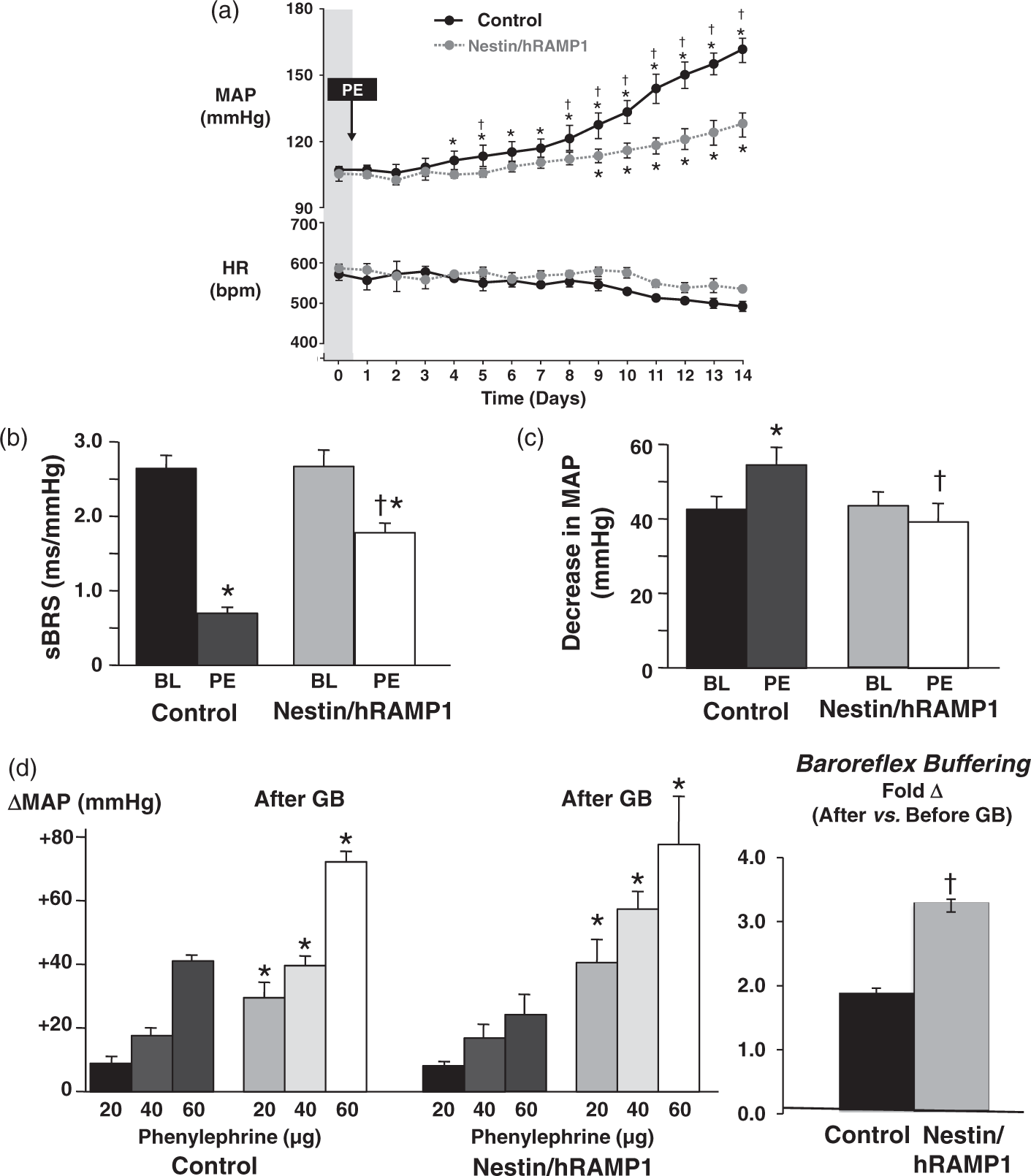
Discussion
Previously, we demonstrated that mice with ubiquitous transgenic upregulation of RAMP1 in all tissues have improved autonomic regulation and are resistant to Ang II-induced hypertension. 30 The increased baroreflex sensitivity and decreased sympathetic vasomotor tone observed in those mice suggested that neural CGRP mechanisms contributed to the attenuation of hypertension. 30 The results of the present study provide direct evidence of a major neural contribution to the antihypertensive influence of CGRP receptors, and extend the findings to another model of hypertension induced by infusion of the alpha-adrenergic receptor agonist PE. The similarities and differences between the two mouse lines, experimental hypertension models, and the relation of our results to previous studies in regards to mechanisms underlying cardiovascular protection by CGRP, along with implications of our findings for emerging therapies for migraine are discussed below.
Cardiovascular and autonomic phenotypes in experimental models
In many respects, the phenotypes in nestin/hRAMP1 mice are essentially the same as those we observed previously in mice with ubiquitous expression of hRAMP1. 30 Both models exhibited normal MAP throughout the day and night, and increased HR variability compared with littermate controls. Ang II-induced decreases in HR variability and sBRS, and increases in sympathetic vasomotor tone and MAP were abrogated in both transgenic models. HR variability actually increased above normal in both models during Ang II infusion. Interestingly, sBRS for control of HR measured at baseline was significantly higher in ubiquitous hRAMP1 mice than in littermate controls, 30 but was not different in nestin/hRAMP1 mice. We speculate that enhanced CGRP receptor signaling in the arterial wall may increase the compliance of large arteries, including carotid sinuses and aortic arch in which baroreceptor afferents terminate, which are an important determinant of BRS. 44 Overall, the similar phenotypes in both models suggest that the favorable autonomic and cardiovascular changes were mediated by neural mechanisms.
We initially focused on the Ang II infusion model of hypertension because of it being well characterized in many species including mice, the known interactions between CGRP and Ang II, 45 and the widely recognized importance of Ang II in human hypertension. 46 Furthermore, chronic administration of Ang II induces oxidative stress, inflammation, and increased sympathetic nerve activity,47–49 hallmarks of human hypertension. The strong neurogenic component to the hypertension in Ang II infused rodents is readily apparent when one considers that acute IV administration of the Ang II AT1 receptor blocker losartan causes only small decreases in MAP in Ang II-infused mice and rats, while injection of a ganglionic blocker causes large decreases in MAP.30,48
Nonetheless, we felt it was important to confirm our findings in another model of hypertension. We chose the PE infusion model because PE, unlike Ang II, is not strongly associated with oxidative stress and sympathoexcitation. Somewhat surprising to us, the autonomic phenotypes were very similar in PE-infused and Ang II-infused mice. Both Ang II infusion and PE infusion decreased sBRS and increased sympathetic vasomotor tone, and these changes were abrogated in nestin/hRAMP1 mice (see Figures 4 and 6). The results suggest that protective actions of neuronal CGRP receptors may be relevant to hypertension in general, and do not simply reflect an interaction with Ang II signaling. Previous studies demonstrating blood pressure lowering effects of endogenous CGRP in deoxycorticosterone-salt hypertension and endothelin 1-induced hypertension support this view.24,25
Mechanisms underlying cardiovascular protection by CGRP and RAMP1
Evidence for CGRP and RAMP1-mediated protection against hypertension comes primarily from studies of CGRP and RAMP1 deficient mice and administration of the CGRP receptor antagonist CGRP8-37 to hypertensive animals. Hypertension associated with inflammation in RAMP1 deficient mice has been reported. 50 CGRP−/− mice have exhibited variable blood pressure phenotypes dependent on methods used and whether the calcitonin exon was also deleted,51,52 or not deleted.23,53,54 Smillie et al. 23 convincingly demonstrated that normotensive CGRP−/− mice exhibit enhanced hypertension, oxidative stress and end-organ damage during infusion of Ang II for two and four weeks. 23 In that model, Ang II-induced changes in gene expression in aorta contributed to the vascular pathology. 23 More recently, Aubdool et al. 55 have shown that chronic infusion of a stable αCGRP analogue can both attenuate and reverse Ang II-induced hypertension, associated with decreases in oxidative stress, inflammation, fibrosis, hypertrophy and dysfunction in aorta, heart and kidney.
In our previous study, we observed changes in gene expression in the nervous system and vasculature that were predicted to play protective roles in hypertension. 30 In particular, Ang II infusion was found to increase CGRP and mRAMP1 RNA levels in the brain of both control and transgenic mice. CGRP and its receptor are expressed in key cardiovascular regulatory regions of the brain, including the nucleus tractus solitarius, hypothalamus, dorsal motor nucleus of the vagus, nucleus ambiguus, and amygdala.26,28,29 The importance of central nervous system (CNS) actions of CGRP in hypertension may have been underestimated in the past due to a preponderance of acute studies and limited ability of peripherally administered CGRP receptor antagonists to rapidly gain access to key central nuclei. We propose that increased CGRP receptor signaling in the nervous system of nestin/hRAMP1 mice improves autonomic regulation and decreases vasomotor sympathetic tone, thereby leading to subsequent lowering of arterial blood pressure. Further studies are needed to define the key anatomical sites of action and the underlying cellular/molecular mechanisms. In addition to the CNS sites mentioned above, the role of circumventricular organs such as area postrema and subfornical organ where circulating Ang II and CGRP may interact with each other and the nervous system should be explored.
Interestingly, MAP remained significantly lower in Ang II- and PE-infused nestin/hRAMP1 mice compared with Ang II- and PE-infused control mice after ganglionic blockade (Table 1). This finding suggests that an additional mechanism besides inhibition of sympathetic tone contributes to the antihypertensive actions of CGRP-RAMP1 signaling. Further studies are needed to explore this possibility. The finding that HR variability and baroreflex blood pressure buffering capacity are increased at baseline in nestin/hRAMP1 mice, in the absence of chronic hypertension, suggests that CGRP-RAMP1 signaling in the nervous system may also reduce risk of arrhythmias and excessive blood pressure variability independent of its antihypertensive actions.
Implications for treatment of migraine
The finding that CGRP receptors in the nervous system can mitigate the increased sympathetic vasomotor tone associated with hypertension may be relevant to current clinical trials for migraine using monoclonal antibodies that block CGRP or the CGRP receptor.56–60 There are valid concerns that complete blockade of CGRP activity could increase the risk of cardiovascular diseases, including hypertension, 61 although to date, there have not been indications of these adverse effects. Presumably, the antibodies are acting in the periphery since they do not readily cross the blood–brain barrier. 62 Our study suggests neural CGRP receptors are sufficient for the protective CGRP receptor actions reported, which could mean that these actions would not be affected by peripheral antibodies if the CGRP receptors are on neurons in the CNS. However, a major caveat is that our study cannot exclude CGRP actions on peripheral nerves and CGRP clearly has a close and complex reciprocal interaction with the sympathetic nervous system.13,63 Indeed, there are CGRP receptors on trigeminal ganglia cell bodies, although relatively few CGRP receptors have been detected on afferent endings.64,65 In this regard, it is important to note that the nestin/hRAMP1 mice, while sensitized to centrally administered CGRP, were not sensitized to peripheral CGRP in a photophobia-like behavioral assay. 66 This suggests that hRAMP1 expression on peripheral nerves may not be rate-limiting. Another caveat is that while there was no enhancement of CGRP-induced vasodilation in the nestin/hRAMP1 mice, we cannot exclude that expression of hRAMP1 in a subset of vessels, while not affecting the systemic blood pressure response to CGRP, could have played a protective role locally.
In summary, we predict that neural CGRP receptors, probably in the CNS, contribute to the protective actions of CGRP against hypertension. Indeed, it seems likely that similar neural CGRP receptor mechanisms could contribute to CGRP protection against not only hypertension, but also CGRP protection against vascular hypertrophy and oxidative stress reported by Smillie and colleagues. 23 Future studies with selective, tissue/organ-specific hRAMP1 overexpression should help define and distinguish CGRP receptor populations important for migraine and protective cardiovascular roles.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by research grants from the NIH (NS075599), U.S. Department of Veterans Affairs (1I01RX002101), and Department of Defense (W81XWH-16-1-0071 and W81XWH-16-1-0211) to AFR; NRSA F31 NS098825 to BNM; U.S. Department of Veterans Affairs (1BX001414) to MWC; and NIH (P01HL014388) to FMA and MWC.
Acknowledgments
The authors thank Anne-Sophie Wattiez and Brandon Rea for assistance and discussions, Larry Agbor and Curt Sigmund for advice on mesenteric artery dissection, and Robert Cornell for use of his microscope.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Russo reports grants and personal fees from Alder Biopharmaceuticals, grants and personal fees from Eli Lilly Pharmaceuticals, non-financial support from Pharmnovo, outside the submitted work. The other authors report no conflicts.
Authors’ contributions
RS performed the cardiovascular studies, BNM and AK performed the genotyping and expression studies, FMA, AFR and MWC helped with experimental design and interpretation; RS, BNM, AFR, and MWC wrote the manuscript.
