Abstract
In patients with major cerebral artery disease, lower blood pressure might reduce blood flow in the collateral pathways, thereby impairing the growth of cerebral collaterals, inhibiting hemodynamic improvement. We evaluated the hemodynamic status twice using positron emission tomography and 15O-gas, over time, in 89 medically treated patients with atherosclerotic internal carotid artery or middle cerebral artery disease that had no ischemic episodes during follow-up (mean, 28 ± 23 months). Changes in the mean hemispheric values of hemodynamic parameters in the territory of the diseased artery at follow-up were correlated with the mean blood pressure values at the baseline and follow-up examinations. There was a positive linear relationship between the degree of hemodynamic improvement and systolic blood pressure. Patients with low systolic blood pressure (<130 mmHg) (n = 18) showed hemodynamic deterioration as indicated by significant decreases in cerebral blood flow, cerebral blood flow/cerebral blood volume ratio, and increases in oxygen extraction fraction during follow-up. In contrast, there were no significant changes in patients without low systolic blood pressure. In patients with atherosclerotic internal carotid artery or middle cerebral artery disease and no ischemic episodes of stroke during follow-up, lower systolic blood pressure was associated with lesser hemodynamic improvement.
Keywords
Introduction
In patients with atherosclerotic internal carotid artery (ICA) or middle cerebral artery (MCA) disease, chronic hemodynamic impairment is a predictor of subsequent ischemic stroke.1–3 Furthermore, hemodynamic deterioration during follow-up also increases the risk of ischemic stroke.2,4,5 Therefore, early detection and appropriate treatment of chronic hemodynamic impairment is essential to improve patient prognosis.
Patients with ICA or MCA disease with hemodynamic impairment at the baseline that received appropriate treatment and suffered no intervening ischemic events during follow-up, show hemodynamic improvement in the long-term,6–9 which may be due to long-term improvement of collateral blood flow.10,11 Patients with hemodynamic impairment necessitating medical treatment have a high risk of subsequent ischemic stroke.5,12 However, if they are successfully treated, the hemodynamic impairment may improve, which could reduce the risk of ischemic stroke. Nevertheless, there is no definitive medical treatment strategy.
Hypertension is a major risk factor for stroke, and antihypertensive therapy is generally beneficial in patients with cerebrovascular diseases. 13 However, the level to which blood pressure (BP) should be lowered to achieve maximal benefit among patients with cerebrovascular disease is not precisely known, although post hoc analysis of PROGRESS 14 suggests a target of < 130 mmHg systolic BP (SBP). However, there were concerns that lowering BP might impair cerebral perfusion in patients with atherosclerotic major cerebral artery disease 15 and thereby exacerbate the degree of hemodynamic impairment and stroke risk.2,16 Our previous studies in patients with ICA or MCA disease showed that the incidence of ischemic stroke in the territory of the diseased artery in patients with low SBP (<130 mmHg) was higher than that in patients without.5,17 This was especially true in patients with impaired perfusion, as indicated by a decreased cerebral blood flow (CBF)/cerebral blood volume (CBV) ratio, determined on performing positron emission tomography (PET). 18 A higher stroke risk was also reported in patients with hemodynamic impairment due to vertebro-basilar artery disease and lower SBP. 19 In addition to possibly causing an acute hemodynamic crisis, aggressive lowering of BP might reduce intravascular flow rates and thereby impair the long-term growth of cerebral collaterals. 20 Thus, low BP might be associated with less hemodynamic improvement during follow-up in patients with atherosclerotic major cerebral artery disease and impaired perfusion, which may be associated with a sustained high stroke risk. However, the relationship between long-term changes in cerebral hemodynamics and BP during follow-up is unclear.
The purpose of this study was to determine whether lower SBP is associated with lesser hemodynamic improvement in patients with ICA or MCA occlusive disease and no ischemic episodes during follow-up.
Methods
Patients
Patient characteristics.
ICA: internal carotid artery; MCA: middle cerebral artery; SD: standard deviation; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker; SBP: systolic blood pressure.
Inclusion criteria were as follows: (1) occlusion or stenosis of the extracranial ICA (>60% diameter reduction according to the NASCET criteria 21 ) or intracranial ICA or MCA (>50% diameter reduction according to the WASID criteria 22 ) as documented by conventional or magnetic resonance angiography, (2) functional independence in daily life (a modified Rankin Scale score <3), (3) for symptomatic patients, history of transient ischemic attack (TIA) or minor completed stroke in the ICA or MCA distributions, (4) medically treated patients with no intervening TIA or stroke since the first PET examination, and (5) availability and willingness to return for follow-up PET examination. TIA was defined as focal symptoms of presumed ischemic cerebrovascular origin lasting < 24 h. Exclusion criteria were: (1) history of vascular reconstructive surgery and (2) presence of potential sources of cardiogenic embolism.
Of 89 patients, 28 were asymptomatic, 21 had TIA, and 40 had completed stroke. In the same patient cohort, 36 had ICA occlusion, 14 had ICA stenosis, 30 had MCA occlusion, and 9 had MCA stenosis. The interval between the first and follow-up PET studies ranged from 3 to 112 months (mean, 28 ± 23 months).
In terms of vascular risk factors, hypertension, diabetes mellitus, ischemic heart disease, hypercholesterolemia, and smoking were evaluated from the patient’s history recorded at the first PET examination. Hypertension, diabetes mellitus, ischemic heart disease, or hypercholesterolemia was judged to be present when there was a history of treatment.
All protocols in this study were approved by the Shiga Medical Center for Adults Institutional Review Board, the Human Study Committee (number 119), and all participants gave written informed consent. All experiments were performed in accordance with the Declaration of Helsinki and Good Clinical Practice.
PET measurements
PET scans were performed in each patient using an Advance whole-body scanner (General Electric Medical Systems, Wauwatosa, WI), which permits the simultaneous acquisition of 35 image slices with an inter-slice spacing of 4.25 mm. 23 Performance tests showed the intrinsic resolution of the scanner to be 4.6 to 5.7 mm in the transaxial direction and 4.0 to 5.3 mm in the axial direction. A transmission scan using 68Ge /68Ga was performed for attenuation correction in each subject before the tracer administration. To reconstruct PET data using filtered back projection, images were blurred to 6.0 mm full width at half maximum in the transaxial direction using a Hanning filter. Functional images were reconstructed as 128 × 128 pixels, with each pixel representing an area of 2.0 mm × 2.0 mm.
After a transmission scan, a series of 15O-gas studies were performed. 23 A small cannula was placed in the left brachial artery for blood sampling. C15O2 and 15O2 were delivered continuously to the patient via a mask for the duration of a 5-min scan. Arterial blood was sampled manually from the brachial artery five times (at 0, 1, 2, 3, and 4 minutes) and three times (at 0, 2, and 4 minutes) during the scanning, respectively. CBV was measured based on bolus inhalation of C15O with scanning for 3 min. Arterial samples were obtained manually twice (at 1 and 2 min) during the scanning. The radioactivity of the radiotracer, oxygen content, and hematocrit was measured. BP was measured through an arterial catheter connected to a pressure transducer. Averaged values of BP during C15O2 and 15O2 scanning were used as the values of BP at the PET examinations.
We used the steady-state method to calculate the CBF, cerebral metabolic rate of oxygen (CMRO2), and oxygen extraction fraction (OEF). 24 The CMRO2 and OEF were corrected based on CBV. 25 The ratio of CBF to CBV was calculated pixel-by-pixel as an indicator of cerebral perfusion pressure.18,26
To obtain normal control values of the 15O-gas PET variables, we performed 15O-gas studies with arterial sampling in seven normal volunteers (four men and three women), aged 47 ± 7 (mean ± SD) years that underwent normal routine neurological examinations and MRI scans.
Data analysis
We analyzed 10 tomographic planes, located 46.25–84.50 mm above and parallel to the orbitomeatal line.
27
The lowest plane corresponded to the level of the basal ganglia and the thalamus, and the uppermost plane corresponded to the level of the centrum semiovale. The software for image analysis was programmed by a member of our research team. A region-of-interest (ROI) was selected for CBF images. Each image was examined by compactly placing 10–12 circular ROIs (diameter, 16 mm), over the gray matter of the outer cortex in each hemisphere (Figure 1). According to the atlas,
28
the ROIs in all 10 images covered the distribution of the MCA as well as the external border-zone regions.
27
The same ROIs were used for the CMRO2, OEF, and CBV images. The mean hemispheric value for the hemisphere affected by ICA or MCA disease was calculated as the average of all the circular ROIs. In a test-retest paradigm, alignment and placement of the same ROIs on the initial and follow-up scans is ideal. Although to this end, the method for three-dimensional MRI-PET imaging coregistration of both the initial and follow-up scans is preferable, we chose the method stated above as we could not obtain three-dimensional MRI images in all patients: we placed the same size and number of ROIs contiguously, on many planes in the initial and follow-up scans and analysed the value in the hemisphere, not in the small region, which could help to diminish the error.
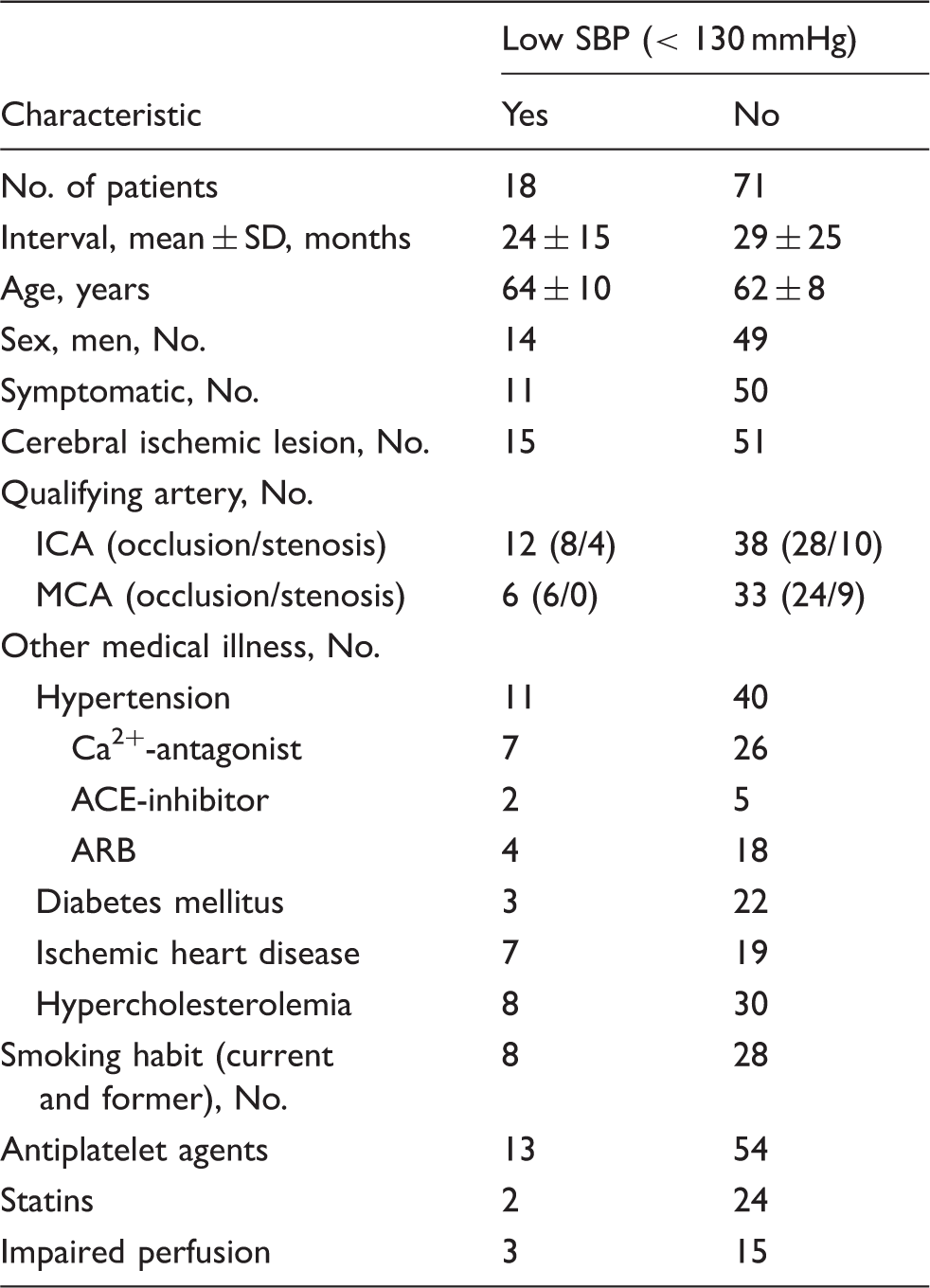
An example of the ROIs on the CBF image.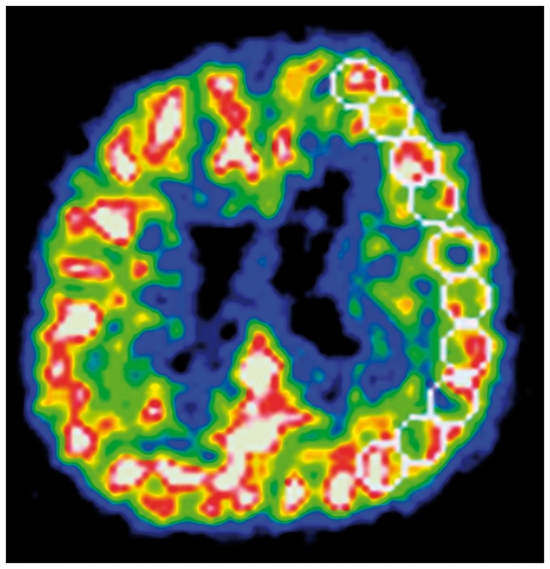
In six patients with infarction in the cerebral cortex, the circular ROIs that overlapped low-intensity areas on T1-weighted magnetic resonance imaging (MRI) were excluded from analysis using a simple method correlating PET images with magnetic resonance images. 29 We used routine MRI imaging (T1-weighted, T2-weighted, or fluid-attenuated inversion recovery imaging) which was only obtained for clinical purpose.
The mean OEF value obtained from the 14 control hemispheres in seven normal volunteers was 44.5% ±3.8%. Hemispheric OEF values beyond the 95% upper limit defined in normal subjects (above 52.9%) were considered to represent increased OEF. Comparative values for CBF and CBF/CBV in normal controls were 44.6 ± 4.5 and 11.4 ± 1.8, respectively. Hemispheric CBF and CBF/CBV values below 35.0 mL/100 g/min and 7.6/min, respectively, were considered abnormal.
Patients with increased OEF, decreased CBF, and decreased CBF/CBV in hemispheres with arterial disease were categorized as having misery perfusion, while patients with decreased CBF/CBV were categorized as having decreased cerebral perfusion pressure (impaired perfusion). Patients were categorized by an investigator who was unaware of their clinical status.
At follow-up examinations, the total change in the CBF, CMRO2, OEF, CBV, or CBF/CBV values in the MCA distribution with arterial disease was calculated by subtracting the values obtained at the follow-up examination from those obtained at the first examination. BP during the follow-up period was defined as the averaged values of BP at baseline and follow-up PET examinations. Low SBP was defined as SBP < 130 mmHg.
Statistical analysis
The clinical background or PET variable values were compared between groups using the Student's t-test or Fisher's exact test, as appropriate. PET variable values between examinations were compared using paired t-tests. The relationships between variables were analyzed using simple or multiple regression analyses. A multiple regression model with a forward stepwise selection procedure was used to test the independent predictive value of the low SBP during follow-up, the presence of impaired perfusion at baseline, and the presence of clinical characteristics (shown in Table 1) with respect to the change in PET variables during follow-up. For all analyses, statistical significance was set at p < 0.05.
Results
Eighteen of the eighty-nine patients had low SBP (<130 mmHg) during follow-up (Table 1). These same patients also had low diastolic BP (<70 mmHg). None of the patient characteristics were significantly different between patients with low SBP and those without. However, the use of statins in the patients with low SBP tended to be lower (p = 0.09) than that in the patients without.
15O-gas PET variables.
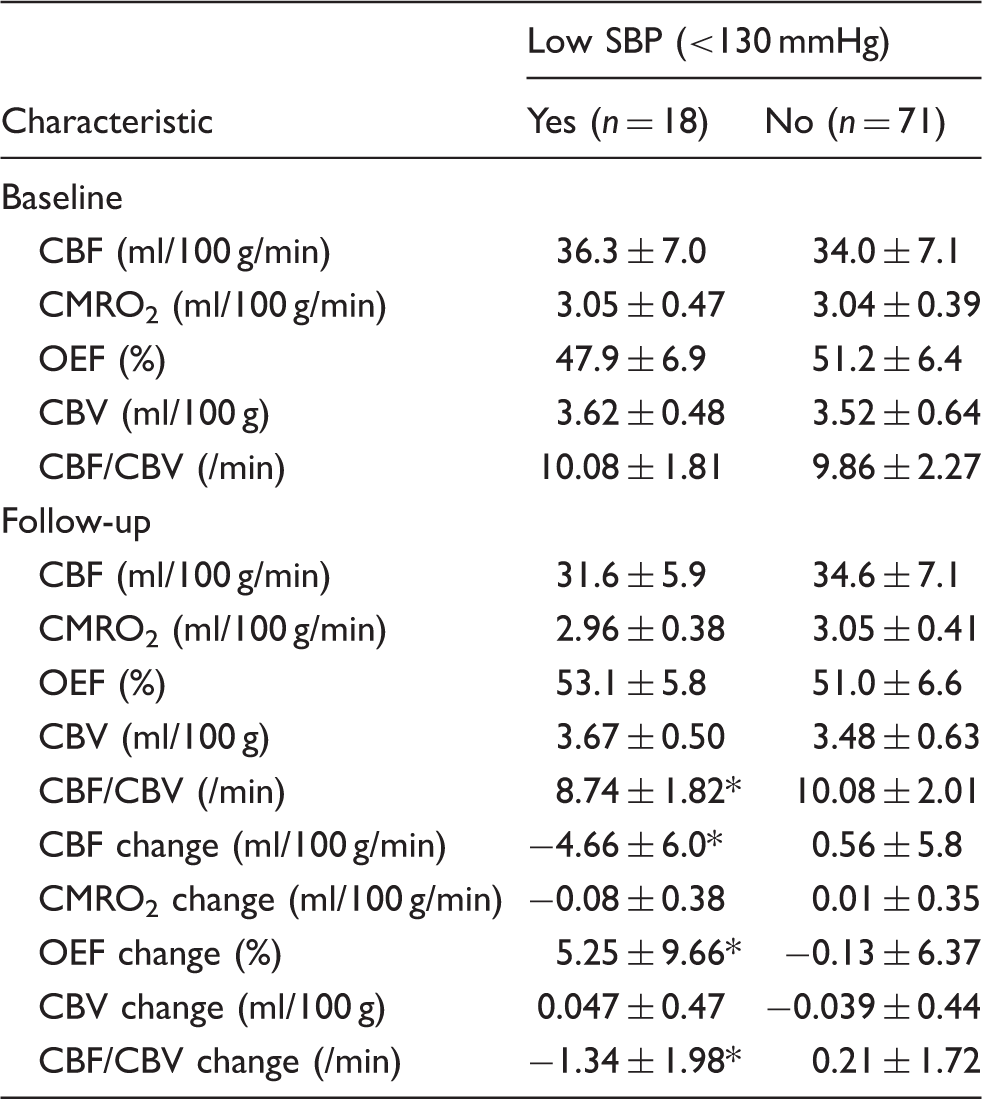
PET: positron emission tomography; CBF: cerebral blood flow; CMRO2: cerebral metabolic rate of oxygen; OEF: oxygen extraction fraction; CBV: cerebral blood volume.
Note: Normal values for CBF, CMRO2, OEF, CBV and CBF/CBV in the seven controls were 44.6 ± 4.5, 3.43 ± 0.33, 44.5 ± 3.8, 3.98 ± 0.48 and 11.4 ± 1.8, respectively.
*p < 0.05 vs. No group.
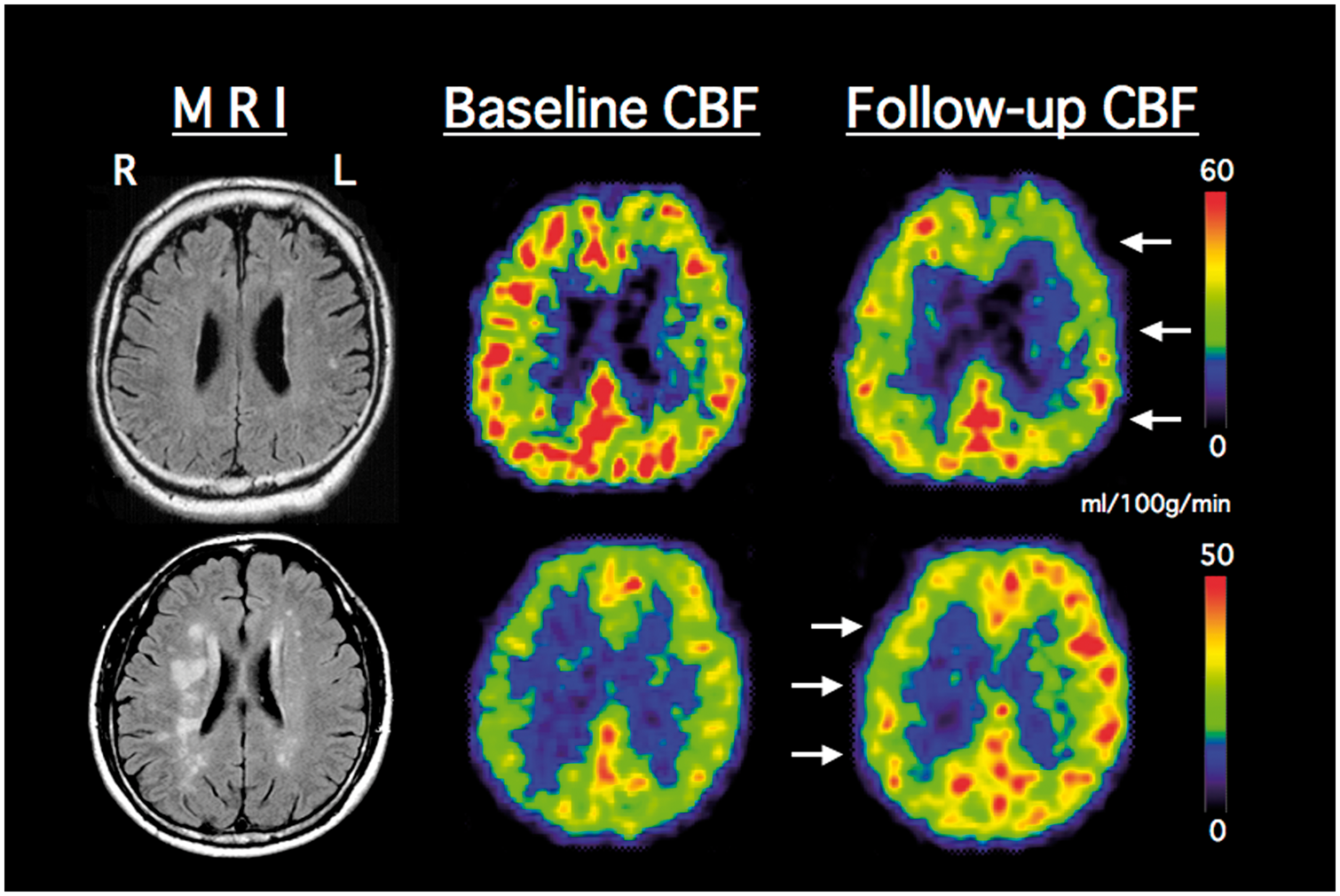
Representative PET CBF images. Upper row, a decrease of CBF at follow-up examination (arrows) 24 months later in a patient with left (L) internal carotid artery occlusion and low systolic blood pressure (117 mmHg). Lower row, an increase of CBF at follow-up examination (arrows) 11 months later in a patient with right (R) internal carotid artery occlusion and high systolic blood pressure (167 mmHg).
Based on the CBF/CBV values in the hemisphere supplied by the diseased artery, 18 patients (20.0%) had decreased cerebral perfusion pressure (impaired perfusion) at baseline, and 71 (80.0%) did not. Of those 18 patients with decreased CBF/CBV, 8 had misery perfusion. At follow up, 10 of the 18 patients still had impaired perfusion, and 5 of the 71 patients had apparent impaired perfusion.
In a simple regression analysis (Figure 3), there was a positive linear relationship between changes in the CBF/CBV during follow-up and SBP (r = 0.322; 95% confidence interval, 0.122–0.497; p < 0.005). In multiple linear regression analysis (forward stepwise selection), low SBP (<130 mmHg) during follow-up and the presence of impaired perfusion at baseline were selected and included in the final model for changes in the CBF/CBV during follow-up (a correlation coefficient of 0.428, p = 0.0002). In this model, low SBP and the presence of impaired perfusion at baseline accounted for 11.2% and 7.2%, respectively, of the variance of the change in the CBF/CBV. Low SBP and the presence of impaired perfusion were independently associated with changes in the CBF/CBV. Furthermore, low SBP (coefficient, −1.504; standard error, 0.454; t = -3.314; p < 0.005) was negatively correlated with the change in the CBF/CBV, whereas the presence of impaired perfusion at baseline (coefficient, 1.243; standard error, 0.454; t = 2.739; p < 0.01) was positively correlated with change in the CBF/CBV.
Scatter plot of the systolic blood pressure and the change of cerebral blood flow (CBF)/cerebral blood volume (CBV) ratio (top), the change of cerebral blood flow (CBF) (middle), or the change of oxygen extraction fraction (OEF) (bottom) in the hemisphere with arterial disease.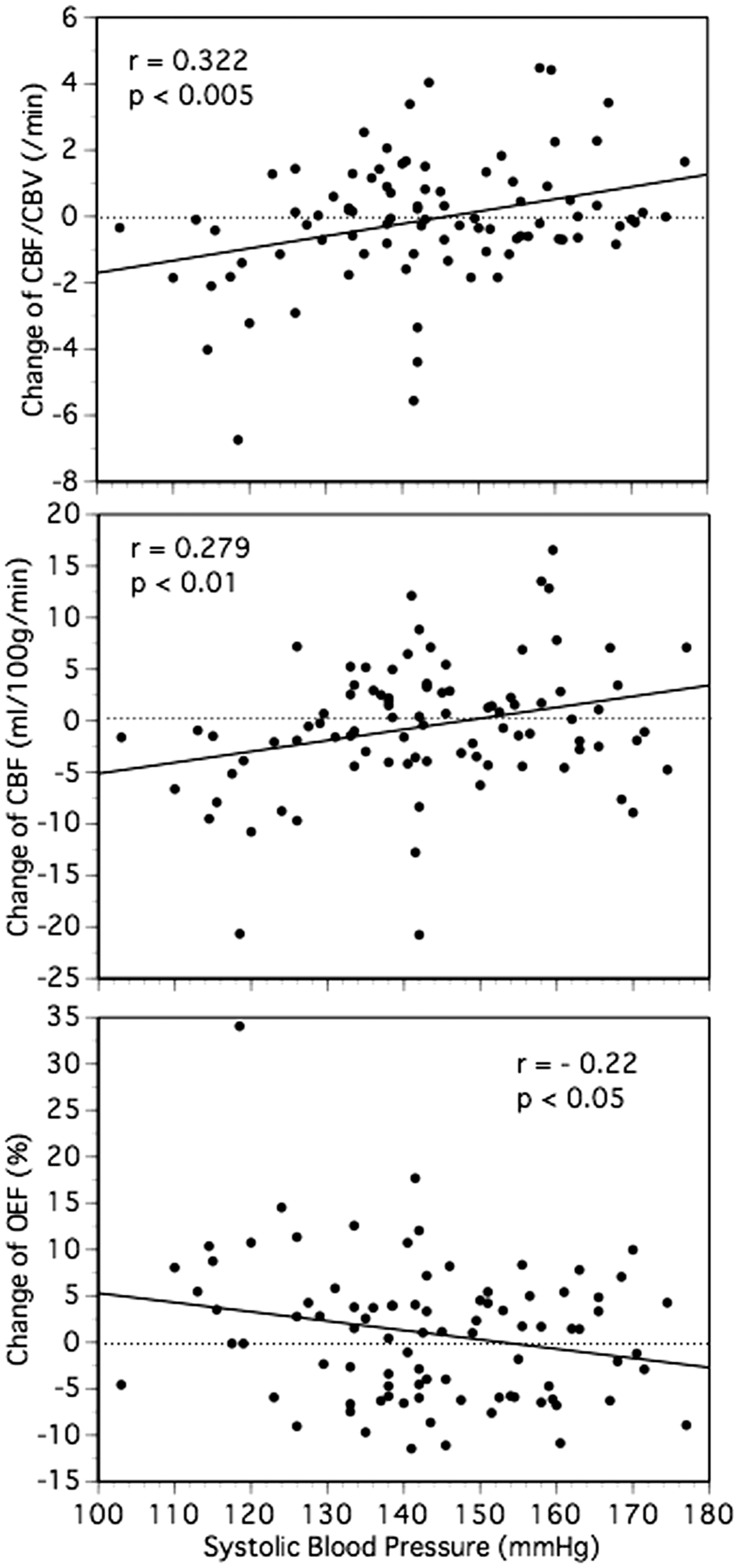
Low SBP during follow-up (coefficient, −1.355; standard error, 0.462; t = −2.93; p < 0.005) and the presence of impaired perfusion at the baseline (coefficient, 1.114; standard error, 0.458; t = 2.432; p < 0.05) were also significant independent predictors of the changes of CBF/CBV ratio, after the inclusion of the interval between the first and follow-up PET examinations and the use of statins in the model.
As a whole, the changes in CBF/CBV were significantly correlated with those in the CBF (r = 0.784; 95% confidence interval, 0.688–0.853) or OEF (r = −0.596; 95% confidence interval, −0.716 to −0.443). In a simple regression analysis (Figure 3), SBP was significantly correlated with changes in the CBF (r = 0.279; 95% confidence interval, 0.028–0.185; p < 0.01) or OEF (r = −0.22; 95% confidence interval, −0.195 to −0.006; p < 0.05) during follow-up. Low SBP during follow-up was also a significant independent predictor of the changes in CBF or OEF.
Discussion
This study investigated the relationship between long-term changes in cerebral hemodynamics on PET and BP during follow-up. We demonstrated that lower SBP was associated with lesser hemodynamic improvement in patients with atherosclerotic ICA or MCA disease and no ischemic episodes of stroke during follow-up. In fact, there was hemodynamic deterioration in patients with low SBP (<130 mmHg). In these patients, significant decreases in CBF and CBF/CBV ratio and increases in OEF were found during follow-up, while there was no significant change in patients without low SBP.
The precise reason for the lesser hemodynamic improvement in patients with low SBP is unclear. However, low SBP might impede the development of collateral flow. In general, growth of cerebral collaterals can lead to long-term improvement of collateral blood flow in patients with atherosclerotic ICA or MCA disease. 10 Vascular remodeling of various mechanisms, including arteriogenesis and angiogenesis, occurs in chronic cerebrovascular occlusive diseases. 30 Although angiogenesis, sprouting of new capillaries from postcapillary venules, is active during chronic hypoxia, arteriogenesis is triggered by fluid shear stress rather than hypoxia. Since arterial occlusion lowers the pressure in the distal vasculature, it increases flow through pre-existing collaterals due to the resulting pressure gradient, and the collateral vessels subsequently grow in diameter. For example, an increase in the diameter of the circle of Willis has been demonstrated after ICA occlusion.10,31 The growth of cerebral collaterals is correlated with the rising intravascular flow rate. 20 Thus, low SBP may reduce the intravascular flow rate and may be associated with lesser development of cerebral collaterals through arteriogenesis.
The studied patients with atherosclerotic ICA or MCA disease had no ischemic stroke episodes during follow-up. As we can expect, impaired perfusion (a decreased CBF/CBV) at baseline was associated with hemodynamic improvement during follow-up. Previous studies showed that medially treated patients with hemodynamic impairment at baseline and no intervening ischemic event had hemodynamic improvement during follow-up,6–9 which might be related to a reduced stroke risk. In the present study, 8 of the 18 patients with impaired perfusion at baseline did not have impaired perfusion at follow-up. However, this was not true for patients with low SBP; low SBP was independently associated with less hemodynamic improvement. Therefore, in patients with impaired perfusion at baseline and low SBP, the risk for ischemic stroke might remain high during the follow-up period. This speculation was consistent with the finding of our previous studies that in patients with impaired perfusion at baseline, a low SBP (<130 mmHg) was associated with an increased risk of ipsilateral ischemic stroke during follow-up.17,32 A previous study also showed that in patients with atherosclerotic ICA occlusion who had no ischemic stroke episodes during follow-up, hemodynamic deterioration during follow-up was associated with subsequent ischemic stroke.4,
Medical treatments to promote collaterals may play an important role in preventing ischemic stroke by improving hemodynamics in patients with major cerebral artery disease.11,30 Treatment of hypertension may prevent progression of large or small cerebral artery disease, which may prevent a decrease in collateral flow. Furthermore, lowering BP may decrease shear stress in the collateral pathways, which may decrease the risk of the development of thromboembolism.16,33,34 However, the results of the present study suggest that controlling the BP may be important for hemodynamic improvement. We do not recommend an aggressive control of SBP (<130 mm Hg) in patients with atherosclerotic ICA or MCA disease, especially in patients with impaired perfusion. In previous studies reporting hemodynamic improvement during follow-up, BP in the patients with hemodynamic impairment might have been controlled at a somewhat higher level than that in those without.8,35 The level to which BP should be lowered to achieve maximal benefits among patients with ICA or MCA disease is not known. In our cohort, the BP was mildly controlled depending on each patient’s specific BP. We suggest that the lower limit of BP control should be individualized based on the presence of impaired perfusion.
In this study, the ratio of CBF to CBV was used as an indicator of cerebral perfusion pressure.18,26 A previous experimental PET study showed the linear relationship between CBF/CBV and mean arterial pressure. 17 In patients with ICA occlusion, decreases of CBF/CBV were early changes in response to decreased perfusion pressure before decreases of CBF and increases of OEF occurred. 25 Therefore, changes in CBF/CBV are considered a sensitive index of hemodynamic changes during follow-up. Decreases in CBF/CBV can also be used as a predictor of subsequent ischemic stroke in patients with ICA and MCA diseases.17,32,36,37 Furthermore, the CBF/CBV is the reciprocal of the expression for vascular mean transit time that can be evaluated by perfusion imaging with MRI or computed tomography in routine clinical practice.38,39
Limitations
This study was a post hoc analysis of an observational study in selected patients with no ischemic episodes of ischemic stroke during follow-up. Therefore, we cannot exclude the possibility that unmeasured confounding variables may explain some of our findings. The interval between first and follow-up PET scans ranged between 3 months and 112 months, which might affect the results, although the effect of the interval on the changes in cerebral hemodynamics was not statistically significant. The patients had a mixture of ICA and MCA disease, which may make it difficult to accurately compare patient groups. The BP was measured at only two PET examinations, which may have led to a misclassification of BP levels during follow-up. We considered that the averaged values of BP at baseline and follow-up PET examinations could reflect BP during the follow-up period. A randomized, controlled trial including direct hemodynamic measurement is needed to determine the level to which BP should be lowered to achieve maximal benefits in hemodynamic improvement or reduced stroke risk in patients with or without impaired perfusion.
Conclusions
In patients with atherosclerotic ICA or MCA disease and no episodes of ischemic stroke during follow-up, lower SBP was associated with lesser hemodynamic improvement during follow-up. There was hemodynamic deterioration in patients with low SBP (<130 mmHg).
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Japan Society for the Promotion of Science KAKENHI [grant numbers 17K09814, 26461323].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
HY designed the study, contributed to PET imaging, analysed the data and wrote the first draft of the manuscript, and modified all subsequent drafts. SK and TH contributed to PET imaging, contributed to interpretation of the data and contributed to all drafts of the manuscript. MT performed much of the scanning, analysed the data and contributed to all drafts of the manuscript.
