Abstract
In atherosclerotic internal carotid artery (ICA) or middle cerebral artery (MCA) disease, hemodynamic compromise may cause selective neuronal damage manifested as loss of central benzodiazepine receptors (BZRs) in the normal-appearing cerebral cortex, without overt episode of stroke. To investigate the association of decreases in cortical BZRs with hemodynamic compromise and the effect of angiotensin receptor blockers (ARBs) on these receptors in patients whose atherosclerotic ICA or MCA disease is asymptomatic, we measured BZRs using positron emission tomography and 11C-flumazenil in 79 patients with asymptomatic atherosclerotic ICA or MCA disease and no cortical infarction. Three-dimensional stereotactic surface projections were used to calculate the BZR index, a measure of abnormally decreased BZRs in the cerebral cortex within the MCA distribution. Multiple regression analysis showed this index to be positively correlated with the value of oxygen extraction fraction, with the presence of silent subcortical infarcts, and with the presence of ischemic heart disease, whereas it was negatively correlated with the treatment of hypertension with ARBs. In asymptomatic atherosclerotic ICA or MCA disease, hemodynamic compromise is associated with selective neuronal damage manifested as decreases in cortical BZRs in the noninfarcted cerebral cortex, whereas ARBs are associated with preservation of cortical BZRs.
Keywords
Introduction
Advances in noninvasive vascular imaging methods have made feasible the detection of asymptomatic atherosclerotic disease of the internal carotid artery (ICA) or middle cerebral artery (MCA), and its management has been debated, especially concerning the indication of vascular reconstruction surgery (Abbott et al, 2007; Qureshi et al, 2009). Major randomized studies suggested a minor benefit of prophylactic carotid endarterectomy in asymptomatic extracranial ICA stenosis, but recent advances in medical management may reduce the need for such surgery in preventing future ischemic events (Abbott et al, 2007). In asymptomatic ICA occlusion or MCA disease, few data have been presented as to the natural history or treatment strategies (Powers et al, 2000; Qureshi et al, 2009). Strict medical management for most patients and prophylactic surgery for patients at particularly high risk of stroke are proposed as the best options for management of asymptomatic patients (Abbott et al, 2007; Qureshi et al, 2009). Several studies have suggested that unstable plaques leading to rupture or embolism in patients with arterial stenosis and hemodynamic compromise distal to arterial disease may represent major factors associated with future risk of stroke (Abbott et al, 2007; Qureshi et al, 2009; Silvestrini et al, 2000). Patients at high risk of stroke may have already suffered ischemic cerebral damage at the time their asymptomatic ICA or MCA disease is discovered. Silent ischemic damage accompanying unstable plaques or hemodynamic compromise would further support these conditions as risk factors for stroke.
Hemodynamic compromise or microembolism may produce silent tissue damage in the cerebral cortex that is not detectable as infarction on computed tomography or magnetic resonance imaging (MRI) (Garcia et al, 1996). In patients with chronic hemodynamic compromise, cortical perfusion may fall below the penumbra threshold for a period of minutes, causing selective cortical neuronal damage without subjective symptoms (Yamauchi et al, 2005, 2007, 2009). Silent ischemic damage because of frequent microemboli may occur especially easily in patients with hemodynamic compromise because of reduced clearance of emboli (Caplan and Hennerici, 1998). However, the pathophysiology of silent ischemic cortical damage in asymptomatic ICA or MCA disease patients has not been specifically investigated.
Most cortical neurons express central-type benzodiazepine receptors (BZRs) and specific imaging of these receptors in vivo has made possible visualization of neuronal alterations induced by ischemia in humans (Guadagno et al, 2008; Hatazawa and Shimosegawa, 1998; Heiss et al, 1998; Kuroda et al, 2006; Nakagawara et al, 1997; Sette et al, 1993). Selective neuronal damage can be detected as a decrease in BZRs in the otherwise seemingly normal cerebral cortex. Our previous studies have shown that in atherosclerotic ICA or MCA disease with mixed symptomatic status, hemodynamic compromise is associated with a decrease in BZRs in the noninfarcted cerebral cortex, suggesting that statins might have beneficial effects against neuronal damage (Yamauchi et al, 2005, 2007, 2009). Those studies likewise demonstrated a decrease in BZRs in the noninfarcted cerebral cortex in a subgroup of patients with asymptomatic atherosclerotic ICA or MCA disease compared with healthy controls, which suggested the presence of selective cortical neuronal damage in asymptomatic patients as well. However, the association of the decrease in BZRs with hemodynamic compromise in asymptomatic patients was uncertain, probably because of a small sample size (Yamauchi et al, 2007). Although chronic hemodynamic compromise is associated with the risk for cerebral ischemic damage in patients with atherosclerotic ICA or MCA disease, the contribution of hemodynamic compromise to a decrease in BZRs may differ between symptomatic and asymptomatic patients and should be investigated separately (Derdeyn et al, 1998). Furthermore, the previous study (Yamauchi et al, 2007) did not investigate the effect of angiotensin receptor blockers (ARBs) on neuronal damage that several experimental studies have suggested (Mogi and Horiuchi, 2009). The purpose of this study was to determine whether decreases in cortical BZRs occur in association with hemodynamic compromise in asymptomatic atherosclerotic ICA or MCA disease, and whether ARBs influence the decreases in BZRs.
Materials and methods
Patients
We studied 79 asymptomatic patients with atherosclerotic occlusion or stenosis of the ICA or MCA (Table 1). Forty-seven patients were part of a previously published data set (Yamauchi et al, 2007, 2009). They were referred to our positron emission tomography (PET) unit for evaluation of hemodynamic effects of ICA or MCA disease using PET and 15O-gas as part of a comprehensive clinical evaluation of the necessity of vascular reconstruction surgery. Inclusion criteria for the 11C-flumazenil (FMZ) PET study were as follows: (1) occlusion or stenosis of the ICA (>60% diameter reduction according to the NASCET criteria (North American Symptomatic Carotid Endarterectomy Trial Collaborators, 1991)) or MCA (>50% diameter reduction (Samuels et al, 2000)) as documented by conventional or magnetic resonance angiography and (2) absence of previous symptoms or signs of ischemia in any vascular territories. The exclusion criteria were (1) cerebral cortical, cerebellar, or brainstem infarct detectable on routine MRI images (T1-, T2-weighted, or fluid-attenuated inversion recovery (FLAIR) images), (2) history of syncope attack, (3) history of vascular reconstruction surgery, (4) unilateral arterial disease with extensive white matter lesions in both hemispheres probably caused by bilateral small vessel disease, (5) history of taking BZR agonists, or (6) presence of potential sources of cardiogenic embolism, including recent myocardial infarction (<3 weeks previous), known atrial fibrillation, mitral stenosis, mitral valve prosthesis, dilated cardiomyopathy, sick sinus syndrome, or acute bacterial endocarditis. Arterial disease was suspected in referred patients based on findings of magnetic resonance angiography or echo angiograms performed as part of screening for cerebral arterial disease in patients presenting with dizziness, vertigo, or headache, or in patients with coronary arterial disease. Among vascular risk factors, status with respect to hypertension, diabetes mellitus, ischemic heart disease, hypercholesterolemia, and smoking were evaluated from patient history recorded at the PET examination. Hypertension, diabetes mellitus, ischemic heart disease, or hypercholesterolemia were judged to be present when there was a history of treatment. The ethics committee of our center approved the protocol of the 11C-FMZ PET study, and each patient gave informed consent in writing.
Patient characteristics
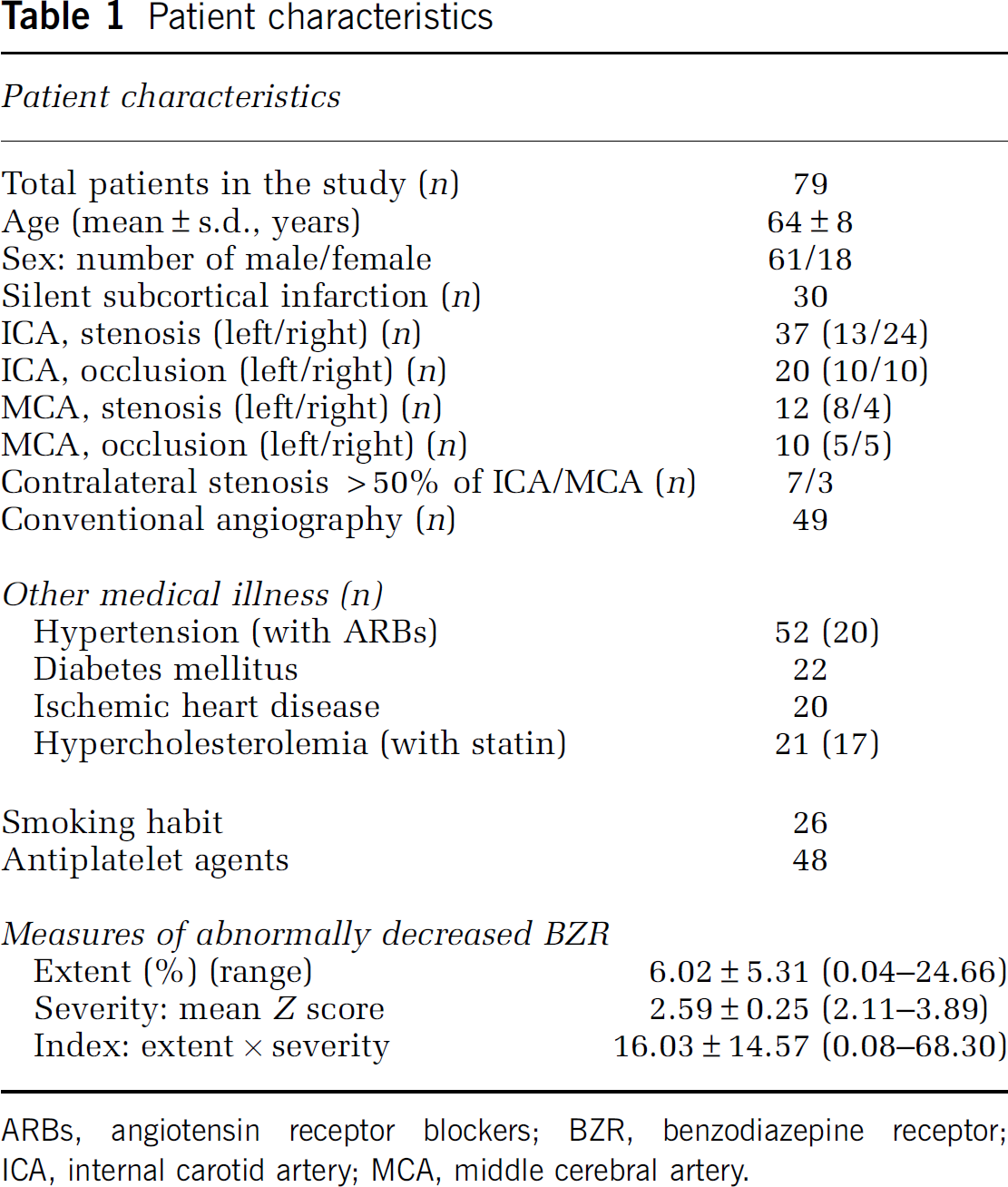
ARBs, angiotensin receptor blockers; BZR, benzodiazepine receptor; ICA, internal carotid artery; MCA, middle cerebral artery.
Positron Emission Tomography Measurements
The PET scans were performed in each subject using an advance whole-body scanner (General Electric Medical Systems, Milwaukee, WI, USA), which permits simultaneous acquisition of 35 image slices with interslice spacing of 4.25 mm (Okazawa et al, 2001). A transmission scan using 68Ge/68Ga was performed for attenuation correction in each subject before the tracer administration. To reconstruct PET data using filtered back projection, images were blurred to 6.0 mm full width at half maximum in the transaxial direction using a Hanning filter.
First, a series of 15O-gas studies was performed (Okazawa et al, 2001). C15O2 and 15O2 were delivered continuously via a mask throughout the 5-minute scan. Cerebral blood volume was measured by bolus inhalation of C15O with scanning for 3 minutes. Arterial blood was sampled two or three times during each procedure of the 15O-gas study. No subject showed significant changes in PaCO2 during scanning.
The 15O-gas study was followed by a study of 11C-FMZ (Okazawa et al, 2004; Yamauchi et al, 2005, 2007), which was synthesized by 11C-methylation of demethylated FMZ (Hoffmann-La Roche, Basel, Switzerland). Specific activities at the time of injection ranged from 63.6 to 120.0 (mean 87.0) GBq/μmol. After slow intravenous injection of 370 to 555 MBq of 11C-FMZ, a 50-minute dynamic PET scan was initiated at the time of tracer administration with frame durations of 10 seconds × 6, 15 seconds × 8, 30 seconds × 4, 60 seconds × 5, 5 minutes × 4, and 10 minutes × 2.
The steady-state method was used to calculate cerebral blood flow (CBF), cerebral metabolic rate of oxygen (CMRO2), and oxygen extraction fraction (OEF) (Frackowiak et al, 1980). The CMRO2 and OEF were corrected on the basis of cerebral blood volume. Binding potential (BPND (nondisplaceable)) of 11C-FMZ was calculated using dynamic data and Logan graphical analysis with reference tissue, by using the time—activity curves obtained from the pons (the reference region) (Logan et al, 1996; Okazawa et al, 2004).
Magnetic Resonance Imaging
The MRI was performed with a Signa unit (General Electric) operating at a field strength of 1.5 T. The imaging protocol consisted of the following imaging series: T2-weighted spin echo (repetition time/echo time [TR/TE]=3,000/88.8 milliseconds), T1-weighted spin echo (TR/T2=550/11.2 milliseconds), and FLAIR (TR/TE=8,002/158 milliseconds, inversion time 2,000 milliseconds).
Silent subcortical ischemic lesions were identified as hyperintense lesions on T2-weighted MRI. The FLAIR images were used to distinguish infarcts from dilated perivascular spaces. All scans were reviewed by one investigator who was masked to patient clinical status including other imaging data.
Data Analysis
For hemodynamic and metabolic parameters, we analyzed 10 tomographic planes, located 46.25 to 84.5 mm above and parallel to the orbitomeatal line. The lowest plane corresponded to the level of the basal ganglia and thalamus, and the uppermost to the level of the centrum semiovale. A region of interest (ROI) was placed on the CBF images. Each image was examined by compactly placing 10 to 12 circular ROIs, each 16 mm in diameter, over the gray matter of the outer cortex in each hemisphere. According to the atlas prepared by Kretschmann and Weinrich, the ROIs in all 10 images would cover the distribution of the MCA as well as the external border-zone regions (Kretschmann and Weinrich, 1986; Yamauchi et al, 1990). The same ROIs were used for the CMRO2, OEF, and cerebral blood volume images. Mean hemispheric value for the hemisphere affected by ICA or MCA disease was calculated as the average of the values for all circular ROIs. In patients with bilateral disease, the hemisphere supplied by the more severely diseased ICA or MCA is referred to here as the affected hemisphere. Normal control values of the OEF were obtained from seven normal volunteers (four men and three women), aged 47±7 (mean±s.d.) years who had normal routine neurologic examinations and MRI scans. The mean±s.d. OEF value in the 14 hemispheres of the normal subjects was 44.5%±3.8%. Absolute hemispheric values beyond the upper 95% limit (the mean plus 13 t 0.05s.d.) defined in normal subjects (>52.9%) were considered abnormal. Previous studies have shown that increased OEF determined by this method is an independent predictor of stroke risk in patients with symptomatic major cerebral arterial occlusive disease (Yamauchi et al, 1996, 1999a ).
The FMZ BP parametric images were analyzed by a three-dimensional stereotactic surface projection (SSP) technique (Figure 1) (Minoshima et al, 1995; Yamauchi et al, 2007). This technique anatomically normalized the individual PET data to the standard brain and compared regional voxel data between patients and controls. This procedure was performed using the interface software iSSP (version 3.5, Nihon Medi-Physics Corporation, Nishinomiya, Japan). First, there was stereotaxic reorientation of each brain into a standard shape by using nonlinear warping with the anterior commissure-posterior commissure line as the anatomic line of reference. During this stereotaxic transformation of each brain, regional information about FMZ BP measured by PET was preserved. Then, a method of data extraction was performed in which the cortical activity was projected onto the brain surface. In the standard stereotactic system, pixels located on the outer and medial surface of both hemispheres were predetermined, along with vectors perpendicular to the three-dimensional surface at each pixel. For each predetermined surface pixel on an individual's anatomically standardized PET image set, the algorithm searched along the vector, six pixels deep into the cortex, for the highest pixel value, and assigned this maximum value to the surface pixel. This surface projection technique in three-dimensional SSP minimizes the effect of cortical atrophy. To correct for fluctuations in whole-brain values, and to extract the change because of ICA or MCA disease, pixel values of an individual's image set were first normalized to the mean cerebellar value. Normalization to the cerebellum was considered appropriate, because several studies have shown that BZRs do not change in crossed cerebellar diaschisis (Hatazawa and Shimosegawa, 1998). Therefore, each brain was stereotactically transformed into a standard surface image format, which enabled us to compare the resultant cortical projections between patients and controls. Z scores calculated for each surface pixel (defined as (mean normalized pixel value for controls−normalized pixel value for the patient)/s.d. for controls) were used to quantify decreases in FMZ BP. Thus, a positive Z score represents reduced FMZ BP in the patient relative to the control group, and Z scores >2 were considered abnormal. The normal control database was based on scans of 10 healthy control subjects, aged 57±7 years, including 7 men, who had no previous history of medical or psychiatric disorder and no history of taking BZR agonists.
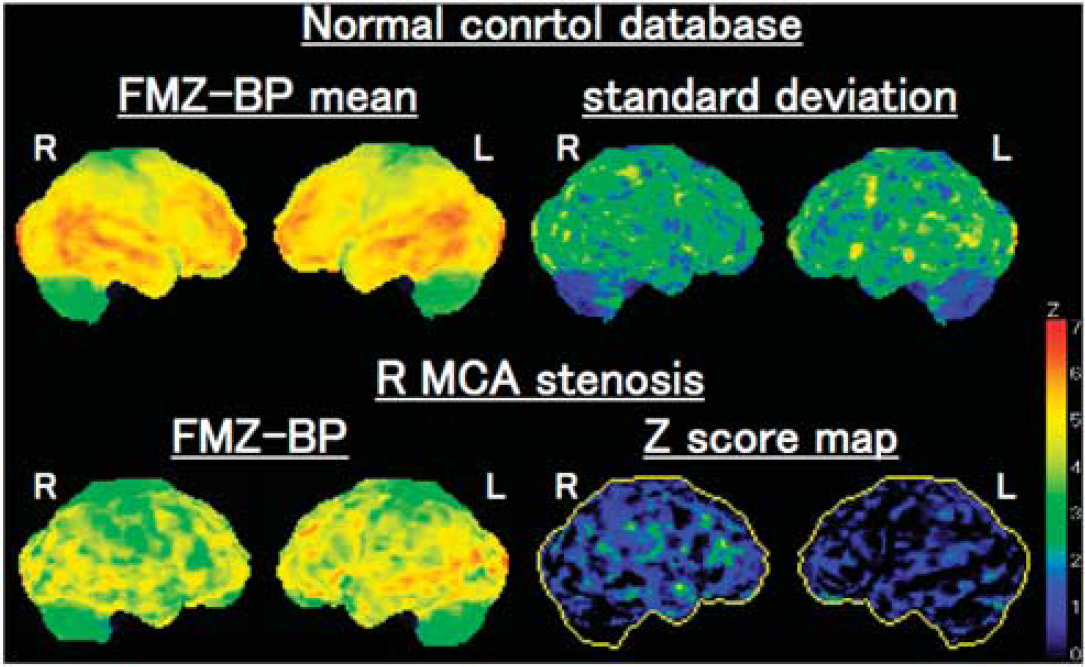
Three-dimensional stereotactic surface projection (3D-SSP) analysis. (Upper row) The 3D-SSP images showing the value of mean and s.d. for normal control database. (Lower row) The 3D-SSP images and Z score maps showing a decrease of flumazenil binding potential (FMZ BP) in the right (R) hemisphere in a patient with stenosis of the R middle cerebral artery (MCA). The extent of abnormally decreased benzodiazepine receptors (BZRs) was 12.0% in the R MCA distribution, the mean Z score in those pixels was 2.54, and the abnormally decreased BZR index was 30.6.
To quantitate the degree of abnormal FMZ BP reduction in each patient, the stereotactic extraction estimation method was used to calculate a BZR index (defined as (the extent (%) of pixels with Z score >2) × (average Z score for those pixels)) for the cerebral cortical MCA distribution affected by ICA or MCA disease (Mizumura et al, 2003). The extent represents the percentage of pixels with Z score >2 in all pixels within the cerebral cortical MCA distribution. By multiplying the extent and the average Z score for pixels with Z score >2 together, the BZR index indicates the severity of the abnormal FMZ BP reduction in the cerebral cortical MCA distribution. As vascular territories vary among individuals, we arbitrarily applied MCA territory according to the atlas prepared by Kretschmann and Weinrich (1986). This MCA distribution included the middle and inferior frontal gyri, the precentral gyrus, the superior and inferior parietal gyri, the angular, postcentral, and supramarginal gyri, the superior, middle, inferior, and transverse temporal gyri, and the superior and middle occipital gyri (Mizumura et al, 2003).
Statistical Analysis
Pairwise comparisons of BZR index values between groups were made using Student's t-test. Stepwise or multiple linear regression analysis was used to test the independent predictive values of OEF, age, sex, the presence of silent subcortical infarction, arterial disease characteristics, the presence of vascular risk factors, and drug treatment history with respect to BZR index values in the hemisphere affected by ICA or MCA disease. Significance was established at P<0.05, unless stated otherwise.
Results
Pixel analysis in all patients indicated abnormally decreased BZR (Table 1). When 10 control subjects were analyzed in the same way, the mean value of BZR index in the bilateral hemispheres was 1.78±1.79 (upper 95% confidence limit (the mean plus 9 t 0.05s.d.)=5.84). The BZR index in 56 patients (71%) was considered abnormal compared with healthy control subjects. The BZR index, which ranged from 0.08 to 68.3 (16.0±14.5), correlated significantly with OEF (r=0.26, P=0.017) (Figure 2) but not with age (r=0.12, P=0.28). The BZR index in patients with increased OEF (>52.9%) (n=17, 22.6±18.0) was significantly higher than those without (n=62, 14.2±13.0) (P<0.05). Values of OEF showed a significant negative relationship with CBF (r=−0.56, P<0.0001). The BZR index also correlated significantly with CMRO2 (r=−0.41, P=0.0001) or CBF (r=−0.36, P<0.005). Patients with male sex, ischemic heart disease, or right-sided arterial disease had higher BZR indexes than those without, but the differences were not statistically significant (Table 2). The BZR index also did not differ significantly between patients with ICA versus MCA disease or between patients with arterial occlusion versus stenosis.
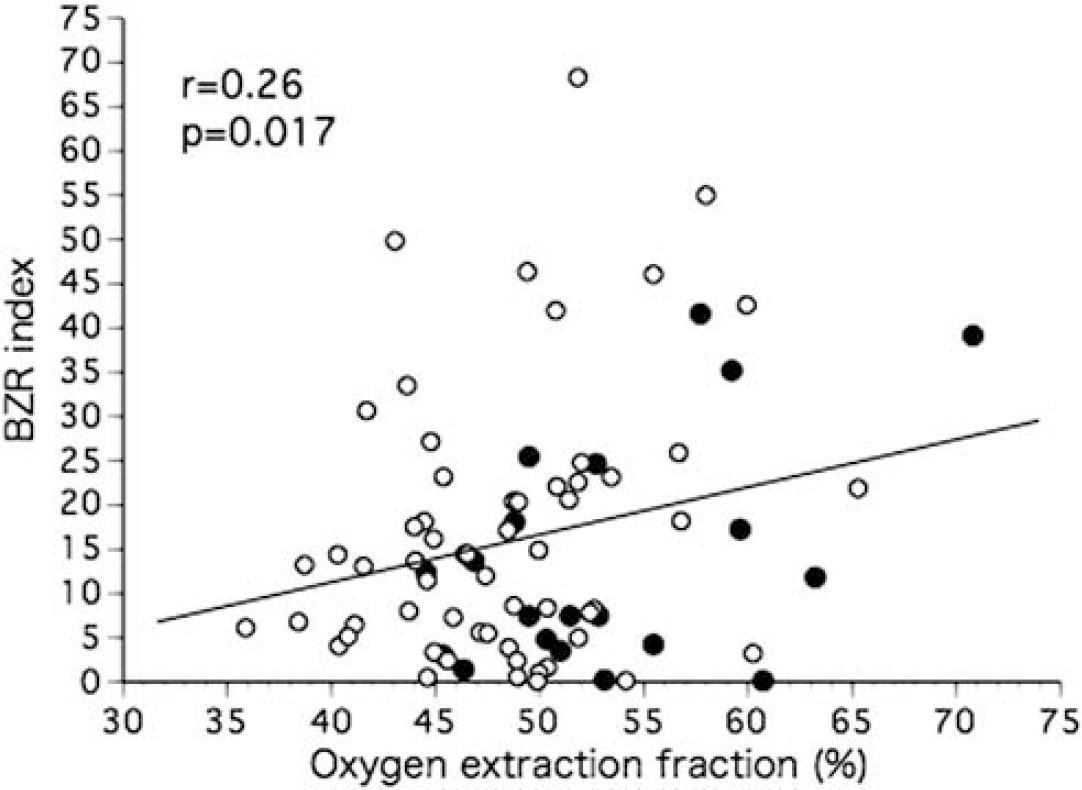
Scatter diagram plotting the benzodiazepine receptor (BZR) index against the value of oxygen extraction fraction in the hemisphere with arterial disease. The closed circles indicate patients taking angiotensin receptor blockers.
Vascular risk factors and BZR index in the hemisphere with arterial disease
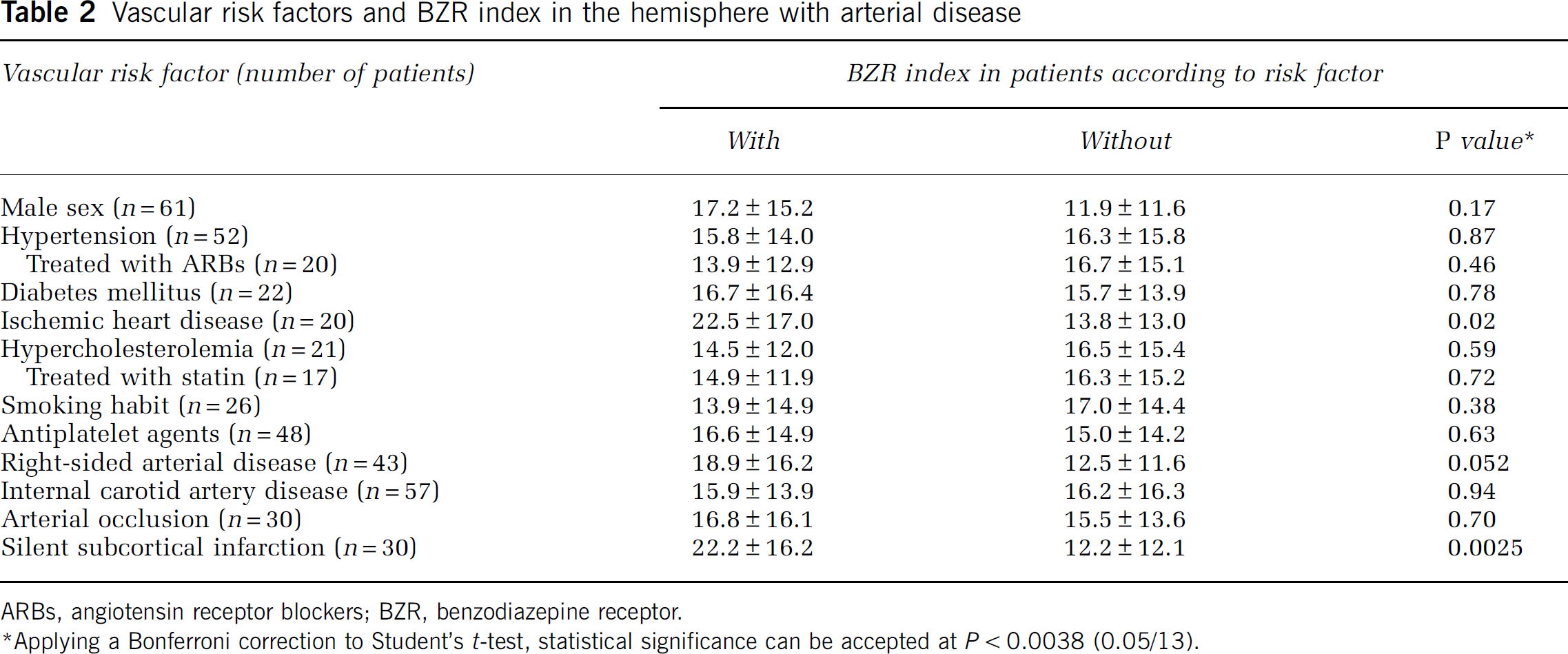
ARBs, angiotensin receptor blockers; BZR, benzodiazepine receptor.
∗Applying a Bonferroni correction to Student's t-test, statistical significance can be accepted at P<0.0038 (0.05/13).
Thirty patients (38%) had a silent subcortical infarct in the hemisphere affected by arterial disease; 16 with deep perforator infarcts (lesions in the territory of the lenticulostriate artery), 7 with superior perforator infarcts (lesions in the territory of the medullary artery), and 7 with both (Table 3). All infarcts were small in size (<1 cm). The BZR index in patients with silent subcortical infarcts was significantly higher than those without (P<0.0025, Table 2).
Distribution of silent subcortical infarcts in the hemisphere affected by arterial disease

When the value of OEF, patient age, gender (male sex), presence of silent subcortical infarction, characteristics of arterial disease (right-sided disease, ICA disease, or arterial occlusion), presence of vascular risk factors (hypertension, diabetes mellitus, ischemic heart disease, hypercholesterolemia, and smoking habit), and history of drug treatments (ARBs, statins, and antiplatelet agents) were entered into a stepwise linear regression analysis (forward selection), it produced a model for the BZR index that included OEF value, the presence of silent subcortical infarction, history of ischemic heart disease, and ARB treatment (correlation coefficient of 0.53, P<0.0001) (Table 4), whereas the remaining variables did not contribute significantly to the magnitude of the correlation. Silent subcortical infarction was the most heavily weighted factor, and accounted for 11.3% of the variance of the change in BZR index, whereas OEF, ischemic heart disease, and ARB accounted for 6.7%, 5.6%, 4.8% of the variance, respectively. These four variables were significant independent predictors of the BZR index, after including patient age and sex in the model (Table 5). In this multiple regression model, the value of OEF was positively correlated with BZR index, whereas treatment with ARBs was negatively correlated.
Stepwise linear regression analysis with BZR index in the hemisphere affected by arterial disease as dependent variable

ARBs, angiotensin receptor blockers; BZR, benzodiazepine receptor; OEF, oxygen extraction fraction.
Multiple linear regression analysis with BZR index in the hemisphere affected by arterial disease as dependent variable
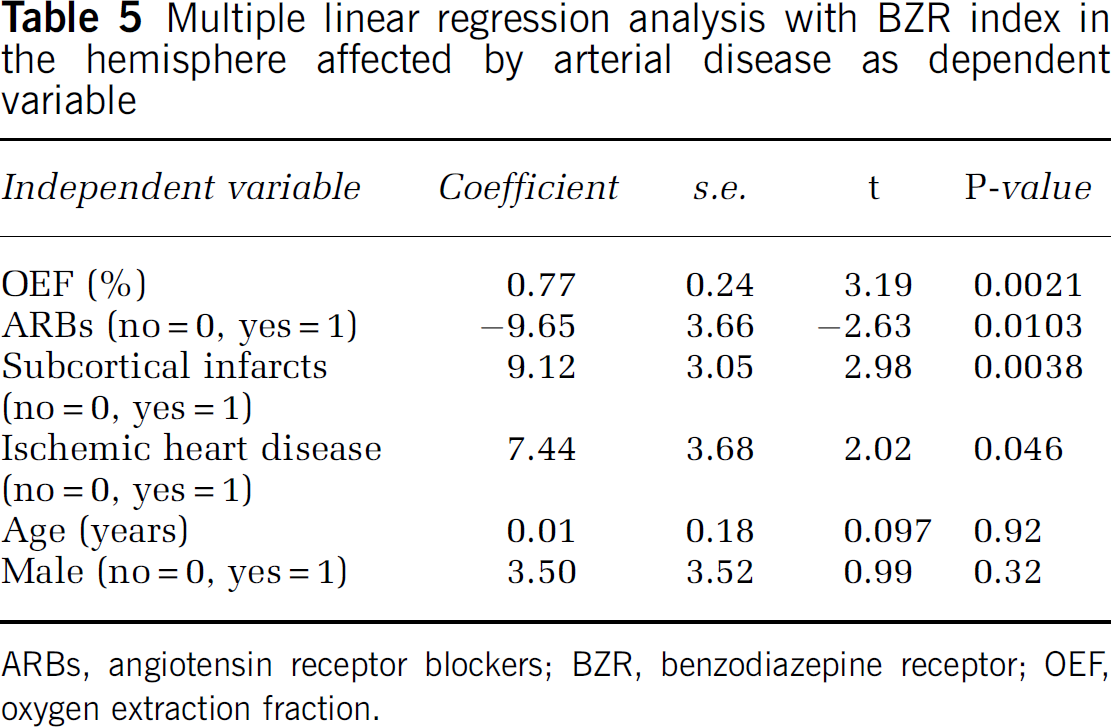
ARBs, angiotensin receptor blockers; BZR, benzodiazepine receptor; OEF, oxygen extraction fraction.
The CMRO2 in patients with silent subcortical infarcts was significantly lower than those without (3.00±0.38 versus 3.26±0.40 mL per 100 g per minute: P<0.0063). After adjustment for the effect of the BZR index by using multiple regression analysis, CMRO2 did not correlate with the presence of silent subcortical infarction.
The treatment with ARBs included 4 or 8 mg of candesartan cilexetil daily in 17 patients, 40 mg of valsartan daily in 2, and 20 mg of olmesartan medoxomil daily in one. The duration of the treatment with ARBs ranged from 3 to 84 (23±24) months at the PET examination. The duration was not correlated with the BZR index (r=−0.12, P=0.59). Separate analysis of the data in patients treated with candesartan (n=17) yielded a better association of ARB treatment with the BZR index. Other class of antihypertensive drugs was taken in combination with ARBs in 11 patients, calcium antagonist in 9, angiotensin-converting inhibitor in 1, and diuretics in 1. Calcium antagonist was also taken in 20 hypertensive patients without ARB treatment and in 1 patient with ischemic heart disease. The BZR index did not differ significantly between patients with calcium antagonist treatment (n=30) and those without, and the results of a stepwise linear regression analysis did not change after the inclusion of calcium antagonist treatment as an independent variable.
Discussion
This study of patients with asymptomatic atherosclerotic ICA or MCA occlusive disease and no cortical infarction has demonstrated the presence of selective neuronal damage manifested as a decrease in BZRs in the noninfarcted cerebral cortex, in association with chronic hemodynamic compromise (increased OEF). The BZRs index was abnormal in 71% of the asymptomatic patients. This abnormality was mild compared with that previously reported for symptomatic patients (the BZR index; 16.0±14.5 versus 48.2±54.1) (Yamauchi et al, 2007). After adjusting for age and gender, the BZR index was independently and positively correlated with OEF, with the presence of silent subcortical infarction, and with the presence of ischemic heart disease, and it was negatively correlated with the treatment of hypertension with ARBs. The effect of ARBs on the BZR index became apparent after adjusting the effects of silent infarction and OEF.
In atherosclerotic ICA or MCA occlusive disease, chronic hemodynamic compromise may increase risk for cerebral ischemic damage (Grubb et al, 1998; Yamauchi et al, 1996, 1999a ), even when patients are asymptomatic (Powers et al, 2000; Silvestrini et al, 2000). The association between decreased BZRs and increased OEF in the present study suggests that hemodynamic cerebral ischemia may have caused selective neuronal damage in patients with increased OEF, without overt episode of stroke. A previous study in patients with atherosclerotic ICA or MCA disease and no intervening stroke during follow-up also showed that cortical BZRs decreased in association with increased OEF (Yamauchi et al, 2007). The reduction in BZRs may be due in part to the cumulative impact of microemboli occurring over a period of years, although the use of antiplatelet agents was not associated with preservation of BZRs in the present study. Transcranial Doppler studies have demonstrated microemboli in the MCA in some patients with asymptomatic severe ICA stenosis (Ritter et al, 2008). In patients with increased OEF, decreased washout of microemboli could easily result in irreversible injury (Caplan and Hennerici, 1998). Therapeutic strategies to prevent neuronal damage, including vascular reconstruction surgery, may be called for in asymptomatic patients with chronic hemodynamic compromise.
The BZR index, CBF, and CMRO2 reflect different aspects of ischemic changes in the brain in the case of patients with atherosclerotic ICA or MCA disease, whereas OEF is a measure of hemodynamic compromise. A decrease in the CBF may be caused not only by a decrease in perfusion pressure but also secondarily by a decrease in the CMRO2 (Powers, 1991). A decrease in CMRO2 is one result of ischemic damage to the tissue and can also be caused by a decrease in neural input from distant regions with fiber connections (deafferentation) (Feeney and Baron, 1986). Therefore, it is reasonable that the BZR index, a measure of cortical neuronal damage, was correlated with CMRO2 better than with CBF or OEF in the present study. As shown in the case of crossed cerebellar diaschisis, a simple decrease of neural input may not be accompanied by a decrease in BZRs (Hatazawa and Shimosegawa, 1998). Thus, the correlation between the BZR index and cortical CMRO2 is affected by the relative contribution of deafferentation and neuronal damage to the decrease in cortical CMRO2.
Silent brain infarcts are often detected in asymptomatic ICA and MCA disease patients and are associated with risk of stroke during follow-up (Kakkos et al, 2009). In the present study, silent subcortical infarcts were associated with decreased cortical BZRs; however, subcortical infarct per se probably does not reduce cortical BZRs (Yamauchi et al, 2007). The cortical CMRO2 in patients with silent subcortical infarcts was decreased. Subcortical infarcts per se may decrease cortical CMRO2 through deafferentation of thalamocortical projections that is not accompanied by a decrease in cortical BZRs (Yamauchi et al, 2007). After adjusting for the effect of the BZR index, cortical CMRO2 was not correlated with the presence of silent subcortical infarcts, suggesting the main contributor to the decrease in cortical CMRO2 was not deafferentation but selective neuronal damage. Many of the silent deep perforator infarcts were bilaterally distributed, suggesting the absence of any relationship to arterial disease, but perhaps reflecting an association with diffuse brain arteriosclerosis leading to silent cortical ischemic damage. Such neuronal damage might be augmented by the combined effect of arteriosclerosis with ICA and MCA disease (Yamauchi et al, 1999b ). In contrast, most medullary artery infarcts were unilateral, suggesting that these may have been caused by microembolic or hemodynamic mechanisms due to arterial disease, with simultaneous damage to cortical neurons (Moustafa et al, 2010). Different types of silent infarcts in asymptomatic ICA or MCA disease may have different pathophysiological backgrounds, but nevertheless lead to an increase in ischemic cerebral damage.
Risk factors for stroke may be associated with selective neuronal damage as well as with infarction. In this study, the presence of ischemic heart disease was correlated with decreased BZRs. The patients with ischemic heart disease had histories of cardiac ischemic attack and angioplasty treatment of coronary arterial disease with or without stenting. Although we excluded patients with syncope attack, cerebral hypoperfusion may occur when cardiac output decreases in the presence of atherosclerotic ICA or MCA occlusive disease. Furthermore, cerebral embolism may occur during coronary angioplasty (Bendszus and Stoll, 2006). Ischemic heart disease should be managed carefully in patients with ICA or MCA disease.
Several experimental studies have shown that ARBs can attenuate ischemic neuronal injury through various mechanisms, including decreased oxidative stress and improved arterial compliance (Mogi and Horiuchi, 2009). In the present study, the treatment of hypertension with ARBs was associated with preservation of BZRs. This relationship became apparent only after adjusting for the effects of silent infarction and increased OEF, because the stronger effects of the presence of silent infarction and increased OEF masked the effect of ARB treatment. Our results would be consistent with protection by ARBs against selective neuronal damage due to ischemia in atherosclerotic ICA or MCA occlusive disease. However, no conclusion regarding cause and effect can be drawn as this is an observational, not an interventional, study. The effects of ARBs on BZRs are not yet fully clear, because the sample size in this study was small. The effect of blood pressure lowering or duration and severity of hypertension has also not been analyzed. Thus, future randomized and masked studies should compare the differences in neuronal damage, evidenced by decreased BZRs, between those patients with ARBs treatment and without.
Statin-treated hypercholesterolemia was previously shown to influence the BZR index in mixed symptomatic status patients (Yamauchi et al, 2007), but its effect was not significant in the present study of asymptomatic patients, perhaps because of limited statistical power caused by small sample size.
Although this study shows selective neuronal damage, manifested as decreased BZRs in the otherwise apparently normal cerebral cortex, the impact of this damage on clinical outcomes in asymptomatic patients remains to be established. Reduced cognitive performance has been demonstrated in patients with asymptomatic ICA disease and hemodynamic compromise without brain infarcts (Silvestrini et al, 2009). That finding, in combination with ours, suggests that hemodynamic compromise leading to silent cortical neuronal damage may contribute to the development of subtle cognitive impairment in atherosclerotic ICA or MCA disease. Although the present study could not include neuropsychological examinations, a relationship between a decrease in BZRs and cognitive impairment was demonstrated in a follow-up study of patients with carotid endarterectomy and hyperperfusion syndrome (Chida et al, 2009).
This study was subject to certain limitations besides limitations from a study design (an observational study). The normal control database was based on scans of only 10 healthy control subjects who are significantly younger than the patients, although our previous study showed no significant change in Z index during the follow-up (mean 41±3 months) in 7 of these 10 control subjects. We were unable to correct the data for partial volume effects, because the MRI studies were conducted for specific clinical purposes. Therefore, the decrease in cortical FMZ binding could at least partly reflect the effect of cortical atrophy and a partial volume effect, although the results of follow-up examinations in patients without apparent changes on MRI indicated that the decrease in cortical FMZ binding did not simply reflect a partial volume effect (Yamauchi et al, 2007). It may be ideal that the data are analyzed by using three-dimensional SSP and stereotactic extraction estimation method after corrections of PET data for atrophy based on coregistration of MR and PET scans (Kato et al, 2008). Although we created FMZ BP parametric maps, large interindividual variations of absolute whole-brain values for FMZ BP necessitated normalization to the mean cerebellar value to carry out subsequent analyses. A Z score threshold of 2 was used to extract abnormal pixels without taking into account the issue of multiple comparisons. Thus, the reassessment of BZR index may be subject to bias, despite the objectivity of this index as a measure of cortical neuronal damage in the individual patient. Although we consider the BZR index a useful quantitative measure of decreases in BZRs, further studies should be conducted to validate it.
In conclusion, in asymptomatic atherosclerotic ICA or MCA disease, hemodynamic compromise is associated with selective neuronal damage manifested as decreases in cortical BZRs in the noninfarcted cerebral cortex, whereas ARBs are associated with preservation of cortical BZRs. In asymptomatic patients, hemodynamic compromise may be associated with risk for ischemic cortical neuronal damage. This suggests that vascular reconstruction surgery might be indicated to prevent cortical neuronal damage, and ARBs might also confer beneficial effects against neuronal damage. As no conclusion regarding cause and effect can be drawn from this observational study, future randomized and masked studies should be performed to determine whether these strategies can prevent ischemic cortical neuronal damage in atherosclerotic ICA or MCA disease.
Footnotes
Acknowledgements
The authors thank Hidehiko Okazawa, MD, PhD (Biomedical Imaging Research Center, Fukui University) and the staff of the PET unit, Research Institute, Shiga Medical Center, for their support and technical help. The authors also thank the staff of the Department of Neurology and the Department of Neurosurgery, Shiga Medical Center, for clinical assistance.
The authors declare no conflict of interest.
