Abstract
The adhesion of both leukocytes and platelets to microvascular endothelial cells has been implicated in the pathogenesis of ischemia/reperfusion (I/R) injury in several vascular beds. The objectives of this study were to (1) assess the platelet–leukocyte–endothelial cell interactions induced in the cerebral microvasculature by middle cerebral artery occlusion (MCAO)/reperfusion, and (2) define the molecular determinants of the prothrombogenic and inflammatory responses in this model of focal I/R. MCAO was induced for 1 hour in wild-type (WT) mice, WT mice treated with a monoclonal antibody (mAb) to either P-selectin or GPIIb/IIIa, and in P-selectin−/−(P-sel−/−) chimeras. Isolated platelets labeled with carboxyfluorescein diacetate succinimidyl ester (CFDASE) were administered intravenously and observed with intravital fluorescence microscopy. Leukocytes were observed after intravenous injection of rhodamine 6G. One hour of MCAO followed by 1 hour of reperfusion resulted in the rolling and adhesion of leukocytes in venules, and after 4 hours of reperfusion, the adhesion of both leukocytes and platelets was detected. Although both the P-selectin and GPIIb/IIIa mAbs significantly reduced the adhesion of leukocytes and platelets at 4 hours of reperfusion, the antiadhesive effects of the P-selectin mAb were much greater. The leukocyte and platelet adhesion responses were significantly attenuated in both P-sel−/−→WT and WT→P-sel−/− bone marrow chimeras, compared with WT→WT chimeras. Neutropenia, induced by antineutrophil serum treatment, also reduced the recruitment of leukocytes and platelets after cerebral I/R. These findings implicate a major role for both platelet-associated and endothelial cell–associated P-selectin, as well as neutrophils in the inflammatory and prothrombogenic responses in the microcirculation after focal cerebral I/R.
Ischemic strokes remain a leading cause of death in North America, but the mechanisms underlying this disease process remain poorly understood. In recent years, a number of studies have implicated a role for inflammatory mediators and leukocyte–endothelial cell adhesion in the pathogenesis of cerebral ischemia/reperfusion (I/R) injury. It has been repeatedly shown that I/R elicits an inflammatory response that is manifested by an accumulation of adherent leukocytes in the cerebral microvasculature, with the subsequent extravasation of these inflammatory cells into the interstitial spaces where they can injure neuronal tissue. The importance of this adhesion-dependent recruitment of inflammatory cells in the pathogenesis of stroke is evidenced by numerous reports that describe protection against tissue injury and improved neurologic function in experimental animals treated with adhesion molecule–specific monoclonal antibodies (mAbs) (Matsuo et al., 1994; Suzuki et al., 1999; Zhang et al., 1995, 1994) and in mutant mice that are genetically deficient in certain adhesion molecules (Connolly et al., 1997; Kitagawa et al., 1998; Soriano et al., 1996). The technique of intravital video microscopy has been used to visualize and quantify the accumulation of circulating blood cells on the endothelial surface of cerebral microvessels after exposing the brain to global porcine (Gidday et al., 1997), gerbil (Uhl et al., 2000), rat (Dirnagl et al, 1994), and mouse (Ishikawa et al., 2003) ischemia or to focal (Ishikawa et al., 1999; Ritter et al., 2000) ischemia, followed by reperfusion. These studies have revealed a time-dependent recruitment of adherent leukocytes in cerebral venules after reperfusion and have helped to define the specific adhesion molecules that mediate this recruitment process.
Using a murine model of cerebral global ischemia and reperfusion, we have recently shown that the accumulation of adherent leukocytes in postischemic venules is accompanied by the adhesion of platelets to the vessel wall (Ishikawa et al., 2003). Mutant mice that are genetically deficient in either P-selectin or intercellular adhesion molecule-1 (ICAM-1) were used to show that both of these adhesion molecules contribute to the I/R-induced recruitment of both leukocytes and platelets in the global I/R model. The results of these experiments suggest that the adhesion of platelets and the induction of a procoagulant state in cerebral microvessels may involve both platelet- and endothelial cell–associated P-selectin, and that leukocyte adherence is required for the I/R-induced platelet accumulation. Two molecular models were proposed to explain the dependence of platelet accumulation on ICAM-1, an adhesion molecule that normally serves to mediate leukocyte, but not platelet, adhesion in postcapillary venules. One model proposed that ICAM-1–bound fibrinogen on endothelial cells may serve as a ligand for GPIIb/IIIa on platelets, whereas a second model proposes that endothelial P-selectin and ICAM-1 mediate the rolling and firm adhesion (respectively) of leukocytes in postischemic venules and that the adherent leukocytes then create a platform onto which platelets can bind using P-selectin. The former model suggests that GPIIb/IIIa blockade would be effective in reducing the I/R-induced recruitment of platelets in venules, whereas the latter model predicts that platelet accumulation would not occur in the absence of circulating leukocytes, and that blockade of either endothelial cell-or platelet-associated P-selectin would blunt both the leukocyte and platelet recruitment responses to I/R.
The objectives of this study were to (1) determine the time course and magnitude of platelet and leukocyte adhesion in mouse cerebral venules exposed to middle cerebral artery occlusion (MCAO)/reperfusion, (2) assess the contributions of P-selectin and GPIIb/IIIa to the I/R-induced recruitment of both platelets and leukocytes in the postischemic cerebral microvasculature, (3) define the contributions of platelet-associated versus endothelial cell–associated P-selectin to the I/R-induced inflammatory and procoagulant state using bone marrow chimeras that are selectively deficient in either platelet or endothelial cell P-selectin, and (4) determine whether I/R-induced platelet adhesion in cerebral venules is altered in mice rendered neutropenic with antineutrophil serum.
MATERIALS AND METHODS
Animal preparations
Experiments were performed on male C57Bl/6J mice, weighing 21 to 25 g. The animals were anesthetized with an intraperitoneal injection of alpha-chloralose (60 mg/kg) and urethane (600 mg/kg), while lidocaine (1%) was used for local anesthesia. All mice were tracheostomized with a polyethylene catheter (PE90, Intramedic; Clay Adams, Parsippany, NJ, U.S.A.) and artificially ventilated (Harvard Rodent Ventilator, Model 683; Harvard Apparatus, South Natick, MA, U.S.A.) with room air during observation with intravital microscopy. In some instances, 0.4 mg/kg pancuronium (Sigma, St. Louis, MO, U.S.A.) was administered (intravenously) to facilitate breathing. The femoral artery and vein were cannulated with a polyethylene catheter (PE10, Intramedic; Clay Adams) to monitor mean arterial blood pressure, sample arterial blood for blood gas analysis, and for intravenous administration of the labeled platelets and rhodamine 6G. Rectal temperature was monitored and maintained at 36.5°C to 37.5°C with an overhead heat lamp throughout the experimental procedure.
MCAO and brain preparation
In the ischemic groups, anesthesia was induced by 4% (and maintained with 3%) isoflurane in air using a facemask. The middle cerebral artery was occluded using the intraluminal filament method (6–0 nylon), as previously described (Hata et al., 1998; Sampei et al., 2000). Briefly, after a midline neck incision, the left external carotid and pterygopalatine arteries were isolated and ligated with 5–0 silk thread. The internal carotid artery was occluded at the peripheral site of the bifurcation of the internal carotid artery (ICA) and the pterygopalatine artery with a small clip and the common carotid artery was ligated with 5–0 silk thread. The external carotid artery (ECA) was cut and a 6–0 nylon monofilament, whose tip was blunted with a coagulator (Geigen-NY, Model-100, Geiger Instrument, Inc., Monarch Beach, CA, U.S.A.) (the tip size was 0.2–0.22 mm), was inserted into the ECA. The ECA and the inserted nylon thread were tightened with a 6–0 silk suture, which prevented bleeding during advancement of the nylon thread and during removal of the nylon at the time of reperfusion, and rotated for its advancement into the ICA. After removal of the clip at the ICA, the nylon thread was advanced until light resistance was felt and the distances from the nylon thread tip to the internal carotid artery-pterygopalatine artery bifurcation was slightly more than 6 mm and the distance to the ICA/ECA bifurcation was slightly less than 9 mm. (During MCAO, the parietal bone becomes pale on the occluded side and laser-Doppler flowmetry reveals that blood flow in this area falls to less than 10% of baseline.) The nylon thread and the common carotid artery ligature were removed after the 1-hour occlusion period. In the sham group, these arteries were visualized but not disturbed. A second control group (designated “sham-filament”) was used to distinguish the effects of MCAO/reperfusion from potential nonspecific effects of transient filament advancement. In this group, the same procedures were applied as in the 1-hour MCAO/4-hour reperfusion group, except that the filament was advanced into the ICA (about 7 or 8 mm from the ICA-ECA bifurcation) but not far enough to elicit cerebral ischemia over the 1-hour period. Four hours after withdrawal of the filament (simulated reperfusion period), the microcirculatory measurements were taken.
The head of each mouse was fixed in a plastic frame in a sphinx position. The left parietal bone was exposed by a midline skin incision, followed by a craniectomy (diameter: 2.5 mm) with a drill at 1 mm posterior from the bregma and 4 mm lateral from the midline. The observation area experiences a more than 90% reduction in blood flow (assessed by laser-Doppler flowmetry) and represents the major infarcted region after MCAO. The dura matter was not cut because the fluorescently labeled platelets and leukocytes are readily be visualized and intracranial pressure is well maintained without cutting this tissue (Ishikawa et al. 2003). A 12-mm glass coverslip was placed over the craniectomy and the space between the glass and dura mater was filled with artificial CSF. These experimental procedures were reviewed and approved by the LSU Health Sciences Center Institutional Animal Care and Use Committee.
Platelet preparation
Approximately 0.9 mL of blood was harvested via a catheter placed in the carotid artery. The blood was collected in polypropylene tubes containing 0.1 mL acid citrate-dextrose ACD buffer (Sigma) and centrifuged at 120g for 10 minutes. Plateletrich plasma and the platelet layer were transferred to a polypropylene tube and centrifuged at 550g for 10 minutes. Carboxyfluorescein diacetate succinimidyl ester (CFDASE; Molecular Probes, Eugene, OR, U.S.A.) was dissolved in dimethyl sulfoxide (final concentration: 0.6%) to a concentration of 14.9 mmol/L. The platelet pellet was resuspended with 1500 μL of phosphate-buffered saline (PBS, pH 7.4) and incubated with 9 μL CFDASE solution for 10 minutes at room temperature. After centrifugation of the incubated platelets at 550g for 10 minutes, the cell pellet was resuspended with 500 μL of PBS, stored on ice, and protected from light. Manual blood cell counts yielded less than 0.05% leukocytes to platelets in the platelet suspension. Platelets were derived from wild-type (WT) mice for all experiments except in one experimental group, where platelets were obtained from P-selectin–deficient (P-sel−/−) mice. In each mouse, 100 × 106 platelets (approximately 30 minutes after final suspension) were infused over 5 minutes using a Harvard Apparatus infusion pump, yielding about 10% of the total platelet count. The platelets were allowed to circulate for a period of 5 minutes before recording (Ishikawa et al., 2003). We have previously shown, using flow cytometric measurements of platelet P-selectin and GPIIb/IIIa, that the isolation procedure produces minimal activation of platelets (Russell et al., 2003; Tailor and Granger, 2003). This assumption is supported by the minimal levels of platelet–vessel wall adhesion observed in the microcirculation of the brain (see Results) and other tissues (Russell et al., 2003; Tailor and Granger, 2003).
Bone marrow chimeras
Three combinations of chimeras were produced (Stokes et al., 2001). WT→WT chimeras were WT mice (CD45.2) that received bone marrow cells from CD45.1 congenic mice, preserving P-selectin function on both platelets and endothelial cells. This resulted in a significant increase in circulating leukocytes that express CD45.1 of donor origin, from less than 5% in WT to more than 90% in the WT→WT chimeras. CD45.1 congenic (WT) bone marrow was transplanted into P-sel−/− mice (CD45.2), yielding WT→P-sel−/− chimeras with normal P-selectin function in circulating blood cells (platelets) but a P-selectin–deficient vessel wall. P-sel−/− bone marrow (CD45.2) was transplanted into CD45.1 congenic (WT) mice, yielding P-sel−/−→WT chimeras with normal vessel wall P-selectin function, but P-selectin–deficient blood cells (platelets).
Bone marrow transfer required the isolation of marrow cells from the femurs and tibias of donor mice and resuspension at 4 × 107 cells/mL PBS. Recipient mice were irradiated with two doses of 500 to 525 rad, 3 hours apart, after which 8 × 106 donor bone marrow cells in 200 μL of PBS were injected into the femoral vein. The chimeras were kept in autoclaved cages, with 0.2% neomycin drinking water for the first 2 weeks. Flow cytometry was used to verify chimera reconstitution by staining for CD45.1 and CD45.2 expression on circulating leukocytes with FITC-labeled anti-CD45.1 and CD45.2 antibodies. The chimera production procedure normally yields greater than 90% penetrance of the transferred marrow at 6 to 8 weeks. Therefore, the mice were studied approximately 3 months after marrow transplantation.
Intravital fluorescence microscopy
An upright Nikon microscope equipped with a silicon-intensified target camera (C2400 to 08; Hamamatsu Photonics, Hamamatsu, Japan) and a mercury lamp was used to observe the cerebral microcirculation. With ×20 and ×40 objectives, the magnifications on the video screen (Sony, Tokyo, Japan) were ×560 and ×1,120, respectively. The microscopic images were received by a CCD video camera that was attached to an image intensifier. The images were recorded on a video recorder (Sony) equipped with a video timer (Time-Date Generator, WJ-810; Panasonic, Tokyo, Japan). CFDASE, which forms the stable fluorochrome carboxyfluorescein succinimidyl ester (absorption peak of 492 nm, emission peak of 518 nm) after reaction with intracellular esterases, and rhodamine-6G (absorption peak of 525 nm; emission peak of 555 nm) imaging required two filter blocks with an excitation filter for 450 to 490 nm (Nikon, B-2A, dichroic mirror for 510 nm, and barrier filter of 520 nm) and an excitation filter for 510 to 560 nm (Nikon, G-2A, dichroic mirror for 580 nm, and barrier filter of 590 nm) (Ishikawa et al., 2003).
Video analysis
The randomly selected venular segments evaluated for platelet and/or leukocyte adhesion were all branches of the middle cerebral artery and were 30 to 40 μm in diameter and at least 100 μm in length. Platelets were classified according to the quality or duration of their interaction with the venular wall as free-flowing, rolling, or adherent. Rolling platelets and leukocytes were defined as cells crossing the 100-μm venular segment at a velocity that is significantly lower than the centerline velocity; their numbers are expressed as cells per 30 seconds per square millimeter. Adherent platelets and leukocytes were stationary for more than 2 seconds and 30 seconds, respectively. Rolling and adherent cells were expressed as the number of cells per square millimeter of venular surface, calculated from diameter and length, assuming cylindrical vessel shape.
Experimental protocols
Labeled platelets were infused and their interactions recorded in five randomly selected venular segments. All venules visualized and used for data collection in our experiments were branches that originate from the middle cerebral artery. These venules were exposed to ischemia during the entire period of MCAO in all experimental groups. Some very low flow is visualized in arterioles in this region during MCAO, which likely reflects retrograde flow from the anterior and/or posterior cerebral artery. Rolling and adherent leukocytes were then monitored and recorded in the same venular segments. The latter was achieved by intravenous administration of 50 μL of 0.02% rhodamine-6G (Sigma), followed by a continuous infusion (2 mL/h) of the fluorochrome at the same concentration for 5 to 10 minutes. WT mice were assigned to one of four experimental groups: (1) 1-hour MCAO and 1-hour reperfusion, (2) 1-hour MCAO and 4-hour reperfusion, (3) 5 hours between sham operation and platelet monitoring, or (4) 30-minute MCAO and 4-hour reperfusion. In those experiments that evaluated the effects of treatment with either a P-selectin (RB40.34, 2mg/kg) or GPIIb/IIIa (1B5, 2mg/kg, kindly provided by Dr. Barry Coller from Rockefeller University) mAb, mice were exposed to the 1-hour ischemia and 4-hour reperfusion protocol (as per group 2) before recording platelet-vessel wall and leukocyte–vessel wall interactions. These blocking mAbs were administered 30 minutes before the induction of MCAO at a dose previously shown to block adhesion molecule function in vivo (Ley et al., 1995; Smyth et al., 2001). A separate group of mice were pretreated (24 hours before the experiment) with antineutrophil serum (150 μg/mouse) for depletion of blood neutrophil count. This treatment reduced the circulating number of neutrophils by more than 95%. In the three groups of bone marrow chimeras, platelets and leukocytes were observed after 1 hour MCAO followed by 4 hours of reperfusion. CFDASE-labeled WT platelets were infused into, and monitored in, WT→WT chimeras and WT→P-sel−/− chimeras. CFDASE-labeled, P-sel−/− platelets were monitored in the P-sel−/−→WT chimeras.
Statistical analysis
Data were analyzed using an analysis of variance and Fisher post hoc test. The data are reported as means ± SD. Statistical significance was set at P < 0.05.
RESULTS
Venular diameter (33 ± 8 μm) as well as blood pressure (82 ± 7 mm Hg), blood pH (7.35 ± 0.06), PO2 (106 ± 12 mm Hg), and PCO2 (33 ± 5 mm Hg) (obtained after platelet and leukocyte adhesion measurements) in the different experimental groups showed no statistically significant differences.
Leukocyte–endothelial cell interactions
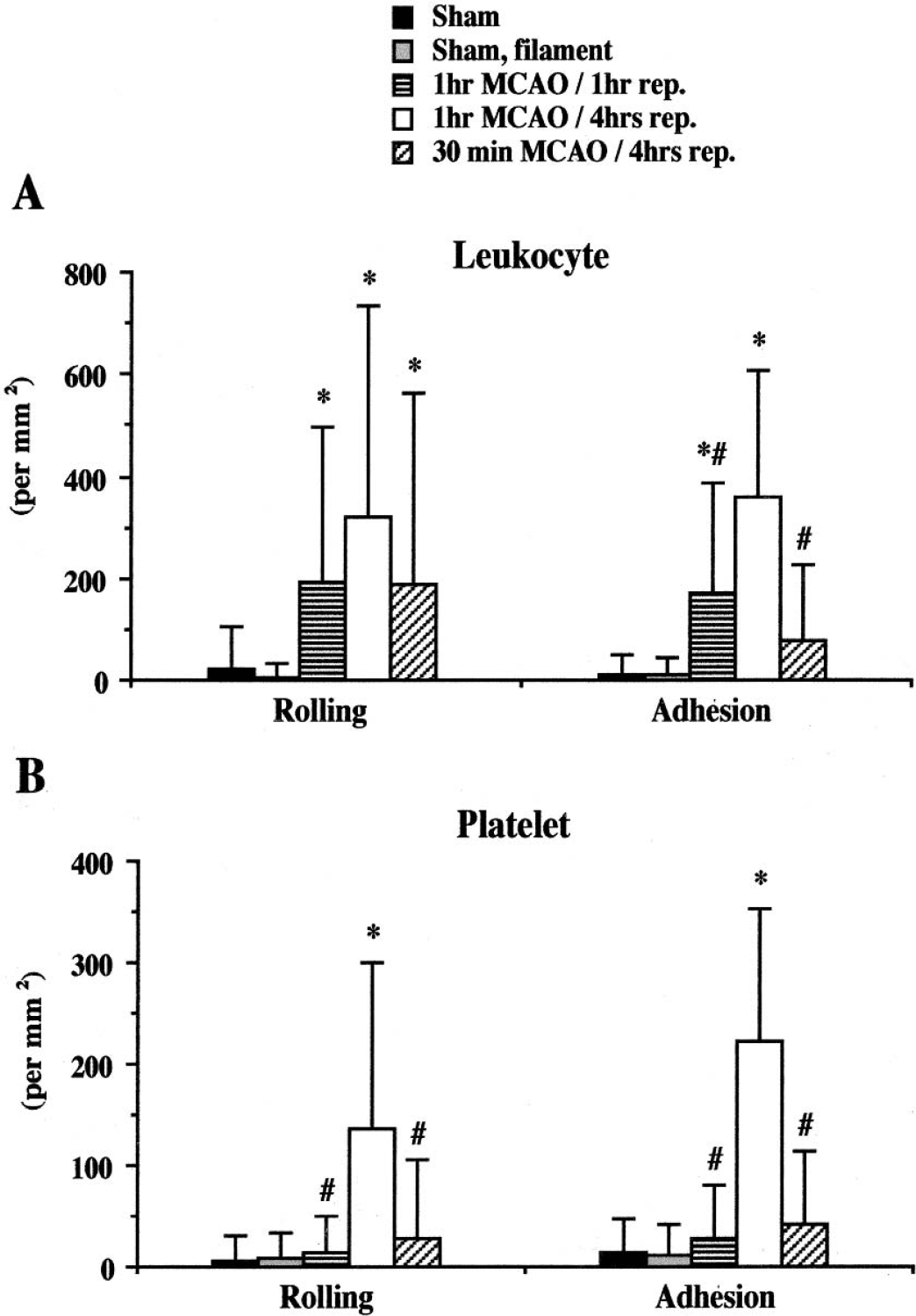
Negligible levels of leukocyte and platelet adhesion were observed in cerebral venules of sham and sham, filament mice, and there were no significant differences in the responses between these two groups. MCAO followed by 1-hour or 4-hour reperfusion resulted in significant increases in the numbers of rolling and adherent leukocytes (Figs. 1A and 2) when compared with the responses measured in sham experiments. The number of adherent leukocytes observed after either 1 hour of MCAO followed by 1 hour of reperfusion or 30 minutes of MCAO and 4 hours of reperfusion were significantly lower than that noted in mice subjected to 1-hour MCAO and 4-hour reperfusion (Fig. 2). Before ischemia and after either 1 hour or 4 hours of reperfusion, no leukocytes were noted to either roll or adhere in cerebral arterioles.
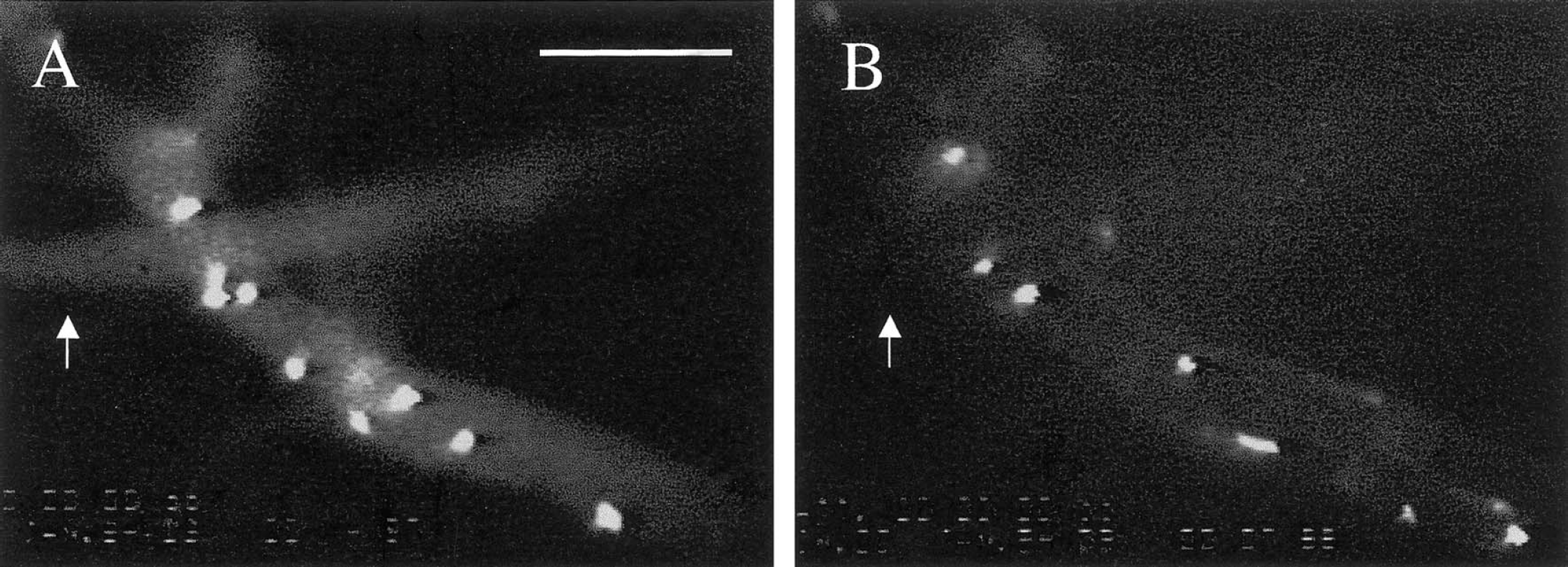
Images depicting leukocyte (
The changes in the rolling and adhesion of leukocytes (
Platelet–endothelial cell interactions
In contrast to the leukocyte adhesion responses described earlier, the numbers of rolling and adherent platelets were unaffected by either 1-hour MCAO/1-hour reperfusion or 30-minute MCAO/4-hour reperfusion when compared with values obtained from a sham experiment. However, large and highly significant increases in platelet rolling and adherence were noted in mice subjected to 1-hour MCAO and 4-hour reperfusion (Figs. 1B and 2). These I/R-induced adhesive interactions were detected only in venules, with no such interactions noted in arterioles of different sizes (10–100 μm).
Adhesion of leukocytes and platelets after treatment with mAb against P-selectin or GPIIIb/IIIa
The I/R-induced recruitment of rolling and adherent leukocytes was significantly attenuated in P-selectin mAb-treated mice, compared with the responses noted in untreated mice exposed to 1-hour MCAO and 4-hour reperfusion (Figs. 3A and 3B). However, the number of adherent leukocytes after P-selectin mAb treatment remained elevated above the level detected in sham experiments. Both the rolling and firm adhesion of platelets was reduced to sham levels in the P-selectin mAb-treated mice. Although treatment with a GPIIb/IIIa mAb significantly reduced the adhesion of both platelets and leukocytes after I/R, the reductions noted were relatively small compared with the responses seen with the P-selectin mAb.
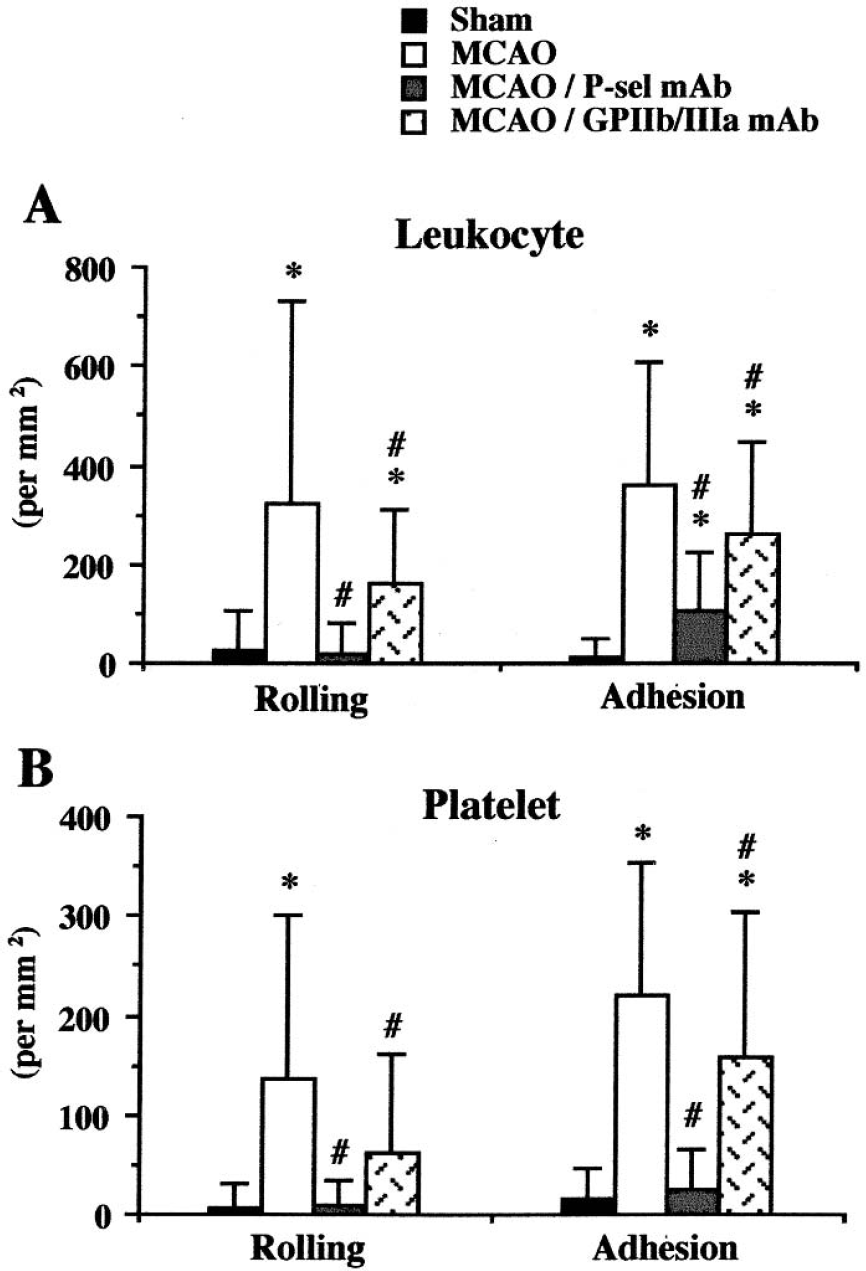
Effects of mAbs against either P-selectin or GPIIb/IIIa on the adhesion of leukocytes and platelets in murine venules after 1-hour MCAO and 4-hours reperfusion. Four to six animals were studied in each group. *P < 0.05 relative to the corresponding sham value; #P < 0.05 relative to the corresponding MCAO group. The P-selectin mAb was more effective in attenuating the I/R-induced blood cell adhesion than the GPIIb/IIIa mAb.
Leukocyte and platelet adhesion in P-selectin–deficient bone marrow chimeras
Our findings related to the role of platelet-associated and endothelial cell–associated P-selectin in mediating the I/R-induced rolling and adhesion of leukocytes and platelets are summarized in Fig. 4. In P-sel−/−→WT chimeras, the number of rolling and adherent leukocytes were significantly but not completely reduced compared with WT→WT chimeras. However, compared with WT→WT chimeras, the rolling and firm adhesion of platelets was virtually abolished after I/R. In WT→P-sel−/− chimeras, the number of rolling and adherent leukocytes and platelets also decreased significantly compare with WT→WT chimeras; however, some adhesion of leukocytes and platelets was still detected after I/R, similar to the responses noted in the group treated with the P-selectin blocking mAb.
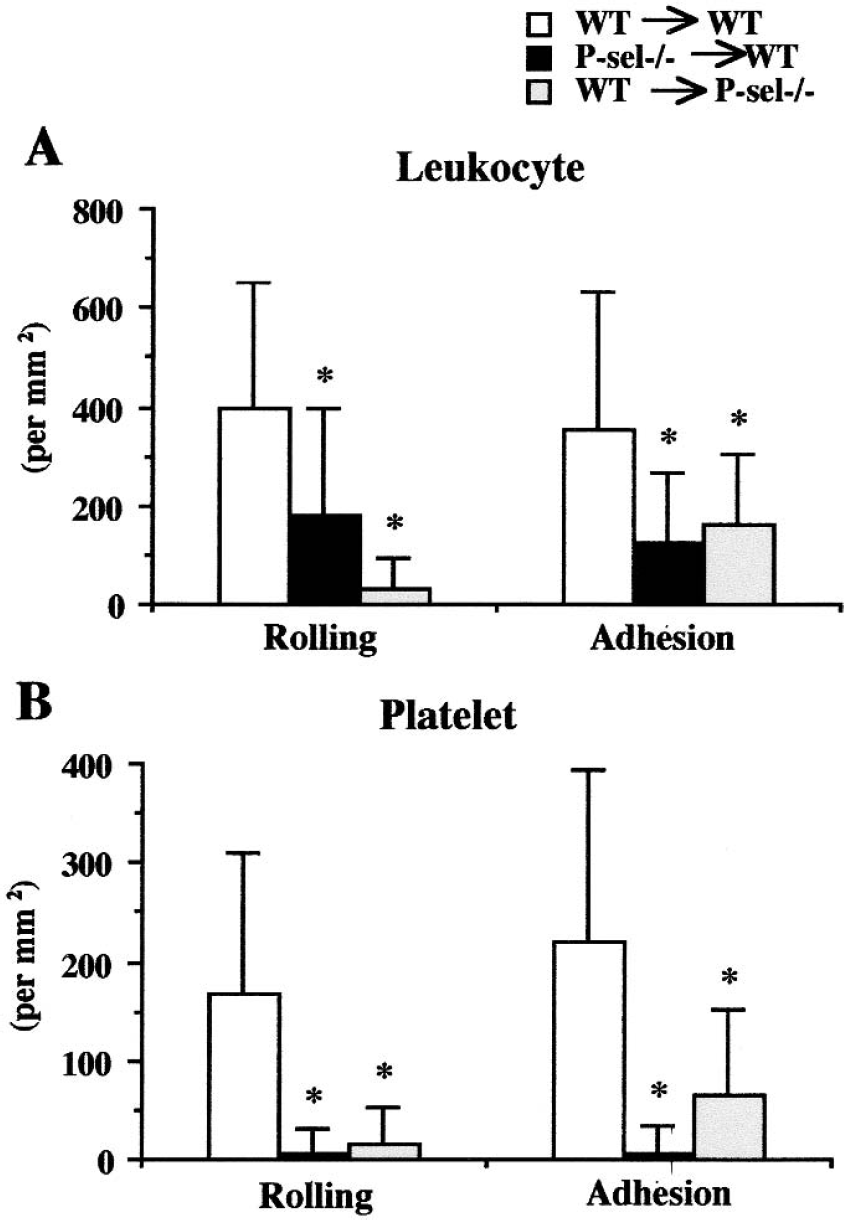
Role of platelet-associated and endothelial cell–associated P-selectin in mediating I/R-induced rolling and adhesion of leukocytes (
Platelet–leukocyte interaction
Platelets were noted to adhere directly onto adherent leukocytes and platelet-bearing leukocytes were occasionally observed rolling in postischemic venules. Some free-flowing platelets were seen to suddenly bind (without rolling) on adherent leukocytes. Some of these platelets detached from the adherent leukocyte whereas others adhered firmly on the leukocyte. Other platelets were seen to roll and adhere directly on venular endothelium.
To quantify the contribution of leukocytes to I/R-induced platelet recruitment, some mice were rendered neutropenic with antineutrophil serum. Although leukocyte rolling and adherence were still observed in cerebral venules of serum-treated mice after I/R, the responses were dramatically reduced. The cerebral venules of neutropenic mice also exhibited large and significant reductions in rolling and adherent platelets after I/R (Fig. 5).
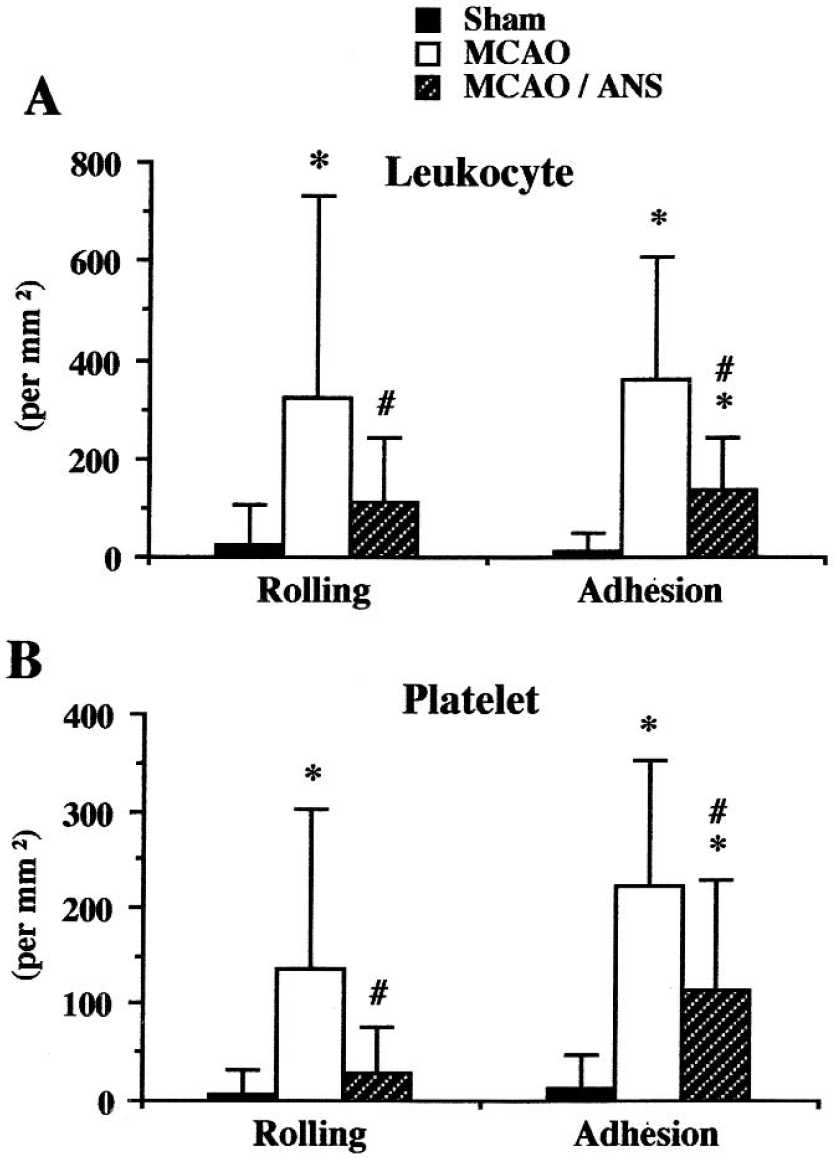
Effect of antineutrophil serum (ANS) on leukocyte and platelet adhesion in pial venules after 1-hour MCAO and 4-hour reperfusion. Six animals were studied in each group. *P < 0.05 relative to the corresponding sham value; #P < 0.05 relative to the corresponding MCAO group. Rolling and adhesion of leukocytes and platelets were significantly decreased, but not to sham levels.
DISCUSSION
Time course of platelet and leukocyte adhesion in focal ischemia
Leukocyte–endothelial cell interactions have been previously described in porcine (Gidday et al., 1997), gerbil (Uhl et al., 2000), rat (Dirnagl et al., 1994), and murine (Ishikawa et al., 2003) models of global I/R, and after focal I/R in the rat (Ishikawa et al., 1999; Ritter et al., 2000). The present study represents the first attempt to systematically evaluate the effects of focal I/R on the adhesive interactions of blood leukocytes and platelets with the walls of postcapillary venules in the cerebral circulation of the mouse. We found that 1 hour, but not 30 minutes, of MCAO followed by 1 hour of reperfusion elicited the recruitment of rolling and adherent leukocytes within cerebral venules. One hour of MCAO followed by a longer period of reperfusion (i.e., 4 hours) was associated with more profound increases in the number of rolling and firmly adherent leukocytes. Platelet rolling and adhesion, however, was only observed after 1 hour of ischemia and 4 hours of reperfusion. This tendency for leukocytes to exhibit a more robust recruitment response than platelets early after the onset of reperfusion is similar to that previously noted in a C57Bl/6 mouse model of global cerebral I/R, i.e., after bilateral common carotid artery occlusion (BCCAO) (Ishikawa et al., 2003). Of interest is that the magnitude of the rolling and adhesion responses of both platelets and leukocytes noted in the focal (MCAO) cerebral I/R model is very similar to the responses seen in the global (BCCAO) I/R model, suggesting that the intensity of the stimulus for leukocyte and platelet recruitment in cerebral vessels is similar for both global and focal ischemia, provided that the venules draining the middle cerebral artery are exposed to similar durations of ischemia and reperfusion. Finally, as noted previously for global I/R in mouse brain (Ishikawa et al., 2003), the recruitment of platelets and leukocytes after focal I/R is limited to the postcapillary segment of the microcirculation, with no significant accumulation noted in arterioles.
The platelet adhesion receptor GPIIb/IIIa (αIIb/β3) has also been implicated in the adhesion of platelets in intestinal postcapillary venules exposed to I/R (Massberg et al., 1999). It was shown that platelets derived from patients with Glanzmann's disease, which are lacking in GPIIb/IIIa, exhibited profoundly lower levels of adhesion in murine intestinal venules after I/R when compared with platelets derived from healthy volunteers. Because the I/R-induced platelet adhesion was also dependent on both fibrinogen and ICAM-1, the authors proposed that ICAM-1–bound fibrinogen on endothelial cells serves as a ligand for GPIIb/IIIa on platelets, thereby allowing for adhesion of platelets to endothelial cells. If such a mechanism contributes to the recruitment of platelets in cerebral venules after focal I/R, then one would expect GPIIb/IIIa blockade to attenuate platelet accumulation in venules after reperfusion. Our finding of a partial reduction in the I/R-induced platelet adhesion (primarily rolling) supports a role for this platelet adhesion receptor. However, the magnitude of the attenuation in platelet adhesion afforded by mAb 1B5, which has previously been shown to protect C57Bl/6 mice against the systemic intravascular thrombosis induced by prothrombotic agents such ADP (Smyth et al., 2001), was significantly less than that observed with the P-selectin blocking mAb, suggesting a larger role for P-selectin than GPIIb/IIIa in this model. Exactly how GPIIb/IIIa contributes to the I/R-induced platelet recruitment in cerebral venules remains unclear. The process of platelet GPIIb/IIIa engagement to ICAM-1–bound fibrinogen on endothelial cells that has been described for postischemic intestinal venules (Massberg et al., 1999) may also apply to the brain microcirculation. However, other mechanisms, such as platelet GPIIb/IIIa adhesion to von Willebrand factor–bound collagen on endothelial cells (Bogousslavsky et al., 2001), may also be involved.
Contributions of platelet-associated versus endothelial cell–associated P-selectin
Another major objective of this study was to determine whether the P-selectin that participates in the recruitment of leukocytes and platelets in cerebral venules after I/R is associated with endothelial cells and/or platelets. To address this issue, bone marrow chimeras were produced that are selectively deficient in either endothelial cell (WT→P-sel−/−) or platelet (P-sel−/−→WT), and the I/R-induced blood cell recruitment responses were compared with those of control (WT→WT) chimeras. This approach was previously used by Carvalho-Tavares et al (2000) to show that although platelets contribute to the leukocyte adhesion seen in tumor necrosis factor-α–stimulated brain microcirculation, platelet P-selectin did not mediate the leukocyte adhesion. Our application of the P-selectin chimeras to a brain model of focal I/R has revealed a role for both endothelial cell–associated and platelet-associated P-selectin in mediating the recruitment of rolling and firmly adherent leukocytes and platelets (Fig. 4). This result is consistent with our previously reported findings in a model of global cerebral I/R (Ishikawa et al., 2003) that used an entirely different experimental strategy to address this issue, i.e., monitoring the adhesion of platelets from WT mice in venules of P-sel−/− mice or P-sel−/− platelets in venules of WT mice. This approach also yielded results that implicate a role for both endothelial cell and platelet P-selectin in mediating the recruitment of platelets. These findings, in combination with data obtained from ICAM-1−/− and P-sel−/− mice, led to the proposal that endothelial P-selectin and ICAM-1 mediate the rolling and firm adhesion (respectively) of leukocytes in postischemic cerebral venules after global I/R, and that the adherent leukocytes then create a platform onto which platelets can bind using P-selectin (Ishikawa et al., 2003). The present study suggests that a similar mechanism may be involved in the recruitment of leukocytes and platelets after focal I/R in the brain.
Dependence of platelet adhesion on circulating leukocytes
The contention that platelet recruitment into cerebral venules is dependent on the recruitment of leukocytes after global I/R was based on evidence that leukocyte and platelet adhesion are both significantly attenuated in ICAM-1−/− mice (Ishikawa et al., 2003). In the present study, we reasoned that if leukocyte adhesion is a prerequisite for platelet recruitment after focal brain I/R, then depletion of the circulating pool of neutrophils (the dominant leukocyte population recruited into postischemic microvessels) should result in an attenuated recruitment of leukocytes as well as platelets. This experimental strategy resulted in the expected reductions in both leukocyte rolling and firm adhesion. This was accompanied, however, by significant reductions in the recruitment of rolling and adherent platelets, indicating that leukocytes do indeed contribute to the platelet adhesion seen in cerebral venules after focal I/R. While the codependence of platelet and leukocyte recruitment is not unique to this model of acute inflammation, the nature of the codependency appears to differ somewhat between models. For example, in the tumor necrosis factor-α–stimulated brain microvasculature, neutrophil recruitment is dependent on platelet accumulation but independent of platelet P-selectin (Carvalho-Tavares et al., 2000). There is also evidence for a dependence of neutrophil recruitment in venules of the postischemic intestine. Salter et al (2001) showed that neutrophil accumulation in the postischemic intestine is attenuated in rats that are rendered thrombocytopenic with antiplatelet serum or that receive monoclonal antibodies directed against P-selectin, GPIIb/IIIa, or fibrinogen. The underlying basis for the different patterns of codependency of leukocyte and platelet recruitment between models of acute inflammation and between regional vascular beds remains unclear and warrants further investigation.
A potential methodological limitation associated with intravital microscopic evaluation of the trafficking of blood cells in venules after focal cerebral ischemia relates to the location of the microvessels studied relative to the penumbral cortex. The venules examined were all branches of the middle cerebral artery and all were exposed to the same ischemic insult and duration of reperfusion. Nonetheless, it is conceivable that genetic and pharmacologic manipulations, as used in this study, change the spatial relationship of the venules with the injured tissue that produces the inflammatory signals that elicit the recruitment of blood cells. Because P-selectin–directed interventions are known to reduce infarct size in the postischemic brain, it possible that the protection afforded by adhesion molecule ablation would also reflect an altered distance between the major tissue sources of the inflammatory signals and the venules monitored under the cranial window. A more difficult scenario to envision would involve an action of P-selectin–directed interventions to skew the location of the penumbral region away from the venules observed under the fixed cranial window. Nonetheless, this possibility cannot be excluded in our study because the location of the penumbra relative to the venules studied was not tracked.
