Abstract
Leukocytes play an important role in the development of ischemia/reperfusion injury. Recent work in our laboratory has demonstrated that a mixture of synthetic fibronectin peptides to leukocyte adhesion molecules reduces ischemic brain damage after transient focal cerebral ischemia. The purpose of this study was to evaluate the efficacy of the individual peptides on leukocyte accumulation, infarct size, and neurological outcome in rats subjected to 1 h of cerebral ischemia and 48 h of reperfusion. Thirty-five animals were divided into five groups: transient ischemia without treatment (Group I), treatment with arginyl-glycyl-aspartic acid (RGD) peptide (Group II), connecting segment (CS)-1 peptide (Group III), fibronectin (FN)-C/H-V peptide (Group IV), and scrambled FN-C/H-V peptide (Group V). Groups III and IV showed a significant decrease in the degree of leukocyte infiltration in the lesion and in the infarct size (p < 0.05) when compared to Groups I, II, and V. The neurological grade of Groups III and IV was significantly better than in Groups I, II, and V at 48 h after reperfusion (p < 0.01). Thus, in addition to demonstrating the potential efficacy of synthetic peptides as therapeutic agents for ischemia-reperfusion, these results also offer new insights into the mechanisms of leukocyte arrest and recruitment in ischemia/reperfusion injury.
Cerebral ischemia represents a major cause of morbidity and mortality, mainly because the cellular mechanisms that lead to primary or secondary brain damage are not completely understood. Recent evidence indicates that leukocytes play an important role in the development of ischemia/reperfusion injury by reducing microvascular blood flow, initiating thrombosis, and releasing free-oxygen radicals (Granger, 1988; Kochanek and Hallenbeck, 1992; Matsuo Y et al., 1995). Reduction of the number of peripheral leukocytes and inhibition of their adhesive interactions by such methods as treating with antibodies to leukocyte adhesion molecules have been demonstrated to ameliorate brain injury after ischemia/reperfusion (Clark et al., 1991; Bowes et al., 1993; Chen et al., 1994; Matsuo et al., 1994).
Leukocyte adhesion to endothelial cells and to extracellular matrix components has a complex molecular basis, and multiple adhesion receptor systems mediate the recruitment of leukocytes from the blood to the central nervous system (CNS) (Butcher, 1991). The initial trafficking of circulating leukocytes to sites of inflammation is mediated by the selectin family of adhesion receptors (Lasky, 1992; Bevilacqua and Nelson, 1993), followed by engagement of additional cellular recognition receptors, including the immunoglobulin superfamily and integrins (Hynes, 1987; Springer, 1990; Ruoslahti, 1991). Of the integrins, those containing β2 subunits are largely involved in cell-cell interactions, whereas β1 integrins usually are associated with mediating adhesion to extracellular matrix constituents (Ruoslahti, 1991; Hynes, 1987; Springer, 1990). One of these extracellular matrix macromolecules is fibronectin, which is found in plasma, cell matrix, and on the cell surface and can support leukocyte adhesion to endothelial cells (Akiyama and Yamada, 1987). Fibronectin possesses multiple domains recognized by integrins, including arginyl-glycyl-aspartic acid (RGD), which interacts with α5β1 [very late antigen (VLA)-5]; α3β1 (VLA-3); and other integrins (Brown and Juliano, 1985; Elices et al., 1991), and the alternately spliced connecting segment (CS)-1 domain, which is recognized by α4β1 (VLA-4) (Guan and Hynes, 1990). In addition to recognizing the CS-1 domain of fibronectin, α4β1 also serves as a receptor for the vascular cell adhesion molecule (VCAM)-1. Along with the integrins, there is also increasing evidence for a role for cell surface proteoglycans in mediating early steps in the inflammatory process (Iida et al., 1992; Woods et al., 1993). In this regard, interest has focused on polypeptides derived from the 33-kDa carboxyl-terminal heparin-binding domain of fibronectin. Synthetic fibronectin peptides, synthesized at our institution from several fibronectin domains for the specific purpose of inhibiting leukocyte adhesion, have shown promise in in vivo studies (Wahl et al., 1994; Hines et al., 1994). We have previously demonstrated that a mixture of synthetic fibronectin peptides reduces ischemic brain damage after transient focal cerebral ischemia in rats (Yanaka et al., 1996).
The purpose of the present study was to evaluate the efficacy of the individual peptides on leukocyte accumulation, infarct size, and neurological outcome in a rat model of transient focal cerebral ischemia.
MATERIALS AND METHODS
General protocol
Forty-two male Sprague-Dawley rats, weighting 270–350 g each, were used in this study. The experiment was approved by the Animal Care Committee of the University of Minnesota and carried out under the auspices of Research Animal Resources, an American Association for the Accreditation of Laboratory Animal Care-approved facility.
Animals were allocated to one of five groups: transient cerebral ischemia without treatment (Group I), treatment with i.v. administration of RGD peptide (Group II), treatment with i.v. administration of CS-1 peptide (Group III), treatment with i.v. administration of FN-C/H-V peptide (Group IV), and treatment with i.v. administration of scrambled FN-C/H-V peptide (Group V). Seven animals were excluded from the study: six did not meet the selection criteria as described below and one died during the period of reperfusion due to unexpected hemorrhage at the operative site. Of the six that did not meet selection criteria, one animal from Group I, one from Group III, and two from Group V were excluded from the study due to the presence of subarachnoid hemorrhage; one animal from Group I and one from Group II were also excluded because of the absence of neurological deficits. Each group was composed of seven animals.
Animal preparation and monitoring
All animal procedures were done under complete, general anesthesia. Anesthesia was induced with i.p. injection of a mixture of ketamine (87 mg/kg) and xylazine (13 mg/kg). After induction, animals were ventilated through a face mask with a mixture of oxygen and air. Arterial Pco2 was maintained between 35 and 40 torr. Rectal temperature was maintained between 37 and 38°C with heating pads. The right femoral artery was cannulated for monitoring arterial blood gas monitoring and peripheral white blood cell counting. Peripheral blood samples were obtained before induction of ischemia and at 48 h after reperfusion for measurement of the number of peripheral white blood cells and differentials. Measurements were made using a Celldyne 3500 cell counter (Abbott Diagnostics, San Jose, CA, U.S.A.). A PE-10 catheter was introduced into the inferior vena cava via the right femoral vein for i.v. administration of synthetic fibronectin peptides.
Transient focal cerebral ischemia in the area perfused by middle cerebral artery (MCA) was induced. Briefly, an incision was made in the midline of the neck and the right carotid bifurcation was exposed. The internal carotid artery (ICA) was followed rostrally, and the pterygopalatine branch identified and ligated. The common carotid artery (CCA) was then occluded, and the branches of the external carotid artery (ECA) were dissected and divided. The occluder, a 4–0 nylon suture with a silicone-coated tip, was then advanced from the ECA into the lumen of the ICA until it blocked the origin of the MCA. The average size of the silicone-coated portion was 0.25 mm in diameter and 12.0 mm in length. We chose an occluder that blocked the MCA at a depth of 19–20 mm. This was chosen after preliminary work showed that this produced the most consistent infarction. Reperfusion was accomplished by withdrawal of the suture. Animals underwent ischemia for 1 h and reperfusion for 48 h. All animals included in the study became awake and lucid and exhibited a movement disturbance of the left forepaw within 60 min after reperfusion. After surgery, animals were allowed free access to food and water. Neurological deficit characterized by left-sided hemiparesis and right Horner's syndrome were used as criteria for ischemic insult. Also excluded were any animals that had subarachnoid hemorrhage or lacked neurological deficits at 3 h after the induction of ischemia.
A neurological examination, as described by Zea Longa (1989), was performed 12, 24, and 48 h after occlusion in a blinded fashion. A standard scoring scale was used: 0, normal; 1, failure to extend the left forepaw; 2, circling to the left; 3, falling to the left; and 4, not walking spontaneously nor exhibiting a consciousness disturbance.
Fibronectin peptides synthesis and treatment
Fibronectin polypeptides containing the RGD domain, the alternatively spliced CS-1 (Asp-Glu-Leu-Pro-Gln-Leu-Val-Thr-Leu-Pro-His-Pro-Asn-Leu-His-Gly-Pro-Glu-Ile-Leu-Asp-Val-Pro-Ser-Thr), and one nonoverlapping sequence corresponding to the FN-C/H region (FN-C/H-V) (Trp-Gln-Pro-Pro-Arg-Ala-Arg-Ile) were synthesized and purified as described (McCarthy et al., 1988, 1990; Wahl et al., 1994; Woods et al., 1993; Hines et al., 1994). Briefly, fibronectin peptides were synthesized at the Microchemical facility of the University of Minnesota using a Beckman system 990 peptide synthesizer (Beckman Instruments Inc., Fullerton, CA, U.S.A.). Lyophilized crude peptides were purified by preparative reverse-phase high-pressure liquid chromatography (HPLC) on a C-18 column and were eluted with a linear gradient of acetonitrile (0–60%) containing 0.1% trifluoroacetic acid in water. Peptide composition was verified by amino acid analysis before use. Peptide purity was >95% based on analytical HPLC. A scrambled version (SC-V) of the FN-C/H-V peptide, (Arg-Pro-Gln-Ile-Pro-Trp-Ala-Arg-Tyr), was also synthesized. RGD peptide interacts with α3β1, α5β1, and β5 integrins (Hemler, 1990), CS-1 peptide interacts with α4β1 integrin and VCAM, and FN-C/H-V has been reported to interact with proteoglycan and to inhibit adhesion to activated endothelial cells (Woods et al., 1993; Wahl et al., 1994).
Animals in Groups II, III, IV, and V received four i.v. administrations of 150 μl of saline containing individual peptide (5 mg/kg): 30 min prior to ischemia, at the time of reperfusion, and 3 and 24 h after reperfusion.
Measurement of infarct size
Four ischemic animals in each group were killed 48 h after reperfusion. Infarct size was measured as reported previously (Yanaka, et al., 1996). Briefly, brains were removed and kept at −80°C for 5 min. Each brain was then cut into 2-mm thick coronal blocks, for a total of seven blocks per brain. Brain slices were incubated at 37°C for 30 min in 2% 2,3,5-triphenyl-2H-tetrazolium chloride (TTC). The regions unstained by TTC, which reflect mitochondrial damage, were quantified as the infarct areas. A tight correlation between histopathology and staining results within 3 days after the onset of ischemia in rat brain has been reported (Clark et al., 1993). The surface of each slice was digitized and the total and infarcted volumes were calculated blindly using three-dimensional reconstruction software (Jandel PC3D, Corte Madera, CA, U.S.A.).
Myeloperoxidase activity assay
The activity of myeloperoxidase (MPO), an enzyme localized in the azurophilic granules of leukocytes, is thought to be a quantitative index of polymorphonuclear leukocyte (PMN) presence (Barone et al., 1991). For the biochemical determination of MPO activity, three ischemic animals in each group were anesthetized and perfused transcardially with 150 ml of physiological saline (25°C at a pressure of 100 mm Hg) at 48 h after reperfusion. Brain samples of the ischemic and contralateral hemispheres were taken from the MCA area, immediately frozen in powdered dry ice, and stored at −80°C for later biochemical analysis. The method used to quantitate MPO activity from rat brain samples was similar to that recently described by Barone et al. (1991), but with minor modifications. The modified MPO assay for brain tissue was conducted blindly as follows: brain samples were thawed on ice, the cortical tissues were dissected from the subcortical portion, and wet weight (in grams) of the cortical tissue measured. Each sample was homogenized (1:20, wt/vol) in 5 mmol/L potassium phosphate buffer (pH 6.0, 4°C) using an Overhead-Stirrer (Wheaton Instruments, Millvill, NJ, U.S.A.) and centrifuged at 30,000 g (30 min, 4°C) (JS-H2 Centrifuge, Beckman Instruments, Palo Alto, CA, U.S.A.). The supernatant was discarded and the pellet was washed again as described above. After decanting the supernatant, the pellet was extracted by suspension in 0.5% hexadecyltrimethylammonium bromide (HTAB) (Sigma Chemical Co., St. Louis, MO, U.S.A.) in 50 mmol/L potassium phosphate buffer (pH 6.0, 4°C) for 2 min at an original tissue wet weight/volume ratio of 1:10. Samples were immediately frozen on powdered dry ice. Three freeze and thaw cycles were performed with sonications (10 s each, 25°C), using a CU-6 ultrasonicator (Branson, Shelton, CT, U.S.A.) between cycles. After the last sonication, the samples were incubated at 4°C for 20 min and centrifuged at 12,500 g (15 min, 4°C). Supernatant MPO activity was assayed as described earlier (Bradley et al., 1982). Briefly, 0.1 ml of supernatant was mixed with 2.9 ml of 50 mmol/L potassium phosphate buffer, pH 6.0, containing 0.167 mg of O-dianisidine dihydrochloride (Sigma) and 0.0005% hydrogen peroxide (Sigma). The change in absorbance at 460 nm was measured at 15-s intervals over 2 min, using a DU-64 spectrophotometer (Beckman). One unit of MPO activity is defined as that which degrades 1 μmol of peroxide/min at 25°C.
Statistics
All values are expressed as mean ± SD. A one-way analysis of variance (ANOVA) was performed on data of MPO activity, size of infarction, and peripheral leukocyte counts. A two-way ANOVA with Tukey multiple comparisons was carried out to compare neurological outcomes. Differences were considered significant if p < 0.05.
RESULTS
MPO activity
Mean MPO activity (U/g wet tissue) in the ischemic cortex was as follows (Fig. 1): Group I, 0.20 ± 0.05; Group II, 0.22 ± 0.05; Group III, 0.09 ± 0.01; Group IV, 0.10 ± 0.06; and Group V, 0.21 ± 0.04. Groups III and IV showed a significant decrease in MPO activity in the lesion when compared to Groups I, II, and V (p < 0.05). Mean MPO activity in the contralateral cortex was as follows: Group I, 0.03 ± 0.02; Group II, 0.04 ± 0.01; Group III, 0.05 ± 0.01; Group IV, 0.06 ± 0.03; and Group V, 0.04 ± 0.02.
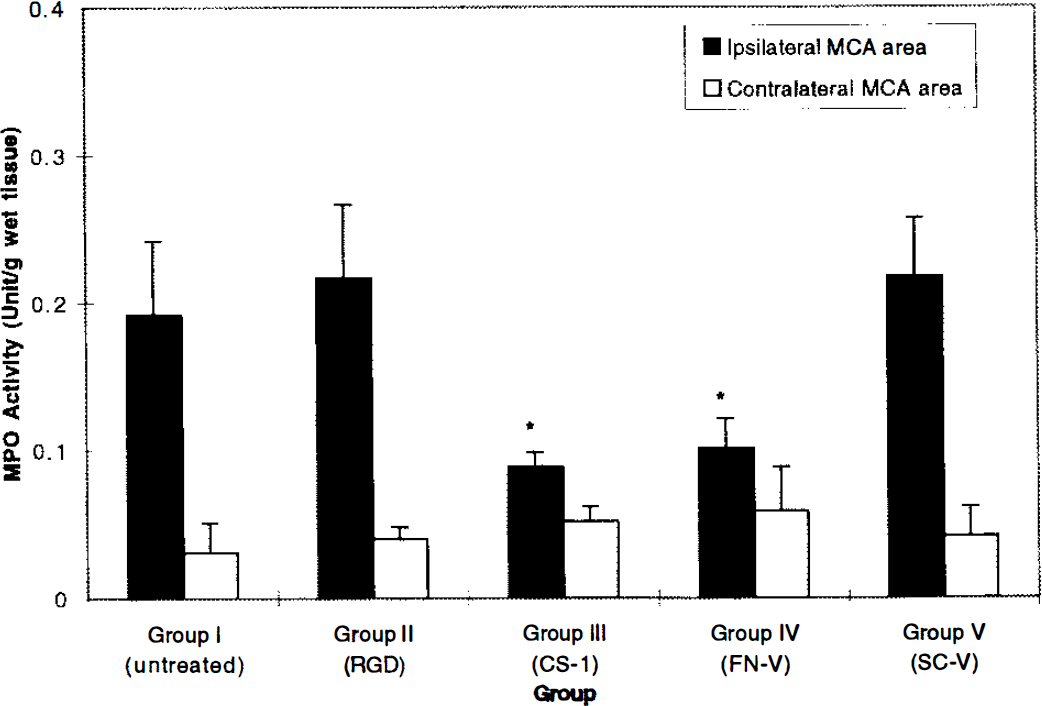
Graph showing MPO activity. Vertical bars indicate SD. MPO activity in the ischemic cortical tissue of Groups III and IV show a significant decrease when compared to that of Groups I, II, and V (p < 0.05) (*).
Infarct volume
Mean infarct size, expressed as a percentage of the total hemispheric volume ± SD, was as follows (Fig. 2): Group I, 46.08 ± 4.74%; Group II, 50.31 ± 7.76%; Group III, 32.61 ± 4.96%; Group IV, 33.78 ± 5.63%; and Group V, 45.03 ± 8.96%. Groups III and IV showed a significant decrease in the size of infarction when compared to Groups I, II, and V (p < 0.05).
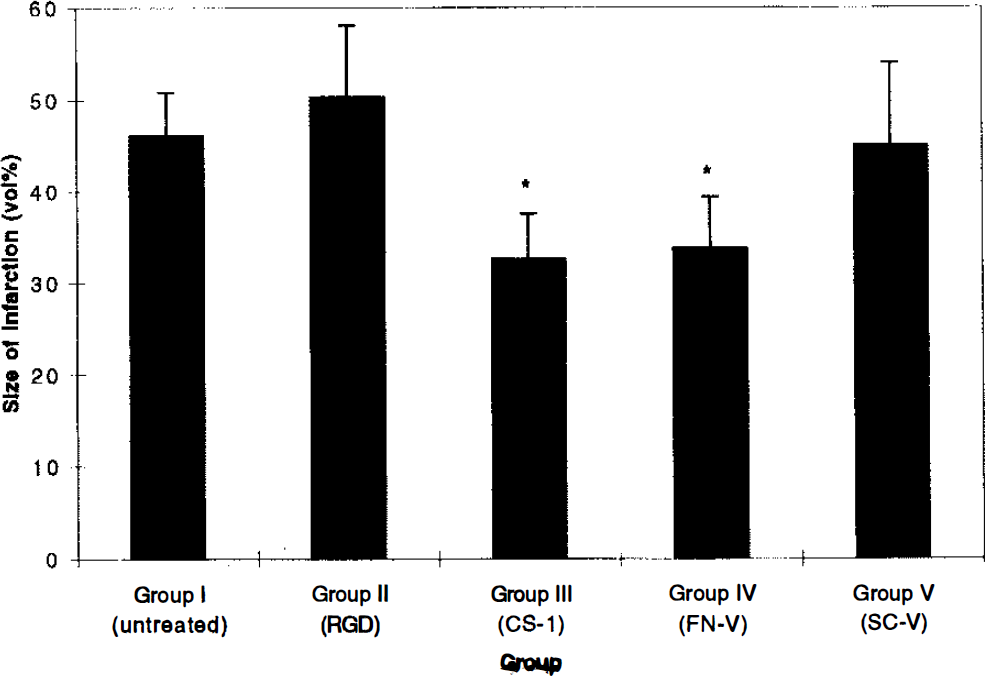
Graph showing mean infarct volume expressed as a percentage of the total hemisphere for each study group. Vertical bars indicate SD. Groups III and IV show a significant decrease in the size of infarction when compared to that of Groups I, II, and V (p 0.05) (*).
Neurological assessment
Neurological grades of the animals are summarized in Fig. 3. The neurological grades of Groups III and IV were significantly better than those of Groups I, II, and V (p < 0.01) at 48 h after reperfusion (Fig. 3).
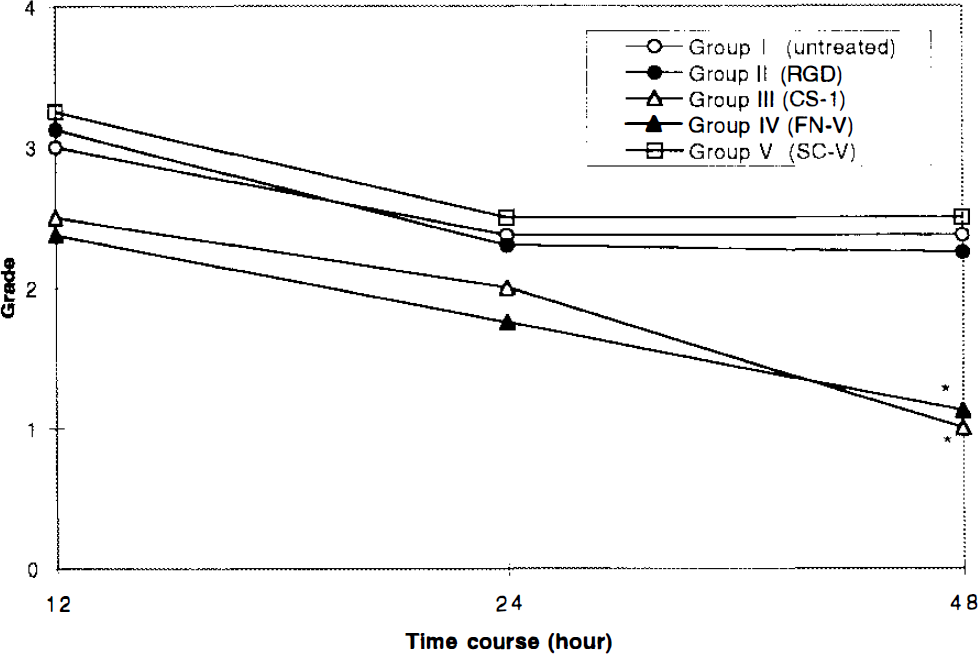
Graph showing the clinical outcome of the animals in each group. See text for definition of grades. Neurological grades of Groups III and IV are significantly better than those of Groups I, II, and V (p < 0.01) (*).
Peripheral leukocyte counting
No significant differences were detected in the number of leukocytes and differentials after peptide administration. Peptide treatment groups showed a tendency towards decreases in the number of leukocytes after administration of peptides, but these decreases were not statistically significant and were within normal physiological range (Table 1).
Peripheral white blood cell and differential counts
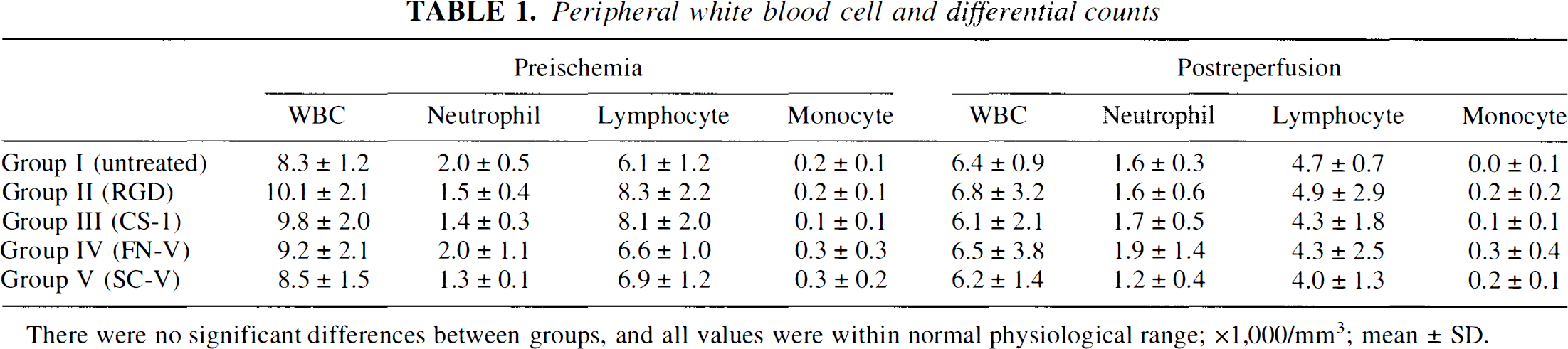
There were no significant differences between groups, and all values were within normal physiological range; ×1,000/mm3; mean ± SD.
DISCUSSION
The current study demonstrates that i.v. administration of certain bioactive adhesive fibronectin peptides significantly inhibits the infiltration of leukocytes into ischemic tissue, reduces the size of infarction, and reduces neurological dysfunction after transient focal cerebral ischemia in rats. Treatment with α4β1 integrin-binding CS-1 peptide or adhesion-promoting, heparin-binding synthetic peptide, referred to as FN-C/H-V, resulted in attenuation of the inflammatory response. However, the scrambled version of FN-C/H-V and α5β1 integrin-binding RGD peptide had no significant effect.
Leukocyte adhesion has been shown to occur via highly specific receptor/ligand-mediated interactions with endothelium and the extracellular matrix. PMNs constitutively express adhesion molecules on their plasma membranes and can rapidly alter their functional state in response to specific stimuli. Much emphasis has been placed on β2 integrin (Mac-1, LFA-1)-intercellular adhesion molecule (ICAM)-1 interactions in mediating leukocyte recruitment to sites of cerebral infarction, and on the potential value of blocking this interaction in the control of PMN and monocyte emigration (Clark et al., 1991; Bowes et al., 1993; Chen et al., 1994). The β1 integrins exhibit considerable specificity in their ability to bind extracellular matrix molecules (Hemler, 1990). Because of their low surface density, β1 integrins were believed to be absent from PMNs, and the function of PMN β1 integrin adherence-related events is largely unknown (Hemler, 1990).
At first glance, the reduction of PMN accumulation by α4β1 integrin-binding CS-1 peptide seems inconsistent, as PMNs are devoid of cell surface α4β1 integrins. PMNs are detected as early as 30 min and reach a peak at 24 h after ischemia in rat brain (Garcia et al., 1994). In contrast, monocytes are first detected in ischemic tissue 4–6 h after cerebral ischemia in rats (Garcia et al., 1994). Recent investigations have revealed a β2 integrin-independent and a β1 integrin-dependent mechanism in PMN accumulation (Hakkert et al., 1991; Winn et al., 1993). β2 integrin-independent PMN migration into inflammatory tissue after recruitment of monocytes has been observed (Mileski et al., 1990), and PMN migration has been thought to be initiated, in part, by a factor released by monocytes (Mileski et al., 1990; Doerschuk et al., 1990; Winn et al., 1993). Monocytes express β1 integrins, including VLA-4, on their surface (Hemler, 1990). There is evidence that synthetic CS-1 peptide can inhibit monocyte adhesion (Wahl et al., 1994; Hines et al., 1994). Therefore, synthetic CS-1 peptides may prevent PMN accumulation by blocking monocyte function.
Although the role of synthetic FN-C/H-V in mediating adhesion of leukocytes is incompletely understood, this peptide has been shown previously to inhibit adhesion of acitvated monocytes to cytokine-activated endothelial cells (Wahl et al., 1994). This suggests that its ability to inhibit adhesion to endothelial cells is through a mechanism similar to that of the CS-1 peptide. Additionally, this peptide adheres to cell surface proteoglycans and modifies leukocyte adhesion (Woods et al., 1993). If proteoglycans are involved in peptide FN-C/H-V action, then this peptide might antagonize selectins, since proteoglycan interactions with selectins have been shown to facilitate leukocyte rolling (Norgard-Sumnichit et al., 1993). Moreover, proteoglycans have recently been implicated in the function of β2 integrins (Diamond et al., 1995). Understanding the mechanism of FN-C/H-V action could yield new information regarding the molecular basis of leukocyte/endothelial interactions.
The synthetic RGD peptide, which interacts with α5β1 integrin, did not reduce leukocyte accumulation in the ischemic tissue. Synthetic RGD peptide may become sequestered or metabolized more rapidly than other peptides, thus accounting for the negative results. However, in contrast to synthetic CS-1 and FN-C/H-V peptides, the RGD peptide does not inhibit adhesion to endothelial cells (Osborne, 1990; Bevilacqua and Nelson, 1993). VCAM-1, on the surface of cytokine-activated endothelials cells, supports the adhesion of monocytes through an interaction with α4β1 integrin (Butcher, 1991; Osborne, 1990). It is possible that the α4β1 integrin-dependent pathway may be more important than the α5β1 integrin-dependent pathway in leukocyte accumulation in ischemic tissue.
Dose-response effect and optimal timing of administration of synthetic fibronectin peptides for brain protection will be discussed in a subsequent article. We have not revealed the multiple interactions of leukocytes with other adhesion receptors whose blockade reduces infarct size. We hypothesized that monocytes would play an important role in PMN accumulation, but these hypotheses have not yet been confirmed. Further experiments are required to resolve these issues, which are presently being addressed in our laboratory. However, the present studies clearly demonstrated the effectiveness of synthetic fibronectin peptides in brain protection following ischemic insult. Thus, although the role of integrins and other cell adhesion molecules in mediating adhesion of cells is complex and incompletely understood, emerging evidence suggests that agents that inhibit or modulate the adhesion process may provide a mechanism by which to regulate inflammation and ischemia/reperfusion injury. Understanding the functional properties of the protective peptides will offer insight into the mechanisms by which the active sequences provide an effective barrier to the pathological events associated with continued recruitment and activation of leukocytes in infarcted/reperfused brain.
Footnotes
Abbreviations used
Acknowledgment:
The authors wish to thank Bina Vachhani (Department of Laboratory Medicine and Pathology, University of Minnesota) for preparing the synthetic fibronectin peptides. The authors also wish to thank Jonathon Coltz (Graduate Program in Neuroscience, University of Minnesota) for helpful advice in statistics. This work was supported, in part, by a Grant-In-Aid from the American Heart Association (K.Y.) and Clinical Investigator Development Award 1K08-NS 01745-01 from the National Institute for Neurological Disorders and Stroke (P.J.C.). L.T. Furcht is a recipient of the Allan-Pardee professorship for cancer biology.
