Abstract
Using positron emission tomography (PET) and microdialysis, the present study showed that neuronal damages after transient focal ischemia was partly induced by hyperactivation of the cyclic adenosine 3′,5′-monophosphate (cAMP) second messenger system through modulations of dopamine D1 and serotonin 5-HT1A receptors in the living brains of cynomolgus monkeys. Occlusion of the right middle cerebral artery for 3 hours suppressed CBF in the striatum, and reperfusion induced hyperperfusion in the neocortex and striatum of the occluded side. Six hours after reperfusion, the activity of the cAMP second messenger system assayed with [11C]rolipram was significantly facilitated in the neocortex and striatum where CBF was lowered more than 40% of normal during occlusion (“ischemic” area). Seven days later, impaired dopamine D1 and 5-HT1A receptor binding, measured with [11C]SCH23390 and [carbonyl-11C]WAY-100635, respectively, was observed in the ischemic area. Microdialysis analysis revealed that the striatal dopamine level provided a transient and marked increased during occlusion and after reperfusion, whereas the cortical serotonin level transiently increased only after reperfusion, and was at an undetectable level thereafter. Administration of rolipram (0.1 and 1 mg/kg, intravenously) during occlusion facilitated reduction of dopamine D1 binding, whereas rolipram administration 6 hours after reperfusion induced a further decrease in 5-HT1A receptor binding. These results suggest that the activation of cAMP second messenger system modulated by dopamine D1 and 5-HT1A receptors could be involved in the neuronal degeneration after transient cerebral ischemic insult.
Neuronal damage after transient focal ischemic insult can be detectable not only by changes in CBF, CMRo2, and CMRglc, but also by changes in neurotransmitter systems in the CNS (Araki et al., 1996, 1997; Globus et al., 1988; Kanai et al., 1993; Kruger et al., 1999; Nagasawa et al., 1992a; Nakata et al., 1997; Prehn et al., 1991, 1993; Semkova et al., 1998; Sette et al., 1993; Shibata et al., 1992; Slivka et al., 1988; Vera et al., 1996; Zhao et al., 2001). Neurons are anatomically and functionally integrated as a network directly (Hattori et al., 1976) or indirectly (Bunney and Aghajanian, 1976) through multisynaptic connections. The binding of neurotransmitters to the specific receptors induces biological actions through the modulation of receptor-specific second messenger systems. Cyclic adenosine 3′,5′-monophosphate (cAMP), one of the second messenger molecules, plays an important role in neuronal signal transmission and transduction pathways in the CNS (Greengard, 1976). The receptors of dopamine D1 (Harada et al., 2002; Kebabian and Calne, 1979; Seeman, 1980; Tsukada et al., 2001a) and serotonin 5-HT1A receptors (Dumuis et al., 1988; Schoeffter et al., 1997; Tsukada, 2002) have been reported to couple with the cAMP second messenger system. Extracellular stimuli with binding of neurotransmitters to their specific receptors elevate the level of cAMP by stimulating adenylate cyclase to convert ATP to cAMP, and the signal transduction is terminated by degradation of cAMP to 5′-AMP by phosphodiesterases (PDEs) (Beavo, 1995). Rolipram is a selective PDE-IV inhibitor that enhances the availability of cAMP in the brain by inhibition of hydrolysis of cAMP to 5′-AMP (Wachtel, 1982). PET has been applied to evaluate several kinds of neuronal receptor binding and their functional responses to physiologic and pharmacologic manipulation in the living brain. Recently, rolipram was labeled with carbon-11 (11C) (DaSilva et al., 1997; Lourenco et al., 1999) and evaluated in vivo as a labeled compound for imaging and quantification of PDE-IV with PET (Gee et al., 1998; Harada et al., 2002; Tsukada et al., 2001a, 2002).
To assess whether neuronal damages after transient focal ischemia is induced by hyperactivation of cAMP second messenger system through modulations of dopamine D1 and serotonin 5-HT1A receptors, animal PET studies were conducted using transient middle cerebral artery (MCA) occlusion model in the cynomolgus monkey (Macaca fascicularis) (Takamatsu et al., 2000, 2001). In addition to PET studies, the effects of transit focal ischemia on the dopamine and serotonin levels in the brain were evaluated with microdialysis measurements.
MATERIALS AND METHODS
Animals and drugs
Six male cynomolgus monkeys (Macaca fascicularis) with body weights ranging from 4.5 to 5.8 kg were used for the PET measurements. Monkeys were maintained and handled in accordance with the recommendations of the US National Institutes of Health and also the guidelines of the Central Research Laboratory, Hamamatsu Photonics (Hamamatsu, Japan). Magnetic resonance images of monkeys were obtained with a MRT-50A/II (0.5 T) (Toshiba Medical Corporation, Tokyo, Japan) under pentobarbital anesthesia.
Isoflurane and pancuronium were purchased from Dainippon Pharmaceutical (Osaka, Japan), and Sankyo Co. Ltd. (Tokyo, Japan), respectively. WAY-100634, the precursor for [carbonyl-11C]WAY-100635, was a kind gift from Professor Christer Halldin (Karolinska Institute). Precursors of [11C]SCH23390, R–SCH23390, WAY-100635, and R/S-rolipram were purchased from RBI (Natick, MA, U.S.A.). Precursors of R- and S- [11C]rolipram were synthesized by ABX (Dresden, Germany).
Middle cerebral artery occlusion and reperfusion
The monkeys were tracheostomized, immobilized with 0.05-mg/kg pancuronium bromide (intramuscularly) every 2 hours, and artificially ventilated. Anesthesia was continued with 0.8% isoflurane in a N2O:O2:N2 gas mixture (1:1:1) during the entire experiment. After transmission and PET scans as a control, the right MCA was occluded with two microvascular clips via a transorbital approach (Hudgins and Garcia, 1970; Takamatsu et al., 2000, 2001). Upon completion of the occlusion procedure, PET scans for the occlusion condition were performed. The clips were removed 3 hours after the start of occlusion, and PET scans for the reperfusion conditions were performed 6 hours after the removal of the clips. Monkeys were allowed to awaken after the scans, and 7 days later they were anesthetized again as described earlier. During the experiments, each animal's body temperature was maintained within normal limits with heating blankets.
Synthesis of [11C]-labeled ligands
[11C]SCH23390 was labeled with 11C by N-methylation of its nor-compound with [11C]methyl iodide (Halldin et al., 1986). [Carbonyl-11C]WAY-100635 was labeled by N-acylayion of WAY-100634 with cyclohexane[carbonyl-11C]chloride (Pike et al., 1996). R-[11C]Rolipram or S-[11C]rolipram was labeled by methylation of the respective nor-compound with [11C]methyl iodide (DaSilva et al., 1997; Lourenco et al., 1999). The radioactive purity of each labeled compound used in this study was greater than 99%. After analysis for identification, the solution was passed through a 0.22-μm (pore size) filter before it was administered intravenously to the monkeys.
Positron emission tomography scan
Data were collected on a high-resolution PET scanner (SHR-7700, Hamamatsu Photonics; Watanabe et al., 1997). After an overnight fast, the animal's head was fixed in a head holder with stereotactic coordinates aligned parallel to the OM line. At day 1 before MCA occlusion, the first set of PET scans with [11C]SCH23390 for 34 minutes (Tsukada et al., 2001a), [carbonyl-11C]WAY-100635, R- and S-[11C]rolipram for 91 minutes (Tsukada et al., 2001a,b), and [15O]gases was performed. The second set with [15O]gases was performed 2 hours after the start of occlusion. The third set of scans was performed with R-and S-[11C]rolipram and [15O]gases 6 hours after the reperfusion. At day 7, the fourth set of PET scans with R- and S-[11C]rolipram, [11C]SCH23390, [carbonyl-11C]WAY-100635, and [15O]gases was performed.
Assessment of CBF, CMRo2, oxygen extraction fraction (OEF), and cerebral blood volume was performed using the steady-state [15O] inhalation method (Frackowiak et al., 1980; Pappata et al., 1993; Sette et al., 1989; Takamatsu et al., 2000, 2001; Touzani et al., 1995; Young et al., 1996), with the successive inhalation of trace amounts of [15O]CO2, [15O]O2, and [15O]CO. The mean values of radioactivity of whole blood and plasma were used for parametric image generation (Sette et al., 1989).
To confirm the involvement of activated cAMP second messenger system on the neuronal damages induced by the ischemic/reperfusion insult, saline or rolipram at doses of 0.1 and 1 mg/kg was intravenously administered 5 minutes after the start of occlusion or 6 hours after reperfusion.
Positron emission tomography data analysis
Regions of interest were identified according to magnetic resonance images of each monkey brain, and the time–activity curves of [11C]SCH23390, [carbonyl-11C]WAY-100635, and R- and S-[11C]rolipram in these regions of interest were obtained.
Specific binding of the ligand to PDE-IV was estimated as the difference of radioactivity of R-[11C]rolipram versus that of S-[11C]rolipram measured from 41 to 91 minutes after injection in each region of all animals. After normalization of regional radioactivity uptake to 1 GBq of injected dose and 5 kg of body weight, areas under the curves (AUC) of R-[11C]rolipram (AUC(R)) and S-[11C]rolipram (AUC(S)) were calculated in each region of the same animal, and specific binding was determined by subtraction of AUC(R) from AUC(S) (Harada et al., 2002; Tsukada et al., 2001a, 2002).
For the quantitation of dopamine D1 and serotonin 5-HT1A receptor binding in vivo, a kinetic two-compartment analysis method was applied as previously described (Huang et al., 1986; Parsey et al., 2000; Tsukada et al., 2001a,b). The cerebellum was used as a reference region because of the low abundance of dopamine D1 and 5-HT1A receptors in this region. The total radioactivity in the cerebellum was used as an estimate of the free and nonspecific radioligands and was subtracted from radioligand binding in the striatal region to determine specific binding. A two-compartment model was fitted to the time activity curve of specific binding in the striatal region. The binding potential of [11C]SCH23390 for dopamine D1 or [carbonyl-11C]WAY-100635 for 5-HT1A was calculated by determining the ratio of the estimated k3 value (association rate) to the estimated k4 value (dissociation rate).
Microdialysis
Two guide cannula were implanted (A: 21, L: 3.0; A: 35, L: 10) according to the individual magnetic resonance images. Two microdialysis probes with a membrane region 250 μm in diameter and 3 mm in length (Eicom A-I-25–03, Eicom, Kyoto, Japan) were inserted into the striatal (17.0 mm below the dura matter) and cortical (3.0 mm below) regions via each guide cannula. The probes were perfused with Ringer solution (pH 6.4) at a rate of 5 μL/min, 75-μL samples were collected every 15 minutes, and the contents of dopamine and serotonin were measured by high-performance liquid chromatography with electrochemical detection (HTEC-500, Eicom). The averaged data obtained from 0 to 120 minutes were used as “baseline” data. Dopamine and serotonin levels in the extracellular fluid (ECF) were expressed as percent of baseline.
Neuropathology
Just after the completion of day 7 scans, monkeys were deeply anesthetized with sodium pentobarbital. The brain was fixed by transcardial perfusion with a 10% formalin neutral buffer solution, pH 7.4, followed by saline perfusion at 100 mm Hg. The brain was then removed, and 12 coronal sections at 2 mm intervals were made using a brain matrix (MBM-2000C, BRC, Japan). Each section was embedded in paraffin wax, and 10-μm-thick sections were cut and stained with hematoxylin and eosin. The neuronal damage in each section was defined (Osborne et al., 1987), and after correction for brain edema (Swanson et al., 1990) the area of neuronal damage was measured using a computerized image analysis system. The volume of neuronal damage was calculated from the areas of damages in the coronal sections and their anteroposterior coordinates.
Statistical analysis
Results are expressed as means ± SD. Comparisons between conditions were carried out using the paired, two-tailed Student's t-test. A probability level of less than 5% (P < 0.05) was considered to indicate statistical significance.
RESULTS
Figure 1A shows PET images of CBF, CMRo2 and OEF before occlusion (control), 2 hours after occlusion, 6 hours after reperfusion, and 7 days after reperfusion in the transient MCA occlusion model. The MCA occlusion decreased CBF and CMRo2 mainly in the striatum, which was the focal area supplied CBF by the MCA. Of interest, increased OEF was observed in lateral site of the striatum and the cortex. Six hours after reperfusion, postischemic hyperperfusion immediately after reperfusion disappeared; however, impaired CMRo2, partially recovered by reperfusion compared with the occlusion condition, was detected not only in the striatum, but also in the cortex (“reperfusion injury”). OEF was markedly reduced in the striatum. At 7 days after reperfusion, postischemic hypoperfusion was observed within an area of more than 40% CBF reduction during occlusion (defined as the “ischemic” area). A further expansion of impaired CMRo2 area was observed 7 days after reperfusion. cerebral blood volume was not significantly affected by occlusion or reperfusion throughout the experiments. Histologic examination revealed that the transient MCA occlusion caused necrotic brain damage 7 days after reperfusion in the cortex and striatum (Fig. 1B).
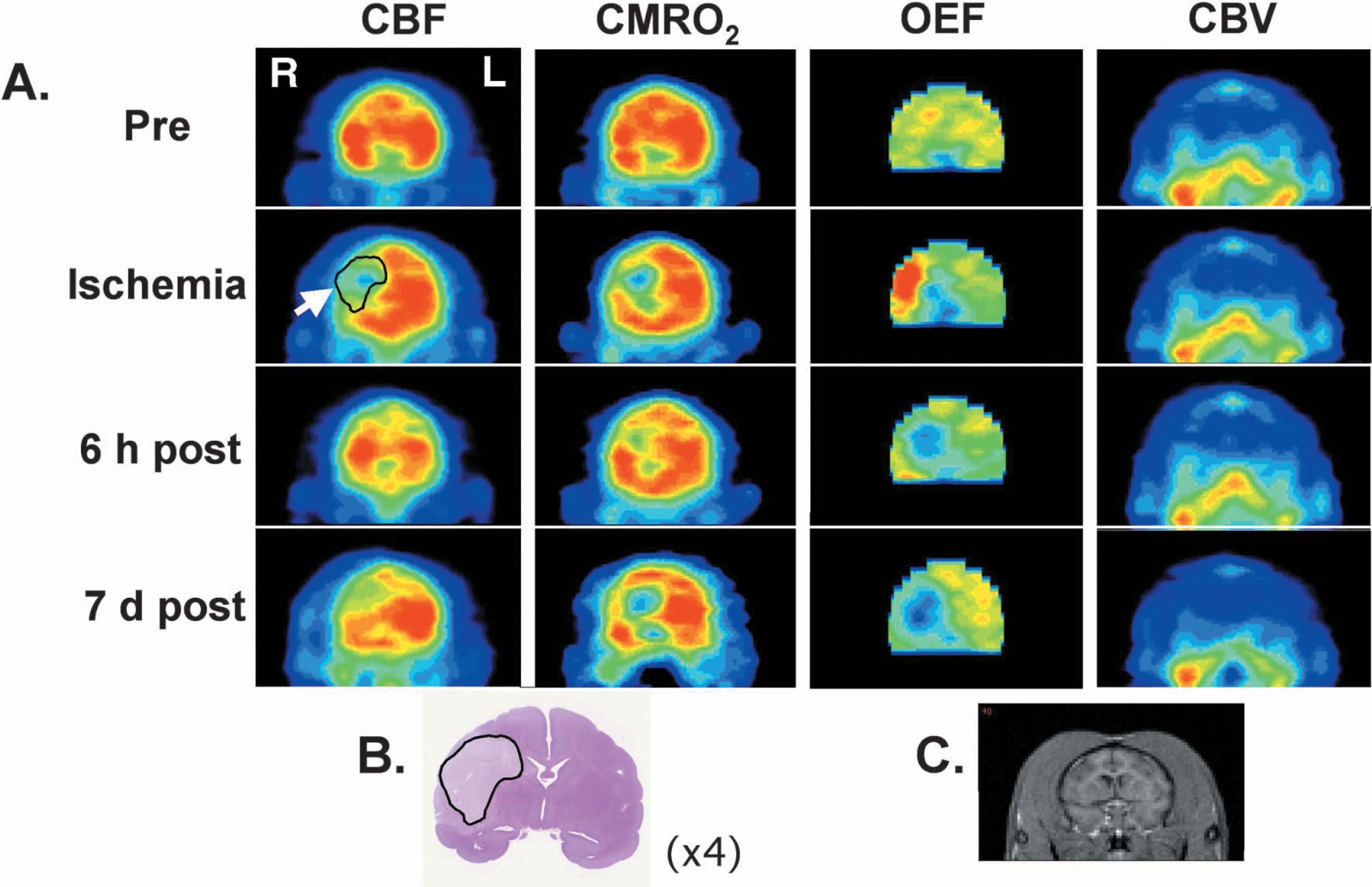
Typical sequential PET images of CBF, CMRo2, and OEF in the living brain (
Figure 2 shows typical coronal section of magnetic resonance and PET images of CBF, R-[11C]rolipram, [11C]SCH23390, and [carbonyl-11C]WAY-100635 in the brain before and during occlusion, and 6 hours and 7 days after reperfusion. Compared to the preocclusion (control) condition, occlusion and reperfusion induced the region-specific facilitation of R-[11C]rolipram uptake in the striatum, as well as in part of the cortex, 6 hours after reperfusion, whereas the uptake showed marked reduction 7 days after reperfusion, which was well correlated with the areas of more than 40% CBF reduction during occlusion (ischemic area) (Fig. 2). No significant changes in S-[11C]rolipram uptake were observed in any cases (data not shown). To determine quantitatively the effects of transient MCA occlusion on PDE-IV activity in the brain, the specific binding of [11C]rolipram was calculated as the difference in radioactivity of R-[11C]rolipram versus S-[11C]rolipram in each cortical region, which was assumed to be an index of enzymatic activity of PDE-IV. Specific binding of [11C]rolipram in the ischemic area were significantly increased 6 hours after reperfusion, followed by marked reduction 7 day after reperfusion in both the striatum (Fig. 3A) and the cortex (Fig. 3B).
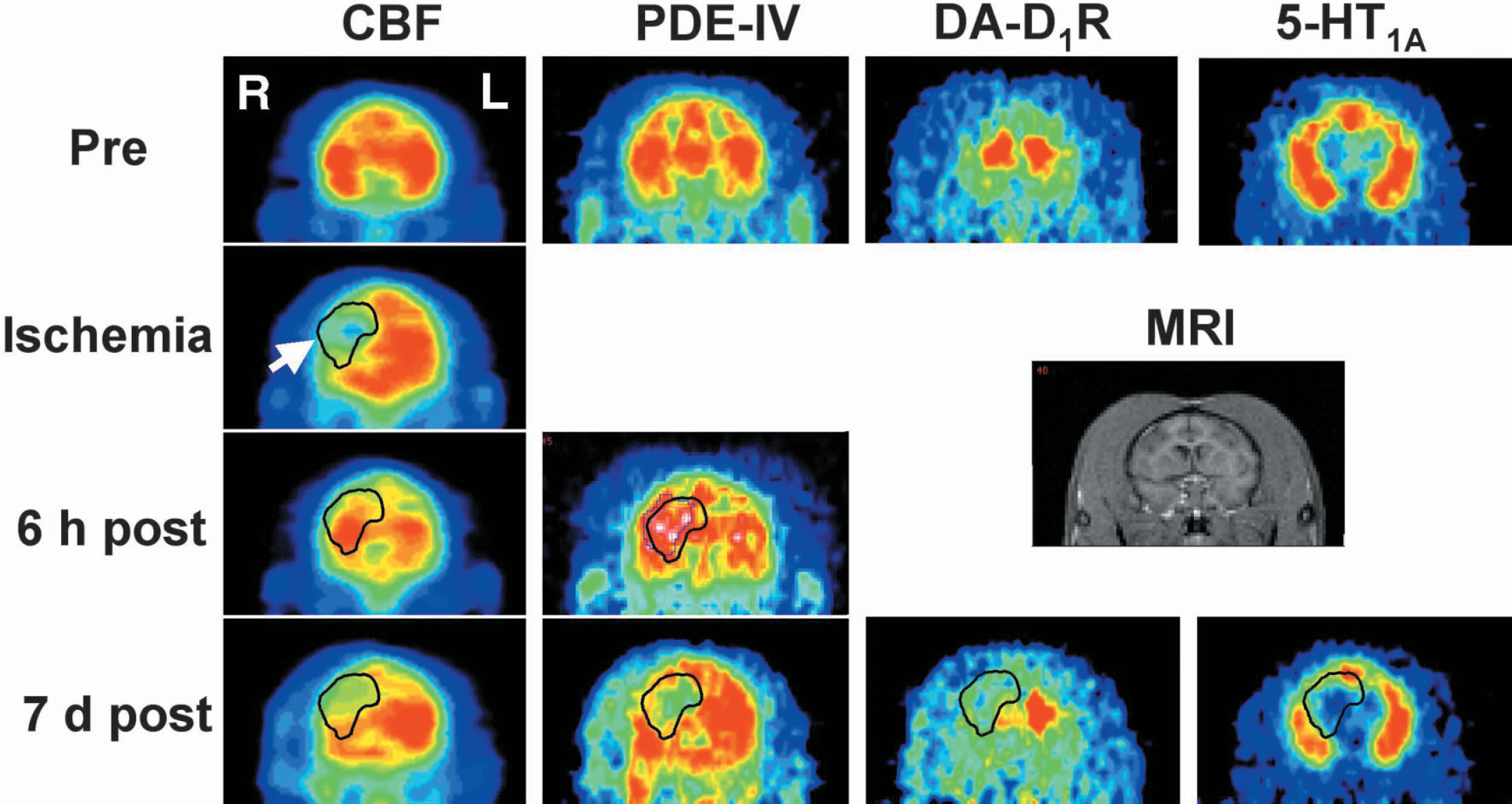
Typical magnetic resonance and PET images of CBF, R[11C]rolipram, [11C]SCH23390 and [carbonyl-11C]WAY-100635 in the living brain of monkey as shown in coronal sections. PET data were collected before MCA occlusion (Pre), 2 hours after MCA occlusion start (Ischemia, [15O]gas only), 6 hours after reperfusion (6 h post), ([15O]gas and [11C]rolipram only) and 7 days after reperfusion (7 d post). The region of interest indicated by the white arrow corresponds to an area of more than 40% CBF decrease during occlusion.
Effects of transient ischemia on PDE-IV activity (
Three-hour MCA occlusion followed by reperfusion also caused region-specific binding impairments of [11C]SCH23390 to dopamine D1 receptors and that of [carbonyl-11C]WAY-100635 to 5-HT1A 7 days after reperfusion (Fig. 2). Transient MCA occlusion induced significant reduction of [11C]SCH23390 binding to dopamine D1 receptors in vivo in the striatum (Figs. 2 and 3C), and also a marked decrease in [carbonyl-11C]WAY-100635 binding to 5-HT1A receptors in the neocortex (Figs. 2 and 3D). These areas of impaired receptor binding were well correlated with the ischemic area, an area showing more than 40% CBF decrease during occlusion 7 days after reperfusion (Fig. 2).
Middle cerebral artery occlusion and reperfusion affected dopamine and serotonin levels in the striatal and cortical regions in the monkey brain. Dopamine in the striatal ECF showed an immediate and transient increase after the start of occlusion, reaching a peak value of approximately 4,400% of the baseline level 1 hours later, followed by a decrease to approximately 800% of the baseline 3 hours after occlusion (Fig. 4A). Reperfusion of MCA after 3 hours occlusion induced an immediate and transient increase in dopamine up to approximately 4,000% of the baseline 2 hours after reperfusion, and then the increased dopamine gradually returned to the baseline level 5 hours after reperfusion, followed by hypoactivity of dopamine less than baseline (Fig. 4A). In contrast, MCA occlusion resulted in no significant changes in serotonin level in the ECF of the cortical region (Fig. 4B), where neuronal damage was observed after reperfusion later on. After MCA reperfusion, serotonin in cortical ECF showed a transient increase up to approximately 1,000% of the baseline 2 hours after reperfusion. The hyper levels of serotonin gradually returned to baseline level 4 hours after reperfusion, and serotonin levels were below the minimal detection level 10 hours after reperfusion and thereafter (Fig. 4B).
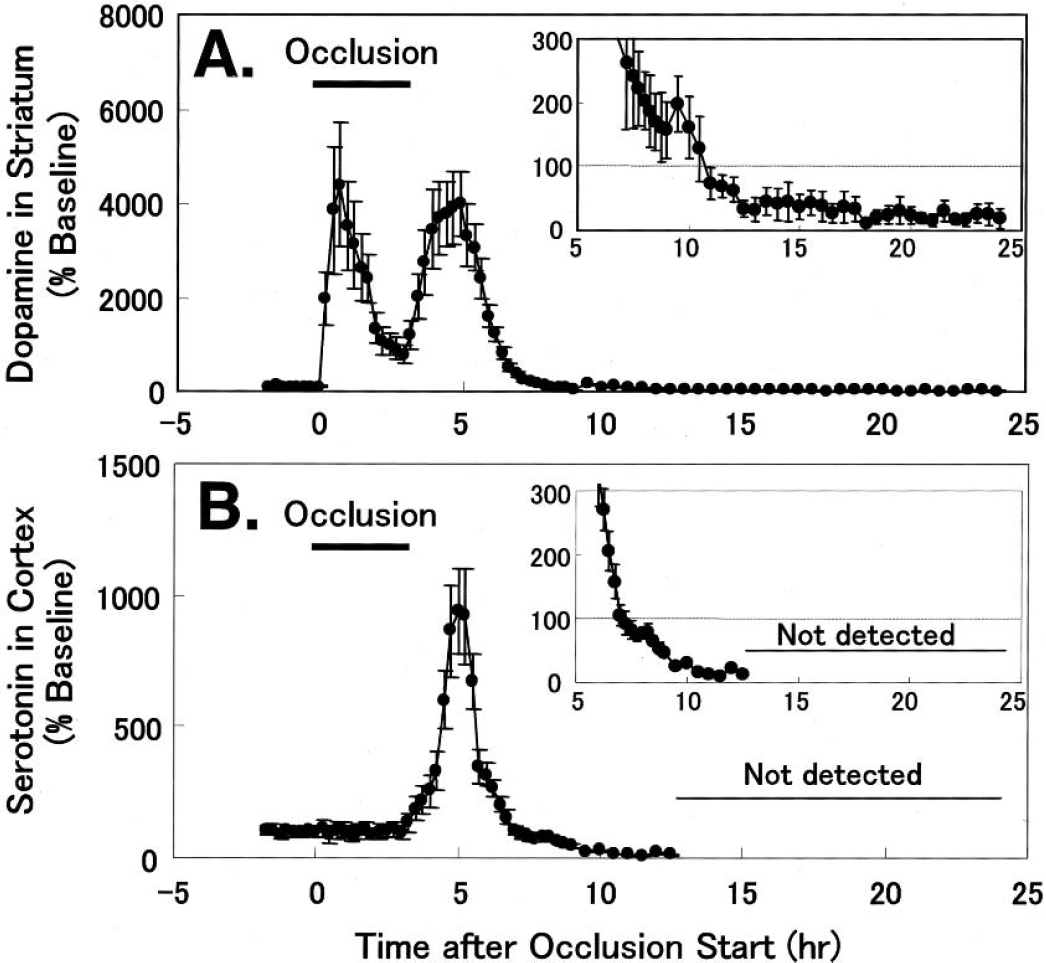
Effects of transient ischemic insult on dopamine (
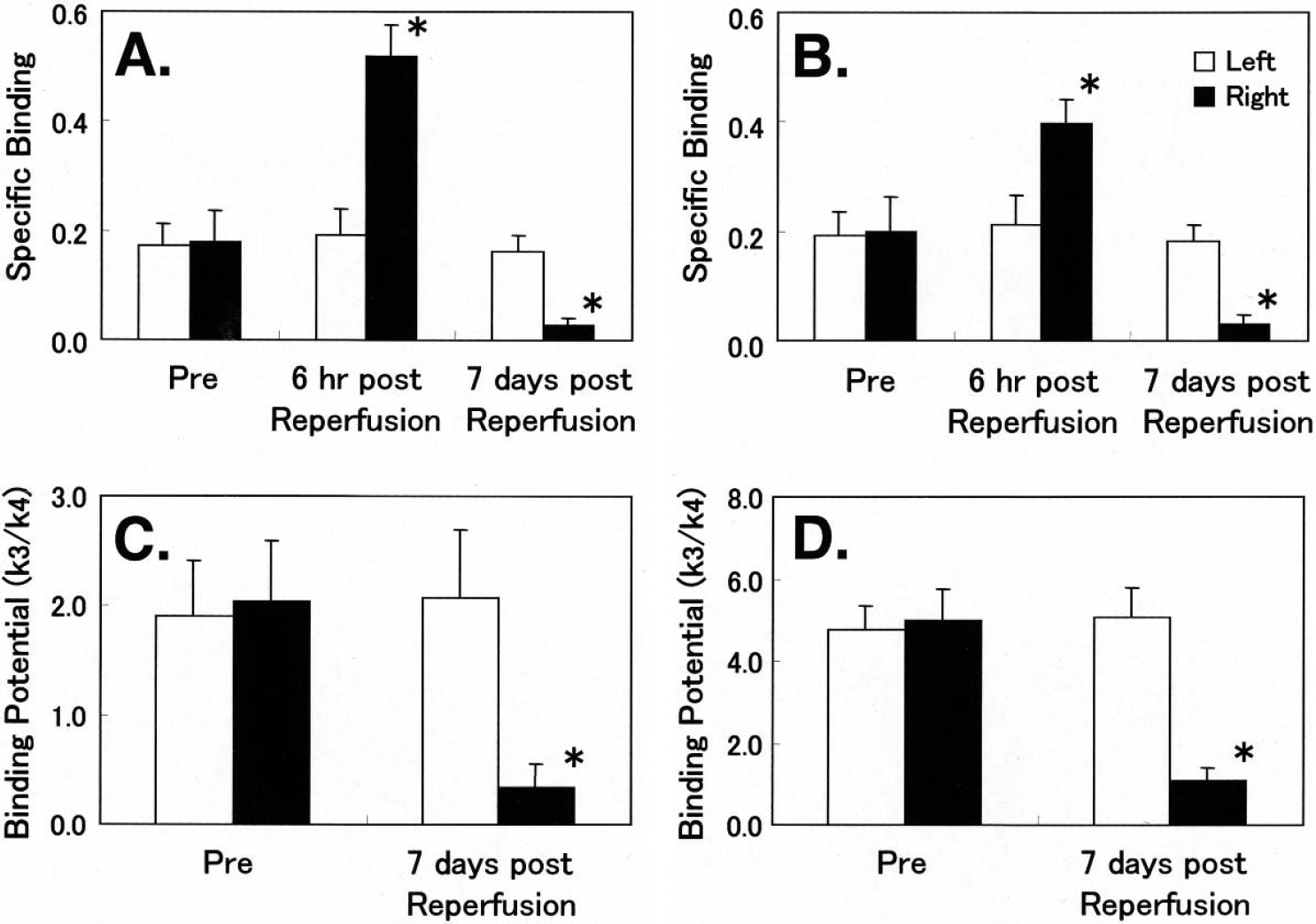
When rolipram at doses of 0.1 and 1 mg/kg was intraperitoneally administered 5 minutes after the start of occlusion, reduction of [11C]SCH23390 binding to dopamine D1 receptors in the striatum was further facilitated compared with the control condition (Fig. 5A), whereas [carbonyl-11C]WAY-100635 binding to 5-HT1A receptors in the cortex was not affected by rolipram (Fig. 5B). In contrast, rolipram (0.1 and 1 mg/kg) administered 6 hours after reperfusion further facilitated the reduction of cortical [carbonyl-11C]WAY-100635 binding (Fig. 5B), whereas no significant alterations of striatal [11C]SCH23390 binding was induced by the late rolipram administration (Fig. 5A). Histologic examinations of the brain slices indicated that rolipram, administered 5 minutes after occlusion start or 6 hours after reperfusion, further increased the extent of damaged volume in the striatum (Fig. 5C) and cortex (Fig. 5D).
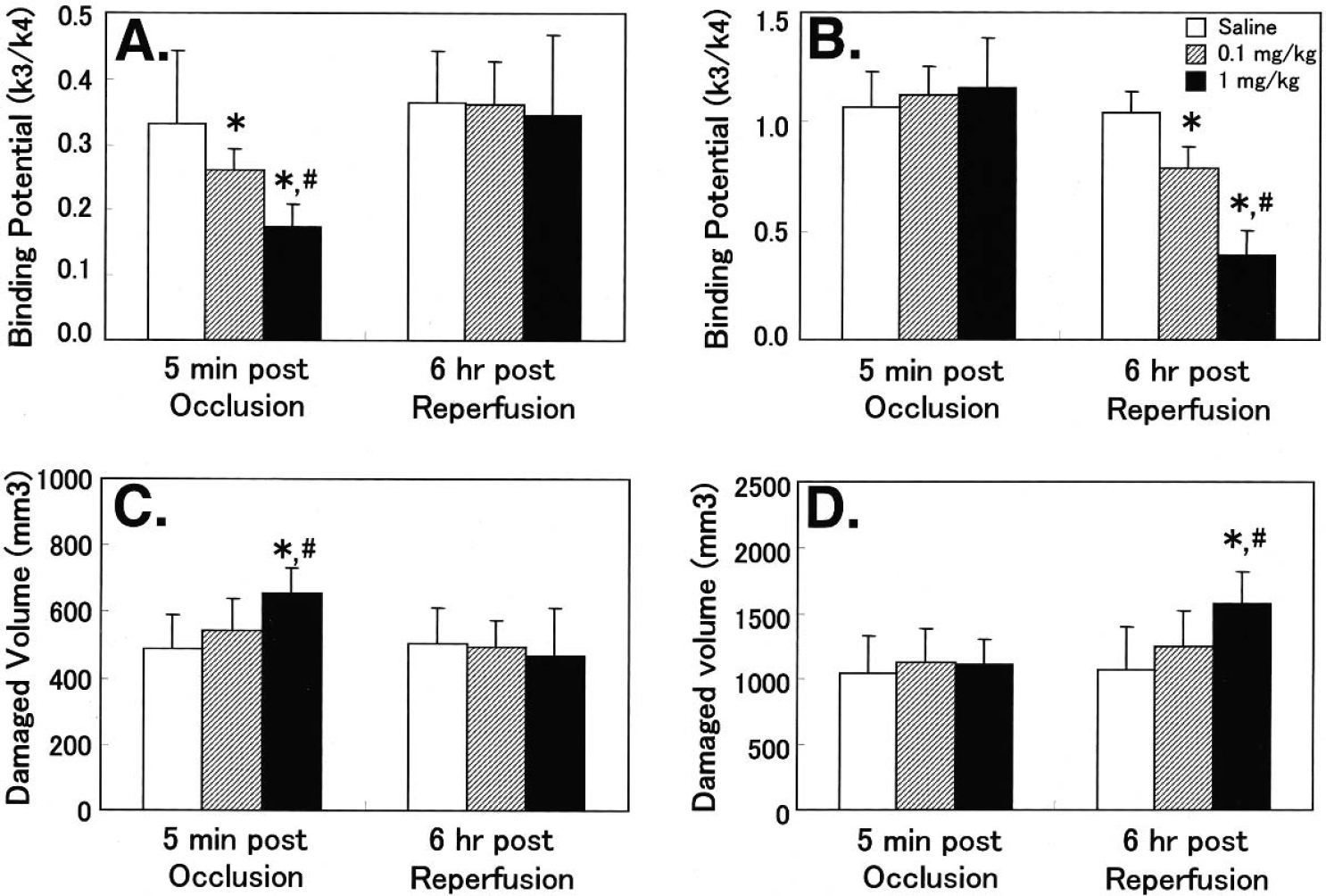
Effects of rolipram on dopamine D1 (
DISCUSSION
The present PET study showed decreased CBF and CMRo2 and increased OEF in the deep MCA territory of the striatum, but not in the neocortex, during MCA occlusion. The OEF increased in an area of mild ischemia (20% to 50% decrease in CBF), which was mainly observed in the neocortex. Previous results showed that immediately after reperfusion, postischemic hyperperfusion was observed in the neocortical area (Takamatsu et al., 2000, 2001). In these hyperperfused areas, decreased CMRo2 was observed thereafter, suggesting that reperfusion may in part contribute to neuronal damage of reperfusion injury. In addition to decreased CMRo2, a marked reduction in [11C]SCH23390 binding to dopamine D1 receptors was observed in deep MCA areas of the striatum, and a significant decrease in [carbonyl-11C]WAY-100635 binding to 5-HT1A receptors was detected in the neocortical area 7 days after reperfusion in the monkey brain. It should be noted that the “ischemic” area including both the striatal and neocortical areas showed a transient facilitation of PDE-IV activity estimated with R- and S-[11C]rolipram 6 hours after reperfusion, followed by a lower PDE-IV activity 7 days after reperfusion.
PDE-IV modulates cAMP signal transduction by controlling free cAMP concentrations in neuronal cells (Beavo, 1995; Reinhardt and Bondy, 1996). Facilitation of cAMP activity induced the increase in PDE-IV level (Manning et al., 1996). In contrast, suppression of cAMP activation by chronic receptor blocking or neuronal lesions provided reduced PDE-IV (Ye and O'Donnell, 1996; Ye et al., 1997). These observations allowed us to predict the possibility of monitoring functional changes in the cAMP-related second messenger system through the assessment of PDE-IV activity with the combination of R- and S-[11C]rolipram (Harada et al., 2002; Tsukada et al., 2001a, 2002). In the present study, PDE-IV activity, measured by R- and S-[11C]rolipram, showed transient facilitation 6 hours after reperfusion in the regions where marked impairments of CMRo2, dopamine D1 receptors (in the striatum) and 5-HT1A receptors (in the neocortex) were determined 7 days after the ischemic/reperfusion insult. During the ischemic/reperfusion process, CBF was chronologically changed throughout the experiment. However, it is unlikely that the changed CBF is related to the altered specific binding of the ligand because the flow dependency of ligands could be compensated by combined use of active and inactive enantiomers as labeled ligands (Kakiuchi et al.2001, Tsukada, 2002). It was previously reported that ischemia induced a significant increase in extracellular cAMP, as measured by a microdialysis assay, in the striatum of the rat brain during ischemia and 4 hours after reperfusion (Prado et al., 1992). Thereafter, PDE-IV activity, as measured by autoradiographic analysis in vitro with [3H]rolipram, was significantly reduced (Nagasawa et al., 1992b). Taken together, these results suggest that the ischemic/reperfusion insult resulted in the transient facilitation of the cAMP cascade with tentative increase in PDE-IV activity, followed by the marked reduction of the activity by resulting neuronal damages, and also that these alterations could be determined by R- and S-[11C]rolipram with PET in the living monkey brain.
A tight link has been reported between the dopamine D1 and 5-HT1A receptors and the cAMP second messenger system. Activation of D1 dopamine receptors increases the cAMP level by stimulating adenylate cyclase (Kebabian and Calne, 1979; Seeman 1980; Tsukada et al., 2001a), whereas activation of 5-HT1A receptors decreases cAMP level by suppression of adenylate cyclase (Dumuis et al., 1988; Schoeffter et al., 1997). Several lines of evidence suggest that a massive release of dopamine is induced in the striatum after cerebral ischemia (Globus et al., 1988; Slivka et al., 1988), and also that intrastriatal injection of dopamine causes a dose-dependent loss of neurons (Filloux and Townsend, 1993). In contrast, depletion of dopamine by inhibition of synthesis or release protected striatal neurons from ischemic damage, despite the excessive release of excitatory amino acid neurotransmitters, glutamate and aspartate, during and after transient ischemic insult (Globus et al., 1987; Phebus and Clements, 1989). The present microdialysis results demonstrate that dopamine levels in the striatal ECF showed a transient and marked increased during MCA occlusion and also just after reperfusion. Furthermore, administration of rolipram at doses of 0.1 and 1 mg/kg during the MCA occlusion induced a further decrease in D1 dopamine receptor binding in the striatum. Rolipram at these doses is pharmacologically active to facilitate cAMP level by PDE-IV inhibition. These present results suggest that the hyperactivation in dopaminergic neural transmission, especially through D1 receptors, plays a key role in the development of ischemic damages in the striatum (Araki et al., 1996; 1997; Kanai et al., 1993; Nagasawa et al., 1992a). In addition to damage due to an amount of oxidative radical forms of dopamine (e.g., 6-hydroxydopamine), the hyperactivation of the cAMP second messenger system might play an important role in the development of neuronal damage after transient ischemic insults at least in the striatum, resulting in degradation of neurons containing D1 dopamine receptors.
Although the results of effects of acute ischemic insult on 5-HT release are controversial, several reports evidenced decreased 5-HT neuronal transmission during and/or after ischemia (Calderini et al., 1978; Cvejic et al., 1990; Harrison et al., 1979; Ishimaru et al., 1993; Jellinger et al., 1978; Kozuka and Iwata, 1995; Luthman et al., 1992; Welch et al., 1977). In contrast, activated serotonergic neural transmission through 5-HT1A receptors has been reported to prevent cortical neurons from ischemic damage (Kruger et al., 1999; Nakata et al., 1997; Prehn et al., 1991, 1993; Semkova et al., 1998; Shibata et al., 1992), which is assumed to be largely mediated by the excitatory and depolarizing action of excitatory amino acid neurotransmitters, illustrating the interactive process between 5-HT1A receptor activity, excitatory amino acid neurotransmissions and the cAMP second messenger system (Basse-Tomusk and Rebec, 1986; Beck et al., 1985; Rowan and Anwyl, 1987). If ischemic/reperfusion insult impaired serotonergic neural transmission to the neocortex through 5-HT1A receptors, the neocortical regions might suffer from further excitatory amino acid action and also the activated cAMP second messenger system. With microdialysis measurements, the present study showed that serotonin levels in the ECF of the neocortical region was significantly increased just after reperfusion, whereas the level of serotonin release became lower than the control level 4 hours after reperfusion and thereafter, suggesting a hyperactivation of cAMP second messenger system in the neocortical region by the inactivation of 5-HT1A receptors. Furthermore, administration of rolipram (0.1 and 1 mg/kg) 6 hours after reperfusion provided further decreased 5-HT1A receptor binding in the neocortex. The hyperactivation of cAMP system induced by inhibition of PDE-IV with rolipram resulted in the dose-dependent degradation of neurons containing 5-HT1A receptors. Although the effects of ischemic-induced changes in the cAMP cascade on the neuronal damage remain to be elucidated (Block et al., 1997; Hara et al., 1990; Kato et al., 1995), the transsynaptic modulations of dopaminergic and serotonergic neuronal transmissions may participate in the ischemic process by amplifying the toxicity of excitatory amino acids, which may involve the activation of the cAMP cascade (Prado et al., 1992).
In conclusion, the present study showed that neuronal damages after transient focal ischemia was partly induced by hyperactivation of cAMP second messenger system through modulations of dopamine D1 and serotonin 5-HT1A receptors. These results suggest that monitoring of cAMP second messenger system using R- and S-[11C]rolipram could be a useful indicators for neuronal degeneration after transient ischemic insult in the living brain as measured by PET.
