Abstract
Antemortem demonstration of ischemia has proved elusive in head injury because regional CBF reductions may represent hypoperfusion appropriately coupled to hypometabolism. Fifteen patients underwent positron emission tomography within 24 hours of head injury to map cerebral blood flow (CBF), cerebral oxygen metabolism (CMRO2), and oxygen extraction fraction (OEF). We estimated the volume of ischemic brain (IBV) and used the standard deviation of the OEF distribution to estimate the efficiency of coupling between CBF and CMRO2. The IBV in patients was significantly higher than controls (67 ± 69 vs. 2 ± 3 mL; P < 0.01). The coexistence of relative ischemia and hyperemia in some patients implies mismatching of perfusion to oxygen use. Whereas the saturation of jugular bulb blood (SjO2) correlated with the IBV (r = 0.8, P < 0.01), SjO2 values of 50% were only achieved at an IBV of 170 ± 63 mL (mean ± 95% CI), which equates to 13 ± 5% of the brain. Increases in IBV correlated with a poor Glasgow Outcome Score 6 months after injury (ρ = −0.6, P < 0.05). These results suggest significant ischemia within the first day after head injury. The ischemic burden represented by this “traumatic penumbra” is poorly detected by bedside clinical monitors and has significant associations with outcome.
Acute head trauma is an important public health problem, with up to 57% of patients continuing to experience unfavorable outcomes after severe head injury (Clifton et al., 2001). Specific neuroprotective interventions have been disappointing (Maas et al., 1999), and management has generally concentrated on the prevention or attenuation of cerebral hypoxia (Chesnut, 1995). However, despite the common observation of ischemic neuropathologic changes in fatal head injury (Graham and Adams, 1971; Graham et al., 1989), efforts to demonstrate when, where, and why true ischemia occurs in the brain have been beset by problems.
Commonly quoted cerebral blood flow (CBF) and CMRO2 thresholds for neuronal survival based upon studies of experimental ischemia and clinical stroke (Heiss et al., 1992; Marchal et al., 1996; Powers et al., 1985) cannot be translated directly to head injury. Both depressed level of consciousness and concurrent sedation can reduce metabolic rate (and hence coupled perfusion) in the injured brain and reduce critical CBF thresholds for ischemia (Bergsneider et al., 2000). Conversely, epileptiform activity, or hypermetabolism associated with excitotoxicity may increase CMRO2 and make “normal” CBF levels inadequate. A clear definition of ischemia in situations where CMRO2 is altered depends upon the demonstration of compensatory decreases in the oxygen saturation of jugular bulb blood (SjO2) or increases in arteriojugular differences in oxygen content (AJDO2) and oxygen extraction fraction (OEF). These considerations are relevant to the interpretation of earlier studies, which showed modest CBF reductions in the first few days after head injury (Obrist et al., 1984). However, the absence of consistent increases in AJDO2 in these studies would seem to exclude significant ischemia. Whereas more than 30% of patients within 12 hours of head injury show global CBF reductions below accepted ischemic thresholds (Bouma et al., 1991), even at this stage, few patients demonstrate increases in AJDO2 above 9 mL/100 mL (a recognized threshold for defining ischemia) (The Brain Trauma Foundation, 2000). Heterogeneous CBF patterns in the injured brain [as demonstrated by Xenon enhanced computed tomography (Xe-CT)] (Bouma et al., 1992; McLaughlin and Marion, 1996) may result in high regional AJDO2 values being diluted by normal physiology in the majority of the brain. Clear identification of true ischemia would require the demonstration that CBF was inappropriately low in the context of CMRO2 in these regions, with a resulting increase in local OEF.
Measurement of regional CMRO2 and OEF traditionally requires imaging with oxygen-15 positron emission tomography (15O PET). However, two previous PET studies have found no evidence of cerebral ischemia, even after the physiologic stress of hyperventilation-induced reduction in CBF (Diringer et al., 2000, 2002). One study used PET to investigate global oxygen supply/demand relationships (Diringer et al., 2000) and found that OEF in patients with head injury was statistically significantly reduced when compared with control values. A more recent study (Diringer et al., 2002) reported no reductions in mean CMRO2 with hyperventilation, even in regions of interest (ROIs) where CBF was reduced to 10 mL 100g−1 min−1. However, these results are based upon data averaging across all ROIs and cannot exclude the possibility that critical ischemia may result in a significant subpopulation of ROIs, especially when the response to hyperventilation is heterogeneous. Furthermore, it would seem inappropriate to select ROIs for analysis based upon perfusion thresholds. Traumatized brain at potential risk of ischemic damage should be defined within a region with increased OEF and maintained or falling CMRO2.
These issues are of more than academic importance. A clear demonstration of the temporal and spatial extent and severity of cerebral ischemia is essential both for the rational management of head injury and to understand the limitations of current neuroprotective interventions. Furthermore, it is important to determine the extent to which common bedside monitors, such as jugular bulb oximetry, miss focal ischemia and whether reliance on such monitors may mislead and potentially result in harm. We have used 15O PET to investigate the incidence and mechanisms of ischemia after early head injury and compared the results with bedside monitoring of jugular bulb oximetry.
METHODS
Subjects
PET studies were undertaken on 10 healthy volunteers (8 men, 2 women) with a mean (range) age of 30 (18–60) years and 15 patients within 24 hours of head injury (11 men, 4 women) with a mean (range) age of 39 (16–70) years (Table 1). Patients had a median (range) postresuscitation Glasgow Coma Score (Teasdale and Jennett, 1974) of 8 (3–12) and developed significant elevation in intracranial pressure (ICP), requiring sedation and control of ventilation. In 13 of these subjects, simultaneous measurement of SjO2 and AJDO2 was available. All volunteers provided informed consent for studies, and assent was obtained from the next of kin for all patient studies. All studies were approved by the Local Research Ethics Committee at Addenbrooke's Hospital, Cambridge, UK, and by the Administration of Radioactive Substances Advisory Committee of the UK.
Patient Characteristics
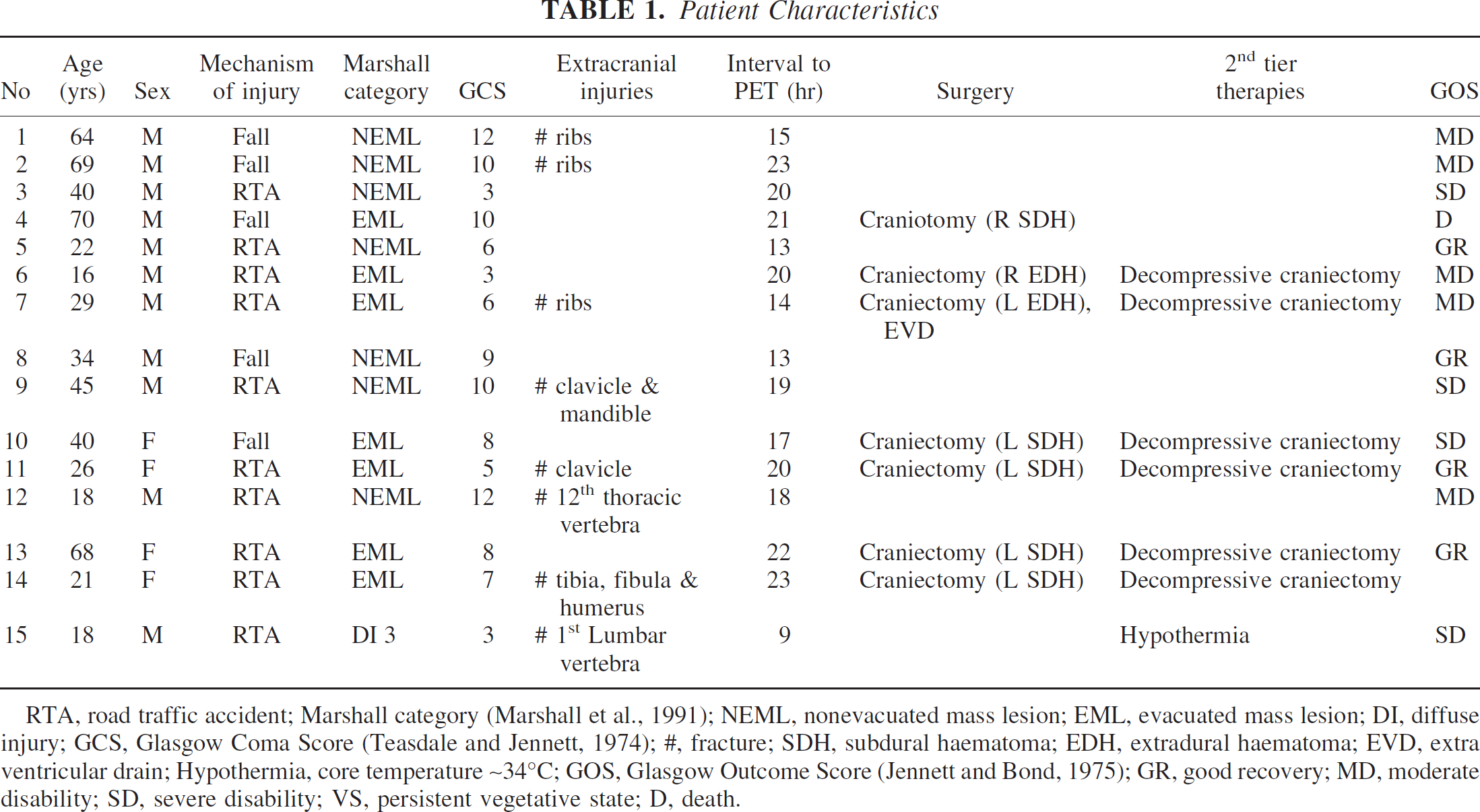
RTA, road traffic accident; Marshall category (Marshall et al., 1991); NEML, nonevacuated mass lesion; EML, evacuated mass lesion; DI, diffuse injury; GCS, Glasgow Coma Score (Teasdale and Jennett, 1974); #, fracture; SDH, subdural haematoma; EDH, extradural haematoma; EVD, extra ventricular drain; Hypothermia, core temperature ~34°C; GOS, Glasgow Outcome Score (Jennett and Bond, 1975); GR, good recovery; MD, moderate disability; SD, severe disability; VS, persistent vegetative state; D, death.
Clinical protocols
Patients were managed with protocol driven therapy aimed at maintaining ICP below 20 mm Hg and cerebral perfusion pressure (CPP) above 70 mm Hg, as previously described (Menon, 1999). Interventions included sedation (propofol up to 8 mg·kg−1·hr−1 and fentanyl 1–2 mg·kg−1·hr−1) and neuromuscular blockade, surgery for space-occupying lesions, drainage of CSF, volume supplementation and vasoactive agents (dopamine and norepinephrine) for CPP augmentation, osmotic diuretics (boluses of 0.5 g/kg of mannitol), and mild hyperventilation (to ~35 mm Hg). Hyperthermia was treated vigorously, and mild hypothermia (~36°C) was commonly used to assist ICP control. Patients who received surgical intervention (CSF drainage or decompressive craniectomy) or second tier medical therapies (barbiturate coma or moderate hypothermia of 33–35°C) before PET imaging are specified in Table 1.
In addition, a fiberoptic right jugular bulb catheter (Baxter, Newbury, UK) was inserted, and its position was confirmed radiologically. Samples of arterial and jugular venous blood were drawn for simultaneous measurement of arterial blood gases and SjO2 and calculation of AJDO2. Using protocol driven therapy (Menon, 1999), SjO2 was continuously measured, and attempts were made to maintain levels above 50%, with regular calibration and measurement of AJDO2.
Whereas hemodynamic stability was ensured during PET studies by titrating fluids and vasoactive agents, sedative infusions were left unchanged. It is important to emphasize that no other material changes in management occurred on the day of the PET study in any of the patients, thus ensuring stable physiology during the acquisition of imaging data.
Positron emission tomography
PET studies were undertaken on a General Electric Advance scanner (GE Medical Systems, Milwaukee, U.S.A.). Emission data were acquired in 3D mode during a 20-minute steady state infusion of 800 MBq of H2 15O (two 5-minute frames at the end of the administration), following a 60-second inhalation of 300 MBq of C15O (single 5-minute frame), and in 2D mode during a 20-minute steady state inhalation of 7,200 MBq of 15O2 (two 5-minute frames at the end of the administration). Just before emission data acquisition, a 10-minute transmission scan was performed using rotating germanium-68 rods (2 ϗ ~300 MBq). These data, in combination with that from a 60-minute blank scan acquired on the same day, were used to correct the emission data for photon attenuation. Images were reconstructed into 2.34 × 2.34 × 4.25 mm voxels using the PROMIS 3D filtered back projection algorithm (Kinahan and Rogers, 1989), with corrections applied for attenuation, scatter, randoms, and dead time. Emission images were smoothed using an isotropic 4 mm Gaussian filter, and subsequently, parametric maps of CBF, cerebral blood volume (CBV), CMRO2, and OEF were calculated by inputting simultaneous PET and arterial tracer activity measurements into standard models (Frackowiak et al., 1980; Lammertsma et al., 1987). We used a blood-brain partition coefficient for H2 15O (ρ) of 0.95 based upon the previous in vitro data (Herscovitch and Raichle, 1985) and a small to large vessel hematocrit ratio (r) of 0.85 (Phelps et al., 1979). Parametric PET images were coregistered to anatomy using magnetic resonance (MR) imaging in volunteers or x-ray computed tomography (CT) in patients.
Image analysis
Images were analyzed using custom designed automated software (PETAN) (Smielewski et al., 2003) incorporating elements of Statistical Parametric Mapping (SPM99, Wellcome Department of Cognitive Neurology, London, UK), Matlab 5.2 (MathWorks, Inc., Natick, Maryland, U.S.A.), Analyze 4.0 (AnalyzeDirect, Inc., Lenexa, Kansas, U.S.A.), and registration by multiresolution optimization of mutual information (mpr) (Department of Radiological Sciences, Guys Hospital, London, UK) (Studholme et al., 1996, 1997). Individual anatomic images were edited to extract a template that identified brain tissue voxels and excluded extracranial tissue, cerebrospinal fluid, and extraaxial hematomas. This brain template was applied to the coregistered parametric PET images and used to generate corrected maps of CBF, CBV, CMRO2, and OEF.
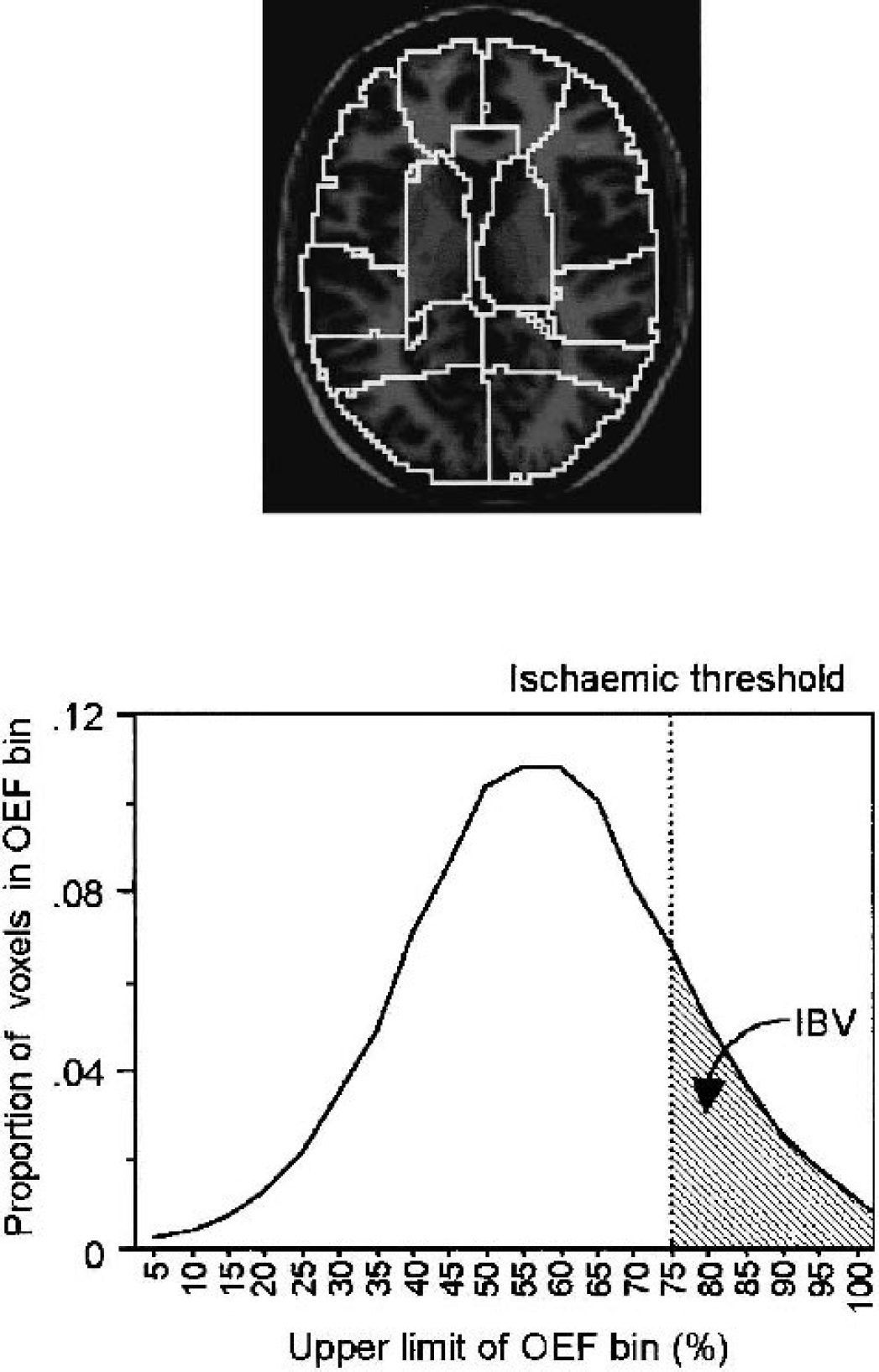
Methodology. (A) T1-weighted image of reference brain, normalized to Talairach space, indicating the 15 ROIs defined in the cerebral hemispheres on the basis of anatomical divisions and vascular territories: right and left medial frontal, lateral frontal, temporal, parietal, occipital, deep grey matter, cerebellum, and the brain stem. (B) OEF histogram from a single patient, showing the distribution of the number of voxels in each OEF bin. Critical OEF thresholds are derived by calculation of the OEF value above which CvO2 would be less than 3.5 mL/100 mL. The summed volume of voxels with OEF values above this threshold is the IBV. ROIs, regions of interest; OEF, oxygen extraction fraction; CvO2, cerebral venous oxygen content; IBV, ischemic brain volume.
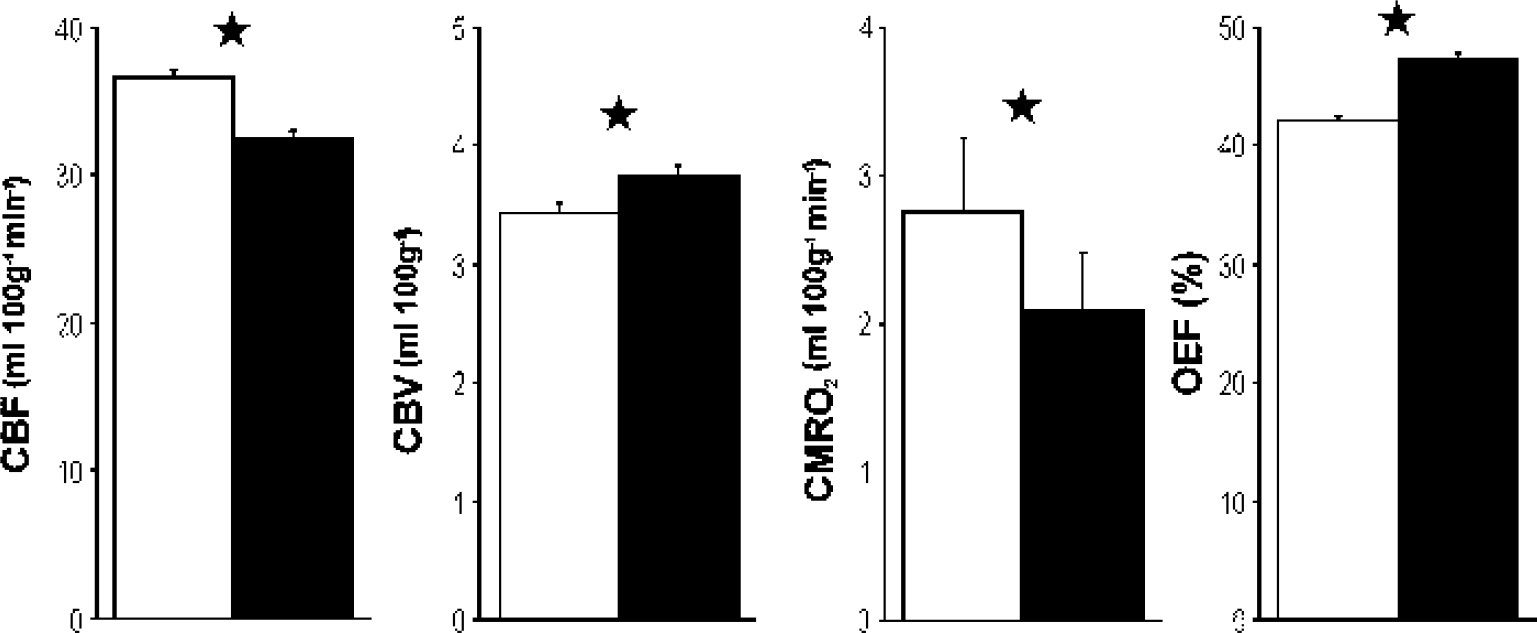
ROI analysis. Mean + SD CBF, CBV, CMRO2, and OEF in ROIs derived from control subjects (white) and from patients with head injuries within 24 hours of ictus (black). (*P < 0.001; unpaired t-tests with Bonferroni correction). CBF, cerebral blood flow; CBV, cerebral blood volume; CMRO2, cerebral metabolic rate of oxygen; OEF, oxygen extraction fraction; ROIs, regions of interest.
Estimation of ischemic burden
We used OEF to assess the burden of ischemia in PET images to avoid the confounding effects of drug and injury induced metabolic suppression upon CBF and CMRO2. It is difficult to find data that identify critical increases in OEF levels that still allow survival in the setting of ischemia, but some guidance is available from both human (Powers et al., 1985; Yundt and Diringer, 1997) and experimental data (Sutton et al., 1990). Sutton and colleagues (1990) identified the cerebral venous oxygen content (CvO2) as the critical predictor of infarction in experimental ischemia, with the highest observed CvO2 threshold for infarction being 2.9 mL/100 mL. Clinical studies (Powers et al., 1985; Yundt and Diringer, 1997) are broadly concordant with these data and suggest that brain regions can survive in the setting of prolonged ischemia with local CvO2 levels as low as 3.5 mL/100 mL.
Consequently, we estimated an individualized critical OEF threshold (OEFcrit; which equated to a CvO2 of 3.5 mL/100 mL) for each subject as follows:
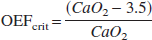
where CaO2 is arterial oxygen content calculated as (1.34 ϗ Hb ϗ (SaO2/100)) + (0.003 ϗ PaO2), Hb = hemoglobin in g/100 mL, and SaO2 = arterial oxygen saturation.
Application of these thresholds to frequency histograms of OEF images allowed us to calculate the volume of voxels with CvO2 values below this threshold and hence allowed estimation of the ischemic brain volume (IBV) (Fig. 1B). We compared the IBV with data derived from jugular oximetry using traditional threshold values for SjO2 and AJDO2 (50% and 9 mL/100 mL, respectively) (Gibbs et al., 1942; Kety and Schmidt, 1948; Obrist et al., 1984; Robertson and Cormio, 1995; Robertson et al., 1989).
Statistical analysis
Statistical analysis was undertaken using Statview© (Version 5, 1998, SAS Institute Inc., Cary, North Carolina, U.S.A.). All data are expressed and displayed as mean ± SD, unless otherwise stated. Global, ROI, and voxel-based PET data from patient groups and control subjects were compared using two-tailed unpaired t-tests. After statistical advice, individual ROIs were treated independently after Bonferroni correction because they represented a clinically relevant method of segmenting the brain to look for regional ischemia, with specific location being irrelevant to this analysis. However, we have not undertaken to detect whether ischemia tends to occur with increased frequency in specific anatomic areas. Relationships between IBV and SjO2, AJDO2, and Glasgow Outcome Score (GOS) (Jennett and Bond, 1975) were explored using linear regression, polynomial fitting, and nonparametric methods (Spearman Rank correlation test) to take account of nonlinear relationships between variables. All P values are quoted after Bonferroni corrections (where appropriate), and corrected P values < 0.05 were considered significant.
RESULTS
Global physiology
The differences between patients and healthy volunteers did not reach significance, except for CMRO2 (Table 2) (P < 0.001 unpaired t-test with Bonferroni correction).
Regional physiology
Mean ± standard deviation for global CBF, CBV, CMRO2, and OEF in healthy controls and patients within 24 hours of injury
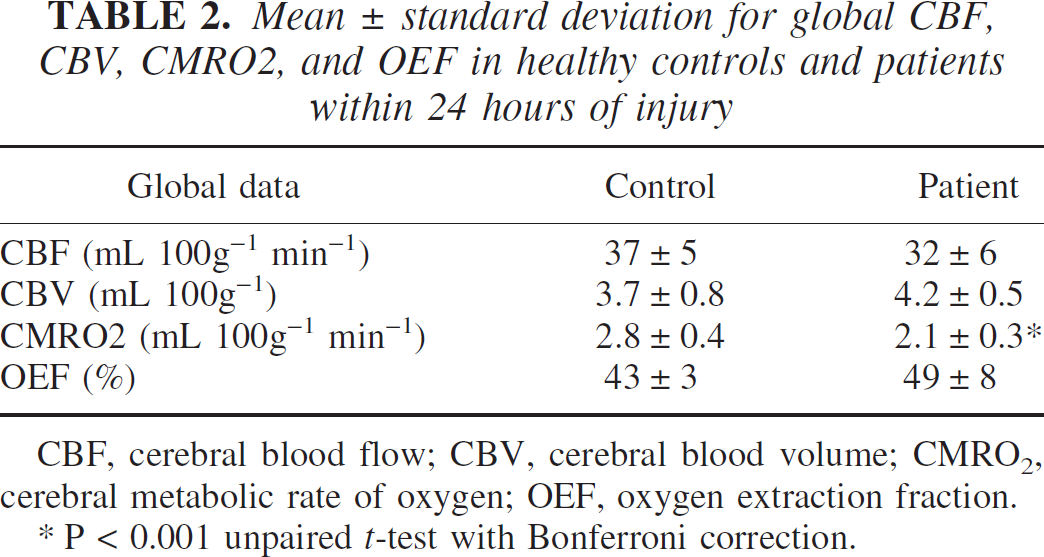
CBF, cerebral blood flow; CBV, cerebral blood volume; CMRO2, cerebral metabolic rate of oxygen; OEF, oxygen extraction fraction.
P < 0.001 unpaired <i>t-test with Bonferroni correction.
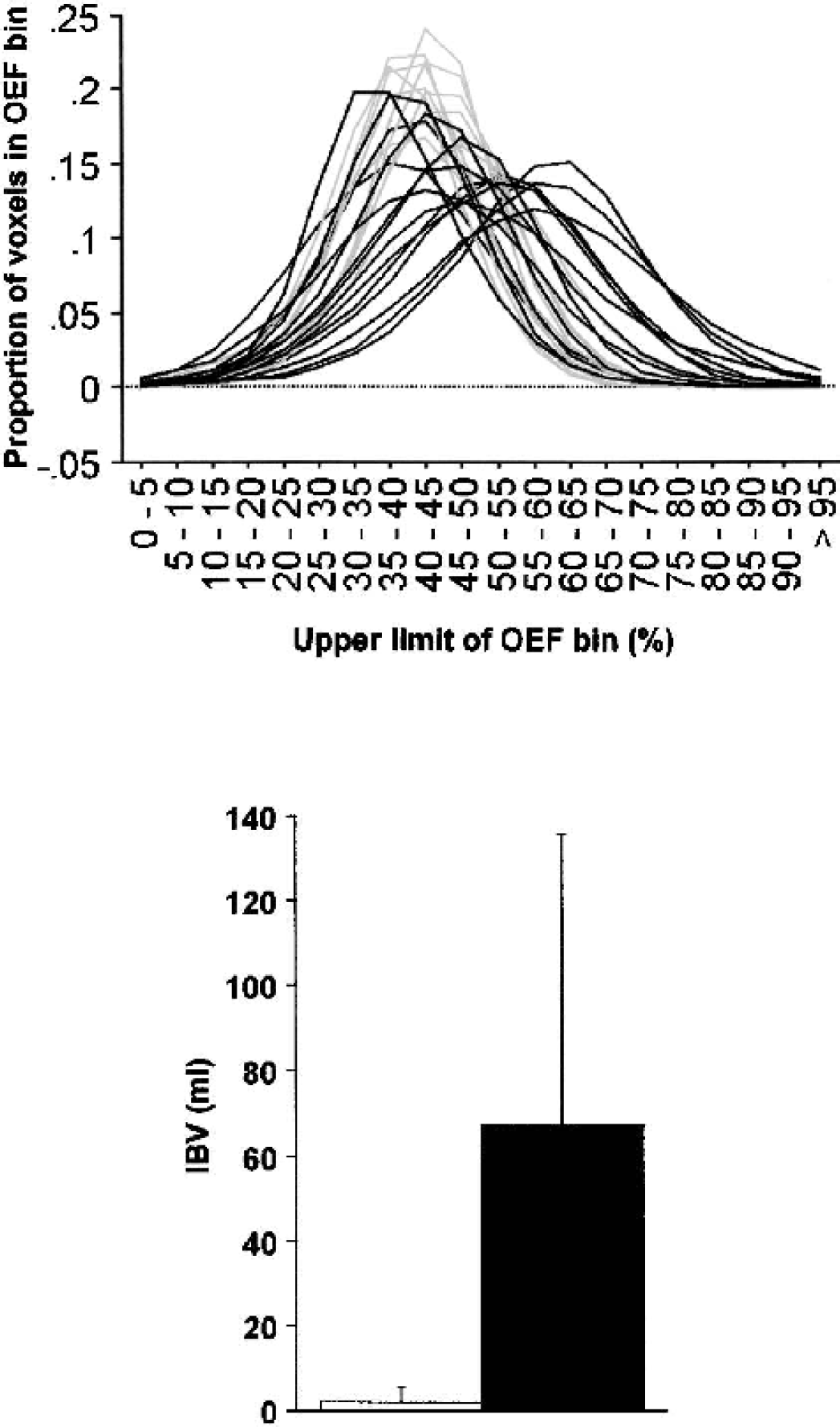
Voxel-based analyses. (A) OEF histograms from 10 volunteers (grey) and 15 patients within 24 hours of injury (black). Note the rightward shift of the patient histograms, which show prominent ischemic “tails” composed of voxels with high OEF values. (B) Mean + SD ischemic brain volumes from control subjects (white) and patients with head injury within 24 hours of ictus (black). (P < 0.01; unpaired t-test). OEF, oxygen extraction fraction.
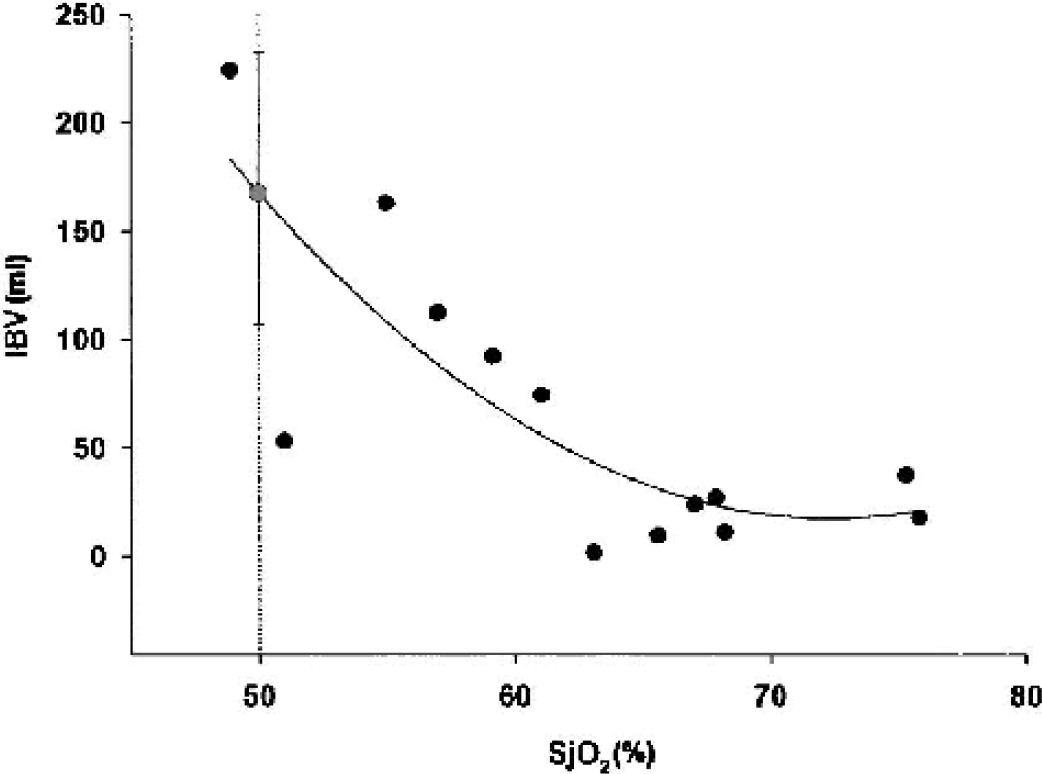
Relationship of PET-derived estimate of IBV to jugular bulb oximetry using SjO2. The ischemic threshold based on SjO2 monitoring is displayed with black dashed lines. The grey circle and error bars represent the mean ± 95% CI for the IBV at which the model indicates the SjO2 threshold is achieved. PET, positron emission tomography; IBV, ischemic brain volume; SjO2, saturation of jugular bulb blood.
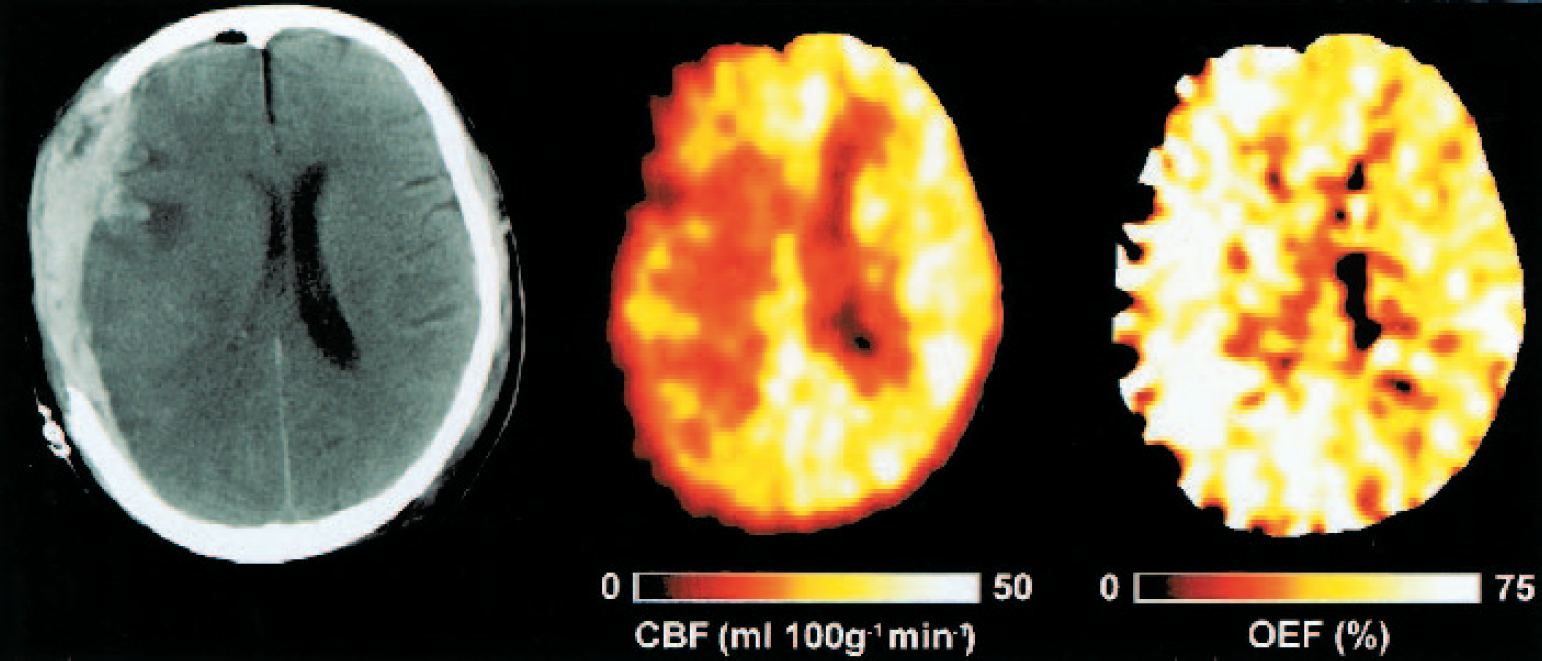
PET images after early head injury. X-ray CT, PET CBF, and OEF images obtained from a 42-year-old woman 16 hours after injury following evacuation of a subdural hematoma. The intracranial pressure was 18 mm Hg, cerebral perfusion pressure was 78 mm Hg, and PaCO2 was 36 mm Hg. Note the small amount of residual subdural blood with minimal midline shift and the marked reductions in CBF and increases in OEF in the cerebral hemisphere underlying the evacuated subdural hematoma. PET, positron emission tomography; CT, computed tomography; CBF, cerebral blood flow; OEF, oxygen extraction fraction.
We were able to obtain follow-up data on 14 of the 15 patients (Table 1). The IBV correlated with a poor GOS (ρ = −0.6; P < 0.05, Spearman Rank) at 6 months after injury.
DISCUSSION
We have used novel techniques to detect and quantify cerebral ischemia in head injury and underline the limitations of a widely used monitoring tool. Conventional management of head trauma is based upon the premise that ischemia is an important pathophysiologic mechanism of secondary injury, and treatment of patients has focused on maintenance of systemic oxygenation and cerebral perfusion pressure as a means of minimizing such injury. Indeed, early removal of space occupying lesions, control of ICP, and maintenance of CPP have been associated with improved clinical outcomes in head injury (Becker et al., 1977; Miller, 1985; Patel et al., 2002; Rosner et al., 1995). Although such interventions remain vital, further improvements in outcome will depend upon the identification and correction of other pathophysiologic mechanisms that contribute to neuronal injury and affect clinical outcome. The regional ischemia that we demonstrate here may represent one such therapeutic target.
Methodologic issues
Conventional functional imaging approaches in clinical and experimental stroke have traditionally used CBF thresholds for ischemia and have succeeded in identifying useful predictive values for tissue survival or death (Heiss et al., 1992; Marchal et al., 1996; Powers et al., 1985). However, the situation in head injury is confounded by the use of sedative agents and by the metabolic effects of trauma (Bergsneider et al., 2000; Verweij et al., 2000), which may cause primary reductions in cerebral metabolism; coupled CBF decreases in this context would not represent ischemia. Under these circumstances, the only true measure of the adequacy of CBF is a measurement of the OEF. In addition, pathophysiology in head injury is complex. Although the primary injury is important in terms of eventual outcome, secondary ischemic insults are responsible for worsening of outcome in many patients (Jones et al., 1994). In this case, it is difficult to predict thresholds for tissue viability based upon early measurements of CBF and CMRO2 and late structural imaging with any certainty.
Additional problems arise with quantifying ischemic burden in the injured brain because of the lack of a priori knowledge regarding the location of ischemia. Ischemia in stroke usually conforms to topographic patterns, with identification of an ischemic core and penumbra (Baron, 1999). Whereas ischemia may be prominent in perilesional areas in head injury, significant ischemia may also be observed in the structurally normal brain (Coles et al., 2002), and this may be significantly modulated by systemic physiology owing to impaired pressure autoregulation and the effect of PaCO2 on the cerebral circulation. Consequently, we needed to identify methods to estimate ischemic burden across the entire brain, making no assumptions regarding the location of such ischemia. The integration of voxels with critically high OEF values provides a novel way of making such estimates in practice, but the identification of ischemic thresholds needs to be based on sound physiologic premises. Both experimental (Sutton et al., 1990) and clinical (Powers et al., 1985; Yundt and Diringer, 1997) literature suggest that the CvO2 may provide the best variable to predict the risk of ischemic injury because it uses oxygen transport data (derived from hemoglobin and arterial oxygen saturation) to individualize the information provided by OEF values. We provide data on ischemic brain volumes with a CvO2 cut-off of 3.5 mL/100 mL based upon the best available estimates of critical CvO2 (Powers et al., 1985; Sutton et al., 1990; Yundt and Diringer, 1997). Moving this threshold up or down results in small changes in the measured ischemic brain volume but does not materially affect the statistical or clinical significance of the result.
The comparison we present between global, ROI, and voxel-based analyses provides some insight into the challenges posed by the interpretation of these data. It is difficult to locate evidence of a “traumatic penumbra,” which is defined as reduced CBF, increased OEF, and relatively preserved CMRO2 (Baron, 1999), based upon whole brain or structurally defined regions. The effect of trauma upon the cerebral circulation and metabolism is both spatially (Bouma et al., 1992; Coles et al., 2002; McLaughlin and Marion, 1996) and temporally variant (Martin et al., 1997). In addition, where regions suggest such evidence, it is difficult to interpret the degree and significance of hypoperfusion and metabolic compromise in the light of drug- and trauma-induced metabolic suppression. In such circumstances, OEF provides the only valid measure of the adequacy of CBF and oxygen delivery and is independent of changes in CMRO2. The availability of defined OEF thresholds for neuronal death would allow the clear definition of critical ischemia, but there are no data on what value of OEF, if sustained for a period of time, leads to neuronal damage. However, even in the absence of such predictive thresholds, the imbalance in flow-metabolism coupling denoted by the high OEF values that we describe characterizes tissue at high risk of ischemic injury. The OEF threshold that we use is based upon the best available data and is clinically relevant in terms of the management of head injury, where we wish to prevent the occurrence of further neuronal injury.
It must also be emphasized that the PET imaging that we undertook was a snapshot of brain metabolic state. Whereas this does provide an indication of the burden of ischemia at the time of imaging, it does not take any account of the duration of such ischemia. We maintained systemic physiology at levels that we considered optimal for brain protection. Consequently, we would expect the distribution of ischemia to represent the physiology seen in patients managed in an intensive care unit with similar therapeutic targets. However, we cannot exclude the possibility that transient events (e.g., an undetected focal seizure) may have temporarily increased oxygen demand during the scan. Under such circumstances, the short-lived period of high OEF may not result in tissue injury, and the IBV may overestimate the burden of ischemia that is of relevance to outcome. Conversely, ischemia may develop at other times during the patient's illness and adversely affect outcome but would be undetected during a single scan. This would lead to the “snapshot” IBV figure, underestimating the integrated burden of ischemia over the course of the patient's illness.
Close matching of flow to metabolism normally results in remarkably little variation in OEF across the brain despite wide regional variations in CBF and CMRO2 (Lebrun-Grandie et al., 1983). The broad-based OEF histograms after head injury (Fig. 3A) suggest that matching of perfusion to oxygen use is impaired after head injury. Although lower emission statistics could produce more extreme OEF values in the setting of ischemia in such voxel-based measurements, we have shown that the intrasubject spread of ROI OEF values is also significantly increased in head injury. This estimate of OEF spread from large ROIs is much less susceptible to noise and provides independent quantitative confirmation of our findings. These data suggest that whereas high OEF values may represent ischemia, the pathophysiology may be more complex than that seen with ischemic stroke. The coexistence of relative ischemia and hyperemia in some patients may represent a more fundamental problem with matching of perfusion to oxygen use after head injury.
The use of voxel-based analysis to define tissue at risk is new in this context but has been used in other settings such as stroke (Marchal et al., 1999). However, statistics in such voxel-based measurements are less robust than those from larger ROIs and likely to be more susceptible to reductions in signal-to-noise ratios. Consequently, the calculated spread of values in metabolic maps will increase when emission counts are reduced, as with low CBF values in patients. The concern is that this would result in more extreme values, which might translate into more voxels with high OEF values and broader OEF distributions. We have examined this issue in experiments using clinical and phantom data (Coles et al., 2003), which assessed whether the increases in IBV and SD of the OEF in patients with head injuries could be attributed to degraded image statistics arising from lower CBF values. We simulated the effects of a global reduction in blood flow in control datasets, from a baseline of 37 mL 100g−1 min−1, to approximately 20, 10, and 5 mL 100g−1 min−1. We then repeated the statistical analysis to see whether differences between patient and control groups were maintained. The calculated IBV within 24 hours of head injury remained significantly higher than the control group in all comparisons, except when global blood flow was artificially set to 5 mL 100g−1 min−1 (67 ± 69 mL in the patient group vs. 3 ± 1, 11 ± 10, and 46 ± 31 mL for simulated control data with a blood flow equivalent to 20, 10, and 5 mL 100g−1 min−1 respectively).
Similarly, the measured SD of the OEF distribution in patients within 24 hours of head injury remained higher than the simulated data with a global CBF of 20 mL 100g−1 min−1 (16.8 ± 3.8% vs. 11.3 ± 0.3%) but was not statistically significantly different from that with a CBF of 10 and 5 ml 100g−1 min−1 (16.8 ± 3.8% vs. 14.1 ± 2.1% and 19.5 ± 2.1%, respectively). Whereas it is clear that a reduction in emission statistics as a consequence of reduced blood flow will lead to an absolute increase in IBV and SD of the OEF distribution, a significant increase in comparison to control data should not be ignored. It is important to note that the effects of poor image statistics do not become apparent until blood flow is reduced below 10 mL 100g−1 min−1 (Coles et al., 2003). In patients, the mean (range) volume of brain tissue with blood flow below 10 and 5 mL 100g−1 min−1 coupled with high OEF (based upon a calculated CvO2 of < 3.5 mL/100 mL) was 7 (0–22) mL and 3 (0–11) mL, respectively. These regions of low perfusion would not account for a mean IBV of 67 mL and a maximum of 224 mL in our patients. Furthermore, it is important to recognize that even CBF values less than 10 mL 100g−1 min−1 do not exclusively represent noise and may still be clinically relevant.
The presence of distributed ischemia in early head injury might be expected to result in distributed tissue loss. However, structural images obtained at follow-up using x-ray CT or MR do not commonly show late evidence of such multifocal ischemia after head injury. However, penumbral levels of ischemia may be associated with selective neuronal loss, rather than pannecrosis (Baron, 1999; Garcia et al., 1996; Marchal et al., 1996). Indeed, clinical proton (1H) MR spectroscopy studies after head injury do show diffuse reductions in N-acetyl aspartate (NAA), even in regions with no structural lesions (Brooks et al., 2000; Friedman et al., 1998; Garnett et al., 2000). Because NAA is recognized as a marker for viable neurons, these changes are fully in keeping with selective neuronal loss. This suggests that 1H MR spectroscopy at follow-up may provide one means of validating the thresholds used to define critical ischemia in these experiments. An alternative approach would be to use phosphorus-31 (31P) MR spectroscopy (Garnett et al., 2001) to define cellular energy failure and relate this to PET-defined indices of ischemia. However, there are no such correlative studies in the acute stage, and future data will need to take account of the relatively low spatial resolution provided by 31P MR spectroscopy.
Clinical correlates
The relationship that we observed between IBV and SjO2 suggests that SjO2 readings may be highly variable in patients with IBV values at the lower end of the range. Whereas several factors may be responsible for this, a significant contributor is the varying degree of hyperemia, which would tend to result in higher SjO2 values and lower mean OEF across the brain, despite similar IBV values. The severity and extent of such hyperemia is represented by the number of voxels at the hyperemic end of the OEF distribution. It is also of clinical relevance that commonly quoted SjO2 thresholds for ischemia (50%) (Gibbs et al., 1942; Robertson and Cormio, 1995) were only reached when a substantial proportion of the brain was potentially ischemic. The model derived from our data indicates that this threshold is only achieved with an estimated IBV of approximately 170 mL, which equates to 13% of the brain volume. The absence of a clear relationship between IBV and AJDO2 and the relatively narrow range of AJDO2 values that we observed (2.6–7.2 mL/100 mL) precluded the assessment of an IBV value that correlated with an AJDO2 of 9 mL/100 mL. However, the absence of AJDO2 values above this threshold despite high IBV values in many of our patients suggests that AJDO2 is an insensitive measure of regional cerebral ischemia.
Could we have refined our use of global cerebrovascular monitoring to provide better discrimination of ischemia? Various groups have suggested more targeted lateralization of SjO2 monitoring and expressed doubt regarding the reliability of unilateral monitoring (Metz et al., 1998; Stocchetti et al., 1994). Furthermore, some authors have used more focused measures of ischemia, such as the lactate oxygen index (LOI) (Robertson et al., 1989), the modified LOI (Cruz et al., 1993), and the cerebral extraction of oxygen (CEO), which uses direct measurement of saturation differences between arterial and jugular blood (Cruz et al., 1993, 1994). Although these approaches may offer some advantages, we do not believe that they can successfully overcome the fundamental failure of a global monitor to detect regional ischemia.
Although it is difficult with the available data in clinical head injury to define any physiologic threshold based on local tissue outcomes, the relationship with clinical outcome based on the 6-month GOS provides some additional information. We were able to obtain follow-up data at 6 months in 14 of the 15 patients that we studied. We show that the burden of ischemia defined by the techniques that we describe correlated with clinical outcome. Although our data provides supportive evidence for the relevance of early physiologic insults in these patients, further confirmation is required before a causal relationship is proven.
Clinical implications
We believe that these findings are important for several reasons. First, they provide evidence that regional ischemia (rather than metabolically coupled hypoperfusion) is present in early head injury, even in patients who achieve suggested targets for CPP and ICP control (The Brain Trauma Foundation, 2000). Although the absolute volume of such tissue may be small, its location within frontal and temporal regions of the brain may be critically important in terms of neurocognitive outcome (Mattson and Levin, 1990). Second, the data raise concerns regarding the use of jugular bulb oximetry. All but one of the patients with significant volumes of critically ischemic brain had SjO2 values that were higher than 50%, which is a widely accepted threshold for defining cerebral ischemia (The Brain Trauma Foundation, 2000). Despite the difficulties in defining tissue at immediate risk of metabolic failure and death, these data suggest that common bedside monitoring techniques, such as jugular bulb oximetry, fail to detect regional ischemia that is clinically important and potentially related to outcome.
Footnotes
Acknowledgements
We thank Dr. J. C. Matthews for help and advice in conducting these studies.
