Abstract
Intracerebral microdialysis (MD) was applied in patients with subarachnoid hemorrhage. The regional CBF, the CMRO2, and oxygen extraction ratio (OER) were measured with simultaneous positron emission tomography (PET). The aim was to directly correlate alterations in dialysate levels of energy-related metabolites (lactate, lactate/pyruvate ratio, hypoxanthine) and excitatory amino acids (EAAs) (glutamate and aspartate) to the energy state in the MD probe region as determined by PET. Regional ischemia was defined according to Heiss et al. and Lassen (Heiss et al., 1992; Lassen, 1966). Whole-brain ischemia was considered present when the OER for the whole brain exceeded the mean whole-brain OER + 2 SD of six reference patients. In general, the presence of whole-brain ischemia and/or regional ischemia within the region of the MD probe was associated with increased levels of energy-related metabolites and EAAs retrieved by MD. Increased levels of energy-related metabolites and EAAs were only occasionally seen when PET did not show any signs of ischemia or when signs of regional ischemia were found remote from the MD probe region. Thus, the energy-related metabolites and EAAs may be used as extracellular “markers” of ischemia. PET may be of use in defining critical ischemic regions (tissue at risk) where the MD probe can be inserted for- chemical monitoring.
Keywords
The main objective of neurosurgical intensive care (NIC) is to maintain normal intracranial dynamics, including normal cerebral energy metabolism, and to avoid secondary ischemic/hypoxic insults to the brain. In clinical practice, the metabolic state of the brain has predominantly been followed by indirect methods such as the surveillance of the clinical neurological state, continuous registrations of intracranial pressure and cerebral perfusion pressure, repeated computed tomography (CT) scans, and measurements of jugular venous oxygen saturation and arteriovenous O2 differences.
The intracerebral microdialysis (MD) technique was introduced for long-term monitoring of brain energy metabolism in patients with acute brain injuries in 1992 (Persson and Hillered, 1992). The method enables the retrieval and analysis of small molecules present in the extracellular fluid (ECF); these include energy-related metabolites (lactate, pyruvate, glucose, adenosine, inosine, and hypoxanthine) and excitatory amino acids (EAAs) (glutamate and aspartate). The first clinical experience of MD in human brain ischemia was performed in the frontal cortex adjacent to a glioma during lobectomy; this served as a model of ischemia (Hillered et al., 1990). The second step was to correlate aN terations in dialysate levels with ischemic/hypoxic events assessed clinically during NIC (Persson and Hillered, 1992). A 25-fold increase in glutamate and aspartate was found to exist under conditions of energy perturbation, caused by intracranial hypertension, as indicated by a high lactate/pyruvate ratio and high levels of hypoxanthine. The chemical ECF alterations largely reflected the overall clinical course. The present study is a further effort to provide basic knowledge about the biochemistry of human cerebral ischemia by intracerebral MD with the ultimate goal of obtaining a monitoring instrument for the detection of imminent ischemia in routine clinical practice.
The specific aim of the study was to directly correlate the cortical dialysate levels of energy-related metabolites and EAAs, i.e., the extracellular reflection of primarily intracellular metabolic events (Hagberg et al., 1987; Hillered et al., 1989a, b ), to regional blood flow and energy metabolism, as measured by positron emission tomography (PET). Thus, the regional CBF (rCBF), the regional CMRO2 (rCMRO2) and the regional oxygen extraction ratio (rOER) were measured by PET simultaneously with intracerebral MD in patients with subarachnoid hemorrhage (SAH). The basic question was whether the presence of ischemia in the MD probe region demonstrated by PET was reflected in the dialysate levels of energy-related metabolites and EAAs.
CLINICAL MATERIAL AND METHODS
Patients
Patients with SAH who were referred immediately after ictus to the NIC unit were candidates for the study, provided that they had a need of a ventriculostomy for intracranial pressure monitoring and CSF drainage. Informed consent was obtained from the patients themselves or their relatives. The study was approved by the Ethics Committee of the Medical Faculty of Uppsala University and the University Hospital Radiation Protection Committee. Six patients were investigated, five women and one man, aged 46–70 years (mean age 57 years). These patients were recruited from a larger group of patients with severe SAH studied with long-term intracerebral MD at the NIC unit. The clinical characteristics of the six patients are presented in Table 1.
SAH patients investigated with simultaneous intracerebral MD and PET: Clinical characteristics, sedation, and physiological parameters during PET
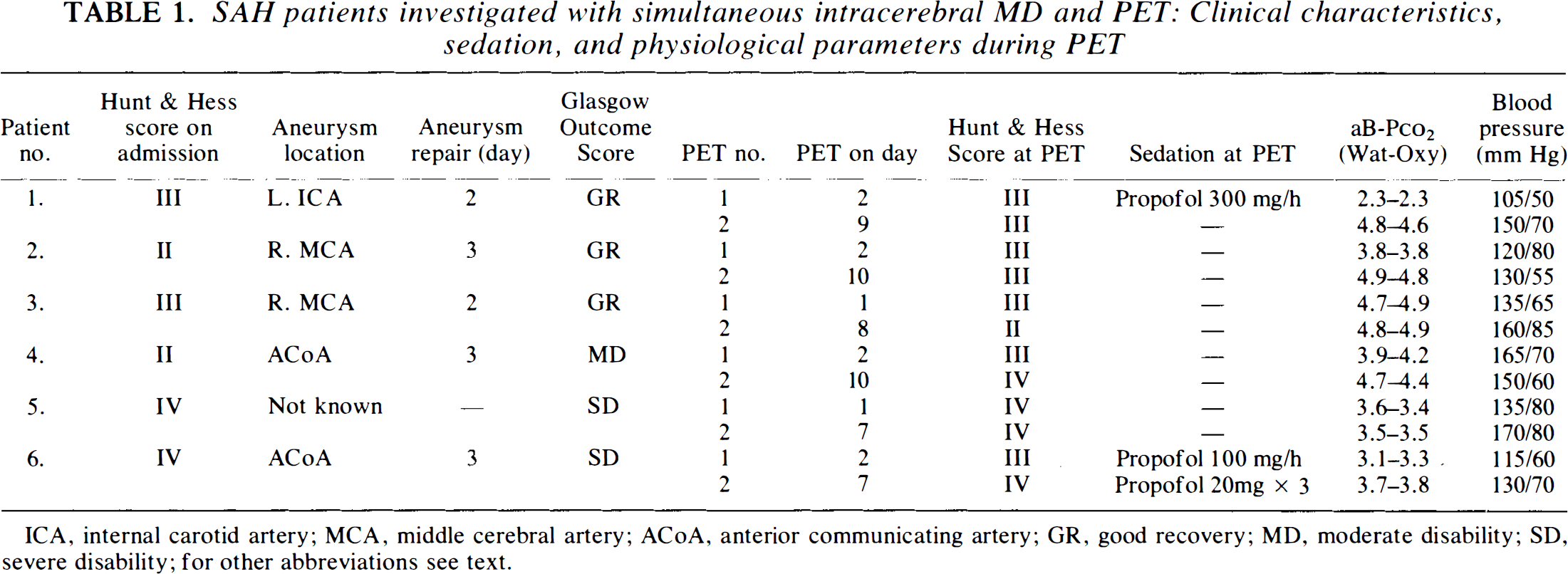
ICA, internal carotid artery; MCA, middle cerebral artery; ACoA, anterior communicating artery; GR, good recovery; MD, moderate disability; SD, severe disability; for other abbreviations see text.
Intracerebral MD
An MD probe with a membrane length of 4 mm (CMA/10; CMA/Microdialysis, Stockholm, Sweden) was inserted in conjunction with the intraventricular catheter ∼2 cm from the midline and 2 cm in front of the coronal suture (Persson and Hillered, 1992). About 5 mm of the MD probe was inserted into the cortex, including the whole membrane length. The perfusion medium was artificial CSF (containing Na+, 148 mM; Ca2+, 1.2 mM; Mg2+, 0.9 mM; K+, 2.7 mM; CI−1, 155 mM) delivered by a microinjection pump at a rate of 2 μl/min. During PET, the samples were collected at 10- or 15-min intervals. Energy-related metabolites (lactate, pyruvate, and hypoxanthine) and EAAs (glutamate and aspartate) were analyzed by HPLC, as described in detail previously (Fredholm and Sollevi, 1981; Hallström et al., 1989; Reid et al., 1990). Since the MD levels of energy-related metabolites and EAAs were stable during the PET measurements, only mean values of all fractions are given in Results.
PET
The PET investigations were performed at the Uppsala University PET Centre (UUPC). The UUPC is situated closely to the NIC unit and intensive care facilities are available at the UUPC; these are of vital importance if patient welfare is not to be compromised. The brain PET camera is equipped with eight rings of 256 detectors, producing 15 axial tomographic slices, which cover 10 cm along the longitudinal axis of the brain (GEMS 2048; General Electric Medical Systems). In-plane resolution is ∼6 mm full width at half-maximum, and slice thickness ∼6.5 mm (Holte et al., 1989). Attenuation correction was based on 10-min transmission scans using a rotating 68Ge pin source. Emission images were reconstructed using an 8-mm Hanning filter. 15O-labeled gases were administered using a specifically designed cart (PETgas 2000; General Electric Medical Systems), in which labeled gas from the radiochemistry production unit is mixed with room air or with oxygen-enriched air from the hospital medical gas supply to an adjustable radioactivity concentration. Patients who were able to cooperate used a mouthpiece and a nonrebreathing valve. In artificially ventilated patients, a small bias flow of labeled gas was fed into the inspiratory hose of the ventilator. Two PET investigations were carried out in each patient, the first within 48 h of hemorrhaging and the second 5–8 days later. Each PET session included measurements of CBF, CMRO2, OER, and cerebral blood volume (CBV).
The autoradiographic H215O method was used for measuring CBF. A bolus of H215O was injected into an antecubital vein at a dose of 15 MBq/kg body wt. Arterial blood was continuously withdrawn via a radial artery catheter and the radioactivity measured. Calculated flow images were created using the autoradiographic method with a predefined distribution volume of 0.95 cm3/cm3 and an integration time of 60 s (Raichle et al., 1983; Heidenreich et al., 1992). The time course of the arterial radioactivity concentration was corrected for delay and dispersion by nonlinear fitting to the total count rate for the scanner (Meyer, 1989).
A bolus administration of C15O was used for CBV measurements. C15O was administered during 3–10 breaths, using the system already described. After allowing arterial and venous C15O to equilibrate for 2 min, the tomograph collected 5 1-min frames. Radioactivity concentrations were measured at 1, 2.5, and 4 min, in whole-blood samples, and blood gas analysis (including CO-hemoglobin) was performed on the same samples. Regional CBV was calculated using the tomographic measurements, determinations of C15O in blood, and a constant correction factor of 0.70 for the difference between peripheral and cerebral hematocrit (Brooks et al., 1986; Heiss et al., 1992).
The steady-state 15O2 method was used to measure CMRO2 and OER. Air mixed with 15O2 was administered at a concentration of 4 MBq/min-kg body wt. When a steady-state level of 15O2 concentration was attained after 7–8 min, scanning of five to eight 1-min frames was initiated. Arterial blood and plasma were sampled at 1, 2.5, and 4 min for radioactivity measurements and for the determination of arterial blood O2 content and Pco2. With use of the previously measured CBF, images of OER and CMRO2 were created using the summation of data from all the frames acquired (Frackowiak et al., 1980). Correction for intravascular radioactivity was based on the CBV measurement (Lammertsma and Jones, 1983).
For the PET measurements, standardized regions of interest (ROIs) were delineated on the H215O total radioactivity images in two adjacent slices. The ROIs were adjusted with the help of the CBV images to exclude the large venous sinuses. ROIs in adjacent image sections were linked together to “volumes of interest.” To obtain whole-brain values, ROIs of the whole brain excluding the ventricular system and visible regional ischemic lesions were delineated. For regional measurements, each of the areas supplied by the anterior cerebral artery (ACA), middle cerebral artery, and posterior cerebral artery, respectively, and the MD probe region were delineated together with the corresponding contralateral regions. Visible ischemic lesions on CT or in either PET investigation were also delineated together with the corresponding contralateral regions.
The following definitions of ischemia were used during PET. The occurrence of regional ischemia was assessed by comparing corresponding contralateral regions to minimize the influence of confounding factors in accordance with Heiss et al. (1992). Regional ischemia was defined as (a) rOER > 125% and rCMRO2 ≥ 45% of the corresponding contralateral region, respectively, suggesting viable tissue with preserved metabolism requiring a blood supply (Heiss et al., 1992); or (b) rCMRO2 < 45% of the corresponding contralateral region, indicating a probable infarction (Lassen, 1966; Heiss et al., 1992). Whole-brain ischemia was defined as whole-brain OER > 53% (mean OER value + 2 SD of six reference patients who served as normal controls). OER reflects the relation between CBF and CMRO2, and an increased OER indicates an insufficient CBF to meet metabolic requirements, i.e., ischemia. By using OER in the definition of whole-brain ischemia, the inevitable problem of confounding effects of variations in the degree of consciousness, hyperventilation, and sedation, respectively, was overcome.
RESULTS
The whole-brain PET measurements varied considerably from patient to patient (Table 2). The mean whole-brain values for all PET examinations were as follows: CBF 39 ml 100 g−1 min−1 (range 22–58 ml 100 g−1 min−1), CMRO2 128 μmol 100 ml−1 min−1 (range 78–210 μmol 100 ml−1 min−1), and OER 49% (range 31–70%). The variability in these parameters appeared, at least partly, to reflect the level of consciousness of the patients, the degree of sedation, and the degree of hyperventilation (Table 1). Ischemic levels in the whole brain were found on four occasions in three patients (Table 2). Regional ischemia was found in several patients, both in the probe region (Table 2) and in areas remote from the probe.
PET values of whole brain and regional measurements of MD probe region
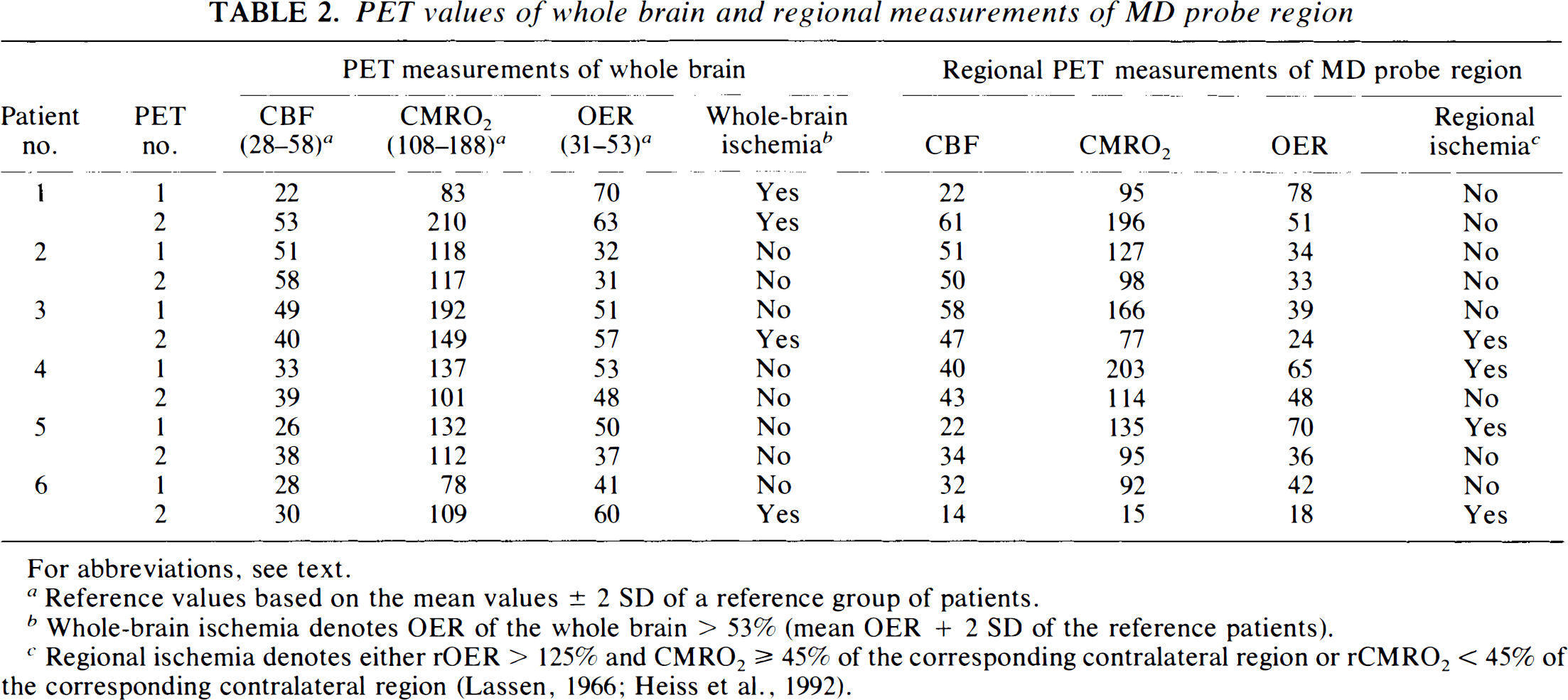
For abbreviations, see text.
Reference values based on the mean values ± 2 SD of a reference group of patients.
Whole-brain ischemia denotes OER of the whole brain > 53% (mean OER + 2 SD of the reference patients).
Regional ischemia denotes either rOER > 125% and CMRO2 ≥ 45% of the corresponding contralateral region or rCMRO2 < 45% of the corresponding contralateral region (Lassen, 1966; Heiss et al., 1992).
The levels of the chemical substances retrieved by MD are presented in Table 3 in relation to the PET findings. In Fig. 1, the MD dialysate level (normal or increased) is related to the occurrence of ischemia in the probe region on PET. Elevated level of the substances retrieved by MD was generally reflected by ischemia on PET. Chemical alterations were sometimes seen when PET showed no signs of ischemia.
Levels of dialysate chemical substances (mean values) during PET related to occurrence of whole-brain ischemia and regional ischemia MD probe region according to predefined criteria
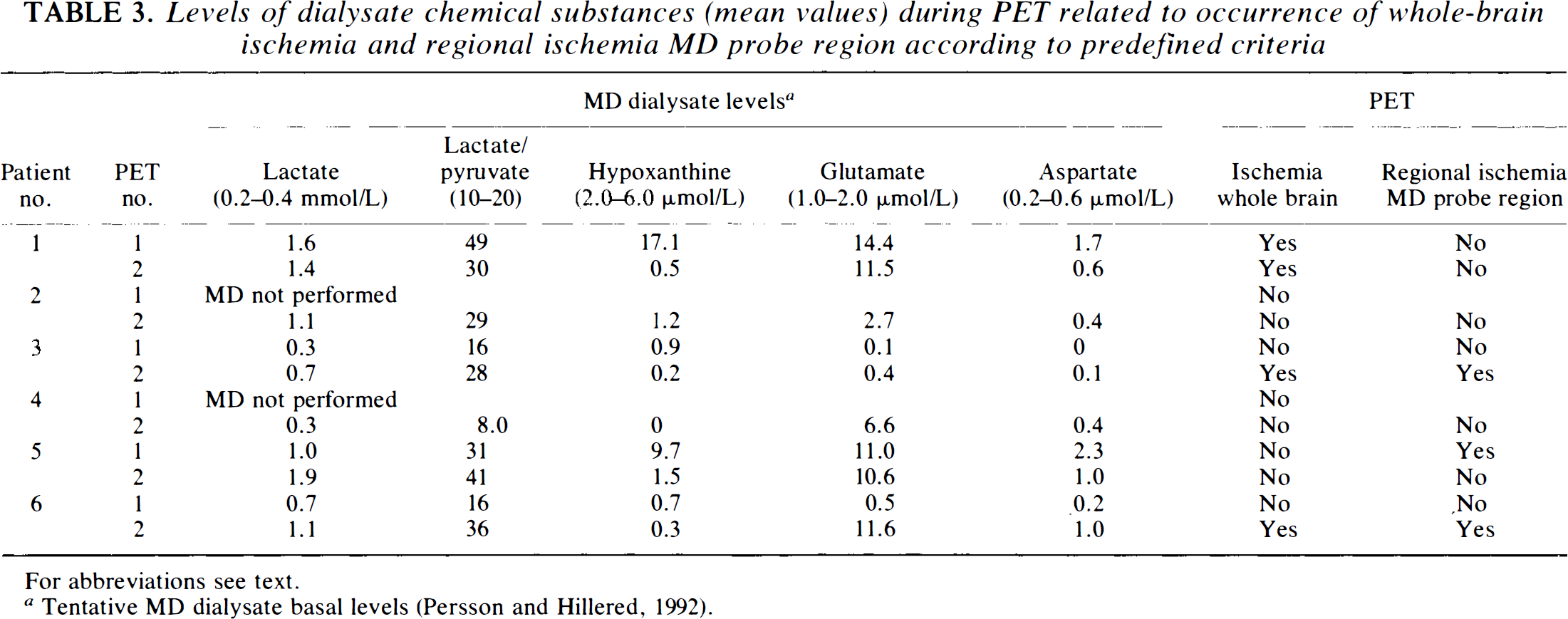
For abbreviations see text.
Tentative MD dialysate basal levels (Persson and Hillered, 1992).
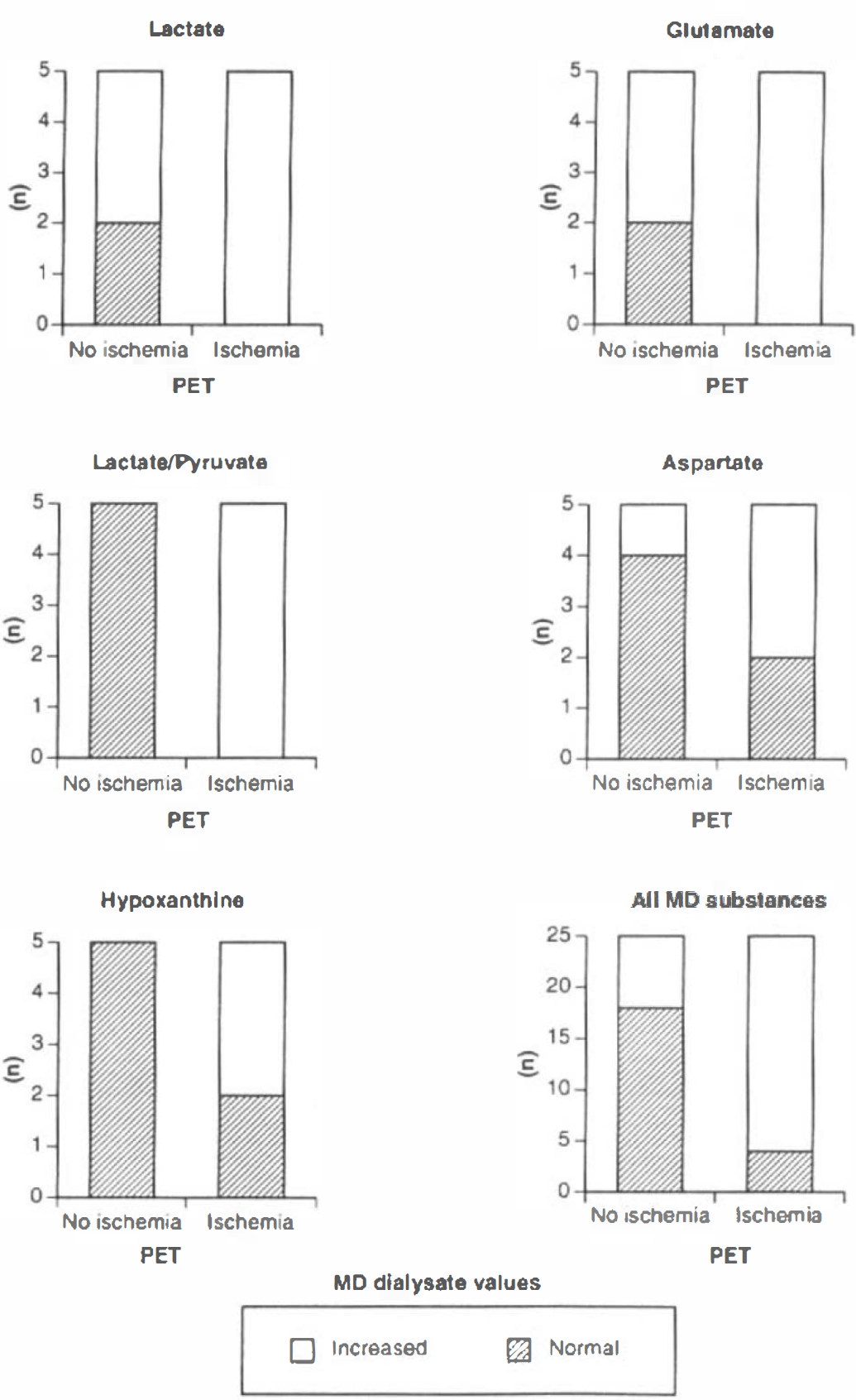
MD dialysate levels (normal or increased) related to the presence or absence of ischemia in the probe region in the 10 PET investigations. Ischemia in the MD probe region was either regional ischemia localized in the probe region or whole-brain ischemia including the probe region. See text for abbreviations.
The material was too small to allow detailed statistical analysis, but it was found that lactate, lactate/pyruvate ratio, and glutamate had the highest sensitivity for detecting ischemia, while aspartate and hypoxanthine were less sensitive (Table 4). The lactate/pyruvate ratio and hypoxanthine had the highest specificity, aspartate intermediate, and lactate and glutamate the lowest (Table 4).
Sensitivity and specificity of chemical substances in MD dialysate with regard to ischemia disclosed by PET
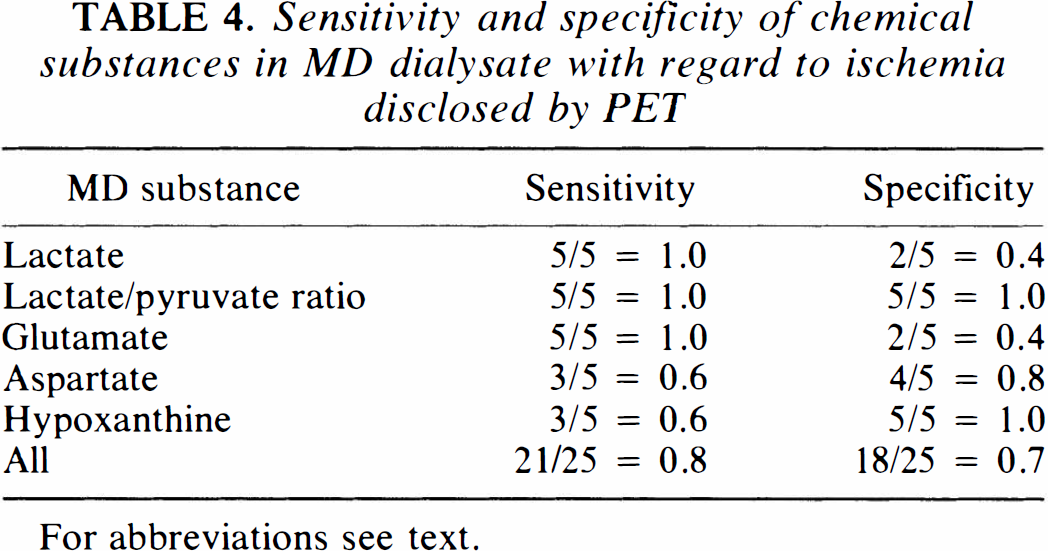
For abbreviations see text.
Illustrative case: Case 6
A 54-year-old woman, with a short transient loss of consciousness, developed epileptic seizures shortly after admission and was therefore intubated immediately. CT showed SAH and an intracerebral hematoma in the left frontal lobe, suggesting rupture of an anterior communicating artery aneurysm. This was confirmed by angiography. A ventricular catheter with an attached MD probe was inserted on day 1. She received propofol sedation and was hyperventilated during PET 1, which took place on the following day. The whole-brain PET values including the probe region were interpreted as approximately normal (Table 2; Fig. 2). Ischemic values were seen in the thalamus on both sides (Fig. 2). The MD values were virtually within the normal range (Fig. 3; Table 3). On day 3 surgery was performed and the aneurysm clipping provided problems. After repeated temporary clipping of the ACAs, the aneurysm was repaired. Postoperatively, the patient deteriorated and CT showed signs of infarction in the territory of the right ACA and in the left caudate nucleus. Follow-up angiography showed signs of moderate diffuse vasospasm and occlusion of the right ACA. In the region corresponding to the right ACA where the MD probe was placed, the PET values indicated manifest infarction (Table 2; Fig. 2). In the dialysate, all parameters except hypoxanthine were increased (Fig. 3; Table 3). The MD monitoring results showed that all MD parameters including hypoxanthine had increased to a maximum on days 3 and 4 and then gradually decreased (Fig. 3). Therefore, hypoxanthine was in this case considered as elevated in the workup of data.
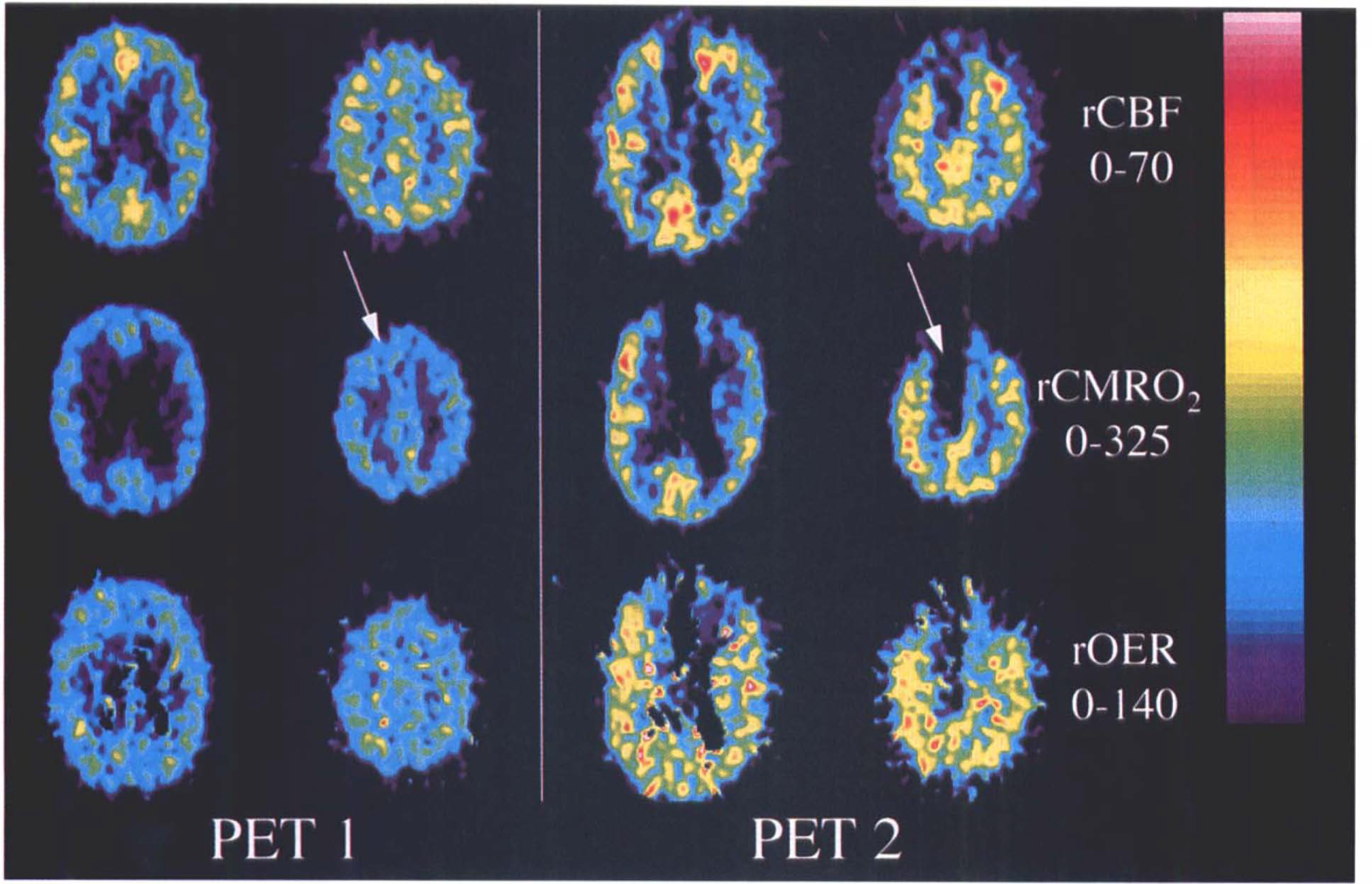
PET scans of Case 6. PET 1: day 2 after SAH. No ischemia in the probe region (arrow). PET 2: day 7 after SAH. Signs of whole-brain ischemia and manifest infarction in the right frontal lobe including the probe region (arrow). See text for abbreviations.
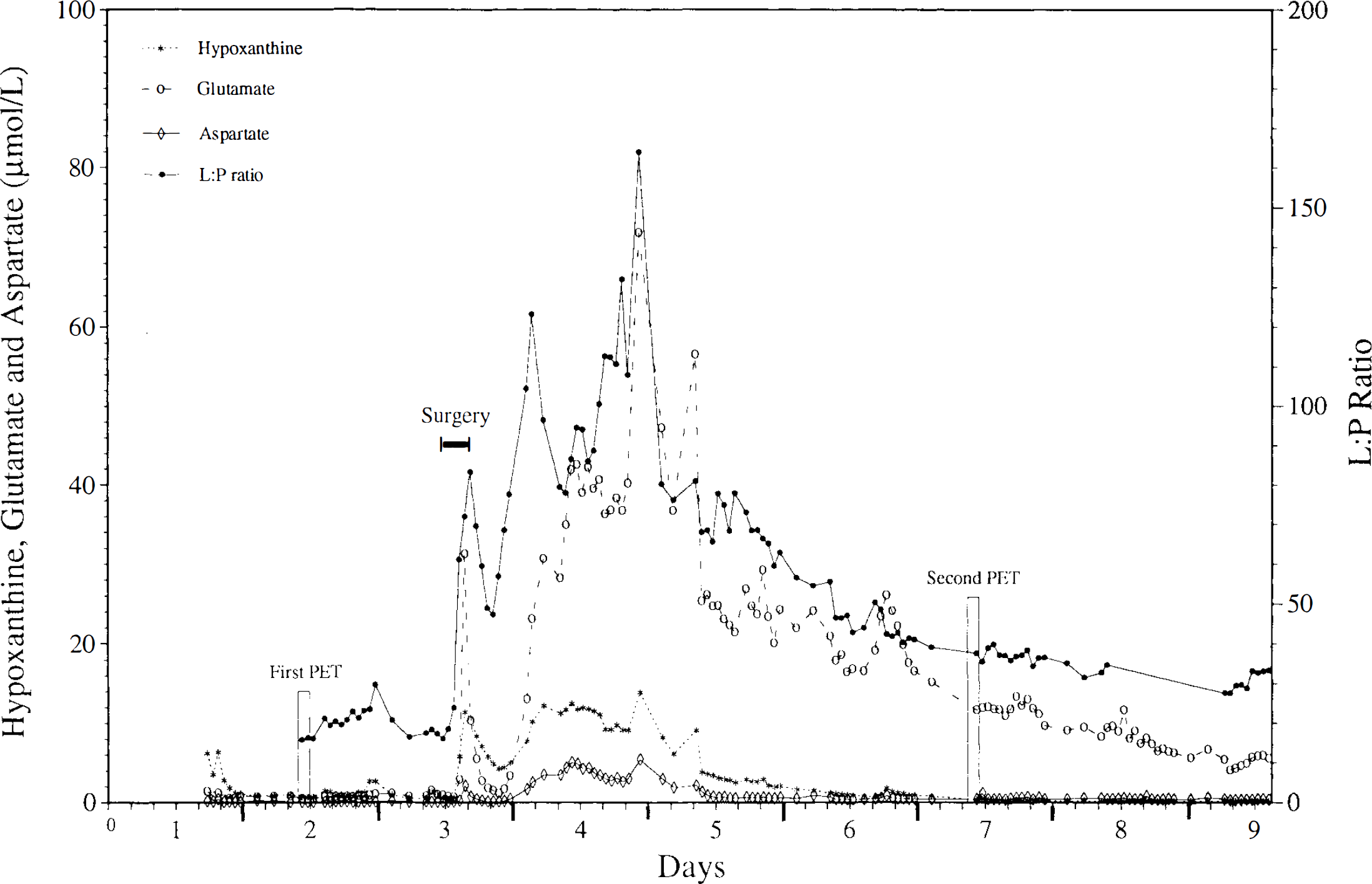
Dialysate metabolite levels over time in Case 6. PET, positron emission tomography; L:P, lactate/pyruvate.
DISCUSSION
MD is a new method of measuring virtually any low molecular weight substance in the ECF of almost any organ. The MD principle was described by Bito et al. (1966) and Delgado et al. (1972) and has been used in experimental studies of many tissues including the brain (Bito et al., 1966; Delgado et al., 1972; see Robinson and Justice, 1991). The first reported study in humans was published in 1987 by Lönnroth et al., who investigated ECF glucose levels in human adipose tissue. The first experiences of MD in the human brain were reported in 1990 (Hillered et al., 1990; Meyerson et al., 1990). In the present study, PET was used to further validate the MD technique with the ultimate goal of developing a tool for chemical monitoring of the brain in NIC patients. Interest was focused on energy metabolism, and it was thus possible to compare directly the energy-related substances retrieved by MD with the presence or absence of ischemia on PET. This is, to our knowledge, the first human study where levels of such substances have been directly related to the current hemodynamic and metabolic state of the brain.
One important practical conclusion of this study is that it is indeed possible to perform MD simultaneously with PET in these severely ill patients without any technical problems or without compromising safety. In such a clinical study, it is impossible to obtain a completely homogeneous patient material, and variations in physiological parameters and levels of consciousness are inevitable. The combination of MD and PET in NIC patients is a complex and demanding task, which restricts the number of patients. However, the results of this study demonstrate a clear agreement between MD and PET in the majority of all measurements.
To overcome interindividual patient variability in the regional analysis of PET results, affected areas were compared with the corresponding contralateral regions according to Heiss and colleagues (1992). Due to the interindividual variation in all whole-brain values, we characterized the regional state of the brain only in terms of ischemia (including infarction) and no ischemia, instead of applying the more detailed description used by Frackowiak et al. (1980). The criteria applied in the regional classification of the state of the brain have also been well substantiated and widely accepted previously (Lassen, 1966; Heiss et al., 1992). Thus, the results of the regional PET interpretations should be relevant as a basis for evaluating the MD results.
To obtain the most accurate whole-brain assessments, a high normal upper reference limit (2 SD above the mean of the reference patients) was used. Further, the diagnosis of whole-brain ischemia was based on alterations in OER, which is less affected than CBF and CMRO2 by confounding factors such as levels of consciousness, Pco2, and sedation because it directly reflects the matching between metabolic demand and oxygen supply.
Lactate and the lactate/pyruvate ratio showed high sensitivity for ischemia, but the specificity of lactate was lower. These findings are in accordance with previous observations that elevated lactate is a rather unspecific finding and may occur in the absence of other signs of ischemia/hypoxia (Persson and Hillered, 1992). Furthermore, no statistical correlation was found between dialysate lactate levels and clinical outcome (Persson et al., 1996). There are other plausible explanations for the observed lactate elevations than nascent ischemia, such as functional activation and low arterial CO2 content. The ECF lactate/pyruvate ratio seems to reflect ischemia more accurately than lactate and is less dependent on methodological limitations (Persson and Hillered, 1992) and has been found to correlate to clinical outcome (Persson et al., 1996).
Hypoxanthine showed a high specificity but a low sensitivity. During energy depletion, the synthesis of ATP stops and the concentration of AMP increases. AMP is degraded to adenosine, inosine, and hypoxanthine, which accumulates owing to a low xanthine oxidase activity in the brain. It is therefore reasonable to assume that pronounced ischemia, or ischemia of prolonged duration, is required to increase the levels of hypoxanthine in the ECF significantly compared with lactate or the lactate/pyruvate ratio, which seem to react more rapidly to ischemia (Persson et al., 1996). This assumption is in good agreement with the higher specificity and lower sensitivity found for hypoxanthine when compared with lactate. The clinical significance of hypoxanthine as a clinical marker of ischemia is underlined by the fact that increased levels correlate with poor clinical outcome (Persson et al., 1996).
Excitotoxicity is a well established mechanism for receptor-mediated neuronal damage where the neurons are destroyed by their own EAAs, i.e., glutamate and aspartate (Olney et al., 1986; Lipton and Rosenberg, 1994). Little is known about the role of excitotoxicity in the human brain. The sensitivity and specificity of glutamate are difficult to predict because increased ECF levels could be caused by several mechanisms, such as transmitter release, inhibited cellular uptake, unspecific leakage from injured cells, or diffusion from the blood through the blood–brain barrier, which may be damaged during the ischemic process. We have hypothesized that diffuse leakage from injured brain tissue or through an impaired blood–brain barrier occurs in severe ischemia/infarction (Persson et al., 1996). The present study showed that glutamate had high sensitivity and low specificity, i.e., behaved similarly to lactate. Tentatively, the low specificity may be attributed to transmitter release and/or inhibited cellular uptake caused by a low grade of ischemia, which could not be identified by the PET criteria used. Aspartate differed somewhat from glutamate by demonstrating intermediate sensitivity as well as specificity. The reason for this is unclear. However, the ECF levels of both glutamate and aspartate have been shown to correlate with clinical outcome (Persson et al., 1996).
CONCLUDING REMARKS
This study demonstrates an agreement between MD and PET, supporting the hypothesis that the ECF substances retrieved by MD can be used as “markers” of ischemia. Intracerebral MD enables the retrieval of these substances in a clinical setting and may thus serve as an instrument for identifying secondary whole-brain ischemia and focal ischemia, provided that the MD probe is located in the ischemic area. PET may be used to define critical ischemic regions (tissue at risk) in which the MD probe can be inserted.
Footnotes
Acknowledgment:
The authors thank Ulla Karlsson, Inger Stjernling, Lena Jansson, and Lena Nalmo for excellent technical assistance. The study was supported by grants from the Swedish Medical Research Council (project nos. 9903 and 7888), the 1987 Foundation for Stroke Research, the Upjohn Company, Selander's Foundation, Astrid Karlsson's Foundation, and Uppsala County Council.
