Abstract
Sex differences have been observed in the outcome after ischemia that are believed to be attributable to sex steroid hormones. The present study investigated the possible benefits of progesterone administration after focal cerebral ischemia. Male mice underwent 60-minute middle cerebral artery occlusion (MCAO) and received progesterone (8 mg/kg, intraperitoneally) or vehicle (dimethyl sulfoxide) 1, 6, and 24 hours after MCAO. The lesion volume at 48 hours after MCAO was significantly reduced (P < 0.05) in progesterone-treated mice compared with vehicle-treated mice. All other mice underwent tests of well being (survival rate and body weight recovery), motor ability (grid test and rotarod), and cognitive ability (water maze) for up to 21 days. MCAO significantly worsened outcome in all of these tests compared with shams. Progesterone treatment was beneficial in that compared with vehicle, it significantly improved survival rate, weight recovery, and motor ability. This improvement was most apparent during water maze testing, where progesterone-treated mice were indistinguishable from shams in terms of acquiring the task. These results indicate beneficial effects of progesterone administration after cerebral ischemia and illustrate the need to further investigate the mechanisms of progesterone action.
Female sex steroid hormones are believed to provide females with an endogenous protection against cerebrovascular events, and it is well documented that premenopausal women have a lower risk of stroke relative to men of the same age (Barrett-Connor and Bush, 1991; Kannel and Thom, 1994). After menopause, the incidence of stroke in women rapidly increases (Wenger et al., 1993), which is coincident with diminished circulating levels of estrogen and progesterone. As a result, hormone replacement therapy (HRT) has been widely used in clinical trials aimed at reducing the occurrence of stroke. However, although the protective role of HRT after cardiovascular disease is well documented (Grodstein and Stampfer, 1995; Grodstein et al., 1996), its role in protection against stroke is less clear.
Some studies have reported a significant reduction in stroke occurrence after HRT (Falkeborn et al., 1993; Finucane et al., 1993; Henderson et al., 1991), whereas many others have reported no significant benefits of HRT (Bushnell et al., 2001; Pederson et al., 1997; Petitti et al., 1998; Viscoli et al., 2001). More recently, the Women's Health Initiative trial, aimed at assessing the effect of HRT on incidence and outcome after stroke, was halted prematurely because increasing health risks outweighing potential benefits (Wassertheil-Smoller et al., 2003). All clinical trials to date have examined either the effects of estrogen alone or estrogen/progesterone-combination HRT. Among trials there is a huge amount of variation in terms of, for example, selection criteria for participants, outcome measures, definition of an HRT user, and examining primary or secondary stroke incidents, as reviewed by Paganini-Hill (2001). Thus, it is difficult to draw overall conclusions with respect to the possible benefits of HRT on the risk of stroke and, as yet, we have no insight into the therapeutic benefit of progesterone-only HRT in reducing stroke risk.
Experimental animal studies have provided much stronger evidence for a neuroprotective role of estrogen after cerebral ischemia (Dubal et al., 1998; Toung et al., 1998). However, as reviewed by Stein (2001), many of these studies have investigated its role in protecting against ischemia injury and, therefore, treatments commenced before injury. Even though preventative treatment would be beneficial, therapies aimed at reducing the damage after stroke are essential. Any treatment concerned with reducing stroke risk should also be applicable in males, who have a risk similar to postmenopausal women. The routine use of estrogen may not be an advisable therapy in males because chronic exposure has undesirable consequences. In premenopausal women, estrogen administration increases the risk of uterine cancer (Grady et al., 1995) and, to counteract this, must be prescribed in combination with progesterone. Thus, estrogen-only treatment would seem applicable in only a subset, albeit significant, of the general population (i.e., postmenopausal women), although the outcome from the estrogen-only arm of the Women's Health Initiative trial is not encouraging (Powledge 2004). Progesterone may be more desirable in terms of clinical practice, and it has been shown to have neuroprotective properties in experimental models of traumatic brain injury (Roof et al., 1996; Shear et al., 2002) and spinal cord injury (Deniselle et al., 2002).
Evidence is beginning to show that progesterone treatment alone is beneficial after global ischemia in cats (Cervantes et al., 2002; Gonzalez-Vidal et al., 1998) and focal ischemia in rats (Murphy et al., 2002). In terms of focal ischemia, these studies have used changes in lesion volume as an indicator that progesterone limits damage after cerebral ischemia for up to 7 days (Chen et al., 1999; Jiang et al., 1996; Murphy et al., 2002). However, lesion volume studies are restricted, and it is important to know whether progesterone administration will have long-lasting functional benefits. Tests of neurologic scoring show that, after cerebral ischemia, progesterone improved outcome at 48 hours (Jiang et al., 1996) and 7 days (Chen et al., 1999). However, no studies have evaluated longer-term functional benefits of progesterone treatment.
The present study was aimed at investigating the beneficial effects of progesterone administration on both motor and cognitive outcome up to 3 weeks after ischemia. To date, no studies with progesterone treatment after cerebral ischemia have been performed in mice. Given the range of genetic variants available, a mouse model should aid our understanding of progesterone's mechanism(s) of action. Male mice were used in the present study, and these have previously been shown to undergo more extensive damage than females after focal cerebral ischemia (Loihl et al., 1999).
MATERIALS AND METHODS
Transient ischemia model
This study was conducted in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 (Project License 40/2206). All mice were adult male C57 BL/6 weighing between 24–32 g at the time of surgery. A total of 44 mice were used in the present study; 1 died during surgery and a further 2 mice were excluded because of inadequate reperfusion. Anesthesia was induced by inhalation of 4% isoflurane (in a NO2/O2 70%/30% mixture) and maintained by inhalation of 1.5% isoflurane. Body temperature was monitored throughout surgery (via a rectal probe) and maintained at 36°C to 38°C using a heating blanket (Harvard Apparatus, Edenbridge, Kent, U.K.). Laser-Doppler flowmetry (Moor Instruments, Sussex, U.K.) was used to monitor cerebral blood flow for 5 minutes before and 5 minutes after middle cerebral artery occlusion (MCAO), and immediately before and after reperfusion. A small incision was made in the skin overlying the temporalis muscle and a 0.7-mm flexible laser-Doppler probe (model P10) was positioned on the superior portion of the temporal bone (6 mm lateral and 2 mm posterior from bregma), secured with Superglue (Loctite). Focal cerebral ischemia was induced by occlusion of the right middle cerebral artery as previously described (Boutin et al., 2002), with the following modifications. A midline incision was made on the ventral surface of the neck, and the right common carotid arteries were isolated and ligated with a 6.0 silk suture. The internal carotid artery and the pterygopalatine artery were temporarily occluded using a microvascular clip (Ohwa Tsusho Co., Tokyo, Japan). As previously described (Boutin et al., 2002), a nylon monofilament (Drennan, Sridgwarer, U.K.: diameter 80 μm) coated with “thermomelting glue” (Bostik, Stafford, U.K.; 2 mm long, 180 μm diameter) was introduced into the intracranial internal carotid artery through an incision in the common carotid artery. The filament was carefully advanced approximately 10 mm distal to the carotid bifurcation, beyond the origin of the middle cerebral artery. Relative cerebral blood flow was monitored for the next 5 minutes and then stopped. The Doppler probe remained attached to the skull while the animals were allowed to recover from anesthesia. During the period of MCAO, drug treatment was randomly assigned (detailed below) and the surgeon was aware of the drug treatment for each animal. After 60 minutes of MCAO, mice were reanesthetized and the occluding filament was withdrawn gently back into the common carotid artery to allow reperfusion to take place. Relative cerebral blood flow was monitored for an additional 5 minutes before the wound was sutured and mice were allowed to recover from the anesthesia. Sham-operated mice underwent the same surgical procedure, except that the filament was not advanced far enough to occlude the middle cerebral artery.
Drug treatment
Mice subjected to MCAO were randomly assigned to receive either progesterone or vehicle treatment. The progesterone group (n = 18) received progesterone (USP; Sigma, St. Louis, MO, U.S.A.) dissolved in dimethyl sulfoxide (16 mg/mL; Sigma), which was injected intraperitoneally in the amount of 8 mg/kg at the onset of reperfusion (i.e., 1 hour after MCAO). Additional injections of progesterone (all 8 mg/kg) were administered at 6 and 24 hours after MCAO. Mice in the vehicle group (n = 20) underwent the same experimental protocol, except that they received the same volume/weight of vehicle only.
Lesion volume
Mice were randomly assigned either to undergo behavioral testing, as detailed below, or to be used for calculation of lesion volume. The experimenter was blinded to the treatment the mice had received before lesion volume analysis and behavioral testing. Mice selected for lesion volume analysis were killed at either 24 hours (progesterone n = 4, vehicle n = 4) or 48 hours (progesterone n = 6, vehicle n = 6) after MCAO. After cervical dislocation, brains were removed and sectioned into 10 × 1-mm coronal slices using a mouse matrix (ASI Instruments, Warren, NJ, U.S.A.). To quantitate ischemic damage, slices were stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC; Sigma) in saline for 30 minutes at room temperature in the dark. They were then stored at 4°C in 10% formalin before analysis. TTC is a marker for mitochondrial function and has been shown to be a reliable indicator of ischemic areas for up to 3 days after ischemia (Bederson et al., 1986; Lin et al., 1993). Digital photographs of all stained slices were taken and the unstained area of infarction was measured on the posterior surface of each coronal section using Scion Image Software. Infarct areas were calculated as previously described (Loihl et al., 1999), which uses an indirect method whereby overestimation of the infarct area due to the contribution of edema is avoided.
Assessment of general animal well-being
Mice assigned to undergo behavioral testing (motor and cognitive) were weighed every day for 7 days after surgery, and also on day 14, as an indicator of their general well-being. Body weights are presented as a percentage change compared with values recorded on the day of surgery. Survival rates of progesterone- and vehicle-treated animals are also presented as a percentage compared with the number of animals undergoing surgery.
Rotarod test
An accelerating rotarod (Letica Scientific Instruments, Barcelona, Spain), adapted from use in rats (Hamm et al., 1994), was used to test the motor function of mice. Mice were placed on the rungs (diameter 29 mm) of the rotarod, which accelerated in speed from 4 revolutions/min to 40 revolutions/min over a 5-minute period. A trial ended if the mouse fell off the rungs, or gripped and spun around for one complete revolution. Mice were acclimatized to the rotarod for five trials each day for 2 days before surgery. On the day of surgery they underwent five trials, the mean of which was used as the presurgery control value for each animal. After surgery, each mouse underwent testing every day for up to 7 days. On each day of testing mice underwent five trials on the rotarod, with an intertrial interval of 1 minute. The data are expressed as the percentage of mean duration per day compared with the presurgery control value.
Foot fault test
Every day for up to 7 days after surgery mice were placed on an elevated grid surface (30 × 35 × 31 cm). The grid openings were 2.5 cm2. During locomotion on the grid, the number of foot faults made by the ipsilateral and contralateral limbs was counted. Each test consisted of three trials lasting 1 minute each, with an intertrial interval of 1 minute. The foot faults are expressed as the number of errors made by the contralateral limbs as a percentage of the total errors made.
Cognitive test: Morris water maze
Spatial reference memory was assessed using the Morris water maze (MWM) (Morris, 1984). A circular tank of 138 cm diameter was filled with opacified water at 21°C (± 1°C) to a depth of 33 cm. The task for the mouse was to escape from the water by locating a hidden platform. A square (10 × 10 cm) Plexiglas platform submerged 1.2 cm below water level was left in the same location (north, east, south, or west, 31 cm from edge) for the duration of each trial. Each mouse was randomly assigned a platform location, which remained constant throughout the duration of testing. Extravisual cues were placed around the room and the lighting was kept constant during testing. Mice were placed into the maze on postsurgery day 14 and underwent two acclimatization trials, whereby they were allowed to swim freely in the maze, without platform, for 90 seconds. After the acclimatization trials, mice were tested each day for 6 consecutive days. Each day consisted of two testing sessions, which were made up of four trials. For each of the four trials mice were placed in the pool facing the wall at one of four starting locations (northeast, southeast, southwest, northwest) and allowed to swim for a maximum of 90 seconds. If the platform was found, the mice were allowed to remain on it for 30 seconds; mice not finding the platform were placed onto it and allowed to remain there for 30 seconds. Intertrial intervals were kept constant at 30 minutes, and a 2-hour interval was permitted between each session. Each trial was videotaped via a ceiling-mounted video camera and the animal's movement tracked using EthoVision software (Noldus Information Technology, Wageningen, The Netherlands), which allows the calculation of various measures such as latency and swim speed. From the data collected over the first 11 sessions it was possible to calculate the latency (in seconds) taken to reach the submerged platform for each day. On the 12th session, mice underwent the “probe trial,” whereby the platform was removed and the mouse placed into the core of the pool where it was allowed to swim freely for 30 seconds. Swim speeds (cm/s) were calculated from the probe trial, along with a measure aimed at investigating how well the task had been learned; i.e., time spent in the goal quadrant (quadrant of pool where platform was previously).
Histology
At 21 days after ischemia, experiments were terminated by administering a lethal overdose of sodium pentobarbitone intraperitoneally. Mice were then transcardially perfused using phosphate-buffered saline followed by buffered fixative (1 mL/g body weight) consisting of 4% paraformaldehyde and 0.2% picric acid in 0.1-mol/L sodium phosphate buffer (pH 7.4). Brains were removed and postfixed in the same solution for 2 hours before being placed into sucrose solution (30% in phosphate-buffered saline) overnight. Sections of brain (30 μm) were cut on a cryostat, mounted onto APES-coated slides, and Nissl stained. Sections were then dehydrated, cleared, and coverslipped in Entellan (Merck & Co., West Point, PA, U.S.A.), and digital photographs were taken.
Statistical analysis
The data are expressed as mean ± SD. All experiments conducted over a series of days (indicators of general well being, motor and cognitive tests) were analyzed by two-way analysis of variance. Data from the probe trial were analyzed using one-way analysis of variance to identify differences according to treatment. Table 1 documents the number of animals in each group at each time point. Post hoc analyses were carried out with the Bonferroni test. Lesion volume data were analyzed at each time point (24 and 48 hours) using the Student's t-test. All data were analyzed using GraphPad Prism Version 3.00 for Windows (GraphPad Software, San Diego, CA, U.S.A.). The criterion for statistical significance was set at P < 0.05.
Number of mice in each experimental group used for behavioral testing that underwent surgery and survived at each time point
RESULTS
Cerebral blood flow
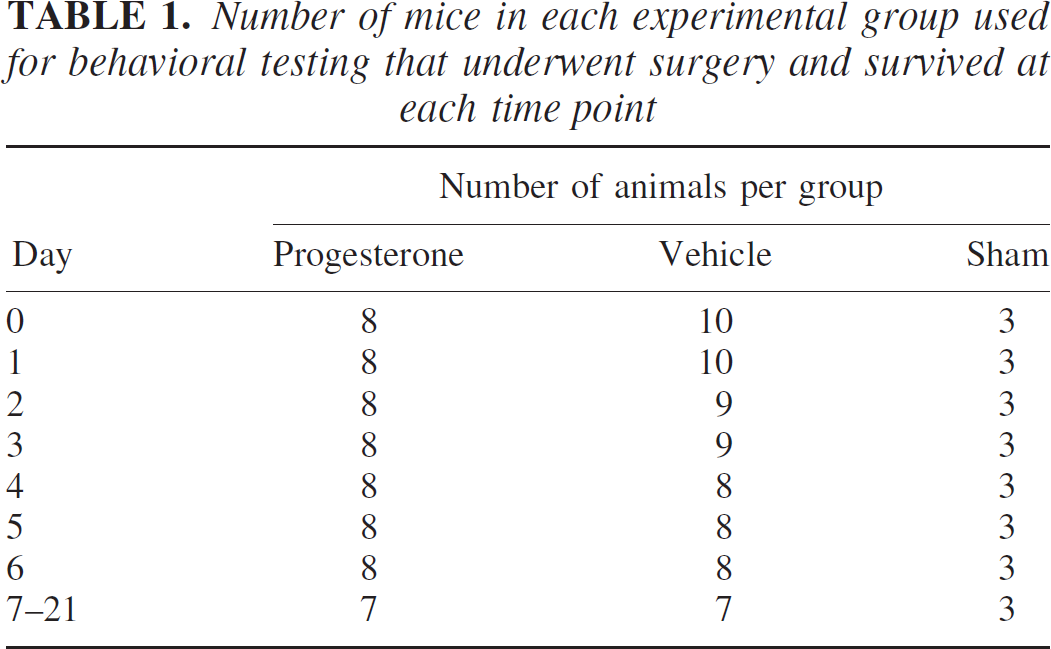
Doppler monitoring showed that, in all mice subjected to MCAO, relative CBF was reduced to 22.8% (± 6.1%) and 18.5% (± 6.7%) of preischemic values within 5 minutes of advancing the filament and induction of MCAO (Fig. 1A) in mice that subsequently received progesterone or vehicle treatment, respectively. After 60 minutes of MCAO and withdrawal of the filament, relative CBF was increased to at least 50% of preischemic values in order for the mice to be included in the study. There were no significant differences in the increase in relative CBF after withdrawal of the filament in the progesterone-treated group (98.6% ± 31.0%) compared with the vehicle-treated group (91.4% ± 16.5%), at least for the 5 minutes after withdrawal of the filament (Fig. 1B).
Laser-Doppler measurement of relative CBF in the right middle cerebral territory for all MCAO mice used in the study (n = 38). Relative CBF was reduced to 22.8% (± 6.1%) and 18.5% (± 6.7%) within 5 minutes of MCAO in mice that subsequently received progesterone or vehicle treatment (
Lesion volume
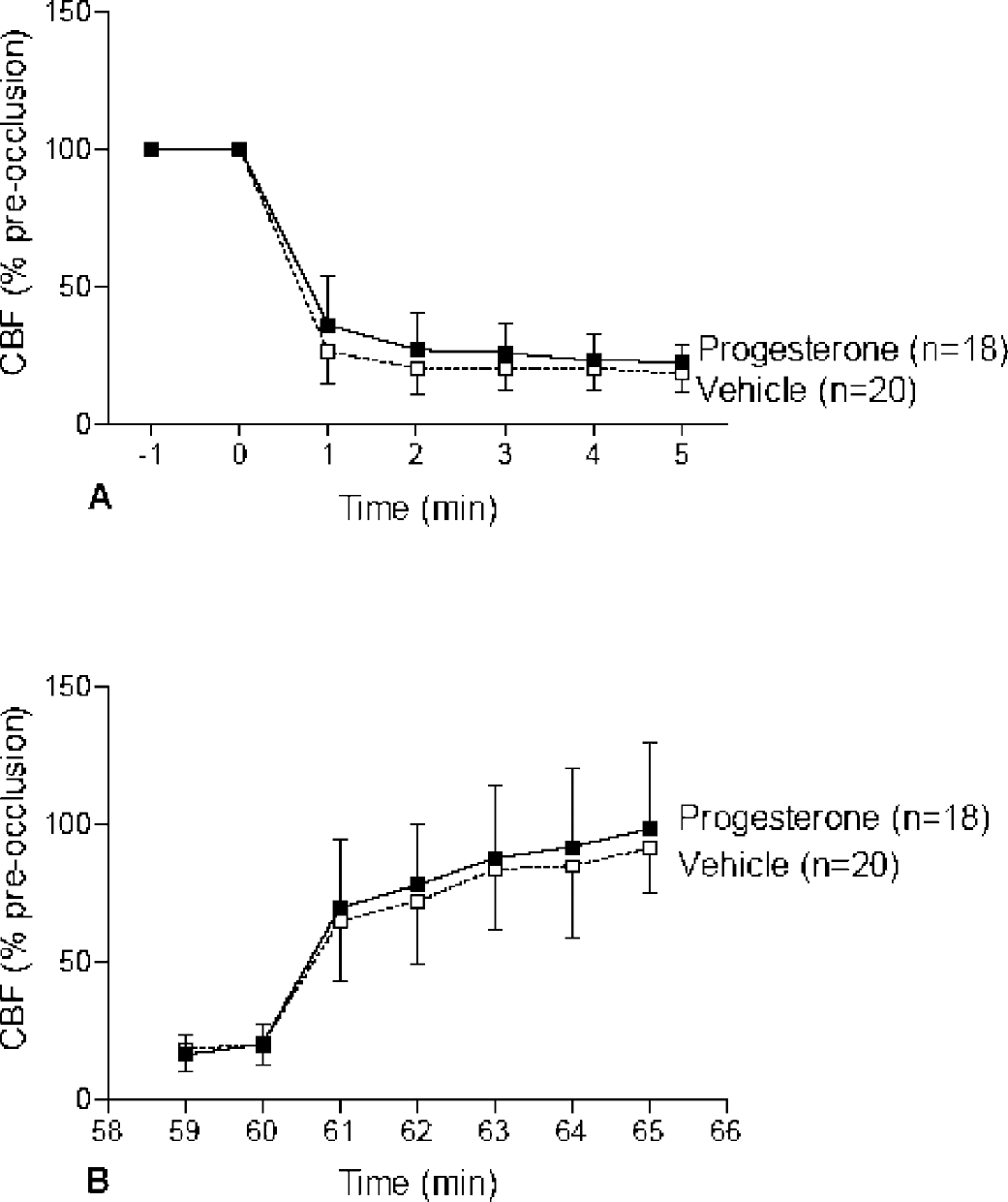
Figure 2 shows a representative brain slice stained with TTC 48 hours after MCAO in vehicle-treated (Fig. 2A) and progesterone-treated (Fig. 2B) mice. The lesion is largely confined to the striatal region of the brain, as indicated by the white area. By day 21 after surgery the lesion can be seen in the striatal area extending into the lateral cortex, as indicated by a lack of Nissl staining (Fig. 2C). Measurements from TTC sections (Fig. 2D) indicated no reduction in lesion volume after progesterone treatment at 24 hours after MCAO (P = 0.5875). However, there was a significant reduction in lesion volume 48 hours after MCAO after progesterone treatment as compared with vehicle (P = 0.0253).
TTC-stained brain slices show that the lesion at 48 hours in both vehicle-treated
Indicators of general “well-being”
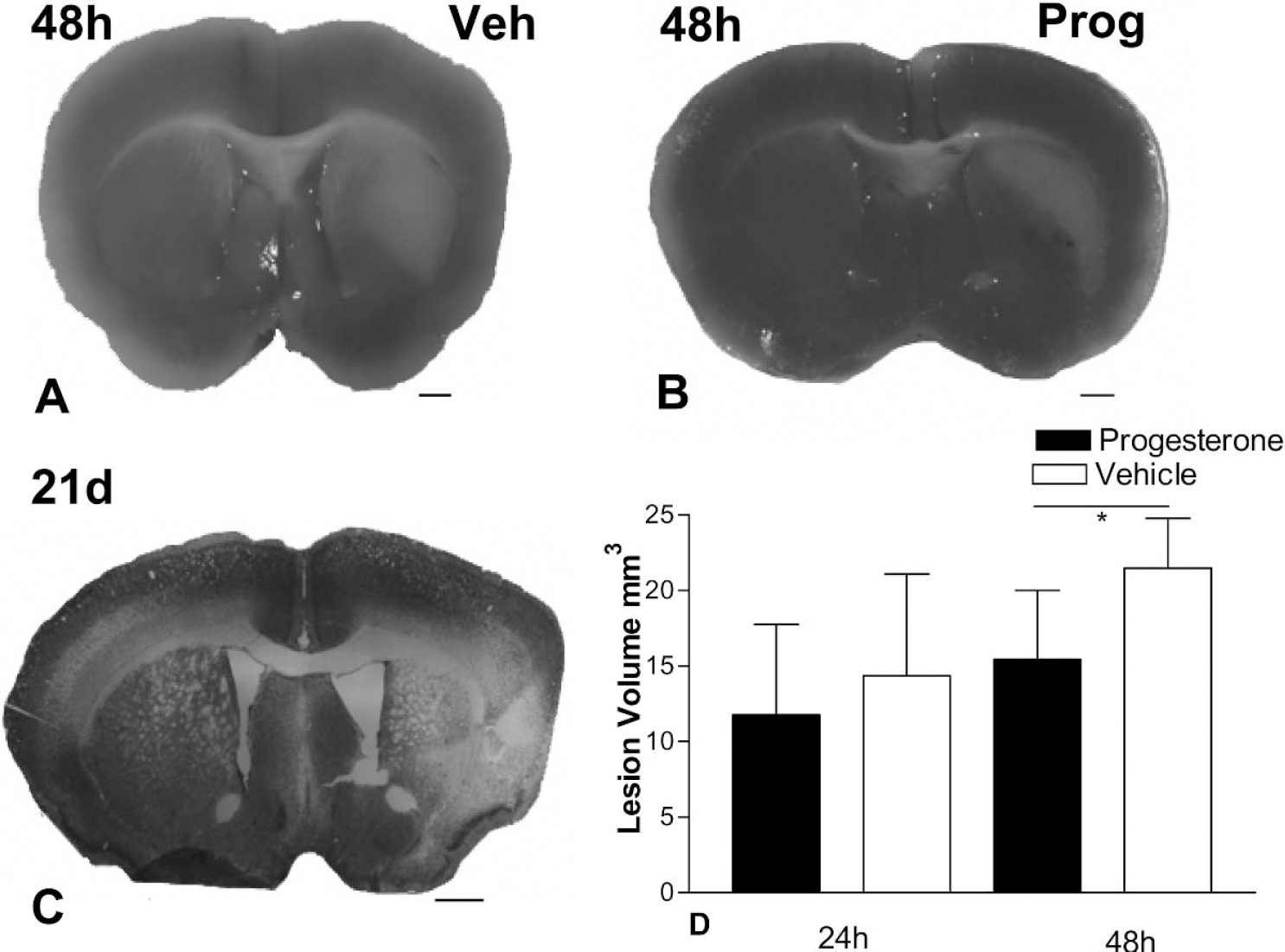
All sham-operated mice survived for the 21 days after surgery, whereas one progesterone-treated animal and three vehicle-treated animals died (Fig. 3A). Progesterone treatment significantly increased the survival rate when compared with vehicle-treated animals (F1,18 = 28.08; P = 0.0005).
As shown in Fig. 3B all experimental groups lost weight over the first 2 days after surgery. Both progesterone-treated and shams gained weight at a similar rate. However vehicle-treated animals gained weight at a significantly slower rate than progesterone-treated animals (F1,18 = 5.006, P = 0.0270).
Survival rates
Motor tasks
Motor function, as assessed using the rotarod, revealed a functional deficit after MCAO regardless of treatment (Fig. 4A). Time spent on the rotarod is expressed as a percentage of presurgery control value, and was significantly decreased in progesterone (F1,11 = 22.76; P < 0.0001) and vehicle-treated animals (F1,13 = 50.01; P < 0.0001) compared with shams. However, progesterone treatment significantly improved the ability to remain on the rotarod as compared with vehicle treatment (F1,18 = 5.294; P = 0.0235).
Unilateral foot faults were expressed by the number of contralateral foot faults as a percentage of the total errors made (Fig. 4B), and a value of 50% represents an equal number of errors made by both sides. Shams revealed no functional deficit because, on average, they made 53.31% (± 4.78%) errors on the contralateral side compared with the ipsilateral side over the testing period. In comparison with shams, after MCAO there was a significant increase in the number of contralateral errors made in the progesterone-treated (66.77% ± 5.35%; F1,11 = 18.57; P < 0.0001) and vehicle-treated (77.5% ± 5.55%; F1,13 = 79.59; P < 0.0001) groups. However, progesterone treatment reduced this functional deficit because it resulted in significantly fewer foot faults as compared with vehicle treatment (F1,18 = 18.26; P < 0.0001) over the first 7 days after surgery.
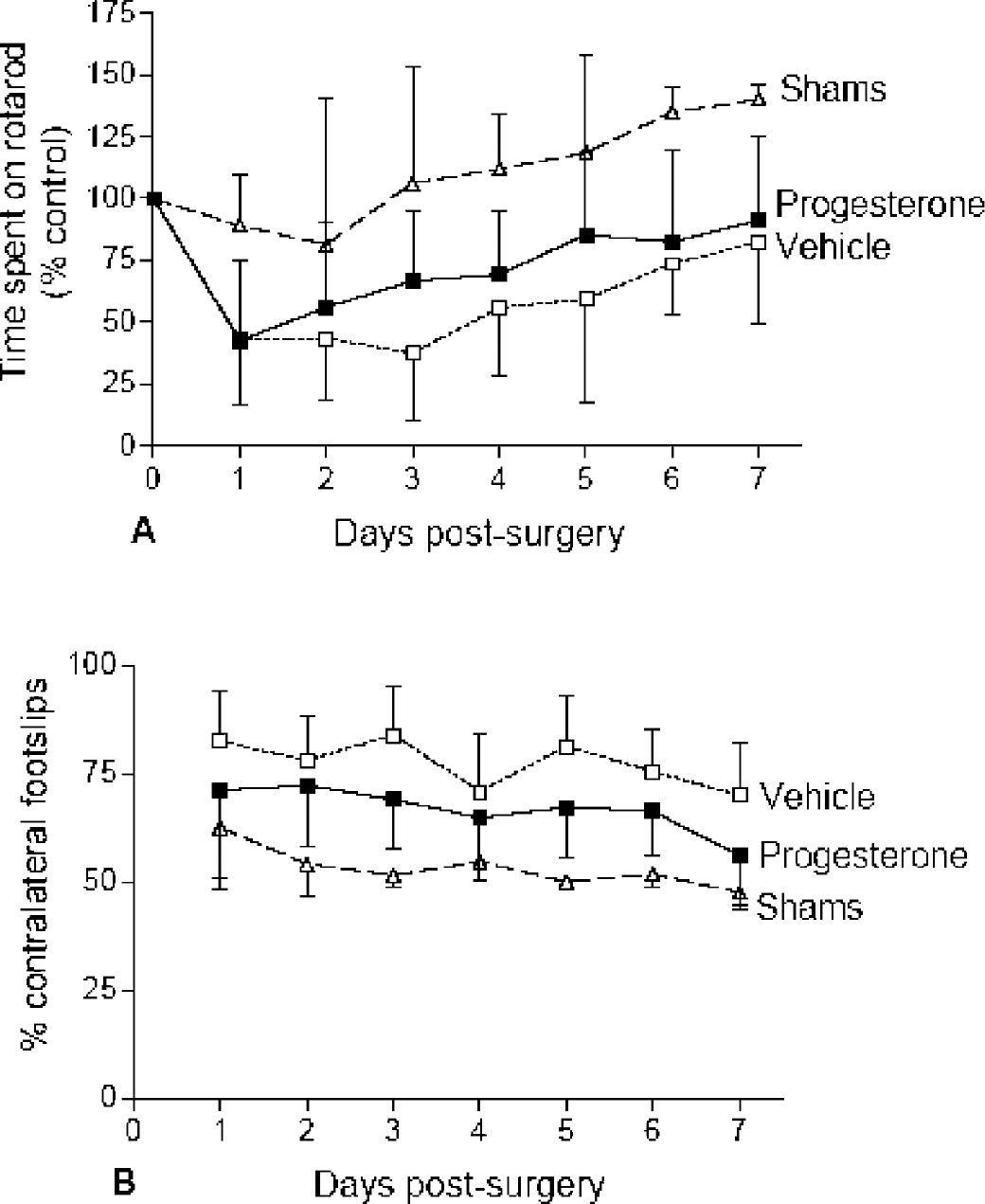
Motor ability as assessed using the rotarod
Cognitive test
Cognitive ability was assessed using the MWM starting at day 15 after surgery. This revealed differences in the ability of mice to acquire the task, hence reducing their latency (i.e., the time taken to find the platform) (Fig. 5A). As expected, all groups exhibited high latencies initially and these reduced as the task was acquired. Shams and progesterone-treated animals did not differ in their ability to learn the task, and showed a similar rate of reduction in latency (F1,10 = 1.109; P = 0.2979). However, vehicle-treated animals were impaired on this task and showed significantly higher latency compared with shams (F1,8 = 8.257; P = 0.0068) or progesterone-treated animals (F1,12 = 7.878; P = 0.0068).
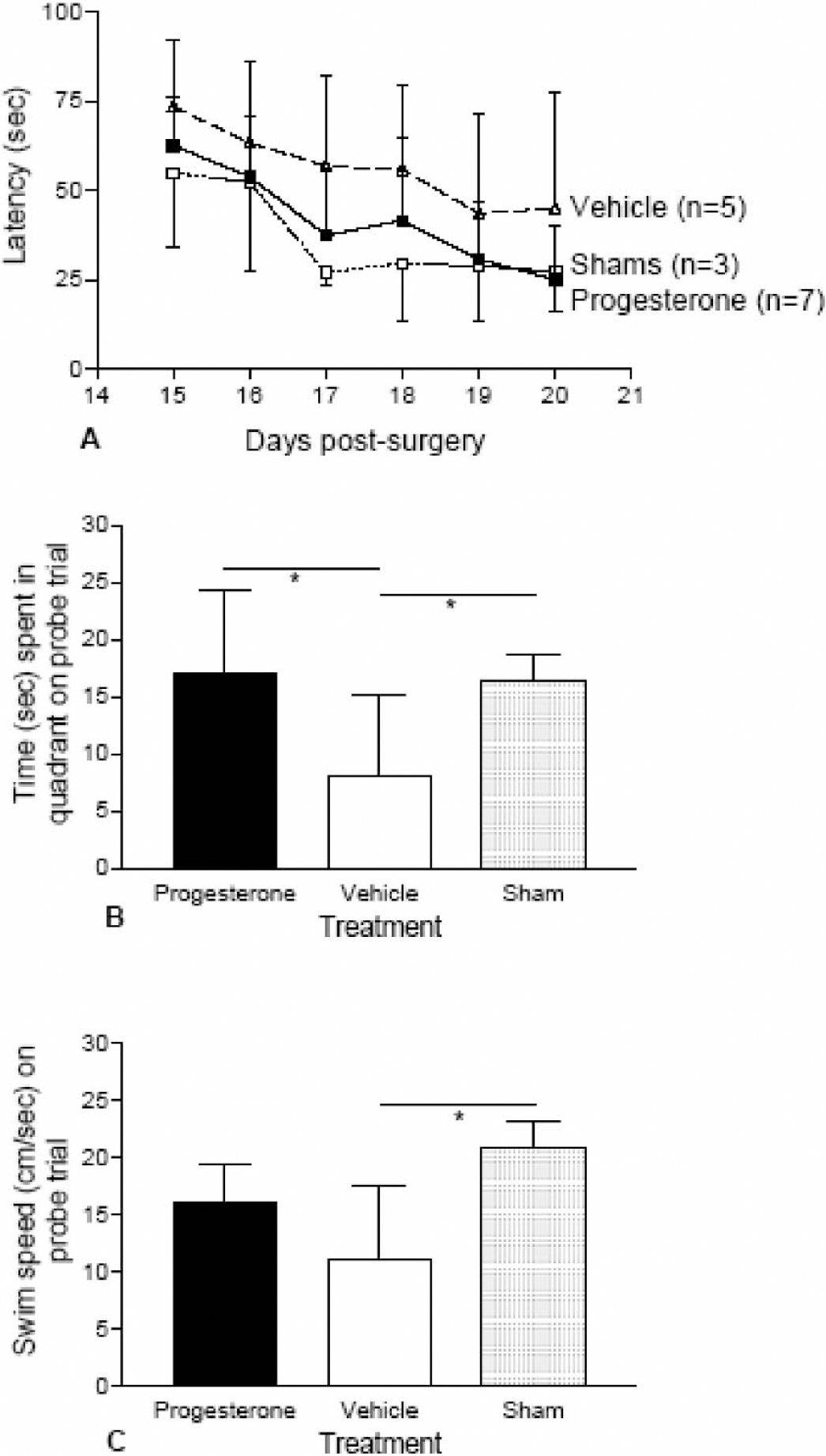
Cognitive performance assessed using the Morris water maze in terms of ability to acquire task and learn platform location (A, escape latency) and ability to recall position of platform on probe trial as indicated by time spent the goal quadrant where the platform had been previously located (B). Progesterone treatment after MCAO improved ability to acquire the task as progesterone treated animals showed no significant differences in escape latency (A) as compared with shams whereas vehicle-treated animals were significantly impaired on acquiring the task as demonstrated by higher escape latency in comparison to progesterone treatment, P < 0.01. On the probe trial both progesterone and sham operated animals spent a significantly higher amount of time compared with vehicle treated animals in the goal quadrant (
On the probe trial, treatment had a significant effect on the amount of time animals spent in the goal quadrant (F2,15 = 5.841; P = 0.0169). Shams and progesterone-treated animals appeared to have learned the task and remembered the platform location better than the vehicle-treated animals. This was evidenced by the finding that both these groups spent a significantly longer period of time in the goal quadrant (Fig. 5B) in comparison with the vehicle group (P < 0.05). Swim speed was also measured on the probe trial (Fig. 5C) and found to be significantly affected by treatment (F2,15 = 4.416; P = 0.0391), with vehicle-treated animals showing significantly slower swim speeds compared with shams (P < 0.05). Although this may indicate a deficit in motor function as compared with cognitive function, it could not account for the improved performance of progesterone-treated compared with vehicle-treated mice because there were no differences in swim speed between these two groups (Fig. 5C).
DISCUSSION
This study shows that progesterone treatment after 60-minute MCAO significantly reduced the size of lesion 48 hours after ischemia. In addition, progesterone treatment reduced a variety of functional deficits in mice. This effect was most apparent in the water maze, a test of cognitive function, where progesterone-treated mice acquired the task just as well as shams.
To date, few studies have concentrated on progesterone administration after ischemia. Of these, even fewer have administered progesterone to naive subjects (i.e., males rather than ovariectomized females). In the present study, we administered progesterone in a dose of 8.0 mg/kg, which has been shown to reduce lesion volume after cerebral ischemia (Chen et al., 1999; Kumon et al., 2000), whereas lower (4 mg/kg) or higher (32 mg/kg) doses failed to have the same effect (Chen et al., 1999). Previous studies also vary with respect to using progesterone in dimethyl sulfoxide for intraperitoneal injection, or water-soluble progesterone for intravenous injection. Jiang et al. (1996) reported that dimethyl sulfoxide—soluble progesterone had significant benefits on lesion volume that were not observed when water-soluble progesterone was administered. This observation, in conjunction with potential difficulties of administering drugs intravenously to freely moving mice, led us to use the route of intraperitoneal administration of dimethyl sulfoxide—soluble progesterone, which has been used successfully in other studies (Jiang et al., 1996; Kumon et al., 2000). A long-term goal of this research is to investigate the possibility of progesterone as a therapy for reducing damage after stroke. Thus, it was considered clinically relevant to initiate progesterone treatment after the ischemic event rather than before it. After the initial progesterone treatment at the onset of reperfusion, subsequent progesterone was administered 6 and 24 hours after MCAO. The inclusion of the two latter time points has been previously reported to have a significant effect on lesion volume after MCAO in male rats (Kumon et al., 2000). Thus, the present study adopted a dosing regimen previously shown to be effective in reducing lesion volume in male rats after MCAO, although no previous studies have administered progesterone to male mice for comparison.
Although we found that progesterone administration had no significant effect on lesion volume when measured 24 hours after ischemia, it did significantly reduce lesion volume 48 hours after ischemia. The reason for this delayed effect may be due to the cumulative effect of three (48 hours) versus only two (24 hours) progesterone injections. However, lesion volume was not measured beyond 48 hours, and so it is impossible to say whether progesterone treatment resulted in a permanent reduction in lesion volume or whether it simply delayed the development of the lesion. From a clinical point of view, it will be interesting to see how long the initial progesterone treatment could be delayed for the treatment still to be effective. Previous work in a trauma model (Roof et al., 1996) has shown that progesterone is still as effective at reducing edema when the initial injection was delayed until 24 hours after the traumatic insult.
Lesion volume calculation is a limited way of investigating whether a treatment is beneficial. Functional outcome, in conjunction with histopathologic outcome, is as important in terms of assessing benefit. Although lesion volume analysis is useful, interpretation of changes in lesion size must be interpreted with caution when investigating potential therapies, because they give little indication of how affected an individual is after stroke. Whereas some studies argue that infarct size correlates well with certain neurologic deficits (Rogers et al., 1997), this is certainly not always the case (Hattori et al., 2000; Reglodi et al., 2003; Wahl et al., 1992). In the present study, we chose to focus on indicators of benefit to mice from progesterone treatment in terms of well-being, motor function, and cognitive function over a longer time period than has been previously reported.
The use of MCAO as a model of focal cerebral ischemia was first described in rats (Tamura et al., 1981) and later adapted for use in mice (Hara et al., 1996). However, much less is known about the functional consequences of experimental stroke in mice compared with rats. Studies also vary with respect to length of ischemia, although 60 minutes appears to be a commonly used duration for mice. We have found that shorter ischemic periods produced infarcts that were less clear, whereas longer ischemic periods were associated with poor survival rates. In the present study, all groups of animals, including shams, displayed weight loss in the first 72 hours after surgery. Shams regained weight at the fastest rate, but all MCAO mice had gained weight by 6 days after ischemia. These body weight changes are similar to other reports after MCAO (Reglodi et al., 2003), and weight loss after sham operation has also been previously reported (Barone et al., 1992). However, we found both survival rate and body weight gain to be significantly improved after progesterone treatment. Progesterone treatment significantly improved the survival rate, although this benefit was not complete because one mouse in the progesterone group died 1 week after ischemia. After 60 minutes MCAO rats have been shown to display significant motor impairments on the rotarod and grid test (Rogers et al., 1997) when compared with shams, a finding replicated in mice in the present study. However, motor deficits on both of these tests were significantly reduced in progesterone-treated mice.
The MWM is a commonly used test of cognitive function in both rats and mice. Mice have been shown to successfully acquire the task, and mice of the C57 BL/6 strain are particularly recommended for the MWM (D'Hooge and De Deyn, 2001) and perform better when compared with other inbred strains (Upchurch and Wehner, 1988). Most studies so far have examined the role of a few well-defined brain regions in MWM learning, and specific deficits are found in animals with damage to hippocampus, striatum, basal forebrain, cerebellum, and several neocortical areas (D'Hooge and De Deyn, 2001). In the present study, we found significant impairments after MCAO in MWM learning, both in terms of time taken to find the hidden platform over the testing period and amount of time spent in the goal quadrant on the probe trial. Progesterone treatment, however, significantly reduced these deficits such that progesterone-treated mice were not significantly impaired compared with shams. The probe trial is designed to test the memory formed over the testing period, and vehicle-treated mice were significantly impaired at this compared with shams or progesterone-treated mice. A motor deficit was still apparent in vehicle-treated mice because on the probe trial they displayed slower swim speed than shams. However, there were no differences in swim speed between progesterone- and vehicle-treated mice; thus, this could not account for the difference in the ability of these two groups to “remember” the platform location on the probe trial.
The present study has shown that progesterone treatment has significant functional benefits to mice that have undergone MCAO, but the mechanism(s) of progesterone action remain unanswered. In studies using models of traumatic brain injury, progesterone has been shown to limit damage by reducing edema formation (Roof et al., 1993, 1994, 1996). Although this is an important component of the cell damage after traumatic brain injury (Beaumont et al., 2000), it has yet to be shown that progesterone acts by reducing edema after cerebral ischemia. A significant proportion of the damage present after cerebral ischemia occurs as a direct consequence of the production of free radicals, leading to oxidative stress (Ginsberg et al., 1988). Progesterone can act as a free radical scavenger and may limit cell damage by reducing peroxidative damage (Roof et al., 1997). Progesterone reduces injury-induced lipid peroxidation in traumatic brain injury models through a membrane-stabilizing type of antioxidant action (Roof et al., 1997). After cerebral ischemia, inflammatory responses are generated at the site of damage, some of which contribute to cell death (Leker and Shohami, 2002). Progesterone may limit this by inhibiting the production of pro-inflammatory cytokines, such as tumor necrosis factor-α (Goodman et al., 1996). After cerebral ischemia, cell death occurs as a direct result of excitotoxicity (Graham et al., 1993; Katayama et al., 1994). Progesterone has been shown to be capable of both enhancing GABAergic neurotransmission (Majewska, 1992) and reducing excitatory amino acid release (Smith et al., 1987; Smith, 1991). Thus, progesterone may limit excitotoxic cell death by enhancing inhibitory synaptic neurotransmission and thereby reducing neuronal excitability (Smith, 1991), excitatory amino acid release, and subsequent excitatory neurotransmission (Goodman et al., 1996). Other possible mechanisms of action by progesterone include increasing blood supply to the ischemic area, because progesterone and its metabolites have been shown to possess vasodilator properties (Molinara et al., 2001; Perusquia et al., 1996). The potential mechanisms of progesterone action appear to be numerous, and it is likely that progesterone acts by a combination of these mechanisms.
Neurogenesis has been shown to occur in discrete regions after cerebral ischemia (Arvidsson et al., 2002; Jiang et al., 2001; Kokaia and Lindvall, 2003). It will be interesting to see if neurogenesis contributes to any of the observed functional recovery in the present study and, if so, whether this is amplified in progesterone-treated animals. Neurogenesis is regulated by steroid hormones in the dentate gyrus, and it is maximal when monthly fluctuating levels of estrogen are at their highest (Tanapat et al., 1999), and estrogen has been shown to directly promote neurogenesis in other studies (Cameron and Gould, 1994; Cameron et al., 1998; Perez-Martin et al., 2003). The effects, if any, of progesterone on neurogenesis await investigation.
Our study has presented, for the first time in male mice, histopathologic evidence that progesterone reduces tissue damage after focal cerebral ischemia and, more pertinent, behavioral evidence indicating long-term functional improvement on a variety of tests. Although the mechanisms underlying progesterone action remain speculative, their investigation must be the focus of future research if progesterone therapy for stroke is to advance.
Footnotes
Acknowledgments
The authors wish to thank Herve Boutin for advice regarding MCAO, Nigel Jones for advice regarding behavioral studies, Philip Bath for helpful consultation throughout the study, and the staff of the BMSU (University of Nottingham) for their care of the experimental animals.
