Abstract
Growth factors possess neuroprotective and neurotrophic properties in vitro, but few have been extensively studied in vivo after stroke. In the present study, we investigated the potential functional benefits of granulocyte colony-stimulating factor (G-CSF) administration after focal cerebral ischemia. Male mice underwent 60-minute middle cerebral artery occlusion (MCAO) and received G-CSF (50 μg/kg, subcutaneously) or vehicle (saline) at the onset of reperfusion. Granulocyte colony-stimulating factor-treated mice killed at 48 hours after MCAO revealed a >45% reduction (P<0.05) in lesion volume. In terms of body weight recovery, and in tests of motor (grid test and rotarod) and cognitive ability (water maze), MCAO significantly worsened the outcome in vehicle-treated mice as compared with shams (P<0.05). However, G-CSF treatment was beneficial as, compared with vehicle, this significantly improved weight recovery and motor ability. This effect was most apparent on the water maze where G-CSF-treated mice were indistinguishable from shams in terms of acquiring the task. These results indicate long-term beneficial effects of a single dose of G-CSF administered on reperfusion, and illustrate the need to further investigate the mechanisms of G-CSF action.
Introduction
Growth factors are able to act directly on the CNS to regulate survival, maturation, and sprouting of developing neuronal cells. Almost all growth factors display endogenous neuroprotective and neurotrophic effects on mature neurons (Larsson et al, 1999; Mattson and Scheff, 1994). More interestingly, such beneficial effects have been reported with their exogenous administration after stroke (Semkova and Krieglstein, 1999), including insulin-like growth factor (Schabitz et al, 2001), brain-derived neurotrophic factor (Schabitz et al, 2000), and basic fibroblast growth factor (bFGF; Li and Stephenson, 2002). However, stroke remains one of the major causes of death and such factors have yet to translate into clinical practice. For example, clinical trials of bFGF were discontinued in phase III because of dose-related adverse side effects that were not anticipated from preclinical studies (Bogousslavsky et al, 2002).
One of the few growth factors currently approved for clinical use is the granulocyte colony-stimulating factor (G-CSF), and it is routinely prescribed to treat neutropenia (Metcalf, 1990). Granulocyte colony-stimulating factor is able to mobilize stem cells from the bone marrow into the peripheral circulation (To et al, 1997) and is a common treatment after hematologic disease, or chemotherapy treatment for cancer, when white blood cell counts tend to be dangerously low and there is risk of infection. Endogenous G-CSF is largely secreted from hematopoietic cells, such as monocytes, macrophages, and lymphocytes (de Koning and Touw, 1996; Nicola et al, 1983; Sallerfors, 1994). Once released, G-CSF plays an essential role in the regulation of granulopoiesis and maintenance of neutrophil number in the peripheral blood (de Koning and Touw, 1996), and stimulates proliferation, survival, and maturation of cells committed to the neutrophilic granulocyte lineage (Nicola, 1987). These actions are mediated via binding to a cell surface receptor (de Koning and Touw, 1996; Demetri and Griffin, 1991), leading to the activation of a variety of intracellular signalling cascades (Avalos, 1996). Granulocyte colony-stimulating factor has been shown to have trophic effects on neuronal cells in vitro (Konishi et al, 1993), and a few studies are now beginning to highlight its neuroprotective action after stroke.
After focal cerebral ischemia, G-CSF administration has been shown to result in significant decreases in lesion volume, both in mice (Six et al, 2003) and the rat (Schabitz et al, 2003). However, these studies have only looked at short-term effects, giving little indication of whether lesion development was prevented or simply delayed. In addition, these previous studies give no clue as to any longer-term benefits of G-CSF treatment after stroke. While some argue that infarct size correlates well with certain neurologic deficits (Rogers et al, 1997), this is certainly not always the case (Hattori et al, 2000; Reglodi et al, 2003). Functional outcomes are more useful in terms of assessing the benefit of a treatment after stroke. The present study was aimed at investigating the beneficial effects of G-CSF on both motor and cognitive outcome up to 3 weeks after ischemia.
Materials and methods
Transient Focal Cerebral Ischemia
This study was conducted in accordance with the UK Animals (Scientific Procedures) Act, 1986 (Project Licence 40/2206). All mice were adult male C57 BL/6, weighing 24 to 32 g at the time of surgery. A total of 35 mice were used, one died during surgery and one was excluded because relative cerebral blood flow (CBF) did not meet the criteria for occlusion. Anesthesia was induced by inhalation of 4% isofluorane (in a NO2/O2 70%/30% mixture) and maintained by inhalation of 1.5% isofluorane. Body temperature was monitored throughout surgery (via a rectal probe) and maintained at 37.0°C±0.6°C using a heating blanket (Harvard Apparatus Ltd, UK). Laser Doppler flowmetry (Moor Instruments, UK) was used to monitor relative CBF for 5 mins before and 5 mins after middle cerebral artery occlusion (MCAO), and immediately before and after reperfusion. At the beginning of surgery, a small incision was made in the skin overlying the temporalis muscle, then a 0.7-mm flexible laser Doppler probe (model P10) was positioned on the superior portion of the temporal bone (6 mm lateral and 2 mm posterior from bregma), and secured with glue (Loctite). Focal cerebral ischemia was induced by occlusion of the right middle cerebral artery as described previously (Boutin et al, 2002), with the following modifications. A midline incision was made on the ventral surface of the neck, and the right common carotid arteries were isolated and ligated with a 6.0 silk suture. The internal carotid artery and the pterygopalantine artery were temporarily occluded using a microvascular clip (Ohwa Tsush Co., Ltd, Japan). A nylon monofilament (Drennan, UK; diameter 80 μm) coated with ‘thermomelting glue’ (Bostik; 2 mm long, diameter 180 μm) was introduced into the intracranial internal carotid artery through an incision in the common carotid artery. The filament was carefully advanced approximately 10 mm distal to the carotid bifurcation, beyond the origin of the middle cerebral artery. Relative CBF was monitored for the following 5 mins, after which monitoring stopped and the animals were allowed to recover from anesthesia with the probe remaining attached to the skull. During the 60 mins of MCAO, drug treatment was randomly assigned to each animal, and the surgeon was aware of the treatment each animal received at the onset of reperfusion. After 60 mins of MCAO, mice were reanesthetized, and the occluding filament was withdrawn gently back into the common carotid artery to allow reperfusion to take place. Relative CBF was monitored for a further 5 mins before the wound was sutured and mice allowed to recover from the anesthesia. Sham-operated mice (n=6) underwent the same surgical procedure, except that the filament was not advanced far enough to occlude the middle cerebral artery.
Drug Treatment
Mice subjected to MCAO were then randomly assigned to receive either G-CSF or vehicle treatment. One group (n=12) received G-CSF (Amgen, The Netherlands) dissolved in saline, and injected subcutaneously (50 μg/kg) at the onset of reperfusion, that is, 1 hour after MCAO. Mice in another group (n=15) underwent the same experimental protocol, except that they received vehicle only.
Lesion Volume
Mice were randomly assigned to undergo either behavioral testing as detailed below, or be used for assessment of lesion volume. The experimenter was masked to the drug treatment of the mice for subsequent lesion volume analysis or behavioral experiments. Two vehicle mice died before they could be killed for lesion volume and were thus excluded from the analysis. Mice selected for lesion volume analysis were killed at 48 hours (G-CSF n=5, vehicle n=5) after MCAO. After cervical dislocation, the brains were removed and sectioned into 10 × 1 mm coronal slices using a mouse matrix (ASI Instruments, Houston, TX). To quantitate ischemic damage, the slices were stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC; Sigma, UK) in saline for 30 minutes at room temperature in the dark. They were then stored at 4°C in 10% formalin before analysis. 2,3,5-Triphenyltetrazolium chloride is a marker for mitochondrial function and has been shown to be a reliable indicator of ischemic areas for up to 3 days after ischemia (Bederson et al, 1986; Lin et al, 1993). Digital photographs of all stained slices were taken and the unstained area of infarction was measured on the posterior surface of each coronal section using Scion Image Software. Infarct areas were calculated as described previously (Loihl et al, 1999), using an indirect method whereby overestimation of the infarct area because of the contribution of edema is avoided.
General Well-Being
Mice assigned to undergo behavioral testing (motor and cognitive) were weighed every day for 7 days after surgery, and also on the 14th day after surgery, as an indicator of their general well-being. Body weights are presented as a percentage change compared with values recorded on the day of surgery. Survival rates of G-CSF- and vehicle-treated animals are also presented as a percentage compared with the number of animals undergoing surgery.
Rotarod Test
An accelerating rotarod (Letica Scientific Instruments), adapted from use in rats (Hamm et al, 1994), was used to test the motor function of mice. Mice were placed on the rungs (diameter 29 mm) of the rotarod, which accelerated in speed from 4 to 40 rev/minute over a 5-min period. A trial ended if the mouse fell off the rungs, or gripped and spun around for one complete revolution. Mice were acclimatized to the rotarod for 5 trials each day for 2 days before surgery. On the day of surgery they underwent 5 trials, the mean of which was used as the presurgery control value for each animal. After surgery, each mouse underwent testing every day for up to 7 days. On each day of testing mice underwent five trials on the rotarod, with an intertrial interval of 1 min. The data are expressed as the percentage of mean duration per day compared with the presurgery control value.
Foot Fault Test
Daily, for the 7 days after surgery, mice were placed on an elevated grid surface (30 L × 35 W × 31 H cm). The grid openings were 2.5 cm2. During locomotion on the grid, the number of foot faults made by the ipsilateral and contralateral limbs were counted. Each test consisted of three trials lasting 1 min each, with an intertrial interval of 1 minute. Foot faults are expressed as the number of errors made by the contralateral limbs as a percentage of the total errors made.
Cognitive Test: Morris Water Maze
Spatial reference memory was assessed using the Morris (1984) water maze. A circular tank of 138 cm diameter was filled with opacified water at 21°C (±1°C) to a depth of 33 cm. The task for the mouse was to escape from the water by locating a hidden platform. A square (10 × 10 cm2) Plexiglass platform submerged 1.2 cm below water level was left in the same location (north, east, south or west, 31 cm from the edge) for the duration of each trial. Each mouse was randomly assigned a platform location, which remained constant throughout the duration of testing. Extravisual cues were placed around the room and the lighting was kept constant during testing. Mice were placed into the maze on the 14th day after surgery and underwent two acclimatization trials, whereby they were allowed to swim freely in the maze, without platform, for 90 secs. After the acclimatization trials, mice were tested each day for six consecutive days. Each day consisted of two testing sessions, which were made up of four trials. For each of the four trials, mice were placed in the pool facing the wall at one of four starting locations (north east, south east, south west, north west) and allowed to swim for a maximum of 90 secs. If the mice found the platform, they were allowed to remain on it for 30 secs; mice not finding the platform were placed on it and allowed to remain there for 30 secs. Intertrial intervals were kept constant at 30 mins and a 2-hour interval was permitted between each session. Each trial was videotaped via a ceiling-mounted video camera and the animal's movement tracked using Ethovision software, which allows the calculation of various measures such as latency and swim speed. From the data collected over the first 11 sessions, it was possible to calculate the latency (seconds) taken to reach the submerged platform for each day. On the twelfth session, mice underwent the ‘probe trial,’ whereby the platform was removed and the mouse placed into the center of the pool, where it was allowed to swim freely for 30 secs. Swim speeds (cm/second) were calculated from the probe trial, along with the time spent in the goal quadrant, that is, where the platform was previously located, used as an indicator of how well the task had been learnt.
Statistical Analyses
Survival data were expressed by applying the Kaplan–Meier curve, followed by the Mantel–Haenszel log-rank test to identify differences between the curves. All behavioral data were found to be normally distributed when tested using the Kolmogorov–Smirnov test (P>0.10). All lesion volume and behavioral data are expressed as mean±s.d., and analyzed using parametric tests. Experiments conducted over a series of days (i.e., weight gain, motor and cognitive tests) were analyzed by two-way analysis of variance (ANOVA). Cognitive data from the probe trial were analyzed using one-way ANOVA to identify differences according to treatment. Table 1 gives the number of mice in each group at each time point. Post hoc analyses were performed with Bonferroni's test. Lesion volume data were analyzed using a two-tailed Student's t test. The data were analyzed using GraphPad Prism Version 3.00 for Windows (GraphPad Software, San Diego, CA, USA). The criterion for statistical significance is P<0.05.
Number of mice in each experimental group used for behavioral testing that underwent surgery and survived at each time point
Results
Cerebral Blood Flow Measurements
Doppler monitoring showed that, in all mice subjected to MCAO, relative CBF was reduced to at least 70% of preischemic values within 5 mins of advancing the filament and induction of MCAO. During the occlusion period, mice were randomly selected to receive G-CSF or vehicle treatment. Doppler data revealed no differences in CBF after occlusion between mice that received subsequent G-CSF (16.48%±8.98%) or vehicle treatment (17.97%±4.86%) for the 5 mins after MCAO. After 60 mins of MCAO, and on withdrawal of the filament, relative CBF had to rise to at least 50% of preischemic values for the mice to be included in the study. There were no significant differences in the increase in relative CBF in the G-CSF-treated group (85.12%±28.9%) compared with the vehicle-treated group (86.63%±25.75%), at least for the five mins after withdrawal of the filament.
Effect of Granulocyte Colony-Stimulating Factor on Lesion Volume
Figure 1 shows a representative brain slice stained with TTC 48 hours after MCAO in G-CSF-treated (Figure 1A) and vehicle-treated (Figure 1B) mice. White areas reflect a lack of staining and hence a loss of viable tissue. Measurements from TTC slices indicate a 46% reduction in lesion volume after G-CSF treatment (14.91±3.5 mm3) compared with vehicle treatment (27.66±8.79 mm3, P<0.05, Figure 1C).
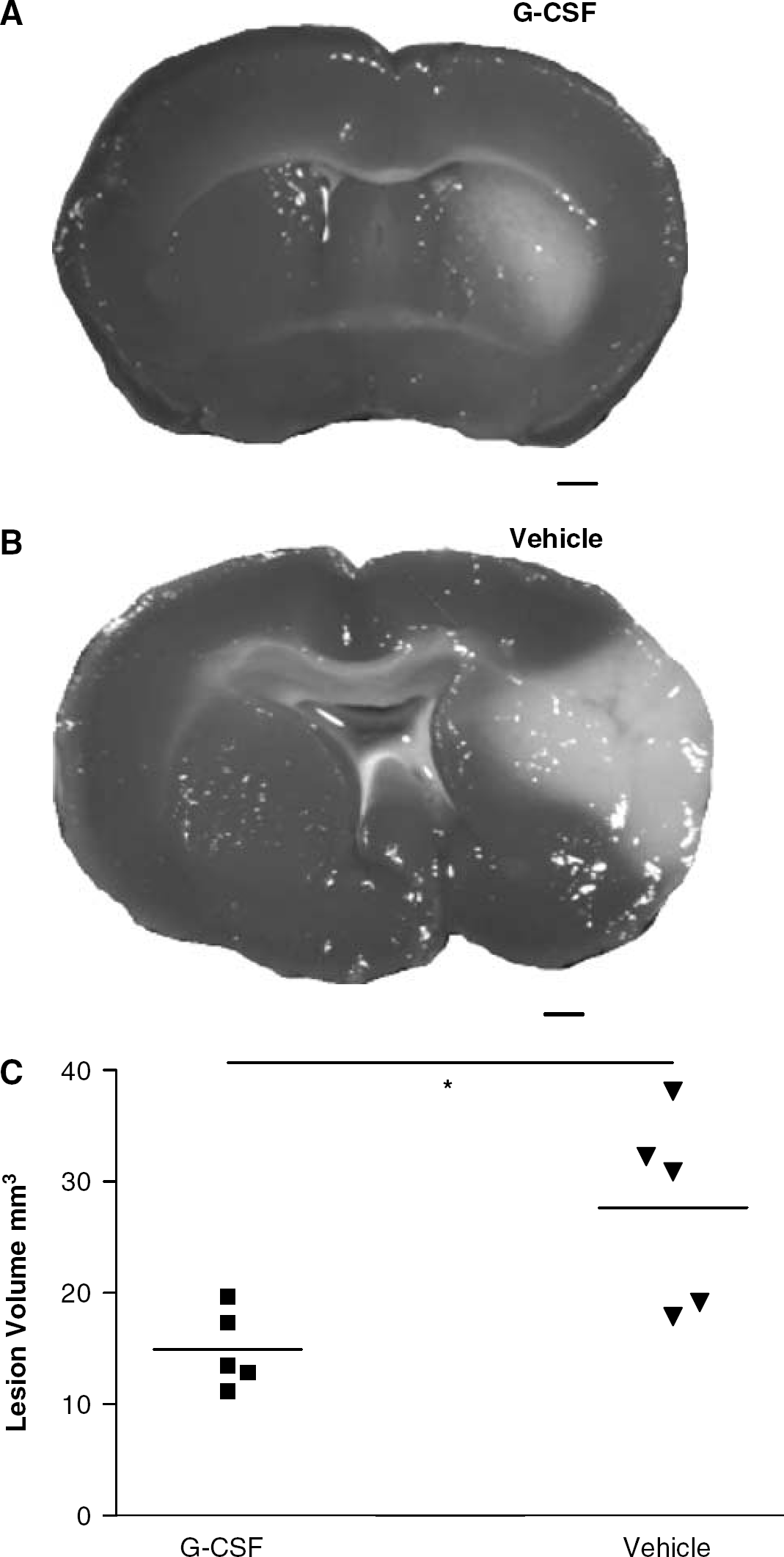
Granulocyte colony-stimulating factor treatment reduces lesion volume. 2,3,5-Triphenyltetrazolium chloride-stained brain slices depict the lesion 48 hours after MCAO in G-CSF- (
Indicators of General ‘Well-Being’
All sham-operated mice survived for the 21 days after surgery, whereas one G-CSF-treated animal and two vehicle-treated animals died (Figure 2A). The survival data are presented by applying the Kaplan–Meier Curve and analyzed using the Mantel–Haenszel log-rank test (χ2=1.772), which revealed there were no statistically significant differences in survival rate between the experimental groups (P=0.418).
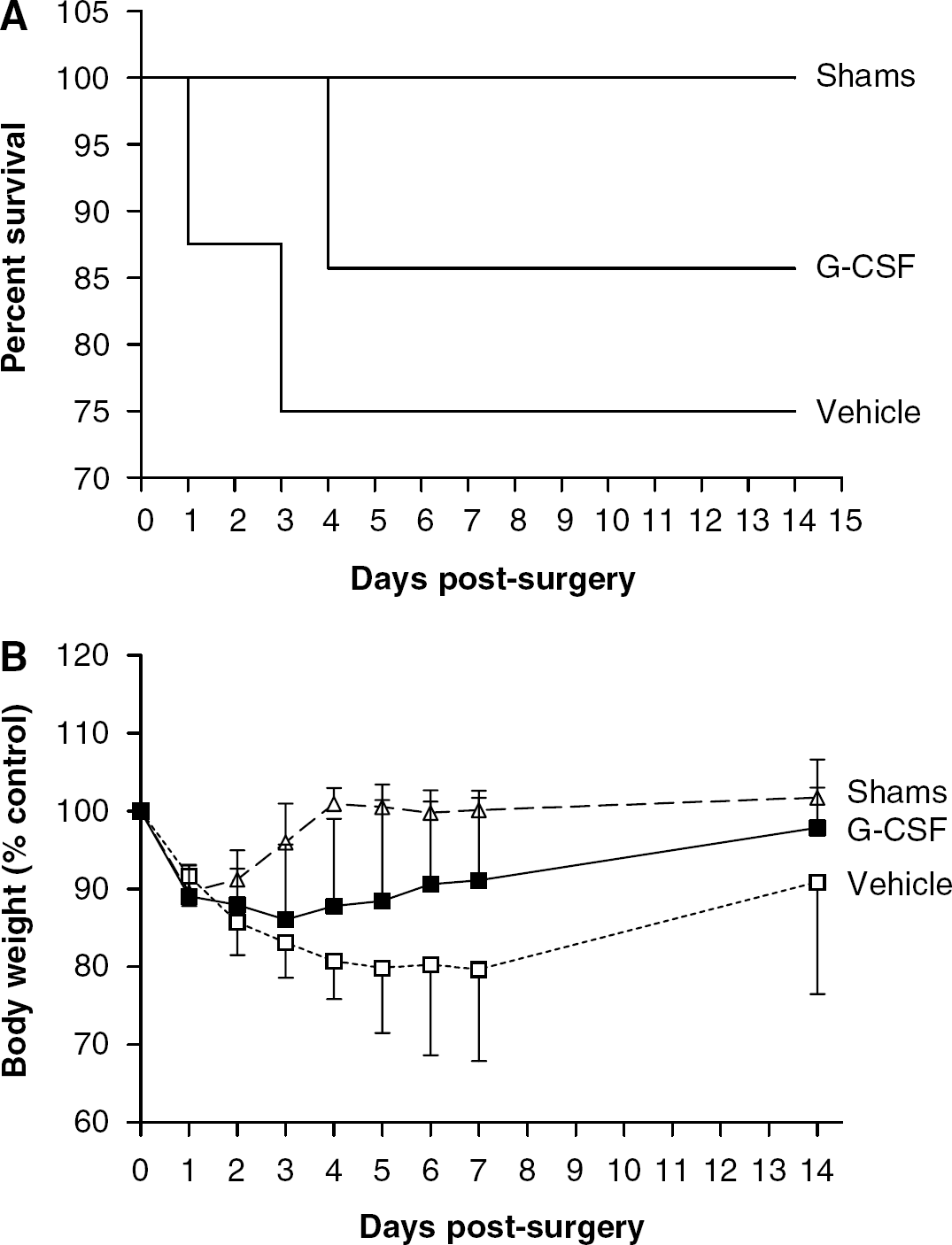
Granulocyte colony-stimulating factor treatment does not affect survival but enhances the recovery of body weight after MCAO. Mortality data were displayed using the Kaplan–Meier Curve (
All experimental groups lost weight over the first 3 days after surgery (Figure 2B). Shams regained weight faster than MCAO animals receiving either G-CSF (F(1,13)=28.39, P<0.0001) or vehicle treatment (F(1,13)=102.4, P<0.0001). However, vehicle-treated animals gained weight at a significantly slower rate than G-CSF-treated animals (F(1,14)=11.31, P=0.0011).
Motor Tasks
Motor function, as assessed using the rotarod, revealed a functional deficit after MCAO regardless of treatment (Figure 3A). Time spent on the rotarod is expressed as a percentage of presurgery control value, and was significantly increased in shams compared with G-CSF- (F(1,13)=30.95, P=<0.0001) or vehicle-treated animals (F(1,13)=73.32, P<0.0001). However, G-CSF treatment significantly improved the ability to remain on the rotarod as compared with vehicle treatment (F(1,14)=9.73, P=0.0025).
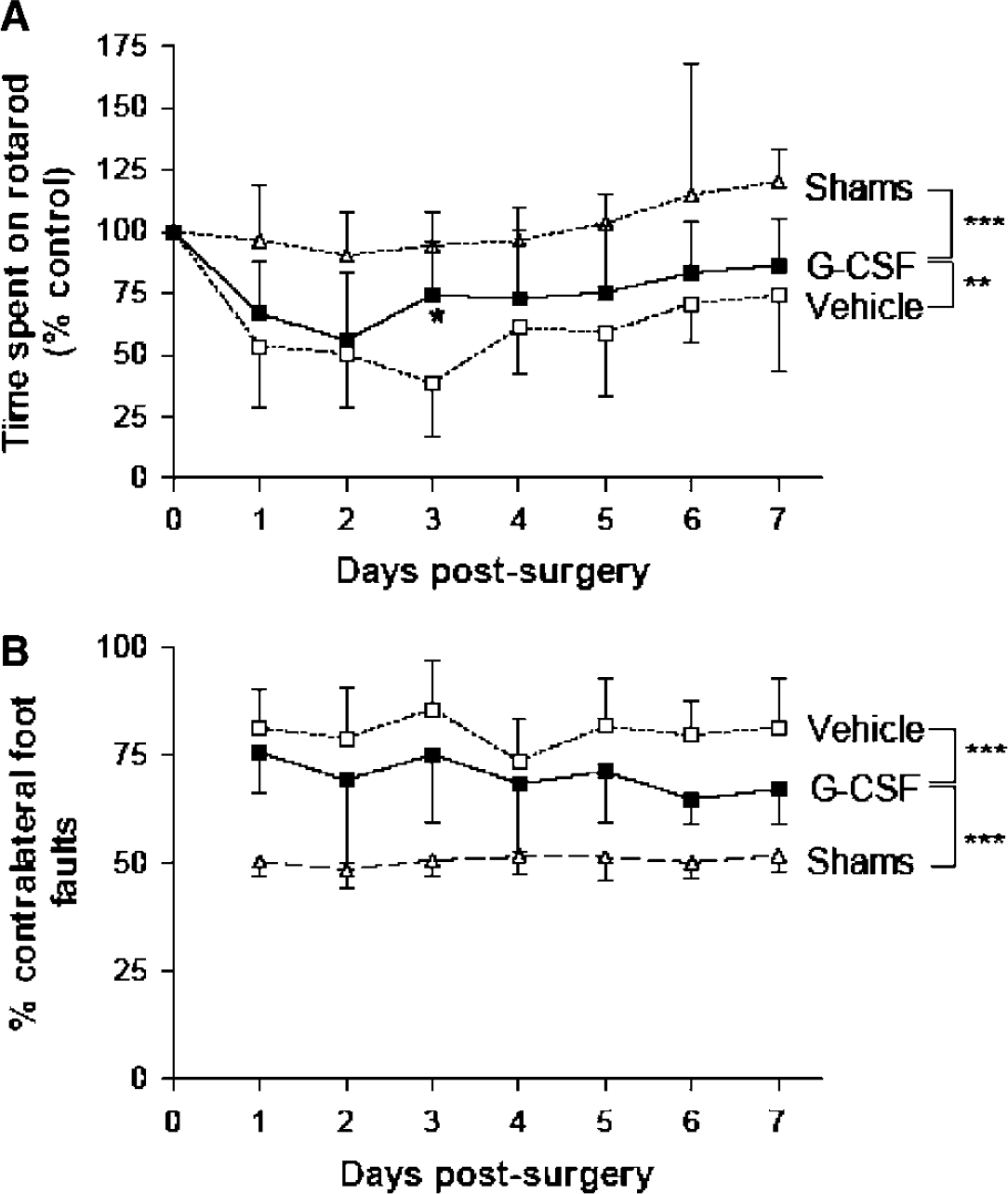
Granulocyte colony-stimulating factor treatment improves motor ability. Granulocyte colony-stimulating factor-treated animals were less impaired with respect to motor function, as they were able to remain on the rotarod for a significantly greater amount of time than vehicle-treated animals as indicated by two-way ANOVA, **P<0.01 (
Unilateral foot faults were expressed by the number of contralateral foot faults as a percentage of the total errors made (Figure 3B), and a value of 50% represents an equal number of errors made by both sides. After sham surgery, no functional deficit was observed as they made approximately the same number of errors on the contralateral side as they did on the ipsilateral side. After MCAO, there was a significant increase in the number of contralateral errors made in the G-CSF- (F(1,13)=85.01, P<0.0001) and vehicle-treated groups (F(1,13)=294.4, P<0.0001) compared with shams. However, G-CSF treatment did significantly reduce this functional deficit, resulting in fewer contralateral foot faults after G-CSF compared with vehicle treatment (F(1,14)=16.55, P=0.0001).
Cognitive Test
Cognitive ability, assessed using the Morris Water Maze, revealed differences in the ability of the different groups to acquire the task, that is, to reduce the latency (seconds) in finding the platform (Figure 4A). As expected, all groups initially exhibited high latencies but these decreased over time as the task was acquired. Shams and G-CSF-treated animals did not differ in their ability to learn the task, showing similar rates in reduction of latency (P>0.05). However, vehicle-treated animals were impaired on this test and showed significantly higher latency compared with either G-CSF-treated animals (F(1,12)=11.80, P=0.0011) or with shams (F(1,12)=29.78, P<0.0001).
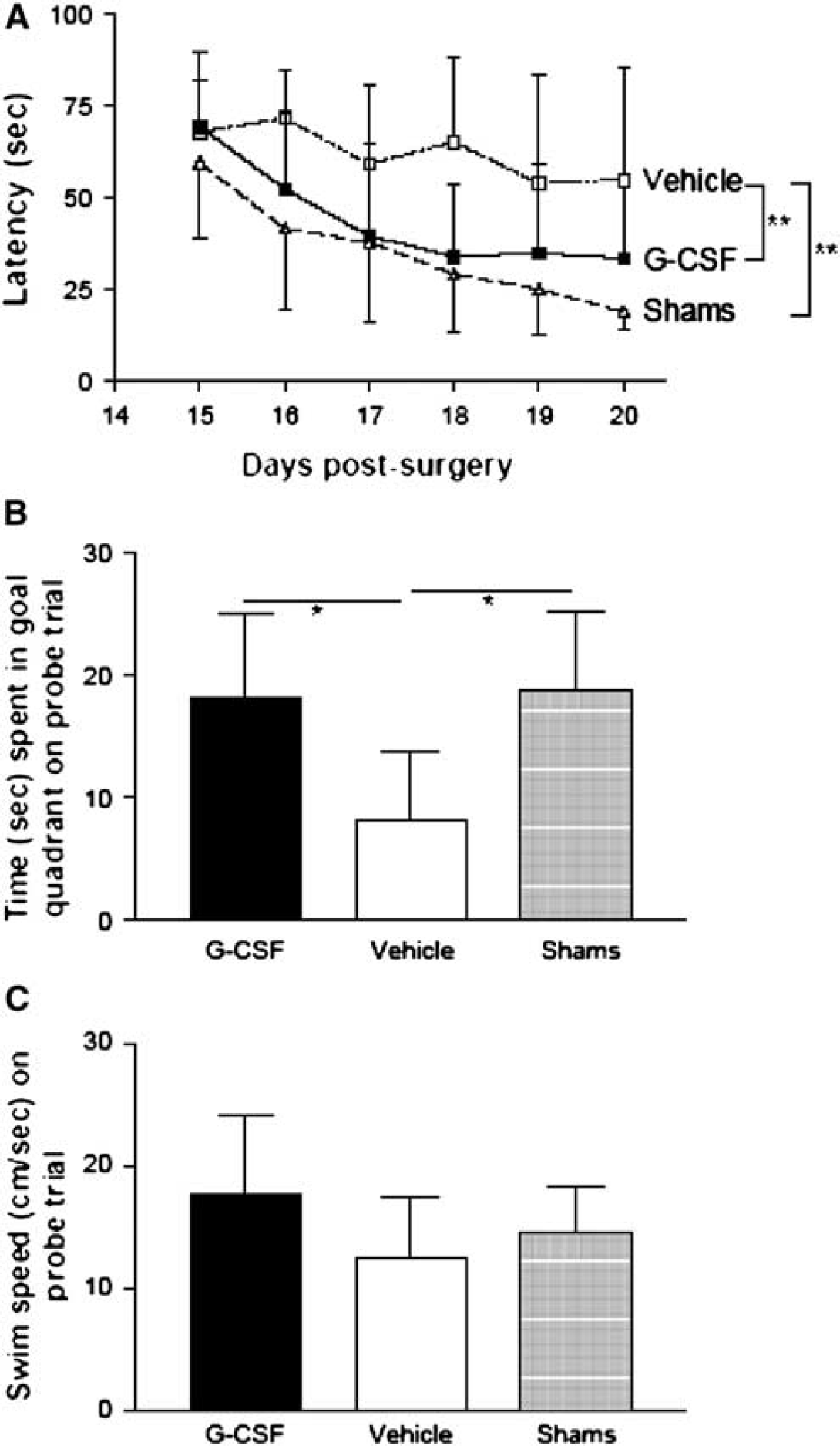
Granulocyte colony-stimulating factor treatment improves cognitive performance. This was assessed using the Morris Water Maze in terms of the ability to acquire the task and learn platform location, and the ability to recall position of the platform on the probe trial (time spent in goal quadrant). Granulocyte colony-stimulating factor treatment after MCAO improved ability to acquire the task, as G-CSF-treated animals showed no significant differences in escape latency as compared with shams. However, vehicle-treated animals were significantly impaired in acquiring the task, as shown by longer escape latency in comparison to either G-CSF treatment, **P<0.01, or shams, ***P<0.0001 (
On the probe trial, treatment had an overall effect on time spent in the goal quadrant (P=0.0185). Both shams and G-CSF-treated animals appeared to have learned the task and remembered the platform location better than the vehicle-treated animals. This was shown by both these groups spending a significantly longer period of time in the goal quadrant (Figure 4B) in comparison to the vehicle group (P<0.05). Swim speed was also measured on the probe trial (Figure 4C), and treatment was found to have no effect. This indicated that the reduced ability of vehicle-treated animals on the water maze task was not the result of impaired motor ability.
Discussion
The present study suggests that G-CSF treatment after 60-minute MCAO significantly reduced the size of the lesion present at 48 hours after ischemia. Additionally, G-CSF treatment was clearly beneficial to the animals, as indicated by weight gain and measures of motor and cognitive performance. The beneficial effect of G-CSF was most apparent in the water maze, where G-CSF-treated MCAO mice acquired the task as well as shams. Apart from our assessment of relative CBF after reperfusion, we did not monitor the effects of G-CSF on physiologic parameters. However, Schabitz et al (2003) reported no effects in the rat of a similar dose of G-CSF on rectal temperature, pH, PCO2, PO2, hematocrit, blood glucose, heart rate, and mean arterial pressure over the subsequent 24-hour period.
Along with other growth factors (Schabitz et al, 2001, 2003), G-CSF has been previously reported to reduce lesion volume after ischemia in mice (Six et al, 2003) and the rat (Schabitz et al, 2003). In these studies, 50 or 60 μg/kg G-CSF was administered via a single subcutaneous injection, and their findings formed the basis for the present study. However, changes in lesion volume do not always represent changes in functional outcome and, while some studies show that lesion size correlates with functional deficit (Rogers et al, 1997), others show it does not (Hattori et al, 2000; Reglodi et al, 2003; Wahl et al, 1992). In the current study, we measured lesion volume at 48 hours, and so it is not possible to rule out the possibility that G-CSF simply delayed the development of the lesion rather than reduced it. Although lesion volume measurement is of interest, unlike behavioral analysis, it gives little indication of the functional benefits of a specific treatment after ischemia.
The MCAO model, first described in rats (Tamura et al, 1981) and later adapted for mice (Hara et al, 1996), is routinely used to mimic focal cerebral ischemia. However, few studies in mice have examined the functional consequences of MCAO. In the present study, all animals displayed weight loss during the first 72 hours after surgery. As expected, shams gained weight at the fastest rate, whereas MCAO animals gained weight more slowly. However, G-CSF treatment increased the rate at which MCAO animals gained weight. Other studies have reported similar weight losses after MCAO (Reglodi et al, 2003) or sham surgery (Barone et al, 1992). Middle cerebral artery occlusion, 60 mins, in rats has been shown to cause significant motor impairments, as assessed using the rotarod and grid test (Rogers et al, 1997). This was found, in the present study, to be true also in mice. However, motor performance on both of these tests was significantly improved in animals that had received G-CSF treatment.
The present study also included a test of cognitive function, conducted 14 to 20 days after surgery to provide a longer-term evaluation of G-CSF treatment. The Morris Water Maze (MWM) is a commonly used test of cognitive function, and mice have been shown capable of successfully acquiring the task (D'Hooge and De Deyn, 2001). Many studies have examined the role of a few well-defined brain regions in MWM learning, and specific deficits occur in animals with damage to the hippocampus, striatum, basal forebrain, cerebellum, and several neocortical areas (D'Hooge and De Deyn, 2001). It is well established that the hippocampal formation is essential for spatial learning, though the importance of one brain region over another in terms of MWM learning remains unclear. For example, there have been reports of deficits in MWM learning in the absence of any hippocampal damage (Okada et al, 1995; Yonemori et al, 1999), whereas lesions restricted specifically to the striatum have been shown to induce deficits (Block et al, 1993). Spatial learning, in particular MWM, tends to be dependent on the coordinated actions of different brain regions acting together to produce a functionally integrated neural network. In the present study, MCAO resulted in a significant inability to learn the MWM with respect to time taken to find the hidden platform over the testing period, and amount of time spent in the goal quadrant on the probe trial. However, G-CSF treatment significantly reduced these deficits, as these mice were not distinguishable from shams. The probe trial, which assessed the memories formed over the testing period, revealed that vehicle-treated mice had not learnt the location of the platform as well as shams or G-CSF-treated mice. On the probe trial, there were no differences in swim speed between the three groups, indicating that deficits in the vehicle-treated animals were not because of impaired motor ability.
The present study reports the use of various functional tests to show the benefits of G-CSF administration after ischemia, and it is interesting to consider through which mechanism(s) G-CSF could potentially act. In vitro studies have shown that G-CSF protects neurons against glutamate-induced neurotoxicity (Schabitz et al, 2003). The main actions of G-CSF are mediated via binding to a G-CSF receptor, present on hematopoietic, neuronal and glial cells (Schabitz et al, 2003). Once activated, the G-CSF receptor can activate proteins such as STAT3 (Shuai, 2000), which in turn activate antiapoptotic proteins such as bcl-2 (Fukada et al, 1996).
It has been shown, in a variety of infections, that G-CSF can mediate antiinflammatory effects (Gorgen et al, 1992; Hebert et al, 1990; Lundblad et al, 1996). This may be through its ability to decrease the levels of proinflammatory cytokines, such as tumor-necrosis factor alpha (Gorgen et al, 1992). Such reduced cytokine activation may, in turn, reduce neutrophil activation and subsequent infiltration; in fact, G-CSF has been shown to reduce neutrophil infiltration in a model of splanchnic ischemia and reperfusion (Squadrito et al, 1997).
While the suppression of inflammation could explain the benefits of G-CSF after MCAO, another effect is to mobilize bone marrow cells. Consequently, G-CSF is administered to patients suffering from neutropenia, most commonly as a result of chemotherapy after cancer. After administration in mice, G-CSF has been shown to increase the number of circulating bone marrow stem cells (BMC) released into the circulation (Orlic et al, 2001). After myocardial infarct, these BMC migrate to the area of damage and contribute to a significant amount of tissue regeneration (Orlic et al, 2001). After cerebral ischemia, it has been observed that either transplanted (Chen et al, 2001) or intravenously administered BMC (Chopp and Li, 2003) infiltrate into the lesion area. Transplantation also results in improved functional outcome, as measured by a variety of neurologic, motor and somatosensory tests (Chen et al, 2001). However, there is debate with regard to the functional contribution of BMC after injury, and the mechanisms by which these cells influence brain repair are still unclear. Survival of BMC for up to 3 months has been shown (Chopp and Li, 2003). While some studies report that these cells are capable of differentiating into neurons (Chen et al, 2001), others do not (Beck et al, 2003). While receiving bone marrow cells from a donor still remains an issue in the clinic (Chopp and Li, 2002), such concerns may be overcome through the administration of drugs, such as G-CSF, that act to stimulate endogenous bone marrow to produce BMC.
Neurogenesis occurs in discrete regions of the CNS continuously during adulthood (Gritti et al, 2002), and is increased after cerebral ischemia (Arvidsson et al, 2002; Jiang et al, 2001; Kokaia and Lindvall, 2003). By increasing the number of circulating BMC, and their infiltration into the CNS, G-CSF could enhance this process. In addition, the antiinflammatory properties of G-CSF could amplify the process of neurogenesis. Inflammation, a major contributor to cell death after cerebral ischemia (Leker and Shohami, 2002), has been shown to be detrimental to neurogenesis. Lipopolysaccharide-induced inflammation strongly impairs the neurogenesis that occurs naturally during adulthood, and also after brain injury (Ekdahl et al, 2003), and suppression of inflammation after brain insult enhances the neurogenesis that occurs (Monje et al, 2003). Thus, G-CSF administration after cerebral ischemia may suppress inflammation and, in turn, create a more permissive environment for circulating BMC to migrate into, populate, survive, and differentiate.
To summarize, here we report a significant decrease in lesion volume, and improved outcome on motor and cognitive tests, with a single treatment of G-CSF after 60-minute MCAO. The mechanisms of G-CSF action after cerebral ischemia, and the possible contribution of mobilized BMC to this functional improvement are subjects for further study.
Footnotes
Acknowledgements
This work was funded by NIH grant NS 29226 (SM). We thank Herve Boutin for advice on MCAO, Nigel Jones for advice on behavioral studies, Annabel Pearson for the lesion volume measurements, and the staff of the BMSU, University of Nottingham, for their care of experimental animals. PMWB is Stroke Association Professor of Stroke Medicine.
