Abstract
Recently the authors have shown that female stroke-prone spontaneously hypertensive rats (SHRSPs) in proestrus (high endogenous estrogen), sustain more than 20% smaller infarcts after middle cerebral artery occlusion (MCAO) compared with SHRSPs in metestrus (low endogenous estrogen). Because estrogen has vasodilator properties, the authors investigated whether the estrous state influences cerebral blood flow (CBF) after MCAO. CBF was measured 2.5 hours after a distal MCAO by [14C]iodo-antipyrine autoradiography in conscious SHRSPs either in metestrus or in proestrus. There were no significant differences in CBF when analyzed either at predetermined anatomic regions or by cumulative distribution analysis of areas with flow <25 mL/100 g/min. As a positive internal control, the authors compared results in SHRSPs with those in their normotensive reference strain, Wistar Kyoto rat. SHRSPs displayed more severe and widespread ischemia than Wistar Kyoto rats. Thus, the absence of demonstrable CBF differences between estrous states appears to be unrelated to the CBF measurement paradigm. In conclusion, the smaller infarct size afforded in proestrus in SHRSPs is unlikely to be due to an influence on CBF.
We have recently observed that the estrogen status of spontaneously hypertensive stroke-prone female rats (SHRSPs) affects the extent of brain damage after a middle cerebral artery occlusion (MCAO). Female SHRSPs with high levels of circulating estrogen (during proestrus) display infarcts more than 20% smaller than those with low levels of circulating estrogen (during metestrus) (209 ± 25 mm3 vs. 273 ± 63 mm3, Carswell et al., 2000). Dubal et al. (1998) and Rusa et al. (1999) have shown that estrogen administered to ovariectomized females significantly reduces infarct size. In the clinic, the value of estrogen replacement in improving stroke risk and outcome in postmenopausal women is less clear. Although Finucane et al. (1993) showed decreased stroke risk, others have shown either no effect (Pedersen et al., 1997) or an increased effect (Wilson et al., 1985) on stroke risk by estrogen replacement.
Estrogen has been reported to induce vasodilatation (Huang et al., 1997; White et al., 1995), possibly mediated by nitric oxide (Pelligrino et al., 1998), prostacyclin (Mendelsohn and Karas, 1994), or an endothelium-independent mechanism (White et al., 1995). The present study investigates whether, through the vasodilatory effects of estrogen, animals in proestrus exhibit improved post-MCAO cerebral blood flow (CBF) compared with animals in metestrus, thereby accounting for the smaller lesion size observed in proestrus.
We incorporated a positive control into our experimental paradigm by comparing CBF after MCAO in SHRSPs with that in its normotensive reference strain, the Wistar Kyoto rat (WKY). SHRSPs display larger infarct sizes (Carswell et al., 1999; Coyle and Jokelainen, 1983) and lower CBF levels (Coyle and Heistad, 1986) compared with WKYs after MCAO.
METHODS
All experiments were carried out under license from the Home Office and were subject to the Animals (Scientific Procedures) Act, 1986. Breeding and housing of SHRSP and WKY rats has previously been described (Clark et al., 1996). Two SHRSP groups were used: females in metestrus (n = 5) and females in proestrus (n = 5). One WKY group (females in metestrus, n = 6) acted as an internal positive control. The stage of the estrous cycle was determined by smear test (Baker et al., 1979). The accuracy of the smear test was confirmed by measurement of plasma estradiol levels during metestrus and proestrus (Coat-a-Count Radioimmunoassay, Euro/DPC Ltd., Wales, U.K.), with an interassay coefficient of variance of less than 10%. However, because a large volume of plasma was required for the radioimmunoassay, a separate group of WKY females was used to provide samples for analysis. Body weights were 180 to 220 g in SHRSPs and 190 to 230 g in WKYs. Anesthesia was induced (5%) and maintained (1% to 2%) by halothane in oxygen and nitrous oxide (30% and 70%). A 2-mm distal segment of the left MCA was occluded by electrocoagulation using the technique of Tamura et al. (1981), with monitoring of physiologic variables throughout the MCAO and before CBF measurement. A temperature probe inserted into the temporalis muscle was used to monitor brain temperature during MCAO. After MCAO, anesthetics were withdrawn and the animals were placed in a plaster cast for restraint and allowed to recover fully. CBF was measured in the conscious rat 2.5 hours after MCAO by quantitative autoradiography using [14C]iodoantipyrine as described by Sakurada et al. (1978) with minor alterations. A dose of 25 to 50 μCi [14C]iodoantipyrine in 0.7 mL saline was administered by ramped intravenous infusion during 60 seconds. During the ramped infusion, 15 to 18 timed arterial blood samples were collected onto preweighed filter paper. On completion of the infusion, the brain was quickly removed, frozen in isopentane (–42°C), sectioned (20-μm sections) coronally, and exposed to autoradiographic film for 1 to 2 days. The absolute CBF was determined by image analysis (MCID, St. Catherine's, Ontario, Canada) from the 14C blood profile, the partition coefficient of the tracer (0.79), and the autoradiographic concentrations of 14C.
The 2.5-hour time interval allowed us to measure dynamic aspects of flow when the lesion is still evolving and when flow is important to outcome. At this time point, alterations in CBF can be produced by intervention (e.g., nimodipine [Jacewicz et al., 1990]), resulting in reduction in infarct size. This time point was early enough to avoid measuring flow when the lesion has been established (e.g., by 4 hours [Kaplan et al., 1991]) and late enough to allow residual halothane to be washed out reliably from both the central nervous system (particularly slow in low-flow areas) and the periphery. Measurement during halothane exposure would not have been practicable because of the potential for anesthetics to compromise CBF to a different extent in the two strains.
For cumulative distribution analysis, CBF levels were set at increments of 5 mL/100 g/min, up to 100 mL/100 g/min, and the cumulative area of cortex at each threshold was measured. Cortical area was then expressed as a percentage of the total cortical area. Measurements were made at two coronal planes. The level of the septal nuclei (7.2 mm from interaural line [Konig and Klippel, 1963]) was chosen to represent blood flow close to the occlusion site. The level of the aqueduct (1.0 mm from the interaural line [Konig and Klippel, 1963]) was chosen to represent blood flow within the MCA territory but distant from the occlusion site. In addition, regional CBF was measured at 10 predetermined neuroanatomic sites in the cortex throughout the MCA territory. The sites, according to Paxinos and Watson (1986), were used irrespective of the flow patterns at that site.
All data are presented as mean ± standard deviation. The cumulative area of cortex with blood flow less than a predetermined threshold was plotted against the cumulative CBF intervals. An overall assessment of the cumulative distribution curves for the three groups was made, rather than the significance of individual points, by a univariate repeated measures analysis of variance, investigating all possible interactions. For each of the regions studied, CBF was examined by one-way analysis of variance, followed by t-tests with Bonferroni corrections for two comparisons (estrous state within SHRSP and WKY vs. SHRSP at metestrus).
RESULTS
Physiologic parameters are illustrated in Table 1 at the time of MCAO (anesthetized) and at the time of CBF measurement (conscious). All parameters were within normal physiologic ranges, except for the higher blood pressure in the hypertensive strain.
Physiologic parameters at time of middle cerebral artery occlusion (anesthetized) and at time of cerebral blood flow measurement (conscious) in SHRSP in proestrus and in WKY and SHRSP in metestrus
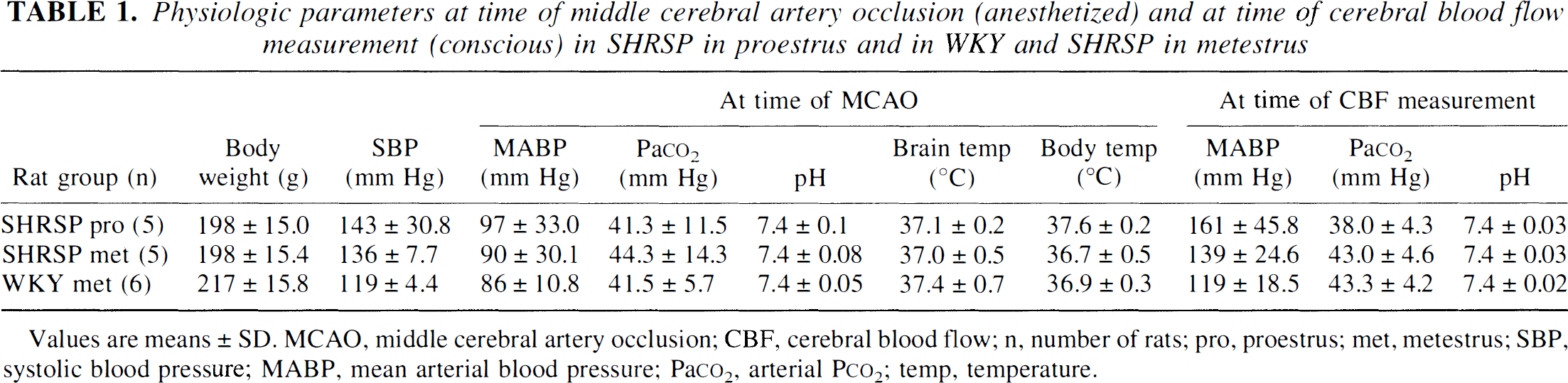
Values are means ± SD. MCAO, middle cerebral artery occlusion; CBF, cerebral blood flow; n, number of rats; pro, proestrus; met, metestrus; SBP, systolic blood pressure; MABP, mean arterial blood pressure; Paco2, arterial Pco2; temp, temperature.
17β-estradiol assay results
Plasma 17β–estradiol levels were 15.4 ± 9.8 pg/mL (n = 5) in animals in metestrus and 167.4 ± 135.1 pg/mL (n = 5) in proestrus (P < 0.05, unpaired two-tailed t-test).
Cumulative distribution of CBF
The cumulative distribution curves were not significantly different between SHRSPs in metestrus and SHRSPs in proestrus at either coronal level examined (Fig. 1). There was, however, a significant difference in the separation of the blood flow distribution curves between SHRSPs and WKYs at both the level of septal nuclei (P = 0.025) and the level of the aqueduct (P = 0.018) (see Fig. 1). In addition, the rate of change in percentage area with increasing CBF was significantly greater in SHRSPs compared with WKYs at the level of the septal nuclei (P = 0.015) but not at the level of the aqueduct (P = 0.097).
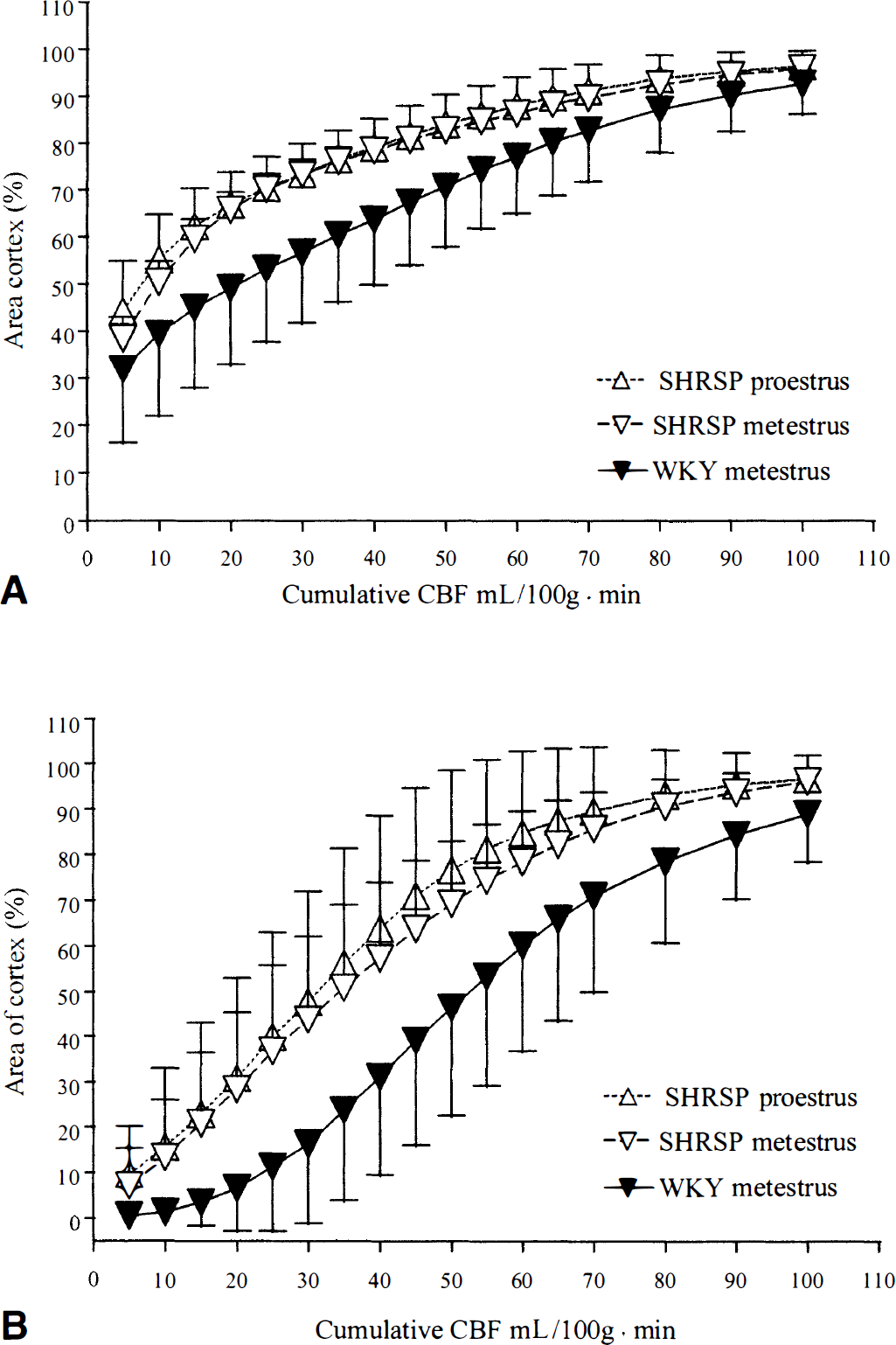
Cumulative distribution analysis of cerebral blood flow (CBF) in female stroke-prone spontaneously hypertensive rats (SHRSPs) in proestrus (n = 5), SHRSPs in metestrus (n = 5), and Wistar Kyoto rats (WKYs) in metestrus (n = 6) in the ipsilateral cortex after middle cerebral artery occlusion (MCAO) at the level of the septal nuclei (
For comparison of neuropathology and ischemia, Fig. 2A shows a significant difference in areas of damage between SHRSPs in the two estrous states and between SHRSPs and WKYs (Carswell et al., 2000), but proportions of cortex with blood flow less than 25 mL/100 g · min differed only between SHRSPs and WKYs. At the level of the septal nuclei, the area with blood flow less than 25 mL/100 g · min in SHRSPs in metestrus and in proestrus was 70.1% ±3.3% and 70.4% ± 6.7%, respectively (n = 5, not significant, unpaired t-test). At the level of the aqueduct (see Fig. 2B), the area of cortex with blood flow less than 25 mL/100 g · min in SHRSPs in metestrus and in proestrus was 36.8% ± 19.0% and 39.9% ± 23.1% respectively (n = 5, not significant, unpaired t-test).
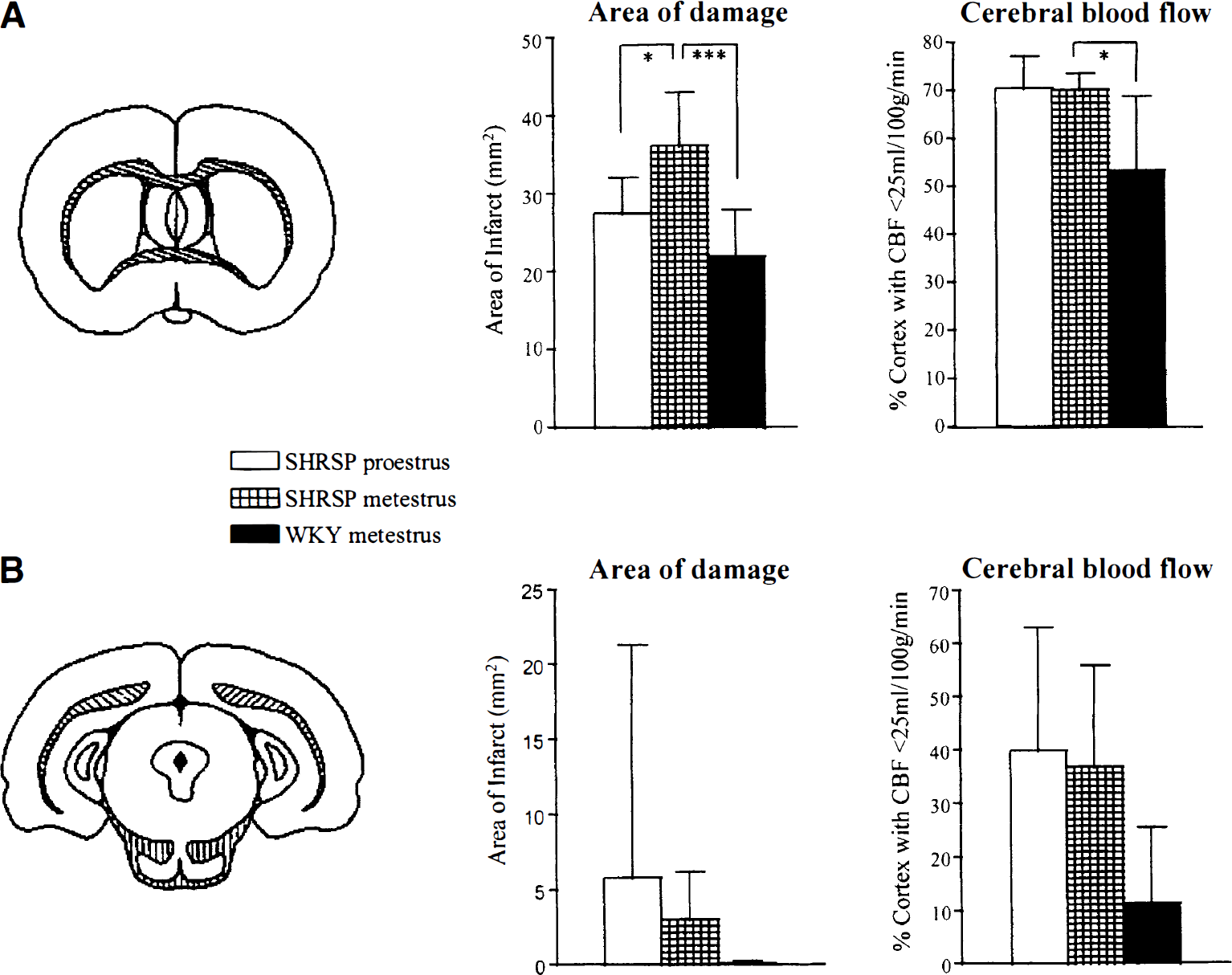
The area of infarct (mm2) (Carswell et al., 2000) and the area of cortex (%) with blood flow less than 25 mL/100 g · min at the level of the septal nuclei
Regional CBF at predetermined neuroanatomic cortical sites
There was no statistical difference in CBF values between SHRSPs in metestrus and in proestrus at any of the regions examined. SHRSPs had lower CBF compared with WKYs at most of the regions examined (Table 2). Contralateral regional blood flow was similar between the estrous states and between the two strains (Fig. 3).
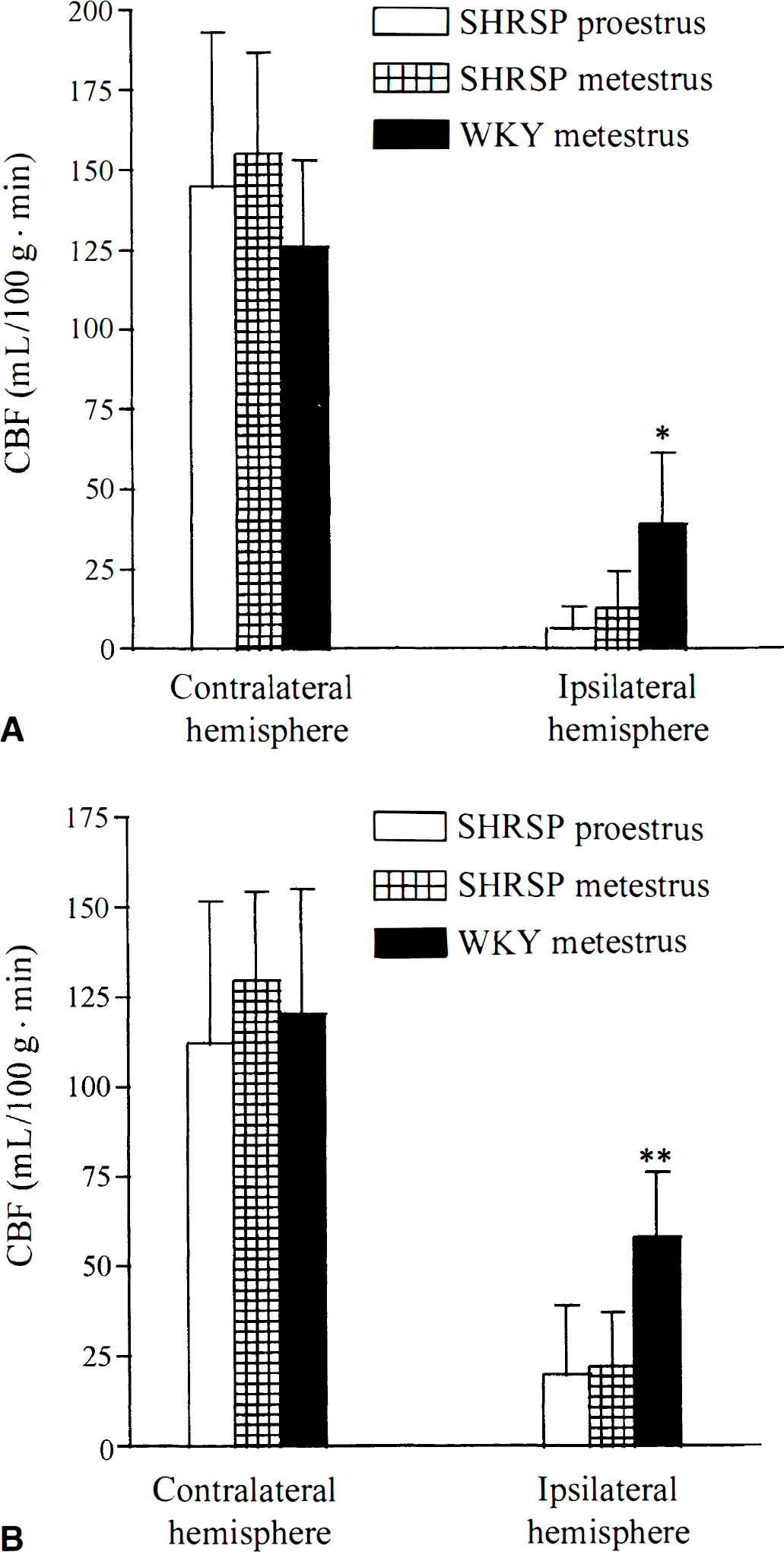
Contra- and ipsilateral blood flow in the frontal cortex
Cortical CBF (mL/100 g · min)
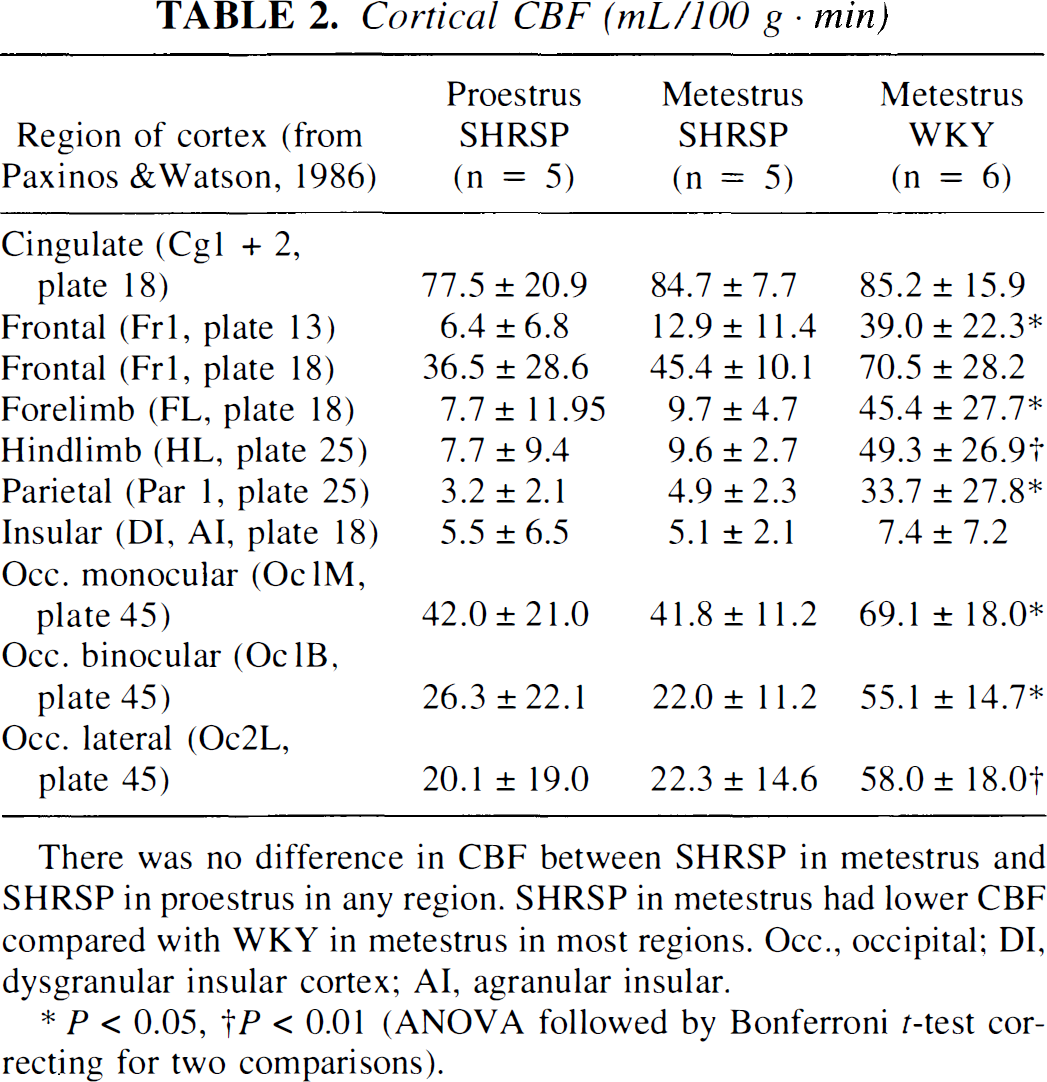
There was no difference in CBF between SHRSP in metestrus and SHRSP in proestrus in any region. SHRSP in metestrus had lower CBF compared with WKY in metestrus in most regions. Occ., occipital; DI, dysgranular insular cortex; AI, agranular insular.
P < 0.05
P < 0.01 (ANOVA followed by Bonferroni t-test correcting for two comparisons).
DISCUSSION
The key finding of the present study is that smaller infarcts recorded during proestrus compared with metestrus in SHRSPs (Carswell et al., 2000) are unlikely to be the end result of improved CBF in the proestrus group. Neither type of analysis used in the present study (cumulative distribution nor regional CBF) detected changes in the blood flow either close to or distant from the occlusion site, between the two estrous states. Even in periinfarct penumbral regions, where cerebral perfusion is less severely compromised, allowing more potential for estrogen to reach and influence the blood vessels, there was no difference in CBF between proestrus and metestrus. However, differences in CBF were clearly detected between SHRSPs and WKYs.
Estrogen-induced reductions in infarct size have been previously observed (Dubal et al., 1998; Rusa et al., 1999). Whether progesterone is important in stroke is less clear. To date, progesterone has been demonstrated to reduce brain damage after focal ischemia in male rats (Jiang et al., 1996) but not in female rats (Murphy et al., 1999).
The effects of estrogen on CBF have been investigated, but the conclusions are controversial. Compared with ovariectomized rats, estrogen replacement with “physiologic” plasma concentrations did not affect CBF after MCAO (Dubal et al., 1998; Rusa et al., 1999) but was shown to enhance CBF in a different model of transient forebrain ischemia (Pelligrino et al., 1998). In addition, Hurn et al. (1995) showed that supraphysiologic levels of estrogen (365 pg/mL) improved CBF during global ischemia in rabbits. These studies assessed intraischemic CBF. The effects of estrogen on reperfusion CBF appear to be equally unclear. Hurn et al. (1995) showed that estrogen prevented hyperemia during reperfusion, whereas Toung et al. (1998) showed no effect of estrogen on reperfusion CBF. Thus, although there is little evidence to indicate that estrogen improves CBF during focal ischemia and subsequent reperfusion, the results from global and forebrain ischemia models suggest that in certain cerebrovascular beds the flow-enhancing effects of estrogen may be uncovered.
The present study indicates that the smaller infarct size recorded in proestrus in SHRSPs is unlikely to be mediated by improved CBF. This is supported by the findings that ovariectomy per se does not influence cortical blood flow under basal conditions (Holschneider and Scremin, 1998) or under focal ischemic conditions (Alkayed et al., 1998). In addition, exogenous estrogen can reduce forebrain ischemia-induced neuronal cell loss by means other than improved CBF (Wang et al., 1999). Such flow-independent mechanisms may be mediated by activation of estrogen receptors that are extensively distributed in the cerebral cortex (Simerly et al., 1990). Genomic effects of estrogen, such as modulation of expression of antiapoptotic genes (Dubal et al., 1999) or of neurotrophins (Sohrabji et al., 1995), and/or nongenomic effects, such as inhibition of lipid peroxidation (Goodman et al., 1996), are examples of flow-independent mechanisms that may contribute to protection sustained in proestrus.
To test our experimental paradigm for CBF measurement and to help interpret whether our result could represent a type II error, we incorporated an internal positive control into the present study. We have previously shown a twofold increase in infarct size in SHRSPs compared with WKYs (Carswell et al., 1999), and in the present study we compared CBF after MCAO in the two strains. A more severe and widespread ischemia was recorded in the ipsilateral hemisphere in SHRSPs compared with WKYs after MCAO. This was shown in the thresholding data and confirmed by analysis of regional CBF. The cumulative distribution curves for SHRSPs were shifted to the left relative to WKY curves, because lower levels of CBF were present throughout a greater fraction of cortex in SHRSPs compared with WKYs. Regional cortical blood flow was lower in SHRSPs in all ipsilateral regions examined except the insular and cingulate cortices. The insular cortex, being in the core of the infarct, displayed virtually no flow in both strains after MCAO. Blood flow to the cingulate cortex is supplied mainly by the anterior cerebral artery, and MCAO had little impact on its flow in either strain. The similar contralateral CBF observed in the present study (see Fig. 3) and the similar basal CBF observed by Coyle and Heistad (1986) between SHRSPs and WKYs indicate that the ipsilateral CBF differences are not due to preischemic differences.
In the present experiments, our analysis has identified a CBF difference where a difference in infarct size exists between SHRSPs and WKYs. Thus, the lack of difference in CBF between estrous states is indeed a valid result and not a type II (false-negative) error. In conclusion, the smaller infarct size sustained in proestrus compared with metestrus after MCAO is unlikely to be due to improved CBF in the proestrus group.
Footnotes
Acknowledgments
The authors thank Delyth Graham, Steven Keenan, Lindsay Gallagher, Duncan MacGregor, Joan Stewart, and Margaret Stewart for their technical expertise and assistance.
