Abstract
To assess the time window for fibroblast growth factor-18 (FGF18)-mediated neuroprotection, FGF18 was administered by intravenous infusion at various times after transient occlusion of the middle cerebral artery (MCAO) in rats. Vehicle or FGF18 (100 μg·kg−1·h−1) was infused at 0.25, 0.5, 1.0, 2.0, 4.0, or 8.0 hours after MCAO with infarct volumes and behavioral deficits measured at 24.0 hours after MCAO. A separate group of animals received the infusions 24 hours after MCAO with endpoints measured at 48 hours after MCAO. Infusion of FGF18 reduced infarct volumes and improved scores in tests of reference and working memory, motor ability, and exploratory behavior. FGF18 was most efficacious when infused within 2 hours after MCAO. Significant reductions in infarct volumes and reductions in deficits of reference memory and motor activity were also observed with FGF18 infused 24 hours after MCAO. Measurements taken at infusion times before 2 hours after MCAO showed that regional cerebral blood flow was increased by FGF18. Administration of vehicle or FGF18 had no significant effect on mean arterial blood pressure, heart rate, brain temperature, blood pH, P
Members of the fibroblast growth factor (FGF) family of proteins have been studied as tissue protectants in animal models of focal or global cerebral ischemia (Ay et al., 2001; Cuevas et al., 1998; Yao et al., 1999). Although efficacious in reducing infarct volumes and behavioral deficits in preclinical studies, results of FGF2 administration in human clinical trials have been disappointing (Bogousslavsky et al., 2002). A new member of the Fgf8/Fgf17 subfamily of FGFs, Fgf18, is expressed in the brain both during development and in the adult (Maruoka et al., 1998; Xu et al., 2000). Hippocampal expression of Fgf18 is upregulated during maze learning in rats, suggesting a role for this factor in the acquisition and retention of spatial memory (Cavallaro et al., 2002). FGF18 has also been reported to stimulate neurite outgrowth from neuronal-derived tissues (Ohbayashi et al., 1998) and is mitogenic for glial cells and astrocytes (Hoshikawa et al. 2002). Exogenous administration of FGF18, moreover, improved spatial learning behavior in normal rats (Cavallaro et al., 2002). These activities suggest that FGF18 might be useful as a neuroprotective or neurorecovery agent in stroke. In support of this hypothesis, we recently reported that administration of FGF18 by intravenous infusion beginning 15 minutes after transient occlusion of the middle cerebral artery (MCAO) in rats reduced infarct volumes and behavioral deficits (Ellsworth et al., 2003). The later studies were extended in the present report by examining the time window for FGF18-mediated neuroprotection.
MATERIALS AND METHODS
Induction of cerebral ischemia
Male Sprague-Dawley rats (250 to 275 g, Harlan, Indianapolis, IN, U.S.A.) were fasted for 4 to 6 hours before experimentation. All procedures were within institutional guidelines for animal use. Rats were anesthetized with halothane (1% in 70%/30% NO2/O2), and their bilateral femoral arteries were cannulated using PE-50 tubing for monitoring of MABP and for blood collection. The right femoral veins were cannulated for infusion of test articles. Body and brain temperatures were monitored using a rectal thermometer and thermistor probe inserted into the temporalis muscle, respectively, and were maintained at 37°C by using a water-jacketed heating pad. Temperatures and MABP were monitored continuously from 1 hour before to 6 hours after ischemia. The left common carotid artery of each rat was exposed through a midline incision in the neck. The superior thyroid and occipital arteries were electrocoagulated and divided. A microsurgical clip was placed around the origin of the external carotid artery (ECA). The distal end of the ECA was ligated with 6-0 silk, transected, and a 6-0 silk was tied loosely around the ECA stump. The clip was removed and the blunted tip of a 4-0 nylon suture was inserted into the ECA stump (Longa et al., 1989). The loop of the 6-0 silk was tightened around the stump and the nylon suture was advanced into and through the internal carotid artery until it rested in the anterior cerebral artery. After the nylon suture had been in place for 2 hours, it was pulled back into the ECA and the incision was closed. Approximately 5% of the rats were not included in the study because of insufficient MCAO.
Experimental groups
At various times after the onset of ischemia, vehicle or FGF18 (100μg·kg−1·h−1) was administered by intravenous infusion at a rate of 0.5 mL/h over a 3-hour period. The contents of the infusion vials were blinded to the investigators performing both the infusions and the analyses. Groups I through VII refer to animals that were infused with either vehicle or FGF18 at 0.25, 0.5, 1.0, 2.0, 4.0, 8.0, or 24.0 hours after MCAO, respectively.
Estimation of infarct volume
Each rat was anesthetized with an intraperitoneal injection of sodium pentobarbital (50 mg/kg) at 24 (groups I-VI) or 48 hours (group VII) after ischemia. The brains were transcardially perfused with 10% phosphate-buffered saline and were removed and chilled for 15 minutes at −20°C and placed in a Rodent Brain Matrix (Electron Microscopy Sciences, Fort Washington, PA, U.S.A.). Coronal sections were prepared and subjected to 2% triphenyltetrazolium chloride staining as described previously (Ellsworth et al., 2003). The infarct area in each section was determined using NIH Image Analysis Software, v. 1.55 (National Institute of Health, Bethesda, MD, U.S.A.). Total infarct volume for each brain was calculated by summation of the infarct areas of nine brain slices [area × slice thickness (1 mm)] for each hemisphere and corrected for edema as follows: infarct volume in cubic millimeters = infarct volume – (TIV – TCV); where TIV is the total ipsilateral volume and TCV is the total contralateral volume.
Behavioral testing
Animals were preconditioned to all tasks and were tested before and 24 hours (Groups I-VI) or 48 hours (Group VII) after MCAO. There were no significant intergroup differences in the pre-MCAO values for any of the behavioral tests. The Morris water maze was used to measure reference and working memory (Corbett and Nurse, 1998; Kraemer et al., 1996). The pool consisted of a 115-cm (diameter) × 57-cm (height) circular metal tank with a removable 14.5-cm-wide, 34-cm-high metal platform. The water level was 1 cm above the surface of the platform with the water temperature maintained at 19°C to 21 °C. Nontoxic black powdered paint was added to the water to obscure the visual appearance of the platform. Video records of each animal's performance were analyzed with a Videomax V (Columbus Instruments, Minster, OH, U.S.A.). In tests of reference memory, video records were analyzed over three 90-second trials for the following: target zone latency, the time taken to reach the former location of the platform; search time, the percent of time during each of three 30-second intervals of the probe trial that the subject spent in the quadrant that contained the platform; and relative target visits, the percent of visits to the target zone out of the total number of visits to all four zones. In the working memory test, video records of two 60-second trials were analyzed for target zone latency.
General locomotion and activity within a novel, open-field environment were assessed in exploratory behavior and general activity tests (Whishaw et al., 1987). The test apparatus consisted of a 60-cm (width) × 60-cm (length) × 100-cm (height) plastic box painted black. A video motion analyzer recorded the activity of each rat during each 15-minute session. The average scores were relative measures: scores of 0, 1, 2, 3 and 4 represented 100%, 75%, 50%, 25%, and 0% of normal, respectively.
Motor performance was evaluated using the beam walk test (Dose et al., 1997). The time required to traverse the beam was converted to an ordinal scale: latencies for all three trials < 10 seconds = 1; latencies > 10 seconds < 25 seconds for all three trials = 2; latencies for all three trials > 25 seconds and < 60 seconds = 3; failure to complete all three trials but completion of one or two with latencies < 25 seconds = 4; failure to complete all three trials but completion of one or two with latencies > 25 seconds = 5; failure to complete all trials = 6.
Measurement of cerebral blood flow
Regional cerebral blood flow (rCBF) was monitored by laser Doppler flowmetry every 30 minutes over the period 1 hour before to 6 hours after MCAO. Animals were anesthetized with halothane (1% in 70%/30% NO2/O2) and a 2-mm hole was drilled in the skull, with the probe was positioned at 0.1 mm above the dura over the cortical surface. In the hemisphere ipsilateral to the occlusion, coordinates were as follows: point A, 1 mm posterior to the bregma and 5.4 mm lateral to the midline; point B, 1 mm posterior to the bregma and 2.1 mm lateral to the midline; point C, 1 mm anterior to the bregma and 3.4 mm lateral to the midline. The mean values of rCBF measured before MCAO were taken as baseline, and the data thereafter were expressed as percentages of this value. The rCBF data presented in the present report were taken from reference point A.
Statistical analysis
The results are expressed as the mean ± SD. Unless indicated otherwise, data from the third trial after MCAO are presented for each test. Two-group comparisons were evaluated by a Student's t-test or the Mann-Whitney test using Bonferroni correction for multiple comparisons. Differences in the monitoring data were analyzed by repeated-measures analysis of variance (Instat, 2.03, GraphPad, San Diego, CA, U.S.A.).
Comparisons between FGF18 and vehicle were evaluated by fitting a linear regression model including the treatment (FGF18 or vehicle), the infusion time, and pre-MCAO blood gas levels as predictors of the outcome variable using STATA 7 (Stata, College Station, TX, U.S.A., 2001). Where given, P values were calculated on the basis of a two-sided likelihood ratio test. Results did not change when the regression model was modified to allow for the treatment group difference to vary by specific time point. Each point in the figures represents the mean ± SD for n = 12 animals per group.
Production of recombinant human FGF18
Fgf18 was expressed in Escherichia coli and purified from culture media as described previously (Ellsworth et al., 2002). The purified FGF18 showed a single species at about 20 kd on reducing sodium dodecyl sulfate – polyacrylamide gel electrophoresis analysis. The preparation was homogeneous as confirmed by N-terminal sequencing and mass spectrometry analyses. The FGF18 was dialyzed into 1.0-mol/L NaCl, 0.01-mol/L edetic acid, and 0.05-mol/L sodium phosphate (pH 7.2) and was stored at −80°C. Before use, FGF18 was bufferexchanged into vehicle solution (0.05-mol/L sodium phosphate [pH 7.2], 0.094-mol/L NaCl, 0.05-mg/mL bovine serum albumin) and stored at −20°C. The bioactivity of FGF18 was evaluated with an alamar blue cell proliferation assay using BaF3 cells stably-expressing Fgfr 3-(IIIc) as described elsewhere (Ellsworth et al. 2002).
RESULTS
Similar to results reported previously (Ellsworth et al., 2003), transient occlusion of the middle cerebral artery in rats produced well-defined infarcts involving the striatum, subcortical white matter, and cortex. Infusion of FGF18 at 100 μg·kg−1·h−1 for 3.0 hours starting 0.25 hours after transient MCAO reduced infarct volumes (Fig. 1). Reductions in infarct volumes were also observed when the FGF18 infusions were delayed for up to 4 hours after MCAO (Fig. 1). Compared to infusion of vehicle, infarct volumes were reduced 32%, 30%, 25%, 17%, 14%, and 0% with FGF18 infusions starting 0.25, 0.5, 1.0, 2.0, 4.0, and 8.0 hours after MCAO, respectively. Relative to animals infused with vehicle at 8 hours after MCAO (with infarct volumes measured at 24 hours after MCAO), lesion volumes were increased in animals infused with vehicle at 24 hours after MCAO (with infarct volumes measured at 48 hours after MCAO) from 115.8 ± 9.9 mm3 to 143.2 ± 9.5 mm3, respectively, (mean ± SD, n = 12 animals per group, P < 0.0009). Lesion expansion over this time frame was abolished by a single infusion of FGF18 24 hours after MCAO (Fig. 1).
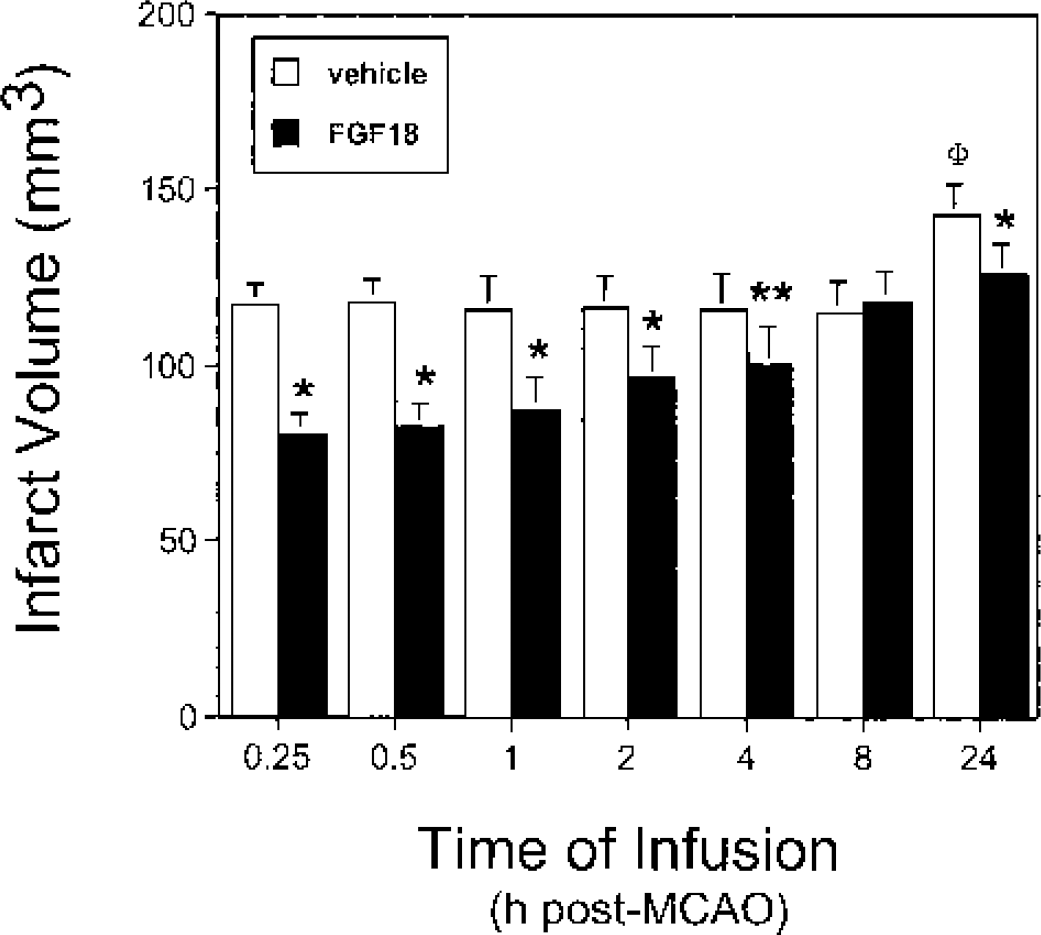
FGF18 reduced infarct volumes in MCAO rats. MCAO animals were prepared, infused with either vehicle (open bars) or FGF18 (solid bars) at various times after ischemia, and infarct volumes were assessed as described in Materials and Methods. The following differences were significant: vehicle vs. FGF18, *P < 0.0009, **P < 0.018; 8 hours vs. 24 hours after MCAO vehicle infusions, ΦP < 0.0009.
Preischemic rats acquired and retained spatial memory as shown by decreasing target zone latencies over the three trials of the reference memory test (34.1 ± 2.2 seconds, 24.7 ± 2.1 seconds, and 13.1 ± 2.6 seconds for trials 1, 2, and 3, respectively, mean ± SD, n = 183 rats per trial, P < 0.0001). Relative to preischemia values, postischemic animals infused with vehicle at various times after MCAO took four to five times longer to find the former location of the platform in the third trial of the water maze test (Fig. 2). Compared to the 0.25-hours infusion time, rats infused with vehicle exhibited a biphasic response over this time course: a reduction in target zone latency was observed in the vehicle treated animals with infusions starting 0.25 to 8.0 hours after MCAO, suggesting a modest degree of recovery over this time, followed by an increase in target zone latency from the 8.0 hours to the 24.0 hours after MCAO infusions (Fig. 2). Infusion with FGF18 starting ≤ 4 hours after the onset of ischemia reduced the target zone latency (Fig. 2), with reductions of more than 60% seen with FGF18 infusions at the earliest times. Compared to infusion of vehicle at 24 hours after MCAO, the target zone latency was also reduced (P < 0.0011) by infusion of FGF18 at this time (Fig. 2). Infusion of FGF18 starting 15 or 30 minutes after the onset of ischemia reduced the search time deficit by 50% to 60% (P < 0.0005, Fig. 3). No significant differences in search time deficit were noted between the groups for infusions delayed longer than 30 minutes after MCAO (Fig. 3). Relative target zone visits for preischemic rats were 51.2% ± 5.7% (mean ± SD, n = 183 rats) of the total visits. None of the post-MCAO rats infused with vehicle had a relative target visit score above 25%, the level of chance (Fig. 4). In contrast, infusion of FGF18 increased the relative number of visits to the target zone to preischemic levels at virtually all infusion time points examined (Fig. 4). The exception was seen for FGF18 infusions initiated at 2 hours after MCAO. Whether the apparent lack of efficacy at this time was due to technical reasons or to a change in the functional status of the animals over this time is not clear. With FGF18 infusions started 24 hours after MCAO, the relative target visits were increased to 80% of the preischemic values (Fig. 4). The above measures of relative target visits were normalized to the total number of visits to all quadrants in the water maze, thus correcting for differences in motor ability between the groups. To assess whether MCAO was also associated with a change in a motor component of reference memory test, the total number of visits to all quadrants was tabulated for each group. Relative to preischemia values of 12.5 ± 1.4 total visits (mean ± SD, n = 183 rats), total visits were reduced to 8.4 ± 1.3 total visits (mean ± SD, n = 84 rats, P < 0.0001) in MCAO rats infused with vehicle (Fig. 5). Infusion of FGF18 at virtually all post-MCAO time points increased the total number of visits to all quadrants. Again, whether the apparent lack of efficacy at the 2-hours infusion time was due to technical reasons or to a change in the functional status of the animals over this period is not clear. Taken together, these data show that infusion of FGF18 reduced the deficits in the acquisition and retention of spatial memory after MCAO in rats.
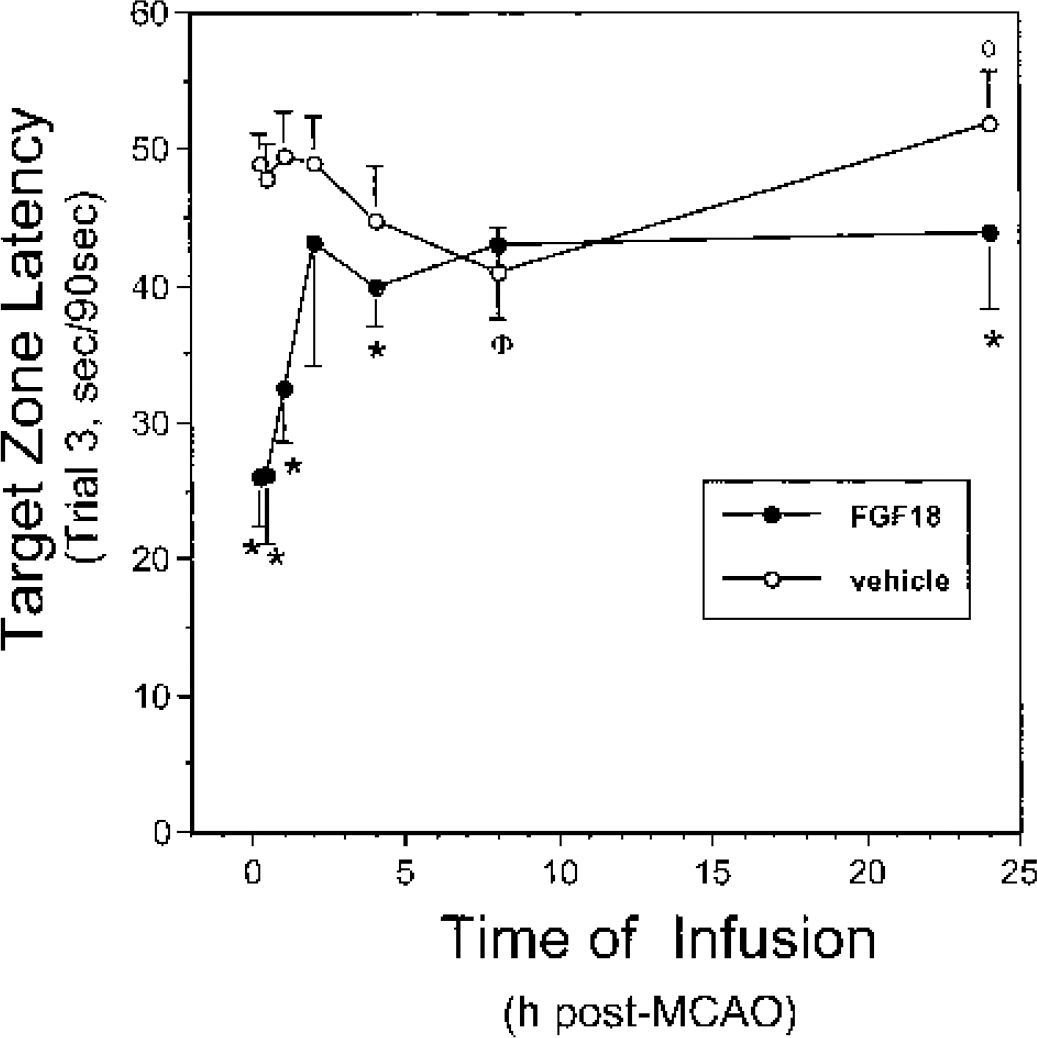
Reduction in the target zone latency by infusion of FGF18. MCAO rats were prepared and infused with either vehicle (open symbols) or FGF18 (solid symbols) at various times after ischemia as described in Materials and Methods. Target zone latencies in the third trial of the reference memory test were measured as described. The following differences were significant: vehicle vs. FGF18, *P < 0.0011; 0.25 hours vs. 8.0 hours after MCAO vehicle infusions, ΦP < 0.0011; 8.0 hours vs. 24.0 hours after MCAO vehicle infusions, φP < 0.0011.
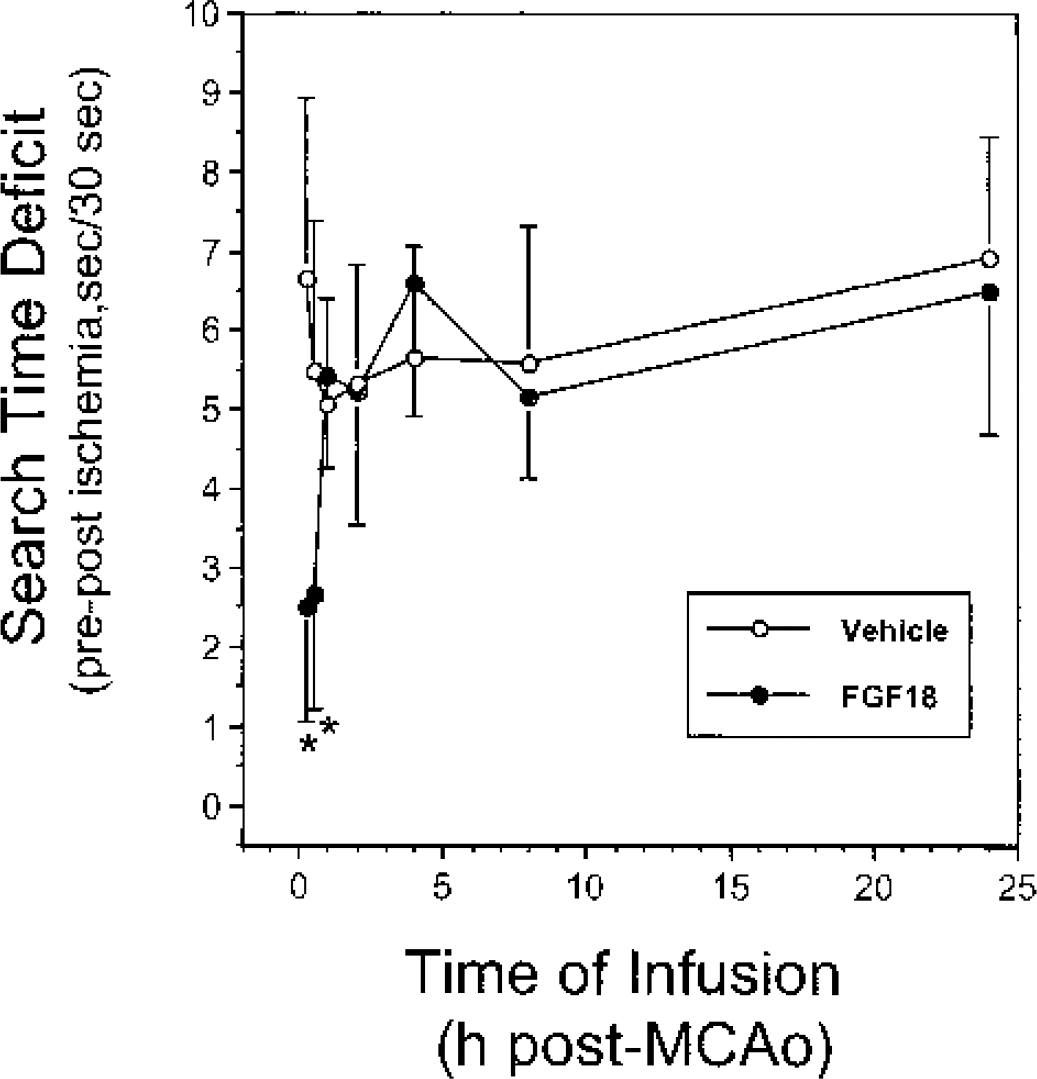
Early administration of FGF18 reduced the search time deficit in rats with MCAO. MCAO animals were prepared and infused with either vehicle (open symbols) or FGF18 (solid symbols) at various times after ischemia as described in Materials and Methods. Search time deficit in the third trial of the reference memory test was measured as described. The following difference was significant: vehicle vs. FGF18, *P < 0.0002.
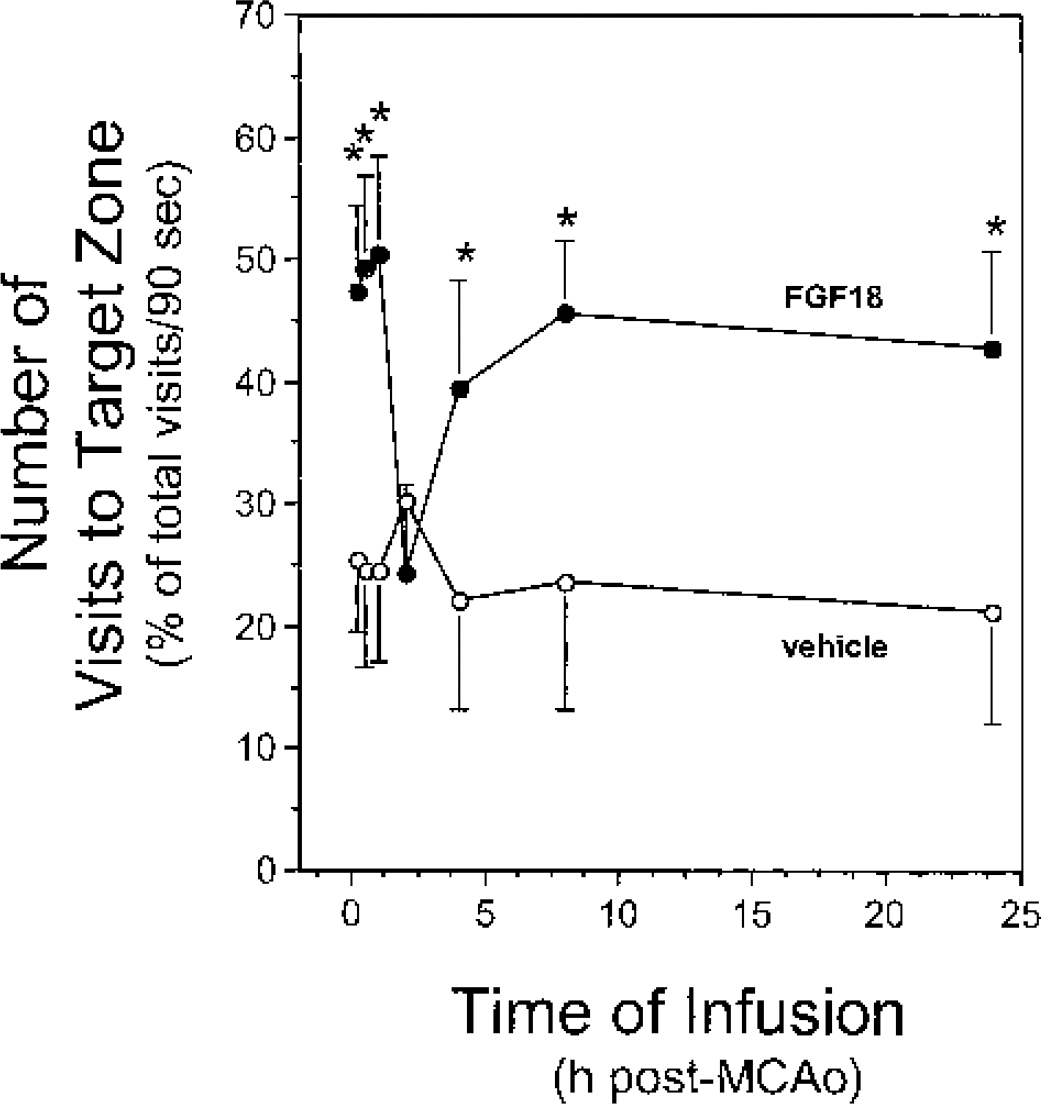
Infusion of FGF18 increased the relative target zone visits in MCAO rats. MCAO animals were prepared and infused with either vehicle (open symbols) or FGF18 (solid symbols) at various times after ischemia as described in Materials and Methods. The relative number of visits to the target zone in the third trial of the reference memory test was assessed for each animal as described. The following difference was significant: vehicle vs. FGF18, *P < 0.0006.
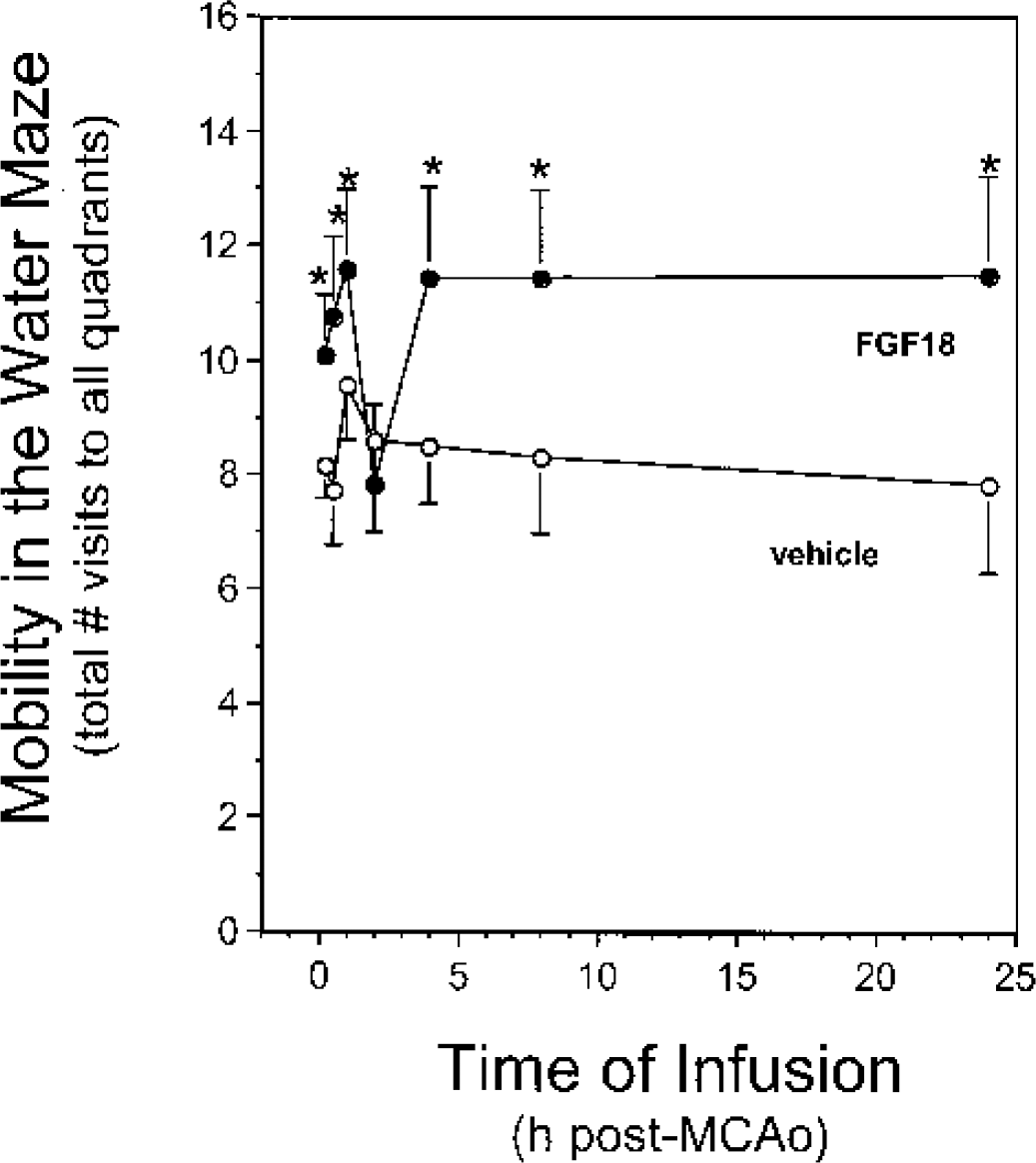
Mobility in the water maze was increased by infusion of FGF18. MCAO animals were prepared and infused with either vehicle (open symbols) or FGF18 (solid symbols) at various times after ischemia as described in Materials and Methods. The total number of visits to all quadrants in the third trial of the reference memory test was measured. The following difference was significant: vehicle vs. FGF18, *P < 0.0006.
For preischemic rats, latency in the working memory test decreased from 34.0 ± 2.5 seconds in the first trial to 22.5 ± 2.8 seconds in the second trial (mean ± SD, n = 183 animals per trial, P < 0.0001), a change consistent with intact short term memory in these animals. In MCAO rats treated with vehicle, trial 1 latency increased to an average of 53.7 ± 2.6 seconds (P < 0.0001, Fig. 6, left panel), values close to the maximum time (60 seconds) allowed in this test. For the vehicle infused rats, latencies in the second trial decreased with time of infusion out to 8 hours after MCAO (Fig. 6, right panel), observations consistent with a modest recovery of short-term memory in these animals. Relative to vehicle infusions, early infusion of FGF18 (≤ 1 hours and ≤ 2 hours after MCAO for trials 1 and 2, respectively) reduced both the trial 1 and trial 2 working memory latencies (Fig. 6, left and right panels, respectively). Little or no improvement in working memory scores was seen for infusions delayed past these times (Fig. 6). These data indicate that early infusion of FGF18 reduced MCAO-induced deficits in working memory.
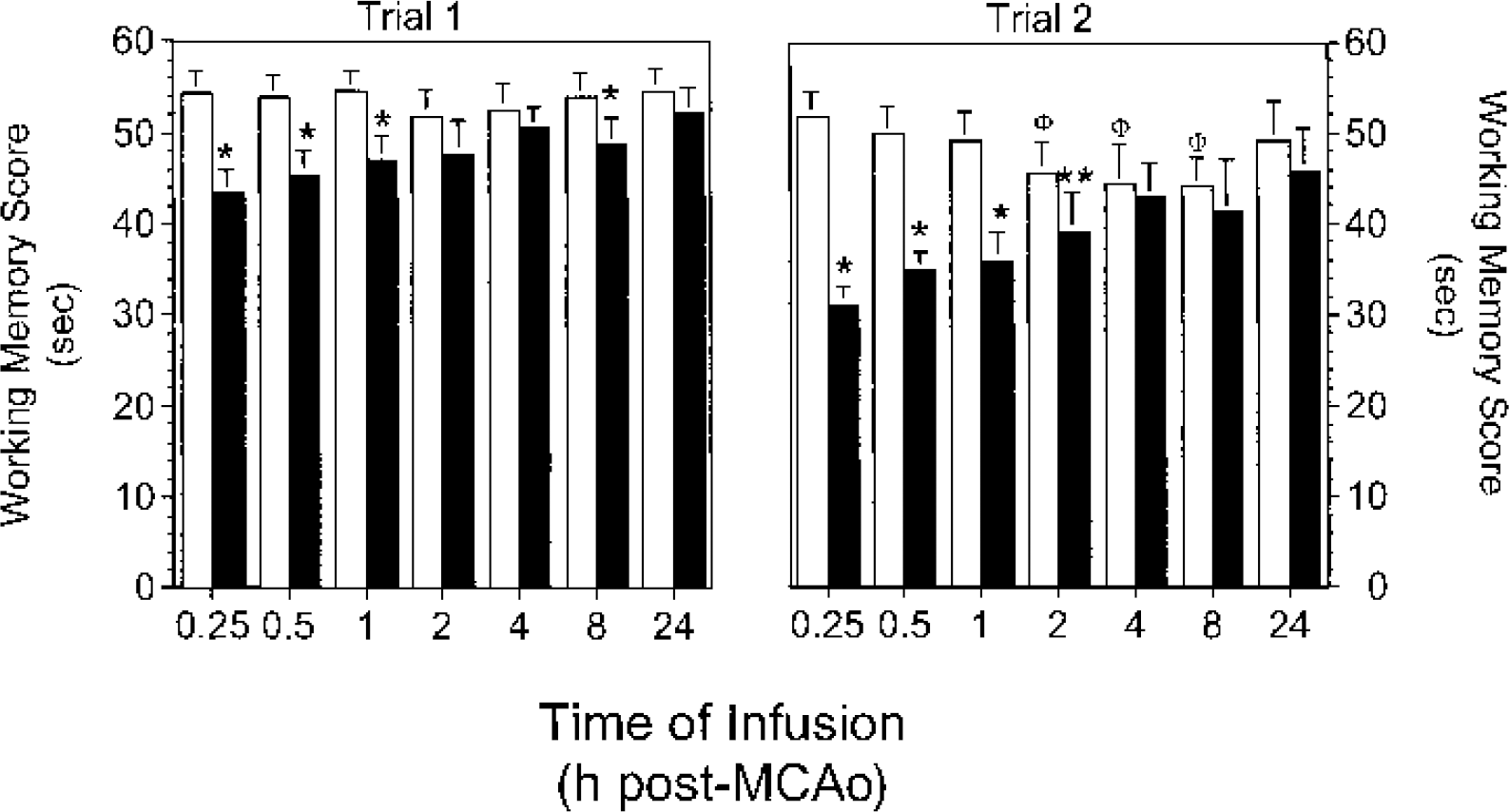
Early infusion of FGF18 reduced the MCAO-induced deficits in working memory. MCAO animals were prepared and infused with either vehicle (open symbols) or FGF18 (solid symbols) at various times after ischemia and working memory was assessed. The following differences were significant: vehicle vs. FGF18, *P < 0.002, **P < 0.01; compared to vehicle infusion at 0.25 hours, ΦP < 0.002.
Relative to preischemic rats, vehicle-treated animals with MCAO exhibited considerable deficits in tests of exploratory behavior and general activity (Fig. 7a) and in the beam walk test (Fig. 7b). The beam walk score worsened for rats infused with vehicle at 24 hours after MCAO compared to those infused at 8 hours after MCAO (5.0 ± 0.4 vs. 3.9 ± 0.3, mean ± SD, P < 0.0006). Early administration of FGF18 reduced MCAO-induced deficits in the exploratory behavior and beam walk tests (Fig. 7). Infusion of FGF18 at 24 hours after MCAO also prevented the increase in beam walk deficit seen in animals infused with vehicle at this time (Fig. 7b). These data indicate that early infusion of FGF18 provided the greatest benefit in reducing the MCAO-induced deficits in exploratory behavior and motor activity.
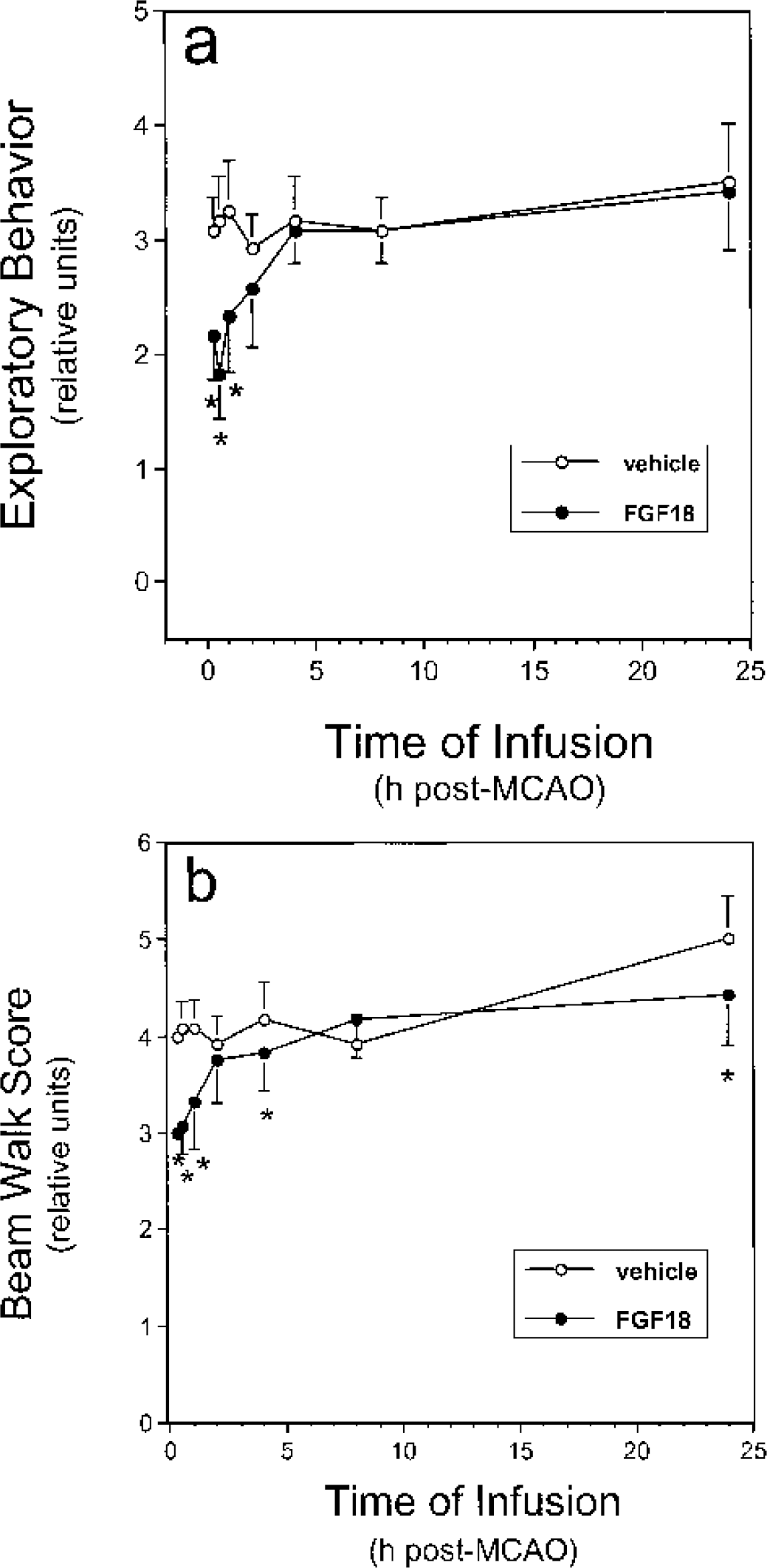
FGF18 reduced deficits in the exploratory behavior and beam walk tests in MCAO rats. MCAO animals were prepared and infused with either vehicle (open symbols) or FGF18 (solid symbols) at various times after MCAO. Exploratory behavior/general activity (
Similar to our previous findings (Ellsworth et al., 2003), MCAO reduced rCBF in the ischemic hemisphere to about 20% of the preischemia values. Removal of the filament after 2 hours increased rCBF and resulted in a reactive hyperemia (10% to 20% above pre-MCAO values) that resolved over the next hour to pre-MCAO levels. Regional CBF was increased during the period of occlusion in rats treated with FGF18 at 15 minutes (Fig. 8a, solid circles), 30 minutes (Fig. 8a, open circles), and 1 hour (Fig. 8a, open triangles) after MCAO. Infusion of FGF18 at 2 hours (Fig. 8a, open diamonds) or at later times (data not shown) after MCAO had no measurable effects on rCBF. Infusion of vehicle had no significant effects on rCBF at any infusion time (Fig. 8b). Similar to data reported previously (Ellsworth et al., 2003), these data show that early administration of FGF18 increased rCBF during the period of MCAO in the ischemic hemisphere.
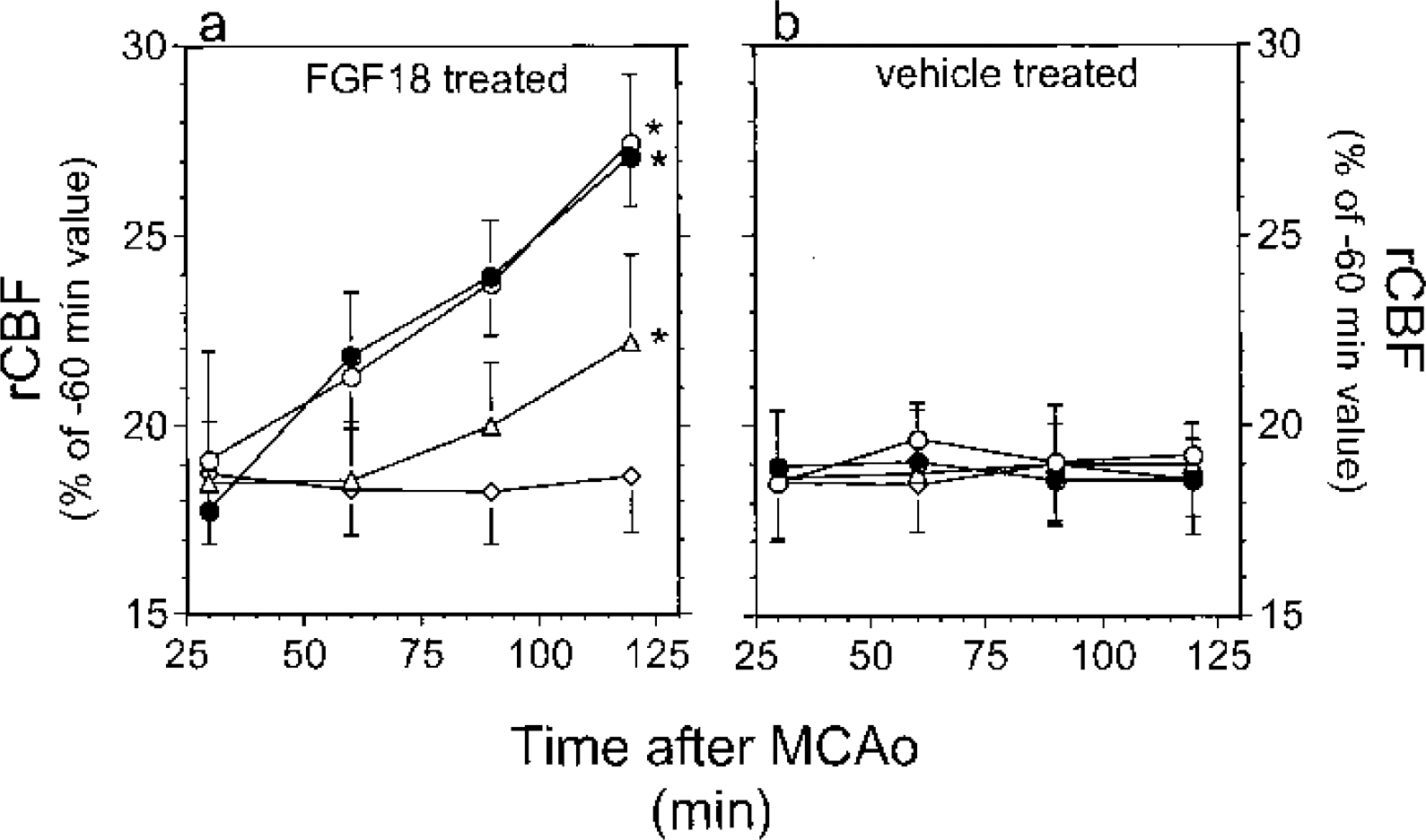
Early administration of FGF18 increased rCBF in MCAO rats. MCAO animals were prepared and infused with either vehicle (right panel) or FGF18 (left panel) at 15 minutes (solid circles), 30 minutes (open circles), 1 hour (triangles), or 2 hours (diamonds) after MCAO. rCBF in the ischemic hemisphere was assessed as described in Materials and Methods. *P < 0.0001.
There were no significant differences in blood pH (Table 1) or brain temperatures (Table 2) between the groups. Neither MCAO nor infusion of vehicle or FGF18 had any significant effects on MABP (data not shown, Ellsworth et al., 2003). Relative to the preischemia values, a small decrease in P
Physiologic parameters in rats before and after MCAO and infusion of FGF18
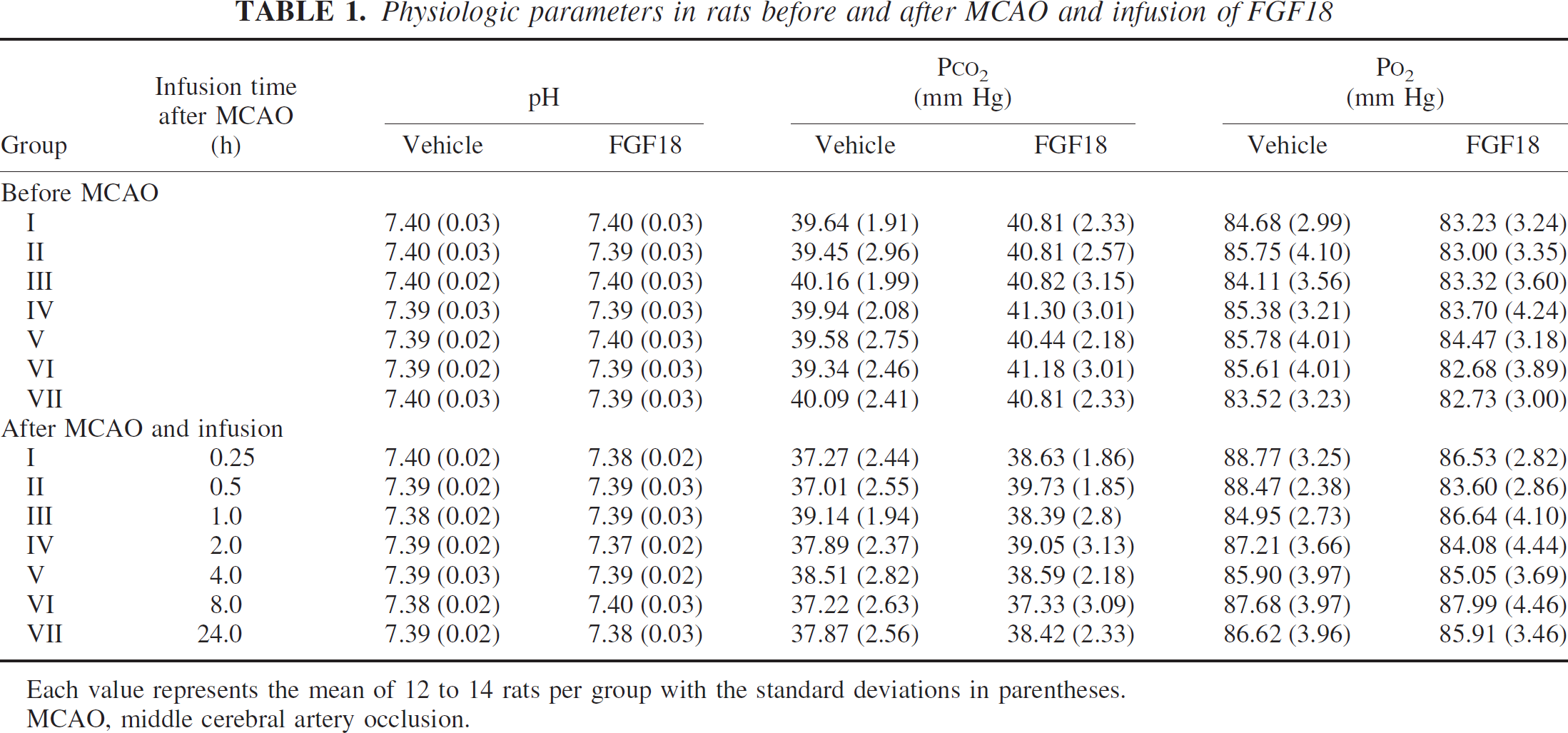
Each value represents the mean of 12 to 14 rats per group with the standard deviations in parentheses.
MCAO, middle cerebral artery occlusion.
Brain temperature in rats before and after MCAO and infusion of FGF18
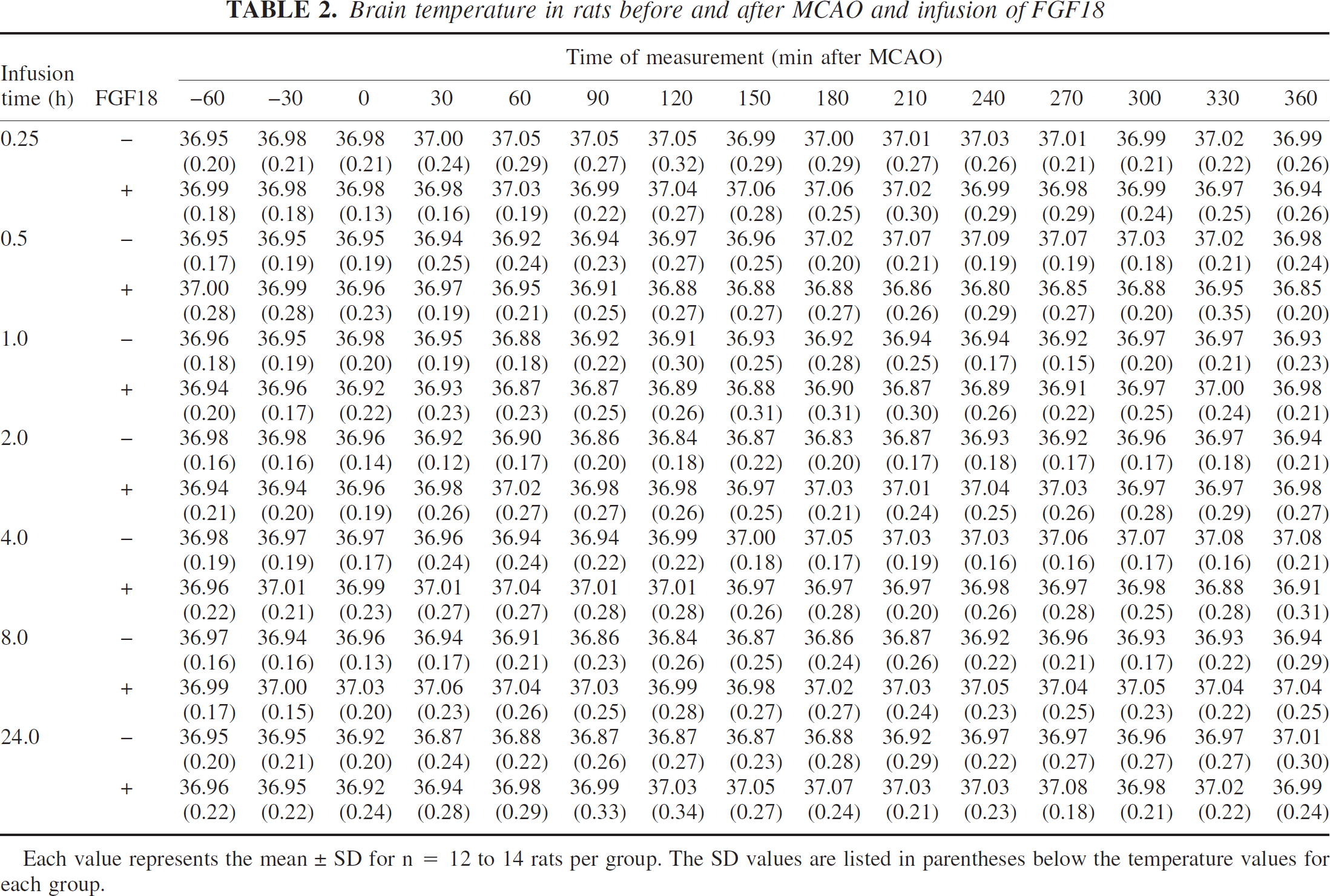
Each value represents the mean ± SD for n = 12 to 14 rats per group. The SD values are listed in parentheses below the temperature values for each group.
DISCUSSION
The results of the present study show that intravenous infusion of FGF18 reduced infarct volumes and behavioral deficits after transient MCAO in rats. As seen with both thrombolytics (Gladstone and Black, 2001; Ringleb et al., 2002) and neuroprotective agents (Fisher and Ratan, 2003; Gladstone et al., 2002), FGF18 was most efficacious when administered within the first several hours after MCAO. Indeed, the largest reductions in infarct volumes and behavioral deficits were seen for FGF18 infusions beginning within the first 2 or 3 hours after induction of ischemia. These data are consistent with those reported for other neuroprotective agents (Gladstone et al., 2002; Lees, 2001; Read, 2000), including FGF2 (Ay et al. 1999), after transient or permanent ischemia in rodents. In addition to the effects observed with early administration, FGF18 also significantly reduced both infarct volumes and deficits in several key behavioral parameters with intravenous infusions beginning at 24 hours after MCAO. These data suggest that the time window for FGF18-mediated neuroprotection may be considerably longer than the first few hours after ischemia.
The time window for neuroprotective or thrombolytic therapy is thought to directly depend on the amount of compromised but salvageable tissue, the “penumbra,” surrounding the infarct core (Dirnagl et al., 1999; Fisher and Ratan, 2003; Ginsberg et al., 1999). Although blood flow to the core has generally been reduced to a sufficient extent and time that irreversible damage has occurred (Iadecola, 1998), the penumbral area receives reduced blood flow that causes loss of function, which may progress to permanent damage if uncorrected (Garcia, 1992). It is believed that the penumbral tissue may recover if blood flow is restored and/or the ischemic cascade is blocked (Fisher and Ratan, 2003; Garcia, 1992). In support of this idea, we observed that infarct volumes and deficits in the beam walk, general activity, and reference memory tests increased in animals infused with vehicle at 24 hours, with analysis at 48 hours after MCAO, compared to those infused with vehicle at 8 hours after MCAO, with analysis at 24 hours after MCAO. These changes were reversed by infusion of FGF18 at 24 hours after MCAO. Although rCBF was not measured in the 8-hour and 24-hour infusion groups, rCBF was increased by early infusion of FGF18, suggesting that part of the tissue protection seen with FGF18 was due to an increase in rCBF. At FGF18 infusion times of 0.25 and 0.5 hours after MCAO, rCBF increased during the period of MCA occlusion from approximately 18% to 28% of the pre-MCAO values in animals. Changes in rCBF of this magnitude have been shown to produce major differences in infarct volumes after cerebral ischemia in several experimental settings (Belayev et al., 1997; Tabrizi et al., 1999). Although the mechanism for FGF18-mediated increases in rCBF is not yet understood, FGF2, which shares some receptorbinding specificity with FGF18 (Ellsworth et al., 2002), appears to be a peripheral and cerebral vasodilator acting through a nitric oxide-dependent pathway (Cuevas et al., 1991; Huang et al., 1997; Rosenblatt et al., 1994). Although disruption of cerebrovascular autoregulation after MCAO can allow for changes in MABP and/or cardiac output to increase rCBF (Iadecola, 1998), FGF18 had no significant effects on heart rate or MABP in MCAO rats (Ellsworth, et al., 2003). Taken together, these data suggest that FGF18 may act locally to produce vasodilatation of the cerebral vasculature.
In addition to increasing rCBF after MCAO, FGF18 may activate other pathways leading to tissue sparing or recovery. Fibroblast growth factors, including FGF18, and their receptors play key roles in development of the mammalian CNS (Dono, 2003). In preliminary in situ hybridization analyses (Yao L, Ellsworth JL, unpublished data, 2001), we observed widespread Fgf18 expression within neurons of adult mouse brain. Hippocampal expression of Fgf18 appears to play a key role in spatial learning in rats, as indicated by the finding that levels of Fgf18 mRNA in this tissue were consistently elevated during water maze training (Cavallaro et al., 2002). Exogenous administration of FGF18, moreover, increased the acquisition and retention of spatial memory in these animals (Cavallaro et al., 2002). These data support the results of the present report and our previous work (Ellsworth et al., 2003), which showed improved reference memory scores in MCAO rats treated with FGF18. Other activities of FGF18 are consistent with a neuroprotective role for this factor, including its ability to promote neurite outgrowth in PC-12 cells (Ohbayashi et al., 1998) and its mitogenic activity for astrocytes and microglia (Hoshikawa et al., 2002). Although the neurotrophic activity of FGF18 appears to be less than that of FGF2 (Hoshikawa et al., 2002), we have consistently observed that FGF18 promoted the survival of primed or unprimed neurite-bearing PC-12 cells and neuronal-like cells derived from newborn or fetal mouse brain cultures (Garrigues U, Ellsworth JL, unpublished observations, 2001). FGF2 promotes neuronal cell survival (Ay et al., 1999) and exhibits a variety of antiischemic effects on neuronal tissues in vivo and in vitro (Ay et al., 1999) possibly through altering the levels of Bcl-2 (Ay et al. 2001) or activin A (Tretter et al. 2000) within brain tissue. FGF2-mediated alterations of these or similar pathways (Barone et al., 2001; Datta et al., 1999; Liou et al., 2003) may explain the reduction of infarct volume seen with FGF2 after MCAO in eNOS-deficient mice (Huang et al., 1997). Through their interaction with a similar subclass of FGF receptor (Ellsworth et al., 2002), FGF18 and FGF2 may regulate tissue protection through similar cellular mechanisms.
It should be noted that the effects observed in the present study were seen after a single intravenous administration of FGF18. The dose used, FGF18 (100μg · kg−1 · h−1), was not maximal (Ellsworth et al., 2003), suggesting that a higher FGF18 dose or repeated administration of the factor may produce more extensive reductions in infarct volumes and improvements in behavioral scores. It is also important to consider that the present studies used a relatively short observation period to assess the efficacy of FGF18. Evidence from animal models and human stroke patients suggests that infarct volume can increase with time after cerebral ischemia (Beaulieu et al., 1999; Garcia, 1992; Matsui et al., 2002; Zhang et al., 1994). Apoptosis may contribute to delayed infarction after mild focal ischemia (Du et al., 1996; Li et al., 2000; Li et al., 1998) and temporary protection by other tissue protective agents has also been reported (Colbourne et al., 1999; Dietrich et al., 1993). In future studies, it will be necessary to thoroughly address the temporal endpoint for measurement of lesion volume and behavioral recovery after FGF18 administration, the dose-response relationship of FGF18, and the FGF18 dosing interval in animal models of cerebral ischemia. This is particularly important in light of the disappointing experience with FGF2 in human clinical trials of stroke (Bogousslavsky et al., 2002).
In conclusion, these data show that early administration of FGF18 provided the greatest efficacy in reducing infarct volumes and behavioral deficits in a short-term model of MCAO in rats. These data are consistent with the evolving concept that stroke should be treated as a medical emergency (American Heart Association, 2001) and for efficacy, thrombolytics and neuroprotectants must be administered within the first several hours after the onset of cerebral ischemia (American Heart Association, 2001; Gladstone et al., 2002). Interestingly, the reduction in tissue damage and behavioral deficit seen at later times in the present study suggests the time window for FGF18-mediated neuroprotection is longer than the first few hours after cerebral ischemia.
Footnotes
Acknowledgements
The authors thank the Analytical Sciences Department at ZymoGenetics for the preparation and analysis of the FGF18 samples for this study.
