Abstract
Large infarcts develop in pinealectomized rats subjected to middle cerebral artery occlusion, which was attributed to loss of antioxidant action of melatonin. However, melatonin also has vascular actions, and pinealectomy may induce hypertension. The authors investigated (I) whether hemodynamic factors contribute to infarct development in pinealectomized rats, (2) whether melatonin administration can reverse the unfavorable effect of pinealectomy on infarct formation, and (3) whether melatonin can reduce the infarct volume in nonpinealectomized rats subjected to focal transient ischemia (2 hours middle cerebral artery occlusion, 22 hours reperfusion). Rats were pinealectomized 3 months before ischemia to eliminate any possible action of pinealectomy-induced hypertension on stroke. Blood pressure and regional CBF values during ischemia and reperfusion were not significantly different between pinealectomized and sham-operated rats, suggesting that pinealectomy-induced increase in infarct was not related to hemodynamic factors. The infarct volume resumed to the level of sham-operated rats on melatonin administration. Injection of melatonin (4 mg/kg) before both ischemia and reperfusion reduced infarct volume by 40% and significantly improved neurologic deficit scores in pinealectomized as well as sham-operated rats subjected to middle cerebral artery occlusion. These data suggest that physiologic melatonin release as well as exogenously given melatonin has a neuroprotective action in focal cerebral ischemia.
Lack of a serious toxitity of melatonin, a powerful antioxidant, has recently evoked hopes that it might be a promising drug for treatment of various diseases thought to be caused by oxidative damage (Reiter et al., 1997b). Melatonin has also been proposed as a neuroprotective agent as it readily penetrates the blood-brain barrier and diffuses into neurons and glia equally well (Reiter et al., 1997a). Supporting the above view, melatonin administration has been demonstrated to decrease kainic acid-induced excitotoxicity and traumatic injury in the brain (Giusti et al., 1996, 1997; Mesenge et al., 1998). Moreover, larger lesions developed when pinealectomized rats were subjected to intracerebral kainic acid injections or to middle cerebral artery (MCA) occlusion (Manev et al., 1996). Although the latter findings suggest a protective action by physiologic levels of melatonin against ischemic brain damage and excitotoxicity, increased vulnerability of the brain might also have been caused by other changes that pinealectomy induced (Moller et al., 1996).
Pinealectomy leads to an increase in systemic arterial blood pressure (Karppanen et al., 1975; Zanoboni et al., 1978; Vaughan et al., 1979) in addition to a drastic reduction in circulating levels of melatonin (Ozaki and Lynch 1976; Lewy et al., 1980; Bubenik and Brown, 1997). Cerebral vessels express melatonin receptors (Viswanathan et al., 1990, 1997), and in vitro, melatonin has been shown to induce concentration-dependent vasoconstriction or dilation (Geary et al., 1997; Mahle et al., 1997). Melatonin administration to 14-day-old rats has been found to cause a reduction in cerebral blood flow (Capsoni et al., 1995). Therefore, determining the arterial blood pressure and cerebral blood flow appears to be warranted to test any possible contribution of hemodynamic factors to the unfavorable effect of pinealectomy on brain damage.
In this study, we monitored the arterial pressure and regional cerebral blood flow (rCBF) in pinealectomized rats subjected to MCA occlusion. We also administered varying doses of melatonin before induction of ischemia or reperfusion to determine whether the unfavorable action of pinealectomy on cerebral ischemia could be reversed by melatonin and to find the optimum dose and timing of melatonin administration. Our data suggest that the large infarcts in pinealectomized rats are caused by changes in brain parenchyma induced by melatonin deficiency but not by hemodynamic factors. The infarct volume resumed to the level of sham-operated rats on administration of melatonin. A combined administration of melatonin before both ischemia and reperfusion led to a significant decrease in infarct in pinealectomized as well as sham-operated rats subjected to MCA occlusion, strongly supporting the role of melatonin as a neuroprotective agent.
METHODS
Experimental groups
Wistar rats weighing 230 to 270 g were housed under diurnal lighting conditions (12 hours darkness and 12 hours light) and fasted overnight but allowed free access to water before the experiment. Animal housing, care, and application of experimental procedures were all done in accordance with institutional guidelines. Rats were anesthetized with ketamine hydrochloride (75 mg/kg) and xylazine (8 mg/kg). Body temperature was monitored by a rectal probe and maintained at 36.5° to 37.5°C by a homeothermic blanket control unit. The tail artery was cannulated for continuous arterial blood pressure monitoring, for obtaining blood samples to determine pH, P
Melatonin (Sigma Chemical Co., St. Louis, MO, U.S.A.) was dissolved in ethanol and further diluted in saline to give a final ethanol concentration of 5%.
Pinealectomy
Pinealectomy was performed as described by Kuszak and Rodin (1977). Briefly, anesthetized rats were placed in a stereotaxic frame and a longitudinal midline incision was made and extended to the occipital ridge. The sagittal and lambdoid sutures were then exposed by scraping the periosteum. A rectangular piece of the calvarium was opened and the superior sagittal vein (SSV), transverse sinuses, and confluens sinuum were exposed. Great care was exercised in drilling and removing the piece of bone so as not to cause hemorrhage from the underlying sinuses. The dura was then cut, and the SSV was double-ligated with 6-0 silk surgical threads, placed under the SSV with a curved atraumatic needle, and resected. The pineal gland was exposed beneath the confluens sinuum by reflecting the caudal portion of the ligated SSV posteriorly and was removed with fine forceps. The procedure was completed by returning the reflected SSV to its original position, reapposing the skin, and closing the incision. The entire procedure was completed within 15 minutes.
MCA occlusion
Proximal occlusion of the right MCA was performed by a nylon filament as described before (Longa et al., 1989; Memezewa et al., 1992). After a midline incision, the right common and external carotid arteries were ligated with a 5-0 silk suture. A 4-0 nylon filament was inserted into the common carotid artery through a small incision 1 to 2 mm proximal to the bifurcation. The filament was prepared by blunting the tip near a flame and its distal 5 mm was coated with cyanoacrylate glue. Middle cerebral artery occlusion was accomplished by further advancing the filament in the internal carotid artery until a faint resistance was felt.
A small burr hole was made over the cortex (6 mm lateral and 2 mm posterior to bregma), and a flexible probe (PF-318 of PeriFlux PF 2B, Perimed, Jarfalla, Sweden) was placed over the dura, away from large pial vessels, to monitor the rCBF by laser-Doppler flowmetry. After obtaining a stable 10-minute period of preischemic rCBF, the MCA was occluded and the rCBF was continuously monitored during ischemia (2 hours) and for the first 20 minutes of reperfusion.
Reperfusion was accomplished by pulling the filament back. Animals that were not successfully reperfused (n = 2, 4.5% of the rats studied) or had subarachnoid hemorrhage (n = 4, 9%) were excluded from the study.
Evaluation of ischemic area by 2,3,5-triphenyl tetrazolium chloride staining
The ischemic area was evaluated by 2,3,5-triphenyl tetrazolium chloride staining (Bederson et. al., 1986b). Briefly, rats were killed by decapitation after 2 hours of ischemia and 22 hours of reperfusion. The brains were quickly removed and placed in ice-cold saline for 5 minutes and then cut into 2-mm coronal slices. Sections were incubated in 2,3,5-triphenyl tetrazolium chloride containing saline solution (2%; Sigma) for 20 minutes and in 10% formalin overnight. The infarction area, outlined in white, was measured by image analysis software (NIH Image 1.59) on the posterior surface of each section, and infarction volume was calculated by summing the infarct volumes of sequential 2-mm thick sections.
Neurologic evaluation
Twenty-two hours after recirculation, neurologic deficits were assessed by an observer naive to the identity of treatment and scored as described previously (Bederson et al., 1986a; Huang et al., 1994) with a minor modification as follows: 0, no observable neurologic deficit (normal); 1, failure to extend left forepaw on lifting the whole body by the tail (mild); 2, circling to the contralateral side (moderate); and 3, leaning to the contralateral side at rest or no spontaneous motor activity (severe).
Statistics
Mean values of arterial blood gases, pH, blood pressure, rCBF, infarct volume, and neurologic deficit scores were compared by analysis of variance and Fisher's protected least-significant difference test was used for post-hoc analysis. Probability values smaller than 0.05 were considered to be significant. Mean values in the text are given with their standard deviations.
RESULTS
Arterial blood gases, pH, and pressure were not significantly different between the experimental groups, although pinealectomized (Px) rats had slightly lower blood pressure values than sham-operated (non-Px) rats. Regional CBF values during MCA occlusion and reperfusion were also not significantly different between the groups (Table 1).
Cerebral blood flow values and physiologic variables in experimental groups
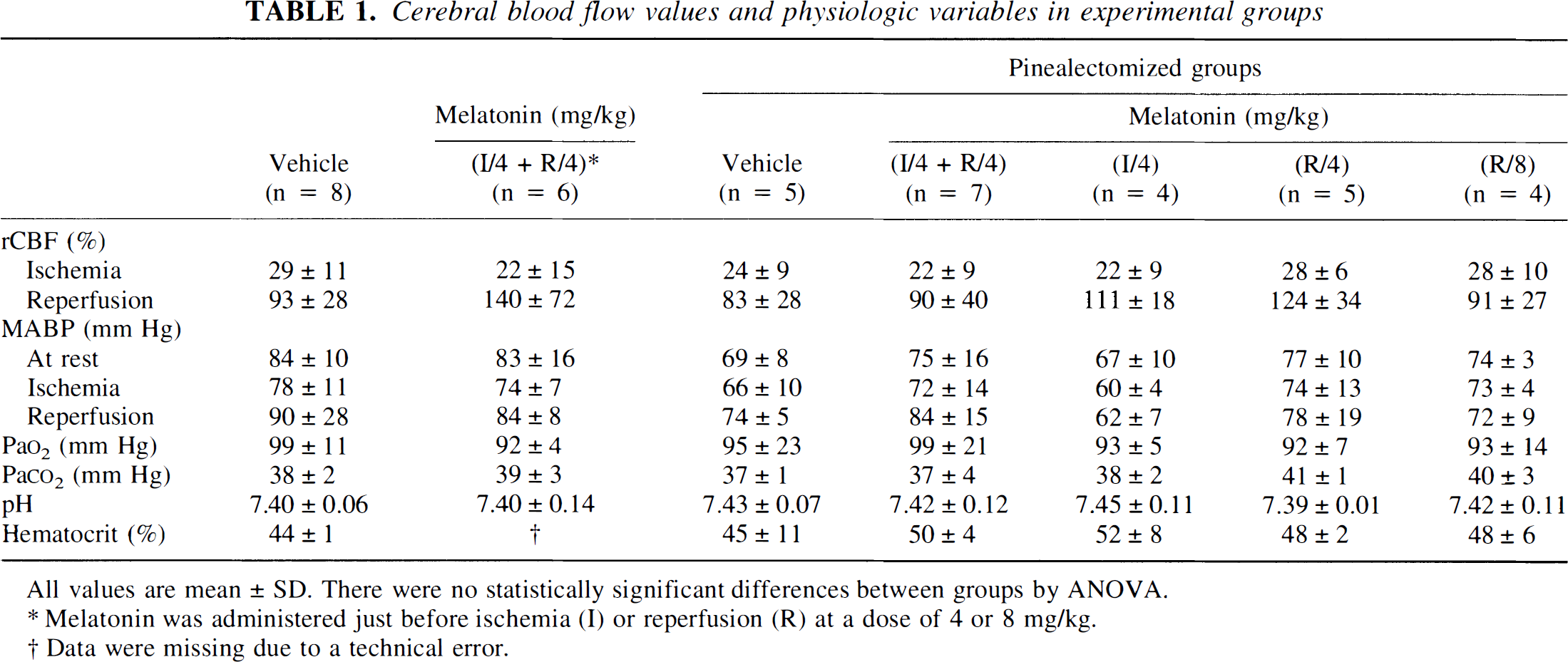
All values are mean ± SD. There were no statistically significant differences between groups by ANOVA.
Melatonin was administered just before ischemia (I) or reperfusion (R) at a dose of 4 or 8 mg/kg.
Data were missing due to a technical error.
Infarct volume
Pinealectomy caused a significant increase in infarct volume. Two-hour MCA occlusion followed by 22 hours of reperfusion led to 232 ± 74 and 306 ± 16 mm3 infarcts in non-Px and Px rats, respectively (Fig. 1A). Administration of 4 mg/kg of melatonin to Px rats just before the induction of ischemia or reperfusion decreased the infarct volume to values comparable to those of the non-Px group. The infarct volumes were 227 ± 66 and 188 ± 58 mm3 for groups given melatonin before ischemia or reperfusion, respectively. Although these values were significantly smaller than the mean infarct volume of the vehicle-treated Px group, they were not significantly different from that of the non-Px group (232 ± 74 mm3). Increasing the dose administered before reperfusion to 8 mg/kg also did not achieve a significantly reduced infarct volume (212 ± 18 mm3). Combined administration of 4 mg/kg melatonin to Px rats before both ischemia and reperfusion, however, led to a further reduction in the infarct volume to 140 ± 58 mm3 (60% of the infarct in the non-Px group). A similar protection with combined melatonin administration was also obtained in the non-Px group subjected to MCA occlusion (151 ± 66 mm3).
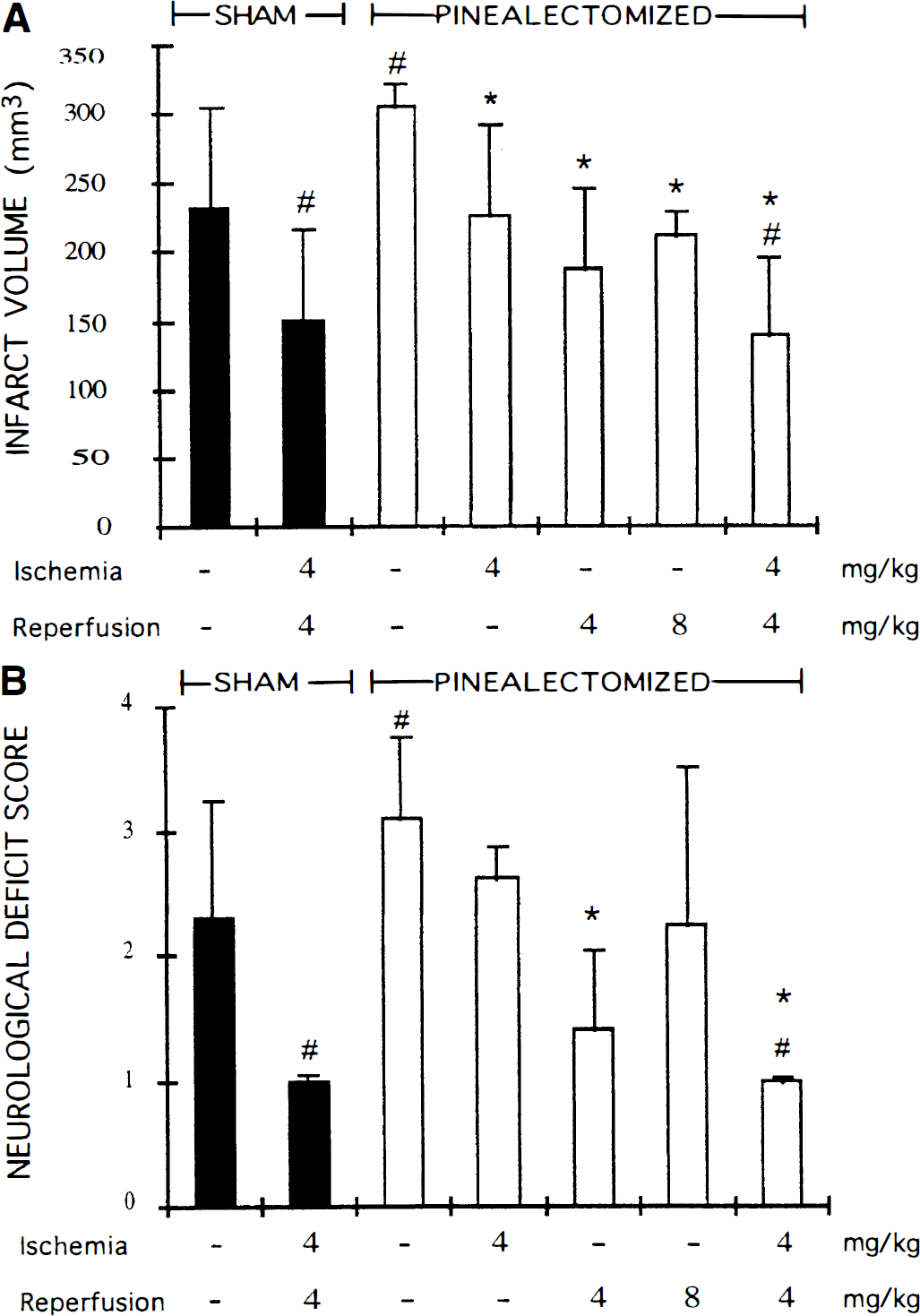
Infarct volumes (
Neurologic evaluation
Mean scores of all groups indicating the neurologic disability of rats 22 hours after reperfusion paralleled the changes in infarct volume (Fig. 1B). Pinealectomized rats had significantly higher disability scores than those of non-Px rats. Melatonin (4 mg/kg) administration before reperfusion significantly reduced the disability of Px animals to the level of non-Px rats. Unlike the infarct volumes, neurologic deficit scores of groups in which 4 mg/kg melatonin was given before ischemia or 8 mg/kg melatonin was given before reperfusion were not significantly lower than scores of the Px group, possibly because of insufficient number of animals in these groups to overcome the variability in neurologic disability. The combined administration of melatonin before both ischemia and reperfusion led to a further reduction in neurologic dysfunction, which was significantly less than that of the non-Px group. Nonpinealectomized rats subjected to 2 hours of MCA occlusion and 22 hours of reperfusion and given combined melatonin treatment also had significantly lower disability scores than vehicle-treated non-Px rats.
DISCUSSION
Manev's group was the first to report that pinealectomized rats were more vulnerable to kainate-induced excitotoxicity and photothrombotic brain injury (Manev et al., 1996). They also demonstrated that larger infarcts developed in pinealectomized rats subjected to 1 hour of MCA occlusion (Manev et al., 1996). However, the arterial blood pressure and cerebral blood flow were not monitored in these pioneering studies. Although the antioxidant property of melatonin might account for its neuroprotective effect, monitoring cerebral blood flow is indispensable in pinealectomized rats because they may be hypertensive for the first 60 days after pinealectomy (Zanoboni et al., 1978; Vaughan et al., 1979) and also because melatonin has been reported to have a regulatory action on cerebral blood flow (Viswanathan et al., 1990, 1997; Mahle et al., 1997; Geary et al., 1997). We used rats that were pinealectomized 3 months before MCA occlusion to eliminate any possible action of pinealectomy-induced hypertension on stroke outcome (Zanoboni et al., 1978). Indeed, the mean arterial pressure was not significantly different between experimental groups. More importantly, rCBF changes during MCA occlusion and reperfusion were also not significantly different between pinealectomized and sham-operated rats, suggesting that observed changes in the infarct volume of pinealectomized rats were not related to hemodynamic factors. However, we cannot totally exclude the possibility that a lower basal cerebral blood flow level in pinealectomized rats may have contributed to the increase in infarct volume because the laser-Doppler flowmetry technique we used detects only relative changes in tissue perfusion. However, this is a very unlikely possibility because melatonin has been reported to cause a reduction in basal rCBF in 14-day-old rats (Capsoni et al., 1995); thus, melatonin deficiency is expected, if anything, to cause an increase in basal rCBF.
The pineal gland is the major source of circulating melatonin, and the extrapineal sources of melatonin do not compensate for melatonin deficiency caused by pinealectomy (Ozaki and Lynch 1976; Lewy et al., 1980; Bubenik and Brown, 1997). Our data show that the unfavorable action of pinealectomy on cerebral infarct development can be reversed by melatonin administration, supporting the idea that melatonin deficiency may be the factor potentiating the ischemic damage in pinealectomized rats. However, more convincing evidence can perhaps be obtained in future studies by chronic nocturnal administration of melatonin in physiologically relevant doses. Sham-operated rats were also protected by melatonin, indicating that exogenously given melatonin has a neuroprotective action in cerebral ischemia in animals with a normal diurnal secretion of melatonin as well. Melatonin reduced infarct volume by 40% and significantly improved neurologic deficit in this transient focal ischemia model. In line with these findings, melatonin administration has also been reported to decrease global ischemia-induced damage in the brain (Cho et al., 1997; Guerrero et al., 1997).
The antioxidant action of melatonin appears to be the most plausible mechanism among its known effects to account for its neuroprotective action. Neuroprotective effect of antioxidants in cerebral ischemia is well established (Hall, 1997). Albeit indirectly, maximal protection obtained with administration of melatonin just before the onset of ischemia and reperfusion, the periods when free radical generation is highest, supports this possibility (Kumura et al., 1996). Melatonin has been demonstrated to scavenge hydroxyl and peroxyl radicals (Tan et al., 1993; Pieri et al., 1994). As an antioxidant, it has been found to be more powerful than vitamin E, mannitol, and glutathione (Tan et al., 1993; Pieri et al., 1994). Melatonin has also been shown to decrease oxidative damage to DNA (Tan et al., 1994), proteins (Abe et al., 1994), and lipids (Melchiorri et al., 1994, 1995) under various in vivo pathologic conditions. Recently, melatonin administration has been reported to attenuate kainic acid-induced oxidative damage to DNA in the brain (Uz et al., 1996). Interestingly, pinealectomy has been demonstrated to increase safrole-induced oxidative DNA damage in the liver, suggesting that physiologic melatonin release might have an antioxidant activity (Tan et al., 1994).
However, loss of a direct antioxidant effect of melatonin in pinealectomized rats cannot be readily associated with the enlarged infarcts because sham-operated rats had also very low plasma melatonin levels when they were subjected to MCA occlusion during the daytime. Nocturnal release of melatonin in sham-operated rats 9 to 15 hours after reperfusion is not expected to have a neuroprotective effect because the majority of cells in the ischemic territory become irreversibly injured by that time (Zhang et al., 1994). There is increasing evidence suggesting that melatonin secretion may modulate the antioxidant enzyme systems, which may account for the ongoing protective effect of melatonin during the daytime. Pharmacologic levels of melatonin have been reported to stimulate glutathione peroxidase and glucose-6-phosphate dehydrogenase activity in the brain (Barlow-Walden et al., 1995, Pierrefiche and Laborit, 1995), and nocturnal melatonin release has been shown to coincide with peak neuronal glutathione peroxidase activity (Pablos et al., 1998). Melatonin increases brain mRNA levels of glutathione peroxidase and copper-zinc and manganese superoxide dismutases (Antolin et al., 1996; Kotler et al., 1998). Melatonin also inhibits the activity of some enzymes whose excessive activity may be harmful under pathologic conditions, such as nitric oxide synthase (Pozo et al., 1994; Guerrero et al., 1997) and 5-lipoxygenase (Uz et al., 1997). Pinealectomy has been shown to upregulate 5-lipoxygenase activity in the brain, and release of a tonic inhibitory effect on this enzyme has been proposed as a potential mechanism to explain the increased brain vulnerability in melatonin-deficient rats (Uz et al., 1997). If melatonin's protective action is indeed related to its regulatory action on pro-oxidant and antioxidant enzyme systems, melatonin seems to be a unique hormone regulating the organism's self-defense against oxidative damage.
In conclusion, these data strongly suggest that exogenously given melatonin as well as physiologic melatonin release has a neuroprotective action in cerebral ischemia, possibly through an antioxidant mechanism, although a causal relationship between the enlarged infarcts in pinealectomized rats and melatonin deficiency could not be established unequivocally.
Footnotes
Abbreviations used
Acknowledgments
Ertuğrul Kilic thanks Ahmet Acet, MD, for his support.
