Abstract
Cerebral vasospasm is a transient, delayed constriction of cerebral arteries that occurs after subarachnoid hemorrhage (SAH). Smooth muscle cells show impaired relaxation after SAH, which may be caused by a defect in the ionic mechanisms regulating smooth muscle membrane potential and Ca2+ permeability. We tested this hypothesis by examining changes in expression of mRNA and protein for ion channels in the basilar arteries of dogs after SAH using quantitative real-time polymerase chain reaction (PCR) and western blotting. SAH was associated with a significant reduction in basilar artery diameter to 41 ± 8% of pre-SAH diameter (P < 0.001) after 7 days. There was significant downregulation of the voltage-gated K+ channel Kv 2.2 (65% reduction in mRNA, P < 0.001; 49% reduction in protein, P < 0.05) and the β1 subunit of the large-conductance, Ca2+-activated K+ (BK) channel (53% reduction in mRNA, P < 0.02). There was no change in BK β1 subunit protein. Changes in mRNA levels of Kv 2.2 and the BK-β1 subunit correlated with the degree of vasospasm (r2 = 0.490 and 0.529 respectively, P < 0.05). The inwardly rectifying K+ (Kir) channel Kir 2.1 was upregulated (234% increase in mRNA, P < 0.001; 350% increase in protein, P < 0.001). There was no significant change in mRNA expression of L- type Ca2+ channels and the BK-α subunit. These data suggest that K+ channel dysfunction may contribute to the pathogenesis of cerebral vasospasm.
Keywords
Rupture of a cerebral aneurysm deposits blood in the subarachnoid space, creating subarachnoid hemorrhage (SAH). The most common cause of morbidity and mortality in survivors of SAH is vasospasm, which is constriction of the cerebral arteries that starts to become significant 3 to 4 days after SAH (Macdonald, 1995; Weir et al., 1978). This constriction becomes most severe 7 days after the hemorrhage and then slowly resolves as vessels return to normal caliber by 14 days. If the constriction is severe, brain infarction can develop in the region supplied by the constricted artery. Clinical effects of vasospasm are manifested in approximately one third of patients with SAH (Macdonald and Weir, 2001). Vasospasm doubles the death rate from SAH and reduces good outcomes by 30% (Dorsch and King, 1994).
Cerebral artery narrowing in vasospasm is caused by sustained smooth muscle contraction rather than by proliferative phenomena (Findlay et al., 1989; Macdonald et al., 1991, 1992; Mayberg et al., 1990). Previous studies from our laboratory demonstrated that there is increasing impairment of smooth muscle relaxation as vasospasm progresses after SAH (Macdonald et al., 1995; Zhang et al., 2001). A major relaxation pathway in cerebral vascular smooth muscle is mediated by nitric oxide (NO) produced in endothelial cells by endothelial NO synthase (NOS), which leads to an increase in cyclic GMP in vascular smooth muscle. This results in activation of cyclic GMP-dependent protein kinase (PKG) and subsequent potentiation of large-conductance, Ca2+-activated K+ (BK) channels (Robertson et al., 1993) and inhibition of L-type Ca2+ channels (Simard et al., 1998). We previously investigated the proximal portion of this pathway and found that several classes of NO donors failed to prevent experimental cerebral vasospasm in primates despite their ability to increase cyclic GMP levels (Aihara et al., 2003; Macdonald et al., 2002). These findings suggest that alteration(s) in vasodilatory mechanisms further downstream may contribute to vasospasm.
One possibility is that membrane ion channels, which are end-targets of relaxation pathways, are themselves affected by SAH, thus leading to vasospasm. Indirect evidence exists for K+ channel dysfunction in vasospasm (Harder et al., 1987; Quan and Sobey, 2000). Vasospastic smooth muscle cells have been reported to be depolarized relative to controls in various animal models of SAH (Harder et al., 1987; Waters and Harder, 1985; Zuccarello et al., 1996). Furthermore, it was suggested that reduced membrane K+ conductance was important in this depolarization although the specific K+ channels responsible were not identified. Smooth muscle membrane potential is a key determinant of cerebrovascular tone by virtue of its role in regulating Ca2+ influx via L-type Ca2+ channels and membrane potential is modulated primarily by K+ conductance (Nelson et al., 1990; Standen and Quayle, 1998). To begin to understand the role of membrane potential and smooth muscle membrane ion channels in vasospasm, we used a quantitative real-time polymerase chain reaction (PCR) assay to measure messenger ribonucleic acid (mRNA) levels of relevant ion channels in a canine double hemorrhage model of cerebral vasospasm. The main K+ channels regulating tone in cerebral arteries were investigated. We report for the first time to our knowledge the main forms of voltage-gated K+ (Kv), inward-rectifier (Kir) and Ca2+-activated K+ channels that are present in dog basilar artery. In addition, we document the new finding of a significant downregulation of Kv 2.2 and of the β1 subunit of the BK channel accompanied by upregulation of Kir 2.1. Changes in Kir and Kv channels were accompanied by corresponding changes in protein, whereas there was no reduction in BK-β1 protein. The decrease in Kv mRNA in particular correlated with the degree of cerebral vasospasm, suggesting a role for K+ channel dysfunction in the pathogenesis of vasospasm.
MATERIALS AND METHODS
Animal model
Thirty-eight mongrel dogs weighing 15 to 25 kg were randomly divided into three groups: (A) control group (no treatment, n = 17), (B) SAH group (SAH with 0.5 mL/kg autologous blood, n = 17), and (C) dilutional SAH group (SAH with 0.5 mL/kg autologous blood plus 1 mL 0.9% saline, n = 4). Methods for angiography and creation of SAH have been described (Macdonald et al., 1995). Briefly, dogs underwent baseline cerebral angiography on day 0. The cisterna magna was punctured percutaneously, and 0.3 mL/kg CSF was allowed to drain spontaneously. Fresh, autologous, arterial, nonheparinized blood (0.5 mL/kg) was injected into the cisterna magna at a rate of 5 mL/min. This was repeated on day 2 and resulted in consistently severe vasospasm by day 7. To generate lesser degrees of vasospasm for molecular correlation studies, autologous blood was mixed with 1 mL of 0.9% saline in four dogs (dilutional SAH group). All dogs were tilted 30° head down during cisternal blood injection and for 15 minutes thereafter. Control animals were killed after angiography on day 0, whereas all other animals underwent repeat angiography 7 days after SAH and were then killed. All procedures performed on animals were approved by the Institutional Animal Care and Use Committee.
RNA and protein processing
Animals were euthanized under general anesthesia by exsanguination and perfusion with 8 L ice-cold phosphate-buffered saline (PBS, pH 7.4) at an intraluminal pressure of 25 to 30 mm Hg to rapidly cool the head. After perfusion, the brain was rapidly excised and placed in 4°C PBS. Connective tissue surrounding the adventitia of the basilar artery was removed under an operating microscope, and any remaining intraluminal blood was flushed out. Basilar arteries were separated from the brain and immediately frozen in liquid N2. Light microscopy of basilar arteries dissected free in a similar manner documented complete absence of adjacent brain tissue. Total RNA and protein were extracted from individual basilar arteries with the use of TRIzol (Gibco BRL; Life Technologies, Rockville, MD, U.S.A.) according to the manufacturer's instructions. RNasefree DNase (Promega, Madison, WI, U.S.A.) was used to eliminate possible contamination of RNA preparations by traces of genomic DNA. RNA reverse transcription was catalyzed using a SUPER SCRIPT First-Strand Synthesis System (Invitrogen Life Technologies, Carlsbad, CA, U.S.A.). RNA (5 μg) was used for reverse transcription in a final volume of 10 μL. The reaction mixture consisted of 50 ng of random hexamers per 1 μg of total RNA. To increase the sensitivity of PCR from first-strand cDNA, we used RNase H to remove the RNA template from cDNA following first-strand synthesis. Protein (150–250 μg per basilar artery) was quantified by spectrophotometry (at 562 nm) with Micro BCA Protein Assay Reagents (Pierce Biotechnology, Rockford, IL, U.S.A.).
Quantitative real time PCR
Canine sequences of mRNAs for the following channels were obtained by searching the NCBI database (http://www.ncbi.nlm.nih.gov/Entrez): BK α and β subunits, the α subunit of the L-type Ca2+ channel, Kv subtypes 1.2, 1.5, 2.2, 3.1, 3.4, 4.3, and Kir 2.1 (kidney and atrial sources). Primers, probes, and length of PCR products were chosen to satisfy the requirements as specified in Primer Express (version 1.5) software (Applied Biosystems, Foster City, CA, U.S.A.) for quantitative RT-PCR (Table 1). Constitutively expressed 18S ribosomal RNA (18S rRNA; TaqMan Ribosomal RNA Control Reagent, Applied Biosystems) was used as an endogenous control to correct for potential variation in RNA loading or in efficiency of the amplification reaction. Quantitative PCR then was performed as described previously with real time TaqMan technology using a Sequence Detection System model 7700 (Applied Biosystems) (Aihara et al., 2001). Expression levels of target genes were evaluated by the ratio of the CT number of target mRNA to 18S rRNA rather than to other commonly used housekeeping genes, such as β-actin, becuase there is evidence that β-actin may be upregulated after SAH in dogs (Ohkuma et al., 2001). The 18S rRNA was not altered based upon quantitative measurement of this RNA compared with the total amount of RNA present in the sample (Wang et al., 2000).
Oligonucleotide sequences of dog-specific primers and probes used for real-time quantitative PCR

PCR, polymerase chain reaction.
Western blotting
Hybond™ P (PVDF) membranes were preincubated in methanol and equilibrated in ice-cold CAPS (3-[cyclohexylamino]-1-propanesulfonic acid) with 10% methanol. Equal amounts of protein were loaded in each lane and fractionated on a gel and transferred to a PDVF membrane at 60V for 2 hours. The membranes were blocked at room temperature for 1 hour in 5% milk in Tris-buffered saline with 0.1% Tween 20 for BK-β1 and 2% milk in Tris-buffered saline with 0.1% Tween 20 for Kv 2.2 and Kir 2.1. Primary antibodies were a rabbit anti-BK-β1 polyclonal antibody (1:500 dilution, Affinity Bioreagents, Golden, CO, U.S.A.), mouse monoclonal anti-Kv 2.2 antibody (1:1000 dilution, a generous of gift from J.S. Trimmer, State University of New York, Stonybrook, NY, U.S.A.) and rabbit anti-human Kir 2.1 antibody (1:200 dilution, Alomone Labs, Jerusalem, Israel). A blocking peptide also was used for the anti-Kir 2.1 antibody. After washing, the membranes were incubated for 1 hour with the following secondary antibodies: goat anti-rabbit alkaline phosphate-linked antibody (1:5000; Amersham Biosciences, Piscataway, NJ, U.S.A.) for BK-β1, horse radish peroxidase-conjugated anti-mouse antibody (1:2000 dilution: Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.) for Kv 2.2, and horse radish peroxidase-conjugated IgG anti-rabbit antibody (1:2500 dilution; Cell Signaling Technology, Beverly, MA, U.S.A.) for Kir 2.1. The BK-β1 membrane was developed using an enhanced chemifluorescence substrate (Amersham Bioscience, Arlington Heights, IL, U.S.A.) and a Storm 860 fluorimager (Molecular Dynamics, Sunnyvale, CA, U.S.A.). The enhanced chemiluminescence Plus western blotting detection system (Amersham) was used to detect Kv 2.2 and Kir 2.1. The total amount of protein extract loaded in each lane was visualized by staining the transferred PVDF membrane with GelCode Blue Stain Reagent (Pierce). Densitometric analysis was performed using a custom procedure written in Igor (Wavemetrics, Lake Oswego, OR, U.S.A.).
Statistical analysis
Comparisons between the two groups were performed using Student's t-test with α = 0.05 and between more than two groups by analysis of variance (ANOVA). Scatterplots were constructed comparing relative mRNA expression levels with angiographic arterial diameters, and the relationship between these was assessed by linear correlation. Statistical analysis was performed using SigmaStat (SPSS, Chicago, IL, U.S.A.) and Microsoft Excel. Values are expressed as mean ± SD.
RESULTS
Basilar artery diameter following SAH
SAH was associated with significant reduction in basilar artery diameter on day 7 (Fig. 1). In the SAH group, the basilar artery was decreased to 41 ± 8% of pre-SAH diameter (P < 0.001, paired t-test). The dilutional SAH group also exhibited significant vasospasm (basilar artery diameter decreased to 63 ± 8% of pre-SAH diameter, P < 0.01, paired t-test), although the reduction in diameter was significantly less than in the SAH group (P < 0.001, unpaired t-test). There were no significant differences between groups in basilar artery diameter at baseline (ANOVA).
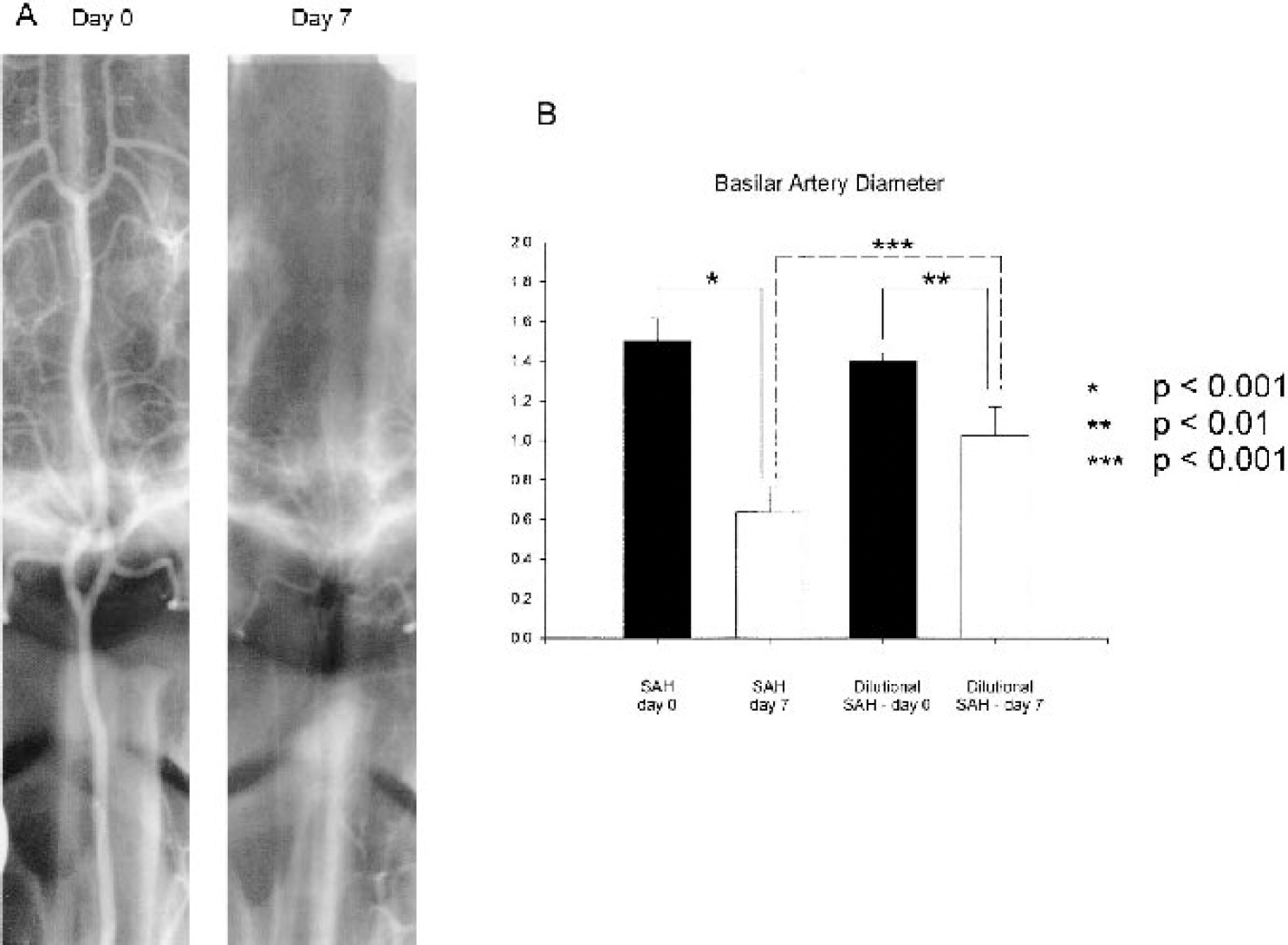
(A) Representative angiograms of dog basilar artery before (Day 0) and 7 days after SAH (Day 7). Severe vasospasm was consistently demonstrated on day 7 in the SAH group. (B) Significant reduction in artery diameter was observed on angiograms on day 7 in animals undergoing SAH or dilutional SAH compared with baseline (n = 4–17 per group, P < 0.01, paired t-tests). In addition, animals in the dilutional SAH had significantly less vasospasm than those in the SAH group (P < 0.001, unpaired t-test). SAH, subarachnoid hemorrhage.
Target gene mRNA expression
The expression level of each target gene mRNA was expressed as a ratio of its CT value to that of 18S rRNA, and normalized ratios were then compared between the control and the SAH group. Normalization to mRNA for commonly used housekeeping genes, such as β-actin and glyceraldehyde 3-phosphate dehydrogenase, was erroneous because expression of these genes was altered after SAH in dogs (R.L. Macdonald, unpublished observations, September 12, 2003). The mRNA for a large-conductance, Ca2+-activated K+ channel was identified in dog basilar artery. The BK channel has been reported to be composed of a pore-forming α subunit and a regulatory β subunit (Brenner et al., 2000). Smooth muscle contains a β1 isoform that was identified along with the α subunit. The expression level of the BK-α subunit was not altered after SAH, but there was a significant reduction in BK-β1 subunit after SAH compared with control (53% reduction, P < 0.02) (Fig. 2). There were no significant differences in the mRNA expression of the L-type Ca2+ channel between control and SAH groups (Fig. 2).
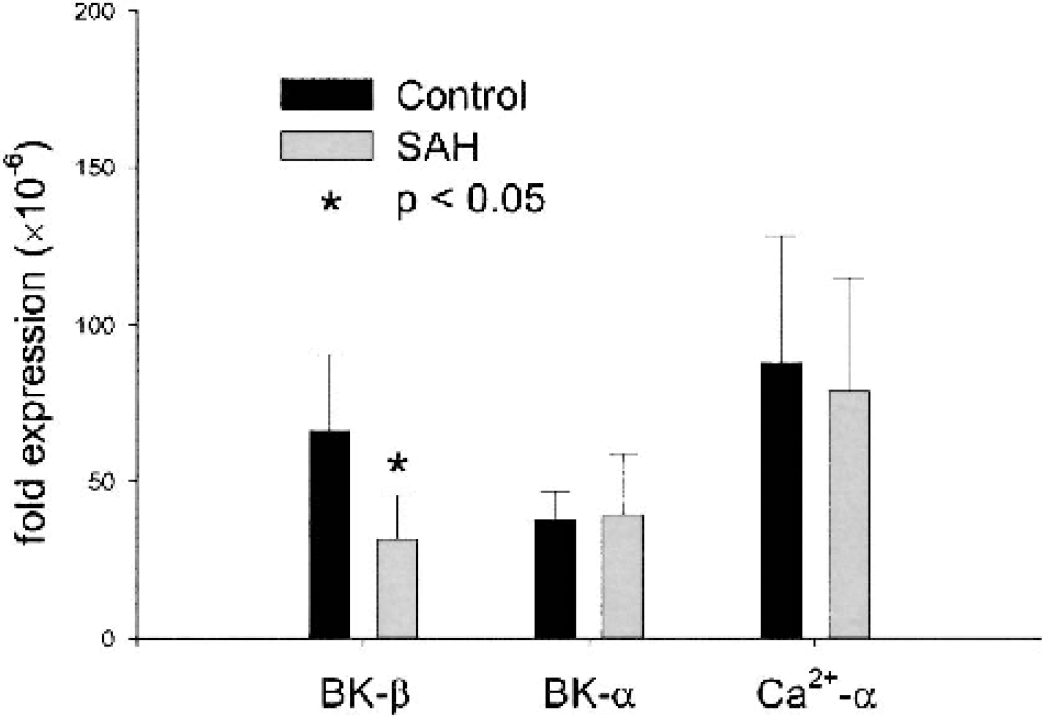
Real-time quantitative PCR of mRNA expression of BK α and β1 subunits and the α subunit of the Ca2+ L-type channel demonstrated significant reduction in expression of the regulatory β1 subunit of the BK channel after SAH (n = 6–7 per group, P < 0.05, unpaired t-test). PCR, polymerase chain reaction; SAH, subarachnoid hemorrhage.
Multiple mRNAs for Kv channels were identified in normal dog basilar artery, although Kv 1.5 and Kv 2.2 were the most abundantly expressed (Fig. 3). The mRNA expression level of other Kv channel family members was generally lower by nearly an order of magnitude. There was a significant reduction in Kv 2.2 and Kv 3.4 mRNA after SAH (65% and 38%, respectively, P < 0.001 and P < 0.05, unpaired t-tests) (Fig. 3). Expression of two Kir 2.1 sequences was examined, and it was noted that mRNA was significantly upregulated when evaluated with primers based upon the complete NCBI sequence from dog kidney tissue (234%, P < 0.001) (Fig. 4). Paradoxically, PCR using the incomplete atrial sequence showed much less amplification.
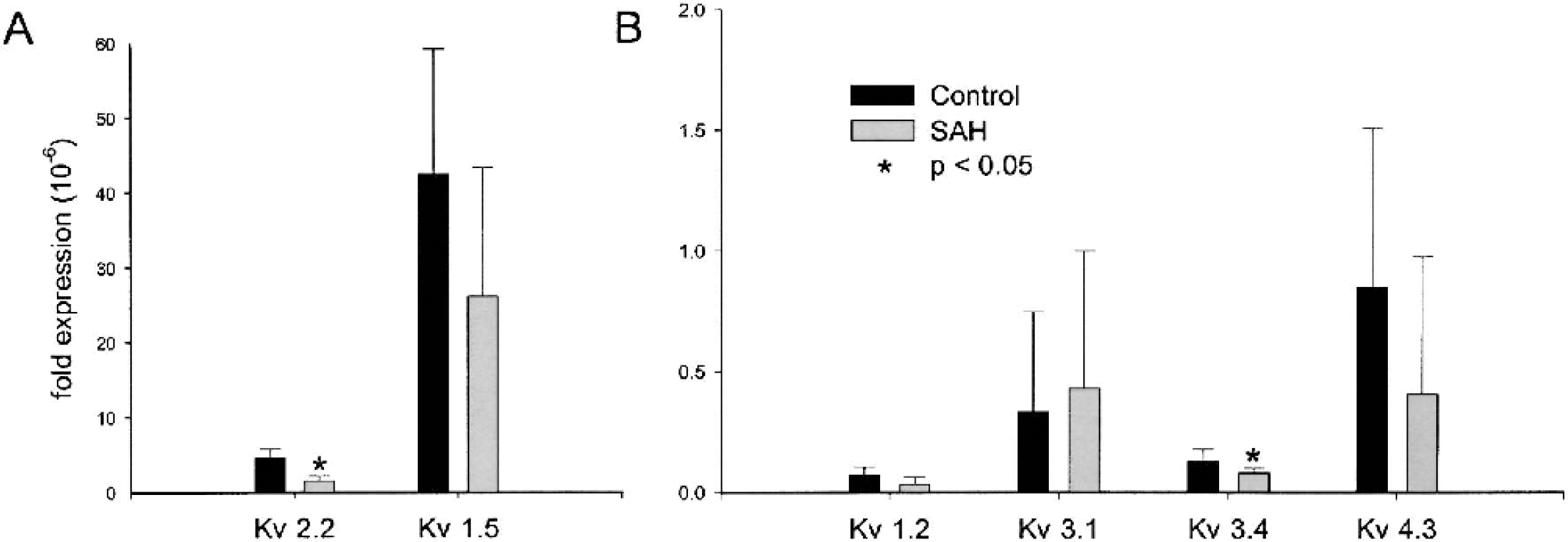
Real-time quantitative PCR of mRNA expression of Kv 1.5 and 2.2 (A) and Kv 1.2, 3.1, 3.4, and 4.3 (B). There is significant reduction in expression of Kv 2.2 and Kv 3.4 mRNA after SAH (n = 6–7 per group, P < 0.05, unpaired t-test). PCR, polymerase chain reaction; SAH, subarachnoid hemorrhage.
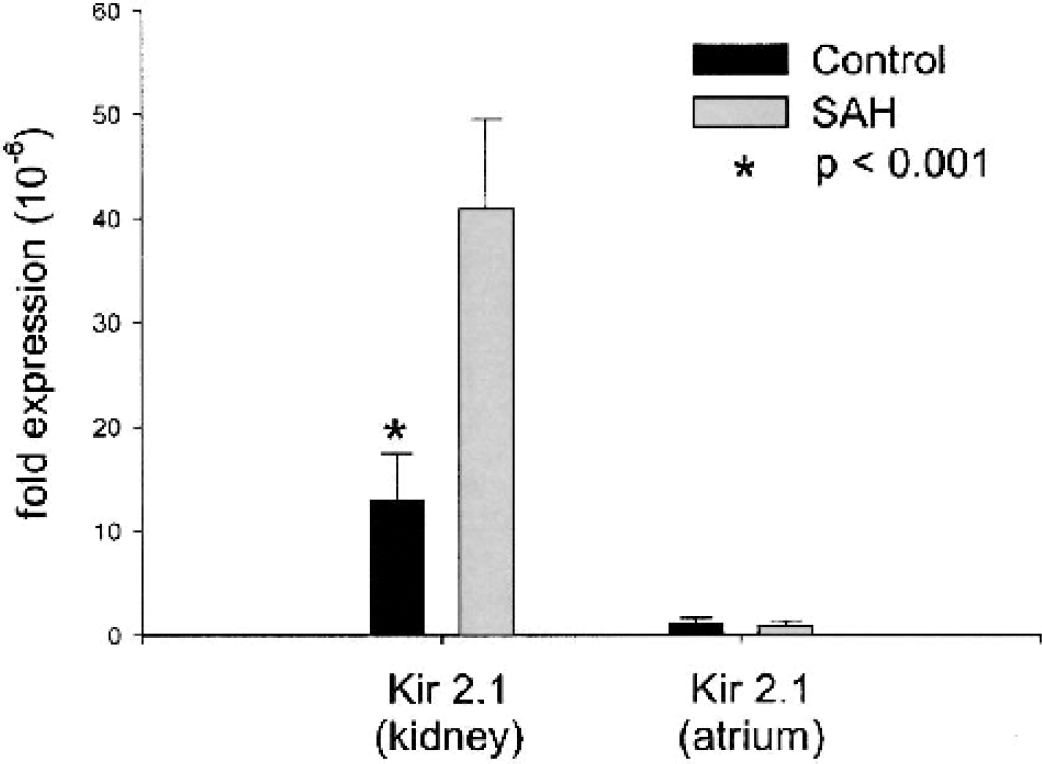
Real-time quantitative PCR of mRNA expression of Kir 2.1 (atrial and kidney sequence sources) found expression of kidney sequence with a significant upregulation of Kir after SAH (n = 6–7 per group, P < 0.001, unpaired t-test). PCR, polymerase chain reaction; SAH, subarachnoid hemorrhage.
Correlation between the degree of vasospasm and the relative change in mRNA expression of target genes after SAH was examined using scatter plots. Because the double hemorrhage canine model consistently reproduced severe (>50%) vasospasm, data from dilutional SAH animals was used to increase the range of vasospasm assessed. There was a significant correlation between the relative expression level of both BK-ß1 subunit and Kv 2.2 mRNA and the degree of vasospasm (n = 10, r2 = 0.53 and 0.49 respectively, P < 0.05) (Fig. 5); this was not observed for other ion channels examined.
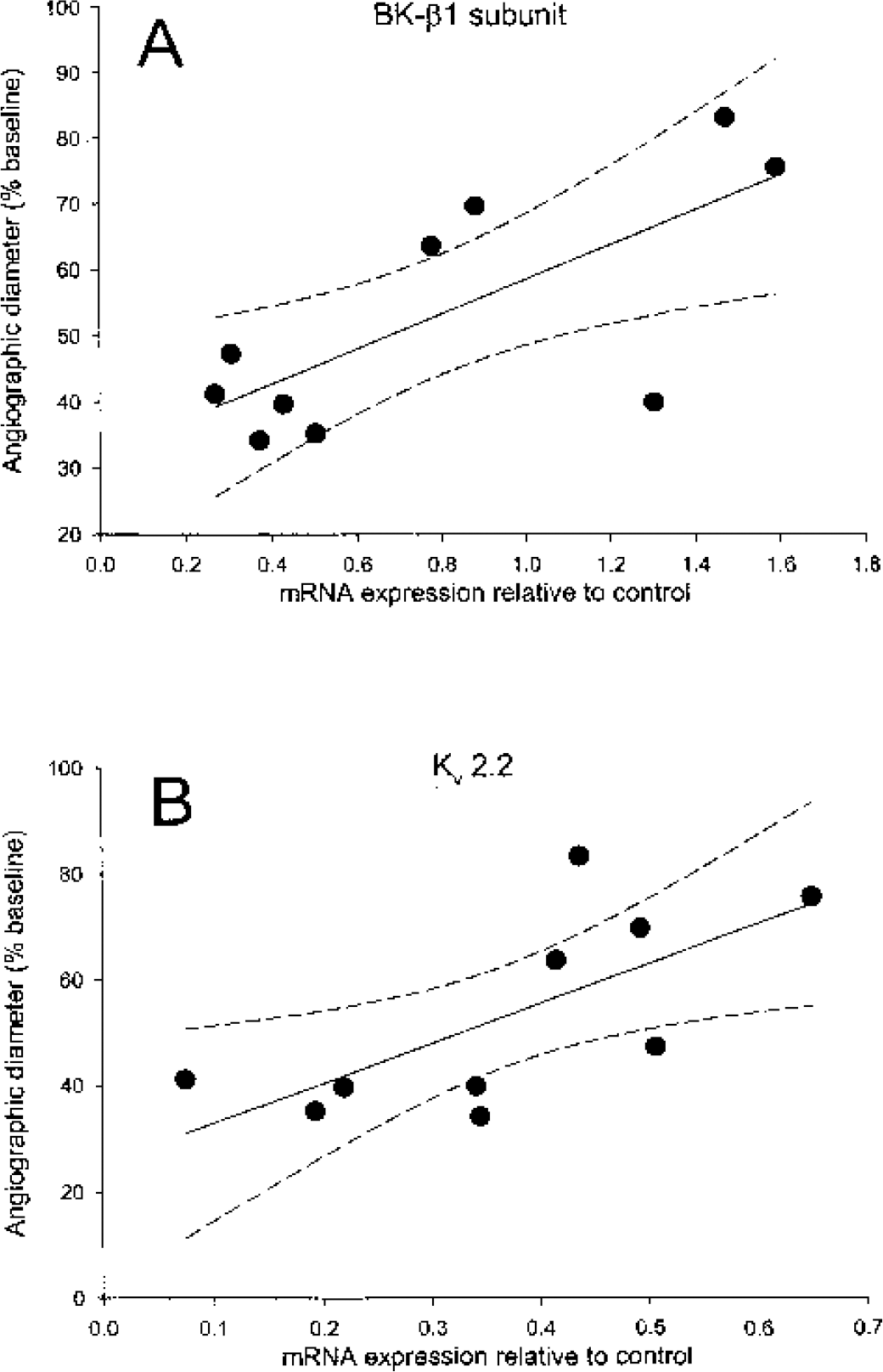
Regression analysis showing correlation between mRNA expression of BK-ß1 subunit (A) and Kv 2.2 (B) (relative to control values) and the degree of vasospasm (n = 10, r2 = 0.53 and 0.49 respectively, P < 0.05). Solid line, regression line; dashed lines, 95% confidence intervals of regression.
Target gene protein expression
Western blots were not normalized to expression of a particular protein such as β-actin because the expression of these proteins was found to vary after SAH (R.L. Macdonald, unpublished observations, September 12, 2003). Therefore, standardization was accomplished by loading identical amounts of total protein per lane. We could detect bands consistent with BK-ß1 subunit (approximately 28 kD), Kv 2.2 (approximately 98 kD), and Kir 2.1 (approximately 60 kD) (Fig. 6). BK-ß1 subunit expression was unchanged between the SAH and control group (data not shown), whereas Kv 2.2 and Kir 2.1 protein levels were altered in the same manner as mRNA for these genes. There was a significant increase in Kir 2.1 protein (350% increase, P < 0.001, unpaired t-test) and a significant decrease in Kv 2.2 protein (49% decrease, P < 0.05, unpaired t-test) in SAH compared to control basilar arteries (Fig. 6).
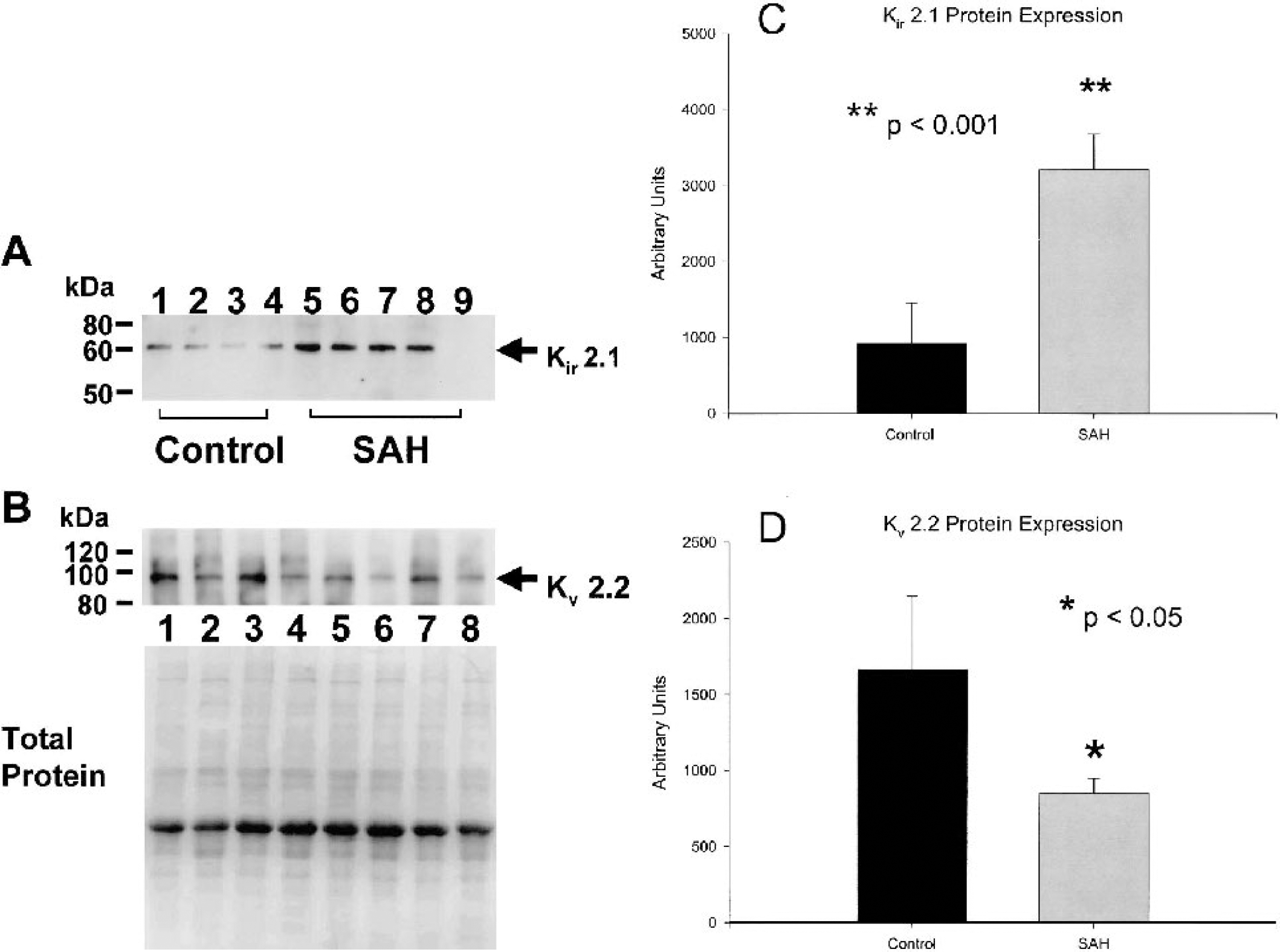
Western blot analysis of Kir 2.1 and Kv 2.2 expression in the dog basilar artery. (A) Kir 2.1 channel protein (60 kD) is greater in SAH group (lane 5 to 8) than control group (lane 1 to 4), and no signal is present when using a blocking peptide (lane 9). (B) Protein levels of Kv 2.2 (98 kD) are down-regulated in SAH (lanes 2, 4, 6, 8) as compared with control basilar artery (lanes 1, 3, 5, 7). Total protein extracts loaded in each lane was visualized by staining the transferred PVDF membrane with GelCode Blue Stain Reagent (Pierce). Each lane contains an equal amount of total protein. Quantification of bands by photometric analysis shows significant upregulation of Kir 2.1 (C) and downregulation of Kv 2.2 (D, n = 4–8 per group, unpaired t-tests). SAH, subarachnoid hemorrhage.
DISCUSSION
We have used quantitative real-time PCR and western blotting to examine changes in membrane ion channels of vasospastic smooth muscle in a canine double hemorrhage model of SAH. We report for the first time the findings of a significant decrease in mRNA and protein expression of Kv 2.2 and of the mRNA for the β1 subunit of the BK channel along with an upregulation of Kir 2.1 mRNA and protein during vasospasm after SAH. Furthermore, there was a significant correlation between the reduction in expression of Kv 2.2 and BK-β1 subunit mRNA and the degree of vasospasm. In comparison, the mRNA expression of the L-type Ca2+ channel was unchanged. These results are consistent with the hypothesis that alterations in K+ channel expression may alter membrane potential and thereby increase cerebral arterial tone, leading to vasospasm after SAH. The real-time PCR amplification method has been used to investigate changes in gene expression in neurologic diseases (Berti et al., 2002; Medhurst et al., 2000) and specifically to study changes in ion channel expression in a canine model of atrial fibrillation (Yue et al., 1999). Aihara and colleagues used a similar technique to determine changes in inflammatory gene expression after SAH in dogs (Aihara et al., 2001).
Reduced function of K+ channels might be expected to be important in vasospasm because a key determinant of vascular tone is smooth muscle membrane potential, and this is tightly regulated by K+ membrane conductance (Nelson et al., 1990; Standen and Quayle, 1998). Changes in vascular smooth muscle membrane potential of only a few millivolts are associated with large changes in vascular tone (Knot and Nelson, 1998). The resting membrane potential in pressurized arterial smooth muscle cells lies approximately 30 to 45 mV positive to the equilibrium potential for K+ (EK) in these cells, and as a result, opening or closing of K+ channels will lead to vasodilation or vasoconstriction, respectively (Standen and Quayle, 1998). The specific K+ channels that have been characterized in cerebrovascular smooth muscle cells include Kir, adenosine-triphosphate-sensitive K+ (KATP), Kv, and BK channels. The latter two play a key role in regulation of membrane potential and vascular tone (Faraci and Sobey, 1998). BK channels in particular have been shown to regulate membrane potential and vascular tone in human cerebral resistance arteries (Gokina et al., 1996;Wellman et al., 2002). They are uniquely situated to modulate vascular tone by virtue of large conductance along with sensitivity to both intracellular Ca2+ concentration ([Ca2+]i) and membrane potential. Membrane depolarization and raised [Ca2+]i activate BK currents, and thus the BK channel is a component of a negative feedback mechanism that opposes vasoconstriction because opening of BK channels results in outward, hyperpolarizing K+ currents that mediate relaxation of cerebral arteries by closing L-type Ca2+ channels and reducing [Ca2+]i (Knot and Nelson, 1998). Increases in [Ca2+]i that mediate such openings may be in the form of focal sparks within the cell that are in close physical proximity to BK channels (Wellman et al., 2002).
The smooth muscle BK channel is composed of a pore-forming α subunit and a regulatory β1 subunit (Brenner et al., 2000). The β1 subunit increases the Ca2+ and voltage sensitivity of the BK channel so that it opens and thereby opposes contraction at lower levels of [Ca2+]i and at more negative membrane potentials. The observed reduction in BK-β1 subunit in our study could make this channel less sensitive to Ca2+ and shift its voltage-dependence to more depolarized potentials following SAH (Cox and Aldrich, 2000; McManus et al., 1995; Nimigean and Magleby, 1999). This would effectively limit its ability to serve as a negative feedback to Ca2+ influx-mediated vasoconstriction, which might contribute to the prolonged vasoconstriction observed in cerebral vasospasm. In support of this hypothesis, BK-β1 knockout mice show increased cerebral vasoconstriction in response to pressure (Brenner et al., 2000). In addition, they demonstrate a shift in the coupling between Ca2+ sparks and spontaneous transient outward K+ currents towards more depolarized potentials (Pluger et al., 2000). On the other hand, no decrease in BK-β1 subunit protein was detected. Additional data are required in appropriate SAH models to determine if there is a functional correlate of the β1 subunit mRNA downregulation observed in this study.
A second key type of K+ channel, the Kv family, also exhibited altered expression after SAH. Four main gene families of delayed rectifier or Kv channels have been identified (Overturf et al., 1994). We identified members of Kv 2 (Kv 2.2) and Kv 1 (Kv 1.5) as the most abundant in dog basilar artery. Pharmacologic characteristics of cerebral arteries have been consistent with presence of Kv channels. Changes in arterial tone in response to 4-aminopyridine were suggested to support the presence of Kv 1.5, although this drug blocks other Kv forms. More direct methods for identifying specific Kv channels in cerebral arteries, such as electrophysiologic studies of isolated cells have seldom been done in detail (Knot and Nelson, 1995). Patch clamp recordings have demonstrated Kv currents in cerebral arterial myocytes from cats (Bonnet et al., 1991), rabbits (Robertson and Nelson, 1994), and dogs (R. L. Macdonald, unpublished observations, July 7, 2003), and basal cerebrovascular tone has been shown to be modulated by Kv conductance as revealed by vasoconstrictor responses to 4-aminopyridine (a Kv blocker) (Knot and Nelson, 1995; Quan and Sobey, 2000). Kv channels also may be targets of second messenger systems that can affect arterial diameter (Beech, 1997; Faraci and Sobey, 1998). Kv 1.5 was identified as a functional channel in rabbit pulmonary vein (Clement-Chomienne et al., 1999), and Kv 2.2 was identified in dog colonic smooth and vascular smooth muscle (Schmalz et al., 1998). These were the two most abundant mRNAs in dog basilar artery. Lower levels of Kv 1.2, 3.1, 3.4, and 4.3 were noted, although some mRNAs, such as Kv 4.3, were only slightly less abundant than Kv 2.2. It is interesting to note that there was a significant reduction in Kv 3.4 mRNA and a trend towards reduction in Kv 4.3. Each of these mRNAs was identified in the heart and may have a role in cardiac electrical function (Yue et al., 1999, 2000). It is impossible to determine, based upon the mRNA data presented here, the functional role of and consequences of alterations in any of the Kv channel isoforms identified here. In any case, reduced Kv mRNA and protein expression observed in vasospastic arteries in this study could result in reduced baseline K+ conductance and the ability to repolarize after vasoconstrictive stimuli, both of which also would favor increasedcerebrovascular tone. Kv channels play a role in the maintenance of resting membrane potential and the myogenic response to increased intraluminal pressure in cerebral arteries (Knot and Nelson, 1995).
Kir channels are believed to mediate cerebral vasodilation that follows activity-mediated increases in extracellular K+ (Edwards et al., 1988). Mild elevations of extracellular K+ (5–15 mM) hyperpolarize and thus dilate pressurized rat cerebral arteries in vitro (Johnson et al., 1998; Knot et al., 1996). Although there are no prior studies of dog basilar artery, Kir 2.1 was expressed in rat basilar artery and vasodilatory responses to external K+ were observed in vivo through a cranial window preparation (Chrissobolis et al., 2000). Knock out of Kir 2.1 in mice abolished external K+-mediated vasodilation in pressurized cerebral arteries and the inwardly-rectifying Ba2+-sensitive current was absent in smooth muscle cells from Kir 2.1 knockout cerebral arteries (Zaritsky et al., 2000). The increase in Kir 2.1 mRNA and protein observed following SAH in this study is interesting in that it may represent a potential compensatory mechanism directed against the sustained smooth muscle cell contraction and the decreased Kv 2.2 and BK-β1 expression observed in cerebral vasospasm. Interestingly, changes in Kir currents have been reported in other cerebrovascular diseases. Bastide and colleagues noted that middle cerebral artery smooth muscle cells exhibited reduced Kir currents after being subjected to ischemia and reperfusion and that this reduction correlated with increasing infarct volume (Bastide et al., 1999). The mRNA detected corresponded to a kidney sequence, whereas we could not detect a partial sequence of atrial Kir 2.1. The latter sequence may represent a different Kir channel that is expressed at lower levels in dog basilar artery. In support of this, the antibody used for detection of Kir 2.1 protein recognized an amino acid sequence that varied by only 1 amino acid from that predicted from the kidney Kir 2.1 sequence.
Our molecular data are consistent with studies of vasospastic smooth muscle cells in cerebral arteries of dogs (Harder et al., 1987), rabbits (Zuccarello et al., 1996), and cats (Waters and Harder, 1985) that showed resting membrane potentials 10 to 20 mV depolarized with respect to control animals. Indirect evidence exists for decreased K+ conductance in vasospastic vascular smooth muscle (Harder et al., 1987; Quan and Sobey, 2000). Vasoconstrictor responses to the Kv antagonist 4-aminopyridine were reduced in rat basilar artery after SAH (Quan and Sobey, 2000) and K+ channel agonists such as cromakalim (Kwan et al., 2000; Sugai et al., 1999; Zuccarello et al., 1996) and nicorandil (Matsui et al., 1994) partially reduce experimental vasospasm, suggesting a K+ channel defect after SAH. The lack of specificity of 4-aminopyridine, cromakalim and nicorandil preclude any definition at this point as to which K+ channels may be involved (Zhang and Cook, 1994). Finally, the BK agonist NS1619 prevented contractions of rabbit basilar artery in response to the putative spasmogen, erythrocyte hemolysate (Kim et al., 1999).
The fourth K channel, the KATP channel, was not examined in this study. Preliminary electrophysiologic data examining smooth muscle cells from dog basilar artery do not show KATP currents (R.L. Macdonald, unpublished observations, July 7, 2003). Pharmacologic studies suggest KATP channels are present in some cerebral arteries although they may be more abundant in smaller resistance arteries than in the large arteries such as the basilar artery (Rosenblum, 2003). This raises the question as to whether the changes observed in basilar arteries in this study are in smooth muscle cells, the endothelium and/or adventitial fibroblasts. It is unlikely that endothelial cells possess BK channels, but they may have Kv and Kir channels of various types (Nilius and Droogmans, 2001). Further work would be needed to delineate the exact location of the changes observed herein.
The dog model used herein has substantial advantages over other models of SAH and vasospasm. Significant, delayed cerebral vasospasm develops which is similar to that developing in humans and which does not occur in mouse, rat and rabbit models of SAH (Macdonald and Weir, 2001; Megyesi et al., 2000). The latter models do not produce severe, delayed vasospasm and are further limited by the difficulty in analyzing small amounts of tissue, which often requires pooling of specimens. Our results nevertheless have some limitations. We restricted our analysis to canine ion channel sequences available in the NCBI database. The question of whether the observed changes are a cause or consequence of SAH cannot be determined from this study. It might be asked how the increase in Kir 2.1 could be important, which was in fact the largest change noted herein, when the artery is actually constricted at the time it is increased. While the correlation between reduced mRNA levels and degree of cerebral vasospasm does not necessarily prove causation, this can be partially addressed by studying different time-points during the development and subsequent abatement of vasospasm. Finally, investigations examining the functional significance of alterations in K+ channels reported here using isometric tension recordings, perfused artery preparations in vitro and patch-clamp recordings of isolated vasospastic arterial myocytes are essential. The data presented here nevertheless provide the first molecular demonstration of a reduction in and correlation of changes in K+ channels with vasospasm. They provide the basis for the future investigation of ion channel dysfunction in cerebral vasospasm.
Footnotes
Acknowledgements
The authors thank Dr. J.S. Trimmer (State University of New York, Stonybrook NY, U.S.A.) for providing the Kv 2.2 antibody.
