Abstract
Hypothermic protection was compared in Long-Evans and spontaneously hypertensive rat (SHR) strains using transient focal ischemia, and in Wistar and SHR strains using permanent focal ischemia. Focal ischemia was produced by distal surgical occlusion of the middle cerebral artery and tandem occlusion of the ipsilateral common carotid artery (MCA/CCAO). Moderate hypothermia of 2 hours' duration was produced by systemic cooling to 32 °C, with further cooling of the brain achieved by reducing to 30°C the temperature of the saline drip superfusing the exposed occlusion site. Infarct volume was determined from serial hematoxylin and eosin-stained frozen sections obtained routinely at 24 hours, or in some cases after 3 days' survival. In the SHR, moderate hypothermia was only effective when initiated before recirculation after a 90-minute occlusion period. In contrast, the same intervention was strikingly effective in the Long-Evans rat even when initiated after as long as 30-minute reperfusion after a 3-hour occlusion. This magnitude and duration of cooling was not protective in permanent MCA/CCAO in the SHR, but such transient hypothermia did effectively reduce infarct volume after permanent occlusions in Wistar rats. These results show striking differences in the temporal window for hypothermic protection among rat focal ischemia models. As expected, “reperfusion injury” in the Long-Evans strain is particularly responsive to delayed cooling. The finding that the SHR can be protected by hypothermia initiated immediately before recirculation suggests a rapidly evolving component of injury occurs subsequent to reperfusion in this model as well. Hypothermic protection after permanent occlusion in Wistar rats identifies a transient, temperature-sensitive phase of infarct evolution that is not evident in the unreperfused SHR. These observations confirm that distinct mechanisms can underlie the temporal progression of injury in rat stroke models, and emphasize the critical importance of considering model and strain differences in extrapolating results of hypothermic protection studies in animals to the design of interventions in clinical stroke.
Hypothermia is arguably the most effective intervention for lesion reduction in animal models of cerebral ischemia (Colbourne et al., 1997). Although focal ischemia does not exhibit the exquisite sensitivity to temperature documented for global ischemia (Buchan and Pulsinelli, 1990; Dietrich et al., 1990; Nurse and Corbett, 1996), the impact of temperature is well recognized as a variable affecting outcome after stroke, both in animals and in humans. The potential negative impact of fever in stroke patients has been increasingly recognized (Castillo et al., 1998; Fukuda et al., 1999; Ginsberg and Busto, 1998; Grau et al., 1999; Kammersgaard et al., 2002; Schwab et al., 1997). Results of clinical trials of hypothermia in stroke are promising, particularly with regard to attenuating delayed edema after large infarcts (Schwab et al., 1998), although complications arising from prolonged systemic hypothermia limit its utility (Georgiadis et al., 2002). Tissue-sparing effects of hypothermia have yet to be documented in clinical stroke. Although early rodent studies clearly showed that hypothermia reduced infarct volume when initiated before or relatively soon after occlusion (Chen et al., 1992; Karibe et al., 1994b; Morikawa et al., 1992; Moyer et al., 1992; Ridenour et al., 1992), defining the optimal magnitude, duration, and therapeutic window for protective cooling has not been straightforward, even in animal models. Apart from variations in sites and methods of vascular obstruction, systematic differences among rat strains could also contribute to model heterogeneity, and an understanding of the mechanisms underlying such differences is critical for valid extrapolation to clinical stroke.
Most studies in rat middle cerebral artery occlusion (MCAO) models indicate that little reduction in infarct volume can be expected in the absence of reperfusion after a range of interventions, including hypothermia (Morikawa et al., 1992; Ridenour et al., 1992). However, there is evident variability among strains, since transient early cooling has been reported to reduce infarct volume even after permanent MCAO in Wistar rats (Baker et al., 1992; Kader et al., 1992), whereas work in SHR has either reported no effect (Ridenour et al., 1992) or found only modest protection with very deep cooling (Onesti et al., 1991). For transient ischemia, effective protection by intraischemic hypothermia has been a consistent finding (Goto et al., 1993; Maier et al., 1998; Xue et al., 1992). This is readily understood in view of the typically well-defined dependence of infarct volume on occlusion duration (Kaplan et al., 1991), and the temperature dependence of diverse pathophysiologic processes during ischemia. The observation that 1 hour of cooling to 33°C initiated at the time of occlusion is as protective as maintaining hypothermia for the duration of 2 hours of MCAO (Maier et al., 1998) is consistent with the interpretation that even the shorter intervention reduced effective insult duration to below the temporal threshold for infarction. There is some evidence that the magnitude of cooling may become increasingly important as the duration of the insult is prolonged (Goto et al., 1993; Maier et al., 1998).
Postischemic cooling paradigms have varied considerably across studies, even apart from strain differences in the models used, but support the generalization that the window for initiating hypothermic protection after focal ischemia is very short. In a relatively comprehensive study in Wistar rats, 32°C hypothermia delayed until the second half of a 3-hour occlusion reduced infarct volume at 24 hours by approximately 50%, and still smaller infarcts were evident when cooling was further extended through the first 90 minutes of recirculation (Xue et al., 1992). Postischemic cooling to 32°C for 3 hours was protective in a Wistar filament occlusion model, whereas 1 hour of hypothermia was not (Zhang et al., 1993b). Observations in a Sprague-Dawley model involving 3-hour MCAO coupled with bilateral carotid occlusion likewise suggested that dramatic protection could be achieved when mild hypothermia was initiated at the time of recirculation and maintained for 21 hours, but that protection was markedly attenuated when cooling was delayed until 30-minute recirculation, and a transient 1-hour cooling period was ineffective (Yanamoto et al., 1996). Moderate protection was reported in the same model with a 3-hour cooling period initiated within 30 minutes of recirculation (Markarian et al., 1996), equivalent to that observed during intraischemic cooling, and a more recent study in this strain also indicated that cooling to 32°C initiated immediately after 2-hour occlusion was as protective as intraischemic hypothermia (Huh et al., 2000). These studies place particular emphasis on the initial recirculation period as a target for protective cooling, although results with an intraluminal filament occlusion model have suggested a somewhat longer temporal window of 1 hour for postischemic intervention (Zhang et al., 1993a). In addition, very prolonged cooling procedures offered lasting protection when initiated as long as 30 minutes or even 1 hour after recirculation in Wistar and spontaneously hypertensive rat (SHR) models, respectively (Colbourne et al., 2000; Corbett et al., 2000). Importantly, hypothermic protection has not yet been examined after transient ischemia in Long-Evans rats, a model that is suggested to involve a distinct component of “reperfusion injury” (Aronowski et al., 1997).
The present study compared the effectiveness of a standardized cooling protocol in established models of transient and permanent focal ischemia in SHR, transient focal ischemia in Long-Evans rats, and permanent focal ischemia in SHR and Wistar strains. Brain temperature control is particularly critical during surgical exposure of the brain surface, and careful attention has been given to avoiding incidental cooling in the course of model development (Brint et al., 1988). However, surface cooling permits supplemental reduction of brain temperature (Barone et al., 1997; Huh et al., 2000), and such an approach was used in the present studies. Initial results replicated the finding of increased infarct volume with delayed recirculation in the Long-Evans strain, and under such conditions identified a relatively prolonged window for hypothermic protection in this model of putative reperfusion injury. Conversely, such intervention was effective only when initiated before reperfusion in the SHR. The Wistar strain showed reduced infarct volumes when transient cooling occurred early during permanent occlusions, whereas SHR did not, which is speculatively interpreted as evidence for a process resembling reperfusion injury in the penumbra of the evolving lesions. Part of this work has been presented in preliminary form (Nowak and Pulsinelli, 1999).
MATERIALS AND METHODS
Ischemia models
Male rats (250–300 g) of SHR (n = 163), Long-Evans (n = 81) and Sprague-Dawley (n = 29) strains were obtained from Harlan Sprague Dawley, Inc. (Indianapolis, IN, U.S.A.) and Wistar rats (n = 130) were obtained from Hilltop Lab Animals (Scottdale, PA, U.S.A.). Focal brain ischemia was produced by tandem occlusion of the right middle cerebral artery (MCA) and ipsilateral common carotid artery essentially as previously described (Brint et al., 1988; Kaplan et al., 1991), according to a protocol approved by the Animal Care and Use Committee, University of Tennessee. After an overnight fast animals were anesthetized and ventilated with 0.5–1% halothane in 70% N2, 30% O2, and fitted with a tail artery cannula to monitor blood pressure and for periodic sampling to measure blood gases, pH, hematocrit, and plasma glucose level. Temperature was monitored with a rectal probe and maintained at 37°C via a feedback-controlled heating pad. The carotid artery was exposed and cauterized in the case of permanent occlusions, or fitted with a Teflon/silastic occluding device to produce temporary flow disruptions. The MCA was exposed by dissection of the temporalis and masseter muscles and a 1-mm burr hole was drilled through the temporal-squamous bones to expose the MCA at the level of the rhinal fissure. Temperature of the brain surface was maintained at 37°C during drilling and all subsequent steps with a warmed saline drip, the temperature of which was appropriately adjusted during cooling procedures (see below). The MCA was snared with a micromanipulatorcontrolled wire hook that was then raised approximately 1 mm to produce transient occlusions, whereas permanent ischemia was produced by cauterization of the artery. After the procedure, wounds were closed with surgical suture and swabbed with povidone/iodine and lidocaine ointment. Anesthesia was discontinued, animals were weaned from the respirator and allowed to recover consciousness and thermal equilibrium, and were then returned to their cages.
Hypothermia
Cooling was initiated at various points during ischemia or reperfusion as indicated for individual experiments. To lower body temperature the control set point was adjusted to 32°C, after which animals were wetted and packed in ice for approximately 20 minutes. In routine experiments the temperature set point of the saline superfusate was simultaneously adjusted to 30°C. Rewarming was initiated by resetting the control point to 37°C and application of a heating lamp, at which point brain surface temperature control was terminated and the surgical site closed. The time course of temperature change is shown for a representative group of animals in Fig. 1. The 5°C transition in rectal temperature required approximately 30 minutes in both cooling and rewarming phases, and the characteristics of the superfusate temperature control and delivery system resulted in a comparable interval for the necessary 10°C decrease. This resulted in a 90-minute period at stable target hypothermic temperatures. As illustrated, a saline temperature of approximately 4°C at the outlet of the saline drip maintained an equilibrium temperature of 37°C in the superfusate pool at the exposed brain surface, whereas a surface temperature of 29.3 ± 0.3°C was maintained during the cooling period. Virtually identical cooling and rewarming profiles were obtained in the several independent experiments that comprise the strain comparisons of this study. Some preliminary studies of transient MCAO in the SHR included groups with a final superfusate temperature of 32°C rather than 30°C as indicated in the text.
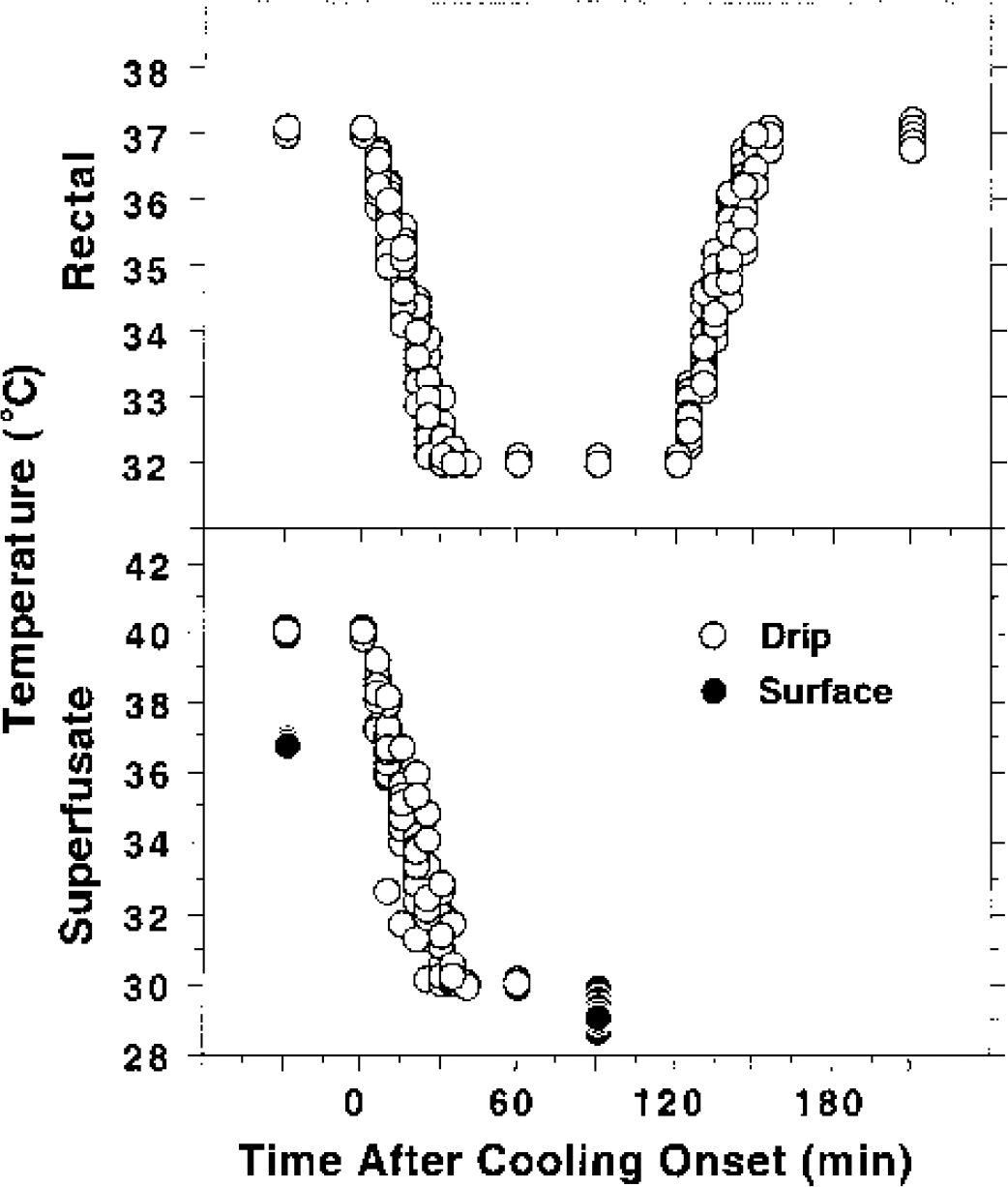
Representative time course of rectal, superfusate, and brain-surface temperature change during cooling and rewarming. Wistar rats (n = 17) were surgically prepared and subjected to permanent focal ischemia and a subsequent interval of hypothermia. Temperatures were recorded from rectal and saline drip probes at the indicated intervals (every 5 minutes during active cooling and rewarming) and plotted for each animal in the study (open circles). Corresponding brain surface temperature during intervals of stable normothermia and hypothermia were also measured and plotted (filled circles).
Infarct measurements
Animals were anesthetized with halothane and decapitated, and brains were rapidly removed and frozen in hexane at −40°C. Frozen sections (20 μm) were collected at 1-mm intervals throughout the extent of the MCA territory, thaw mounted on slides, and stained with hematoxylin-eosin. Lesion areas were determined in digitized video images (NIH Image) and summed across all sections to determine lesion volume in cubic millimeters.
Statistical analysis
All group values are stated as mean ± SD. Comparisons involving two groups used an unpaired t-test, whereas analysis of variance and Scheffé F test were used for evaluation of multiple comparisons, both implemented in StatView 5.0 (SAS Institute, Inc., Cary, NC, U.S.A.). A value of P < 0.05 was considered statistically significant.
RESULTS
Strain differences in infarct thresholds and evidence for reperfusion injury
The effect of occlusion duration on infarct volume in SHR, Long-Evans, Wistar, and Sprague-Dawley rats is shown in Fig. 2. In the hands of Investigator A, the SHR yielded progressively increasing infarct volumes of 103 ± 60, 147 ± 31, and 180 ± 37 mm3 after 90-minute, 2-hour, and permanent occlusions, respectively, when evaluated at 24 hours. Investigator B obtained a somewhat lower threshold for maximal infarction with equivalent lesion size after 90-minute and permanent occlusions (153 ± 43 vs. 158 ± 32 mm3). Occlusion of the middle cerebral artery and tandem occlusion of the ipsilateral common carotid artery (MCA/CCAO) in Long-Evans rats produced smaller infarcts and required longer durations of occlusion to achieve maximum infarct volume than did the SHR. Transient 3-hour occlusions in the Long-Evans rat resulted in significantly larger and more reproducible infarcts than found after permanent MCA/CCAO (83 ± 25 vs. 21 ± 21 mm3). Only small infarcts were obtained in Sprague-Dawley rats with this occlusion method regardless of occlusion duration, although occasional animals exhibited larger infarcts after intervals of transient focal ischemia. Wistar rats present an intermediate response, in that maximum infarct volumes approximated those seen in the SHR, but permanent occlusions produced more variable and on average slightly smaller lesions than seen after long temporary occlusions. As for the SHR, the precise relationship between occlusion duration and infarct volume varied somewhat between investigators, and retrospective analysis of physiologic variables revealed that Investigator A maintained a systematically lower brain surface temperature (37.2 ± 0.3°C vs. 37.4 ± 0.2°C), as well as a more ventilated respiratory state (Pa
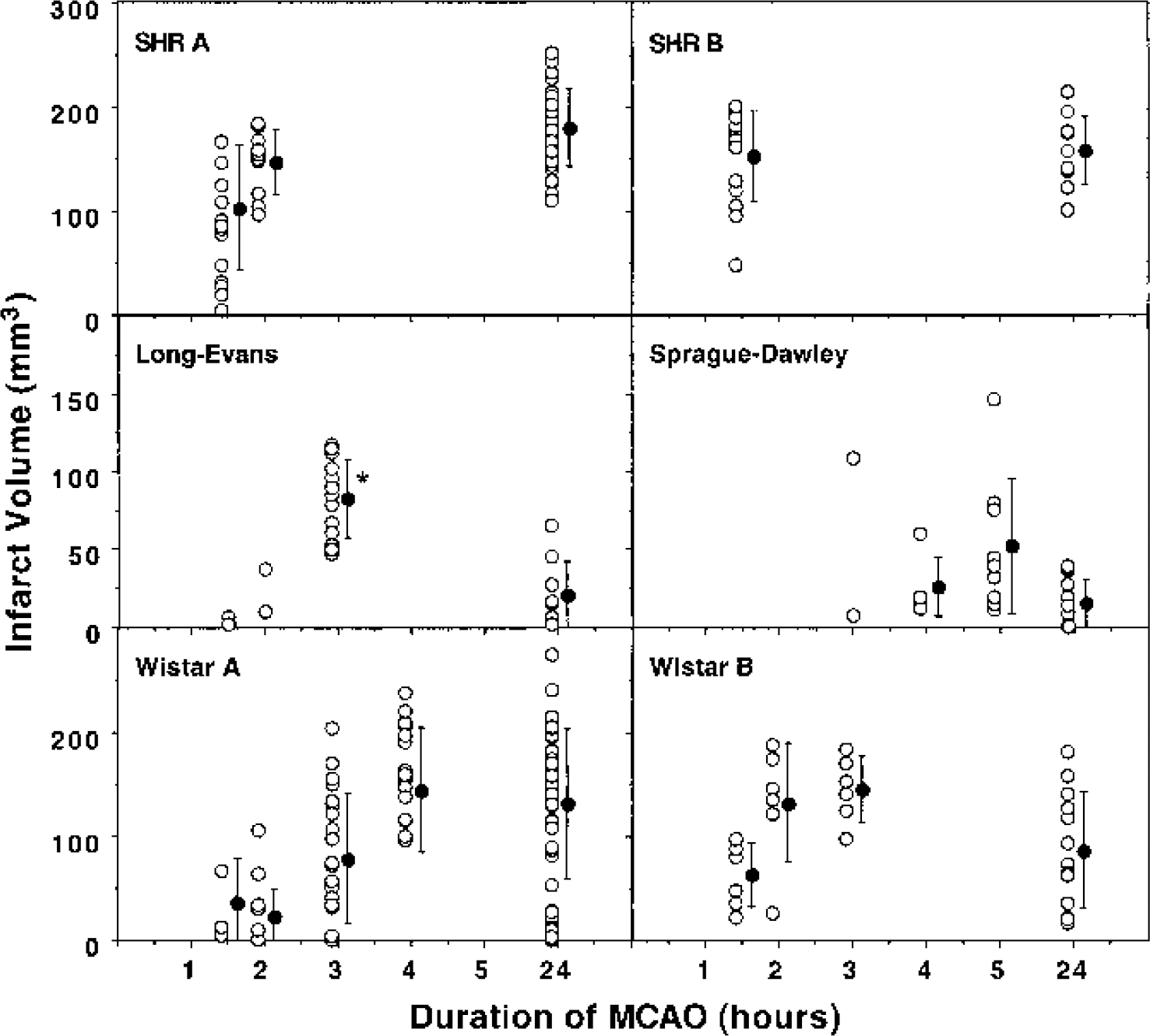
Strain comparisons of the relationship between occlusion duration and infarct volume. Rats were subjected to the indicated durations of distal MCA/CCAO and infarct volumes were determined 24 hours after initiation of ischemia. Spontaneously hypertensive rats exhibited consistent lesion size after occlusions longer than 1.5 to 2 hours, with a slight difference in threshold between investigators A and B. The Long-Evans strain showed small lesions after permanent occlusions but significantly larger infarcts after 3-hour temporary occlusions (asterisk indicates a significant difference from permanent occlusions, unpaired t-test), whereas Sprague-Dawley rats showed inconsistent damage under all conditions tested (note difference in scale for infarct volumes in these strains). Wistar rats presented an intermediate pattern, showing reproducible infarcts after temporary occlusions and a tendency toward smaller lesions after permanent MCAO in the hands of two separate investigators. Open symbols indicate values for individual animals; filled symbols indicate group means and standard deviations.
Hypothermic protection during transient ischemia in the spontaneously hypertensive rat
Preliminary work had demonstrated that a 3-hour cooling to 32°C initiated within 30 minutes of a 90-minute ischemic insult in the SHR conferred significant protection, whereas no reduction in infarct volume was achieved when the same hypothermic intervention was delayed until immediately after recirculation (Ren et al., 1998). Initial experiments therefore examined in more detail the window for hypothermic protection in the model of 90-minute transient MCAO in the SHR, with cooling initiated at varied intervals during ischemia and extended through 2-hour recirculation. As shown in Fig. 3, cooling to 32°C with superfusate temperature at the same set point provided nearly complete protection if initiated at 30 minutes of occlusion, but was no longer effective if delayed until 60 minutes. Lowering superfusate temperature to 30°C during the cooling period markedly extended this window, resulting in significant protection even if initiated just 5 minutes before recirculation, and this effect was maintained at a survival interval of 3 days. Cooling delayed until after recirculation was no longer protective (data not shown).
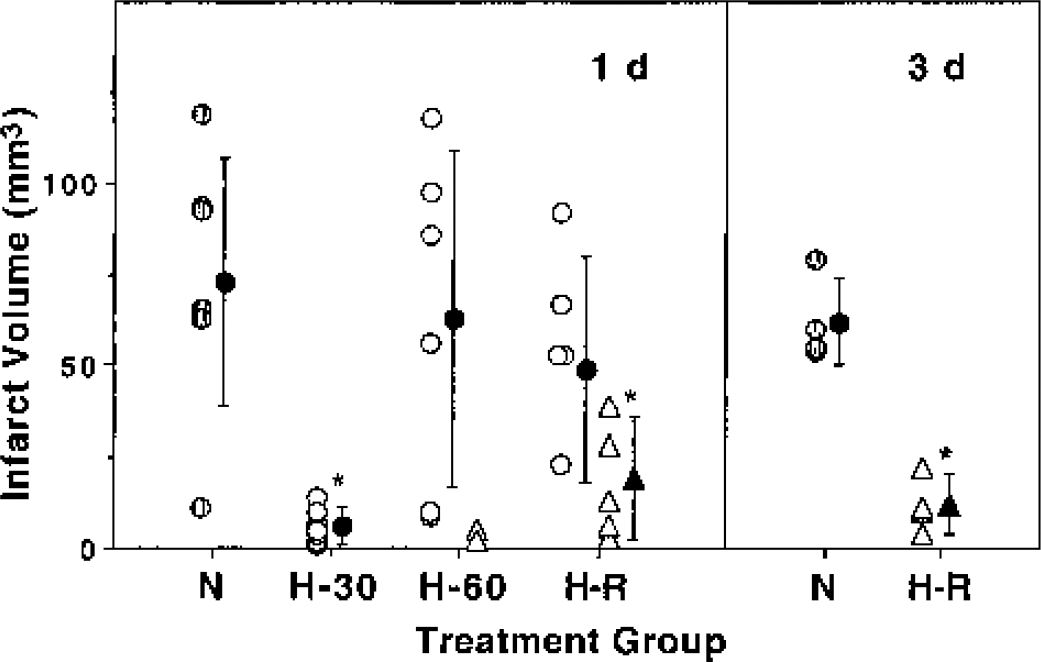
Temporal threshold for hypothermic protection in SHR. Rats were subjected to 90-minute focal ischemia and either maintained normothermic (N, shaded circles) or subjected to systemic 32°C hypothermia initiated after 30-minute (H-30), 60-minute (H-60), or 85 min occlusion just before release (H-R), in all cases continuing through 2 hours of recirculation. The saline drip controlling brain surface temperature was adjusted to either 32°C (open circles) or further lowered to 30°C (open triangles). Infarct volumes were evaluated at 24 hours (n = 6–9 animals per group), with the indicated groups (n = 4 each) examined at 3 days. Two animals with surface cooling to 30°C at 60 minutes and exhibiting no infarct are shown for illustration, but were excluded from the statistical analysis. Infarct volumes were reduced only when 32°C cooling was initiated early in the ischemic period (H-30), but supplemental brain cooling extended this window until the time of recirculation (H-R), and this protection remained evident at 3 days (asterisk indicates a significant difference from corresponding normothermic group, Scheffé F test for 24-hour groups, unpaired t-test for 3-day groups). Filled symbols indicate group means and standard deviations.
Physiologic variables measured in this SHR study are provided in Table 1. Rectal temperatures did not differ significantly among groups except during the interval of controlled hypothermia, and the magnitude of this cooling was identical in all groups. None of the measured parameters differed among normothermic and hypothermic groups at any sampling time, when measured at the analyzer temperature of 37°C. This indicates maintenance of acid-base status appropriate to the temperature dependence of pH neutrality (Ream et al., 1982). Systematic decreases in Pa
Physiologic variables during hypothermic protection in spontaneously hypertensive rats
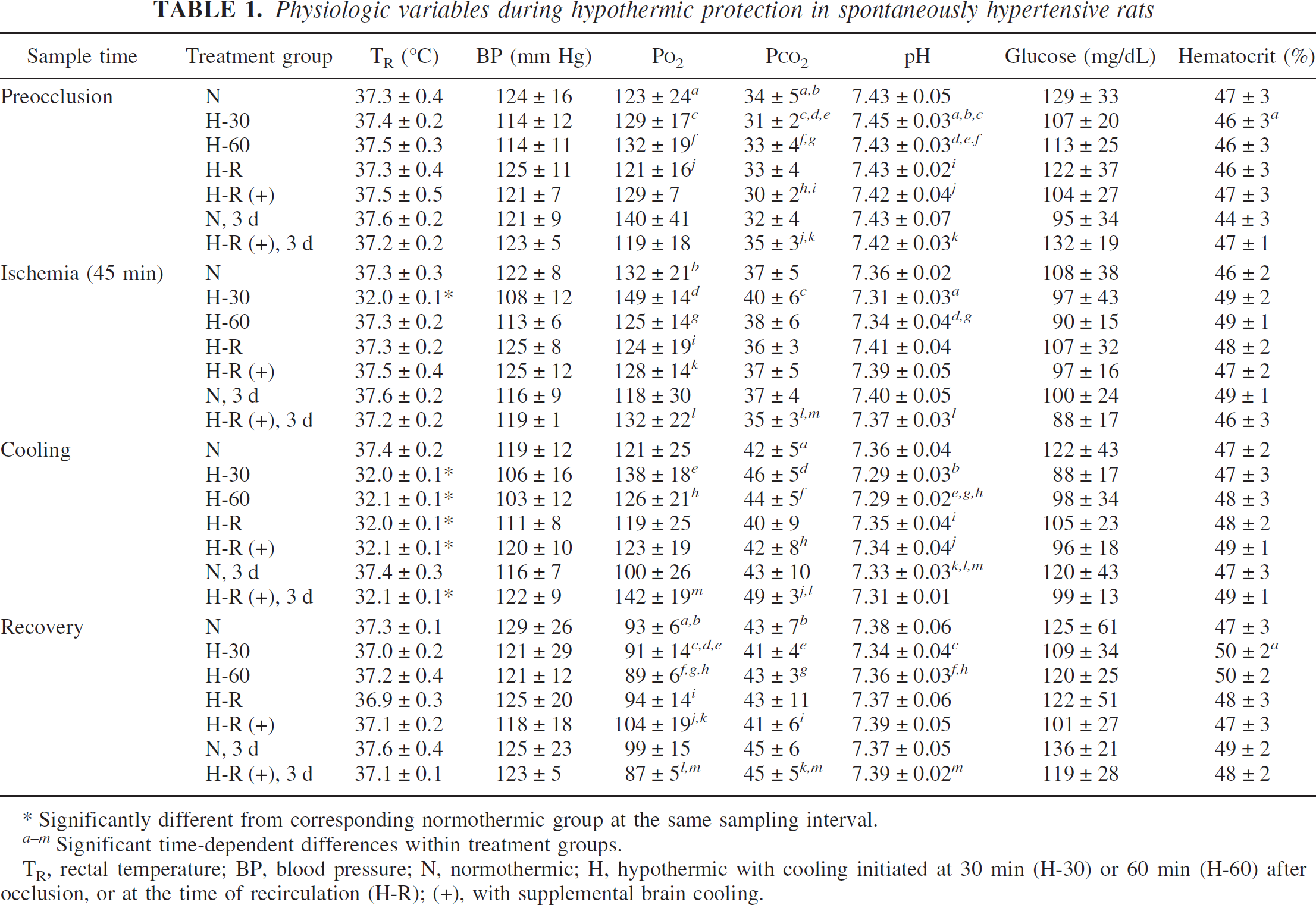
Significantly different from corresponding normothermic group at the same sampling interval.
Significant time-dependent differences within treatment groups.
TR, rectal temperature; BP, blood pressure; N, normothermic; H, hypothermic with cooling initiated at 30 min (H-30) or 60 min (H-60) after occlusion, or at the time of recirculation (H-R); (+), with supplemental brain cooling.
Hypothermic protection in the Long-Evans model
A longer window for hypothermic protection could be demonstrated in the Long-Evans model. As shown in Fig. 4, 2 hours of systemic hypothermia combined with supplemental brain cooling significantly reduced infarct volume in this model when initiated as long as 30 minutes after release of a 3-hour occlusion, and several animals showed smaller lesions even when cooling onset was delayed until 60-minute recirculation. Physiologic variables are provided in Table 2. Rectal temperature was identically lowered in all hypothermic groups during cooling, and temperature did not otherwise vary significantly among treatment groups. The slight rise in hematocrit was less prominent in the normothermic group, this difference reaching statistical significance when compared with one of the hypothermic groups at the final time point evaluated.
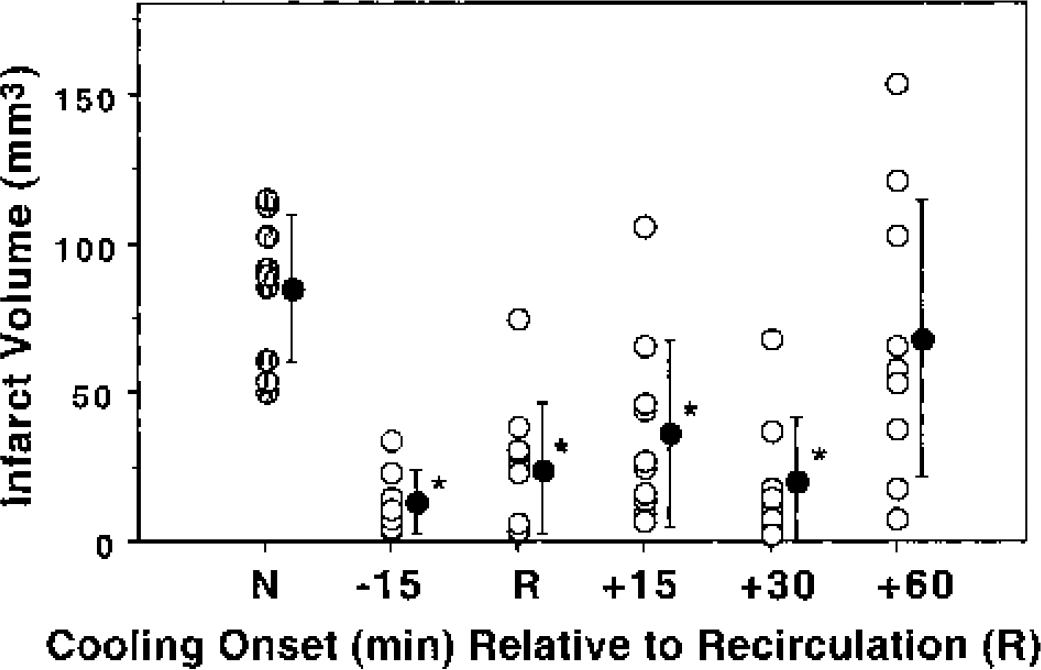
Temporal threshold for hypothermic protection in Long-Evans rats. Animals were subjected to 3-hour transient MCAO under conditions of maintained normothermia (N) or with the standard 2-hour cooling protocol (32°C systemic, 30°C brain surface) initiated at the indicated time points before or after recirculation (n = 8–10 animals per group). Hypothermia was equally effective whether initiated 15 minutes before or as long as 30 minutes after recirculation (asterisk indicates a significant difference from normothermic animals, Scheffé F test). Filled symbols indicate group means and standard deviations.
Physiologic variables during hypothermic protection in Long-Evans rats
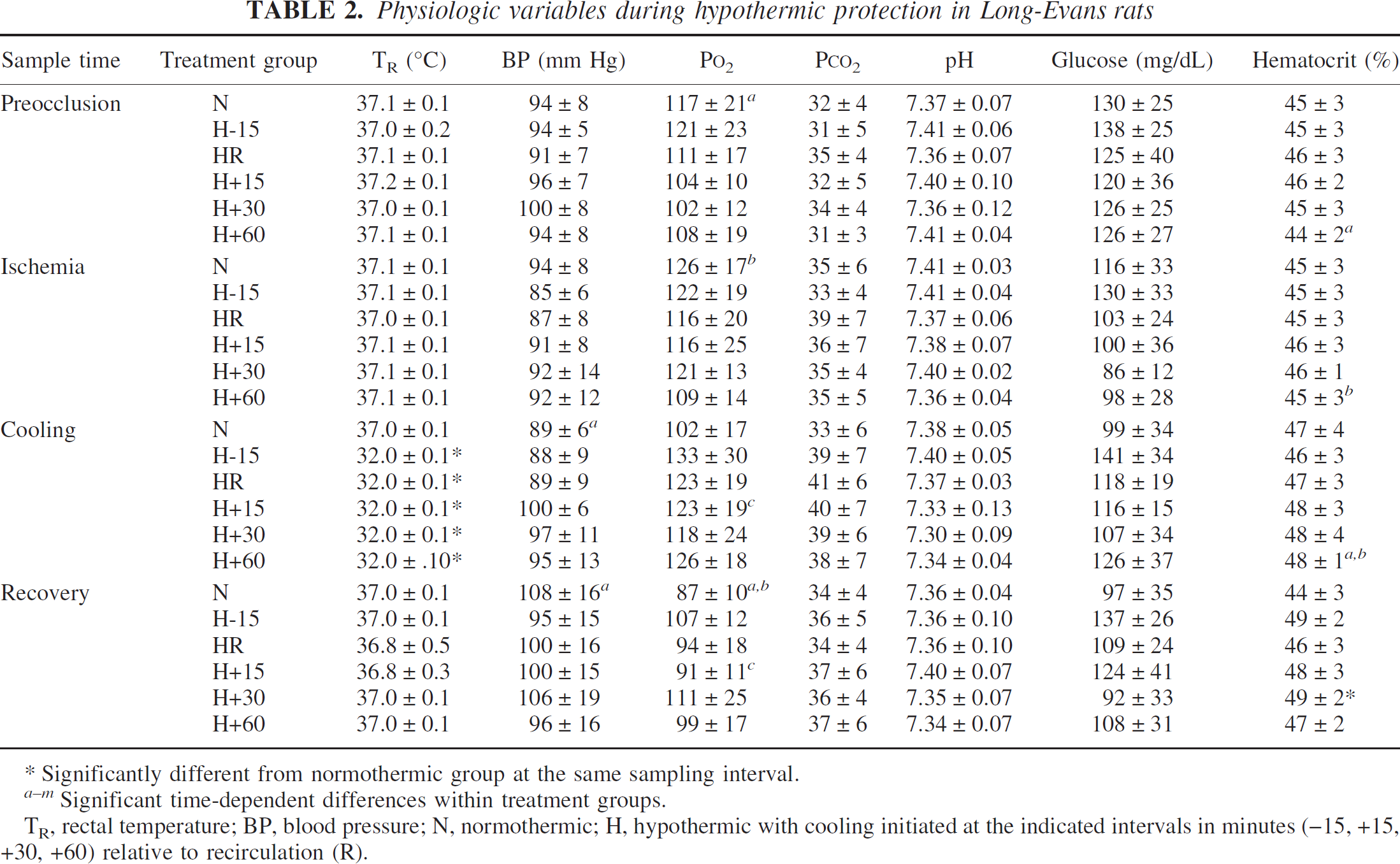
Significantly different from normothermic group at the same sampling interval.
Significant time-dependent differences within treatment groups.
TR, rectal temperature; BP, blood pressure; N, normothermic; H, hypothermic with cooling initiated at the indicated intervals in minutes (–15, +15, +30, +60) relative to recirculation (R).
Transient hypothermia in permanent focal ischemia
The effectiveness of a transient cooling protocol was also examined in models of permanent focal ischemia in SHR and Wistar rats. In these studies, animals were subjected to permanent right MCA/common carotid artery cauterization, with the standard 2-hour hypothermia protocol initiated 30 minutes after the onset of ischemia. Normothermic groups experienced the same duration of sustained anesthesia. Infarct volumes at 24 hours were significantly reduced by transient hypothermia in Wistar rats, from 160 ± 68 to 99 ± 40 mm3 (Fig. 5). This result represents the pooled data of independent experiments conducted by two investigators, each of which demonstrated statistically significant protection. Such an effect was not seen in SHR, for which lesion volumes in normothermic and hypothermic groups were 171 ± 30 and 173 ± 56 mm3, respectively.
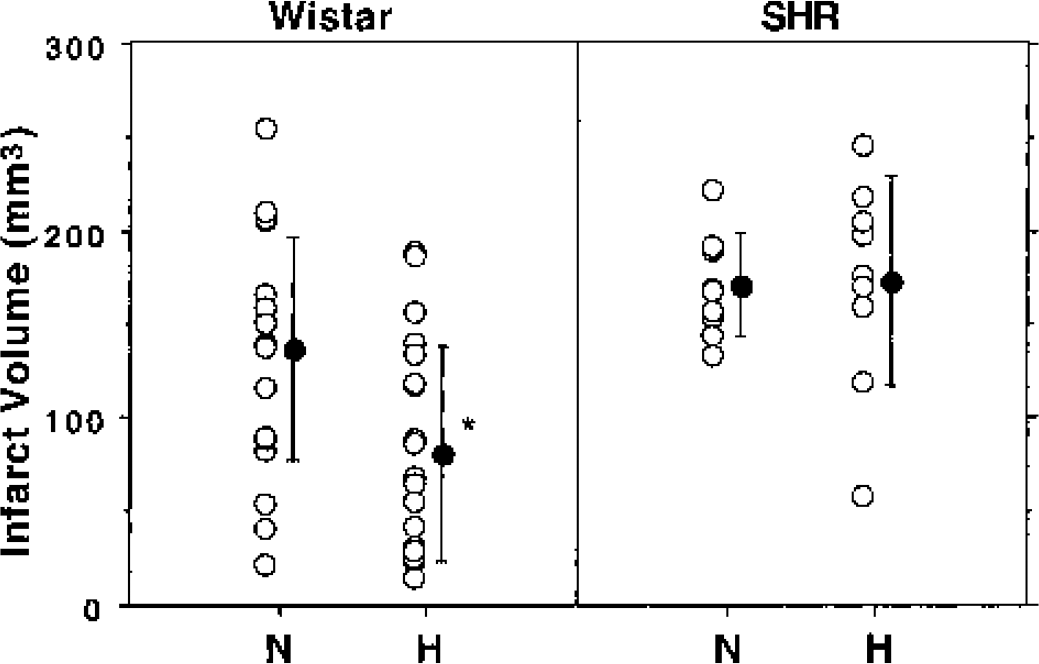
Strain differences in protection by transient cooling during permanent focal ischemia. Wistar rats (n = 41) and spontaneously hypertensive rats (n = 18) were subjected to permanent MCAO. Animals of each strain were then subjected to the standard cooling protocol beginning 30 minutes after occlusion or maintained for an equivalent interval under sustained normothermic anesthesia. Transient hypothermia reduced infarct volume at 24 hours in the Wistar strain but not in SHR (asterisk indicates a significant difference from normothermic animals, unpaired t-test). Filled symbols indicate group means and standard deviations.
DISCUSSION
These results illustrate striking differences in the temporal window for hypothermic protection among rat focal ischemia models. The main findings are that (1) sufficient brain cooling can extend the window of transient hypothermic intervention until the end of a 90-minute occlusion period in the SHR; (2) there is an expanded window for protection after 3-hour occlusion in a Long-Evans model of focal ischemia and reperfusion injury, extending until 30 to 60 minutes of recirculation; and (3) infarct volume can be reduced by early, transient cooling during permanent MCAO in Wistar rats, but not SHRs. These findings emphasize the critical importance of considering strain differences in evaluating hypothermic protection, and in extending such interventions to clinical trials in stroke.
Some general comments are in order before discussing results in the individual models. First, all of these studies used a consistent, relatively rapid cooling and rewarming protocol, with only 90 minutes at the minimum hypothermic temperature (Fig. 1). This permits the identification of narrow temporal windows for protective intervention, but may also contribute to the observed requirement for deeper brain cooling. Some studies have indicated adverse effects of rapid rewarming (Nishio et al., 2002), although a more gradual return to normothermia also implies an extended interval of hypothermia that could contribute to added protection. Moderate cooling can be tolerated for prolonged periods, but there may be practical advantages to deeper cooling during discrete intervals, and the identification of such critical periods is also mechanistically important. As reviewed in the introduction, there is a strong basis in the literature for a focus on the early reperfusion period, leading to the experimental design of the studies presented here. A second issue concerns the long-term efficacy of protection. Most of the present results were obtained at 24 hours' survival, but the critical observation of beneficial cooling just before reperfusion in the SHR was also confirmed at 3 days (Fig. 3). Although very delayed infarct progression has been described after brief periods of focal ischemia in one study (Du et al., 1996), we have established that no such phenomenon occurs in SHRs under the occlusion conditions used here (Hawkins BT, Howell RD, Nowak TS Jr, unpublished observations, 1997). Evidence for eventual injury progression after an initial phase of apparent hypothermic protection arises primarily in the context of global ischemic insults (Colbourne et al., 1997; Dietrich et al., 1993), in which selective neuron loss evolves over many days even under normothermic conditions. Nevertheless, it remains important that future studies examine the long-term pathological and behavioral consequences of optimal short-term cooling after focal ischemia, as has been done for prolonged hypothermia (Colbourne et al., 2000; Corbett et al., 2000).
Long-Evans rats
Results in the Long-Evans model are perhaps most straightforwardly interpreted. The increase in lesion volume after prolonged transient versus permanent MCAO (Fig. 2) reproduces previous observations (Aronowski et al., 1997), and supports the conclusion that events specifically occurring during delayed reperfusion contribute to infarct evolution in this model. A 3-hour occlusion was necessary to obtain a substantial infarct in the present study, which is somewhat longer than the 2-hour threshold previously reported, but this may reflect our use of younger animals and sustained anesthesia throughout the period of occlusion. The observation that a 2-hour cooling interval initiated within 30-minute recirculation was highly protective in this model (Fig. 4) is entirely consistent with the interpretation that considerable pathology evolved subsequent to recirculation, and defines a relatively narrow temporal window during which temperature-sensitive steps in this injury process must occur. The precise hemodynamic and biochemical features that distinguish transient and permanent occlusions in Long-Evans rats remain to be defined. As an important cautionary note, recent studies in this laboratory indicate that investigator-dependent variables such as position of the occlusion site along the MCA, as well as anesthesia duration and control of physiologic parameters after surgery, can markedly impact the distinction between temporary and permanent occlusions in this strain (Hashimoto and Nowak, 2003). Whatever the cause of such discrepancies, ongoing experiments indicate that there is no longer a significant postischemic protection window in Long-Evans rats under conditions of proximal occlusion that yield identical infarct volumes after permanent and prolonged transient ischemia (Hashimoto M, Zhao L, Nowak TS Jr, manuscript in preparation).
Spontaneously hypertensive rats
Infarct volume in the SHR was a well-behaved function of occlusion duration, increasing progressively to equal that obtained after permanent occlusions (Fig. 2), as previously described (Kaplan et al., 1991). Such characteristics do not directly distinguish a component of injury attributable to reperfusion. However, the finding that substantial infarct reduction could be achieved with 2-hour cooling of sufficient magnitude when initiated in the final minutes of the occlusion period (Fig. 3) is strongly suggestive of a brief, temperature-sensitive phase of injury propagation during early recirculation in the SHR. Subsequent studies have examined in more detail the magnitude and duration of hypothermia needed to achieve optimal protection in this model, and clearly establish a requirement for brain cooling below 30°C (Kurasako T, Pulsinelli WA, Nowak TS Jr, manuscript in preparation). This is in agreement with a previous study applying a longer 6-hour cooling interval immediately before recirculation that also indicated a requirement for deeper cooling to achieve significant protection (Huang et al., 1999). It is of note that striking protection has been described in the SHR with moderate but prolonged (48 hour) cooling even when delayed until 1-hour recirculation after 90-minute MCAO (Colbourne et al., 2000), which would seem to suggest both an extended protection window and a less stringent cooling requirement in the context of prolonged hypothermia. However, the ipsilateral common carotid artery remained permanently occluded in that study, introducing a component of persistent hypoperfusion and perhaps prolonging lesion evolution, and intraischemic temperatures also tended to be lower in the group later cooled during recirculation. Further studies are necessary to examine the impact of these methodologic details on the inferred hypothermic protection window.
In the present work, we also confirm the observation that transient hypothermia initiated soon after occlusion is not protective after permanent occlusions in the SHR (Ridenour et al., 1992), even with the more substantial brain temperature decrease employed in the present study. However, it should be noted that modest protection has been reported in one early study in this strain in which animals were deeply cooled (20°C rectal, 24°C temporalis muscle) before permanent occlusion and subsequent rewarming (Onesti et al., 1991). It therefore remains possible that very transient mechanisms operating at the onset of occlusion may be accessible to modulation, or that very deep cooling may have some lasting metabolic impact.
Wistar rats
Available evidence suggests that the Wistar strain provides a model with features intermediate between Long-Evans and SHR strains. Permanent occlusions in the Wistar rat were variable in our hands (Figs. 2 and 5), consistent with earlier results comparing rat strains using the same occlusion method (Brint et al., 1988), reflecting heterogeneity in vascular supply (Herz et al., 1996). However, there was a consistent tendency for long, transient occlusions to yield more reproducible infarcts than permanent occlusions (Fig. 2), suggesting by analogy to the Long-Evans model that a component of “reperfusion injury” may contribute to infarct expansion after transient occlusions in this strain. This comparison has also been noted in a recent study showing protective effects of mild hypothermia initiated 1 hour after release of a 2-hour occlusion in Wistar rats, in which magnetic resonance imaging was used to document lesion expansion during the subsequent 24 hours (Kollmar et al., 2002). The latter experiment replicated protection seen in an earlier study involving a similarly timed intervention in Wistar rats (Zhang et al., 1993a), although interpretation is somewhat complicated by the use of an intraluminal filament occlusion method in both studies (see below). Very prolonged cooling initiated at 30 minutes of recirculation after a short 30-minute filament occlusion has been confirmed to be protective at long survival times (Corbett et al., 2000).
Previous reports had indicated that transient cooling could be protective even in models of permanent surgical MCAO in Wistar rats (Baker et al., 1992; Kader et al., 1992), and results shown in Fig. 5 provide confirmation that infarct volume is significantly reduced by 2-hour hypothermia in this strain, whereas identical intervention in the SHR is ineffective. The present results also indicate that moderate cooling is as protective as profound (24°C) cooling (Baker et al., 1992). Clearly, temperature-sensitive mechanisms operate during a substantial postocclusion window, with or without overt reperfusion, that determine the extent of subsequent injury expansion in Wistar rats. One such mechanism could involve the transient metabolic challenges posed by periinfarct depolarizations (Back et al., 1994, 1995; Higuchi et al., 2002; Takeda et al., 1993). Pharmacologic manipulations that reduce such depolarizations have been reported to reduce infarct volume (Iijima et al., 1992; Mies et al., 1994), whereas additional depolarizations increase injury (Back et al., 1996). A dramatic reduction in periinfarct depolarizations has been reported during hypothermic ischemia in one Wistar study involving an intraluminal filament occlusion (Chen et al., 1993), although this attenuation was not seen in a Sprague-Dawley model combining surgical occlusion with hypotension (Sick et al., 1999). This raises the possibility that such interactions between hemodynamic and metabolic factors that potentially impact infarct expansion may differ with strain and occlusion method. Model-dependent differences in temperature regulation are considered in detail below.
Sprague-Dawley rats
The absence of consistent infarction in Sprague-Dawley rats precluded an examination of hypothermic protection using the occlusion method applied in these studies. However, cooling effects have been studied under more consistent insult conditions in this strain when MCA/CCAO was combined with additional occlusion of the contralateral common carotid artery (Goto et al., 1993; Markarian et al., 1996), or with systemic hypotension (Sick et al., 1999), or when ischemia was produced by an intraluminal filament occlusion method (Karibe et al., 1994a; Maier et al., 1998). Even mild hypothermia (33°C) reduced infarct volume when applied during a long (3 hour) interval of temporary surgical occlusion in Sprague-Dawley rats, and a further temperature reduction (30°C) was more protective (Goto et al., 1993). Delaying the initiation of mild intraischemic cooling for more than 30 minutes resulted in loss of protection in the same model (Markarian et al., 1996), although only 15% protection was obtained with cooling throughout occlusion and 1-hour recirculation, indicating the severity of this long ischemic insult. Mild cooling beginning at recirculation and extending through 24 hours was reported to be protective in this model, whereas a 1-hour cooling period was not (Yanamoto et al., 1996). A recent study showed that an extended period of moderate to mild hypothermia through 48 hours achieved 50% reductions in infarct size even after permanent occlusions, remaining evident through 21 days' survival (Yanamoto et al., 2001). Both of the latter studies also documented persistent temperature increases in the “normothermic” groups similar to those typically seen after intraluminal filament occlusions (see below), indicating that procedures designed to reduce collateral flow in Sprague-Dawley rats amplify damage in part as a result of hyperthermic effects, and thereby increase the sensitivity of such models to prolonged cooling. As noted previously, occlusions produced by such methods fail to permit attenuation of periinfarct depolarizations during hypothermia (Sick et al., 1999), raising the possibility that hemodynamic factors play a significant role in this phenomenon.
Intraluminal filament models
The present observations may not be straightforwardly generalized to studies involving intraluminal filament occlusions, but in view of the widespread application of this methodology it is important that some comparisons be considered. Such models invariably employ strains with substantial collateral flow to reduce mortality, particularly with permanent occlusions, and a majority of studies involving brain cooling having been carried out in Sprague-Dawley rats. Postischemic hyperthermia is a common feature of intraluminal occlusions (Kuluz et al., 1993; Zhao et al., 1994), apparently secondary to ischemic injury in hypothalamus (Ábrahám et al., 2002; He et al., 1999; Li et al., 1999), whereas we found no evidence of a temperature increase after surgical occlusion of the distal MCA, consistent with previous results (Yamashita et al., 1997). There is a reported temporal threshold of 90 to 120 minutes of occlusion for this effect (Li et al., 1999), although the magnitude and time course of temperature change in such models appear to be quite sensitive to methodologic variables (Ábrahám et al., 2002). Although the sometimes modest elevation in temperature produced under these conditions may not necessarily affect outcome after permanent ischemia (Li et al., 1999), its impact after transient ischemia must certainly be considered. Hyperthermia and occlusion duration clearly synergize to exacerbate insult severity, and attenuation of hyperthermia has been suggested to contribute particularly strongly to pharmacologic protection in this stroke model (Britton et al., 1997).
A second and interrelated difference between surgical and intraluminal filament approaches involves the vascular territories impacted by the respective occlusions. It is well recognized that there is heterogeneous CBF reduction outside the MCA distribution in suture occlusion models (Kanemitsu et al., 2002). Therefore, in addition to impacting hypothalamus and thereby temperature regulation, this expanded ischemic territory potentially increases the volume of penumbral tissue experiencing intermediate perfusion levels and an extended time course of infarct evolution, and such regions may exhibit sustained temperature sensitivity (Hakim, 1998). An early study in Sprague-Dawley rats showed a relatively short window of protection using mild (32–33°C) hypothermia, which was no longer effective if delayed until the second hour of a 2-hour occlusion (Karibe et al., 1994a); however, a study using similar methods found that cooling during the first hour was as protective as 2 hours of intraischemic hypothermia, whereas cooling for the first 30 minutes alone was not protective (Maier et al., 1998). Postischemic cooling for 3 hours was not itself protective after a 3-hour suture occlusion, and provided no further protection when it followed intraischemic cooling (Huang et al., 1998). However, similar postischemic cooling was found to reduce cortical infarct volume after a shorter (2 hour) filament occlusion (Huh et al., 2000). Interestingly, a striking protective effect was seen after a rapid saline flush through a hollow-bore occluding filament immediately before reperfusion, to which unrecognized cooling could have contributed (Ding et al., 2002). Long-term temperature changes after recirculation were not indicated in any of the above reports. A recent study documented postischemic temperature elevation in normothermic animals, and showed that dramatic protection could be achieved with an extended period of hypothermia initiated just 30 minutes before recirculation (Kawai et al., 2000), although the animals never regained normal temperatures in this experiment. As noted previously, a series of studies applying intraluminal filament occlusions in Wistar rats also showed substantial protection with either intraischemic hypothermia (Chen et al., 1992) or when applied during a delayed, 1-hour postischemic window (Kollmar et al., 2002; Zhang et al., 1993a). Although none of these studies evaluated delayed postischemic temperature changes, it is quite possible that this variable was also impacted by the manipulations. Conversely, Sprague-Dawley rats subjected to brief, 1-hour suture occlusions that did not cause overt hyperthermia showed increased damage if subjected to an interval of severe (40°C) brain heating 24 hours later (Kim et al., 1996). It remains to be established whether such effects can be generalized to other models in which a significant volume of ischemic penumbra might be expected. It should be noted that an extreme sensitivity to intraischemic temperature has been inferred from one study in which suture occlusions were produced in animals maintained at various set points appropriate to the diurnal temperature rhythm (Vinall et al., 2000). Diurnal variations in subsequent temperature responses during recirculation were not investigated, nor were expected variations in blood glucose levels monitored. Nevertheless, this report identifies yet another potential source of model variability impacting temperature. As a final comment, a trend toward smaller infarct volumes after permanent occlusion has also been noted in one study using suture occlusions in Sprague-Dawley rats (Kawai et al., 2000), suggesting that this could be a general finding in rat strains other than SHR, although such an effect has not always been observed (Li et al., 1999).
CONCLUSIONS
Perhaps the most important observation of the present study is that a brief period of relatively deep brain cooling initiated at or soon after recirculation offers effective protection after transient focal ischemia in SHR and Long-Evans models in which overt postischemic hyperthermia is not a confounding variable. This holds particular promise for a role of acute hypothermia in extending the window for tPA or other therapies targeting reperfusion after clinical stroke. In view of the magnitude of temperature reduction required, injury mechanisms impacted by this early postischemic intervention appear to differ substantially from those attenuated by intraischemic cooling. Further, the effectiveness of very transient cooling identifies a phase of injury progression during early recirculation that may be distinct from mechanisms impacted by delayed temperature manipulation in some models. It would appear that delayed injury mechanisms, perhaps comparable to those occurring during “reperfusion injury,” can operate during permanent occlusions in a Wistar model that permit substantial collateral flow. The rational design of cooling interventions in the spectrum of conditions presented in stroke patients clearly requires further studies comparing the requirements for optimal protection during different phases of infarct evolution in well-defined animal models. In this regard, it will likely be important to distinguish between benefits derived from the avoidance of hyperthermia versus those produced by overt cooling. Furthermore, although technical challenges currently limit its clinical applicability, selective brain cooling provides an important adjunct to systemic hypothermia in experimental studies. Finally, although a comprehensive assessment of the literature on model differences in pharmacological protectability is beyond the scope of this discussion, it is important to consider that variations in the therapeutic window for hypothermic protection among rat strains are likely to extend to other interventions.
Footnotes
Acknowledgements
Technical assistance was provided by Robert Howell.
