Abstract
The purpose of the present set of studies was to develop a new primate model of focal ischemia with reperfusion for long-term functional assessment in the common marmoset. Initially, the cerebral vascular anatomy of the marmoset was interrogated by Araldite-cast and ink-perfusion methods to determine the feasibility of an intravascular surgical approach. The methods showed that the internal carotid artery was highly tortuous in its passage, precluding the development of an extracranial method of inducing temporary middle cerebral artery occlusion in the marmoset. A pilot dose-response study investigated an intracranial approach of topically applying endothelin-1 (ET-1) to the M2 portion of the middle cerebral artery in a small sample of marmosets for up to 6 hours (n = 2 or 3 per group). Dose-dependent reductions in middle cerebral artery vessel caliber followed by gradual reperfusion were inversely related to increases in corrected lesion volume after ET-1 treatment, relative to vehicle control application. Finally, the functional consequences of ET-1–induced lesions to the M2 vascular territory were assessed up to 24 hours after surgery using the optimal dose established in the pilot study (2.5 nmol/25 μL). ET-1–treated marmosets (n = 4) showed marked contralateral motor deficits in grip strength and retrieval of food rewards and contralateral sensory/motor neglect towards tactile stimulation, relative to their ipsilateral side and vehicle-treated marmosets (n = 4). Strong correlations were shown between contralateral impairments and histopathologic parameters, which revealed unilateral putamen and cortical damage to the middle cerebral artery territory. No deficits were shown on general mobility, and self-care was promptly resumed in ET-1 marmosets after surgery. These results show that this novel model of ischemia with reperfusion in the marmoset has the potential to assess long-term function and to gauge the efficacy of novel therapeutic strategies targeted for clinical stroke.
Recent recommendations concerning the development of putative neuroprotective and restorative therapeutics highlight the importance of their evaluation in primate models of cerebral ischemia with assessment of longterm function (STAIR, 1999). Although significant progress has been made with a permanent middle cerebral artery occlusion (pMCAO) model in the New World common marmoset (Callithrix jacchus) (Marshall and Ridley, 1996), alternative models that incorporate reperfusion after MCAO need to be addressed on functional outcome and neuropathology in this nonhuman primate species.
Historical surgical techniques for inducing MCAO and subsequent reperfusion in nonhuman primates date back to over 30 years ago (Fukuda and del Zoppo, 2003). The majority of studies have used transorbital intracranial approaches to access the proximal portion of the middle cerebral artery (MCA) with subsequent temporary application of surgical clips (Crowell et al., 1970; Garcia and Kamijyo, 1974; Sundt et al., 1969; Young et al., 1997), ligatures (Crowell et al., 1981; DeGirolami et al., 1984; Marcoux et al., 1982), or inflation of cuffs (del Zoppo et al., 1986; Spetzler et al., 1980) in large, Old World monkeys (baboons and macaques) or New World monkeys (squirrel monkeys). However, interanimal variability in collateral blood flow and infarct volume, high incidences of mortality, and no reports of long-term objective functional assessment have underscored the “true” utility of these models to be predictive of the clinical context and assessing the efficacy of antiischemic compounds. Recently, a modified transorbital approach using a “triple-vessel” occlusion method in the baboon to restrict collateral blood flow during the occlusion period of 1 hour has been evaluated (Huang et al., 2000). Large subcortical and cortical infarcts within the MCA perfusion territory (∼32% of ipsilateral hemisphere) were robustly produced, as visualized by T2-weighted magnetic resonance imaging (MRI) and end-point 2,3,5-triphenyltetrazolium chloride staining, which were significantly correlated with a quantitative neurological assessment at 48 to 72 hours after MCAO. However, all these intracranial surgical approaches are quite invasive and require intensive care unit management within the immediate postoperative period. Furthermore these surgical methods ultimately prevent realistic long-term behavioral assessment because removal of the eye for access of the MCA confounds any functional measures for identifying visuospatial neglect, a key impairment associated with stroke.
Extracranial endeavors to induce MCAO and reperfusion in nonhuman primates have been championed by the development of autologous clot embolism techniques (Kito et al., 2001; Kuge et al., 2001), as opposed to microsphere or photochemically induced thromboembolism. These authors injected a large, single nonfragmented blood clot into the left internal carotid artery of macaques. This induced robust occlusion of the proximal MCA resulting in contralateral motor and sensory deficits that correlated with 24-hour end-point infarct volume. However, there was no evidence of recanalization or clot breakdown as shown by serial CBF measurements and visual inspection of the circle of Willis at end-point. Clearly, thromboembolic models can be hindered by the lack of control over the distribution of the clots, the onset and degree of spontaneous recanalization, and the variability in the size and location of the ensuing infarcts. In addition, the high degree of postoperative care required after recovery from surgery, potential incidence of mortality, and the feasibility of assessing long-term function beyond the classical 24-hour end-point in large primates makes this procedure arduous because the animals are debilitated after surgery.
To this end, alternative approaches were investigated in the small, laboratory-bred, New World monkey, the common marmoset. The common marmoset breeds readily in captivity, and weighs between 300 and 400 g when mature (at ∼18 months). The marmoset has a nongyrencephalic brain, yet has a brain size approximately eight times the size of, and a greater white matter to grey matter ratio relative to, rodents. The marmoset is small and relatively easy to handle, making them very practical for behavioral testing and for administering postoperative care. Furthermore the cerebrovascular anatomy, neuroanatomy, and behavioral repertoire of the marmoset are viewed to be more advanced than rodents because of its closer proximity to humans within the phylogenetic tree. From a functional perspective, the marmoset provides a significant advantage over rodents when assessing hand movement and hand-to-eye coordination, impairments that provide great distress to human stroke patients. Therefore, this places the marmoset, a New World monkey, in an ideal intermediate position between rodents and humans when assessing stroke pathophysiology, functional outcome, and the efficacy of antiischemic agents.
The current pMCAO approach in the marmoset, developed by Marshall and Ridley (1996), involves turning a bone flap in the skull to access the M1 and M2 portion of the MCA. This method obviates the need for a transorbital approach, but does require carefully lifting the frontal lobe to expose the proximal portion of the MCA, at a point at which it enters the lateral sulcus adjacent to the olfactory tract. This surgical method produced a consistent large area of infarction within the MCA perfusion territory, extending from the motor cortex, dorsal temporal cortex, and parietal cortex after M2 occlusions (Marshall and Ridley, 1996; Marshall et al., 1999), to the subcortical regions of the caudate, putamen, and underlying white matter after M1 occlusions (Marshall et al., 2000a, 2001). In addition, stable and long-lasting unilateral motor deficits were seen for up to 20 weeks after surgery (Marshall et al., 2001). Although contralateral neglect has also been successfully shown, improvements in this deficit are observed with time (Marshall and Ridley, 1996; Marshall et al., 2000b, 2001). This surgical model is clearly a logical step for determining long-term functional outcome in nonhuman primates after MCAO providing a robust MCA territory lesion volume that correlates with functional impairment (Marshall et al., 2001). However, the surgical intervention and postoperative care makes this procedure challenging, and the nature of the model does not allow an assessment of the contribution of reperfusion to functional parameters and to the size and distribution of the evolving infarct. Indeed, an investigation of reperfusion after occlusion in the common marmoset is warranted because recanalization can be instigated either spontaneously in a large percentage of clinical stroke patients (Ringelstein et al., 1992) or via thrombolytic therapy for those who meet the criteria for treatment. It is therefore very appropriate to study a reperfusion model in a nonhuman primate, such as the marmoset. Such a model may allow the identification of both acute and chronic functional changes after MCAO that may relate in some degree to the pMCAO model, which are just as important end-points as quantifying brain damage within the MCA territory.
The intraluminal thread model in the rat has been shown to be a reliable and robust model for inducing transient MCAO with long-term functional impairments that correlate with neuropathology within the MCA territory, as visualized by MRI and conventional histopathologic techniques (Modo et al., 2000; Virley et al., 2000). Moreover, the rat thread model, whether permanent or transient, is regarded as a valid “first-pass go/no-go” model to assess putative therapeutic agents targeted for stroke clinical trials. With reference to developing an intraluminal thread model in the marmoset, it is important to consider the highly tortuous and variable kinking of the internal carotid artery seen in higher primates. If the marmoset possessed this variable cerebral vascular anatomy then use of the “thread” technique would be precluded. Therefore, a thorough examination of the cerebral vasculature in the common marmoset is viewed to be an essential prerequisite before any surgical intervention. If the thread model was not viewed to be viable approach then appropriate alternatives should be considered, such as topical administration of the potent vasoconstrictor peptide, endothelin-1 (ET-1), to a proximal or distal branch of the MCA (Macrae et al., 1993), or stereotaxic infusion of ET-1 onto the proximal M1 segment of the MCA via guide cannulae (Sharkey and Butcher, 1995), as has been well characterized in the rat. Long-term functional impairments and the therapeutic potential of a number of putative neuroprotective compounds have also been assessed in these rodent ET-1–induced MCAO and reperfusion models (Dawson et al., 1995; Marston et al., 1995; Sharkey et al., 1996).
Therefore, the primary objective of this study was to establish an optimal method of inducing MCAO and reperfusion in the marmoset. Initially, we investigated the cerebral vascular anatomy of the common marmoset to assess the feasibility of an intraluminal thread approach for inducing MCAO and reperfusion. The results from this experiment indicated that this surgical approach would not be practical, so we decided that an assessment of dose response and the neuropathological consequences of ET-1–induced MCAO and reperfusion in a small sample of marmosets within the nonrecovery context was required to assess the reliability of the method. Finally, recovery after ET-1–induced MCAO for up to 24 hours after surgery, using an optimal dose of ET-1 established from the previous experiment, was investigated. Both functional outcome and end-point histopathology were assessed to confirm validity and reproducibility of the model for MCA territory infarction. Therefore, the ultimate objective of these experiments was to develop a reproducible model of focal ischemia with reperfusion for long-term recovery without compromising the welfare of the marmoset, which would be highly relevant to human stroke.
MATERIALS AND METHODS
All procedures were carried out under a project license in accordance with the UK Animals (Scientific Procedures) Act of 1986 and approved by an internal ethical review panel at GlaxoSmithKline Pharmaceuticals. The overall plan of work was to develop a reproducible model of MCAO and reperfusion in the marmoset with minimal debility, in terms of cost of welfare to the animal.
Investigation of the cerebral vascular anatomy
Araldite corrosion casts
Pilot studies were performed on four terminally anesthetized (sodium pentobarbital [Euthatal, 200 mg/mL, Merial Animal Health Ltd, UK]) male Sprague-Dawley rats (300–350g, Charles River, UK), using a 1:1:0.1 ratio of Araldite (MY750): Hardener (HY951): Red Color Paste (Mikrolith pigment paste, DW0133; all supplied by B & K Resins, Kent UK). This provided a standardized viscosity for the rate of infusion into a biological system. Transcardial perfusion was achieved by cannulating the aorta with polyethylene tubing (3 FG, outer diameter of 1.02 mm, Portex, England) via the left ventricle. Once the right atrium was cut, a prewash of heparinized 0.9% saline was delivered first followed by the Araldite mix, via an infusion pump, to the cerebral circulation. Because this method proved to be very effective, the protocol for infusion was adopted for assessment of the cerebral vascular anatomy in the common marmoset (n = 2, male and female, 350–400g). Animals were terminally anesthetized with sodium pentobarbital (Euthatal, 200 mg/mL, Merial Animal Health), and then the thoracic aorta was cannulated with polyethylene tubing (8 FG, outer diameter of 2.66 mm, Portex) via the left ventricle. Purse sutures were introduced around the ventricle of the heart, where the tubing was inserted, to support the tubing and to prevent any back flow of the Araldite mixture. The right atrium was then cut and heparinized 0.9% saline was initially infused as a prewash into the ascending aorta, followed by the mixture of Araldite: Hardener: Red Color Paste (1:1:0.1). The Araldite mixture was infused at a rate of 1.0 mL/min via an infusion pump. After 30 minutes, the Araldite mixture increased in temperature and subsequently formed an insoluble mass that prevented further infusion. The head was then carefully removed and placed in 20% potassium hydroxide for 3 or 4 days to remove skin, muscle, and bone tissue. Then the remaining cast was immersed in 50% potassium hydroxide and left for 14 days to remove any remaining debris. The solution of 50% potassium hydroxide was then changed and the cast was left immersed for a further 14 days. Once this time had elapsed, the cast was much cleaner from tissue debris and the branching patterns could clearly be discerned. The cast was finally rinsed with distilled water and then examined under a macroscope and photographed using a macro lens. Scale bars (in millimeters) were placed near the specimen to estimate of the distance of the MCA from the internal carotid artery (ICA) and the diameter of the ICA.
Ink perfusions
Two additional common marmosets (one male and one female, 350–400 g) were terminally anesthetized with sodium pentobarbital (Euthatal, 200 mg/mL, Merial Animal Health), and the thoracic aorta was cannulated with polyethylene tubing (8 FG, outer diameter of 2.66 mm, Portex). Purse sutures were again introduced around the ventricle of the heart, where the tubing was inserted and heparinized 0.9% saline was then infused as a prewash into the ascending aorta, followed by infusion of Quink black ink (Solv-x, Parker, UK) with neutral buffered formaldehyde in a ratio of 1:5 at a rate of 1.0 mL/min. After 30 minutes, the head was carefully removed and immersed in neutral buffered formaldehyde. After an additional 48 hours, the brain was carefully removed and the intracranial arterial anatomy could be clearly visualized and subsequently measured. Scale bars (in millimeters) were placed near the specimen to estimate of the diameters of the ICA and MCA.
ET-1–induced MCAO and reperfusion (nonrecovery)
Subjects and surgical procedure
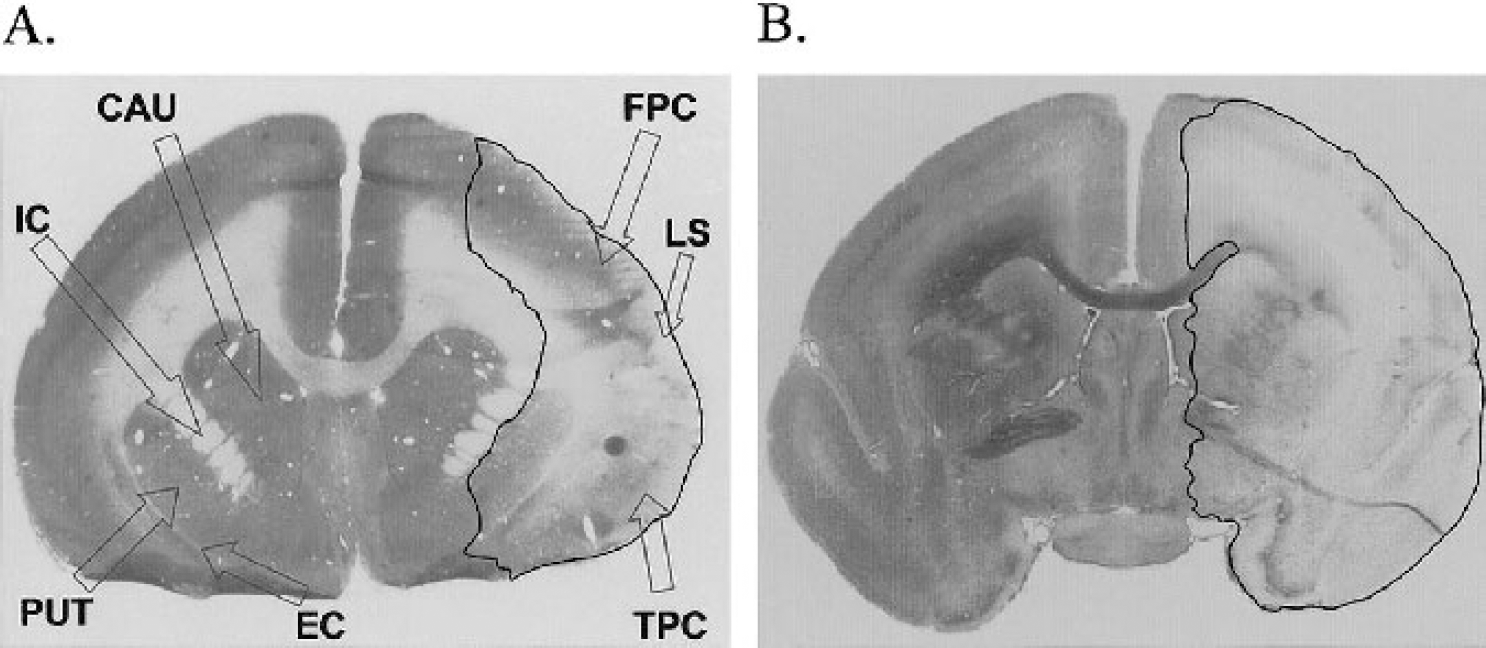
Ten common marmosets (C jacchus, eight males and two females, 365–400 g) underwent general anesthesia, induced by i.m. injection of alphaxalone/alphadolone (Saffan, 12 mg/mL, 0.15 mL/100g, Schering-Plough Animal Health, Schering-Plough Ltd, UK) and maintained by intravenous infusion (1.2–1.8 mL/h for anesthesia) via a tail vein cannulation, with the infusion rate adjusted to maintain adequate anesthesia for the duration of the experiment. Marmosets were also given fluid supplementation throughout the course of the experiment infusing normal saline (total intake was 1 mL/100g) into the tail vein via a three-way tap. All marmosets received dexamethasone (Dexadreson, 0.1 mL/100 g of a 5-mg solution, i.m., Intervet UK Ltd, UK) to minimize the possibility of life-threatening cerebral edema. Body temperature was thermostatically controlled using a rectal temperature probe with feedback to a heating blanket. Endotracheal intubation was performed to maintain a patent airway to facilitate artificial ventilation with oxygen and nitrous oxide (1:2), because of the duration of the experimental procedure without recovery. ECG, pulse oximetry, and capnography were used to monitor physiological status and depth of anesthesia. After an incision to the right side of the scalp, the periosteum of the skull and associated muscle were reflected to reveal anatomical landmarks on the skull. Topical application of local anesthetic (mepivacaine hydrochloride [Intra-Epicaine, Arnolds Veterinary Products Ltd, Shrewsbury, UK], 2.0% wt/vol) and hydrogel (Intrasite gel, Smith & Nephew Medical Ltd, UK; used to prevent dehydration) to the overlying muscle was performed before muscle incision and reflection. A small craniotomy was performed under an operating microscope to expose the M2 portion of the right MCA territory and facilitate viewing of the cerebral vasculature (Fig. 2). The dura and arachnoid membrane were pierced, via a stellate incision, in the vicinity of the M2 portion to permit a single abluminal application of ET-1 (Sigma UK, dose range of 0.1 [n = 3], 0.05 [n = 3], and 0.01 [n = 2] nmol/μL; rate of 5 μL/min for 5 minutes) or vehicle (water, at the same infusion rate, n = 2). Carefully controlled application of ET-1 or vehicle onto the MCA was achieved from a stylet that was attached to a cannula at one end. The other end of the cannula was connected to a graduated Hamilton syringe that was primed within an infusion pump for continuous delivery. Doses of ET-1 were selected on the basis of work established in the rat (Macrae et al., 1993). A micromanipulator was used for correct positioning of ET-1 or vehicle deposition on the exposed MCA. Successful MCAO was indicated by vasoconstriction and blanching of the cortex. In one marmoset per group, video footage of the procedure was recorded and later analyzed off-line in order to assess changes in MCA caliber (φ, in millimeters) over time. All marmosets remained under anesthesia for 6 hours after ET-1 or vehicle topical application and were continuously monitored throughout the experiment to ensure that the appropriate depth of anesthesia was maintained. At 6 hours after ET-1 or vehicle application, each marmoset was terminally anesthetized with sodium pentobarbital (Euthatal, 250 mg/kg i.v., Merial Animal Health) followed by a transcardiac perfusion-fixation using heparinized 0.9% saline and 4% paraformaldehyde. Brains were carefully removed and immersed in 4% paraformaldehyde. Coronal brain sections (60 μm) were cut at 180-μm intervals using a cryostat from the rostrocaudal axis of the brain and stained with cresyl violet and Luxol fast blue. Lesion volume analysis was then conducted using Datacell image analysis system with Optimas software, adopting Simpsons rule and correcting for hemispheric swelling. The total volume of the contralateral and ipsilateral hemisphere from the anteriorposterior axis in all marmosets were also measured to provide indices of percentage hemispheric swelling and total lesion as percentage of the ipsilateral and contralateral hemispheres.
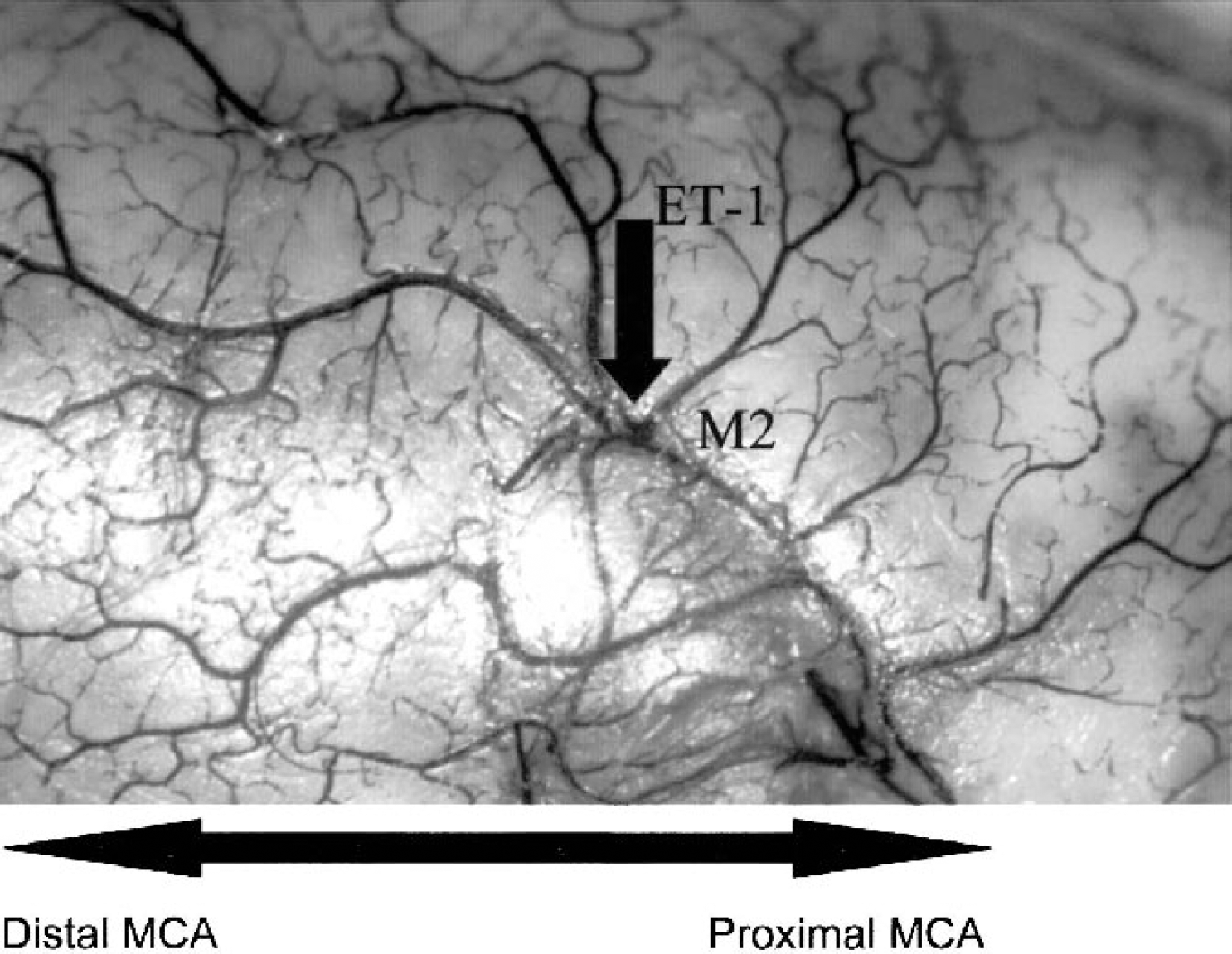
Ink perfusion showing the lateral sulcus and the M2 portion of the middle cerebral artery (MCA). Notice that the M2 aligns the lateral sulcus and then dives inwards and downwards, out of site, to the more proximal portion of the MCA. Topical application of endothelin-1 (ET-1) is indicated for clarification of surgical procedures. Large arrow indicates direction of MCA (i.e., distal to proximal location).
ET-1–induced MCAO (recovery)
Subjects and surgical procedures
Eight common marmosets (C jacchus, four males and four females, 347–488 g) were selected for this experiment. Each of these marmosets was well acclimatized to handling before the start of this experiment. Marmosets underwent general anesthesia, induced by i.m. injection of alphaxalone/alphadolone (Saffan, 12 mg/mL, 0.15 mL/100 g, Schering-Plough Animal Health) and maintained by intravenous infusion (1.2–1.8 mL/h for adequate anesthesia) via a tail vein cannulation, with the infusion rate adjusted to maintain adequate anesthesia for the duration of the surgical procedure. Marmosets were also given fluid supplementation throughout the course of the experiment infusing normal saline (total intake, 1 mL/100 g) into the tail vein via a three-way tap. All marmosets received dexamethasone (Dexadreson, 0.1 mL/100g of a 5-mg solution, i.m., Intervet) to minimize the possibility of life-threatening cerebral edema. Surgery was performed under aseptic techniques. Body temperature was thermostatically controlled using a rectal temperature probe with feedback to a heating blanket. Endotracheal intubation was performed to maintain a patent airway, and marmosets were allowed to spontaneously breathe throughout the surgical procedure with supplementation of oxygen with artificial ventilation if required. Because the duration of the surgical procedure was significantly less than that of the nonrecovery experiment, and because of the potential hazards of recovering from anesthesia, marmosets spontaneously breathed throughout the surgical procedure. ECG, pulse oximetry, and capnography were used to monitor physiological status and depth of anesthesia. A cannula was also secured in a tail artery and exteriorized to allow the withdrawal of blood samples for blood gas assessment. Surgical preparation to expose the M2 portion of the MCA was similar to that explained in the previous section. Once the dura and arachnoid membrane were pierced (via a stellate incision) in the vicinity of the M2 portion, a single abluminal application of ET-1 (dose of 0.1 nmol/μL at a rate of 5 μL/min for 5 minutes, for a total dose of 2.5 nmol/ 25 μL, n = 4) or vehicle (water, at the same infusion rate, n = 4) was delivered to the MCA from a graduated Hamilton syringe, attached to an infusion pump. The craniotomy was closed by using a thermoplastic convex disc and then sealed with bone wax, and the muscle and skin sutured closed. Physiological monitoring continued until each marmoset recovered from anesthesia. The endotracheal tube was then removed and a facemask was then introduced to provide oxygen to facilitate complete recovery from anesthesia. Postoperative buprenorphine (0.01-mg/kg Vetergesic, Alstoe Ltd, UK) was then administered subcutaneously. Each marmoset was then placed in a thermostatically controlled intensive care unit (an incubator lined with soft bedding material [VetBed, Petlife International Limited, UK]) in a darkened and quiet postoperative room for the initial recovery phase of the experiment (i.e., 16 hours), with access to soft food and water. The postoperative room was in close proximity to the marmoset colony room so that audible vocal exchanges between marmosets could be maintained. Routine checks were made by veterinary staff and experimenters during the immediate postoperative period and during the night to check on the welfare of the marmosets, and the use of further analgesic administration was available if required. The following morning, each marmoset was placed in a suitably padded (with VetBed) postoperative cage, located within the postoperative room, containing a perch located horizontally within the center of the cage and nest box (placed on the padded cage floor), with access to soft food and water. A piece of VetBed that was scent marked by the marmoset a few days before surgery was also placed within the incubator and transferred to the postoperative cage to facilitate recovery.
Behavioral assessment
Marmosets placed in the padded postoperative cage were observed for neurological status and simple behavioral tasks. Measurements were recorded preoperatively to provide measurements at baseline and at 24 hours after surgery. A neurological grading system that was developed to assess marmosets subjected to the pMCAO surgical procedure (Marshall and Ridley, 1996) was used to measure the presence or absence of particular movements and postural abnormalities at 24 hours after surgery. Each marmoset was closely observed for the occurrence of four abnormal signs on each side of the body: (1) a hand or foot slipping off the perch; (2) a hand left dangling below the level of the perch; (3) uncharacteristic postures in which the marmosets hold their arm across the midline of their chest; and (4) uncoordinated arm movement and hand waving. The total number of signs was totalled to give a maximum score of 4 for each side of the body. In addition, freely emitted behaviors such as vocalizations (frequency and call type: phee, long-distance contact call; tsik, predator/alarm call; Stevenson and Poole, 1976) and mobility (time spent on horizontal perch, cagefront, or cage floor, in seconds) were recorded during a 5-minute test session.
At the end of the test session, the marmosets were coaxed to the front of the cage for marshmallow or small pellet-pink reward (Baby rice, Farleys, UK) mixed with vitamin B12 (Cytacon syrup, Goldshield Pharmaceuticals, UK) and vitamin D3 oil (Vetoquinol) to form a soft dough. The experimenter provided five pieces of food reward to each forelimb, in a random order at the center of the cage and the successful retrieval of each food reward by each forelimb was recorded. The purpose of this simple test was to provide a useful index of fine-motor retrieval in central space similar to the more sophisticated staircase test and the two-tube choice and six-tube search tests adopted by Marshall and Ridley's group (Marshall and Ridley, 1996; Marshall et al., 1999, 2000a,b, 2001) to assess retrieval in pMCAO marmosets.
Each marmoset was then carefully caught and removed from the postoperative cage to assess sensorimotor function. Assessment of grip strength for each forepaw and hindpaw was subsequently made by gently allowing the marmoset to grip the shaft of a paintbrush while the experimenter pulls it away. This procedure was repeated three times to each forepaw and hindpaw in a random fashion and scored as follows: 2 = clearly noticeable grip to the stimulus; 1 = modest grip but lacking strength; 0 = no grip. Assessment of a forelimb and hindlimb tactile response to the hairs of a paintbrush was then conducted. Each forelimb and hindlimb were stimulated 3 times in a random fashion and scored as follows: 2 = clearly noticeable reflex to the stimulus; 1 = modest reflex to stimulus; 0 = no response to stimulus. A response to tactile stimulation of the ears was finally conducted by a modification of the Von Frey hair test (Schallert et al., 1982). Essentially, the marmoset was held around the torso facing the experimenter, who reached from behind the marmoset and, without intruding into the visual field, lightly stroked one ear tuft/ear with a paintbrush and recorded the response (2 = clearly noticeable response to the stimuli, e.g., attempting to bite the brush; 1 = mild almost negligible response to the stimuli, e.g., turn of the eyes; 0 = no observable effect). The order of stimulation of the ears in pairs was random and was repeated for a total of three pairs (after Marshall and Ridley, 1996).
Quantification of ischemic damage
At 24 hours after ET-1 or vehicle application, the marmosets were terminally anesthetized with sodium pentobarbital (Euthatal, 250 mg/kg i.p., Merial Animal Health) followed by a transcardiac perfusion fixation using heparinized saline and 4% paraformaldehyde. The brains were then carefully removed and immersed in 4% paraformaldehyde and coronal brain sections (60 μm) were then cut on cryostat at 180-μm intervals and stained with cresyl violet and Luxol fast blue to assess the integrity of grey and white matter, respectively. Lesion volume analysis was conducted using a Data cell image analysis system with Optimas software, adopting Simpsons rule and correcting for hemispheric swelling. An index of the longitudinal extent of the lesion was also performed to assess the number of sections that the lesion encompassed. The total volume of the contralateral and ipsilateral hemisphere from the anterior-posterior (12.5–4.5) axis (Stephen et al., 1980) in all marmosets were also measured to provide indices of hemispheric swelling and total lesion as percentage of the ipsilateral and contralateral hemispheres.
Statistical analysis: ET-1–induced MCAO (recovery) Behavior
The behavioral data were analyzed using multivariate analysis of variance (MANOVA). Essentially, between-subject factors (group and sex) and within-subject factors (preoperative and postoperative assessment; ipsilateral and contralateral side) were assessed as main effects and interactions within the analysis of variance. Where statistical significance was observed, two-tailed post hoc Newman-Keuls t-tests were applied to compare specific differences between groups and ipsilateral and contralateral sides at both preoperative and postoperative assessments. All analyses were performed using the Statistica (Statsoft Inc., Tulsa, OK, U.S.A.) statistical package. In all analyses P < 0.05 was considered statistically significant.
Histopathological analysis
All histopathological parameters were compared between ET-1–treated and vehicle-treated marmosets at 24 hours after surgery by independent t-tests (two-tailed). In all analyses, P < 0.05 was considered statistically significant.
Correlational analysis
Correlational analyses using Pearson r were performed to determine relationships between behavioral impairments observed at 24 hours in ET-1–treated marmosets with histopathological measures. In all analyses P < 0.05 (two-tailed) was considered statistically significant.
RESULTS
Cerebral vascular anatomy
Araldite corrosion casts
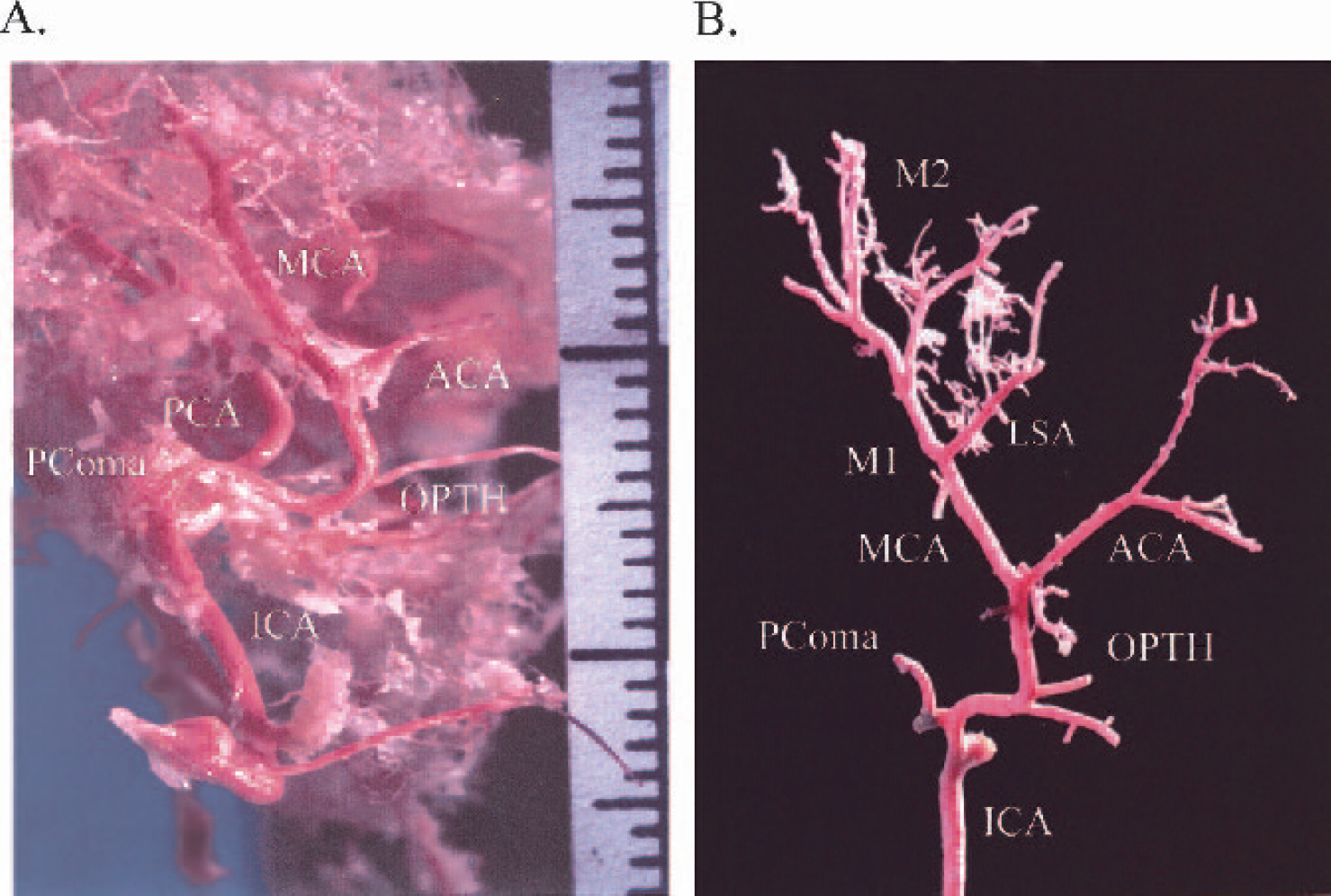
The common carotid artery was clearly delineated, as was the carotid bifurcation, giving rise to both the external carotid artery and ICA in both animals (Figs. 1A and 1B). Each ICA (n = 4, two from each animal), approximately 1 mm in diameter, began its course by ascending vertically approximately 8 mm, before making an S-shaped bend in an upward horizontal direction to curve to a point that passes vertically and laterally to bifurcate into the anterior cerebral artery (ACA) and MCA (see Figs. 1A and 1B). The distance from the end of the S-shaped bend (carotid siphon) to the origin of the MCA was approximately 6 mm. The ophthalmic artery was also visible on all marmoset casts and was located 3 mm from the end of the S-shaped bend (Figs. 1A and 1B). The posterior communicating artery departed from the ICA at the carotid siphon (see Fig 1A and 1B). The diameter of the MCA was approximately 0.5 mm. The intracranial vascular anatomy of the marmoset was very similar to that of the human arterial anatomy in terms of course and tortuosity.
(
Ink perfusions
The ICA ascended after a tortuous bend to a juncture that gave rise to the MCA and ACA. The diameter of the ICA at the end of the S-shaped carotid siphon was approximately 1 mm. The origin of the MCA (M1 segment) clearly departed from the arterial circle, 2 mm medial to the olfactory tract, to enter into the lateral sulcus, with a diameter of approximately 0.5 mm. The lenticulostriatal arteries were also visualized as S-shaped complexes with diameters of approximately 0.1 mm. The MCA continued to run inwards and upwards within the inferior frontal region, until it turned 90 degrees and emerged from deep within the brain parenchyma at the cortical surface aligning the lateral sulcus (M2 segment; Fig. 2). The M2 segment then extended forwards and backwards to supply the frontoparietal and temporoparietal cortices, respectively (Fig. 2).
ET-1–induced MCAO (nonrecovery)
Physiological parameters and changes in MCA caliber
Physiological monitoring throughout the procedure showed that parameters were within acceptable limits (min-max range) for animals that were artificially ventilated; i.e., body temperature (35–36°C), heart rate (180–220 beats per minute), O2 (95–100%) and EtCO2 (36–46 mm Hg) for each treatment group. Dose-dependent vasoconstriction followed by reperfusion was shown in all the ET-1–treated marmosets (Table 1 and Fig. 3), but not in vehicle-treated marmosets.
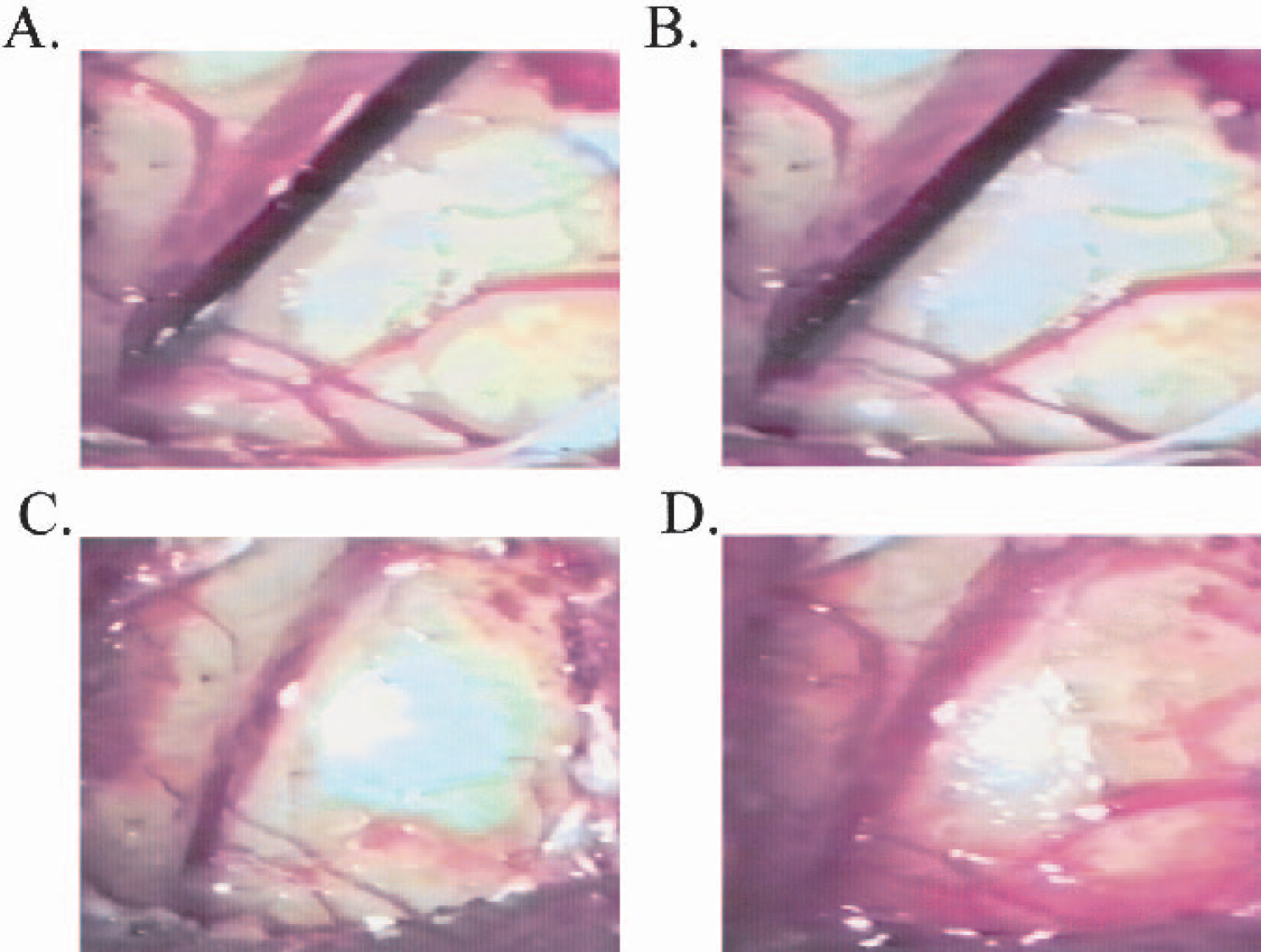
Images of surgical procedure using ET-1 (0.1 nmol/μL). (
ET-1 induced MCA caliber changes

Percentage (%) change in MCA caliber, from pre-application diameter over time following ET-1/Vehicle application. ET-1, endothelin-1.
Neuropathology
Dose-dependent induction of corrected lesion volume (in cubic millimeters) by ET-1 was shown at 6 hours, consistently affecting the M2 vascular territory (mean ± SD, n = 2 or 3: ET-1 0.1 nmol/μL, 390.70 ± 38.00; ET-1 0.05 nmol/μL, 362.96 ± 140.89; ET-1 0.01 nmol/μL, 173.75 ± 85.82; vehicle, 50.18 ± 36.90; Figs. 4 and 5). This was further corroborated by assessing the lesion extent (number of sections) across the anterior-posterior axis (mean ± SD, n = 2 or 3: ET-1 0.1 nmol/μL, 35.00 ± 2.65; ET-1 0.05 nmol/μL, 28.33 ± 6.11; ET-1 0.01 nmol/μL, 23.50 ± 9.19; vehicle, 19.00 ± 7.07). No major differences were shown for percentage hemispheric swelling across the groups as end-point (mean ± SD, n = 2 or 3: ET-1 0.1 nmol/μL, −1.43 ± 1.65%; ET-1 0.05 nmol/μL, + 0.34 ± 2.84%; ET-1 0.01 nmol/μL, −4.98 ± 2.81%; vehicle, −4.55 ± 2.24%). Additional assessment of corrected lesion volume as a percentage of the ipsilateral hemisphere showed a dosedependent effect of ET-1 (mean ± SD, n = 2or 3: ET-1 0.1 nmol/μL, 31.15 ± 4.25%; ET-1 0.05 nmol/μL, 25.43 ± 1.80%; ET-1 0.01 nmol/μL, 20.10 ± 3.27%; vehicle, 5.49 ± 2.57%). This also accorded well with corrected lesion volume as a percentage of the contralateral hemisphere (mean ± SD, n = 2 or 3: ET-1 0.1 nmol/μL, 30.67 ± 3.73%; ET-1 0.05 nmol/μL, 29.99 ± 4.72%; ET-1 0.01 nmol/μL, 19.05 ± 2.54%; vehicle, 5.21 ± 2.33%). Cresyl violet and Luxol fast blue staining consistently revealed extensive disorganization within cortical cell layers of ET-1–treated marmosets, relative to the contralateral side (Figs. 6 and 7), and vehicle-treated control marmosets. Furthermore, high-power microscopy revealed neuronal cell swelling, pyknosis, and a vacuolated neuropil within and around the ipsilateral lateral sulcus (Figs. 6A and C) of ET-1–treated marmosets that extended into the ipsilateral frontoparietal cortex (Fig. 7A) and temporoparietal cortex. Interestingly, one marmoset given the middose of ET-1 (0.05 nmol/μL) sustained an M1 territory infarction (525 mm3) as the caudate and putamen were shown to be severely damaged (Figs. 4 and 5B), possibly due to ET-1 diffusing down to the proximal M1 segment of the MCA. This effect was not reproduced in the next two marmosets administered the same dose of ET-1.
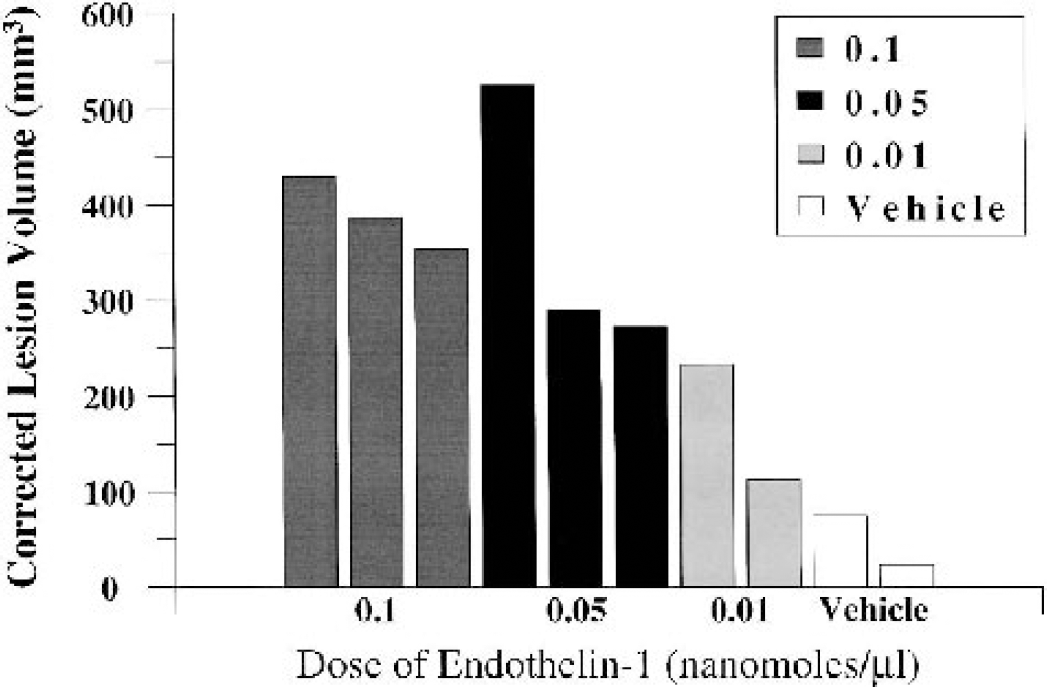
Corrected lesion volume (in cubic millimeters) for marmosets treated with ET-1 (0.1, 0.05, and 0.01 nmol/μL) or vehicle (n = 2 or 3 per group) at 6 hours after application (nonrecovery experiment). Individual data points for each marmoset are plotted. Notice the dose-dependent induction of corrected lesion volume, other than the one marmoset treated with the middose (0.05 nmol/μL) of ET-1 who sustained an M1 territory infarction.
(
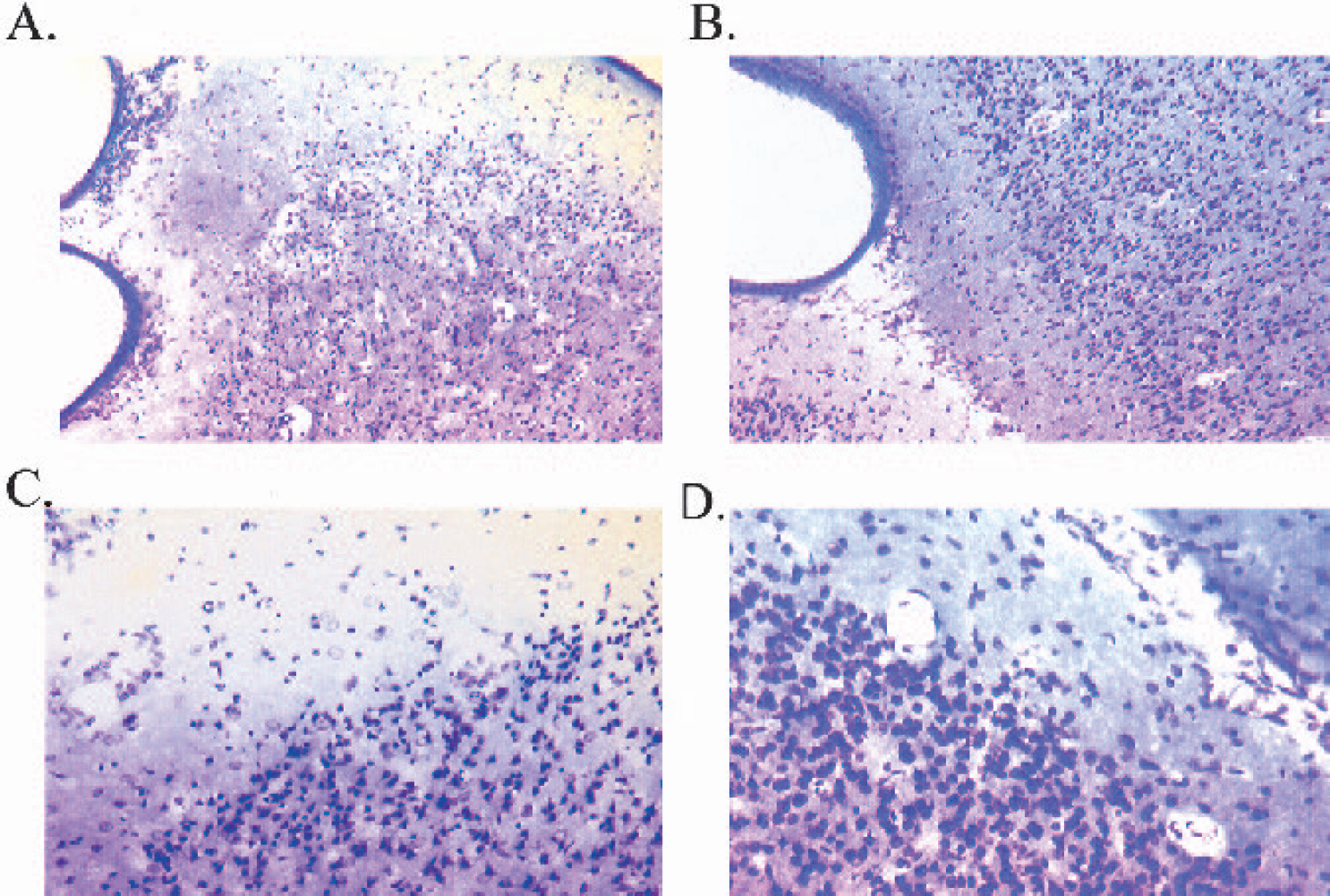
High-power light microscopy photomicrographs showing a typical lesion around the ipsilateral lateral sulcus (
High-power light microscopy photomicrographs (×10 magnification) of the ipsilateral frontoparietal cortex (
ET-1–induced MCAO (recovery)
Physiological parameters
Physiological monitoring throughout the procedure showed parameters to be within acceptable limits (min-max range) for animals that were breathing spontaneously, supplemented with oxygen, and not artificially ventilated; i.e., body temperature (35–36°C), heart rate (173–255 beats per minute), O2 (90–99%), and EtCO2 (45–70 mm Hg) for each treatment group. Arterial blood gases also showed acceptable limits (min-max range) for both ET-1–treated and vehicle-treated marmosets: pH (7.28–7.32), P
Behavioral assessment
Immediate postoperative neurological deficits. All marmosets recovered their righting reflex approximately 30 minutes after the cessation of anesthesia. For the first 6 hours after recovery from ET-1–induced MCAO surgery, marmosets displayed obvious signs of neurological dysfunction, including a modest deviation of the head and rotation/circling towards the side of the lesion. Each ET-1–treated marmoset also showed an unsteady gait. Indeed, marmosets that were treated with ET-1 had a tendency to hold their contralateral forearm close to their chest in the immediate postoperative phase. In addition, when a sudden auditory stimulus (hand clap) was initiated in contralateral space by an observer (out of sight from marmoset), the ET-1–treated marmosets did not respond, which is indicative of neglect. This response was in direct contrast to that elicited when the auditory stimulus was initiated in an ipsilateral space, to which all ET-1–treated marmosets responded. Moreover, when ET-1–treated marmosets were gently stimulated with the shaft of a paintbrush, they showed no grip or reflex with their contralateral forearm or hindleg, unlike their ipsilateral forearm and hindleg, which showed a normal grip and reflex to the stimulus. Marmosets began to drink from the water bottle provided within a few hours after recovery from anesthesia, whereby they used their ipsilateral hand to position themselves against the side of the incubator to obtain the water from the spout, demonstrating weakness in the contralateral forearm and hindleg. Indeed, they shifted their weight onto the ipsilateral side to compensate for the weakness on the contralateral side. ET-1–treated marmosets all started to use their ipsilateral forearm and hindlimb to scratch and groom, but did not use their contralateral limbs for this purpose. Generally, all ET-1–treated marmosets were quiet after surgery and emitted few if any vocalizations, and frequently showed periods of sedation and lethargy with their heads faced down on the soft bedding within the incubator. This may have been due to the surgery, analgesia, and/or recovery from anesthesia. Before the end of the day, all marmosets were provided with bread, a small piece of banana, and baby rice mixed with drinking water. ET-1–treated marmosets took the piece of banana and bread using their ipsilateral forearm and ate some of baby rice provided in a small dish. In direct contrast to the above findings, all four vehicle-treated marmosets showed no abnormal signs of neurological dysfunction, showed equivalent use of both ipsilateral and contralateral limbs, and responded to sounds initiated within both the ipsilateral and contralateral hemispaces. Furthermore, they tended to be much more mobile and vocal within the incubator. However, all vehicle-treated marmosets did show signs of sedation near the end of the day, as indicated by placing their heads face down on the soft bedding within the incubator, which again may have been due to the surgery, analgesia, and/or recovery from anesthesia.
Total neurological score
During the preoperative session, none of the marmosets exhibited abnormal neurologic signs that were part of the grading scale. At 24 hours after surgery, two of the abnormal signs were observed in the majority (three out of four) of ET-1–treated marmosets; i.e., (1) hands and/or feet slipping off the perch and (2) hands left dangling below level of the perch. In one of the ET-1–treated marmosets, only the first abnormal sign was evident (i.e., hands and/or feet slipping off the perch). Clearly, ET-1–treated marmosets had problems with posturing and fine coordination using their contralateral limbs. None of these signs was shown in the vehicle-treated group of marmosets at 24 hours after surgery. A MANOVA showed a significant group by time by side interaction (F1,4 = 49.00, P < 0.005). Post hoc comparisons revealed a significant main effect of group between the vehicle and ET-1 contralateral side in terms of abnormal signs at 24 hours after surgery (P < 0.001; Table 2). Furthermore, post hoc comparisons revealed a significant neurological impairment on the contralateral side in ET-1–treated marmosets relative to their preoperative contralateral score and their postoperative ipsilateral score (P < 0.01; Table 2). No sex effect was shown, suggesting that there was no difference in neurological scores between males and females, irrespective of treatment group.
Total neurological score
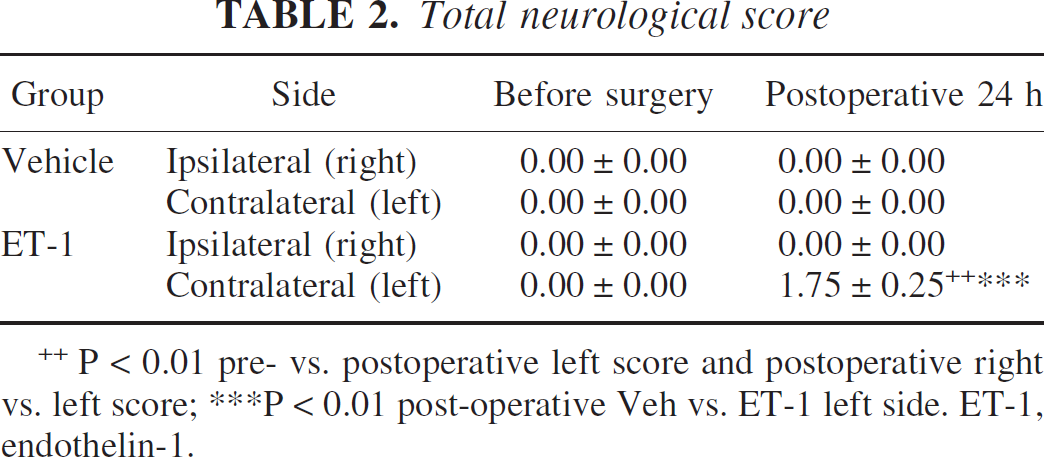
P < 0.01 pre- vs. postoperative left score and postoperative right vs. left score; ***P < 0.01 post-operative Veh vs. ET-1 left side. ET-1, endothelin-1.
General activity and vocalization frequency
During the preoperative session, all marmosets spent the majority of their time either on the perch or on the cagefront during the 5-minute test period. Most of the animals spent little time on the cage floor, suggesting that the marmosets were fairly active in the novel environment. Some of the marmosets freely emitted long-distance contact calls (phees) and novel stimulus alarm calls (tsiks), whereas others were essentially silent. At 24 hours after surgery, ET-1–treated marmosets tended to spend less time on the perch, shifting their position to the cagefront, whereas one marmoset remained on the cage floor for the duration of testing. However, ET-1 marmosets were able to climb and jump from different locations within the cage but clearly exhibited postural abnormalities highlighted by occasional slips of contralateral limbs off the perch and on the front of the cage. Vehicle-treated marmosets showed a similar propensity to sit on the cagefront and perch with respect to their preoperative baseline test period, although one marmoset did spend some time on the cage floor but not as long as the ET-1–treated marmoset. The frequency of vocalizations tended to diminish in those marmosets that were highly vocal in the preoperative session, irrespective of treatment, suggesting that habituation to the test environment may have attenuated the number of contact calls and novel stimulus calls emitted. Similarly, those marmosets that did not emit calls within the preoperative session remained silent during the postoperative test period. A MANOVA showed no significant differences in terms of interactions or main effects (F1,4 < 4.0, P > 0.05; Tables 3A and 3B) on each of the general activity and vocal frequency parameters. These results suggest that the group of ET-1–treated marmosets recovered well from surgery and did not differ in terms of general activity and vocalization repertoire from vehicle-treated marmosets and their respective preoperative baseline measures.
General activity(s)
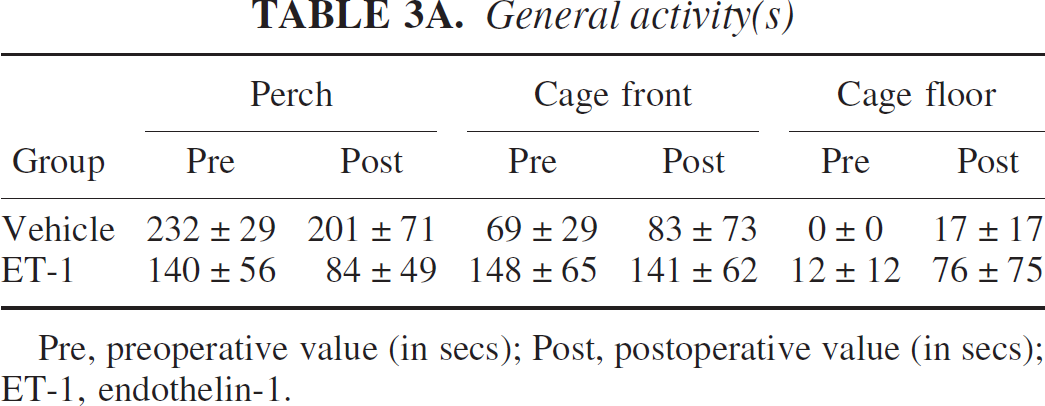
Pre, preoperative value (in secs); Post, postoperative value (in secs); ET-1, endothelin-1.
Frequency of call types

Pre, preoperative value (number of calls); Post, postoperative value (number of calls); ET-1, endothelin-1.
Forelimb retrieval of food rewards from cagefront
During the preoperative session, all marmosets approached the experimenter from the center of the cage avidly and successfully retrieved food rewards with both the ipsilateral and contralateral forearm. Before surgery, all marmosets were matched for ability, in that they were able to retrieve all the food rewards equally with their left and right hands. At 24 hours after surgery, ET-1–treated marmosets had significant difficulty in using their contralateral forearms to successfully retrieve food rewards. Only one of the marmosets was able to retrieve just one of the food rewards with its contralateral hand out of five random trials. All the remaining ET-1–treated marmosets could not initiate or execute control in their contralateral forearm to retrieve rewards coaxed to their contralateral hands in each of the five random trials. However, when rewards were presented in close proximity to their ipsilateral hands the majority (three out of four) ET-1–treated marmosets successfully retrieved all food rewards. One of the ET-1 marmosets retrieved only three out of the five rewards presented to the ipsilateral hand, possibly because of reduced motivation during the specific test session. An interesting observation was noted when rewards were presented at other times (i.e., not during the test session) in contralateral space where the ET-1–treated marmosets did not respond, but as the rewards moved from contralateral to ipsilateral space, the rewards seemed to come into view and the marmosets immediately responded by avid retrieval with their ipsilateral hand. This suggests that ET-1–treated marmosets not only have a contralateral motor impairment but also a contralateral spatial neglect. Vehicle-treated marmosets showed equivalent performance with both ipsilateral and contralateral hands for food rewards presented at the center of the cage. When food rewards were presented at other times (not during the test session) in contralateral space, the vehicle-treated marmosets responded quickly to acquire the food reward using their ipsilateral and contralateral forearms equally. A MANOVA showed a significant group by time by side interaction (F1,4 = 25.00, P = 0.005). Post hoc comparisons revealed a main effect between the vehicle and ET-1–treated group on the contralateral side at 24 hours after surgery, in terms of an impaired contralateral retrieval displayed by the ET-1–treated animals (P < 0.001; Fig. 8). Furthermore, post hoc comparisons also revealed a significant difference between preoperative and postoperative contralateral forelimb retrieval and between postoperative ipsilateral and contralateral forelimb retrieval in ET-1–treated marmosets (all P < 0.01; Fig. 8). No sex effect was shown, suggesting that there was no difference in forelimb retrieval for males and females, irrespective of treatment group.
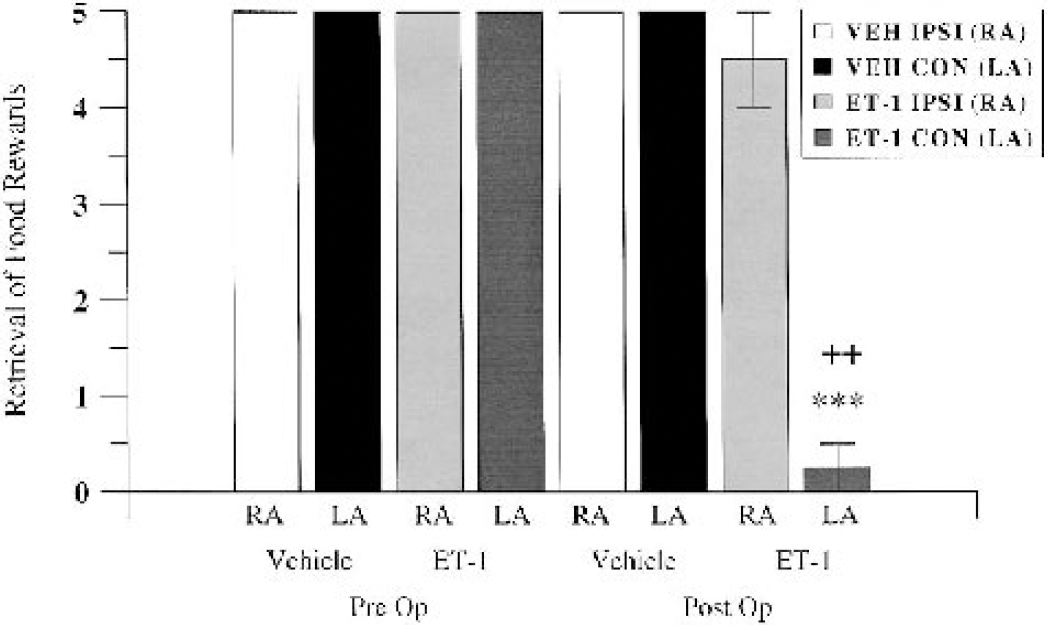
Retrieval of successful food rewards presented at the center of the cage by vehicle-treated (VEH) and ET-1–treated marmosets at preoperative baseline and 24 hours after surgery (n = 4 per group). RA and LA refer to right (ipsilateral) and left (contralateral) arms. ***P < 0.001 between vehicle and ET-1 contralateral left arms at 24 hours; ++P < 0.01 between ipsilateral right arms and contralateral left arms for the ET-1 group at 24 hours. Pre Op, preoperative; Post Op, postoperative.
Forelimb grip strength
During the preoperative session, all marmosets showed a clearly noticeable grip to the shaft of the paintbrush with both ipsilateral and contralateral hands. At 24 hours after surgery, the majority (three out of four) of ET-1–treated marmosets showed either a modest grip lacking strength or no grip at all with the contralateral hand. One marmoset showed a modest to noticeable grip with its contralateral hand over the three random trials during the test session. All ET-1–treated marmosets showed a noticeable strong grip to the shaft of the paintbrush when it was presented to the ipsilateral hand. Vehicle-treated marmosets showed a strong noticeable grip of the paintbrush whether it was presented to the ipsilateral or contralateral hand during the test session. A MANOVA showed a significant group by time by side interaction (F1,4 = 128.00, P < 0.005). Post hoc comparisons revealed a significant main effect between the vehicle and ET-1 contralateral side at 24 hours after surgery in terms of an impaired forelimb grip strength displayed by the ET-1–treated animals (P < 0.001; Fig. 9A). Furthermore, post hoc comparisons revealed a significant difference between preoperative and postoperative contralateral forelimb grip strength and between ipsilateral and contralateral forelimb grip strength during the postoperative session in ET-1–treated marmosets (all P < 0.01; Fig. 9A). No sex effect was shown, suggesting that there was no difference in forelimb grip strength for males and females, irrespective of treatment group.
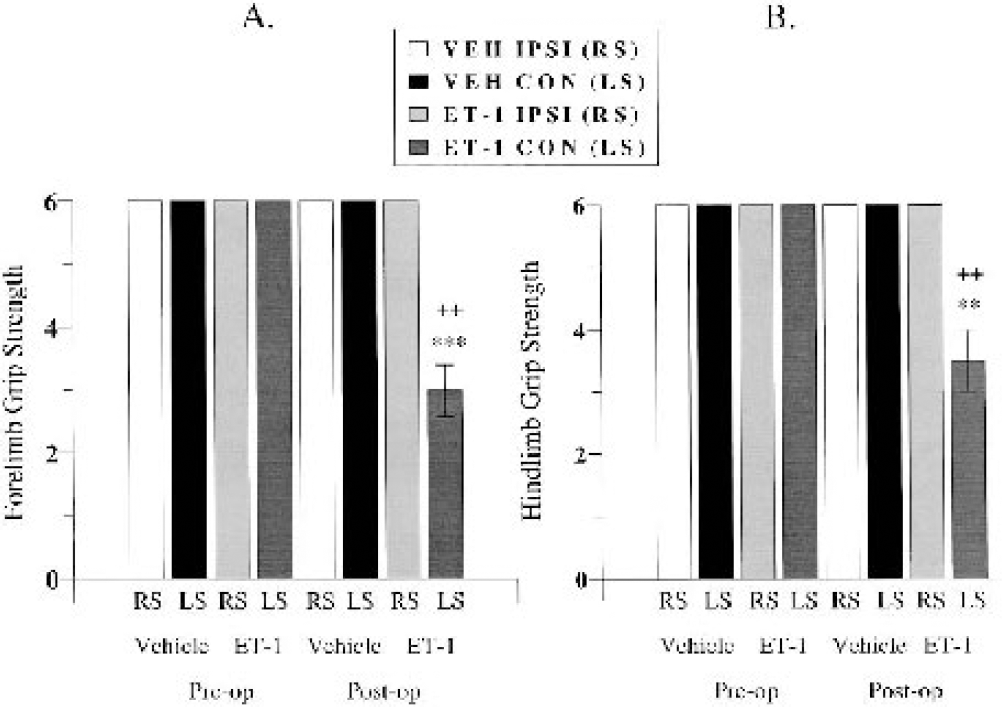
Graded forelimb (
Hindlimb grip strength
During the preoperative session all marmoset showed a clearly noticeable grip to the shaft of the paintbrush with both ipsilateral and contralateral feet. At 24 hours after surgery, all of ET-1–treated marmosets showed either a modest grip lacking strength or noticeable strong grip with the contralateral foot. The grip strength in the contralateral foot of ET-1 marmosets was clearly better than that shown by the contralateral hand. All ET-1–treated marmosets showed a noticeable strong grip to the shaft of the paintbrush when it was presented to the ipsilateral foot. Vehicle-treated marmosets showed a strong noticeable grip of the paintbrush whether it presented to the ipsilateral or contralateral foot during the test session. A MANOVA showed a significant group by time by side interaction (F1,4 = 25.00, P = 0.007). Post hoc comparisons revealed a significant main effect between the groups on the contralateral side at 24 hours after surgery in terms of an impaired hindlimb grip strength displayed by the ET-1–treated animals (P < 0.01; Fig. 9B). Furthermore, post hoc comparisons revealed significant differences between preoperative and postoperative contralateral hindlimb grip strength and between postoperative ipsilateral and contralateral hindlimb grip strength in ET-1–treated marmosets (all P < 0.01; Fig. 9B). No sex effect was shown, suggesting that there was no difference in hindlimb grip strength for males and females, irrespective of treatment group.
Forelimb tactile reflex
During the preoperative session, all marmosets showed a clearly noticeable reflex to the hairs of the paintbrush with both ipsilateral and contralateral forelimbs. At 24 hours after surgery, half (two out of four) of the ET-1–treated marmosets showed either a modest tactile reflex or no response at all with the contralateral forearm, whereas the other half showed either a modest or noticeable response with the contralateral forearm to the hairs of the paintbrush. All ET-1–treated marmosets showed a noticeable tactile reflex when the hairs of the paintbrush gently stroked the ipsilateral forearm in a random fashion. Vehicle-treated marmosets showed a strong noticeable tactile reflex when either the ipsilateral or contralateral forearm was stimulated during the test session. A MANOVA showed a significant group by time by side interaction (F1,4 = 98.00, P = 0.0006). Post hoc comparisons revealed a significant main effect of group on the contralateral side at 24 hours after surgery, in that an impaired reflex was displayed by the ET-1–treated animals (P < 0.001; Fig. 10A). Furthermore, post hoc comparisons revealed a significant difference between preoperative and postoperative contralateral forelimb reflex and between postoperative ipsilateral and contralateral forelimb reflex in ET-1–treated marmosets (all P < 0.01; Fig 10A). No sex effect was shown, suggesting that there was no difference in forelimb reflex for males and females, irrespective of treatment group.
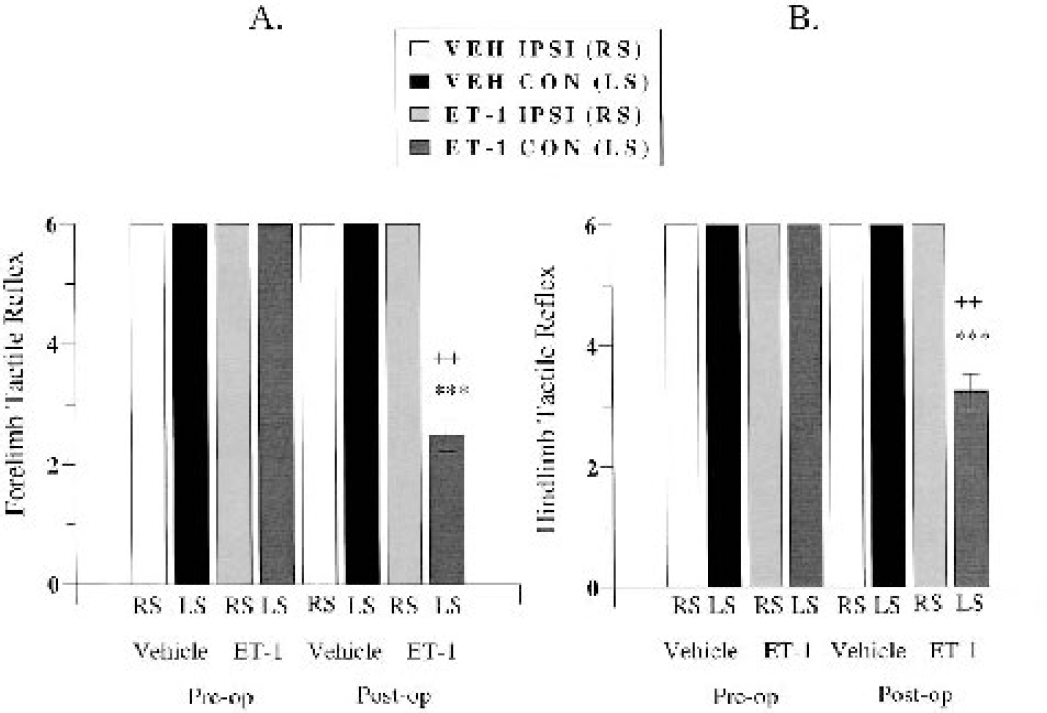
Graded forelimb (
Hindlimb tactile reflex
During the preoperative session, all marmosets showed a clearly noticeable reflex to the hairs of the paintbrush with both ipsilateral and contralateral hindlimbs. At 24 hours after surgery, the majority (three out of four) of ET-1–treated marmosets showed a modest tactile reflex with the contralateral hindlimb, whereas one ET-1–treated marmoset showed either a modest or noticeable response with the contralateral hindleg to the hairs of the paintbrush. All ET-1–treated marmosets showed a noticeable tactile reflex when the hairs of the paintbrush gently stroked the ipsilateral hindlimb in a random fashion. Vehicle-treated marmosets showed a strong noticeable tactile reflex when either the ipsilateral or contralateral hindleg was stimulated during the test session. A MANOVA showed a significant group by time by side interaction (F1,4 = 98.00, P = 0.0006). Post hoc comparisons revealed a significant main effect between groups on the contralateral side at 24 hours after surgery in terms of an impaired reflex displayed by the ET-1–treated animals (P < 0.001; Fig. 10B). Furthermore, post hoc comparisons revealed a significant difference between preoperative and postoperative contralateral hindlimb reflex and between postoperative ipsilateral and contralateral hindlimb reflex in ET-1–treated marmosets (all P < 0.01; Fig. 10B). No sex effect was shown, suggesting that there was no difference in hindlimb reflex for males and females, irrespective of treatment group.
Ear stimulation
During the preoperative session, all marmosets found stroking their ear tufts quite provocative, responding by biting the brush when presented to either the ipsilateral or contralateral ear. At 24 hours after surgery, all ET-1–treated marmosets elicited either a mild almost negligible response or no observable response at all to the stimulus presented to the contralateral ear. In contrast, the ET-1–treated marmosets showed either an aggressive response (biting the brush) or a mild response when the ipsilateral ear tuft was stroked. However, vehicle-treated marmosets did show habituation to this test in the postoperative testing session. The majority of vehicle-treated marmosets showed either a mild or marked response to the stimulus, regardless of whether it was presented to the ipsilateral or contralateral ear. None of the vehicle-treated marmosets failed to show a response, indicating that this test highlights the contralateral neglect in tactile response of ET-1–treated marmosets. A MANOVA showed a significant group by time by side interaction (F1,4 = 128.00, P = 0.0003). Post hoc comparisons revealed a significant main effect between groups on the contralateral side at 24 hours after surgery, in that the ET-1–treated animals displayed an impaired reflex (P < 0.001; Fig. 11). Furthermore, post hoc comparisons revealed significant differences between preoperative and postoperative contralateral ear reflex and between postoperative ipsilateral and contralateral ear reflex in ET-1–treated marmosets (all P < 0.001; Fig. 11). No sex effect was shown, suggesting that there was no difference in ear reflex for males and females, irrespective of treatment group.
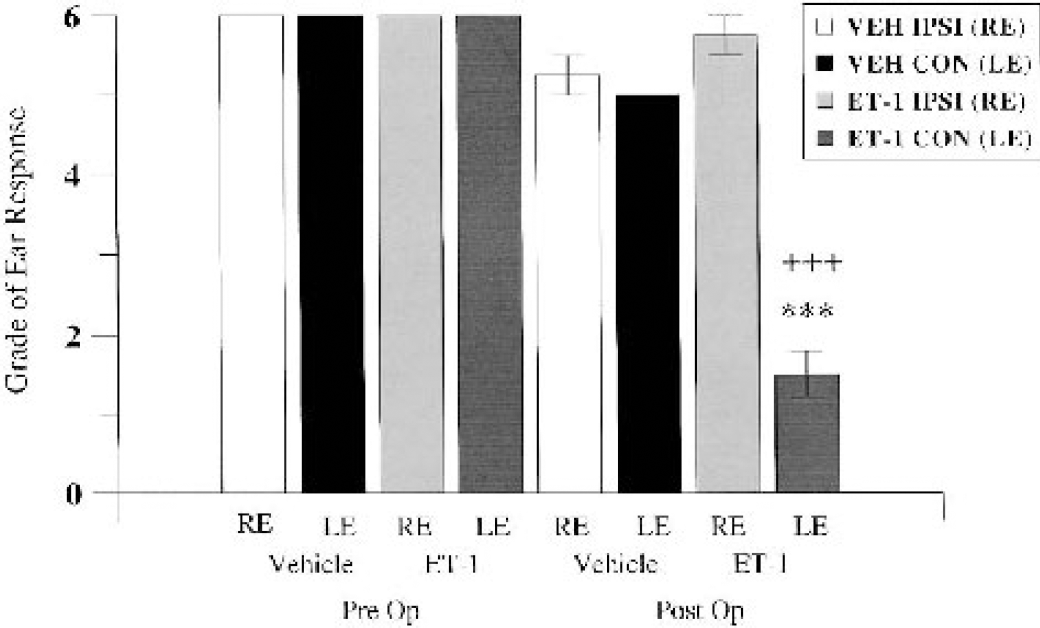
Ear stimulation. Graded ear response to tactile stimulation by vehicle-treated (VEH) and ET-1–treated marmosets at preoperative baseline and 24 hours after surgery (n = 4 per group). RE and LE refer to right (ipsilateral) and left (contralateral) ears. ***P < 0.001 between vehicle and ET-1 contralateral ear response at 24 hours; +++P < 0.001 between ipsilateral right and contralateral left ear response for the ET-1 group at 24 hours. Pre Op, preoperative; Post Op, postoperative.
Neuropathology
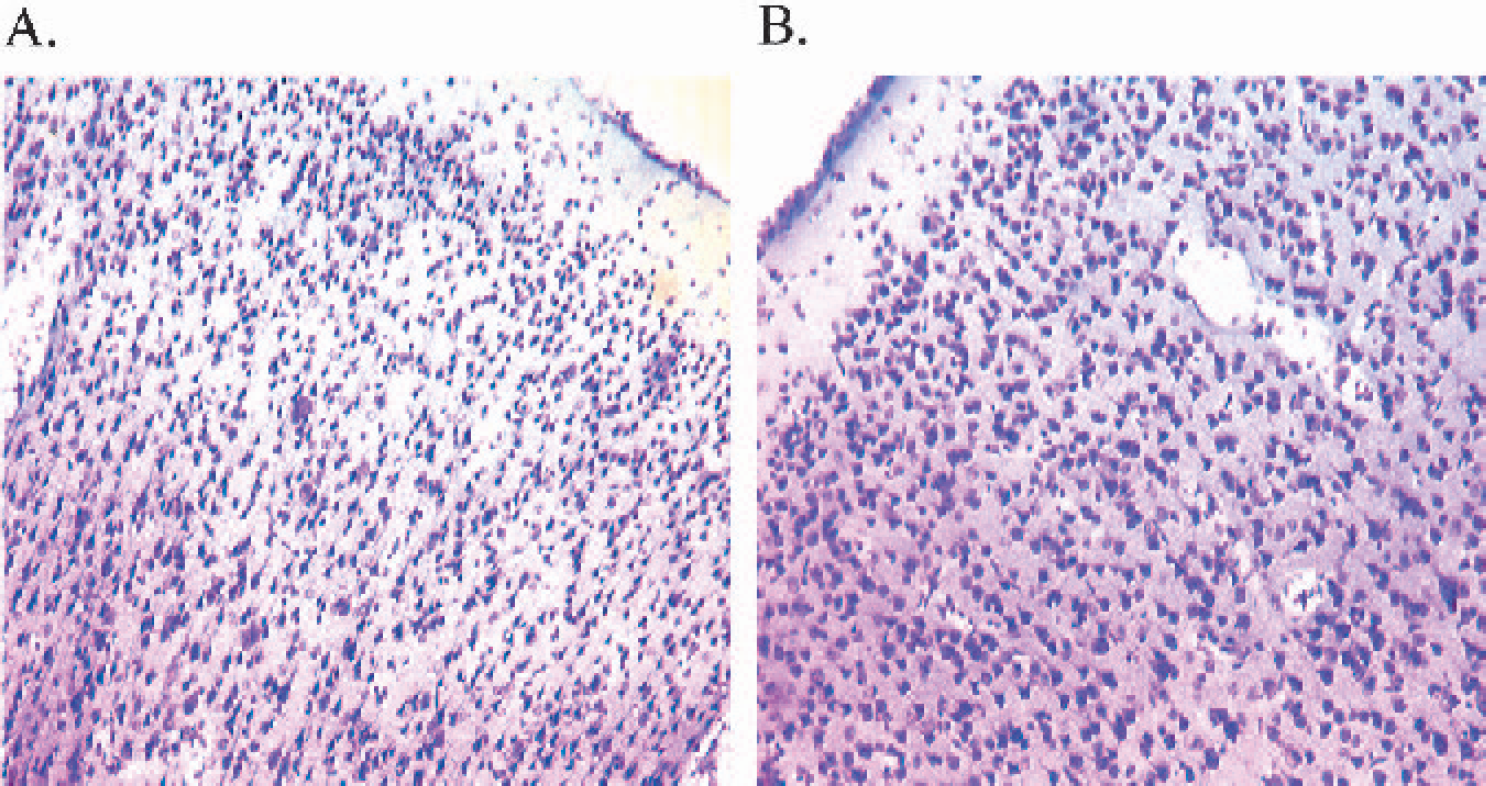
At 24 hours after surgery, all ET-1–treated marmosets had sustained substantial unilateral damage to the right hemisphere cortical areas surrounding the lateral sulcus, where ET-1 was topically applied to the MCA (Figs. 12A and 12C). Both frontoparietal and temporoparietal cortices, as well as some external capsule and lateral putamen damage, were visible in all ET-1–treated marmosets. No caudate damage was shown in all four marmosets, suggesting that ET-1 had not occluded the M1 segment of the MCA. Pockets or “hot-spots” of necrosis were shown to extend back to more posterior regions of the frontal and temporal cortex, which were surrounded by neuronal cell swelling (Fig 13.A). Clearly, ET-1 reproducibly affected the M2 vascular territory that extended back to more posterior regions supplied by the perfusion territory of the distal segments (M3-M4) of the MCA. Cresyl violet and Luxol fast blue staining consistently revealed extensive disorganization within cortical cell layers of ET-1–treated animals, relative to the contralateral side (Figs. 12 and 13) and vehicle-treated control animals. Furthermore, high-power microscopy revealed neuronal cell swelling, pyknosis, and a vacuolated neuropil within and around the ipsilateral lateral sulcus (Figs. 12A and 12C) of ET-1–treated marmosets that extended into the ipsilateral frontoparietal cortex(Fig. 13A) and temporoparietal cortex.
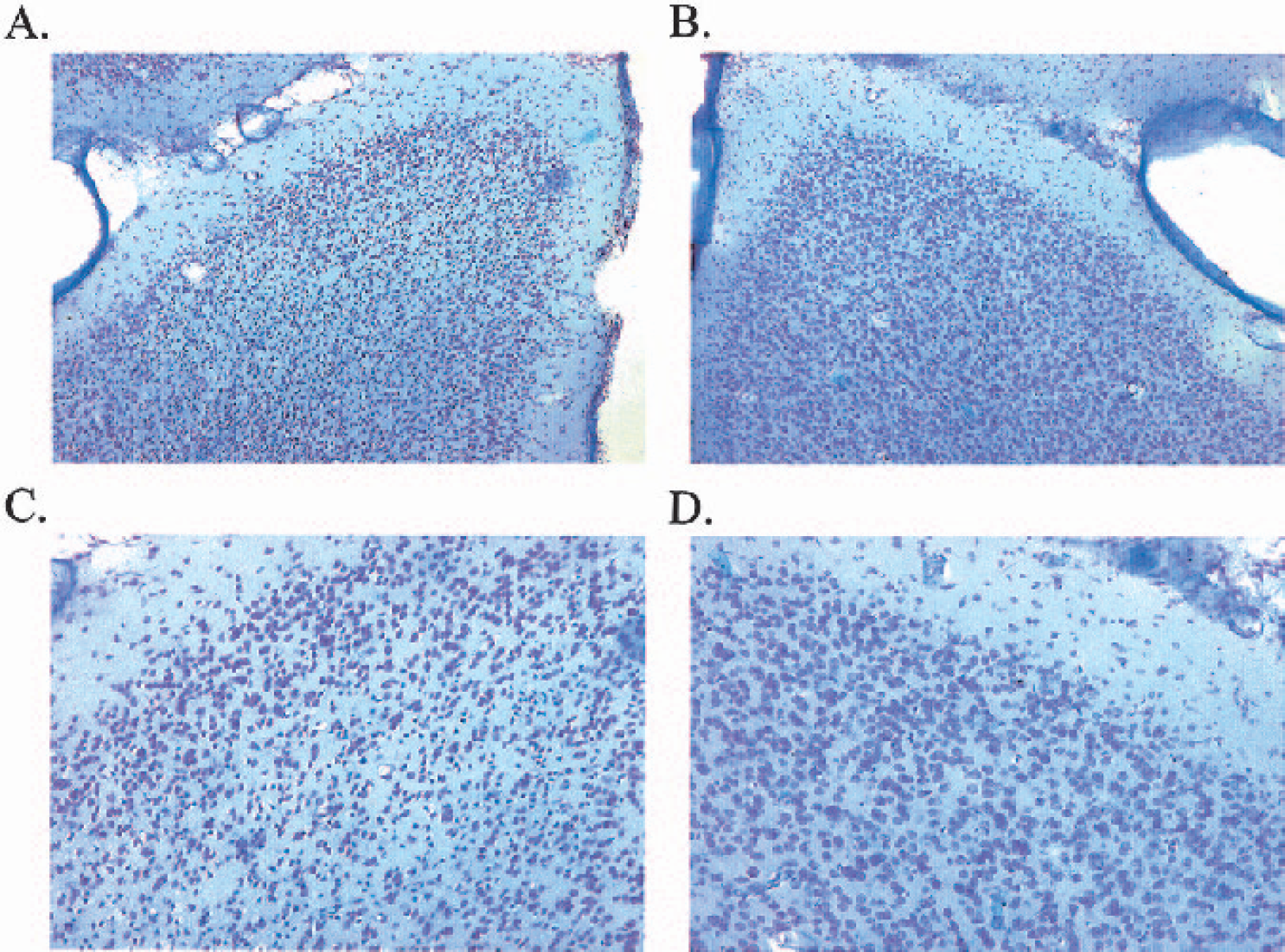
High-power light microscopy photomicrographs showing a typical lesion around the ipsilateral lateral sulcus (
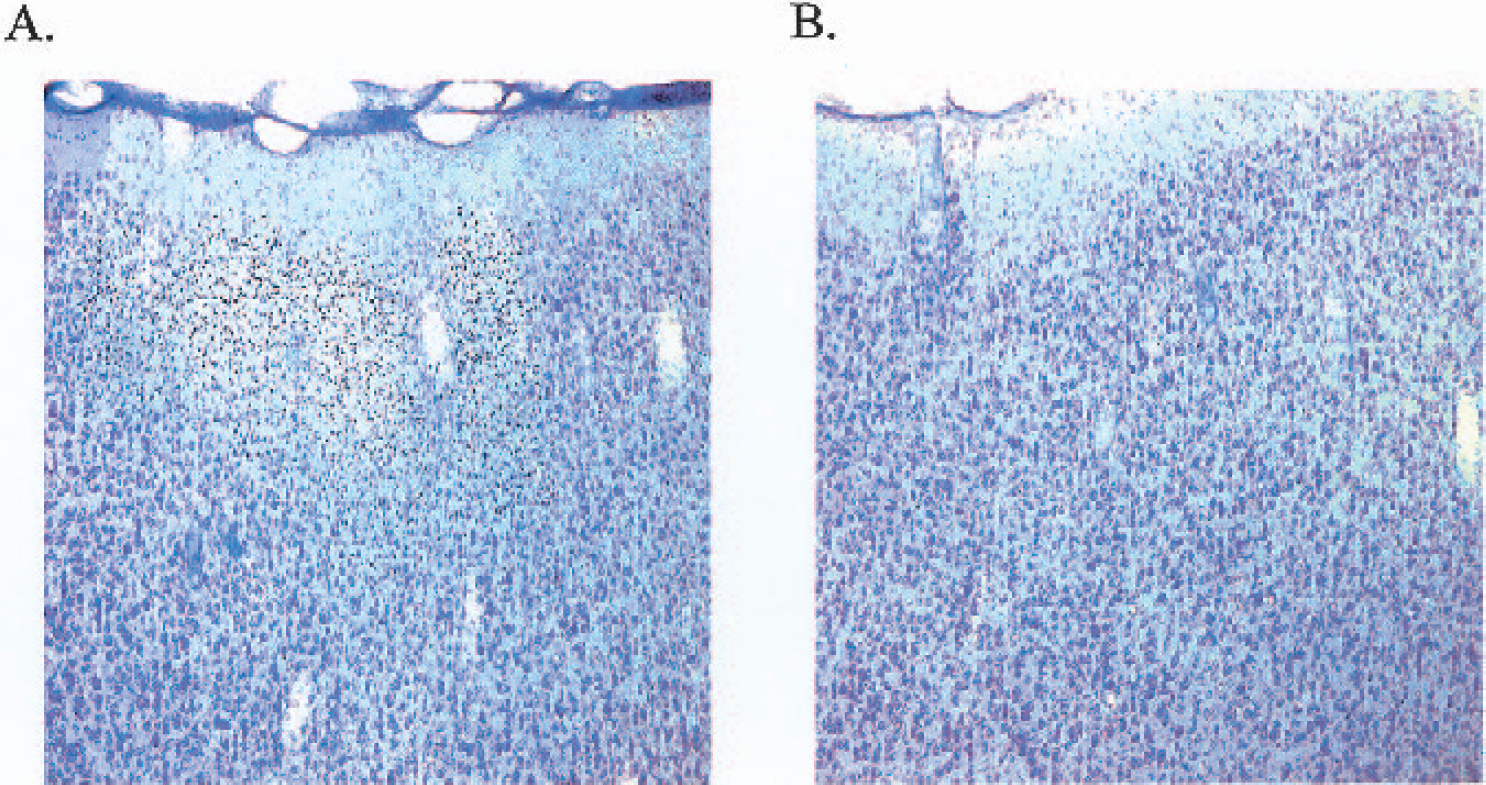
High-power light microscopy photomicrographs (×10 magnification) of the ipsilateral frontoparietal cortex (
Corrected lesion volume
Mean ± SD corrected lesion volume for ET-1–treated marmosets was 329.40 ± 41.83 mm3, whereas vehicle-treated marmosets had damage of 2.96 ± 1.12 mm3 (see Fig. 14 for individual data points). An independent t-test revealed a highly significant difference between ET-1 and vehicle treatment applied to the MCA in terms of corrected lesion volume at 24 hours (t6 = 15.60, P < 0.001, two-tailed). Therefore, unlike the vehicle-control application, the optimal dose of ET-1 was shown to consistently (n = 4) induce large unilateral lateral putamen and cortical (frontoparietal and temporoparietal) damage.
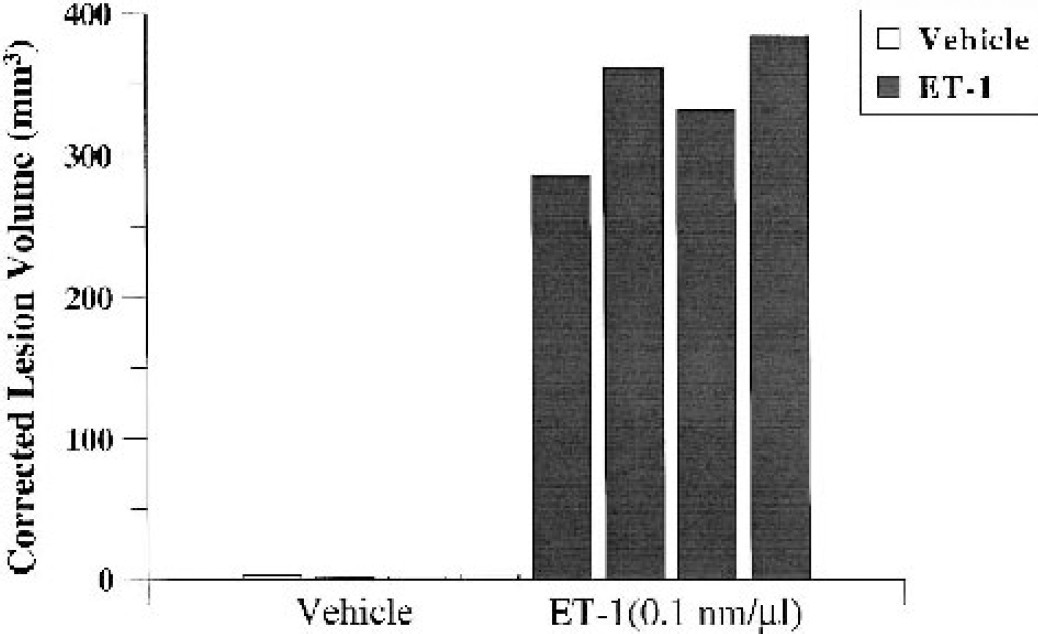
Corrected lesion volume (in cubic millimeters) for marmosets treated with ET-1 (0.1 nmol/μL) or vehicle (n = 4 per group) at 24 hours after application (recovery experiment). Individual data points for each marmoset are plotted. Notice the minimal lesions in vehicle-treated marmosets relative to the consistent lesion volumes in ET-1–treated marmosets.
Corrected total lesion volume as a percentage of ipsilateral and contralateral hemispheric volume
Mean ± SD corrected lesion volume as a percentage of the ipsilateral hemisphere for ET-1–treated marmosets was 19.86 ± 0.55, whereas vehicle-treated marmosets had minimal damage (0.18 ± 0.08). Corrected lesion volume as a percentage of the contralateral hemisphere for ET-1–treated marmosets was 20.13 ± 0.74, whereas vehicle-treated marmosets sustained minimal damage (0.18 ± 0.08).
Statistically significant differences in total lesion volume as percentage of ipsilateral (t6 = 71.3, P < 0.001) and contralateral (t6 = 53.5, P < 0.001) hemispheric volume were found between ET-1–treated animals and vehicle-control treated animals at 24 hours after surgery. Essentially, on average 20% of the hemisphere was damaged after ET-1 application to the M2 portion of the MCA, whereas an average of 0.2% of the hemisphere was damaged after vehicle-control application.
Swelling
Mean ± SD percentage hemispheric swelling for ET-1–treated marmosets was 1.40 ± 3.6, relative to vehicle-treated marmosets (1.58 ± 2.14). No statistically significant differences in percent of hemispheric swelling at 24 hours were found between ET-1–treated and vehicle-treated marmosets (t6 = −0.08, P = 0.935771).
Longitudinal extent of lesion
Mean ± SD longitudinal extent of the lesion for ET-1–treated marmosets was 34.25 ± 6.34, relative to vehicle-treated marmosets (8.00 ± 1.63). A statistically significant difference in the lesion extent was shown between ET-1–treated and vehicle-control treated marmosets at 24 hours (t6 = 8.01, P < 0.001). Clearly, ET-1 induced damage in significantly more histological sections that encompassed the M2-M4 vascular territory than did vehicle.
Correlational analyses
There were strong negative correlations between corrected lesion volume and postoperative contralateral forelimb grip strength (r = −0.963, P < 0.05), longitudinal extent of lesion and postoperative contralateral forelimb grip strength (r = −0.965, P < 0.05), corrected lesion volume as a percentage of the contralateral hemisphere and postoperative contralateral hindlimb grip strength (r = −0.966, P < 0.05), postoperative contralateral hindlimb tactile reflex (r = −0.966, P < 0.05), and postoperative contralateral forelimb retrieval of food rewards (r = −0.966, P < 0.05) in ET-1–treated marmosets. These results imply that MCA territory lesions were strongly associated with poor contralateral forelimb and hindlimb function in ET-1–treated marmosets. No statistically significant relationships were obtained for vehicle-treated animals when attempting to correlate histopathological and functional parameters at 24 hours after surgery (r < 0.50, P > 0.05).
DISCUSSION
These experiments show that the well-controlled topical application of ET-1 to the M2 portion of the MCA in the common marmoset can induce consistent reduction of MCA caliber suggestive of occlusion, followed by a gradual reperfusion, and provide a new reproducible model of focal stroke induction in a nonhuman primate. Consistent contralateral deficits on a number behavioral tests were shown, which were attributable to M2 territory infarction and which complement those seen with the pMCAO approach in the marmoset (Marshall and Ridley 1996; Marshall et al., 1999). Marmosets recovered well from surgery and were in sufficiently good health to be moved from an incubator to a postoperative cage within 16 hours after surgery. General activity measures in ET-1-treated animals at 24 hours after surgery were not significantly different from their respective preoperative baseline measures or from those of vehicle-treated animals tested at 24 hours. Clearly, the surgical procedure had no deleterious effect on general mobility or feeding. Self-care was resumed promptly after surgery, suggesting that marmosets can be rehoused into the colony room to resume and maintain socialization for long-term assessment. However, selective contralateral deficits were sensitively identified by the battery of behavioral tests used, which provides significant progress towards developing a long-term functional model of ischemic stroke with reperfusion in a nonhuman primate.
Cerebral vascular anatomy
The Araldite corrosion cast and ink perfusion experiments show that the vascular anatomy of the arterial circle of common marmoset can be clearly delineated and are similar to those previously described for the yellow-handed marmoset (Bugge, 1963, 1974, 1978, 1980) and human (Lee et al., 1995; Salamon et al., 1985). The casts showed the ICA to be highly tortuous in its passage, forming the well-known S-shaped bend or carotid siphon before terminating into the ACA and MCA. The opthalmic artery and posterior communicating artery were also visualized in anatomical locations similar to those reported for the yellow-handed marmoset (Bugge, 1978, 1980) and humans (Lee et al., 1995; Salamon et al., 1985). The ACA and MCA formed the terminal branches of the ICA in the common marmoset; the MCA was the largest branch of the ICA, consistent with descriptions derived from observations in humans (Lee et al., 1995; Salamon et al., 1985). Measurements taken from Araldite casts and ink perfusion specimens for ICA and MCA diameters were equivalent, suggesting that both techniques provided complementary information concerning vessel caliber and anatomical distribution. The twisting passage of the ICA is well conserved from nonhuman primates to humans, in contrast to rodents, which in evolutionary terms relates to the enlargement and forward movement of the ICA in relation to the tympanic cavity. The extensive kinking of the ICA before it terminates into the ACA and MCA bifurcation precludes the introduction of an intraluminal thread at the origin of the carotid bifurcation. Thus, the alternative approach of topically applying ET-1 onto the M2 portion of the MCA was used to induce MCAO and subsequent reperfusion.
ET-1–induced MCAO (nonrecovery)
ET-1 is a potent vasoconstrictor peptide that has been shown to override cerebral autoregulatory mechanisms to constrict a number of cerebral vessels in vivo and reduce CBF below the ischemic threshold to induce infarction. ET-1 is believed to act at the ETA/B receptor on the adventitial surface, and studies have found that ET-1 injected intraluminally to cerebral arteries induces minor changes in vessel caliber, unlike abluminal applications (Mima et al., 1989; Ogura et al., 1991). The abluminal application of ET-1 to the exposed rat MCA induces rapid and severe dose-dependent reductions in cerebral blood flow (70–96%) with a prolonged duration of action that is related to ischemic brain damage (Macrae et al., 1993). Gradual recanalization of the MCA has been reported in the rat ET-1 model, although the degree of reperfusion is incomplete on the basis of evidence from laser-Doppler flowmetry and assessments of ipsilateral hemispheric swelling during the first 4 hours. Furthermore, as brain swelling resolved the degree of reperfusion improved, suggesting an inverse relationship between edema and recanalization of the MCA territory (Gartshore et al., 1997). These experimental findings are in agreement with the results from our pilot ET-1 doseranging study in the marmoset. After abluminal application of ET-1 to the M2 portion of the MCA, a rapid and marked dose-dependent reduction in MCA caliber size was observed (60–80%), as signified by constriction of the MCA and arterial branches and concomitant blanching of the surrounding cortical parenchyma, unlike vehicle application. The highest dose of ET-1 induced sustained vasoconstriction on the basis of vessel caliber measurements, although gradual recanalization was observed with time after application. Caution must be exercised on the basis of these quantitative vessel caliber estimates because only one marmoset per group was assessed. However, qualitative observations from the remaining animals within each of the dose groups indicated that rapid cessation of blood flow and blanching of the cortex followed by a gradual reperfusion induced by ET-1 was a true phenomenon. In some instances we observed relative brain swelling around the lateral sulcus after ET-1, although this observation tended to resolve by end-point (as confirmed by percentage hemispheric swelling estimates). We administered dexamethasone before surgery in each of the marmosets to obviate problems associated with potential edema, which may well explain why there were no marked differences in brain swelling at end-point.
Histopathological analysis revealed that corrected lesion volume was also generally related to the dose of ET-1 applied to the MCA, corroborating the findings observed in earlier rat ET-1–induced MCAO studies. However, one marmoset sustained a very severe M1 territory infarction that extended more medially, encroaching on the whole of the caudate and putamen (Fig. 5B). Possible reasons for this event are that the MCA was very accessible in this marmoset or that the volume of ET-1 applied may have diffused more readily downwards and inwards to the more proximal portion, inducing M1 infarction. Lesions tended to encompass the M2 territory in the other ET-1–treated marmosets, with the highest dose (2.5 nmol in 25 μL) producing the largest and most reproducible lesions; this is equivalent to the dose used in the rat ET-1 MCAO studies (Gartshore et al., 1997; Macrae et al., 1993). It is conceivable that if a larger volume of ET-1 (at the same concentration) was used, then an M1 infarction may be more consistently produced. On histopathological inspection, the lesions around the lateral sulcus were indicative of “necrosis,” with characteristic hallmarks of neuronal cell swelling, pyknosis, and a vacuolated neuropil. These characteristics were also apparent within the lateral putamen and the frontoparietal and temporoparietal cortices. However, the elements of “selective” necrosis were also observed within the frontoparietal cortex, where lesions had a multiple stellate or punctate appearance, similar to the “fleabitten” type profile reported by Marcoux et al. (1982) and DeGirolami et al. (1984) within the cortex of macaques that underwent temporary MCAO with reperfusion. The grey matter was clearly more vulnerable than the white matter, although the external capsule that surrounded the putamen from the surrounding cortex was mildly affected. Variations in the ET-1 model in the rat have also been shown to provide a reproducible penumbral area of at risk tissue that may be amenable to neuroprotective strategies (Fuxe et al., 1997), suggesting that this novel primate model may be highly relevant in assessing treatments that have unique properties in protecting potentially salvageable tissue.
The technical approach for inducing reproducible ET-1–induced lesions in the common marmoset requires further comment. Although a small craniotomy exposed the M2 portion of the MCA, this major vessel sometimes lay deep within the lateral sulcus and hence careful exposure was required to enable the topical application of ET-1. Alternative methods that have been adopted for ET-1 application in rats, such as stereotaxic application via guide cannula (Sharkey et al., 1993; Sharkey and Butcher, 1995), would not be appropriate in this context because extensive variability in the location and passage of the MCA within the lateral sulcus of the common marmoset was seen. Therefore, it was essential to visualize the M2 portion of the MCA and apply the ET-1 topically to confirm reduction in vessel caliber size and blanching of the surrounding parenchyma, and hence to induce successful MCAO.
ET-1–induced MCAO (recovery)
Using the higher dose of ET-1 selected from the pilot study, consistent contralateral neglect and motor deficits were shown in marmosets within the immediate postoperative period and at 24 hours after ET-1 application, unlike vehicle-treated control marmosets. These behavioral results are in agreement with the initial results reported with M2 territory infarction using the pMCAO approach in the common marmoset (Marshall and Ridley, 1996; Marshall et al., 1999). Although the tests used in this study were not as extensive as the battery of objective tasks adopted by Marshall and Ridley's group, they do provide useful information on the functional status of ET-1–treated marmosets that can relate to the assessments made in pMCAO-treated marmosets. ET-1–induced M2 territory lesions significantly impaired contralateral grip strength and responses to contralateral stimuli up to 24 hours after surgery. Contralateral forelimb retrieval of food rewards was also markedly impaired in ET-1–treated marmosets, which can be directly extrapolated to the sustained impairments with the contralateral hand in pMCAO-treated marmosets assessed on the “Hill” and “Valley” staircase tests (Marshall and Ridley, 1996; Marshall et al., 1999, 2000a, 2001). Moreover, when the rewards were presented within contralateral hemispace, ET-1–treated marmosets failed to respond, although they showed rapid responses with their ipsilateral “good” hand when the rewards were presented in ipsilateral space. These findings relate well to the contralateral spatial neglect shown with the “Valley” staircase, two-tube choice and six-tube search tests used to assess contralateral spatial neglect in pMCAO-treated marmosets (Marshall and Ridley, 1996; Marshall et al., 1999, 2000a, 2001). Furthermore, small unilateral parietal ablations in marmosets, which disrupted the forward flow of information from the striate cortex to the dorsal territory of the MCA, also produced contralateral neglect on the above tasks at 2 and 10 weeks after surgery (Marshall et al., 2002). Clearly, the simple food reward test adopted in this study provides a very suitable primer for subsequent objective assessment with the batteries of tests used by Marshall and Ridley's group to assess long term functional outcome in ET-1–treated marmosets. General mobility and self-care improved within the immediate postoperative phase in ET-1–treated marmosets, in that they did not differ from vehicle-control treated marmosets at 24 hours after surgery. Although contralateral impairments of the forelimb and hindlimb predominated, these deficits did not hinder general wellbeing and mobility. The marmosets spontaneously ate and drank, resumed grooming, and climbed, jumped, and ran along perches around the postoperative cage in a almost normal manner. Furthermore, all ET-1–treated marmosets survived the surgical procedure with no mortality. This information suggests that this surgical model induces a very specific contralateral deficit that does not interfere with general welfare, allowing ET-1 marmosets to be rehoused into the colony room with other marmosets to resume and maintain socialization for long-term assessment. We believe this novel model improves existing primate models of stroke by balancing the ability to assess the efficacy of putative neuroprotective and neurorestorative therapies with significant refinements that minimize functional impairment and surgical intervention.
Histopathological analysis showed that all ET-1–treated marmosets had sustained consistent M2 territory infarction that reproducibly recruited the lateral putamen and frontoparietal and temporoparietal cortices to the lesion. The corrected lesion volume of ET-1–treated marmosets was 329.40 ± 41.83 mm3, which is very similar to the M2 lesion reported for pMCAO-treated marmosets at later time points, represented as cavitation (i.e., 370.8 ± 37.4 mm3 at 10 weeks after surgery; Marshall et al., 1999). However, one must consider that the degree of recanalization afforded in this model may have potentially protected further regions of damage attributable to pMCAO M2 lesions from maturing into infarcted tissue. Again it was clear that the ET-1 lesions consisted of “total” necrosis around the lateral sulcus and more “selective” necrosis within the frontoparietal and temporoparietal cortices, as was shown in the pilot study. Correlational analyses showed strong negative associations between ET-1 lesions, which encompassed the putamen, frontoparietal and temporoparietal cortices, and contralateral motor and sensory/neglect deficits. Recently, unilateral excitotoxic lesions to the putamen of common marmosets were shown to produce very robust contralateral motor impairments on the staircase test for up to 9 months of testing (Kendall et al., 2000). Deficits in skilled contralateral forelimb retrieval and spatial neglect have been shown in pMCAO-treated marmosets, when the lesion severely intrudes into the parietal cortex and the underlying caudate and putamen (Marshall et al., 2000a). Furthermore, our own group has shown that damage to the parietal cortex, visualized by serial MRI, significantly contributes to both contralateral motor impairments and neglect in the transient intraluminal thread MCAO rat model (Virley et al., 2000). It is evident also in stroke patients that damage to the frontal and parietal cortex plays a fundamental role in neglect (Heilman et al., 1985; Husain and Kennard, 1997; Husain et al., 2001) and reaching movements (Kalaska et al., 1997; Mattingley et al., 1998). Clearly, unilateral right-sided damage to the putamen, frontal cortex, and parietal cortex in ET-1–treated marmosets is consistent with evidence from preclinical lesion studies and stroke patients who show impairments in contralateral neglect and fine motor coordination.
In summary, this new focal model of ischemia and reperfusion in the common marmoset produces a robust M2 territory infarction that is correlated with contralateral motor and sensory/neglect impairments. The ET-1 model in the marmoset provides a novel opportunity to assess the efficacy of novel neuroprotective and restorative treatments that have shown efficacy in rodent models of MCAO and that are targeted towards clinical trials. However, one must consider agents that may directly interact with ET receptors, and hence may have the propensity to alter the ET-1–induced mechanism of ischemia and reperfusion. Further work is required to quantitatively assess regional CBF after ET-1 administration in the marmoset and to determine how perfusion status in this model determines irreversible and potentially salvageable ischemic tissue via the utility of serial MRI. In essence, the results from this study suggest that this novel primate model of stroke has the potential to assess long-term functional outcome and to gauge the efficacy of novel therapeutic agents targeted for clinical stroke trials.
Footnotes
Acknowledgements
The authors thank Mrs. L. Ives, Mr. M. Goodey, and Mrs. A. Bryan for technical assistance, Dr. P. Woodhams (Neurology and GI CEDD, GlaxoSmithKline Pharmaceuticals) for histopathological assistance, and J.W.B. Marshall and R.M. Ridley for kind advice regarding techniques and aftercare of marmosets at the start of this project.
