Abstract
The therapeutical potential of transplantation of undifferentiated and predifferentiated murine embryonic stem cells for the regeneration of the injured brain was investigated in two rodent stroke models. Undifferentiated embryonic stem cells xenotransplanted into the rat brain at the hemisphere opposite to the ischemic injury migrated along the corpus callosum towards the damaged tissue and differentiated into neurons in the border zone of the lesion. In the homologous mouse brain, the same murine embryonic stem cells did not migrate, but produced highly malignant teratocarcinomas at the site of implantation, independent of whether they were predifferentiated in vitro to neural progenitor cells. The authors demonstrated a hitherto unrecognized inverse outcome after xenotransplantation and homologous transplantation of embryonic stem cells, which raises concerns about safety provisions when the therapeutical potential of human embryonic stem cells is tested in preclinical animal models.
Keywords
Embryonic stem (ES) cells as well as adult stem cells may provide cures for a variety of diseases (Arvidsson et al., 2002; Björklund and Lindvall 2000; Björklund et al., 2002; Brüstle et al., 1999; Gage et al., 1995; Isacson et al., 1995; Kim et al., 2002; McKay 2000; Reubinoff et al., 2001; Savitz et al., 2002; Zhang et al., 2001). It has been shown that transplantation of undifferentiated ES cells into the intact animal produces teratomas or even highly malignant teratocarcinomas (Reubinoff et al., 2000; Thomson et al., 1998), but the tumorigenic potential of ES cells seems to be greatly reduced when cells are predifferentiated in vitro before implantation. Successful examples of this approach include intracerebral implantations of ES cell–derived neural, neuronal, or glial progenitor cells, all of which did not produce brain tumors (Arnhold et al., 2000; Brüstle et al., 1999; Kim et al., 2002; Reubinoff et al., 2001; Zhang et al., 2001). Obviously, in vitro differentiation by currently applied protocols does not fully prevent the persistence of some undifferentiated cells, but this fraction does not seem to build up the oncogenic potential required for the induction of tumor growth (Reubinoff et al., 2001; Zhang et al., 2001).
In a recent magnetic resonance imaging investigation on stem cell migration in an experimental stroke model of rat, we made the surprising observation that transplantation of murine ES cells did not produce tumors even in the absence of any predifferentiation (Hoehn et al., 2002). Instead, ES cells migrated across the midline towards the periinfarct surrounding, and on the way to the lesion began to differentiate into neural precursor cells. We therefore wondered whether the absence of tumorigenesis in this model was unrelated to the state of ES differentiation and reflected a tumor-suppressive effect of the xenologous host tissue. To test this hypothesis, we compared the outcome of murine ES transplantation in the xenologous rat with that in a homologous mouse stroke model.
MATERIALS AND METHODS
Cell cultures
Murine ES cells of the D3 cell line, stably transfected with the pCX-(β-act)-EGFP expression vector (Arnhold et al., 2000), were cultivated in Dulbecco modified Eagle medium (DMEM) containing 15% fetal calf serum (FCS), nonessential amino acids (stock solution 1:100), penicillin-streptomycin (stock solution 1:100), 50-μmol/L β-mercaptoethanol, and 100-nmol/L leukemia-inhibiting factor (Williams et al., 1988). For in vitro predifferentiation of ES cells, hanging drops were established by plating a suspension of 200 ES cells in 20 μL DMEM plus 10% FCS with leukemia-inhibiting factor on the lids of bacterial dishes. After 2-day incubation in 5% CO2 at 37°C, the growing embryoid bodies were washed out with 10 mL Iscove modified DMEM (Gibco, Karlsruhe, Germany) plus 20% FCS and incubated for 2 more days in bacterial dishes. Then, embryoid bodies were plated separately on gelatin-coated 60-mm tissue culture dishes (20–30 embryoid bodies per dish), kept for 1 more day in Iscove modified DMEM plus 20% FCS, and then for 6 days in B1 medium (Gibco DMEM/F12 supplemented with 5-μg/mL insulin, 30-nmol/L sodium selenite, 50-μg/mL transferrin, and 5-μg/mL fibronectin), which was changed every second day. At day 8 after plating, embryoid bodies were dissociated and replated on poly-D-lysin/laminin-coated 60-mm dishes in B2 medium (DMEM/F12 supplemented with 20-nmol/L per mL progesterone, 100-μmol/L putrescine, 1-μg/ml laminin, 25-μg/mL insulin, 50-μg/mL transferrin, and 30-nmol/L sodium selenite) for 4 more days. Epidermal growth factor (20 ng/mL) and basic fibroblast growth factor (10 ng/mL) were added each day after replating.
Western blotting and immunocytochemistry
Undifferentiated or predifferentiated murine ES cells were harvested and boiled in loading buffer containing 20% glycerol, 3% SDS, 3% 2-mercaptoethanol and bromophenol-blue for 10 minutes. Proteins (50 μg/lane) were separated on a 12% SDS-polyacrylamide gel and tank blotted onto a nitrocellulose membrane (Amersham, Braunschweig, Germany). The membranes were incubated at 4°C for about 12 hours with a polyclonal Oct4 antibody (1:500; Santa Cruz Biotechnology, Santa Cruz, U.S.A.), a monoclonal nestin antibody (1:500; Chemicon, Hofheim, Germany), or a polyclonal GFP antibody (1:1000; Santa Cruz Biotechnology). Blots were incubated with horseradish-coupled antirabbit or antimouse secondary antibody (1:2000; Amersham) at room temperature for 1 hour and developed by using the Amersham ECL-system according to the manufacturer's instructions. Immunocytochemistry was performed in undifferentiated or predifferentiated murine ES cells that were air dried and fixed in ethanol/acetone (1:1 vol/vol) on glass slides. Slides were incubated for 24 hours at 4°C with a polyclonal Oct4 antibody (1:100; Santa Cruz Biotechnology) or a monoclonal nestin antibody (1:150; Chemicon) in phosphate-buffered saline (PBS) containing 5% normal goat serum and 0.3% Triton X-100. Thereafter, cells were washed and incubated with a biotinylated goat antirabbit or antimouse antibody (1:150; DAKO Diagnostics, Glostrup, Denmark). After further washing, streptavidin-horseradish peroxidase (VECTASTAIN Elite ABC; Vector Co., Burlingame, CA, U.S.A.) was added and immunoreactivity was visualized via detection of the biotin-streptavidin-peroxidase complex.
Focal brain ischemia
All experiments were carried out in accordance with NIH animal protection guidelines, and were approved by the local governmental authorities. Adult male Wistar rats (body weight, 260–300 g; age, 12–16 weeks) and adult male C57-BL-6 or SV129 mice (body weight, 22–28 g; age, 10–12 weeks) were anesthetized with 1–1.5% halothane in a 7:3 mixture of N2O:O2. In rats, the middle cerebral artery (MCA) was transiently occluded for 1 hour using the intraluminal thread insertion method (Kohno et al., 1995). In mice, the MCA was exposed by a temporal approach and permanently occluded by bipolar coagulation (Backhauss et al., 1992).
Stem cell implantation
In rats, ES cells were grafted 2 weeks after MCA occlusion under light halothane anesthesia into the contralateral cortex at coordinates 0.5 mm anterior from bregma, 3.0 mm lateral, and 2.0 mm below the dorsal surface. In mice, cortical implantation coordinates were 0 mm from bregma, 2 mm lateral and 1 mm below the dorsal surface. Intrastriatal transplantations were also carried out at 0.5 mm anterior, 3 mm lateral, and 5 mm deep in some rats, and at 0 mm anterior, 2 mm lateral, and 2.3 mm deep in the mouse. The immunosuppressive compound cyclosporine A (Novartis, Basel, Switzerland) was applied intraperitoneally immediately before implantation and every other day before animals were killed with a 10-mg/kg dose. Cell suspensions were slowly injected with a Hamilton syringe; the needle was retained in the brain for several minutes to avoid reflux.
At the end of the anticipated survival time, animals were reanesthetized with an overdose of halothane and transcardially perfused with 2% paraformaldehyde in modified PBS (pH 7.4). Brains were removed, postfixed for 1 week in the same solution, embedded in paraffin, and processed for histologic staining and immunohistochemistry.
Immunohistochemistry
Coronal brain sections were air dried on glass slides and fixed in ethanol/acetone (1:1 vol/vol). Slides were incubated for 24 hours at 4°C with a polyclonal GFP antibody (1:100; Santa Cruz Biotechnology), monoclonal NeuN antibody (1:100; Chemicon), polyclonal Oct4 antibody (1:100; Santa Cruz Biotechnology), or monoclonal glial fibrillary acidic protein antibody (1:100; Santa Cruz Biotechnology), in PBS containing 5% normal goat serum and 0.3% Triton X-100. Thereafter, sections were washed and incubated with a biotinylated goat antirabbit or antimouse antibody (1:150; DAKO Diagnostics, Glostrup, Denmark). After washing, streptavidin-horseradish peroxidase (VECTASTAIN Elite ABC; Vector Laboratories, Burlingame, CA, U.S.A.) was added and immunoreactivity was visualized via detection of the biotin-streptavidin-peroxidase complex. A TRITC-labeled second antibody (1:64; Sigma, Taufkirchen, Germany) was used to detect immunofluorescence.
RESULTS AND DISCUSSION
Rats submitted to 1-hour transient MCA occlusion developed well-demarcated infarcts located mainly in the peripheral part of the MCA territory (Fig. 1A). Contralateral implantation of 80,000 undifferentiated murine ES cells stably expressing green fluorescent protein (GFP) under the control of the constitutively active β-actin promoter led to migration and differentiation of the transplanted cells in immunosuppressed rats.
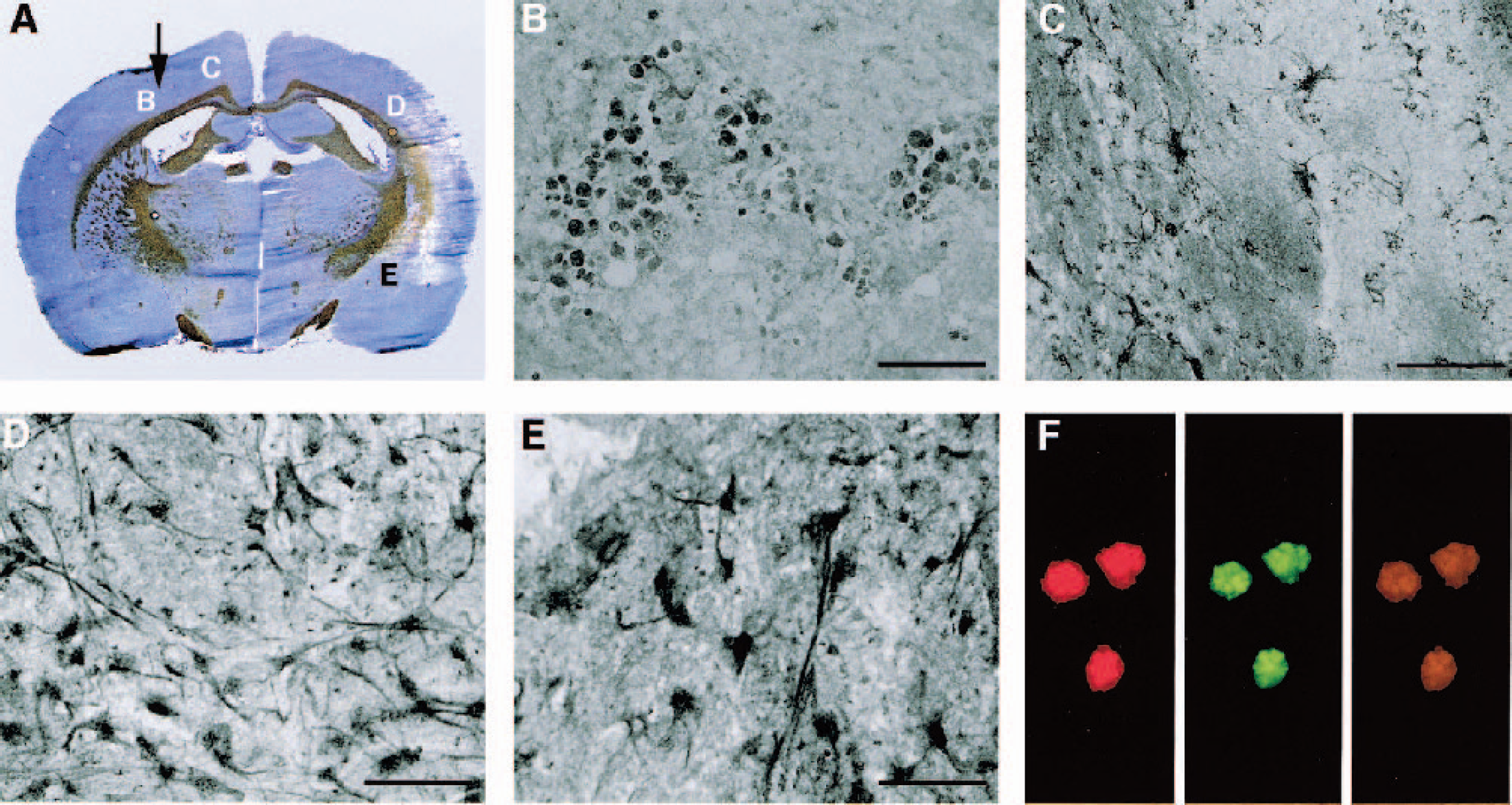
Migration and differentiation of undifferentiated murine ES cells in the ischemia damaged rat brain. (
GFP-positive cells of the globular shape typical of undifferentiated ES cells were detected at the implantation site (Fig. 1B); however, clusters of GFP-expressing cells that were morphologically changing into more differentiated shapes could be seen along the corpus callosum towards the lesion (Fig. 1C). Migration was mainly targeted towards the brain lesion, and only a few GFP-positive cells could be detected beyond this stream in the healthy hemisphere. A massive infiltration of GFP-immunoreactive cells was observed in the border zone of the ischemia-induced lesion (Figs. 1D and 1E). In this area, GFP-expressing cells displayed appearance of neuronal differentiation with axonlike structures as confirmed by colocalization of GFP and the neuronal marker NeuN (Fig. 1F; Hu and Reichardt, 1999). Despite the use of undifferentiated ES cells, tumorigenesis in rats was rare. Small, microscopically visible tumors near the place of implantation were observed in only 2 out of 22 animals (Table 1). Therefore, xenotransplantation of undifferentiated murine ES cells into the ischemia-damaged rat brain results in intense migration and neuronal differentiation, supporting a potential curative effect in this injury model.
Incidence of tumor development after intracerebral implantation of murine embryonic stem (ES) cells and predifferentiated neural precursor (NP) cells

Different results were obtained when the same ES cell line was implanted into the homologous mouse brain. In these animals, brain infarcts were produced by coagulation of the MCA, and 2 weeks later approximately 50,000 undifferentiated murine ES cells expressing GFP were implanted into the cortex of the hemisphere opposite to the lesion. When animals were killed 3 weeks after implantation, 10 out of 11 brains exhibited large, macroscopically visible tumors within the hemisphere of implantation (Fig. 2A; Table 1). Histologically, these tumors exhibited the characteristics of highly malignant teratocarcinomas (Fig. 2B). Diverse structures resembling primitive neural tubes, cartilage, muscle, or mesenchymal tissue were arranged heterogenously (Figs. 2B, 2C, and 2E). Typical and atypical mitoses were abundant, and large parts of the tumors were necrotic, reflecting a mismatch between cell proliferation and angiogenesis (Fig. 2D). Tumor growth was highly invasive, and micrometastases outside the tumor were repeatedly detected. GFP immunohistochemistry revealed the presence of GFP-positive cells within tumor tissue (Fig. 2F), suggesting that they were derived from the implanted ES cells. Positive stainings were also obtained with Oct4, NeuN, and glial fibrillary acidic protein antibodies, demonstrating the presence of both undifferentiated ES cells, and of cells differentiated into neuronal and astroglial lineages (Figs. 2G–2I). GFP immunoreactivity was strictly confined to tumor tissue. No immunoreactivity was detected along the corpus callosum or near the ischemic brain lesion, indicating that after homologous transplantation, murine ES cells generate tumors but do not display migrational or regenerative activity.
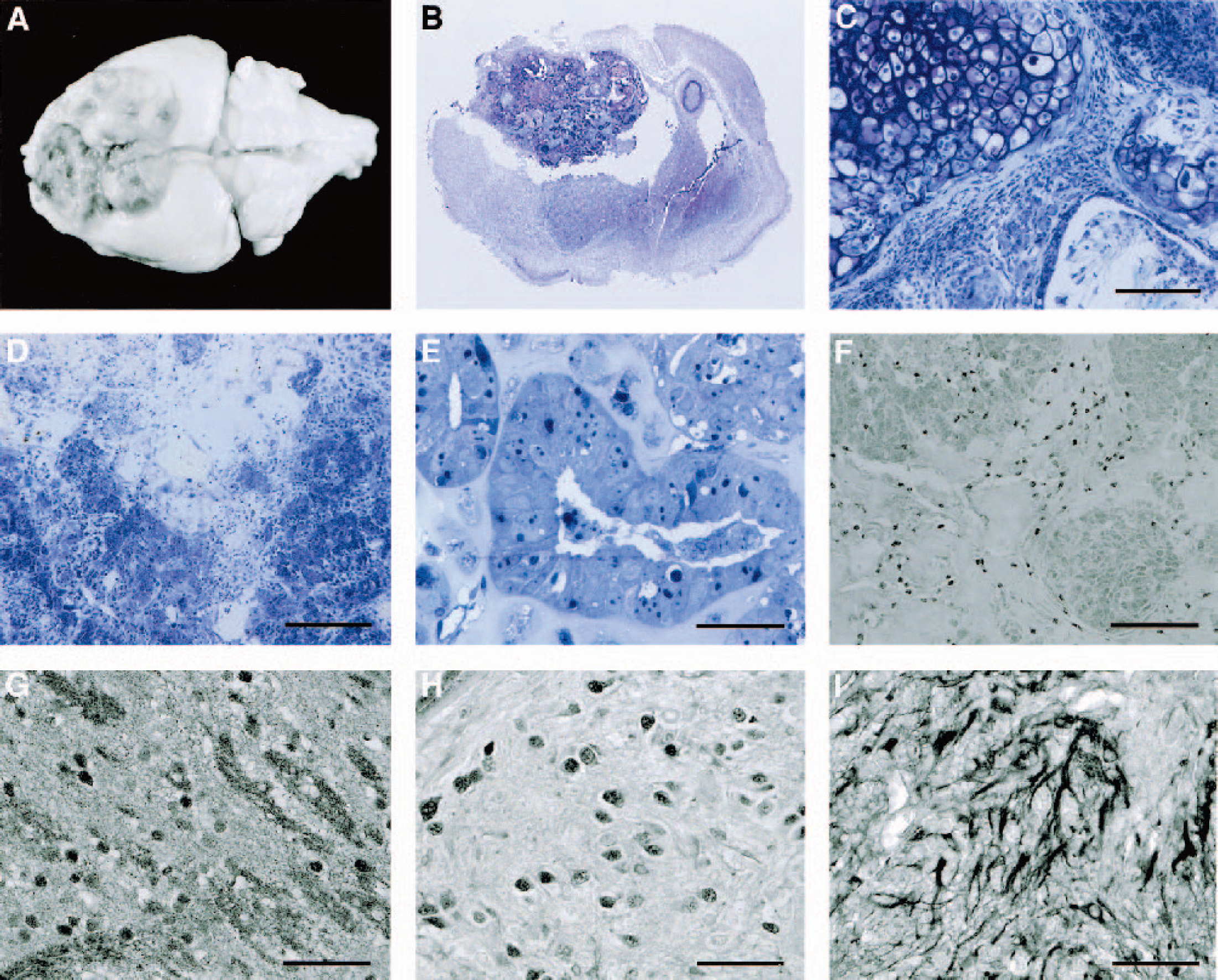
Tumor development after implantation of undifferentiated ES cells in the ischemia-damaged mouse brain. (
Intracerebral implantation of undifferentiated ES cells was also studied in nonischemic healthy animals. In the rat, cortical implantation of approximately 80.000 murine ES cells did not induce tumor formation when investigated histologically 2 weeks later (Table 1). In contrast, implantation of undifferentiated murine ES cells into the healthy mouse brain resulted in tumor formation in most animals (Table 1). Tumorigenesis was induced by as few as 500 cells and was independent of the location of implantation, presence or absence of immunosuppression by cyclosporine A, and gender, age, and strain of the mice (data not shown).
Next, we investigated whether in vitro predifferentiation of ES cells to neural progenitor cells may alter the outcome after homologous transplantation. A protocol based on the formation of embryolike aggregates in hanging-drop preparation was employed that results in a high degree of neural predifferentiation (Arnhold et al., 2000; Bain et al., 1995; Okabe et al., 1996; Sanchez-Pernaute et al., 2001). Western blotting (Fig. 3C) as well as immunocytochemistry (Fig. 3D–3G) of predifferentiated ES cells revealed strong induction of nestin, a well-known marker protein for neural progenitor cells. In these cells Oct4, a transcription factor specifically expressed in undifferentiated ES cells, was below the limit of detection by Western blotting. Immunocytochemistry revealed that a very small number, well below 0.5%, were Oct4 immunoreactive, indicating highly efficient in vitro differentiation. Implantation of approximately 10,000 of such predifferentiated neural progenitor cells into the mouse brain 2 weeks after MCA occlusion resulted in tumor development in 86% of the animals (Table 1). Within the tumor tissue, Oct4 immunohistochemistry showed clusters of undifferentiated cells (Fig. 3B) that far exceeded the number of undifferentiated cells that could be detected before implantation. It is therefore likely that these few cells strongly proliferate in situ before transforming into malignant tumor cells.
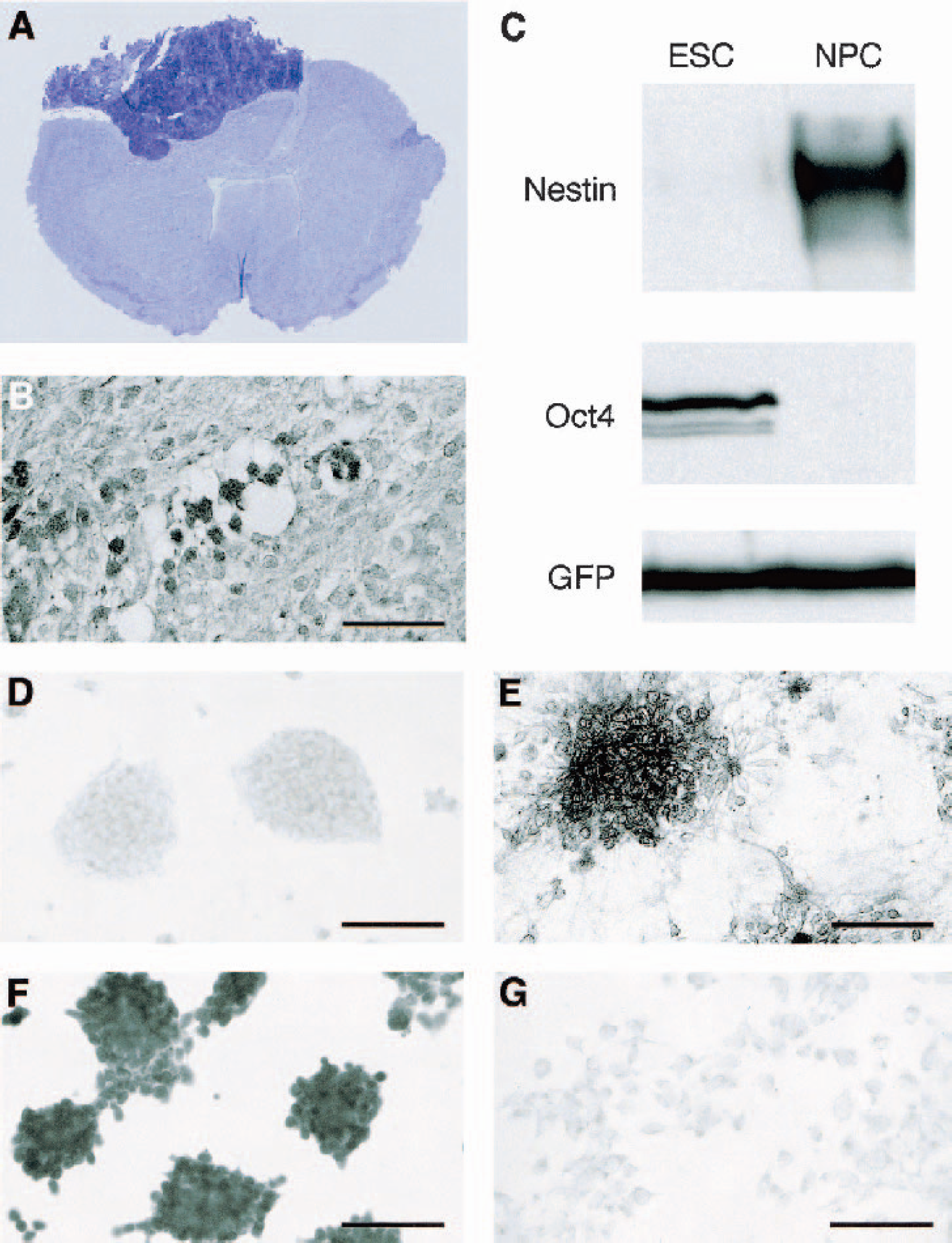
Neural predifferentiation of ES cells does not prevent tumorigenesis in mice. (
The results of this study demonstrate a hitherto unrecognized fundamental difference in the outcome after xenotransplantation and homologous transplantation of murine ES cells, suggesting that the interaction of host-specific factors with ES cells determines whether regeneration or tumorigenesis is promoted. Obviously, this observation raises safety concerns regarding the use of ES cells for clinical purposes. In the past, xenotransplantation of predifferentiated ES cells has been successfully used to enhance regeneration in animal models of various neurologic diseases (Björklund and Lindvall, 2000; Björklund et al., 2002; Brüstle et al., 1999; Kim et al., 2002; McKay 2000). The absence or low incidence of tumor development in these studies was attributed to predifferentiation and not to a potential suppressive effect of the xenologous host tissue. The present investigation clearly shows that this interpretation has to be revised, since it appears that in the homologous system even minor contaminations of undifferentiated ES cells promote tumorigenesis. Obviously, as common differentiation protocols based on the application of differentiation factors cannot exclude the presence of impurities of undifferentiated ES cells, future investigations have to clarify what kind of fate-switching molecules are responsible for the different oncogenic potential of ES cells in different species.
In summary, our data provide evidence for the remarkable migrational and regenerative potency of murine ES cells in the treatment of experimental stroke in rat, but also raise serious safety concerns about the use of such cells in the homologous system. If this conclusion can be generalized, new strategies have to be developed to prevent the risk of tumorigenesis in preclinical studies.
Footnotes
Acknowledgments:
The authors thank U. Beckmann, I. Hölker, P. Janus, and D. Schmiegelt for technical assistance and A. Lorig for the preparation of the manuscript.
