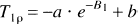Abstract
The impact of brain imaging on the assessment of tissue status is likely to increase with the advent of treatment methods for acute cerebral ischemia. Multimodal magnetic resonance imaging (MRI) demonstrates potential for selecting stroke therapy patients by identifying the presence of acute ischemia, delineating the perfusion defect, and excluding hemorrhage. Yet, the identification of tissue subject to reversible or irreversible ischemia has proven to be difficult. Here, the authors show that T1 relaxation time in the rotating frame, so-called T1ρ, serves as a sensitive MRI indicator of cerebral ischemia in the rat. The T1ρ prolongs within minutes after a drop in the CBF of less than 22 mL 100 g−1 min−1. Dependence of T1ρ on spin-lock amplitude, termed as T1ρ dispersion, increases by approximately 20% on middle cerebral artery (MCA) occlusion, comparable with the magnitude of diffusion reduction. The T1ρ dispersion change dynamically increases to be 38% ± 10% by the first 60 minutes of ischemia in the brain region destined to develop infarction. Following reperfusion after 45 minutes of MCA occlusion, the tissue with elevated T1ρ dispersion (yet normal diffusion) develops severe histologically verified neuronal damage; thus, the former parameter unveils an irreversible condition earlier than currently available MRI methods. The T1ρ dispersion as a novel MRI index of cerebral ischemia may be useful in determination of the therapeutic window for acute ischemic stroke.
There is a growing need to develop imaging methods capable of identifying the pathophysiologic status of brain tissue as early as possible after the onset of ischemia, since ischemic stroke currently is subject to treatment with thrombolytic therapy (The National Institute of Neurological Disorder and Stroke rt-PA Study Group, 1995; Hacke et al., 1998). Before applying any therapeutic interventions, imaging data are needed to exclude contraindications for given treatments, but it would be equally important to demonstrate the presence of salvageable regions within the flow-compromised tissue (Heiss et al., 1999). Whereas long-term consequences of brain ischemia (that is, infarction) can be revealed either with computed tomography or magnetic resonance imaging (MRI), the task of unambiguously identifying tissue at risk of infarction remains an elusive goal for imaging modalities. Despite limitations such as decision-making from single time point data, imaging methods are expected to have a tremendous impact on management of patients with acute stroke in the future.
The diffusion coefficient of water extensively drops within minutes from the collapse of CBF (Moseley et al., 1990; Minematsu et al., 1992; Sotak and Li, 1992; Davis et al., 1994) below the levels of 20 mL 100 g−1 min−1 (Busza et al., 1992; Kohno et al., 1995; Gröhn et al., 2000). As a result of extensive experimental stroke studies, diffusion MRI is a standard procedure in clinical stroke diagnosis (Warach et al., 1995; Welch et al., 1995; Marks et al., 1996). Regarding the predictive value of diffusion MRI in the clinical setting, reduced diffusion in most stroke patients indicates irreversible tissue damage (Baird and Warach, 1998). Perfusion imaging with bolus tracking MRI has been successfully used to reveal hemodynamic defects in acute brain ischemia (Sorensen et al., 1996). These studies show that the area with hindered perfusion is larger than that displaying reduced diffusion in as many as in 70% of cases of acute stroke (Sorensen et al., 1996; Baird and Warach, 1998; Albers, 1999). The condition, termed as perfusion/diffusion mismatch, appears to predict stroke lesion enlargement during the subsequent days. Perfusion/diffusion mismatch could be an index for salvageable tissue, but more work is required to substantiate this hypothesis.
It is commonly accepted that T1 and T2 MRI contrasts can be used only for identification of the irreversible ischemia. However, as previously pointed out, several physiologic, physical, and methodologic factors influence tissue T1 and T2; thus, interpretation of cerebral status from these relaxation times may not be so straightforward in acute ischemia (Gröhn et al., 1998; Calamante et al., 1999; Gröhn et al., 2000). We have previously shown that T1 in the rotating frame—so-called T1ρ as quantified with an on-resonance spin-lock method—is a more sensitive indicator of irreversible ischemia than elevated T2 (Gröhn et al., 1999). Our data suggest that T1ρ apparently probes tissue status without direct contribution from either blood oxygenation level-dependent effects or CBF. Here, we use two rat models of acute cerebral ischemia to study CBF dependency of T1ρ and the interrelationship of early T1ρ change with neuronal density on focal ischemia–reperfusion damage. Our data show that T1ρ increases at similar flow thresholds as diffusion drops. Dependence of T1ρ on the spin-lock field strength—so-called T1ρ dispersion—changes rapidly on ischemia, indicating developing ischemia–reperfusion damage after middle cerebral artery (MCA) occlusion under conditions where diffusion remains ambiguous. The current data suggest that discrimination of irreversible ischemia may become more accurate in multimodal MRI, thus expanding its usefulness in the assessment of acute stroke.
MATERIALS AND METHODS
Animals
Animal experiments were approved by the Ethical Committee of the National Laboratory Animal Center, University of Kuopio. Male Wistar rats (250 to 350 g) were anesthetized with 0.8% to 1.5% halothane in 70/30 N2O/O2 for MCA occlusion experiments or by injecting 1.2 to 1.5 g/kg intraperitoneal urethane for CBF threshold experiments. Arterial blood gases and pH were analyzed (ABL-5, Radiometer, Inc., Copenhagen, Denmark). Blood pressure (CardioCap II, Datex, Helsinki, Finland) and core temperature were monitored online during surgery and MRI, and temperature was maintained at 37° ± 0.5°C by feedback-controlled electrical heating blanket or by circulating thermostat-controlled water in the heating element.
Focal cerebral ischemia
The intraluminal thread model (Longa et al., 1989) was used for MCA occlusion followed by reperfusion. Bifurcation of the internal carotid and external carotid arteries was exposed through a midline cervical incision. A nylon thread (diameter 0.22 mm) was inserted through external carotid artery into the internal carotid artery and advanced until it reached the origin of the MCA, and occlusion was maintained either for 45 (n = 6) or 90 minutes (n = 7). The thread was retracted remotely, with animals remaining in the holder inside of the magnet. Animals exposed to 45 minutes of MCA occlusion were reanesthetized 48 hours after reperfusion with halothane for MRI assessment. Sham-operated animals (n = 6) underwent the same procedure without introducing the occluding thread and served as controls for brain histologic study.
Five animals underwent the surgical procedure for MCA occlusion, but without removal of occluder, and were used together with five unoperated rats in brain temperature experiments. At the end of each experiment, unoperated rats were exposed to mild hypothermia (core temperature reduced to 33° to 34°C) by circulating cool water (10° to 20°C) in the heating element to assess the feasibility of the brain temperature determination and to test the direct influence of hypothermia on cerebral T1ρ dispersion (see later).
Determination of CBF and exposure of rats to graded hypoperfusion
Cerebral blood flow of the rats (n = 25) was quantified using the hydrogen clearance method (Crockard et al., 1980) as described previously (Gröhn et al., 2000). Briefly, two platinum/iridium electrodes were inserted into the cerebral cortex 2 mm caudal to bregma and 3 mm lateral from midline to both sides of the brain. The Ag/AgCl reference electrodes were inserted subcutaneously in the flank. A hydrogen clearance curve was recorded after loading with a mixture of H2/N2/O2 10/65/25 and analyzed using the initial slope procedure (Olesen et al., 1971).
Graded reduction of CBF was obtained as previously described (Gröhn et al., 2000). Briefly, both arterial vertebrales were occluded and either 24 or 48 hours later both common carotid arteries (CCAs) were exposed, and remote-graded arterial occlusions were performed using silicon-coated snares joined to a controllable screw (Allen et al., 1993). These occluding devices made it possible to produce graded, reversible occlusions of the CCAs from outside of the magnet (Gröhn et al., 2000). Two baseline CBF values and two sets of control images were measured, after which CCAs were partially occluded to produce a controlled CBF decrease. The T1ρ was measured between 12 and 15 minutes of CCA occlusions simultaneously with hydrogen clearance curve, after which time the CCAs were released. This protocol was repeated one to five times for each animal with more severe flow reductions until either the CBF threshold of 30 mL 100 g−1 min−1 was reached or CBF did not normalize after release of the CCAs.
Protein phantoms
Bovine serum albumin (BSA, Fraction V, Sigma, St. Louis, MO, U.S.A.) was dissolved in a 100-mmol/L solution of TRIS-buffer (Tris-hydroxymethyl-aminomethane) pH 7.5 and cross-linked with glutaraldehyde as described previously (Koenig et al., 1993), yielding the final BSA concentration of 8% and varying glutaraldehyde concentration between 0.0% and 1.0%.
Magnetic resonance imaging
The MRI experiments were performed using a 4.7-T horizontal magnet, bore size 310 mm (Magnex Scientific, Abingdon, U.K.), equipped with actively shielded field gradients (Magnex) interfaced to a SMIS console (Surrey Medical Imaging Systems, Guildford, U.K.). Round triple-loop (18-mm diameter) and oval dual-loop (27 × 30 mm) transmit/receive surface coils were used in phantom and in vivo measurements, respectively. Rats were fixed into a custom-built head holder from the front teeth and with ear bars and was tilted so that the tilt angle in the axial images corresponded to that used in the stereotactic rat brain atlas (Paxinos and Watson, 1996). This procedure was chosen to match the MRI and histologic data as carefully as possible (Gröhn et al., 1999). Typical water line widths of 35 to 45 Hz were obtained after global shimming.
Axial pilot images were acquired using a multislice spin-echo sequence with a matrix size of 256 × 64, field of view (FOV) of 35 mm, echo time (TE) of 18 milliseconds, repetition time (TR) of 800 milliseconds, and slice thickness (ST) of 1 mm. The horizontal imaging slice was chosen according to the pilot images so that the center of the slice was 4.1 mm from the top of the skull.
Absolute diffusion images were obtained using a spin-echo sequence (TR 1500 milliseconds, TE 52 milliseconds, FOV 40 mm, matrix size 128 × 64, ST 1.5 mm) with four bipolar gradient pairs in each direction. This achieves weighting by the trace of diffusion tensor (Dav = ⅓ Trace) in a single acquisition (Mori and van Zijl, 1995). Data from three acquisitions with different diffusion weighting (b-values of 70, 956, and 1420 s/mm2, respectively) were used to calculate the absolute Dav images.
On-resonance T1ρ measurements were performed using a single adiabatic spin-lock pulse consisting of two 4-millisecond hyperbolic secant adiabatic half-passages interrupted by a variable length spin-lock time ranging between 5 and 50 milliseconds. For in vivo experiments, seven spin-lock fields between 0.17 and 1.70 G were chosen (the B1 amplitude was determined across the imaging slice) in front of a fast low-angle shot imaging scheme acquired in a center-out fashion. Imaging parameters were as follows: TR 4.8 milliseconds, TE 2.3 milliseconds, FOV 40 mm, matrix size 128 × 64, flip angle 18°, ST 1.5 mm, eight averages per image, and 3.5-second delay between scans, resulting in total imaging time of 13 minutes for each T1ρ dispersion data set. The MRI data for T1ρ dispersion were acquired in an interleaved manner with respect to different spin-lock fields to minimize the possible influence of dynamically changing T1ρ relaxation after induction of MCA occlusion. No gradient in T1ρ values across the imaging slice, positioned parallel to the coil, was detected, indicating good homogeneity of the spin-lock field. The BSA phantoms were measured at room temperature (22° to 24°C) using 10 to 12 spin-lock fields between 0.26 and 2.55 G followed by the same fast low-angle shot imaging scheme sequence with five spin-lock times between 30 and 70 milliseconds and a delay of 12 seconds between the scans.
The effect of the radiofrequency radiation deposition by the spin-lock sequence on brain temperature was determined both in normal and ischemic tissue by probing the water resonance frequency (Cady et al., 1995; Corbett et al., 1995). Non–water-suppressed 1H spectra were acquired using STEAM (stimulated-echo acquisition mode) sequence with parameters as follows: TR 2200 milliseconds, TE 60 milliseconds, TM 60 milliseconds, spectral width 2.5 kHz covered with 4096 data points, 64 to 128 averages. A localized isotropic voxel of (3.5 mm)3 was positioned in ipsilateral hemisphere so that it covered areas in the putamen and parietal cortex.
Histology
Rats exposed to 45 minutes of MCA occlusion, as well as the sham-operated ones, were transcardially perfusion-fixed 48 hours after the insult, postfixed, and processed for sectioning as previously described (Gröhn et al., 1999). Brains were sectioned in the horizontal plane at a thickness of 30 μm with a sliding microtome, and the sections were stored in a cryoprotectant tissue-collecting solution (30% ethylene glycol, 25% glycerol in 0.05 mol/L sodium phosphate buffer) at −20°C until processed. Estimation of neuronal densities in parietal cortex and the striatum was done from thionin-stained sections by using Stereo Investigator software in a Neuro Lucida morphometry system (MicroBrightField, Colchester, VT, U.S.A.). These areas were chosen for analysis because they consistently show 60% to 70% neuronal loss 24 hours after 90 minutes of MCA occlusion (Gröhn et al., 1999). The mean neuronal densities were derived from representative three thionin-stained sections spaced at 150-μm intervals. The first section counted was chosen from the horizontal level that corresponds to the midlevel of the slice analyzed by MRI (that is, the level 4.10 mm ventral from bregma according to the rat stereotaxic atlas) (Paxinos and Watson, 1996). A 425-μm wide counting frame extending through all layers was placed on parietal cortex areas 1 and 2 (Paxinos and Watson, 1996). To estimate the neuronal density in the striatum, the entire striatal area was digitized under low-power magnification. The sections were inspected according to a systematic random sampling scheme. This scoring method identifies remaining neurons with intact plasma membrane and nucleus. Neuronal damage percentage was determined as the neuron density difference between ipsilateral and contralateral side divided by neuron density in contralateral side.
Magnetic resonance imaging data analysis
Absolute T1ρ and Dav images were calculated on a pixel-by-pixel basis by fitting the MRI data to a single exponential as a function of spin-locking time or b-values, respectively, using a Matlab routine (Mathworks, Natick, MA, U.S.A.). the T1ρ dispersion and T1 dispersion at the low magnetic field strength are well documented in native and cross-linked protein solutions (Koenig and Brown, 1993; Bryant, 1996), reflecting the low-frequency processes such as slow molecular tumbling, water molecule exchange, and proton exchange. Lorentzian dispersion is expected for the relaxation rate in these preparations, with the inflection point indicating the correlation time of the contributing process. The situation in tissue becomes complex because considerable distribution of water spin lifetimes prevails, and consequently, relaxation dispersion is inevitably broadened. Additional factors influencing T1ρ values include (1) saturation transfer effects close-to-resonance resulting from saturation of spins by the locking pulse (Caines and Schleich, 1991), and (2) contribution of broadened macromolecular lines as the spin-lock field approaches internal magnetic fields of immobile proteins (Brown and Koenig, 1992). Consequently, our reported figures for T1ρ have inherent contributions from several processes. Physical modeling of these factors exceeds the scope of this work; consequently, we have analyzed dispersion of T1ρ (ms) as a function of spin-lock field (B1, G) using an exponential fit of the format:
This treatment yields two coefficients, a and b, which are interpreted so that a describes the T1ρ dispersion (dT1ρ) while b-a estimates T1ρ as B1 approaches zero (T01ρ asymptotically approaching T2). In ischemic tissue, ΔdT1ρ is calculated as the relative change of coefficient a compared with the same brain area in contralateral hemisphere. The contrast-to-noise ratios of the parametric maps were calculated as a difference between ipsilateral and contralateral brain regions divided by the SD of values within the contralateral region of interest.
Brain temperature was calculated from the frequency difference between water and N-acetyl aspartate using the quantitative interrelationship determined by Cady et al. (1995). The resonance frequency of the water was determined in the time domain using a variable projection method (Knijn et al., 1992). The water signal was removed from the spectra using Henkel-Lanczos singular-value decomposition (van den Boogaart et al., 1994), after which resonances from choline-containing metabolites, creatine plus phosphocreatine, N-acetyl aspartate, and lactate (only in ischemic brain) were fitted in the time domain to obtain their respective resonance frequencies.
Statistical significance was estimated using analysis of variance or Student's t-test. All results are indicated as means ± SD. Six animals from the 45-minute MCA occlusion group, together with six sham-operated rats, were used in the correlation analysis. Pearson correlation coefficients (r) between histologically quantified neuronal score and MRI parameters acquired both 25 to 35 minutes of ischemia and 50 to 60 minutes of reperfusion were calculated in striatum and parietal cortex.
RESULTS
T1ρ and cerebral blood flow
Physiologic parameters were within the normal range in all animals undergoing MRI (Table 1). The ΔT1ρ, determined using a spin-lock field of 0.4 G, is shown as a function of CBF (Fig. 1). The drop of CBF from the control value of 75.3 ± 13.9 to less than 22 mL 100 g−1 min−1 prolonged T1ρ significantly from the preocclusion value of 76.2 ± 1.0 milliseconds, and further reduction in CBF was associated with increasing T1ρ. Notice that the CBF threshold for the extensive drop of diffusion has been determined to be approximately 20 mL 100 g−1 min−1 (Busza et al., 1992; Kohno et al., 1995; Gröhn et al., 2000).
Physiologic parameters in each experimental group
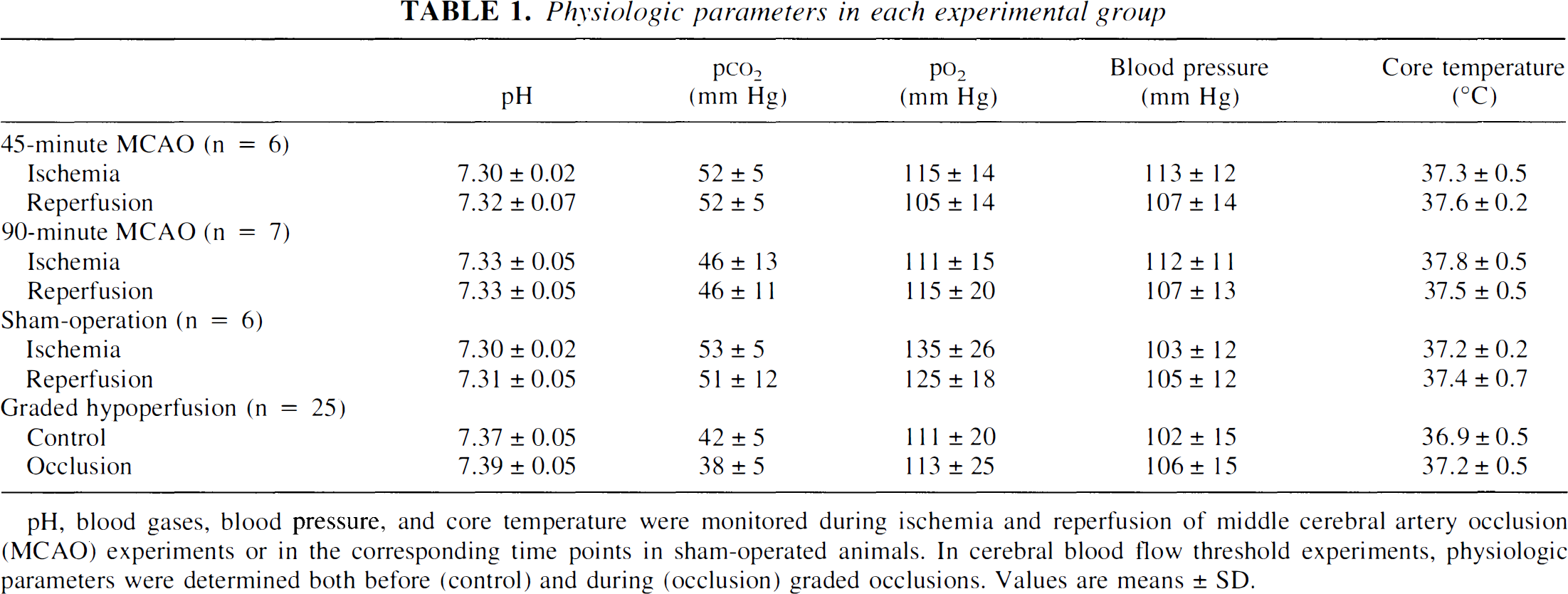
pH, blood gases, blood pressure, and core temperature were monitored during ischemia and reperfusion of middle cerebral artery occlusion (MCAO) experiments or in the corresponding time points in sham-operated animals. In cerebral blood flow threshold experiments, physiologic parameters were determined both before (control) and during (occlusion) graded occlusions. Values are means ± SD.
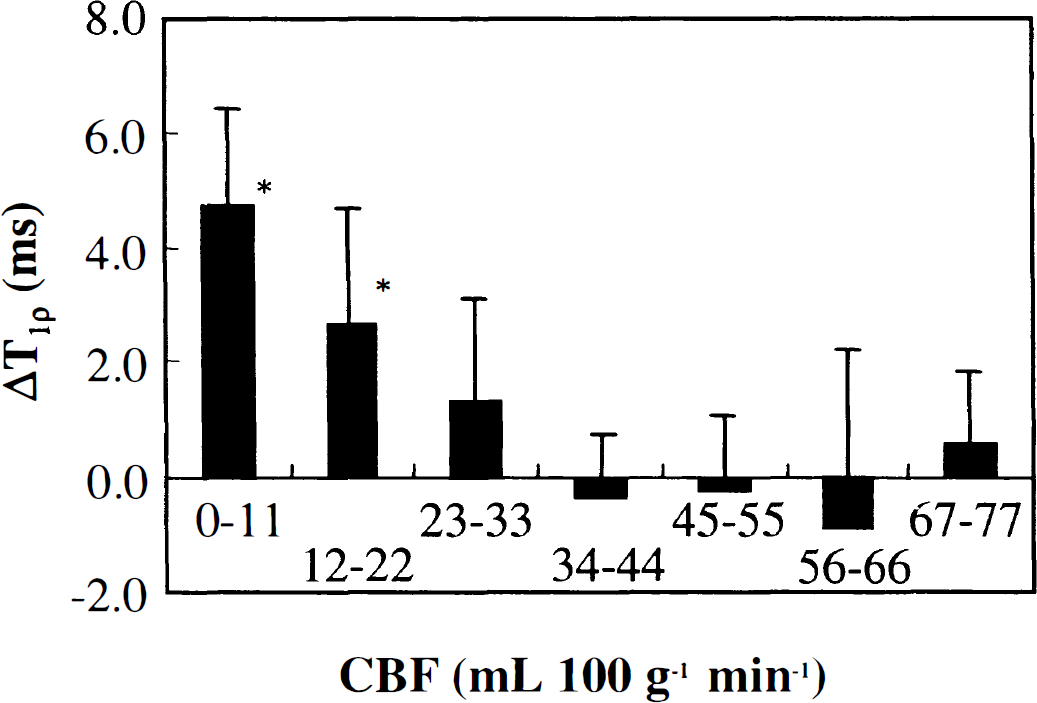
ΔT1ρ as a function of cerebral blood flow (CBF). T1ρ data are divided into 7 groups according to CBF as follows: 0 to 11 (n = 14), 12 to 22 (n = 9), 23 to 33 (n = 11), 34 to 44 (n = 20), 45 to 55 (n = 20), 56 to 66 (n = 14), and 67 to 77 (n = 11) mL 100 g−1 min−1. ΔT1ρ are the differences between preocclusion and occlusion values (mean ± SD). Statistical significance was evaluated using analysis of variance with Tukey's HSD post hoc test (* P < 0.01).
Ninety minutes of middle cerebral artery occlusion
The Dav value decreased by 27.3% ± 0.5% during MCA occlusion in the ipsilateral putamen as a sign of severe ischemia, and after 90 minutes of occlusion, no recovery of the diffusion coefficient on removal of occluder was detected (Fig. 2A). In the unexposed caudate putamen, T1ρ changed from 74.4 ± 2.8 to 115.3 ± 5.2 milliseconds as spin-lock field increased from 0.17 to 1.7 G, indicating an inherent T1ρ dispersion (Fig. 3A). Analysis of relaxation dispersion (dT1ρ) in the unexposed putamen gave dT1ρ = 61.4 ± 6.6 and T01ρ = 64.1 ± 2.6 milliseconds. Visual inspection of a T1ρversus B1 field plot indicates that dT1ρ was greater in ischemic than in normal putamen (Fig. 3A). Indeed, dT1ρ had risen by 28.0% ± 6.6% compared with contralateral putamen, and dT1ρ gradually increased both during and after MCA occlusion, reaching a value that was 48.4% ± 24.1% higher than in contralateral putamen (Fig. 2B). These data show that the increase in dT1ρ in acute cerebral ischemia was of the same or greater magnitude than the decrease in absolute Dav and that dT1ρ dynamically changed both during evolution of ischemia and ischemia–reperfusion damage.
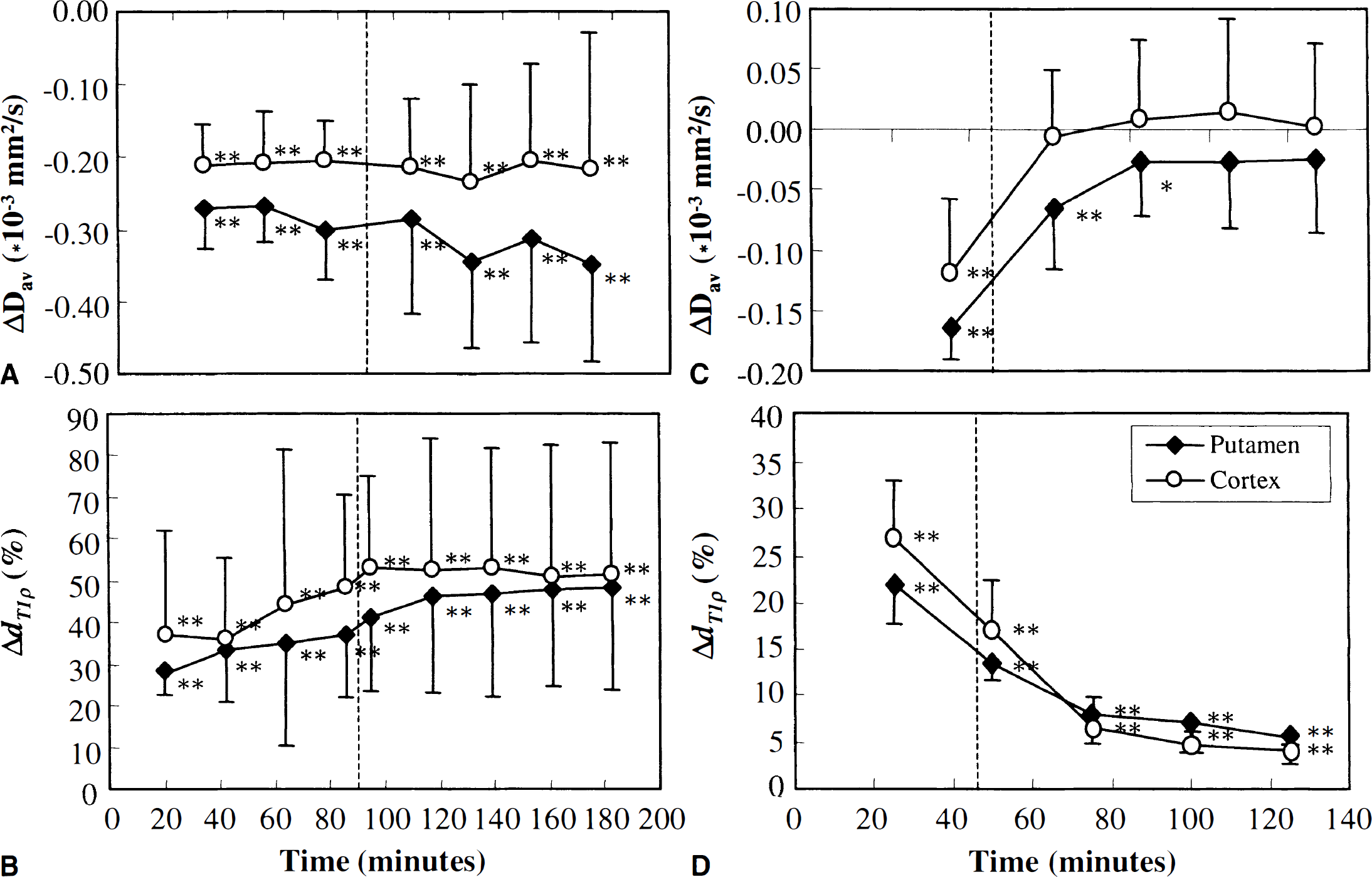
Diffusion and ΔdT1ρ data during and after middle cerebral artery occlusion (MCAO).
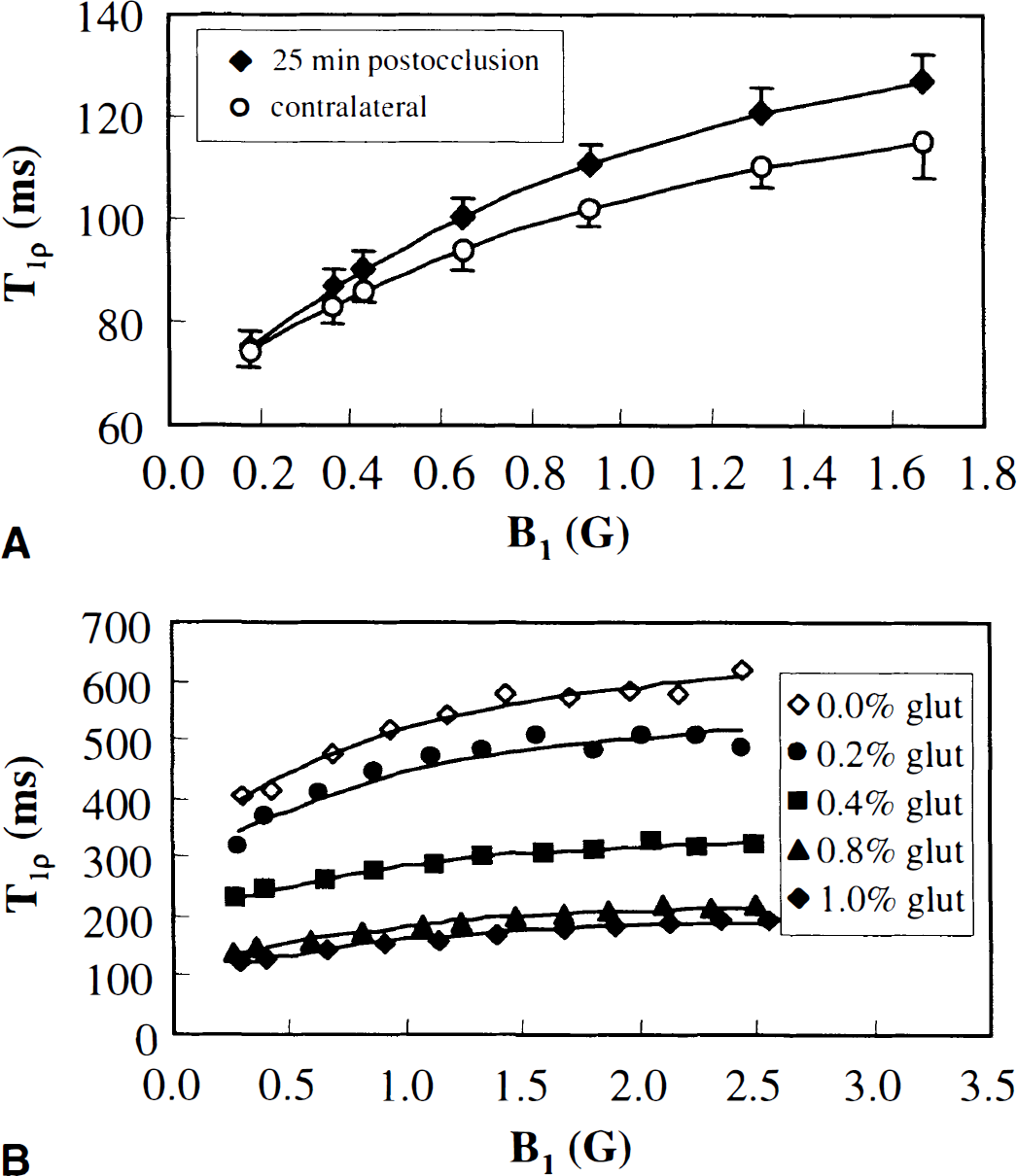
T1ρ relaxation time as a function of spin-lock field in the brain and bovine serum albumine (BSA) phantoms.
The T1ρ relaxation times at given locking field strengths increased as a consequence of ischemia; the greatest increase in T1ρ relaxation and the highest contrast-to-noise ratio were detected with the highest spin-lock field throughout the ischemic region as shown for putamen (Fig. 4). Yet, dT1ρ was a more sensitive indicator of ischemia than any individual T1ρ relaxation time, although the contrast-to-noise ratio of the dispersion was comparable with the highest spin-lock field used.
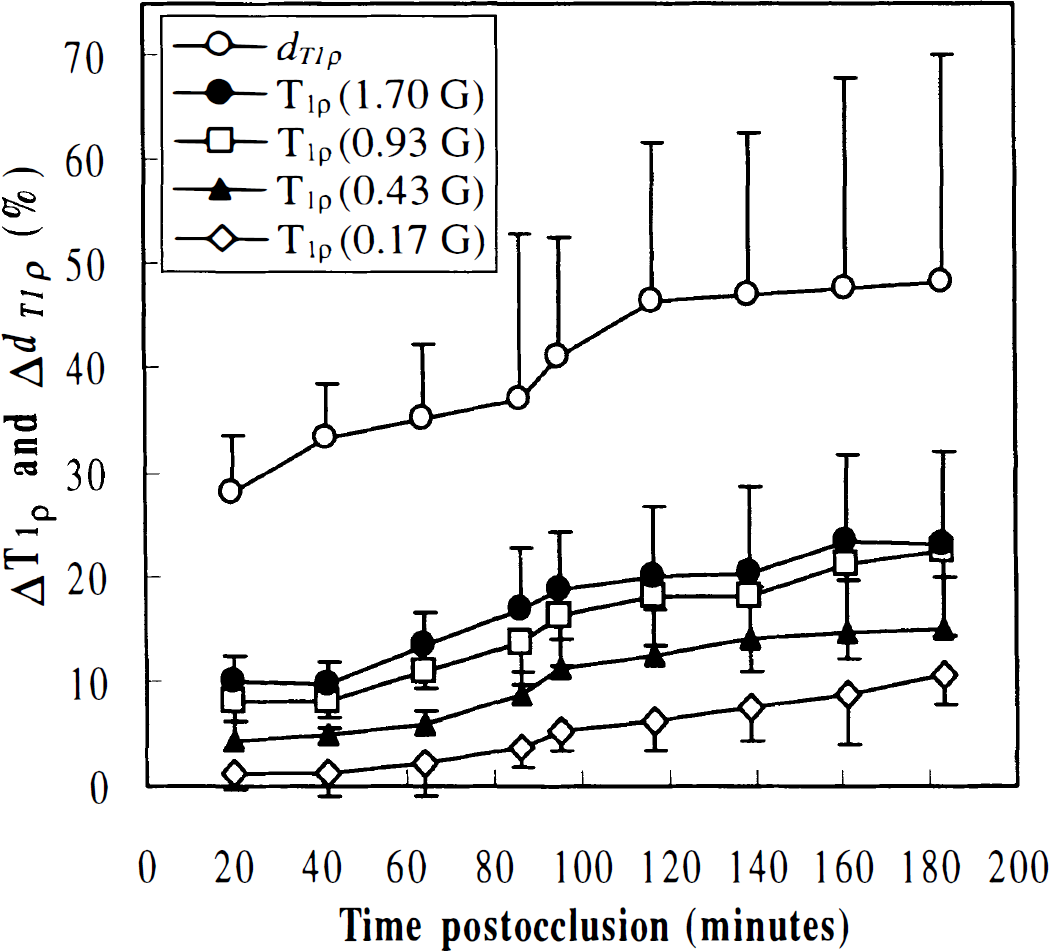
Percentage increase in T1ρ and dT1ρ in ipsilateral relative to contralateral putamen during and after 90 minutes of middle cerebral artery occlusion. T1ρ data from 4 of 7 B1 fields studied are shown for clarity. The contrast-to-noise ratios (see Methods) were 7.1 ± 0.6, 6.4 ± 0.4, 4.9 ± 0.3, 4.0 ± 0.2, and 2.3 ± 0.2 in dT1ρ and T1ρ maps with B1 fields 1.7, 0.93, 0.43, and 0.17 G, respectively.
Forty-five minutes of middle cerebral artery occlusion
After 45 minutes of MCA occlusion, Dav completely recovered on reperfusion (Figs. 2 and 5). Before removal of the occluding thread, dT1ρ had increased by 21.8% ± 4.4% and 26.7% ± 7.3% above the figures determined in the contralateral putamen and parietal cortex, respectively. After reperfusion, dT1ρ partially recovered but stabilized to a level that was 6.0% ± 2.4% above the inherent dT1ρ in the contralateral hemisphere. The T2-weighted images showed hyperintensity 48 hours later as a MRI sign of irreversible damage in both putamen and cortex of all animals. The dT1ρ had elevated by 64% ± 17% and diffusion decreased by 15% ± 7.3% 48 hours after exposure to 45 minutes of MCA occlusion.
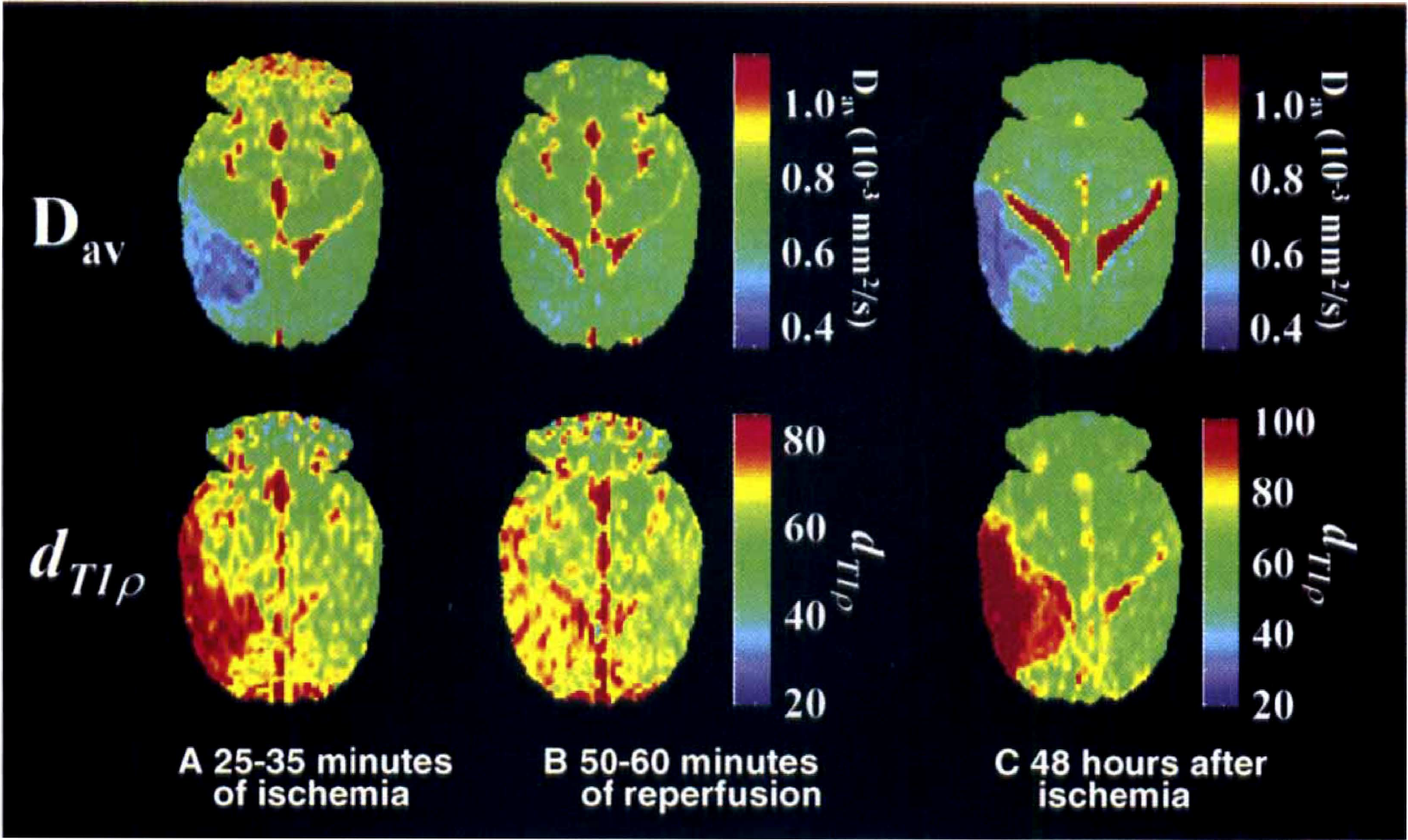
Representative absolute Dav and dT1ρ images during and after 45 minutes of middle cerebral artery occlusion (MCAO). Magnetic resonance imaging data were acquired
Histology
Neuronal counting was performed from animals exposed to 45 minutes of MCA occlusion, since the outcome of the tissue after ischemia of this duration may vary (Li et al., 1999), but 90 minutes of MCA occlusion results in a severe drop of neuronal density in these regions (Gröhn et al., 1999). Neuronal loss ipsilateral to MCA occlusion by 77% ± 12.2% (P < 0.01) and 55% ± 22% (P < 0.01) were detected in striatum and parietal cortex, respectively, but no significant differences in neural densities were detected in sham-operated animals. No signs of subarachnoid hemorrhage were seen in any of the brains analyzed.
During ischemia, dT1ρ and Dav significantly correlated with histologically determined degree of neuronal damage (Table 2). The T1ρ values displayed a trend toward higher correlation values as a function of spin-lock strength, and r values of T1ρ with spin-lock amplitudes of 1.3 G or more were comparable with dT1ρ and Dav. During reperfusion, T1ρ and dT1ρ correlated highly with neuronal outcome in striatum, whereas in the cortex, dT1ρ was the only MRI parameter capable of predicting neuronal score. Under these conditions, Dav failed to indicate significant correlation with neuronal damage either in striatum or cortex.
Pearson correlation coefficient between neuronal damage and MRI parameters
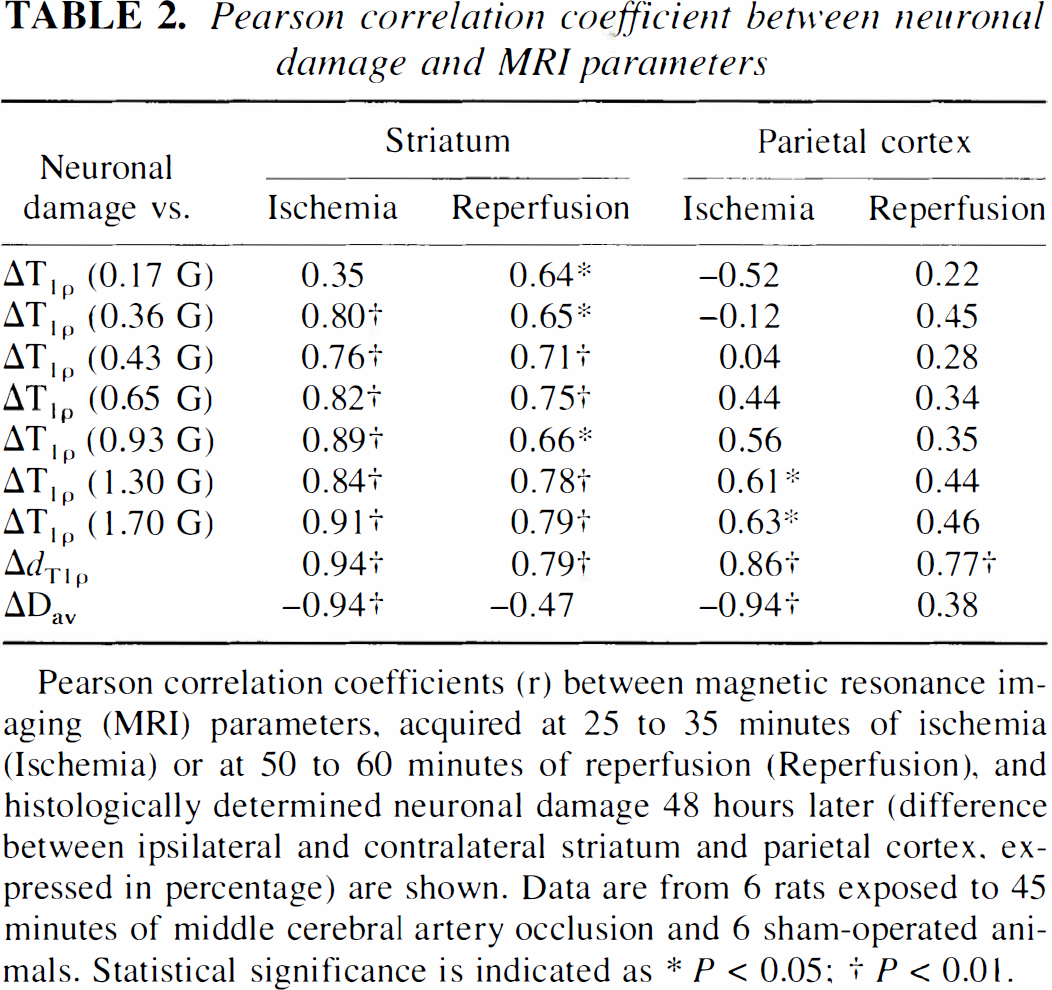
Pearson correlation coefficients (r) between magnetic resonance imaging (MRI) parameters, acquired at 25 to 35 minutes of ischemia (Ischemia) or at 50 to 60 minutes of reperfusion (Reperfusion), and histologically determined neuronal damage 48 hours later (difference between ipsilateral and contralateral striatum and parietal cortex, expressed in percentage) are shown. Data are from 6 rats exposed to 45 minutes of middle cerebral artery occlusion and 6 sham-operated animals. Statistical significance is indicated as * P < 0.05
P < 0.01.
Protein phantom studies
It is well established that protein solutions show dispersion of T1ρ (Koenig and Brown, 1993; Bryant, 1996). To reconcile the effects of molecular mobility on MRI relaxation, we quantified T1ρ in protein phantoms both in native and cross-linked states (Brown and Koenig, 1992). It is evident that T1ρ was shorter and dT1ρ smaller in immobilized cross-linked protein samples than in native protein solution, suggesting that protein mobility influences both T1ρ and dT1ρ (Fig. 3B). A previous study has shown that dT1ρ apparently is insensitive to protein concentration, although it influences the individual T1ρ values both in native and cross-linked solutions (Virta et al., 1997).
Brain temperature and T1ρ dispersion
Temperature was 37.3° ± 0.7°C and 35.2° ± 0.4°C in normal and ischemic brain tissue, respectively (Fig. 6), a difference consistent with a previous report obtained in focal cerebral ischemia (Morikawa et al., 1992). No increase in brain temperature could be detected immediately after repeated dT1ρ MRI either in normal or ischemic brain, showing that the MRI method used did not produce adverse heating effects. The deliberate hypothermia decreased core and brain temperature to 33.2° ± 0.7°C and 33.8° ± 0.9°C, respectively. In normothermic and hypothermic brain, dT1ρ was 63.2 ± 2.2 and 62.8 ± 1.3, respectively, showing no effect of temperature on dispersion at this temperature range.
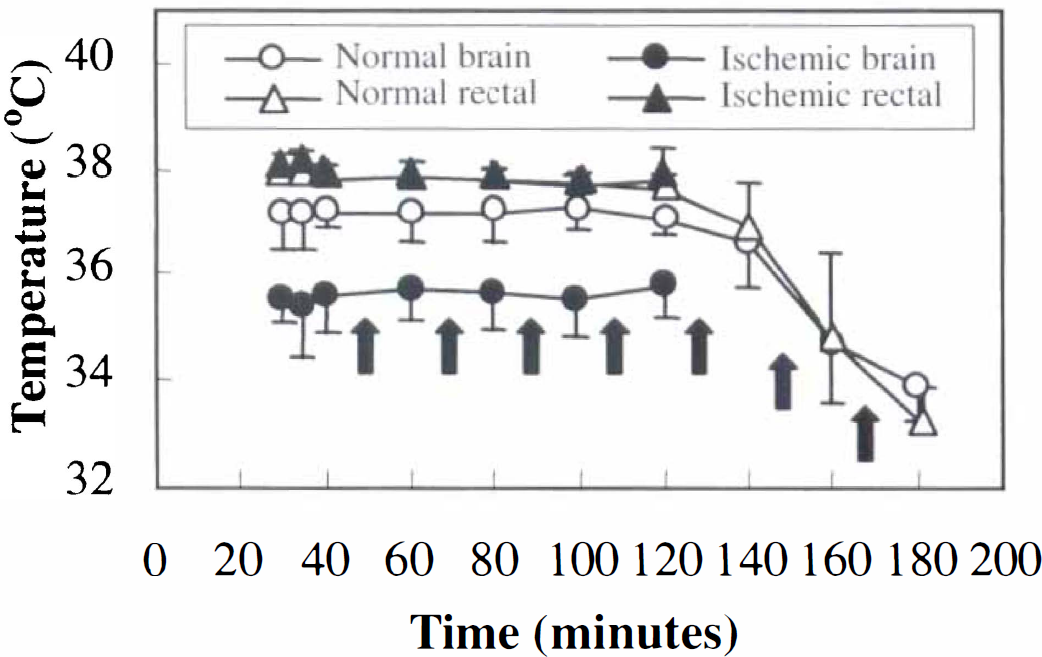
Temperature in normal, ischemic, and hypothermic rat brain. Brain temperature was determined as described in the Methods section using localized 1H NMR spectroscopy during repeated T1ρ dispersion measurements and hypothermia. Arrows indicate the onset of T1ρ dispersion data acquisitions. Core temperatures in both experimental groups are also shown. Hypothermia was induced for normal animals in the end of each experiment. Values are means ± SD.
DISCUSSION
We have characterized T1ρ changes during acute cerebral ischemia and demonstrated that both T1ρ and dT1ρ can be used as novel, sensitive MRI indicators of tissue alterations in acute stroke. After 25 minutes of MCA occlusion, dT1ρ is elevated by approximately 25%, which is of the same magnitude commonly reported for the apparent diffusion coefficient (Moseley et al., 1990; Davis et al., 1994) or Dav (Miyabe et al., 1996; Gröhn et al., 2000). An important difference between the dynamics of diffusion and dT1ρ is that the latter MRI variable increases during evolution of the damage process. For instance, 3 hours after onset of 90 minutes of MCA occlusion, dT1ρ is elevated by approximately 45%, a value that renders it an excellent endogenous MRI contrast for acute ischemia. Notice that in the permanent MCA occlusion models, diffusion drops extensively by 30% to 40% during the first hour, showing only a minor change over the subsequent 4 to 5 hours (Knight et al., 1994; Hoehn-Berlage et al., 1995). A further important observation here is that dT1ρ remains elevated in the early phase of reperfusion in the tissue destined to loss of neurons despite complete recovery of the diffusion. It appears that T1ρ senses pathophysiologic factors associated with irreversible ischemia, whereas diffusion shows potential for full recovery on restoration of CBF. Thus, this relaxation parameter may be an index of irreversible ischemia with improved sensitivity over diffusion in predicting tissue outcome (Li et al., 1999).
Our data show that T1ρ increases at similar CBF levels that are known to cause extensive drop in the apparent diffusion coefficient of water (Busza et al., 1992; Kohno et al., 1995) and Dav (Gröhn et al., 2000) and that the response of T1ρ to reduced flow occurs within minutes. The latter aspect clearly is different from the time course of T2, and increase of T2 relaxation is detected only after hours of ischemia (Knight et al., 1994). Notice that under experimental conditions identical to those just described, T2 relaxation is initially decreased at CBF of less than 50 mL 100 g−1 min−1, predominantly because of the blood oxygenation level-dependent effect (Gröhn et al., 2000). It is likely that energy-dependent factors, such as membrane depolarization, shift of ions and water across the plasma membrane, alterations in cytoskeletal organization (Jenkins et al., 1979), and acidification influence T1ρ and diffusion with distinct contributions. Therefore, these two MRI variables clearly possess a complementing value in predicting long-term tissue status.
We used a spin-lock method to quantify T1ρ, allowing relaxation to be probed at low spin-lock field and to take advantage of the high signal-to-noise ratio provided by main magnetic field. It is generally considered that under these conditions, the major contribution to the relaxation time comes from the slow processes taking place in the macromolecules or in the macromolecule–water interface (Sepponen et al., 1985; Rommel and Kimmich, 1989; Brown and Koenig, 1992). Consequently, T1ρ probes specific spin populations, insignificantly influencing T1 in the laboratory frame, but rather senses similar processes affecting T2. Consistent with the latter idea, tissue T1ρ and T2 are of similar magnitude, and T1ρ approximates T2 as spin-lock field approaches zero. However, the spin-lock method used for quantification of T1ρ inherently imposes contributions from magnetization transfer and related processes (Brown and Koenig, 1992). It has been shown that T1ρ and magnetization transfer contrasts are similar in the brain and brain tumors (Markkola et al., 1996). The exchange of OH-and NH-protons in amino acids proceeds at 103 to 104 1/s (Liepinsh and Otting, 1996). The exchange rates are close to the condition ω1 = γB1 of data acquisition used here, and thus the exchange may significantly contribute to T1ρ as it does to the magnetization transfer (Liepinsh and Otting, 1996). As the proton exchange rates are strongly pH dependent, acidification associated with anaerobic conditions in ischemia may contribute to the observed T1ρ changes. Notice that magnetization transfer contrast increases during the first 40 minutes of MCA occlusion (Ewing et al., 1999).
The current data highlight three important mechanistic aspects of potential factors affecting T1ρ in hyperacute ischemia. First, the method used for T1ρ quantification is not directly sensitive to flow, since CBF can be reduced by 50% without effect on the relaxation time (Fig. 1). This is consistent with our previous study showing that diffusion recovery on removal of MCA occluder was not associated with a sudden change in T1ρ (Gröhn et al., 1999). Second, reduction of brain temperature per se does not influence dT1ρ, thus excluding the temperature drop associated with ischemia as a major contributor to the observed dT1ρ change. This observation is interesting because temperature is a well-known factor affecting T1 relaxation (Youl et al., 1992) as well as exchange processes, presumably underlying the T1ρ relaxation. However, notice that temperature variation in vivo is small compared with temperature range used in protein phantom or tissue sample studies (Bottomley et al., 1984; Bryant, 1996). Third, protein mobility affects dT1ρ in BSA phantoms. The increase in dT1ρ on acute ischemia is similar to that detected with decreasing cross-linking density, implying that intracellular destructive processes leading to, for instance, cytoskeletal alterations within minutes after onset of ischemia (Jenkins et al., 1979) may affect dT1ρ. As glutaraldehyde links the protein molecules together, predominantly through NH-groups, T1ρ relaxation changes in BSA phantoms with different degrees of cross-linking also may reflect difference in proton exchange rates. However, the water-to-protein (or macromolecule) ratio inevitably affects T1ρ relaxation, and consequently, net water gain into the tissue during the course of ischemia affects T1ρ (Gröhn et al., 1999), as it does T1 and T2, and becomes the predominant mechanism under these conditions.
The T1ρ MRI places requirements on the coil performance as well as on the radiofrequency system, but as a benefit over diffusion MRI, no need for high-performance gradients exists. It has been shown that spin-lock field of 0.35 G can be technically obtained with a head coil at 0.1 T (Markkola et al., 1996) and 2.5 G at 0.02 T using a local coil (Lamminen et al., 1993). The deposition of radiofrequency radiation is a drawback of spin-lock MRI and must be dealt with in clinical MRI. We show here that even at a B0 of 4.7 T, no detectable tissue heating by the T1ρ method using high B1 is observed.
To conclude, we have shown that the on-resonance T1ρ is a sensitive indicator of acute cerebral ischemia changing at a similar CBF threshold to diffusion. Furthermore, dispersion of T1ρ reveals irreversible ischemia with better accuracy than diffusion, suggesting that different cellular factors affect these MRI variables. Our data suggest that multimodal MRI may be useful for detection of irreversible ischemia early in the disease process.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Dr. Robert Bryant (University of Virginia, Charlottesville, VA, U.S.A.) and Dr. Juhani Lounila (University of Oulu, Oulu, Finland) for fruitful discussion concerning T1ρ dispersion. The authors also thank Ms. Niina Kuhmonen for expert technical assistance.