Abstract
Transplantation of stem cells has the possibility of restoring neural functions after stroke damage. Therefore, we transplanted neuronal progenitors generated from monkey embryonic stem (ES) cells into the ischemic mouse brain to test this possibility. Monkey ES cells were caused to differentiate into neuronal progenitors by the stromal cell-derived inducing activity method. Focal cerebral ischemia was induced by occluding the middle cerebral artery by the intraluminal filament technique. The donor cells were transplanted into the ischemic lateral striatum at 24 h after the start of reperfusion. The cells transplanted into the ischemic brain became located widely around the ischemic area, and, moreover, the transplanted cells differentiated into various types of neurons and glial cells. Furthermore, at 28 days after the transplantation, over 10 times more cells in the graft were labeled with Fluorogold (FG) by stereotactic focal injection of FG into the anterior thalamus and substantia nigra on the grafted side when compared with the number at 14 days. From these results we confirmed the survival and differentiation of, as well as network formation by, monkey ES-cell-derived neuronal progenitors transplanted into the ischemic mouse brain.
Introduction
Stroke, especially cerebral infarction, strikes a great number of people, over 500,000 new patients a year in the United States alone, and the incidence rate is increasing every year. As the sequelae of stroke directly and negatively influence the daily life of the patient, a strategy to restore to normal the neuronal population reduced by the ischemic event is highly desired. However, present therapies are mainly for primary or secondary prevention. Therefore, cell replacement therapies, such as those involving embryonic stem cells (ES cells; Erdo et al, 2003), neural stem cells (Modo et al, 2002a, b; Nelson et al, 2002; Savitz et al, 2002; Watson et al, 2003), and bone marrow-derived stem cells (Beck et al, 2003; Chen et al, 2001, 2003; Li et al, 2001, 2002; Zhao et al, 2003), have the possibility of offering a novel potential treatment for stroke damage (Bjorklund and Lindvall, 2000; Kondziolka et al, 2002).
Embryonic stem cells are self-renewing, pluripotent cells derived from the inner cell mass of the preimplantation blastocyst. They can be expanded to large numbers while maintaining their potential to differentiate into various somatic cell types of the three germ layers, and have many of the characteristics required of a cell source for cell-replacement therapy, including proliferation and differentiation capacities. Indeed, ES cells have been found to differentiate in vitro into many clinically relevant cell types, including hematopoietic cells, cardiomyocytes, insulin-secreting cells, osteocytes, hepatocytes, keratinocytes, glial cells, and neurons (Bjorklund and Lindvall, 2000; Kondziolka et al, 2002). After transplantation into the central nervous system (CNS), ES-cell-derived neuronal progenitors have been shown to become integrated into the host tissue, which, in some cases, results in functional improvement (Wei et al, 2005). A few years ago, we discovered a strong neural differentiating activity affecting ES cells and named it stromal cell-derived inducing activity (SDIA; Kawasaki et al, 2000). By use of PA6 stromal cell monolayers, we could cause mouse ES cells to differentiate efficiently into neural precursors and neurons on the surface of these stromal cells in the absence of exogenous bone morphogenic protein-4 (BMP-4) within a week. Originally, SDIA was used to cause efficient differentiation of ES cells into dopaminergic neurons for investigation in Parkinson models (Kawasaki et al, 2000). Since then, SDIA induction has also been applied to primate ES cells, which generate large numbers of neuronal progenitors and postmitotic neurons when cocultured with PA6 cells for a couple of weeks (Kawasaki et al, 2001). Further, we very recently showed the survival and differentiation of mouse ES-derived neural progenitors transplanted into the ischemic mouse brain (Takagi et al, 2005a), and we also just reported that dopaminergic neurons generated from monkey ES cells function in a Parkinson primate model (Takagi et al, 2005b). Now we are interested in focusing on the behavior of primate ES-derived neuronal progenitors in a brain environment recently subjected to ischemia. In the present study, we generated neural progenitors from monkey ES cells, induced an ischemia-reperfusion injury by the intraluminal thread method (Hara et al, 1996), which results in a stable focal cerebral infarction, and then transplanted these cells into the postischemic brain. This is the first report, to our knowledge, of the use of primate ES-cell-derived neurons for transplantation into the postischemic brain.
Materials and methods
Maintenance of Primate Embryonic Stem Cells
Cynomolgus monkey ES cell lines CMK6 and CMK9 were established, and the pluripotency of the cells was confirmed by teratoma formation in mice with severe combined immunodeficiency, as described previously (Kawasaki et al, 2001). Undifferentiated ES cells were maintained on a feeder layer of mitomycin C (WAKO, Osaka, Japan)-treated mouse embryonic fibroblasts in DMEM (Sigma, St Louis, MO, USA)/F-12 (Invitrogen Corp., Carlsbad, CA, USA) supplemented with 0.1 mmol/L 2-mercaptoethanol (Sigma)/1000 U/mL leukemia inhibitory factor (LIF; Chemicon, Temecula, CA, USA)/20% knockout serum replacement (Invitrogen Corp.)/4 ng/mL fibroblast growth factor-2 (FGF2; Upstate Biotechnology, Lake Placid, NY, USA). Subculturing of ES cells was performed by using 0.25% trypsin (Invitrogen Corp.) in PBS containing 20% knockout serum replacement (Invitrogen Corp.) and 1 mmol/L CaCl2 (WAKO), as described earlier (Kawasaki et al, 2001).
Induction of Neuronal Progenitors from Primate Embryonic Stem Cells
PA6 cells were plated on type I collagen-coated chamber slides (Becton Dickinson Labware, Franklin Lakes, NJ, USA) or gelatin (Sigma)-coated dishes (Becton Dickinson Labware) and used as a feeder cell layer. To strictly avoid contamination by incidentally differentiating cells, we manually selected undifferentiated ES cell colonies with stem cell-like morphology (tightly packed cells with a high nucleus/cytoplasm ratio). Undifferentiated ES cell colonies were first washed twice with GMEM medium (Sigma) supplemented with 10% knockout serum replacement/1 mmol/L pyruvate (Sigma)/0.1 mmol/L nonessential amino acids (Invitrogen Corp.)/0.1 mmol/L 2-mercaptoethanol (Sigma). After trypsinization for 5 mins at 37°C, the partially dissociated ES cell clumps (10 to 50 cells/clump) were plated on PA6 cells at a density of 1000 clumps/10-cm dish and cultured in the differentiation medium for 2 weeks. The differentiated colonies were detached from the feeder cells by using a papain dissociation system (Worthington Biochemical Corporation, Lakewood, NJ, USA).
Immunofluorescence Study
Single- and double-immunofluorescence studies were performed on the differentiated cells after permeabilization and blocking for nonspecific binding with 0.3% Triton X-100 (Sigma) and 10% normal donkey serum (Jackson Immunoresearch Laboratories, Inc., West Grove, PA, USA), respectively. The cells were stained overnight at 4°C with the following primary antibodies: anti-NCAM (1:300; Chemicon), anti-Musashi-1 (l:200;gift from Dr H Okano, Keio University, Japan), and anti-β III tubulin (1:300; Babco, Richmond, CA, USA). They were subsequently reacted with donkey Cy3-labeled secondary antibodies (1:100; Jackson Immunoresearch Laboratories) or donkey fluorescein isothiocyanate (FITC)-labeled secondary antibodies (1:100; Jackson Immunoresearch Laboratories), at room temperature for 120 mins. The percentage of immunopositive cells after papain treatment was evaluated by using a Fluoview FV300 laser confocal microscope (Olympus Optical Co., Tokyo, Japan). For quantification, cells in five randomly selected fields, each of which included 100 to 500 cells, were counted in 3 to 5 independent cultures.
Induction of Focal Ischemia
Animal experiments were performed in accordance with our institutional guidelines. C57BL/6 mice (20 to 25 g) were anesthetized with 2% halothane and maintained on 1.5% halothane in 70% nitrous oxide and 30% oxygen. Regional cerebral blood flow (rCBF) was measured as described previously (Hara et al, 1996, 1997) by using a laser-Doppler (Omegawave, Tokyo, Japan) equipped with a flexible skull probe. Rectal temperature was maintained between 36.5°C and 37.5°C with a homeothermic blanket. Focal cerebral ischemia was induced by occluding the middle cerebral artery (MCA) by the intraluminal filament technique, as described previously (Hattori et al, 2004; Takagi et al, 2002). After surgery, the mice were kept at 37°C for 30 mins, and then the filament was withdrawn to allow reperfusion.
Transplantation
Under inhalational anesthesia with 2% halothane, the mice were placed in a stereotaxic frame (Narishige, Tokyo, Japan) 24 h after the ischemia-reperfusion. Each animal received an injection of 2 μL, (1 μL/min, 5 to 20 × 104 cells/μL) of neuronal progenitor colonies derived from ES cells into the lesioned striatum (from bregma: A -0.5 mm, L + 2.5 mm, V + 3.0 mm, incisor bar 0) via a Hamilton microsyringe fitted with a 26-gauge blunt needle.
Immunosuppression
The mice were administered cyclosporine A (Carbiochem, San Diego, CA, USA) contained in their drinking water (200 mg/L) from 3 days before transplantation until the period of transcardiac perfusion.
Retrograde Tracer Labeling
At 7 days before transcardiac perfusion, the mice were anesthetized inhalationally with 2% halothane and then received an injection of 0.2 μL (0.05 μL,/min) of hydroxystilbamidine, a retrograde tracer for neurons (2% solution in saline; equivalent to FG; Molecular Probes, Eugene, OR, USA) into the anterior thalamus (from bregma: A -0.5 mm, L + 0.7 mm, V + 3.5 mm, incisor bar 0) or substantia nigra (from bregma: A + 3.0 mm, L + 1.5 mm, V + 4.25 mm, incisor bar 0) on the grafted side via a Hamilton microsyringe fitted with a 26-gauge blunt needle.
Immunohistochemistry
At 14 (n = 5) or 28 days (n = 5) after the transplantation, the animals were euthanized with pentobarbital and perfused with PBS, followed by 4% paraformaldehyde. Their brains were postfixed overnight and equilibrated in graded solutions of sucrose from 15% to 30%. Coronal 40-μm sections were cut serially on a freezing microtome and post-fixed in 4% paraformaldehyde for 10 mins. Then the mounted samples were completely dipped in 10 mmol/L sodium citrate buffer, pH 6.0, and irradiated at 500 W for 1 min in a microwave apparatus for antigen retrieval.
After several washes in 0.1 M PBS, the sections were incubated for 1.5 h at room temperature with 4% skim milk (Becton Dickinson and Company, Sparks, MD, USA) containing 0.3% Triton X-100 (Sigma). Thereafter, immunohistochemical staining was performed at 4°C for 16 h with antibodies against human nuclei (HN) (1:100; Chemicon), HuC (1:200, Chemicon), HuD (1:200; Chemicon), glial fibrillary acidic protein (GFAP; 1:1500, Chemicon), glutamate decarboxylase (GAD; 1:100, Sigma), glutamate (1:200, Chemicon), choline acetyltransferase (1:100, Chemicon), tyrosine hydroxylase (TH, 1:60, Chemicon), serotonin (1:20,000, Dia Sorin, Stillwater, MN, USA), calbindin (1:500, Chemicon), DARPP-32 (1:500, Chemicon), parvalbumin (1:500, Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA), MAP-2 (1:100, Santa Cruz) synaptophysin (1:50, ZYMED Laboratories Inc., South San Francisco, CA, USA), and FG (1:5000, Chemicon), with 2% skim milk and 0.3% Triton X-100 in PBS used as the diluent. Then, after 3 rinses in PBS, the sections were incubated for 2 h at room temperature with FITC-labeled (1:200; Jackson Immunoresearch Laboratories), Cy3-labeled (1:300; Jackson Immunoresearch Laboratories), or Cy5-labeled (1:500; Jackson Immunoresearch Laboratories) secondary antibodies. Immunostained sections were evaluated by observation under a confocal microscope. The area of immunopositive cells was quantified in every fourth section throughout the graft and its surroundings. All values were expressed as mean plus or minus 1 s.d. Statistical analyses were performed by use of the unpaired Student t-test. P-values of less than 0.05 were considered statistically significant.
Infarct Measurement
Serial coronal brain sections separated by 1-mm intervals were stained with cresyl violet. Infarct volumes were quantified by summing the infarct area of each section with an image analysis software (Scion Image beta 4.02, Scion Corporation, Frederic, MD, USA).
Results
Physiologic Parameters and Volume of Infarction
In randomly selected animals (n = 5), rCBF during middle cerebral artery occlusion (MCAO) and reperfusion were as follows: 12.5% ± 5.2% during ischemia and 87.0% ± 9.9% after ischemia. The infarct volume of mice after a 30-min MCA occlusion was 10.1 ± 0.3 mm3. When ES-derived neuronal progenitors were transplanted, the infarct volume at 14 days (after transplantation) was 10.0 ± 1.1 mm3 and that at 28 days 10.0 ± 0.3 mm3.
Induction of Neuronal Differentiation of Primate Embryonic Stem Cells on Plasminogen Activator 6 Cells
We applied the SDIA method to primate ES cells as described previously by Kawasaki et al (2001). After having been cultured on a monolayer of PA6 cells for 2 weeks, the majority of the primate ES cell colonies showed extensive neurite formation. These colonies contained a large number of cells positive for Musashi-1 (neural progenitors; Figure 1A, 90.4% ± 7.5%), neural precursors and neurons (NCAM; Figure 1B and 1C, 97.5% ± 4.6%), and neuronal precursors positive for class III β-tubulin (TuJ 1; Figure 1D, 83.8% ± 10.3%). At this time point, 78.3% + 7.5% of the total cells were immunoreactive for NCAM, and 75.0% + 15.4% for Musashi-1.
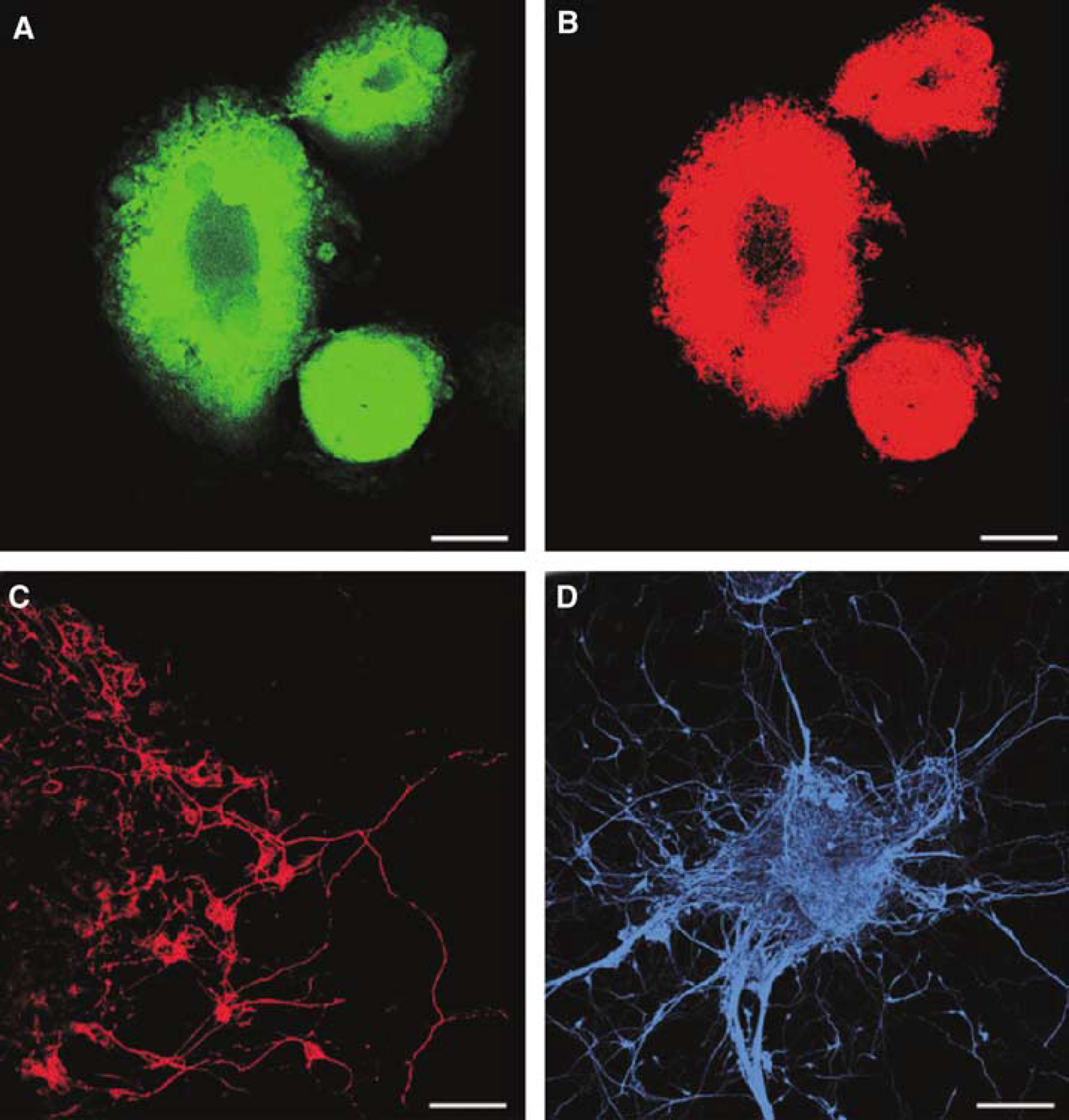
Neuronal progenitors derived from monkey ES cells cocultured for 2 weeks on a monolayer of PA6 cells. Musashi-1 (
Transplantation of Embryonic Stem-Derived Neuronal Progenitors into the Ischemic Brain
Middle cerebral artery occlusion for 30 mins resulted in brain infarction that was restricted to the lateral striatum. Neuronal progenitors from primate ES cells were transplanted into this area by a stereotactic method (Figures 2A to 2F). The population of donor cells was 1.0 to 4.0 × 105, and the grafts were detected by their immunoreactivity toward antibodies against HN. Human nuclei antibody recognizes monkey cells in vivo and HN immunoreactivity was not detected in mouse cells in vivo (Supplementary Figure a and b). Thus, we used HN antibody for detection of the grafts (Supplementary Figure c). The cells transplanted into the ischemic brain became located widely around the ischemic area (Figures 2C-E). The grafted cells occupied 11.5 ± 7.9 mm3 (8.7 × 104 ± 2.6 × 104 cells) at 2 weeks (Figures 2C and 2F) and 19.1 ± 8.4 mm3 (14.3 × 104 ± 1.5 ×104 cells) at 4 weeks after transplantation (Figures 2D to 2F). On the contrary, in sham-operated animals, ES-derived cells moved laterally along the corpus callosum. As shown in Figures 2A and 2B, surviving cells in the sham controls occupied only a small area (3.0 ± 2.2 mm3, 4.7 × 104 ± 1.7 × 104 cells at 2 weeks and 4.9 ± 0.3 mm3, 6.7 × 104 ± 2.9 × 104 cells at 4 weeks after transplantation).
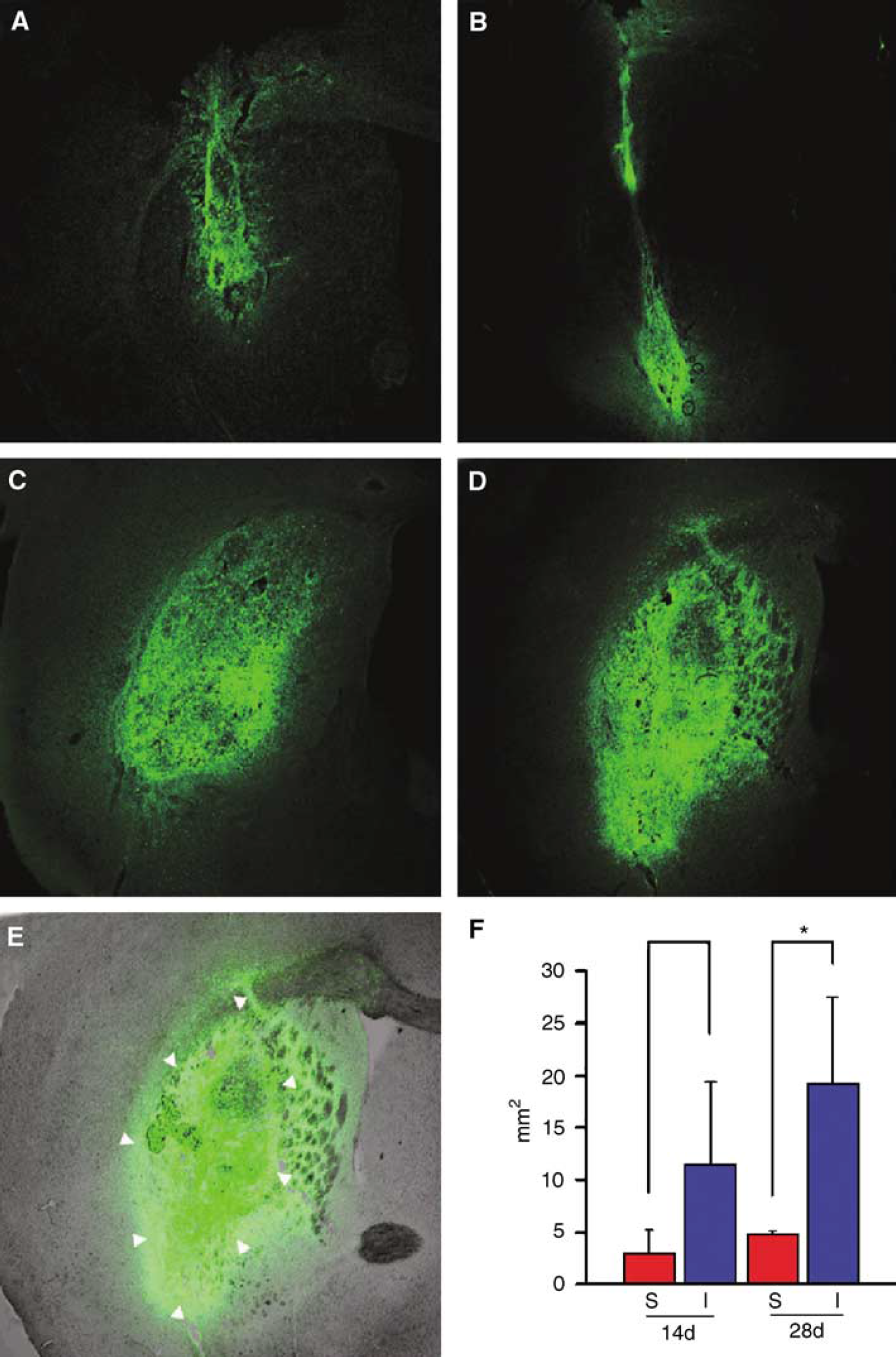
Neuronal progenitor grafts from monkey ES cells transplanted into the lateral striatum of a mouse subjected to MCAO. (
Differentiation of Transplanted Embryonic Stem-Derived Neuronal Progenitors
Next we characterized the transplanted cells in the brains immunohistochemically at 4 weeks after transplantation by examining various markers. The grafts expressed HuC (a marker of immature neurons; Figures 3A and 4A, 73.4% ± 22.0% at 14 days and 62.0% ± 9.7% at 28 days after transplantation) and HuD (a marker of mature neurons; Figures 3B and 4A, 30.9% ± 6.8% at 14 days and 60.5% ± 6.1% at 28 days after transplantation), which revealed them to have neuronal characteristics. The expression of HuD at 28 days after the grafting was significantly stronger than that at 14 days after it. However, astroglias positive for GFAP (Figures 3C and 4A, 1.3% ± 1.0% at 14 days and 1.4% ± 0.5% at 28 days after transplantation) were few in the grafts. In addition, we analyzed various neuronal transmitter-related markers of the transplanted cells. At 14 days after transplantation, 9.6% ± 6.9% of the grafted cells expressed TH (Figure 4C); 29.0% ± 19.0%, glutamate (Figure 4C); 23.8% ± 15.1%, GAD (Figure 4C); 8.4% ± 2.9%, ChAT (Figure 4C); and 1.6% ± 1.4%, serotonin (Figure 4C). As for striatal neuron-related markers, 5.1% ± 3.1% expressed calbindin (Figure 4B); 1.4% ± 0.5%, DARPP-32 (Figure 4B); and 3.1% ± 1.4%, parvalbumin (Figure 4B). By 28 days after transplantation, 14.9% ± 8.1% were TH-positive (Figure 4C); 26.6% ± 6.0%, glutamate-positive (Figures 3D and 4C); 20.0% ± 0.7%, GAD-positive (Figure 4C); 12.9% ± 1.4%, ChAT-positive (Figure 4C); 0.9% ± 0.1%, serotoninpositive (Figure 4C); 5.4% ± 2.6%, calbindin-positive (Figure 4B); 4.8% ± 1.8%, DARPP-32-positive (Figures 3E and 4B); and 5.7% ± 2.5%, parvalbumin-positive (Figures 3F and 4B).
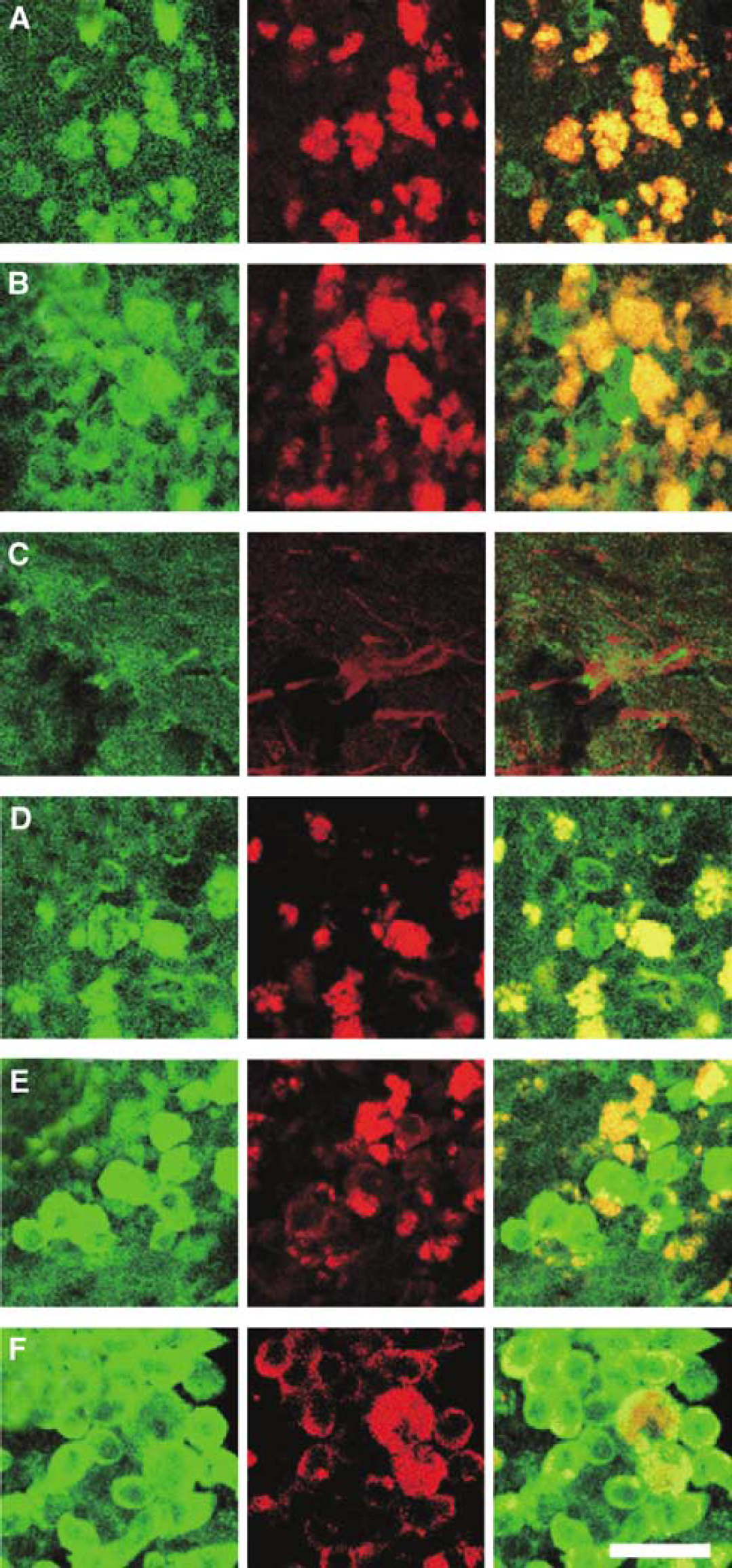
Differentiation of transplanted neuronal progenitors generated from ES cells. Transplanted primate cells were detected by HN immunoreactivity (
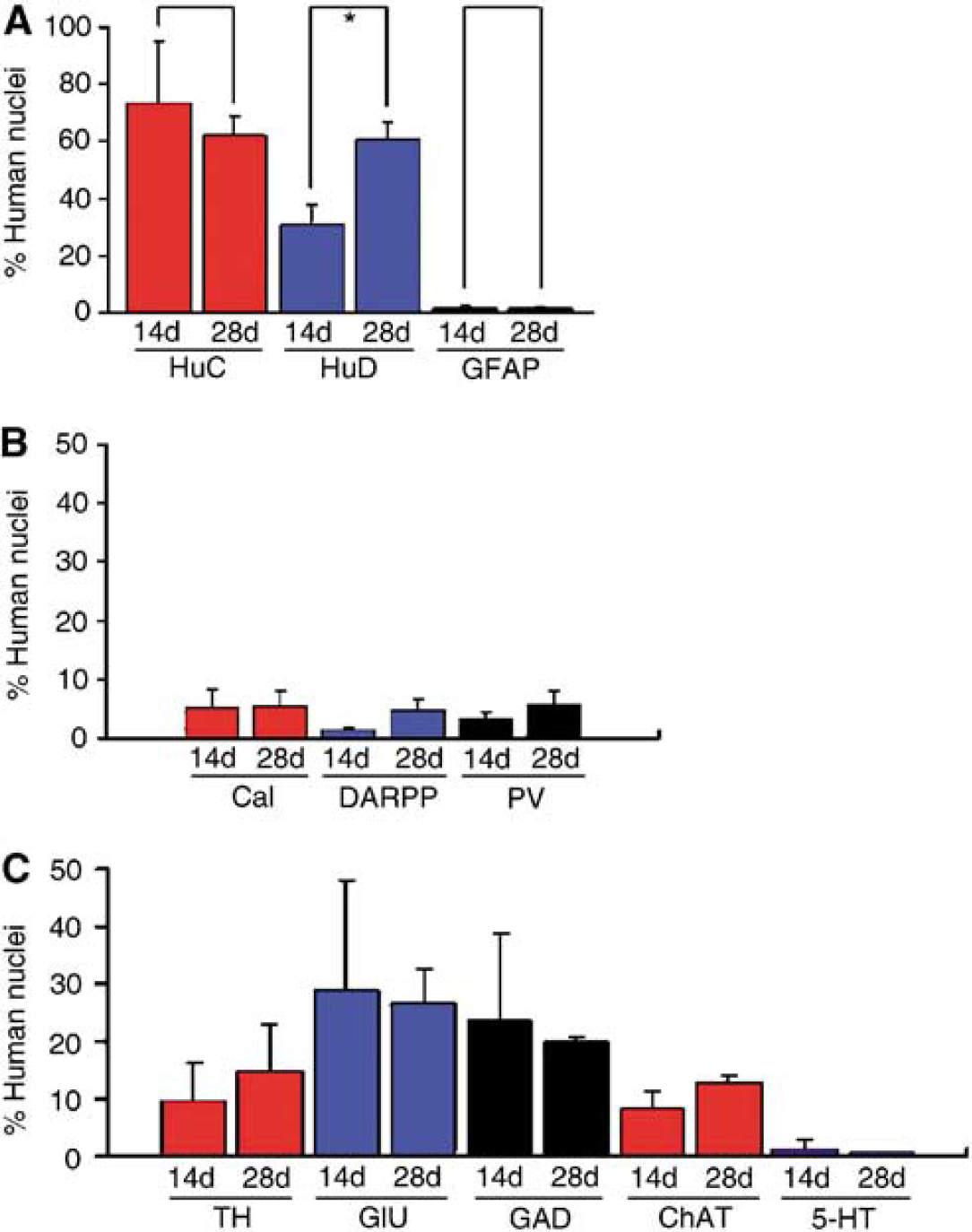
The proportions of cells in the ES-derived graft expressing differentiated neural and glial (
Neuronal Connections in Xenografts of Embryonic Stem-Derived Neural Progenitors
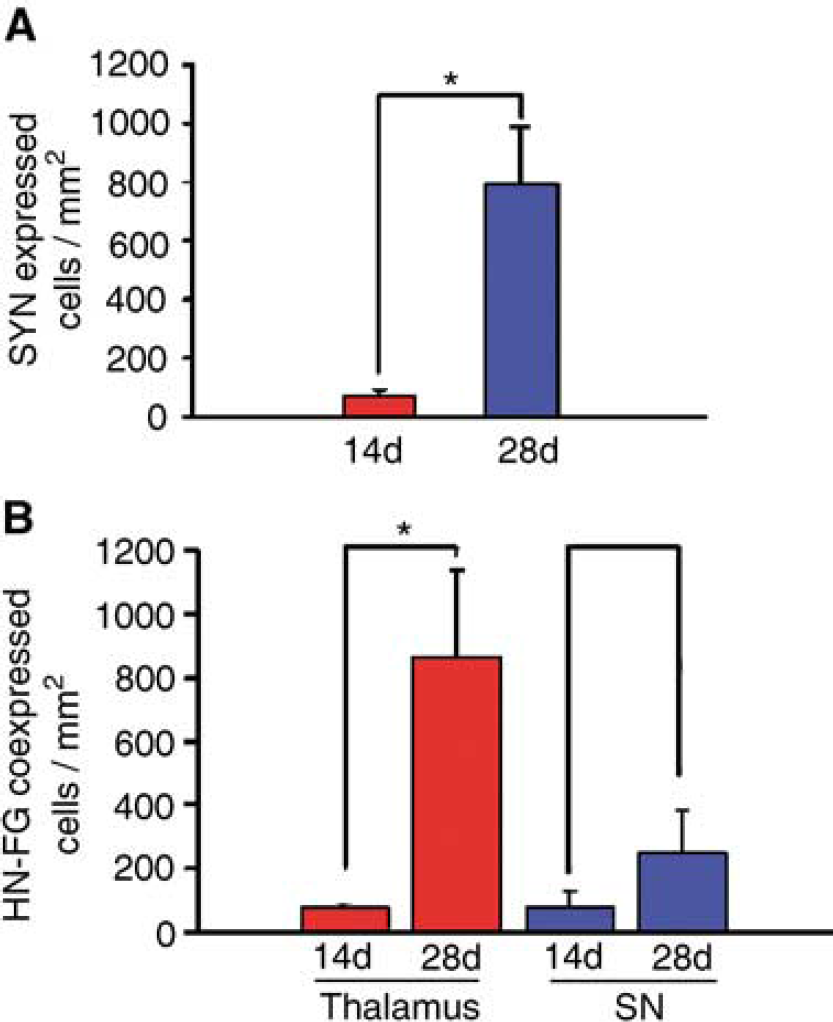
Finally, we investigated neuronal network formation in the grafts by analyzing the expression of a presynaptic vesicle-related marker, synaptophysin, and the subfunctional connection between the host and the graft by a retrograde labeling study. At 14 days after transplantation, a few synaptophysin-positive cells were detected (75.2 ± 16.8 cells/mm2, Figures 5A and 6A), but by 28 days posttransplantation, an over 10 times larger number of these cells (791.8 ± 191.4 cells/mm2) were expressed in the graft (Figures 5B, 5C and 6A). In the mice injected into their substantia nigra with FG, there were 80.5 ± 50.4 FG-positive cells/mm2 at 14 days and 252.2 ± 130.4 cells/mm2 at 28 days after transplantation (Figure 6B). Moreover, at 28 days after transplantation, over 10 times more cells in the graft were labeled with FG (862.9 ± 275.4 cells/mm2) by stereotactic focal injection of FG into the anterior thalamus on the grafted side when compared with the number at 14 days after transplantation (75.9 ± 14.9 cells/mm2; Figures 5D to 5F and 6B).
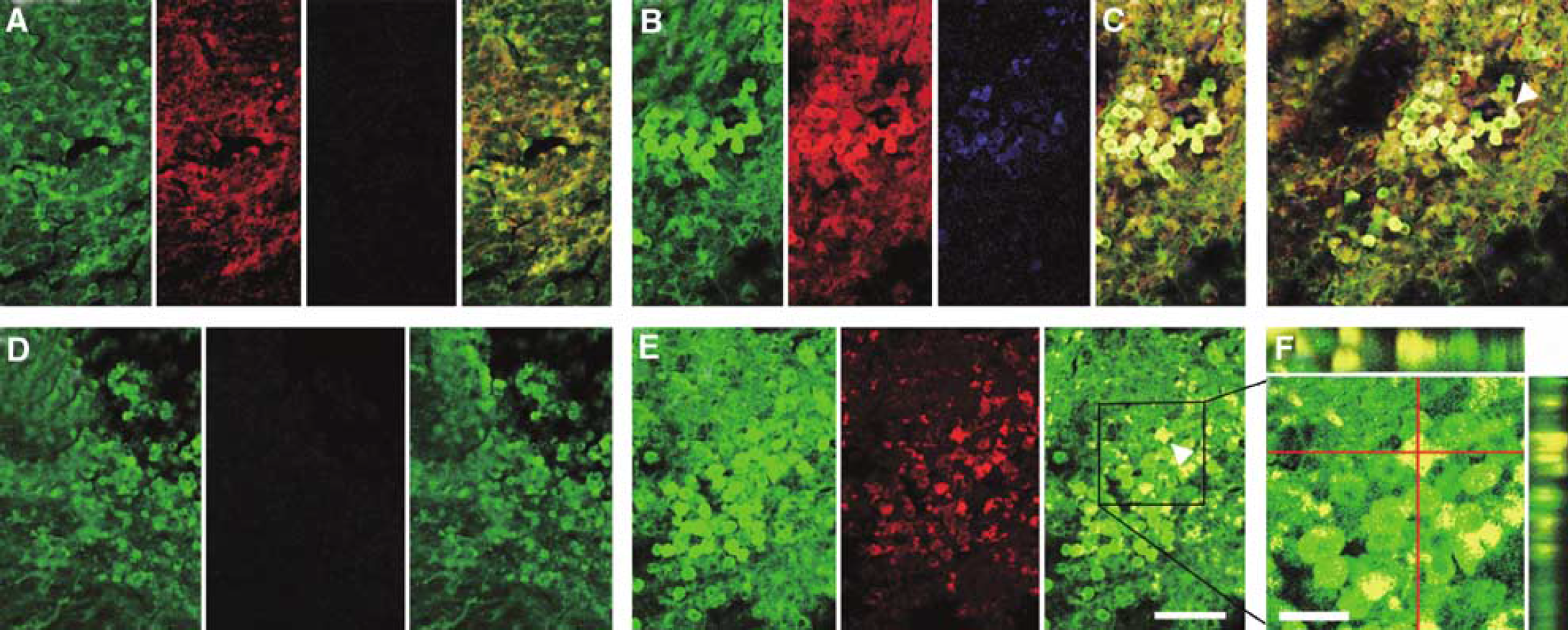
Expression of synaptic vesicle-related protein (
(
Discussion
In this study, we generated multipotent neuronal progenitors from monkey ES cells by the SDIA method and transplanted them into the postischemic mouse brain. These cells could survive in the acute postischemic environment, and differentiated into various types of neuronal cells. Furthermore, they could make synaptic connections with each other and interconnections between the host and the donor neurons, suggesting the possibility that the grafted ES-cell-derived neurons could form networks reflecting function in the host even though they were a xenograft. There are several studies in the literature on neuronal xenotransplantation, and some of them showed network formation between the host and the donor (Kawasaki et al, 2001; Sanders et al, 1998; Uchida et al, 2003).
In various types of brain injury, including traumatic brain injury, degenerative disease, intracerebral hemorrhage, hypoxic damage, and cerebral infarction, for a return to normal it is favorable that newly generated neurons are formed to repair networks and to recover lost functions. Various insults result in various grades of neuronal injury, and thus the quantity of neuronal replacement can be different in each case.
Now, there are several methods for replacement therapy, including enhancement of endogenous neurogenesis (Moskowitz and Lo, 2003; Yoshimura et al, 2001, 2003) and transplantation therapy. The approach of using endogenous neural stem cells has the advantage of being a noninvasive one. However, it remains unclear whether endogenous neurogenesis can provide a sufficient number of newborn neurons to recover the functions or not. However, ES cells, neural stem cells, and bone marrow-derived stem cells can differentiate into neuronal cells and can provide a large number of cells for transplantation therapy (Abe, 2000; Beck et al, 2003; Chen et al, 2001, 2003; Hoehn et al, 2002; Li et al, 2001, 2002; Modo et al, 2002a, b ; Morizane et al, 2002; Takagi et al, 2005a, b ; Zhao et al, 2003).
The use of neuronal transplantation for the treatment of neurological diseases was first tested by Bjorklund and Perlow in 1979 (Bjorklund et al, 1981; Kondziolka et al, 2002; Perlow et al, 1979). They transplanted dopamine-containing neurons into the rat striatum and recognized improvement of the functional deficits induced by damage to the nigrostriatal pathway. Since that time, various investigations using animal models have led to the human model, which has achieved various degrees of success (Kondziolka et al, 2000, 2002; Meltzer et al, 2001; Nelson et al, 2002). A wide variety of models of disease from degenerative diseases to trauma or stroke have been established, and the donor cells used for transplantation have ranged from fetal tissue to ES cells (Modo et al, 2002a, b; Nelson et al, 2002; Savitz et al, 2002). Successes in animal models have led to transplant trials in the human population. Clinical trials for Parkinson's disease, Huntington's chorea, spinal cord injury, and stroke were performed previously (Bjorklund and Lindvall, 2000; Kondziolka et al, 2002).
In this study, we used ES-cell-derived neuronal progenitors as the source of transplantation. The use of ES cells has an advantage because they can be expanded easily compared with neural stem cells. We previously reported the survival and functioning of mouse ES-cell-derived neuronal progenitors in animals with cerebral ischemia and in a primate model of Parkinson's disease (Morizane et al, 2002; Takagi et al, 2005b). As a continuation of those investigations, we used monkey ES cells as a donor and mouse focal ischemia as a recipient at first for the basis of further investigations.
By our observation, the grafted cells interestingly occupied only a small area in the sham control brains. On the contrary, the transplanted cells became widely distributed around the ischemic area in the ischemic brain. This result indicates that the fate of the graft is dependent on the host environment, as we reported previously (Takagi et al, 2005a). In the acute phase of cerebral ischemia, various cytokines, chemokines, and trophic factors are produced mainly by the microglia and astrocytes. Especially, 24 h after ischemia, when the ES-cell-derived neuronal progenitors were grafted in our study, cytokines such as interleukin 6, which is considered to protect neurons against stresses, are predominantly expressed. Moreover, some growth and trophic factors such as ciliary neurotrophic factor (CNTF), platelet-derived growth factor (PDGF), brain-derived neurotrophic factor (BDNF), glial-derived neurotrophic factor (GDNF), and neurotrophin 3 are considered to support the proliferation and differentiation of grafted cells (Abe, 2000; Moskowitz and Lo, 2003). We also confirmed the differentiation of ES-cell-derived neuronal progenitors in the postischemic condition of the brain. When a brain tissue is exposed to an ischemic event, various types of neurons and glial cells are lost. In this study, we showed that ES cells could supply these cells to the ischemic brain.
In this study, we used the 30-mins MCA occlusion model for focal cerebral ischemia. A benefit of this model is that it is possible to conduct long-term observation, but this model has fewer neurological deficits for analyzing functions because the infarct area is limited to the lateral striatum. So, we analyzed synaptic connections of grafted cells by investigating the expression of synaptophysin and analyzed the integration between the graft and host cells by analyzing the intake of the retrograde tracer FG (Jinno and Kosaka, 2004), referred to as the nigra-striatum or thalamus-striatum network in the literature (Salin and Kachidian, 1998; Samuel et al., 1988).
In summary, we confirmed the survival and differentiation of monkey ES-cell-derived neuronal progenitors transplanted into the postischemic mouse brain and also confirmed the formation of a neuronal network by them. The SDIA method, which we used, has the strong activity for inducing neuronal differentiation by ES cells and is also applicable to human ES cells. Embryonic stem cells have a great potential for use in replacement therapy for the treatment of stroke.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
