Abstract
The relation between relative cerebral blood flow (relCBF), relative cerebral blood volume (relCBV), and mean transit time (MTT) changes was examined in 20 patients with acute cerebral ischemia (<6 h) using perfusion-weighted magnetic resonance imaging. Regions of interest (ROI) were selected on MTT maps covering the entire MTT abnormal cortical area. These ROIs were transferred to the relCBF and relCBV maps to analyze the relation between relCBF, relCBV, and MTT on a voxel-by-voxel basis. On the unaffected side, a tight coupling of relCBF and relCBV was found with little variation of MTT. In hypoperfused cortex, relCBV was increased at all investigated relCBF categories, and there was greater relCBV variability than on the unaffected side. Only a severe decrease of relCBF, to less than 0.3, in comparison with the unaffected side was associated with a reduction of relCBV less than 1.0. In contrast to the unaffected side, a power law function (relCBV = 2.283 × relCBF0.549) resulted in a better fit than using a linear function for the correlation of relCBF and relCBV. MTT ratios increased steadily with decreasing CBF values. In conclusion, there is a clear relation between different perfusion-weighted magnetic resonance imaging parameters in acute ischemia, reflecting both the degree of hemodynamic failure as well as compensatory mechanisms including vasodilation.
Acute stroke is characterized by a severe reduction of regional cerebral blood flow (CBF) in the ischemic core and a critical decrease of CBF in the penumbra. In the past, most studies focused on the detection of thresholds of CBF or other perfusion parameters, differentiating between irreversible tissue damage, critical hypoperfusion of viable regions, and oligemia (Hossmann, 1994; Baron, 1999; Heiss, 2000).
Relatively few studies examined the complex interaction between different perfusion parameters in acute cerebral ischemia. Most available data highlighting the interaction of different perfusion parameters are from studies using positron emission tomography (PET) in patients with cerebrovascular disease, particularly patients with chronic high-grade carotid artery disease. Mainly based on these studies of patients with chronic hemodynamic alteration, a two-stage model of hemodynamic impairment was proposed: stage I is characterized by autoregulatory vasodilation and stage II by reduced CBF and an increased oxygen extraction fraction (OEF) (Baron et al., 1981; Powers, 1991). According to this model, which was slightly modified recently (Derdeyn et al., 2002), reductions in CBF occur when autoregulatory mechanisms fail to compensate for the decrease in local cerebral perfusion pressure. An increase in the oxygen extraction fraction may then compensate for the decrease in CBF, before the integrity of cerebral tissue is at risk. In acute stroke, most PET studies analyzed CBF, the regional metabolic rate of oxygen, and the OEF (Wise et al., 1983; Hakim et al., 1987; Heiss et al., 1992; Marchal et al., 1996). Particularly in the few hyperacute studies (symptom onset <6 h), regional cerebral blood volume (CBV) was not routinely reported (Heiss et al., 1998; Heiss et al., 2001). Therefore, the relevance of autoregulatory vasodilation in acute stroke is not as well established as in chronic cerebrovascular disease.
Over the last years, perfusion-weighted magnetic resonance imaging (PWI) has become available in many centers and has already been extensively used in acute stroke patients (Baird and Warach, 1998; Neumann-Haefelin et al., 2000a), including patients with symptom onset less than 6 h previously (Schellinger et al., 2001; Parsons et al., 2002). With PWI, several parameters of perfusion can be determined, including relative CBF (relCBF), relative CBV (relCBV), and mean transit time (MTT). In contrast to PET, these parameters are analyzed only semiquantitatively by most investigators because of theoretical problems with absolute quantification techniques in acute stroke patients (Calamante et al., 2002). Changes of PWI parameters have been reported by several investigators (Schreiber et al., 1998; Neumann-Haefelin et al., 1999; Schlaug et al., 1999; Sorensen et al., 1999; Liu et al., 2000; Grandin et al., 2001; Thijs et al., 2002), but the interaction of these parameters has been addressed in only a few studies (Caramia et al., 1998; Hatazawa et al., 1999; Sakoh et al., 2000). Severe reductions in relCBF have been shown to be associated with progression to infarction, whereas relCBV was shown to be decreased or normal in the ischemic core and elevated in penumbral and oligemic regions (Schreiber et al., 1998; Hatazawa et al., 1999; Schlaug et al., 1999; Liu et al., 2000; Grandin et al., 2001). However, in most of the studies, relCBF and relCBV were assessed on a region-of-interest (ROI) basis rather than on a voxel-by-voxel approach, and little is known about the relation between these parameters and MTT. The purpose of the present study was, therefore, to systematically investigate the relation between relCBF, relCBV, and MTT as measured by PWI using a voxel-by-voxel approach in acute ischemic stroke patients.
MATERIALS AND METHODS
Patients
From our database of acute stroke patients examined with a standardized magnetic resonance imaging (MRI) protocol, 20 patients (13 men and 7 women) were selected with the following inclusion criteria: (1) symptom onset less than 6 h before the acute MRI study, (2) ischemic stroke in the territory of the middle cerebral artery, and (3) good quality PWI (few motion artifacts, sufficient signal drop at bolus arrival). Time of onset was determined by information obtained from the patients themselves or their relatives. The study was approved by the local ethics committee and informed consent was obtained from all patients.
Magnetic resonance imaging protocol
The MR measurements were performed on a 1.5-T echo-planar imaging–capable whole-body MR scanner (Siemens Magnetom Vision) equipped with a gradient overdrive with the standard head coil. In each patient, the MRI examination included diffusion-weighted imaging (DWI), PWI, magnetic resonance angiography, and conventional imaging.
Diffusion-weighted imaging was performed with a single-shot echo-planar imaging spin-echo sequence: echo time (TE), 123 milliseconds; flip angle, 90°; field of view, 230 × 230 mm; matrix, 128 × 128; slices 19; slice thickness, 6 mm; gap, 1.2 mm; and b = 1,000 with the diffusion gradient in one direction. For bolus-tracking PWI, we used a gradient-echo echo-planar imaging sequence (TE, 60.7 milliseconds; field of view, 240 × 240 mm; matrix 128 × 128; slice thickness, 5 mm; gap, 1 mm). Forty T2*-weighted images for each of the 12 slices at intervals of 2 seconds were obtained. The contrast agent (0.1-mmol/kg Gd-DTPA; Magnevist, Schering, Berlin, Germany) was injected into an antecubital vein using a power injector at a rate of 5 mL/s followed by a flush with 10 mL saline.
Three-dimensional time-of-flight angiography was performed with a repetition time, 35 milliseconds; TE, 6.6 milliseconds; flip angle, 20°; field of view, 142 × 190 mm; matrix, 200 × 512; slab thickness, 70 mm; number of partitions, 40; acquisition time, 3 minutes 45 seconds; and maximum intensity projection reconstruction.
Postprocessing and data analysis
For further analysis, all MR images were transferred to a Unix workstation (Sun Microsystems). Images were displayed and processed with the software package MRVision (MRVision Co., Winchester, MA, U.S.A.). Perfusion-weighted MRI raw images were processed on a pixel-by-pixel basis to generate maps of the time-to-peak, relative MTT, relCBF, and relCBV. For calculation of MTT, relCBF, and relCBV maps, we used the model-independent SVD method (nonparametric singular value decomposition deconvolution) described by Ostergaard et al. (1996a, b ). Relative CBF is determined by deconvolution of the tissue concentration-time curve with an arterial input function. We determined the shape of the arterial input function in each patient by manually choosing 5 to 10 pixels over the first two segments of the middle cerebral artery of the unaffected hemisphere, showing an early and large decrease in signal intensity after the contrast injection. Relative CBV was defined as the area under the concentration-time curve between two time points (t1 and t2), which were determined for each patient separately. t1 was chosen from the arterial input function as a time point one or two images (2 to 4 seconds) before the arterial arrival of the contrast agent, and t2 was the time point 20 images later (40 seconds), when the signal had again completely or almost completely returned to baseline (including hypoperfused areas). Mean transit time was then calculated from these measurements as the ratio of relCBV/relCBF. Since we were only interested in evaluating relative changes of CBF and CBV in ischemic tissue as compared with normal tissue, no calibration procedure was used for absolute quantification of CBF and CBV.
Diffusion-weighted imaging and time-to-peak lesion volumes were measured as previously described (Neumann-Haefelin et al., 1999). Briefly, the areas of hyperintensity on DWI images were manually traced on each slice, summed, and multiplied with the slice thickness and interslice gap to obtain the DWI lesion volume. The volume of tissue with a time-to-peak delay of ≥2 seconds was determined as a measure of the total perfusion abnormality (Neumann-Haefelin et al., 2000b).
For the analysis of the interactions between relCBF, relCBV, and MTT, identical axial slices at the ventricular level were selected. Since MTT is highly sensitive to alterations of perfusion and provides good contrast between normal and hypoperfused areas, MTT maps were chosen to place an ROI covering the entire hemodynamically disturbed cortical area. The ROI was then mirrored to the contralateral, unaffected hemisphere. By registering the signal intensities for relCBF, relCBV, and MTT at identical slices using the same ROI, we obtained relCBF, relCBV, and MTT values for each voxel. Hemispheric ratios were calculated for relCBF, relCBV, and MTT, dividing each voxel of the affected cortex by the median ROI value of the unaffected contralateral cortex. Figure 1 illustrates the perfusion-weighted maps of one patient and the placing of the ROI.
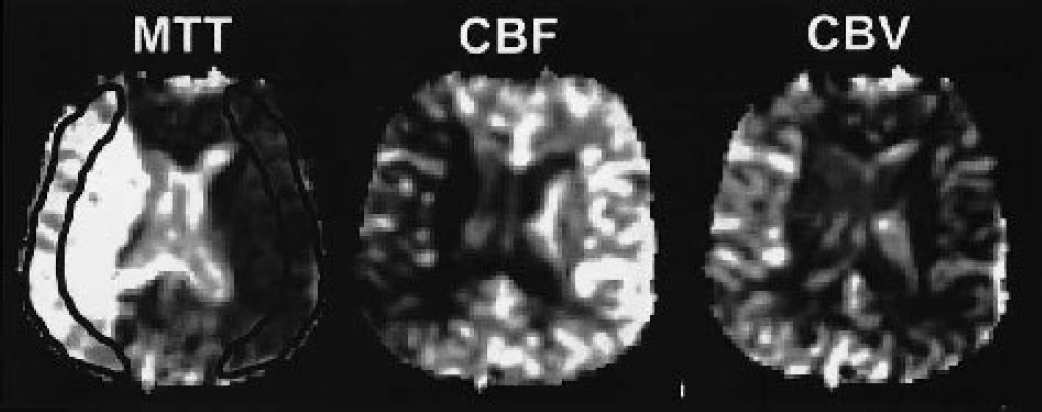
Mean transit time (MTT), relative cerebral blood flow (relCBF), and relative cerebral blood volume (relCBV) maps of a single patient. The MTT map depicts an extensive slowing of the bolus passage in the left hemisphere. The region of interest (ROI) was placed according to the hyperintense cortical areas on MTT, mirrored and adjusted to the contralateral hemisphere. Afterward, the ROIs were placed on identical regions of relCBF and relCBV maps.
In total, the data pool consisted of 20,071 pixels (ischemic and contralateral cortex). On average, each patient (n = 20) contributed 1,003 ± 248 (mean ± SD; range 456 to 1,394) pixels to the data set. Relative CBV and MTT data were categorized by the relCBF value with intervals of 0.1. To avoid unequal subject weighting, each patient's mean relCBV and MTT values were used to calculate the group mean relCBV and MTT value for each of the relCBF categories (Fig. 2). Individual mean relCBV and MTT values were excluded if <10 pixels contributed to a single category, which lead to exclusions at very low and very high relCBF categories (<0.2 and >1.1); this was done to exclude mean values based on too few pixels to be considered valid.
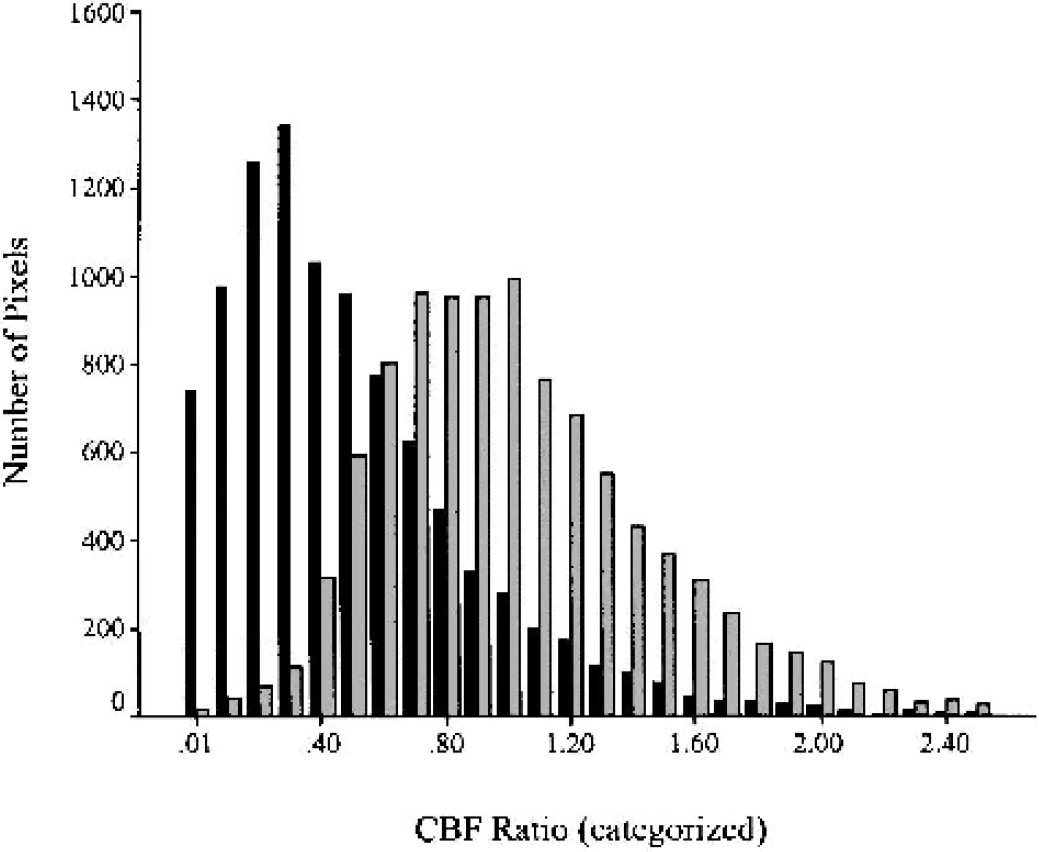
Distribution of pixels in hypoperfused (black bars) and contralateral unaffected (gray bars) cortex according to relative cerebral blood flow (CBF) categories. Relative CBF categories >2.5 (including 88 pixels out of a total of 20,071 pixels) are not displayed.
Statistical analysis
For correlation analysis between relCBF and relCBV values, Spearman's test was used. Regression analysis was performed for the ischemic and contralateral regions to determine the best fitting function, assuming either a linear or a power law relation. Figures are limited to relCBF values ≤1.5, since higher values did not contribute substantially to the data pool (Fig. 2). Results are presented as mean ± SD unless indicated otherwise.
RESULTS
Table 1 summarizes the demographic and imaging data. Mean age was 66 years (range, 39 to 79) and imaging was performed on average 2.6 h (range, 1.2 to 5.8 h) after symptom onset. Average DWI lesion volume at initial MRI was 78 mL (range, 3 to 322 mL), which is relatively large and is explained by the high proportion of patients with proximal vessel occlusion.
Demographic data and imaging results
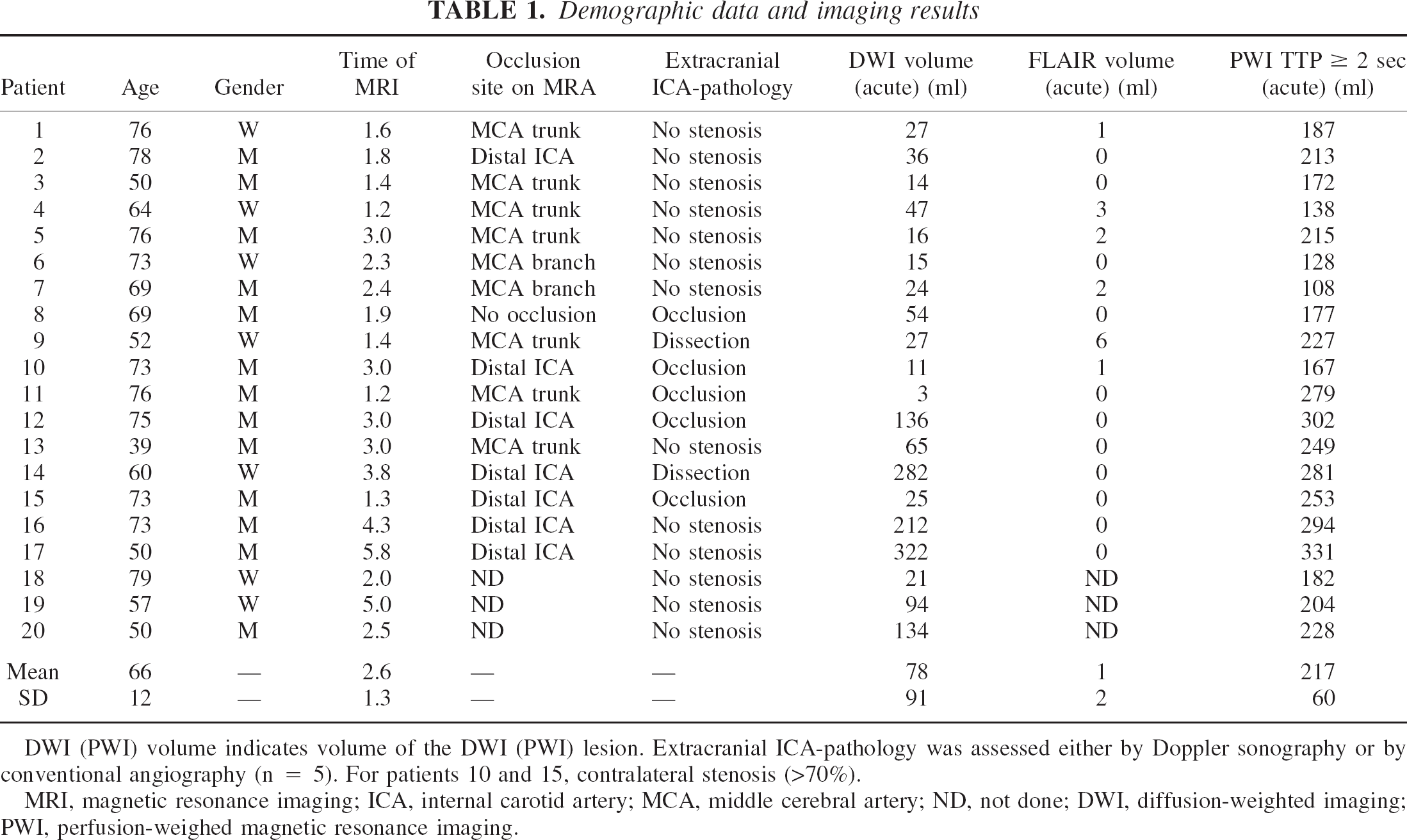
DWI (PWI) volume indicates volume of the DWI (PWI) lesion. Extracranial ICA-pathology was assessed either by Doppler sonography or by conventional angiography (n = 5). For patients 10 and 15, contralateral stenosis (>70%).
MRI, magnetic resonance imaging; ICA, internal carotid artery; MCA, middle cerebral artery; ND, not done; DWI, diffusion-weighted imaging; PWI, perfusion-weighed magnetic resonance imaging.
Average relCBF and relCBV values in the perfusion abnormal cortical region were 0.55 ± 0.25 (range, 0.07 to 1.09) and 1.49 ± 0.62 (range, 0.17 to 2.9), respectively. Mean relative MTT ± SD was 3.18 ± 0.98 (range, 1.83 to 5.11). Mean TTP delay ± SD in hypoperfused tissue was 9 seconds ± 5.16 (range, 0.82 to 23.98).
Figure 3 illustrates the relation between relCBF and relCBV in ischemic and contralateral normal cortex. In unaffected cortex, there was substantial variation of relCBF values in individual voxels (SD, 0.45). As expected, relCBV was approximately 1 at a CBF ratio of 1 and 0.5 at a CBF ratio of 0.5, respectively. The best fitting regression curve was determined using both a linear function (relCBV = 0.965 × relCBF + 0.122; r2 = 0.901; P < 0.001) as well as a power law formula (relCBV = 1.099 × relCBF0.943; r2 = 0.918; P < 0.001), indicating a nearly linear relation and a tight coupling between relCBF and relCBV in normal cortex (Table 2).
Linear and power law regression results for relCBF and relCBV in control (contralateral to ischemia) and ischemic tissue
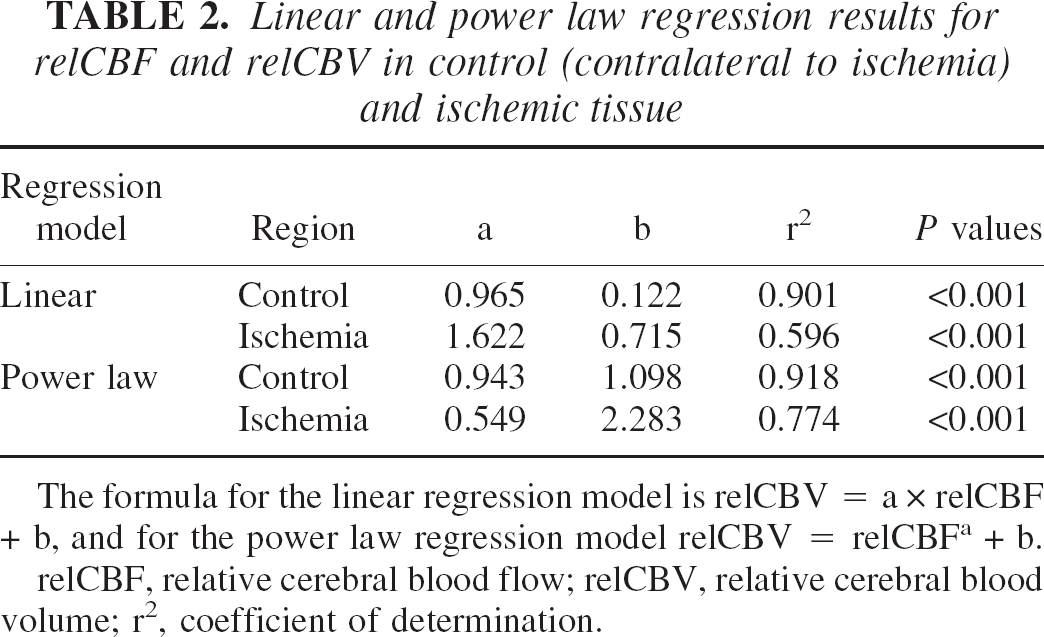
The formula for the linear regression model is relCBV = a × relCBF + b, and for the power law regression model relCBV = relCBFa + b.
relCBF, relative cerebral blood flow; relCBV, relative cerebral blood volume; r2, coefficient of determination.
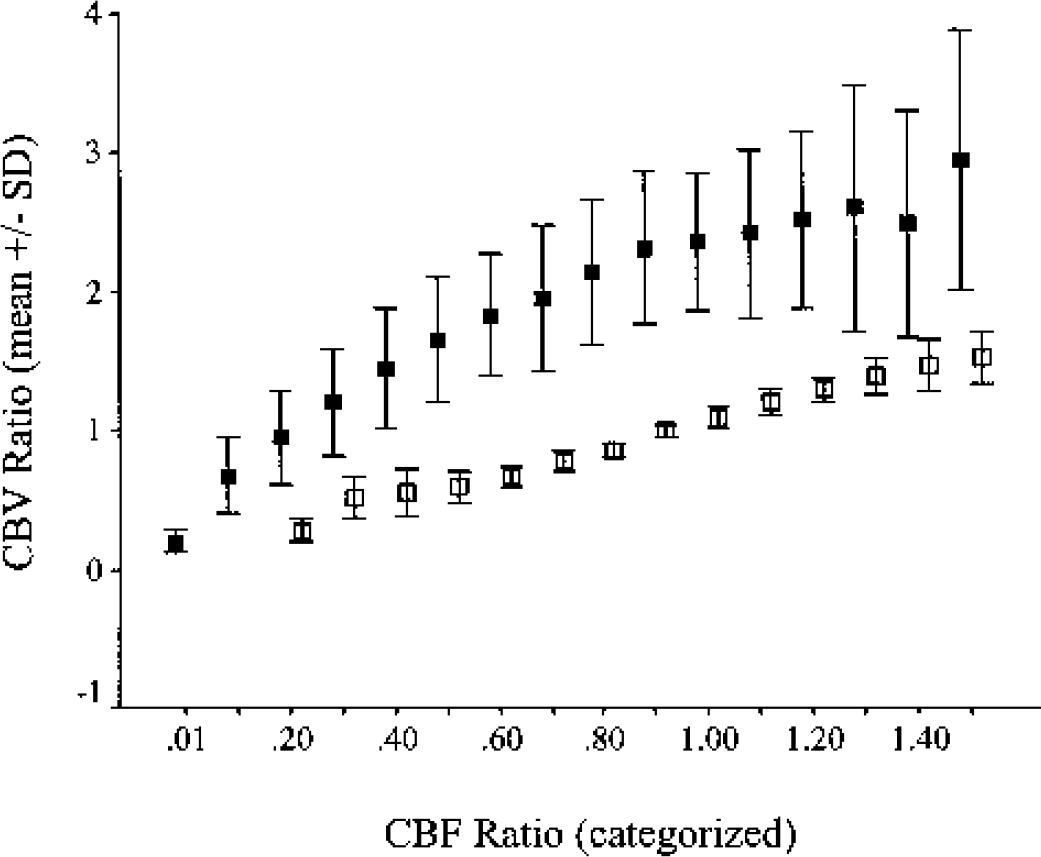
Relation between relative cerebral blood flow (CBF) and relative cerebral blood volume (CBV) in ischemic (▪) and contralateral (□) tissue. Relative CBF was categorized as described in Materials and Methods.
In ischemic cortex, relCBV was higher (and there were greater relCBV variations) at most relCBF categories as compared with normal cortex. In perfusion abnormal regions with apparently normal relCBF (ratio = 1.0), CBV ratio was 2.37 ± 0.5 (mean ± SD). Even in regions with relCBF values as low as 0.3, relCBV was still in the normal range (ratio ≈1). Only a drop of relCBF below 30% of normal median relCBF was associated with a reduction of relCBV below 1.0. The relation between relCBF and relCBV showed nonlinear characteristics, with a steeper slope at lower relCBF values than at higher relCBF values. This is also evident when comparing the regression curves, which showed a better fit when using the power law relation (relCBV = 2.283 × relCBF0.549; r2 = 0.774; P < 0.001) than the linear regression (Table 2). A subgroup analysis including the 15 patients without arteriosclerotic carotid disease revealed nearly identical results (data not shown in detail). Therefore, a relevant bias due to chronic preexisting perfusion abnormalities can be excluded.
In normal cortical tissue, relative MTT was constant over a broad range (0.5 to 1.5) of relCBF values indicating that a decline in relCBF was accompanied by a proportional reduction in relCBV (Fig. 4). In ischemic tissue, MTT was generally prolonged; the MTT ratio was 2.27 ± 0.43 (mean ± SD) at apparently normal relCBF values (ratio ≈1.0) and increased to 3.05 ± 0.81 and 4.23 ± 1.48 (mean ± SD) at a CBF ratio of 0.50 and 0.10, respectively. A decline in relCBF below 0.10 was accompanied by a drop in MTT ratio to 2.97 ± 1.42 (mean ± SD).
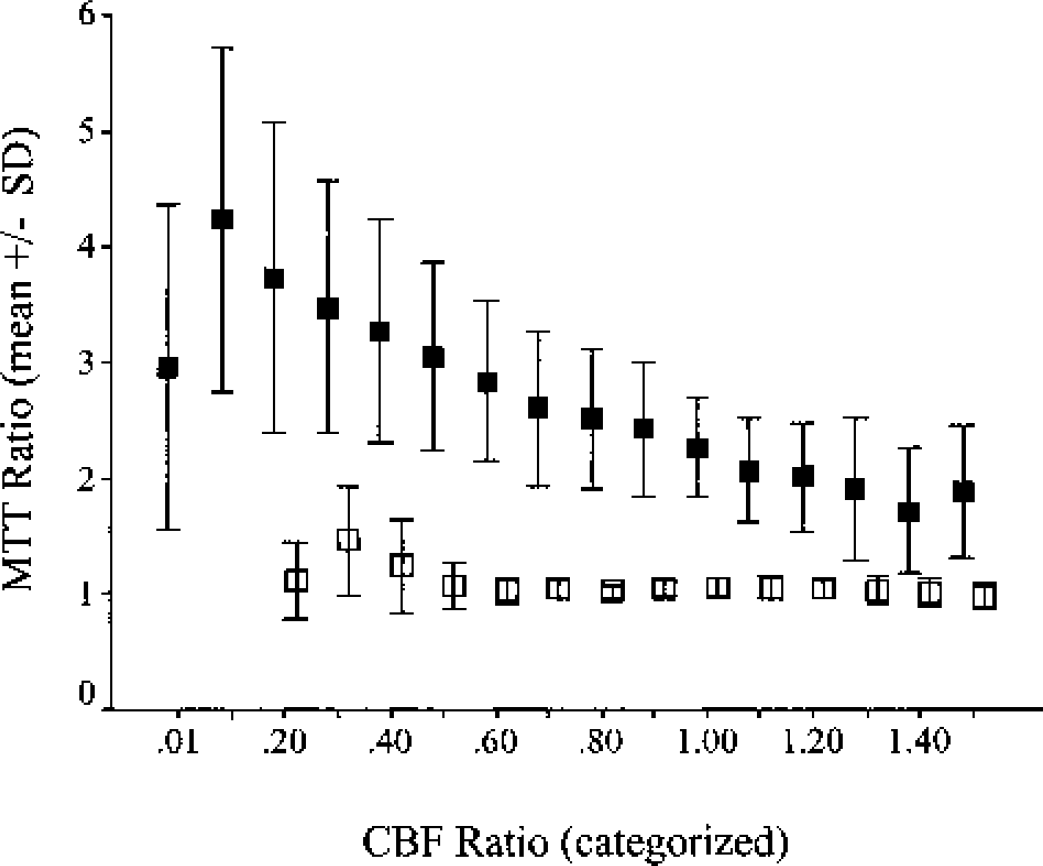
Mean transit time (MTT) ratio in ischemic (▪) and contralateral (□) tissue plotted against the corresponding relative (CBF) value. MTT ratio is indicated as mean ± SD of five or more patients contributing with at least 10 data points to the particular CBF ratio category.
DISCUSSION
In this acute stroke MRI study, the interactions between relCBF, relCBV, and MTT were analyzed for both hypoperfused as well as unaffected contralateral tissue.
Relative CBF and relative CBV in unaffected tissue
The large range of relCBF values found in unaffected contralateral tissue most likely reflects regional differences in metabolic demand. This assumption is supported by PET studies showing that CBF is tightly linked to regional metabolism (Raichle et al., 1976; Powers, 1991; Silva et al., 1999; Li et al., 2000). For relCBV, we found a similar variability in unaffected cortical tissue and a tight coupling to relCBF. Accordingly, no relevant MTT changes were observed in unaffected cortex as a function of relCBF. The stability of MTT, which was calculated as MTT = relCBV/relCBF, indicates that relCBV changed proportionally with relCBF. These findings are in line with the results reported by Sette et al. (1989), who found a positive linear correlation between CBF and CBV in five healthy subjects using PET, whereas MTT remained stable over a broad CBF range.
Relative CBF and relative CBV in hemodynamically compromised tissue
In hypoperfused (i.e., MTT-abnormal) tissue, relCBV was increased compared with the unaffected side at all investigated relCBF categories and the relation between relCBF and relCBV showed nonlinear characteristics, with a steeper slope at lower compared with higher relCBF values. The relation between these PWI parameters has not been studied in detail in humans. However, our results are in general agreement with experimental data obtained in a pig MCA occlusion model (Sakoh et al., 2000) showing a steeper slope of the correlation curve in the lower relCBF range. In this study, which also included PET measurements for comparison, Sakoh et al. (2000) found elevated relCBV values at a relCBF >70% (as compared with >40% in our study) and decreased relCBV at relCBF values <60%. Similarly, in a human study correlating relCBV (as measured by MRI) with relCBF measured by single-photon emission computed tomography (SPECT) in 9 stroke patients, Hatazawa et al. (1999) found a nonlinear relation between relCBF and relCBV; elevated relCBV values (>100%) were observed, with relative relCBF values >60% and a decline of relCBV below normal at a CBF ratio below 0.6.
Several factors may explain the lower relCBF range (<0.3) associated with low relCBV (<1.0) measured in our study. First, in the study by Hatazawa et al., relCBF was measured with SPECT, which tends to yield higher relCBF values than PWI (Liu et al., 2000). Second, the time lag between SPECT and MRI was up to 2.5 h. Third, the calculation of relCBF and relCBV maps from PWI raw data are not completely standardized yet, and minor differences between investigators may certainly explain some variability. Finally, as shown in our study, the relation between relCBF and relCBV is different in hemodynamically compromised and unaffected tissue; therefore, compromised and unaffected tissue must be analyzed strictly separately to avoid underestimation of relCBV values in affected regions.
The finding of reduced relCBV in areas with severely impaired relCBF (ratio < 0.3) is presently not well understood. In theory, the severe relCBF reduction should be associated with maximum vasodilation and consequently elevated relCBV. Sette et al. (1989), using PET, found a similar reduction of CBV in patients with cerebrovascular disease in areas with severe reductions of CBF and metabolic failure as indicated by an increased OEF and a reduced cerebral metabolic rate for oxygen. They speculated that the metabolic depression associated with a loss of function contributed via an ischemia-induced release of vasoactive substances to a reduction of CBV, thus counterbalancing autoregulatory vasodilation. In addition, there is experimental evidence that slight increases in extracellular potassium induce vasodilation, whereas higher concentrations (i.e., caused by cell death) can lead to vasoconstriction (Kuschinsky and Wahl, 1978). However, the decrease in relCBV in the low relCBF range may also be a consequence of microvascular failure associated with low-flow conditions and may thus be unrelated to metabolic depression. In cats with induced severe systemic hypotension, an increasing number of capillaries with either intermittent flow or no flow was found with increasing degrees of hypotension (MacKenzie et al., 1979). In focal cerebral ischemia, factors contributing to capillary obstruction (and thus possibly contributing to low relCBV) include plugging of capillaries by leukocytes, endothelial swelling, formation of microvilli, platelet aggregation, and external compression of capillaries because of edema or perivascular astrocyte swelling (Hudetz, 1997). In summary, there are several factors that may explain the relCBV reduction in low-flow regions, but the relevance of these factors remains unknown.
Relative CBF and mean transit time in normal and hypoperfused tissue
The reduction of relCBF in hypoperfused tissue was accompanied by a progressive prolongation of MTT in our study, reaching its maximum value at a relCBF ratio of 0.1. This is in striking contrast to the situation in contralateral normal tissue, where no variations in MTT occurred over a broad relCBF range (0.5 to 1.5). Increases in MTT reflect relative changes in the relCBV/relCBF ratio and occur in areas where relCBF is more severely decreased than relCBV, or where relCBV is relatively more increased compared with reduced, normal, or even elevated relCBF (Thijs et al., 2001). Mean transit time has also been shown to be a sensitive marker of reductions in cerebral perfusion pressure. Because there is little regional variation of MTT in unaffected regions, the relatively marked relCBF differences in these areas are unlikely to be related to regional differences in perfusion pressure. However, the prolongation of MTT in hemodynamically impaired regions indicates substantial changes in perfusion pressure, which is most likely a consequence of vessel obstruction in the feeding arteries.
Two aspects of the MTT/relCBF relation (Fig. 4) require further discussion. First, MTT values were higher in hypoperfused than in normal tissue for all relCBF categories, including “normal” relCBF categories. This is certainly in part because we used MTT maps to select the ROIs for the analysis of “hypoperfused” tissue. In addition, however, we believe that this finding reflects vasodilation in areas with reduced cerebral perfusion pressure, increased transit times, but still normal relCBF. Most likely, these regions are located in the periphery of the hypoperfused regions and are not as much at risk as regions with significant flow reductions, which is in line with previous reports (Caramia et al., 1998; Schlaug et al., 1999; Neumann-Haefelin et al., 2000b). Second, the progressive increase in MTT that is associated with decreasing relCBF values was also associated with decreases in relCBV (when compared with values obtained for “normal” relCBF categories; Fig. 3). This decrease in relCBV partially counterbalances the effect of relCBF decreases on MTT. Assuming that the relCBV decrease was at least in part the effect of microvascular failure, this indicates that the transit time could be even longer with fully accessible microvessels.
Limitations
Our study has several limitations. First, the precision of CBF and CBV measurements in acute stroke using PWI is still under discussion (Calamante et al., 2002). Bolus delay and dispersion caused by vessel obstruction and collateral flow may lead to underestimation of CBF and overestimation of MTT (Ostergaard et al., 1998; Calamante et al., 2000). Second, MTT maps were used for selecting ROIs of hypoperfused tissue because differentiation of normal from hemodynamically compromised tissue is easier on these maps than on CBF or CBV maps (Schlaug et al., 1999; Sorensen et al., 1999; Liu et al., 2000; Grandin et al., 2001). However, the differentiation between gray and white matter is slightly more difficult; therefore, we cannot exclude that the ROIs comprised some adjacent subcortical white matter. Third, for relative quantification of perfusion parameters, we used the contralateral hemisphere as reference tissue. However, contralateral perfusion may not be normal because of transhemispheric diaschisis (or contralateral carotid artery disease), leading to a CBF reduction and consequently to an underestimation of ipsilateral hypoperfusion in relative measurements (Andrews, 1991; Meyer et al., 1993). Finally, because the study was limited to a single PWI examination during the hyperacute stage of cerebral ischemia, we cannot exclude that the relation between different PWI parameters may change at later time points.
CONCLUSIONS
In summary, our study focused on interactions between relCBF, relCBV, and MTT in acute stroke patients with symptom onset less than 6 h previously. In hypoperfused tissue, relCBV was increased compared with the unaffected side, and the relation between relCBF and relCBV showed nonlinear characteristics, with a steeper slope in the lower relCBF range. Only in regions with severely impaired relCBF, relCBV dropped to below normal, most likely reflecting microvascular failure or ischemia induced metabolic vasoconstriction.
