Abstract
Vasodilator effects of glutamate in the cerebral circulation are, in part, mediated by carbon monoxide (CO), which is formed from heme via the heme oxygenase (HO) pathway. The hypothesis addressed was that glutamate receptors (GluRs) in cerebral microvascular endothelium are functionally linked to HO. Using a radioligand binding and immunoblotting, GluRs were characterized in cerebral microvascular endothelial cells (CMVEC) from newborn pigs. High-affinity (80 nmol/L) reversible binding of [H]Glutamate ([3H]Glu) was detected in CMVEC membranes. The N-methyl-D-aspartate (NMDA) receptor ligands—NMDA, quinolinic acid, (±)1-aminocyclopentane-cis-1,3-dicarboxylic acid (cis-ACPD), AP5, 4C3HPG, and CPP—and the (RS)-α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA)/kainate receptor ligands—AMPA, kainic acid, quisqualic acid, DNQX, and CNQX—displaced 20% to 30% of bound [3H]Glu in CMVEC membranes. Metabotropic GluRs antagonists (4CPG, PHCC, and CPPG) did not displace bound [3H]Glu. L-Aspartate, an agonist of GluRs and glutamate transporters, displaced 80% or more of bound [3H]Glu. Ionotropic (NR1 and GluR1) and metabotropic (mGluR1α) GluRs were detected in CMVEC by immunoblotting. Glutamate, aspartate, cis-ACPD, AMPA, (RS)-2-amino-(3-hydroxy-5-tert-butylisoxazol-4-yl)propanoic acid (ATPA), and kainate (10−5 mol/L) increased HO-directed CO formation by isolated cerebral microvessels and by cultured CMVEC. These data in newborn pigs suggest that CMVEC express ionotropic GluRs that are functionally linked to HO. GluR-mediated increases in CO formation by vascular endothelium may result in increase in cerebral blood flow.
Keywords
L-Glutamic acid (glutamate), an excitatory neurotransmitter, is a vasodilator in cerebral circulation in vivo (Bari et al., 1997; Busija and Leffler, 1989; Faraci and Breese, 1993; Faraci et al., 1994; Fergus and Lee, 1997; Meng et al., 1995). Release of glutamate from presynaptic neurons may result in increasing blood flow to the affected brain areas. Neurons are primary cellular targets for glutamate in the brain. Blood vessels in the brain are extensively innervated, and neuronal influences are important in the regulation of cerebral blood flow (Busija, 1993). Neuronal activation in response to excitatory neurotransmitters may affect brain microvessels via neurally derived vasorelaxant factors, including nitric oxide (NO) (Faraci and Breese, 1993; Faraci et al., 1994; Meng et al., 1995; Snyder and Ferris, 2000; Yang and Iadecola, 1996) and carbon monoxide (CO) (Baranano and Snyder, 2001). In cerebral circulation of newborn pigs, CO is a potent vasodilator (Leffler et al., 1999), whereas NO is of less importance (Zuckerman et al., 1996). Inhibition of heme oxygenase (HO), a key enzyme in CO formation, reduced cerebral vasodilation in response to glutamate (Robinson et al., 2002) and glutamatergic seizures (Pourcyrous et al., 2002) in newborn pigs. Neurons express HO (Baranano and Snyder, 2001; Dore et al., 1999, 2000; Zakhary et al., 1996), indicating that cerebral vasodilation to glutamate in vivo can be mediated, in part, by neurally derived CO. Recent evidence, however, suggests that cerebral microvasculature can be also directly targeted by glutamate. Cerebral microvessels from newborn pigs highly express HO (Leffler et al., 1999). Furthermore, glutamate stimulates CO production by cerebral microvascular endothelial cells (CMVEC) from newborn pigs (Parfenova et al., 2001). These data suggest that cerebral vascular endothelium may express glutamate receptors (GluRs) functionally linked to HO.
Effects of glutamate involve activation of GluRs. GluRs in the central nervous system are divided into the ligand-gated ion channel receptors (ionotropic GluRs) and the G protein—coupled receptors (metabotropic GluRs) (Monaghan et al., 1989; Nakanishi, 1992). Ionotropic GluRs are classified into three groups based on their pharmacology and structural properties. N-methyl-D-aspartate (NMDA)-type receptors are heteromeric complexes comprised of NR1 subunit combined with one or more NR2 subunits (Nakanishi, 1992). In the brain, NMDA receptors were detected in cerebral cortex and most other structures. Non-NMDA ionotropic GluRs include (RS)-)-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA; GluR1–GluR4) and kainate receptors (GluR5–GluR7) that have similar pharmacologic properties. Multiple metabolic GluRs (mGluR1–mGluR5) that are coupled to diverse second messenger systems, including phosphoinositides, phospholipase D, and cyclic AMP (cAMP), are also widely distributed in the brain (Schoepp and Conn, 1993).
Cerebral blood vessels express receptors for dopamine (Bacic et al., 1991), acetylcholine (Elhusseiny et al., 1999), and serotonin (Cohen et al., 1999) and, therefore, can be directly targeted by these neurotransmitters. However, the question whether cerebral microvessels express receptors for glutamate remains highly controversial. Several studies concluded that GluRs are not expressed in cerebral microvasculature from several animal species (Beart et al., 1988; Morley et al., 1998; Wending et al., 1996). Other authors, however, did identify GluRs in CMVEC from rat and human brain (Collard et al., 2002; Krizbai et al., 1998; Sharp et al., 2002).
In the present report, we addressed the hypothesis that GluRs expressed in cerebral vascular endothelium of newborn pigs are functionally linked to HO. We characterized GluRs in CMVEC using several approaches. In a pharmacologic approach, a competitive radioligand binding technique was used to test the ability of distinct GluR ligands to displace [3H]Glutamate from the high-affinity binding sites. Glutamate receptor expression was also detected by immunoblotting. As a positive control in these studies, we used piglet brain cortex membranes, which are known to express diverse GluRs. Finally, we investigated the ability of GluR agonists to increase CO production by freshly isolated cerebral microvessels and by cultured CMVEC.
MATERIALS AND METHODS
Isolation of cerebral microvessels and endothelial cell cultures
Brain cortex was obtained from ketamine- and acepromazine—anesthetized newborn pigs (1 to 5 days of age). Cerebral microvessels (60 to 300 μm) were collected by filtration of the brain cortex homogenate through 300- and 60-μm nylon mesh screens consecutively. Cerebral microvascular endothelial cells were cultured on Matrigel-coated plates in Dulbecco's Modified Eagle Medium with 20% fetal bovine serum (Parfenova et al., 2001). Radioligand binding experiments were performed on confluent quiescent cells in first passage. To achieve quiescence, cells were exposed to a glutamine-free low-serum medium (0.1% fetal bovine serum) for 15 to 20 hours before the experiment. For detection of CO production, CMVEC were cultured on collagen-coated dextran microcarrier beads (Sigma, St. Louis, MO, U.S.A.). Primary cultures of CMVEC at confluence were reseeded onto microcarrier beads (5 × 103 cell/cm3) into Bell-Flo spinner flasks (Waters, 1996). Beads were stirred with intermissions overnight to promote attachment and then maintained at 60 rpm continuously for 2 days to reach confluence.
Tissue and cell fractioning
Brain cortex was homogenized in 20 mmol/L N-(2-hydroxyethyl)piperazine-N'-2-ethane-sulfonic acid (HEPES) and 10 mmol/L EDTA (pH 7.4) with protease—phosphatase inhibitors (homogenizing buffer). Cerebral microvascular endothelial cells were scraped into ice-cold phosphate-buffered saline, collected by centrifugation, and disrupted by sonication (3 pulses for 20 seconds each) in homogenizing buffer. Cell homogenate was centrifuged at 3,000g for 5 minutes at 4°C, and the P1 pellet (crude membrane fraction) was collected. The supernatant was centrifuged at 17,000g for 30 minutes, and the P2 pellet (microsomal fraction) was collected. The pellets resuspended in 20 mmol/L HEPES, 2 mmol/L MgCl2, and 2 mmol/L CaCl2 (pH 7.4, assay buffer) were used immediately in ligand-binding experiments. The amount of protein was determined by Bradford assay.
Radioligand binding
Ligand binding assays were performed using L-[G-3H]-glutamic acid ([3H]Glu, 42 Ci/mmol). Membrane suspension (30 to 50 μg protein) was incubated on ice for 1 hour with 80 nmol/L [3H]Glu in the absence and presence of 1 mmol/L glutamate (total and nonspecific binding, respectively) in the assay buffer (total volume, 100 μL). For competitive binding, the ligands of distinct GluRs (10−4 mol/L) were added to the media. To characterize NMDA receptors, we used the receptor agonists, NMDA, (±)1-aminocyclopentane-cis-1,3-dicarboxylic acid (cis-ACPD), quinolinic acid, and NMDA receptor antagonists, D(−)-2-amino-5-phosphonopentanoic acid (D-AP5), R-4-carboxy-3-hydroxyphenylglycine (4C3HPG), 3-((R0–2-carboxypiperazin-4-yl)-propyl-1-phosphonic acid) (CPP), and LY235959. To characterize AMPA/kainate receptors, we used the receptor agonists, AMPA, (2S,3S,4R)-carboxy-4-(1-methylethenyl)-3-pyrrolidineacetic acid (kainic acid), and (L)-(+)-α-amino-3,5-dioxo-1,2,4-oxadiazolidine-2-aminoethane (L-quisqualic acid). 6-Cyano-7-nitroquinoxaline-2,3-dione disodium (CNQX) and 6,7-dinitroquinoxaline-2,3-dione (DNQX) were used as AMPA/kainate receptor antagonists. (S)-4-carboxyphenylglycine (4CPG), N-phenyl-7-(hydroxyimino)cyclopropa[b]chromen-1a-carboxamide (PHCCC), and (RS)-α-cyclopropyl-4-phosphonophenylglycine (CPPG) were used as antagonists of metabolic GluRs. The reaction was terminated by dilution with ice-cold buffer A and rapid filtration through glass microfiber (GF/C) filters (Whatman Paper, Gaithersburg, MD, U.S.A.). The filters were washed three times with 5 mL of ice-cold buffer. The filters were air dried, placed in vials containing Opti Fluor cocktail (Packard Instruments, Meriden, CT, U.S.A.), and counted in a beta scintillation counter.
Detection of carbon monoxide production by gas chromatography—mass spectrometry
Cerebral microvascular endothelial cells on microcarrier beads or cerebral microvessels were placed into vials containing artificial cerebrospinal fluid (in mmol/L: 3.0 KCl, 1.5 MgCl2, 1.5 CaCl2, 132 NaCl, 6.6 urea, 3.7 dextrose, and 24.6 NaHCO3) equilibrated with 6% CO2/6% O2 to pH 7.4. Glutamate and GluR agonists (10−7 to 0−4 mol/L) were added to the vials. In the experiments using the HO inhibitor Zn protoporphyrin (ZnPP; 2 · 10−5 mol/L), the microvessels were pretreated for 30 minutes. Isotopically labeled 13CO was used as an internal standard for measurements by gas chromatography—mass spectrometry (Robinson et al., 2002). The vials were sealed and incubated for 30 minutes at 37°C. To terminate incubation, the samples were placed in hot water (75°C). Gas chromatography—mass spectrometry analysis of the headspace gas was performed by Hewlett-Packard 5970 ion detector interfaced to a Hewlett Packard 5890A gas chromatograph. The separation of CO was carried out on a Varian-5A Molesieve capillary column (30 m, 0.32 mm internal diameter; Varian, Walnut Creek, CA, U.S.A.) with a linear temperature gradient from 35°C to 65°C. Ions at the mass-to-charge ratio (m/z) 28 and 29 corresponding to 12CO and 13CO were recorded via selective ion monitoring. The amount of CO was calculated from the ratio of the peak areas of m/z 28 and m/z 29. The results were normalized to protein detected by the Lowry assay.
Western immunoblotting for glutamine receptors
Membrane fractions of CMVEC and brain cortex were solubilized in Laemmli sample buffer (100°C, 10 minutes); proteins (20 to 50 μg protein/lane) were separated by 9% sodium dodecyl sulfate—polyacrylamide gel electrophoresis, transferred to nitrocellulose membranes, and nonspecific binding was blocked with 5% bovine serum albumin–0.1% Tween 20 (Parfenova et al., 2001). The membranes were probed with polyclonal antibodies raised against (1) C-terminus peptide of rat NR1 (at 1:1,000 dilution; Sigma), (2) C-terminus peptide of rat GluR1 (at 1:1,000 dilution; Chemicon, Temecula, CA, U.S.A.), or (3) peptide of rat mGluR1α (1:2,000; Chemicon), followed by peroxidase-conjugated donkey anti—rabbit IgG (1:10,000; Jackson Immunoresearch, West Grove, PA, U.S.A.). Bands were visualized with the Renaissance chemiluminescence kit (NEN Life Science Products, Boston, MA, U.S.A.).
Materials
Cell culture reagents were purchased from Life Technologies (Gaithersburg, MD, U.S.A.), and Amersham Pharmacia Biotech, Piscataway, NJ, U.S.A. Fetal bovine serum was from Hyclone (South Logan, UT, U.S.A.). Matrigel was from Becton Dickinson (Bedford, MA, U.S.A.). Glutamate receptor ligands were from Tocris Cookson (Ellisville, MO, U.S.A.). 13CO was from Isotech Inc., Miamisburg, OH, U.S.A. L-[G-3H]Glutamic acid was purchased from Amersham Pharmacia Biotech. Zn protoporphyrin was from Porphyrin products (Logan, UT, U.S.A.). All other reagents were from Sigma (St. Louis, MO, U.S.A.).
Data analysis
Data are presented as means ± SEM of absolute values. Statistical significance was assessed using Student's t test. A level of P < 0.05 was considered significant.
RESULTS
High-affinity [3H]Glutamate binding with cerebral microvascular endothelial cells and brain cortex
We compared high-affinity binding of [3H]Glu (free ligand, 80 nmol/L) to the membrane fractions from CMVEC and from the brain cortex. In endothelial membranes, specific binding accounted for over 80% of total [3H]Glu binding (Fig. 1). In CMVEC, the amount of specific glutamate binding in fractions P1 and P2 was 6.9 ± 1.5 and 3.5 ± 0.9 pmol/mg protein, respectively. In the brain cortex, [3H]Glu-specific binding accounted for approximately 50% of total of [binding (Fig. 1). The amount of high-affinity [3H]Glu binding sites in the brain was lower than in CMVEC.
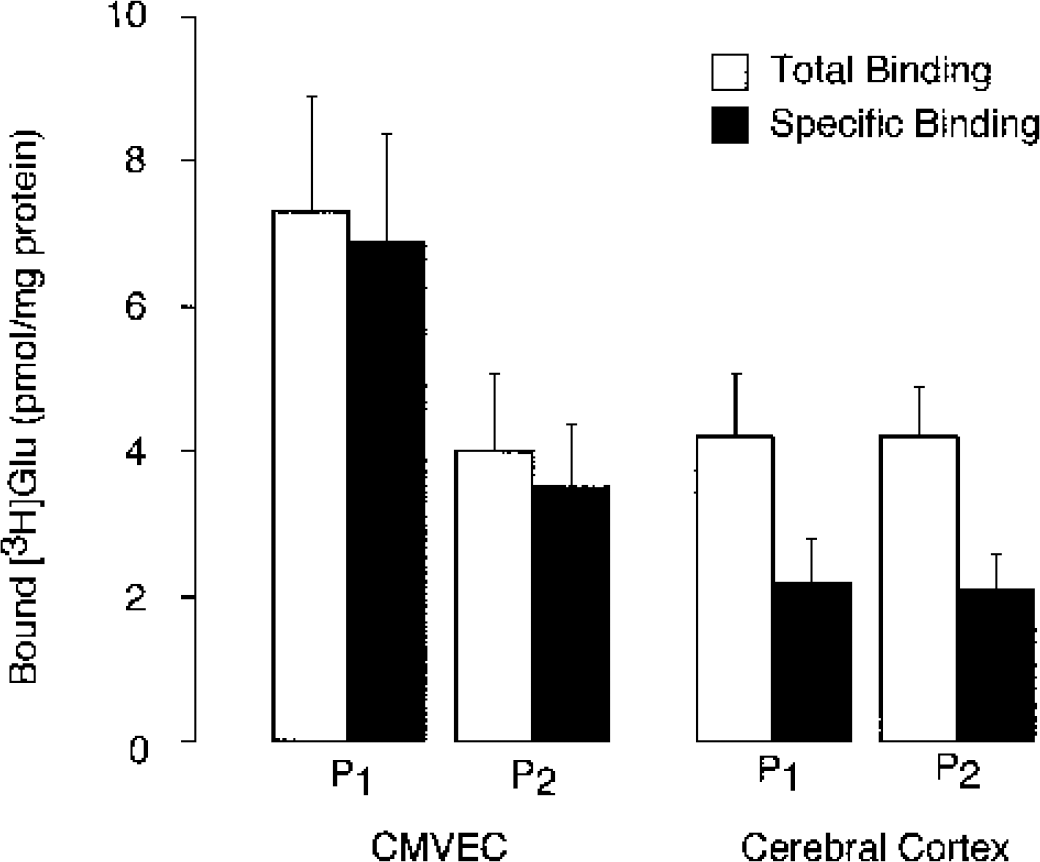
L-[3H]Glutamic acid binding with membrane fractions from cerebral microvascular endothelial cells (CMVEC) and brain cortex from newborn pigs. Crude membrane fraction P1 and microsomal fraction P2 were obtained from confluent CMVEC. Membranes (50 μg protein) were incubated with 80 nmol/L [3H]Glutamic acid ([3H]Glu) in the absence or presence of 1 mmol/L unlabeled glutamate (total and nonspecific binding, respectively). Specific binding was calculated as the difference between total and nonspecific binding. Data represent average of six independent experiments. Values are means ± SE.
Displacement of [3H]Glutamate with glutamate receptor ligands
We investigated the ability of ligands of distinct GluRs to competitively displace [3H]Glu from the high-affinity binding sites (free ligand, 80 nmol/L). To characterize NMDA receptors in a competitive binding assay, we used the receptor agonists, NMDA, quinolinic acid, and cis-ACPD as well as antagonists, D-AP5, 4C3HPG, CPP, and LY235959. Most of NMDA receptor ligands (100 μmol/L), with the exception of LY235959, decreased the amount of [3H]Glu bound to the CMVEC membranes (both P1 and P2 membrane fractions) by 20% to 30% (Figs. 2A and 2B). The ability of NMDA receptor ligands to displace bound [3H]Glu suggests the presence of NMDA-type GluRs in CMVEC. NMDA receptors identified in P1 and P2 fractions account for 20% to 30% of high-affinity [3H]Glu binding to CMVEC. AMPA/kainate receptor agonists, AMPA, kainic acid, and quisqualic acid, as well as AMPA/kainate receptor antagonists DNQX and CNQX (100 μmol/L) decreased binding of [3H]Glu with the CMVEC membrane fraction P2 by 20% to 30% (Fig. 2B). In membrane fraction P1, only quisqualic acid displaced [3H]Glu, whereas other ligands were not effective (Fig. 2A). These results suggest that CMVEC express AMPA/kainate receptors that are localized mainly in P2 membrane fraction; these receptors account for 20% to 30% of high-affinity [3H]Glu binding sites. Antagonists of metabotropic GluRs, 4CPG, PHCCC, and CPPG (100 μmol/L) did not displace [3H]Glu in either P1 or P2 fractions of CMVEC (Fig. 2A and 2B).
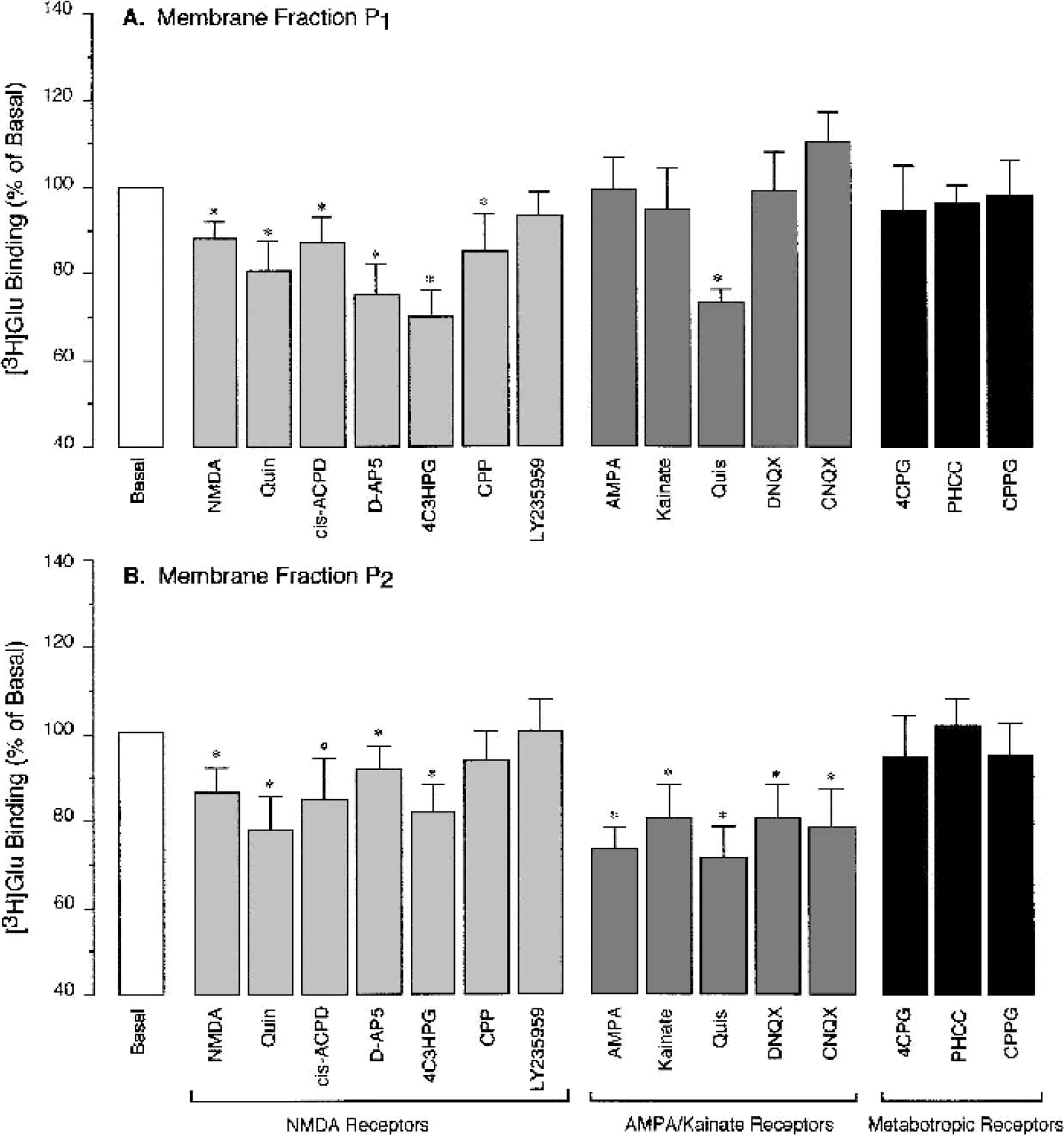
Displacement of L-[3H]Glutamic acid from the high-affinity binding sites in cerebral microvascular endothelial cells (CMVEC) from newborn pigs. Crude membrane fraction P1
We compared pharmacologic profiles of GluRs in CMVEC with those in the brain cortex. In the brain, NMDA, quinolinic acid, D-AP5, and 4C3HPG (100 μmol/L) displaced 50% to 60% of specific binding of [3H]Glu from P1 and P2 membranes (Fig. 3A and 3B). AMPA, kainic acid, quisqualic acid, DNQX, and CNQX (100 μmol/L) displaced 50% or more of [3H]Gluspecific binding (most effectively, in P1 fraction). 4CPG, PHCC, and CPPG displaced 40% to 60% of [3H]Glu from P1 and P2 brain membrane fractions. Therefore, ionotropic (NMDA and AMPA/kainate subtypes) and metabotropic GluRs were detected in P1 and P2 membrane fractions from the brain cortex.
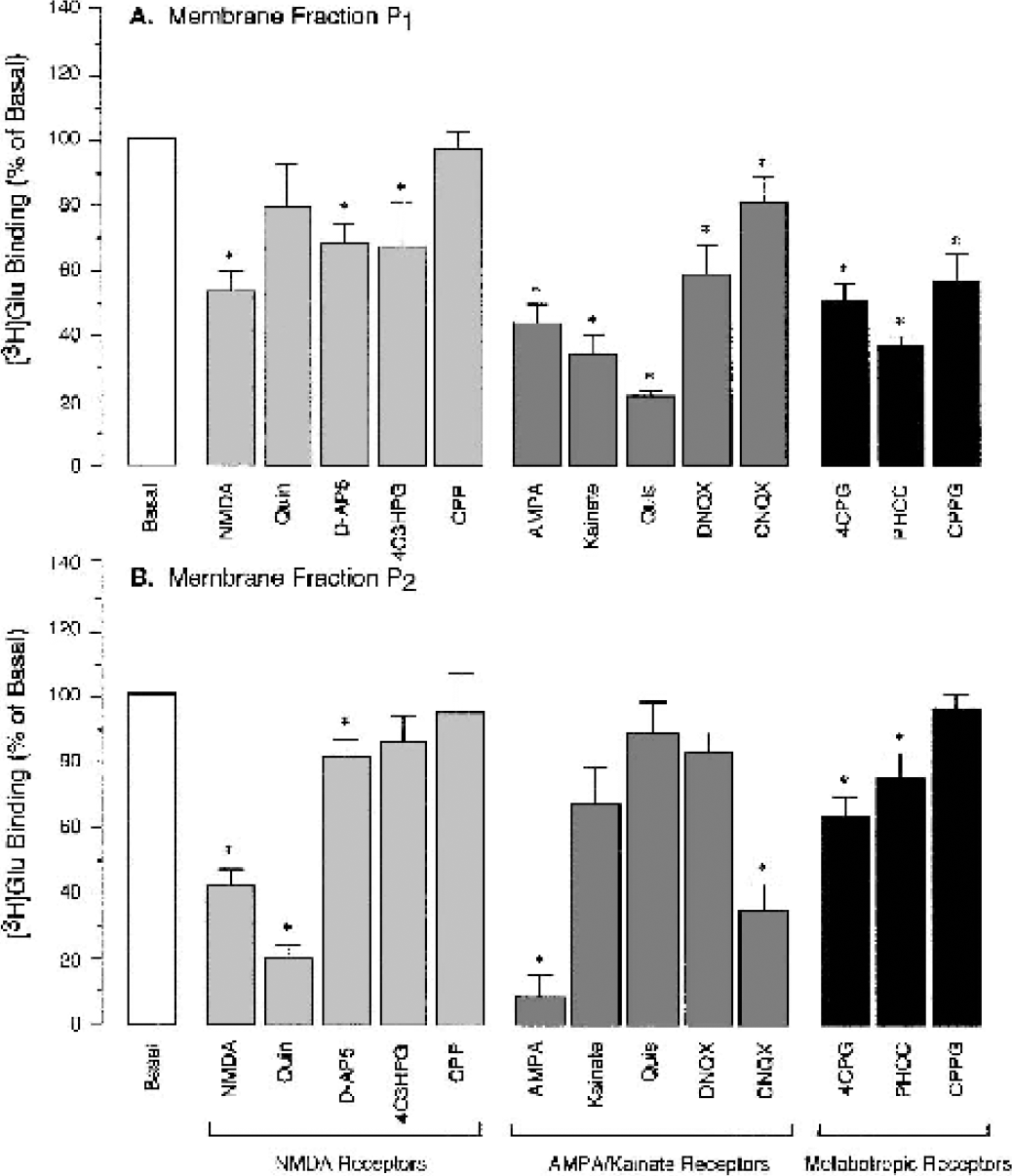
Displacement of L-[3H]Glutamic acid from the high-affinity binding sites in the newborn pig brain cortex. Crude membrane fraction P1
Displacement of [3H]Glutamate from high-affinity binding sites with L-aspartate
In CMVEC, aspartate was as effective as glutamate in displacing [3H]Glu from high-affinity binding sites. Based on dose-response displacement curves (Figs. 4A and 4B), KI for aspartate (5 and 6 μmol/L for P1 and P2 membranes, respectively) was only two times higher than KI for glutamate (2 and 3 μmol/L for P1 and P2 membranes, respectively). At concentration 100 μmol/L or more, aspartate as well as glutamate displaced approximately 80% of bound [3H]Glu from P1 and P2 membranes (Figs. 4A and 4B). Because other GluR ligands displaced less than 30% of bound [3H]Glu, the greater effectiveness of aspartate may indicate that it also binds to glutamate transporter sites in piglet CMVEC.
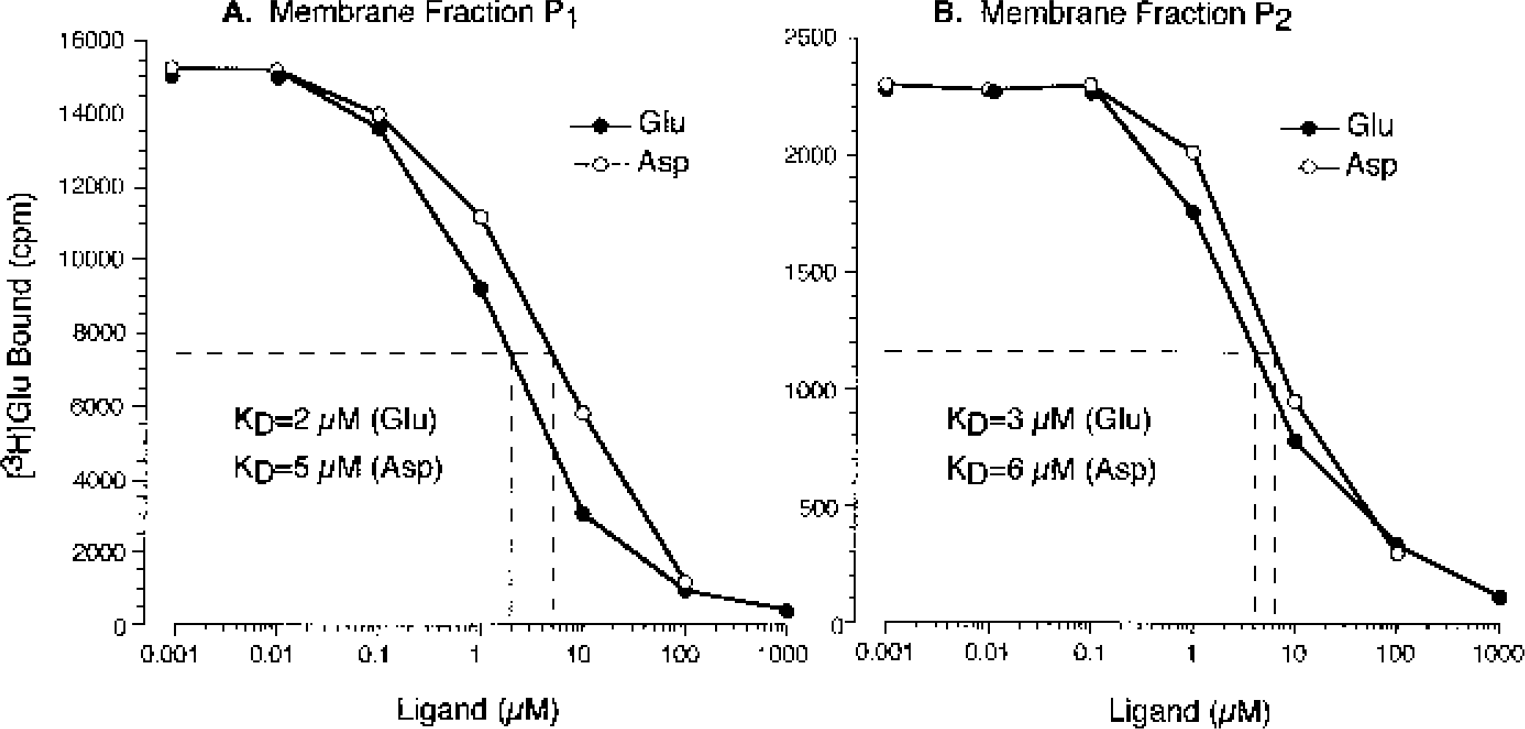
Displacement of L-[3H]Glutamic acid from high-affinity binding sites in cerebral microvascular endothelial cell membranes by L-aspartic acid (Asp) and L-glutamic acid (Glu). Crude membrane fraction P1
Immunodetection of distinct GluRs in cerebral microvascular endothelial cells and in the brain cortex
We tested several antibodies on their ability to recognize distinct GluRs in CMVEC from newborn pigs. As a positive control, we used newborn pig brain cortex (Fig. 5). Antibody against rat NR1 (116 kd) recognized several bands in the newborn pig brain and CMVEC: a 170-kd band and two major low-molecular-weight bands (36 to 38 kd). The antibody against rat GluR1 (108 kd) recognized one major approximately 110-kd band in the brain and CMVEC. The antibody against rat mGluR1α (160 kd) recognized a 160-kd band in the brain and CMVEC (Fig. 5).
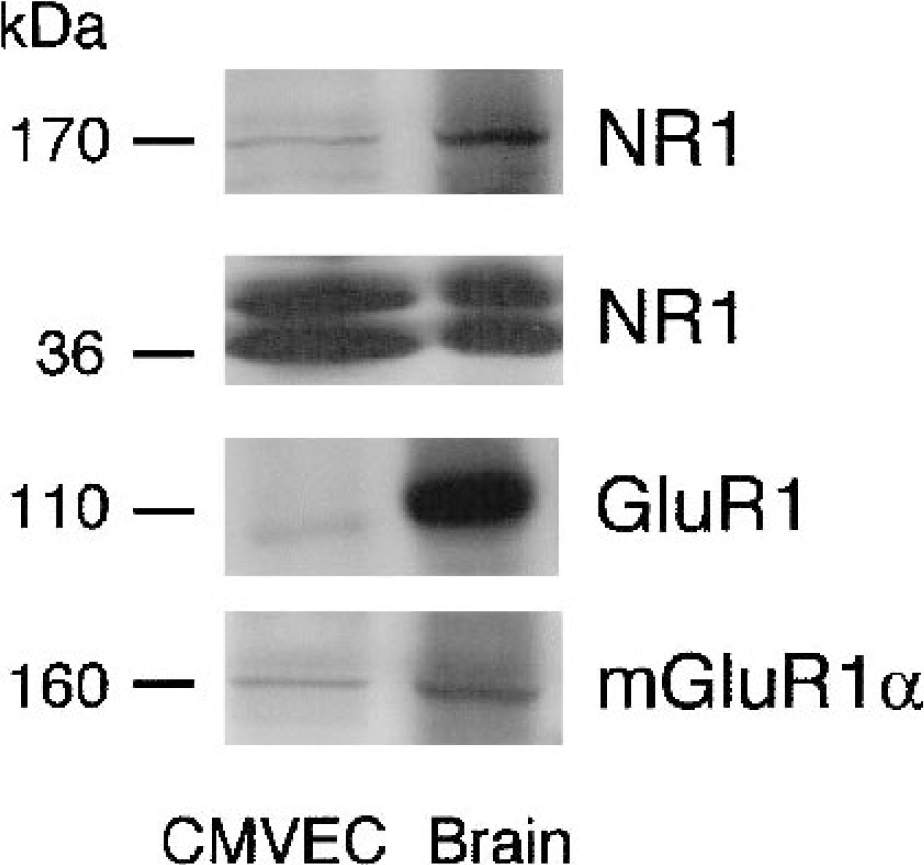
Western immunoblots for glutamate receptor (GluR) detection in cerebral microvascular endothelial cells and in the brain cortex of newborn pigs. Microsomal fraction (P2) (20 μg protein/lane) was resolved by 8% sodium dodecyl sulfate—polyacrylamide gel electrophoresis and transferred to nitrocellulose membrane. Membranes were probed for NR1 (1:1,000), GluR1 (1:2,000), and mGluR1α (1:2,000). Blot is representative of three independent experiments. CMVEC, cerebral microvascular endothelial cells. NR1 and GluR1, ionotropic GluRs; mGluR1α, metabotropic GluR.
Effects of glutamate and glutamate receptor agonists on carbon monoxide production
We tested the hypothesis that activation of endothelial GluRs may increase HO activity in cerebral microvasculature. Cerebral microvessels freshly isolated from newborn pig brain cortex produce CO from endogenous heme under basal conditions (Fig. 6). Glutamate (10−5 to 10−3 mol/L), ATPA (10−6 to 10−4 mol/L), and AMPA (10−6 to 10−4 mol/L) stimulated CO production by microvessels twofold to threefold (Fig. 5). In microvessels pretreated with ZnPP (2 × 10−5 mol/L) for 30 minutes, the stimulation of CO production by GluR agonists was completely abolished (Fig. 6). Cerebral microvascular endothelial cells from newborn pigs also produce CO under basal conditions from endogenous heme (Fig. 7). Glutamate (10−5 mol/L) increased CO production from threefold to fourfold (Fig. 7). Agonists of ionotropic GluRs, cis-ACPD, aspartate, AMPA, and kainate (10−5 mol/L) increased CO production by CMVEC twofold to fourfold (Fig. 7). trans-ACPD, an mGluR agonist, did not alter endothelial CO production (Fig. 7).
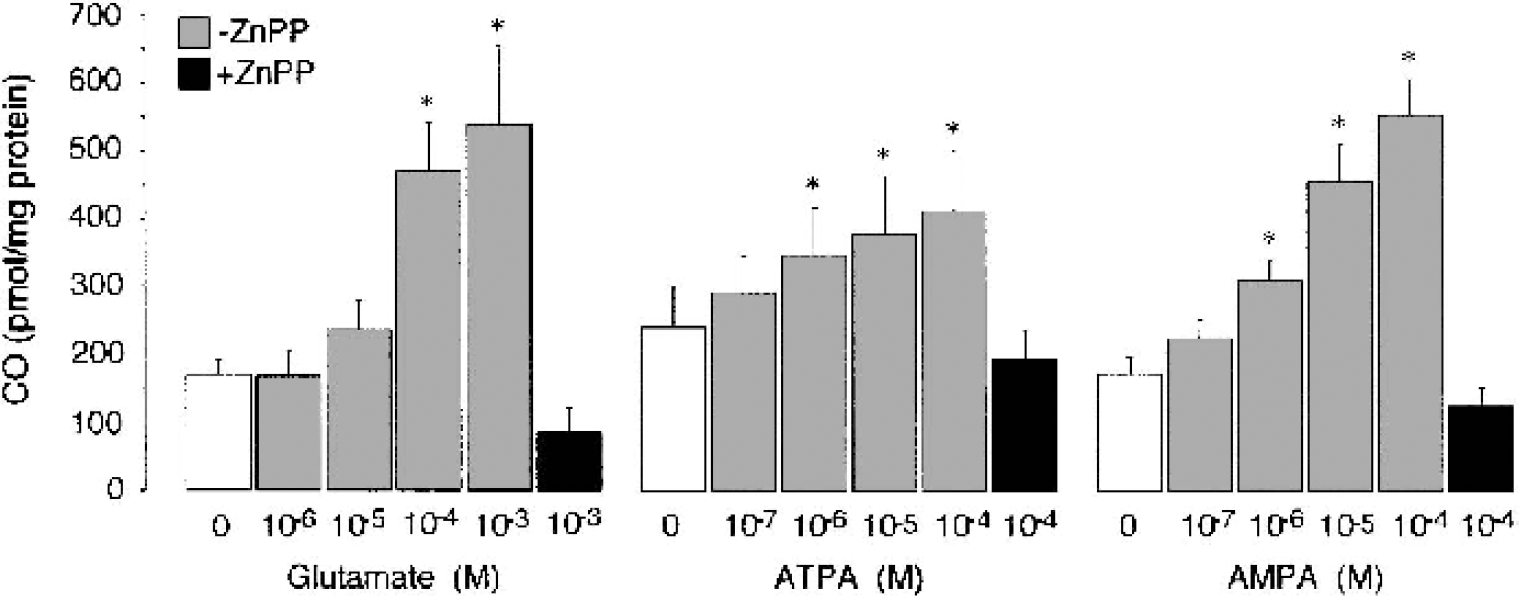
Dose-dependent effects of glutamate and glutamate receptor agonists on carbon monoxide (CO) production by freshly isolated cerebral microvessels. Cerebral microvessels (200 μg protein) were incubated with glutamate (10−6 to 10−3 mol/L), ATPA (10−7 to 10−4 mol/L), and AMPA (10−7 to 10−4 mol/L) in the absence or presence of Zn protoporphyrin (2 × 105 mol/L) for 1 hour at 37°C. Carbon monoxide production was detected by gas chromatography—mass spectrography. Data are normalized to the protein amount. Data represent average of five independent experiments. Values are means ± SE. * P < 0.05 compared with the basal value. ATPA, (RS)-2-amino-(3-hydroxy-5-tert-butylisoxazol-4-yl)propanoic acid; AMPA, (RS)-)-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid; ZnPP, Zn protoporphyrin.
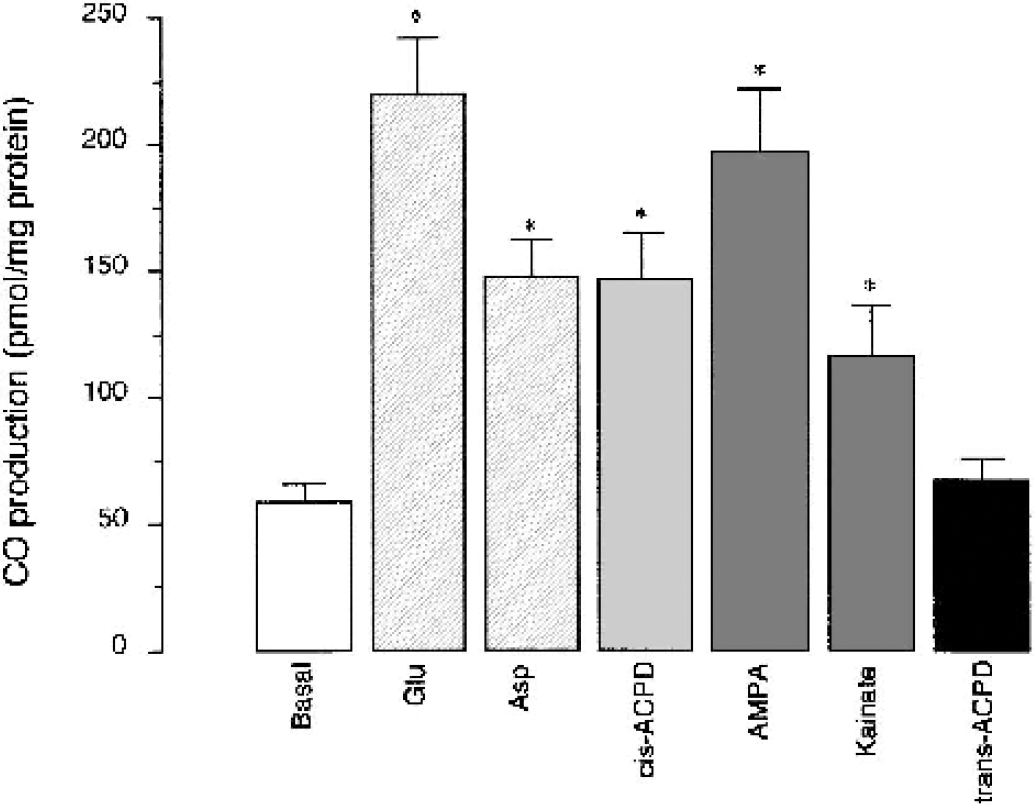
Effects of glutamate and glutamate receptor agonists on carbon monoxide (CO) production by cerebral microvascular endothelial cells (CMVEC). CMVEC from newborn pigs (200 μg protein) were incubated with glutamate (Glu, 10−5 mol/L), aspartate (Asp, 10−5 mol/L), cis-ACPD (10−5 mol/L), AMPA (10−5 mol/L), kainate (10−5 mol/L), or trans-ACPD (10−5 mol/L) for 1 hour at 37°C. Carbon monoxide production was detected in head space by gas chromatography—mass spectrography. Data are normalized to the protein amount. Data represent average of three independent experiments. Values are means ± SE. * P < 0.05 compared with the basal value. AMPA, (RS)-)-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid; cis-ACPD and trans-ACPD, (±)1-aminocyclopentane-cis-1,3-dicarboxylic acid and (±)1-aminocyclopentane-trans-1,3-dicarboxylic acid, respectively.
DISCUSSION
We present here evidence that endothelial cells from cerebral microvessels of newborn pigs express ionotropic GluRs functionally linked to CO production via the HO pathway. NMDA- and AMPA/kainate-type GluRs were identified by competitive radioligand binding, immunoblotting, and by the ability of the receptor agonists to increase CO production by isolated cerebral microvessels and cultured CMVEC. The implications of these novel findings are as follows: (1) cerebral microvessels can be directly targeted by excitatory neurotransmitters glutamate and aspartate via a GluR-mediated mechanism, (2) activation of ionotropic GluR increases CO production by vascular endothelium, and (3) endothelium-derived CO can contribute to CO-mediated responses of cerebral microvessels to excitatory neurotransmitters.
Are cerebral microvessels among targets for glutamate in the brain? In rat and human CMVEC, some investigators detected ionotropic and metabotropic GluRs as mRNA and protein (Collard et al., 2002; Krizbai et al., 1998; Sharp et al., 2002), whereas evidence from other investigators suggest that GluRs are not expressed in CMVEC (Morley et al., 1998). Glutamate receptors in cerebral microvessels and CMVEC have been linked to protein phosphorylation (Krizbai et al., 1998) and blood—brain barrier function (Collard et al., 2002; Koenig et al., 1992; Sharp et al., 2002). GluR agonists, however, failed to increase levels of inositol triphosphate, cAMP, or Ca2+ in CMVEC (Morley et al., 1998). Although expression of kainate receptors was detected in cerebral microvessels and in CMVEC (Morley et al., 1998), kainate did not increase Ca2+ in CMVEC (Morley et al., 1998) and had no vascular effects in vitro (Faraci et al., 1994).
To characterize GluRs in CMVEC from newborn pigs, we combined pharmacologic, immunochemical, and functional approaches. Radioligand binding assay is commonly used to pharmacologically characterize receptors for biologically active substances. High-affinity glutamate binding sites suggestive of GluRs are detected in CMVEC from newborn pigs (Parfenova et al., 2001). The amount of [3H]Glu receptor binding sites in CMVEC is higher than in the brain cortex. NMDA receptors in CMVEC were identified by the ability of the receptor agonists (NMDA and quinolinic acid) and antagonists (D-AP5, 4C3HPG, and CPP) to displace [3H]Glu from membrane binding sites. As indicated by the ligand potencies, NMDA receptors in CMVEC and in the brain appear to have distinct pharmacologic profiles. NMDA and quinolinate were more effective competitors of [3H]Glu in the brain, whereas CPP was more effective in CMVEC; D-AP5 and 4C3HPG were of equal potency in CMVEC and in the brain. AMPA/kainate receptors were identified in CMVEC by the ability of the receptor agonists (AMPA, kainic acid, and quisqualic acid) and antagonists (DNQX and CNQX) to displace [3H]Glu. In CMVEC, all AMPA/kainate receptor ligands had similar potencies in displacing glutamate, whereas in the brain, AMPA, kainate, and quisquilate were most effective. In CMVEC, the majority of AMPA/kainate receptor sites are found in microsomal membranes, whereas in the brain, these receptors are identified mainly in the low-speed membrane fraction. As indicated by the ligand effectiveness, the amount of AMPA/kainate receptors in CMVEC is significantly lower than in the brain cortex. Metabotropic GluR antagonists, 4CPG, PHCC, and CPP, while displacing 40% to 60% of [3H]Glu from the brain cortex, were not effective in CMVEC.
Ionotropic and metabotropic GluRs are detected in piglet CMVEC by immunoblotting. Antibodies against the rat NR1 subunit recognized three protein bands in CMVEC and in pig brain cortex: a 170-kd band and two low-molecular-weight bands, 36 to 38 kd. It has been reported that, in addition to the NR1/NR2 complexes (120 to 170-kd subunits) (Nakanishi, 1992), other NMDA receptors have been identified in the brain, including a 33-kd glutamate-activated ion channel protein pharmacologically identical to NMDA receptors (Smirnova et al., 1993). Antibodies against rat GluR1 (108 kd) recognized a major approximately 110-kd band in CMVEC and in the brain of newborn pigs. In CMVEC, we immunodetected metabotropic GluRs as a 160-kd band using antibodies againsts rat mGluR1α (160 kd). Recent evidence indicate that mGluR1 (140 kd), mGluR4 (102 kd), and mGluR5 (140 kd) are expressed in human brain endothelial cells and are important in blood—brain barrier permeability (Collard et al., 2002). Identification of mGluR1 protein, in spite the fact that mGluR antagonists did not displace bound [3H]Glu, suggests that characteristics of these receptors in CMVEC and in the brain are distinct. Immunochemical identification of NR1 and GluR1, in concert with our radioligand binding data, strongly suggests that ionotropic GluRs are expressed and functionally active in CMVEC.
L-Aspartic acid, another excitatory neurotransmitter, exhibits its effects via GluRs (Dreier and Honoré, 1988). In cerebral circulation of newborn pigs, aspartate dilates cerebral arterioles as effectively as glutamate and NMDA (Busija and Leffler, 1989), suggesting the engagement of GluRs. Our present data show that aspartate is an extremely effective competitor of glutamate in the radioligand assay. L-Aspartate displaced more than 80% of bound [3H]Glu and was as effective as glutamate in displacing the radioligand. The effectiveness of aspartate in competing for high-affinity glutamate binding sites in CMVEC cannot be explained solely by its interaction with NMDA and AMPA/kainate receptors. It seems that aspartate also interacts with other glutamate binding sites in CMVEC, presumably, high-affinity glutamate transporters.
Endothelial ionotropic GluRs are functionally coupled to HO. Glutamate increases endothelial CO production from endogenous heme via the HO pathway (Parfenova et al., 2001). As our present data show, agonists of NMDA- and AMPA/kainate receptors also increase CO production by freshly isolated cerebral microvessels and by cultured CMVEC. In the cerebral circulation of newborn pigs in vivo, agonists of ionotropic GluRs, AMPA, ATPA, and ACPD, are codependent vasodilators (Robinson et al., 2002). Therefore, rapid activation of endothelial HO via GluR-mediated mechanism is essential in vasodilator responses to glutamate in the cerebral circulation. Mechanisms by which activation of ionotropic GluRs increase HO activity in CMVEC remain to be elucidated. Heme oxygenase is expressed in two isoforms: constitutive HO-2 and inducible HO-1 (Maines, 2000). In freshly isolated cerebral microvessels from healthy piglets, HO is represented only by the constitutive isoform HO-2 (Leffler et al., 1999; Parfenova et al., 2001), whereas in cultured CMVEC, both HO-2 and HO-1 are expressed under low-serum conditions (Parfenova et al., 2001). HO-1 is a heat shock protein that is rapidly upregulated in response to cellular stress (Maines, 2000). Because cerebral dilation to glutamate develops immediately (Busija and Leffler, 1989; Robinson et al., 2002), involvement of new HO synthesis in the vascular response is unlikely. Therefore, it appears that glutamate rapidly increases activity of HO-2, which is the only HO isoform expressed in cerebral microvessels in vivo. Overall, mechanisms that may account for immediate increase in CO production-HO activity are poorly understood. Heme oxygenase can be activated by phosphorylation (Dore et al., 1999). Endogenous substrate delivery can be a rate-limiting step in CO production: (1) HO substrate, heme-L-lysinate, is a dilator in cerebral circulation (Leffler et al., 1999); (2) aminolevulinic acid (heme precursor) stimulates CO production by CMVEC (unpublished observations, 2002). Other possibilities such as regulation of HO activity by intracellular Ca2+ level need to be explored.
In summary, ionotropic GluRs (NMDA- and AMPA/kainate types) were detected in CMVEC from newborn pigs pharmacologically by displacement of radiolabeled ligand and by immunoblotting. Furthermore, activation of NMDA and AMPA/kainate receptors increased CO production by cerebral microvessels and by CMVEC. These results suggest that CMVEC express functional GluRs and, therefore, may directly respond to glutamatergic stimulation.
Footnotes
Acknowledgments
The authors thank Dr. C. Waters, for help in establishing CMVEC cultures on microcarrier beads; Svetlana Sarkisova, for excellent technical support; and Danny Morse and Greg Short, for helping with preparation of the figures.
