Abstract
1-ARA-35b (35b) is a cyclized dipeptide that shows considerable neuroprotective activity in vitro and improves neurologic recovery after fluid percussion–induced traumatic brain injury in rats. The authors evaluated the effects of treatment with 35b in mice subjected to controlled cortical impact brain injury. Animals treated with intravenous 35b after traumatic injury showed significantly enhanced recovery of beam walking and place learning functions compared with vehicle-treated controls, in addition to reduced lesion volumes. Beneficial effects were dose related and showed an inverted U-shaped dose–response curve between 0.1 and 10 mg/kg. Protective actions were found when the drug was administered initially at 30 minutes or 1, 4, or 8 hours, but not at 24 hours, after trauma. In separate experiments, rats treated with 35b on days 7 through 10 after injury showed remarkably improved place learning in comparison with injured controls. These studies confirm and extend the neuroprotective effects of this diketopiperazine in traumatic brain injury. In addition, they show that 35b has a relatively wide therapeutic window and improves cognitive function after both acute and chronic injury.
The concept of secondary injury after CNS trauma has been firmly established on the basis of experimental studies (Panter and Faden, 1992). Traumatic CNS injury initiates a complex series of metabolic and other biochemical changes that begin within seconds of injury and may continue for days to weeks after the insult (Faden, 1996; McIntosh, 1994). Collectively, these changes ultimately cause death of neuronal cells adjoining the area of immediate physical insult.
Based on this concept of secondary biochemical injury, many potential treatments have been proposed and tested experimentally (Faden, 1996; McIntosh et al., 1998). Most of these strategies have been directed toward inhibiting a single component or factor in the secondary injury cascade. Although there is considerable experimental support for the use of these kinds of targeted strategies to reduce histologic damage and improve neurologic outcome after CNS trauma in experimental animal models, beneficial effects in humans have proved quite limited. For example, methylprednisone and naloxone have shown comparable and significant protective effects when administered within the first 8 hours after spinal cord trauma in humans (Bracken and Holford, 1993). These beneficial effects, however, are relatively modest and their therapeutic window is relatively narrow. Treatment of human brain trauma has proved less effective in several recent clinical trials, despite promising experimental results (Faden, 2002; Maas, 2001). Given the fact that tissue injury after neurotrauma is thought to be highly multifactorial, it may not be surprising that strategies aimed at only a single injury component have shown limited effectiveness.
Optimal treatment for traumatic CNS injury may therefore require either single drugs that modulate multiple factors within the secondary injury cascade or the use of combination drug-treatment strategies (Faden AI, 2001). Thyrotropin-releasing hormone (TRH), a tripeptide named for its hypophysiotropic function, has considerable neuroprotective properties and has been found to modulate a number of proposed secondary injury factors (Faden, 1996; Faden et al., 1999b). However, TRH and traditional TRH analogs have several physiologic actions—autonomic, analeptic, and endocrine (Yarbrough, 1979)—that may limit their clinical utility. We have developed a series of tripeptide derivatives of TRH (Faden et al., 1999a) and related cyclic dipeptides, which are devoid of autonomic, analeptic, and endocrine actions, yet have striking neuroprotective actions. Among these, 35b, a cyclohexyl diketopiperazine, has shown good neuroprotective activity in in vivo and in vitro rat models (Faden et al., 2003). The present studies were designed to examine the effects of 35b in a different head injury model (controlled cortical impact [CCI]) in a different species (mouse). Dose–response and therapeutic-window studies were examined, with both behavioral and lesion volume outcomes. In addition, the potential cognitive-enhancing effect of the compound was examined relatively late after injury (7 to 10 days). 35b was found to have striking neuroprotective effect, with a relatively large therapeutic window of at least 8 hours, as well as nootropic actions in chronic injured animals.
MATERIALS AND METHODS
Animals
Male C57Bl/6 mice (20 to 25 g) were obtained from Taconic Farms (Germantown, NY, U.S.A.) and housed in an area directly adjoining surgical and behavioral rooms for at least 1 week before any procedures. A constant temperature (22°C ± 2°C) and 12-hour light/dark-cycle environment were maintained, and all behavioral testing was performed during the light cycle (6 a.m. to 6 p.m.). All mice had free access to food and water.
Controlled cortical impact device
Our injury device was designed and built at the Georgetown Institute for Cognitive and Computational Sciences and consists of a microprocessor-controlled pneumatic impactor with a 3.5-mm-diameter tip (Fox et al., 1998). The impactor is vertically mounted on a mill table (Sherline, Vista, CA, U.S.A.) and allows for precise adjustment in the vertical plane above the mouse head, which is secured in a stereotaxic apparatus (David Kopf Instruments, Tujunga, CA, U.S.A.). The core rod of a linear voltage differential transducer (Serotec, Raleigh, NC, U.S.A.) is attached to the lower end of the impactor to allow measurement of velocities between 3.0 and 9.0 m/s. Velocity of the impactor is controlled by fine tuning both positive and negative (back) air pressures. An oscilloscope (Tektronix, Beaverton, OR, U.S.A.) records the time–displacement curve produced by the downward force on the linear voltage differential transducer, allowing precise measurement of the impactor velocity.
Surgery
Surgical anesthesia was induced and maintained with 4% and 2% isoflurane, respectively, using a flow rate of 1.0 to 1.5 L/min oxygen. Depth of anesthesia was assessed by monitoring respiration rate and palpebral and pedal-withdrawal reflexes. The animal was then placed on a heated pad and core body temperature was monitored and maintained at 38°C ± 0.2°C. The head was mounted in a stereotaxic frame and the surgical site clipped and cleansed before surgery. A 10-mm midline incision was made over the skull, the skin and fascia were reflected, and a craniotomy was performed (4 mm) on the central aspect of the left parietal bone (Roboz, Gaithersburg, MD, U.S.A.). Great care was taken during removal of the parietal bone to avoid injury to the underlying dura mater, which was continuously bathed in sterile normal saline warmed to 37.5°C. The impounder tip of the pneumatic injury device was sterilized, extended its full stroke distance (44 mm), positioned to the surface of the exposed dura, and reset to impact the cortical surface at a moderate level (6.0-m/s velocity, 1-mm tissue deformation). After injury, the incision was closed with interrupted 6–0 silk sutures, anesthesia was discontinued, and the mouse was placed in a heated cage to maintain normothermia during a 45-minute recovery period. All animals were monitored carefully for at least 4 hours after surgery and then daily. To minimize variation between animals due to anesthesia during acute neurologic testing, 20 minutes was allowed for surgery and 5 minutes for suturing for each animal.
Acute and chronic neurologic evaluation
After cessation of anesthesia, each mouse was placed into a heated cage in the supine position. Acute neurologic recovery was assessed in all mice by recording the time to recovery of hind paw flexion after application of pressure, an indicator of simple somatomotor function. Similarly, latency to recovery of the righting reflex, an indicator of somatosensory function, was recorded for each animal.
Chronic neurologic recovery was evaluated for all animals using a beam-walking task, a method which is particularly good at discriminating fine motor coordination differences between injured and sham-operated animals (Fox et al., 1998). The device consists of a narrow wooden beam 6-mm wide and 120mm long suspended 300 mm above a 60-mm-thick foam rubber pad. The mouse was placed on one end of the beam and the number of foot faults for the right hind limb recorded during 50 steps counted in either direction on the beam. A basal level of competence at this task was established before surgery with an acceptance level of <10 faults per 50 steps.
Spatial learning and memory function
A Morris water maze paradigm was used to access spatial learning and working memory by training mice to locate a hidden submerged platform using extramaze visual information, as previously detailed (Fox et al., 1998). The apparatus consists of a large, white, circular pool (900-mm diameter, 500-mm high, water temperature of 24°C ± 1°C) with a 76-mm-diameter plexiglas platform painted white and submerged 15 mm below the surface of the water (225-mm high), which was rendered opaque with the addition of white nontoxic paint. During training, the platform was hidden in one quadrant of the maze 14 inches from the side wall. The mouse was gently placed into the water facing the wall at one of four randomly chosen quadrants separated by 90 degrees. The time required (latency) to find the hidden platform with a 90-second limit was recorded by a blinded observer. Mice failing to find the platform within 90 seconds were assisted to the platform. Animals were allowed to remain on the platform for 15 seconds on the first trial and 10 seconds on all subsequent trials. There was an intertrial interval of 30 minutes, during which time the mice were towel-dried and placed under a heat lamp. A total of 16 training trials were conducted in blocks of four per day on days 7 to 10 (studies 1 and 4) or 14 to 17 (study 3) after surgery. To control for visual discriminative ability, animals were subjected to an additional four visuomotor trials 2 hours after the last training trial (trial 16). For visuomotor trials, a clearly visible black platform, raised 5 mm above the water surface, was placed at a different location than that for the submerged platform. Time required to find the visible platform was recorded.
Drug administration
Conscious mice were placed in a mouse restrainer and injected via the lateral tail vein with normal saline or 35b at various doses and times after CCI injury. The investigators were blinded to drug treatment, both at the time of surgery and for neurologic and behavioral scoring. Four separate studies were performed: (1) administration of a single intravenous bolus (1.0 mg/kg) of 35b thirty minutes after trauma; (2) dose–response evaluation of 35b, administered as a single intravenous bolus injection (0.1, 1, or 10 mg/kg) 1 hour after trauma; (3) time-course (therapeutic window) study with 35b (1.0 mg/kg) administered as a single intravenous bolus injection 1, 4, 8, or 24 hours after trauma; and (4) daily injection of an intravenous bolus of 35b (1.0 mg/kg) administered 30 minutes before mice were subjected to four place-learning trials on days 7, 8, 9, and 10 after injury.
Magnetic resonance imaging measurement
Proton density and T2-weighted magnetic resonance imaging (MRI) has been shown to be a reliable method of measuring lesion volume in other models of brain injury, most notably ischemia (Hockings et al., 1995; Loubinoux et al., 1997) where lesion size at 24 hours correlated well with histologic measures of damage. Diffusion and perfusion imaging have also been performed at 24 hours and 7 days after TBI (Assaf et al., 1999). We used T2-weighted MRI at 21 days to measure lesion volume after TBI, as previously detailed (Faden et al., 2001).
Twenty-one days after TBI, all animals were anesthetized with sodium pentobarbital (70 mg/kg, intraperitoneally) and subjected to MRI using a Bruker 7T/21 cm Biospec-Avance system (Bruker, Karlsruhe, Germany). Briefly, animals were placed within a Plexiglas animal bed with a 37°C heating pad to maintain body temperature. Respiratory gating to reduce motion artifacts was achieved using a respiratory monitor as described previously (Albensi et al., 2000). The animal bed was positioned with the head in the center of the magnet within a 72-mm 1H birdcage resonator (Bruker). Field homogeneity across the brain was optimized and a sagittal scout image was acquired (RARE [rapid acquisition relaxation enhancement pulse sequence] image, field of vision, 4 × 4 cm; 128 × 128 resolution; repetition time (TR) to echo time (TE), 1,500/10 milliseconds with a RARE factor of 8, making the effective TE 40 milliseconds). Multislice T2-weighted images were then acquired to obtain eight contiguous slices commencing at the end of the olfactory bulb and working caudally (field of vision, 3 × 3 cm; slice thickness, 2 mm; 128 × 128 resolution; TR/TE, 1,500/20 milliseconds; four echo images and two averages). Each scan took approximately 8 minutes. Lesion volume was estimated from the summation of areas of hyperintensity on each slice, multiplied by slice thickness, for both the ipsilateral and contralateral hemispheres. Average lesion volume (± SEM in μL) was calculated for each treatment group.
Histologic evaluation
Twenty-one days after injury, animals were killed with an overdose of sodium pentobarbital (100 mg/kg body weight) and perfused transcardially with physiologic saline and heparin followed by 4% paraformaldehyde in phosphate-buffered saline. The brains were removed, postfixed in the same fixative for 24 hours, dehydrated in a graded series of ethanols, and embedded in a series of celloidin (2% to 12%) dissolved in a 50:50 mixture of ethanol and ethyl ether. Embedded brains were exposed to chloroform vapors and then submerged in ethanol until serial (40-μ) sections were cut coronally on a microtome. Every fifth section was stained with modified Giemsa stain. A 0.4% w/v Giemsa stock was prepared in buffered methanol at a pH of 6. The stock was diluted 1:4 with water, and six drops of 1% glacial acetic acid were added per 100 mL diluted stain. Sections were stained in Giemsa (60 minutes) and rinsed in 95% (45 minutes) and 100% (2 minutes) ethanol. Sections were then immersed in isopropyl ethanol (20 minutes) and cedar wood oil (5 minutes), washed in xylene, mounted on slides, and cover-slipped with permount. The lesion volume was assessed blindly with respect to treatment group using a systematic sampling principle (Gundersen and Jensen, 1987). On average, 15 sections per animal were used for lesion volume measurement: section periodicity was 5, the space between used sections was 200 microns. In selected sections, the area that contained damaged tissue or cavity was carefully circumscribed at low magnification (1.5× objective lens) using the Neurolucida program (MicroBrightField, Williston, VT, U.S.A.).
Data analysis
Beam-walking and Morris water maze data were analyzed by two-way repeated measures ANOVA, followed by post hoc one-tailed Dunnett tests with CCI + vehicle-treated animals serving as controls. In the dose–response and therapeutic-window studies, raw beam-walk scores were not normally distributed; these data were normalized with a square root transformation and then assessed as described previously. Data are expressed as mean ± SEM, except for beam-walk scores in the therapeutic window study (which are expressed as individual animal scores and medians for each group). Data points greater than three standard errors from the mean were considered outliers and not included for statistical analysis. Lesion volume data were analyzed by one-tailed t-tests and regression ANOVA, and graphed as mean ± SEM. Histologic data were analyzed with GB-Stat Version 6 statistical package (Dynamic Microsystems, Silver Spring, MD, U.S.A.). The relation between lesion volume measured by MRI and histologic assessment was analyzed by linear regression.
RESULTS
Physiology and acute recovery
There was no significant difference in body weight and core temperature among groups. After cessation of anesthesia, no significant differences in either pedal withdrawal or righting reflexes were found in any of the treated groups when compared with the control group (data not shown).
Beam walking
Treatment with 35b (1.0 mg/kg) 30 minutes after injury (study 1; Fig. 1) significantly reduced the number of foot faults compared with vehicle-treated controls at 14 and 21 days after injury (P < 0.05 for CCI + 35b vs. CCI + saline on days 14 and 21). In addition, when given over a range of doses (0.1 to 10 mg/kg) at 1 hour after injury (study 2; Fig. 2), 35b improved beam walking 21 and 28 days later (P < 0.05 for 0.1, 1.0, or 10 mg/kg 35b vs. control scores from the same day on days 21 or 28). The 1.0-mg/kg dose was most robust, and significantly reduced foot faults 7, 14, 21, and 28 days after injury (P < 0.05 for CCI + 1.0 mg/kg 35b vs. control scores from the same day). This dose was therefore selected for a therapeutic-window study (study 3; Fig. 3), where long-term improvement in beam walking was observed when treatment was delayed as long as 4 or 8 hours after injury (on day 14, P < 0.05 for CCI + 35b at 1 or 4 hours after injury vs. saline at 1 hour after injury; on day 21, P < 0.05 for CCI + 35b at 1, 4, or 8 hours after injury vs. saline at 1 hour after injury).
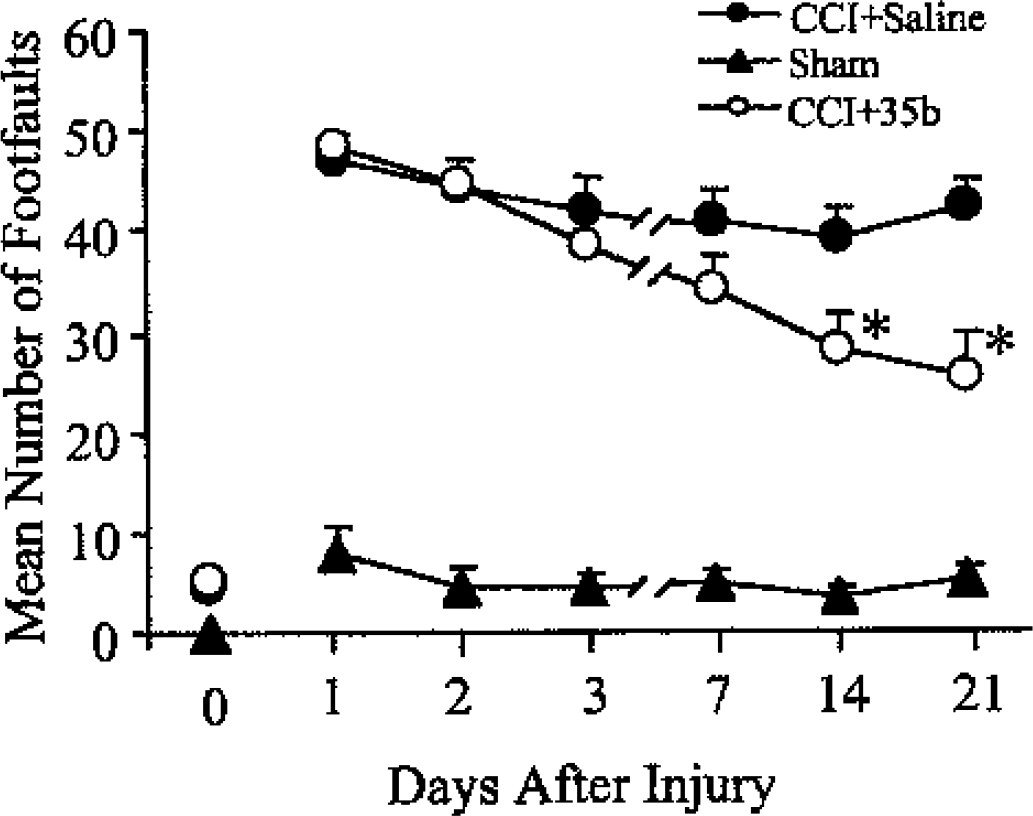
Effect of 35b on motor recovery over time after controlled cortical impact (CCI) in the mouse. 35b (1.0 mg/kg) or an equivalent volume of saline vehicle was injected intravenously at 30 minutes after injury. Animals were tested to determine the number of errors (foot faults) on a beam-walking task 1 to 21 days later. The 0 indicates beam scores before injury. Sham mice received surgery but no injury. Dots represent the mean ± SEM number of foot faults for each group as shown in the legend (*P < 0.05 vs. CCI + saline scores from the same day).
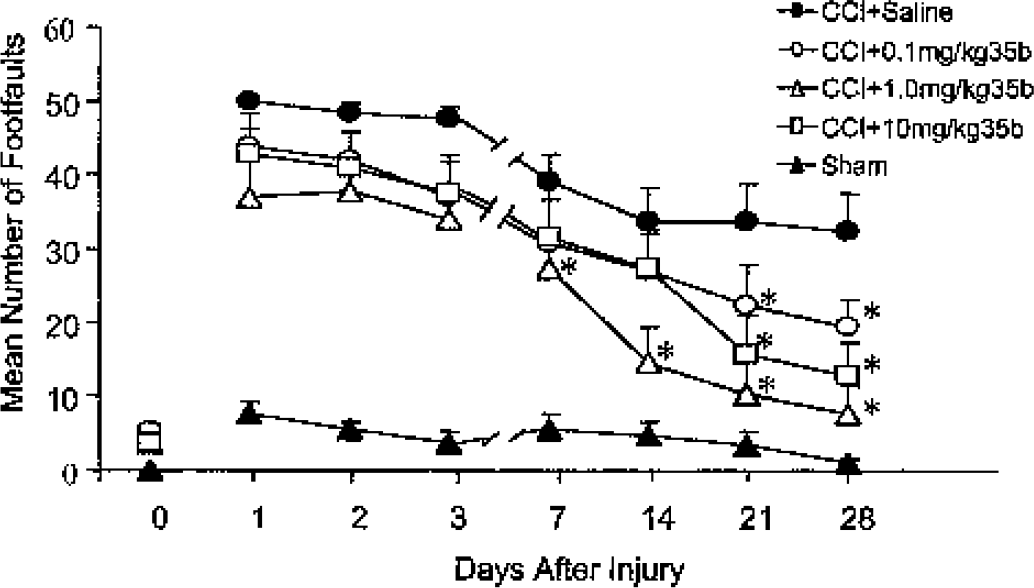
Dose–response of 35b on motor recovery after controlled cortical impact (CCI) in the mouse. Either 0.1, 1.0, or 10 mg/kg 35b or an equivalent volume of saline was injected intravenously 1 hour after CCI. Beam-walking ability was tested 1 to 28 days later (x-axis). Sham mice received surgery but no injury. The 0 indicates beam scores before injury. Dots represent the mean ± SEM number of foot faults for each group, as shown in the legend. *P < 0.05 versus CCI + saline scores from the same day.
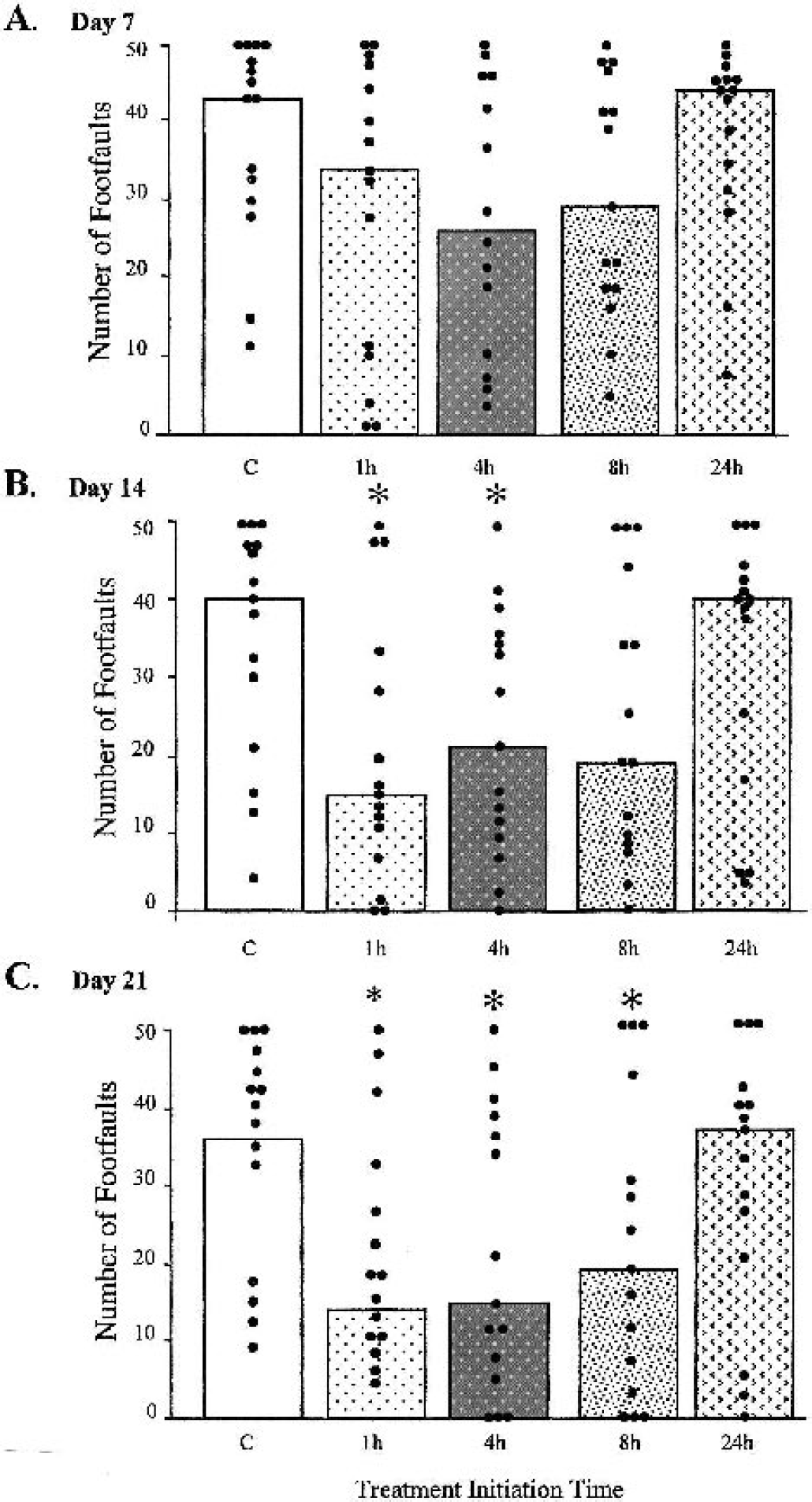
Therapeutic window of 35b effect on motor recovery after controlled cortical impact (CCI) in the mouse. A single dose of 35b (1.0 mg/kg) was injected intravenously 1, 4, 8, or 24 hours after injury. Controls received equivolume injections of saline 1 hour after injury. The number of foot faults for each individual animal is represented as a dot, whereas bars represent the median score for each treatment group. A, B, and C show scores 7, 14, and 21 days after injury, respectively. *P < 0.05 versus control scores from the same day.
Place learning
The effect of 35b on cognitive recovery (study 1; Fig. 4) was assessed by comparing the time required to find a submerged resting platform in a Morris water maze task (goal latencies). Treatment with 35b (1.0 mg/kg) 30 minutes after injury significantly reduced mean latencies on three of four test days (P < 0.05 for CCI + 35b vs. control scores on the same day on days 8, 9, or 10). This performance was similar to that seen in sham animals. Delayed administration of 35b was associated with similar improvements. Treatment at 1 or 4 hours after injury (study 3; Fig. 5) reduced mean latencies on two of four test days (P < 0.05 for CCI + 35b at 1 or 4 hours after injury vs. saline at 1 hour after injury, scores from the same day on days 16 and 17). Treatment delayed as long as 8 hours after injury (Fig. 5) significantly reduced latencies on one day of testing, and showed strong trends toward reduction on three of four test days (on day 16, P < 0.05 for CCI + 35b at 8 hours after injury vs. saline at 1 hour after injury scores from the same day). Trends were also observed in mice treated 24 hours after injury (Fig. 5).
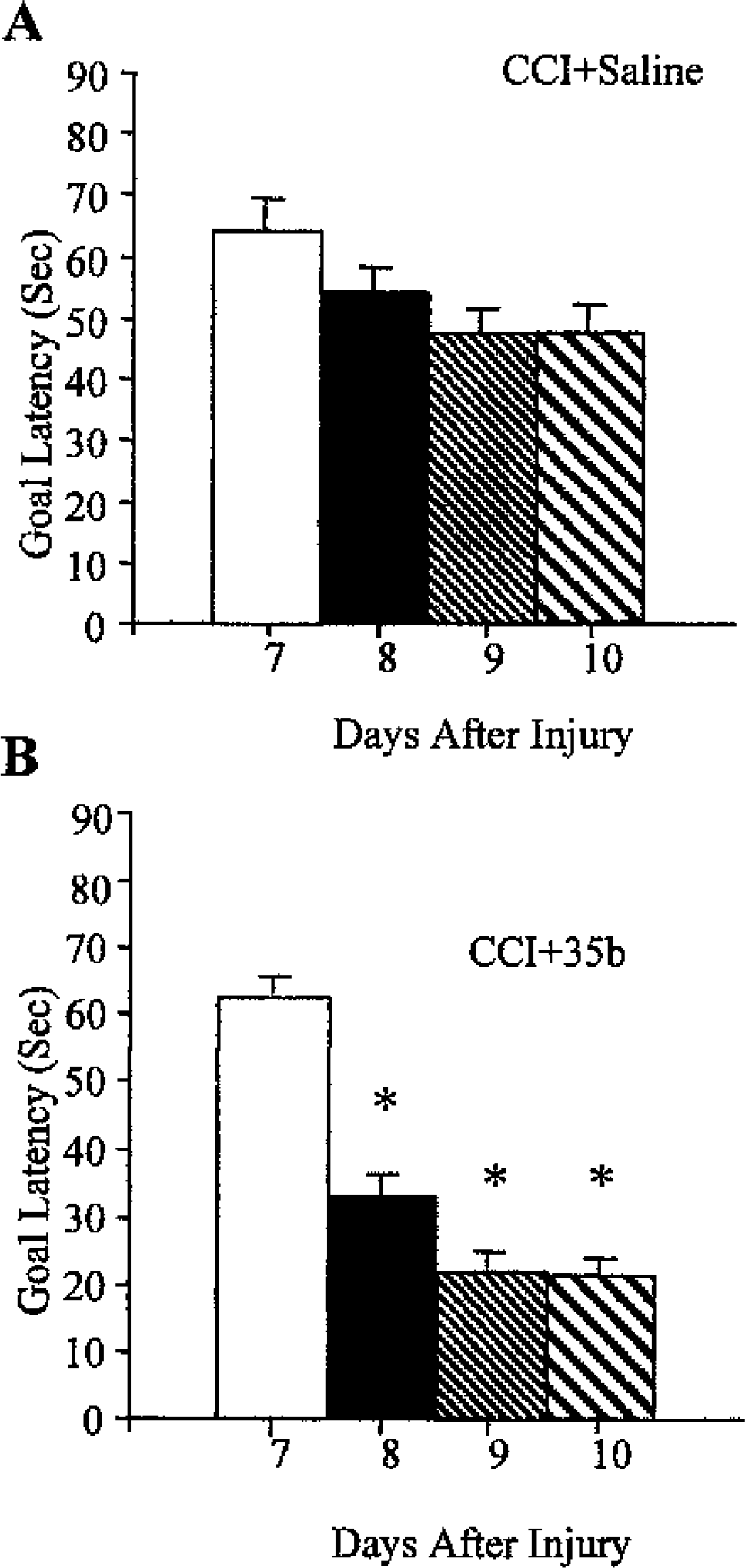
Effect of 35b (
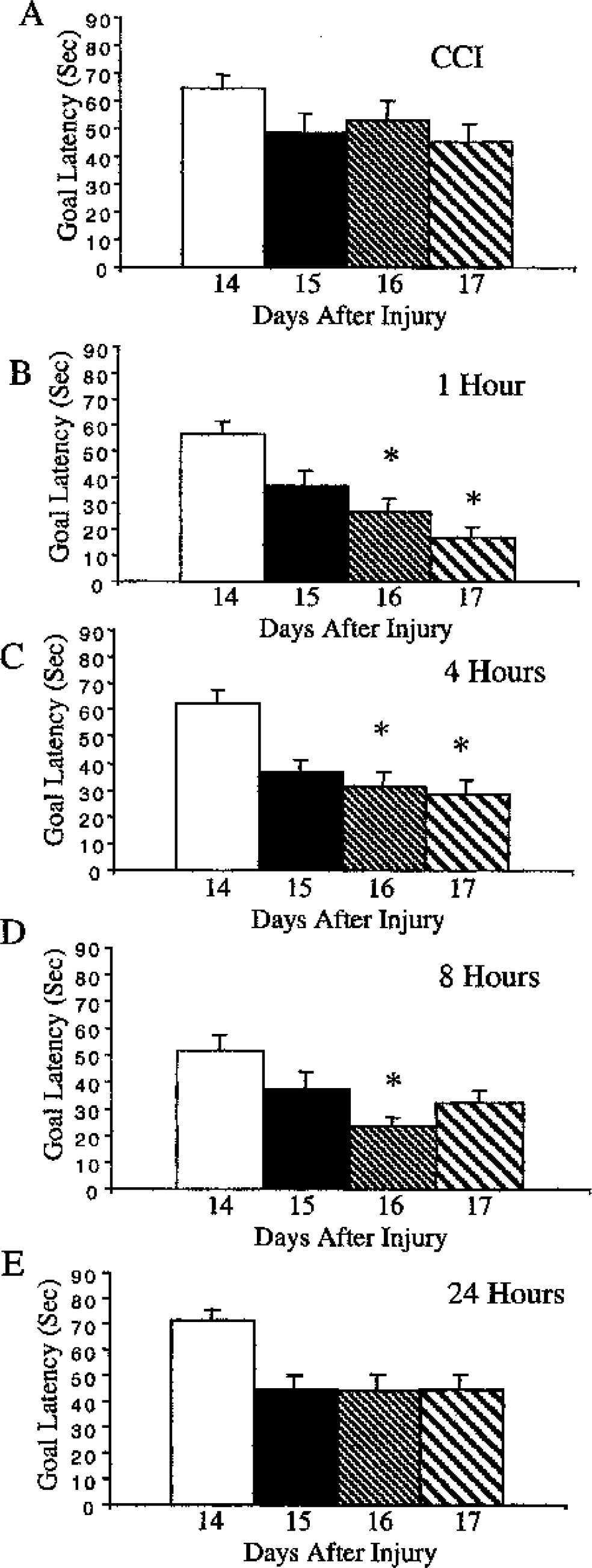
Therapeutic window of 35b effect on cognitive recovery after controlled cortical impact (CCI) in the mouse. Data are from the same mice injured and treated as described in Fig. 3. Animals experienced 4 training trials 30 minutes apart on days 14 through 17 after injury, as indicated on the x-axis.
In a study designed to test the effect of 35b on cognition in animals with an established brain lesion (study 4; Fig. 6), 35b (1.0 mg/kg) was administered once daily before water maze testing on each of four consecutive days, beginning 1 week after injury. This treatment reduced latencies in the water maze task compared with saline-treated injured controls on three of four testing days (P < 0.05 for CCI + 35b vs. CCI + saline on days 8, 9, and 10). There was no difference in swimming ability between the treated and saline groups on the visuomotor test because both groups had similar latencies to find a visible platform (mean latency ± SEM for CCI + 35b or CCI + saline groups was 19.6 ± 3.78 and 20.2 ± 4.14, respectively).
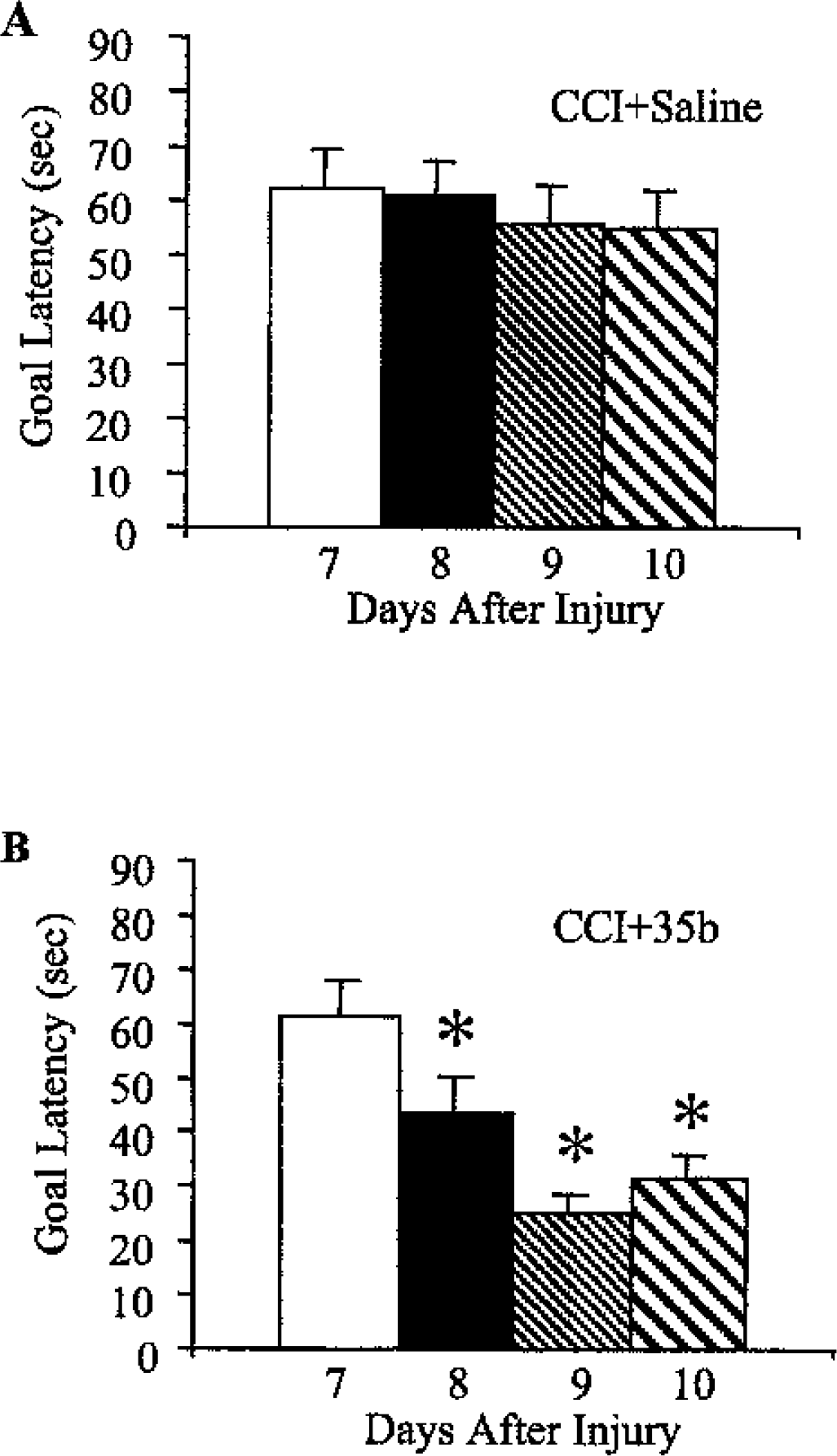
Effect of 35b on Morris water maze performance in mice with an established controlled cortical impact (CCI) lesion. Mice were injured and allowed to recover. Seven to 10 days after injury, a single dose of 35b was injected intravenously (1.0 mg/kg) on each day 30 minutes before four trials of water maze testing began. Blocks represent the mean ± SEM score across the four trials for each day. Treatment with saline or 35b is shown in
Lesion volume measurements
The T2-weighted MRI images used to assess lesion volume 21 days after injury showed a trend toward reduced lesion size in animals treated with 35b one hour after injury compared with vehicle-treated controls, though this reduction just missed significance cutoff value (P = 0.06; Fig. 7A). Histologic assessment of lesion volume in the same animals (Figs. 7B and 8), however, showed a similar decrease in lesion volume that was statistically significant (P < 0.05). Plotted together, the MRI and histologic data were highly correlated (correlation coefficient 0.7529, P = 0.0012, by simple regression ANOVA; Fig. 7C).
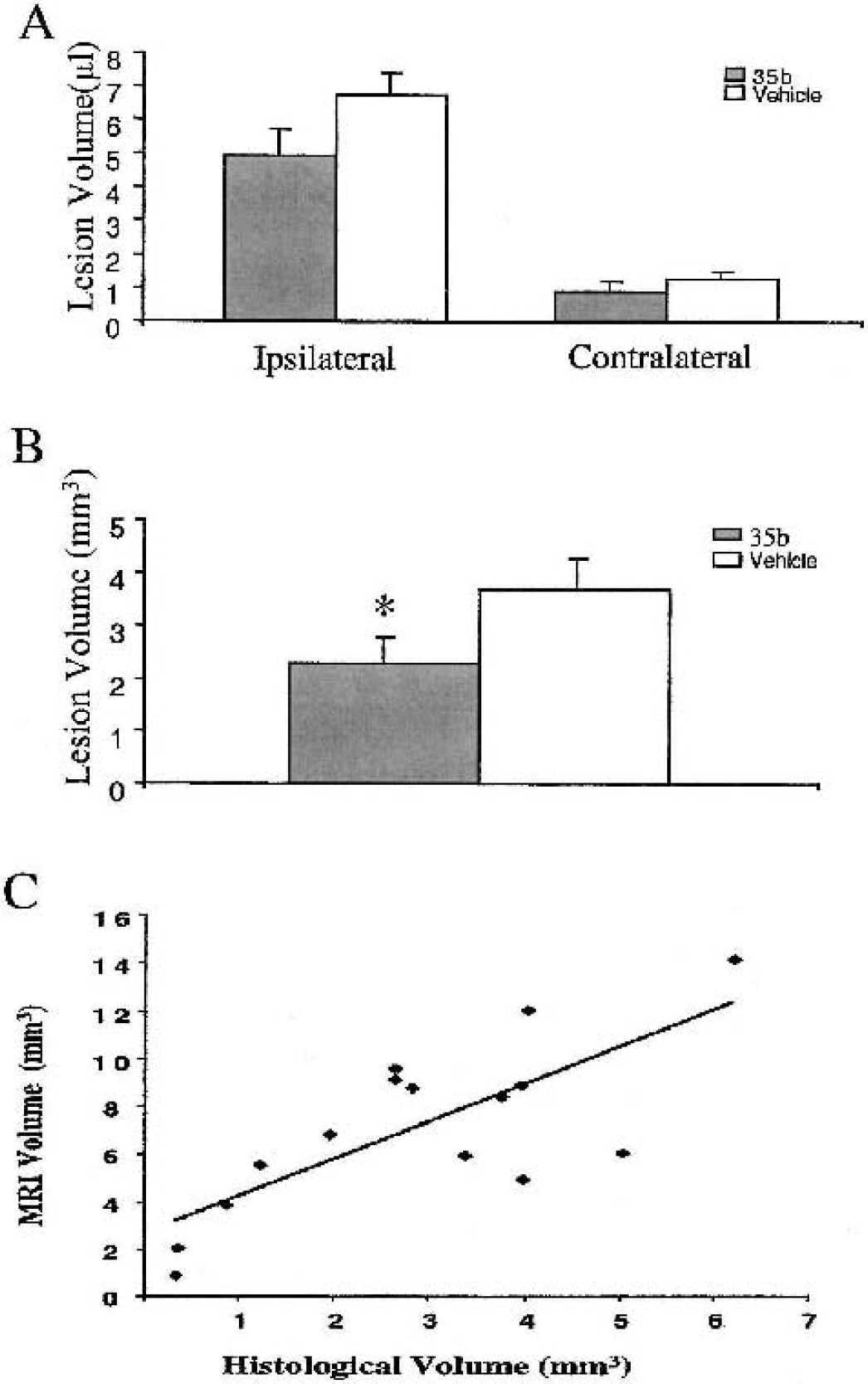
Effect of 35b on controlled cortical impact (CCI)–induced lesion volume 21 days after injury in the mouse. Data are from the same mice treated 1 hour after injury with 35b or saline vehicle, as detailed in Fig. 3.
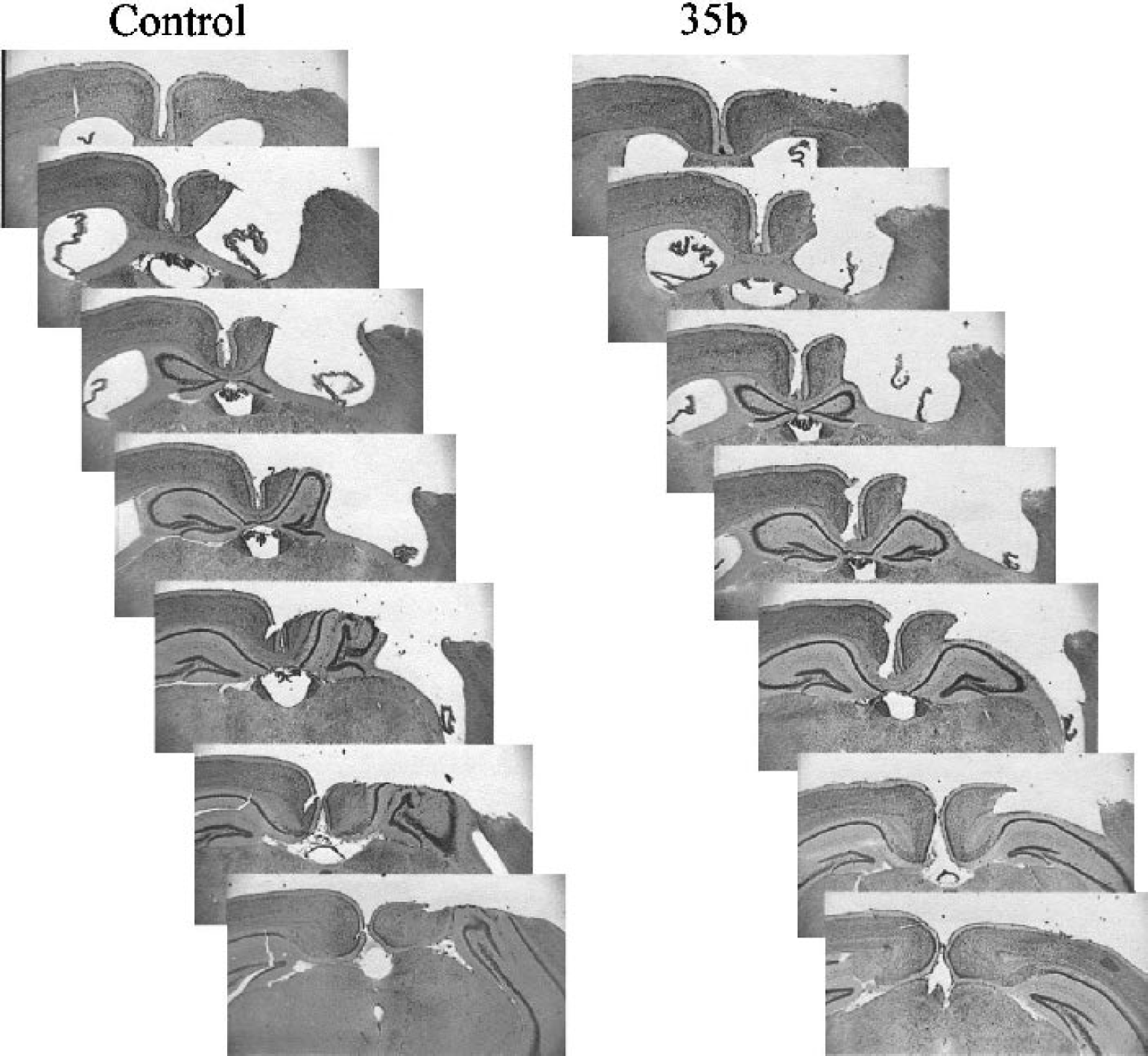
Cresyl violet–stained sections through the anterior to posterior aspect of the lesion from representative mice in the control and 35b treatment groups. Data are from animals treated 1 hour after injury in the therapeutic-window study.
DISCUSSION
Thyrotropin-releasing hormone and closely related peptides have substantial neuroprotective actions across a variety of experimental models of CNS trauma in a number of species (Akdemir et al., 1993; Arias MJ, 1987; Behrmann et al., 1994; Ceylan et al., 1990, 1992; Faden, 1989, 1993; Faden et al., 1981, 1990; Fukuda et al., 1979; Puniak et al., 1991; Takami et al., 1991; Wang and Zhu, 1991). A small clinical study confirmed these beneficial effects in human spinal cord injury (Pitts et al., 1995). However, TRH and traditional TRH analogs have other physiologic actions that may limit their clinical utility (Faden et al., 1999b). For example, pressor actions may be undesirable in a head-injured patient with significant intraparenchymal hemorrhage. Euthermic actions may preclude the use of hypothermia and may serve to elevate body temperature above desirable levels. Analeptic effects may antagonize the effects of sedatives (e.g., barbiturate coma) or anesthetics required for surgical intervention. Finally, endocrine interactions may be problematic if longer-term administration is considered.
Compound 35b was identified as part of a drug-discovery effort to develop peptide and nonpeptide analogs of TRH that preserve its neuroprotective actions but eliminate the other major physiologic actions of this hormone (Faden et al., 2003). Previously, we reported a novel tripeptide analog that showed considerable neuroprotection and was devoid of autonomic or analeptic effects, although it possessed some endocrine actions (Faden et al., 1999b). Thyrotropin-releasing hormone is metabolized to a cyclized dipeptide (cyclo-his-pro) that retains some physiologic activity, although it has not been reported to be neuroprotective (Prasad, 1995). Other cyclized dipeptides (diketopiperazines) have also been shown to have physiologic activity (Prasad, 1995).
Compound 35b is a relatively lipophilic diketopiperazine that has structural similarities to cyclo-his-pro. The partition coefficient for 35b between n-octonol and water (logP), calculated using the Hint program (eduSoft, Ashland, VA, U.S.A.), was 1.17; thus it is several orders of magnitude more hydrophobic than TRH or previous TRH analogs. This superior hydrophobicity would be expected to enhance its cellular permeability, thereby improving its accessibility to the brain. 35b has no autonomic, analeptic, or endocrine activity and, indeed, does not have appreciable activity at either of the two identified, high-affinity TRH receptors (Faden et al., 2003). In parallel studies, this compound showed considerable neuroprotective activity in vitro—including models of trauma, apoptosis, and necrosis. Moreover, rats subjected to lateral fluid percussion–induced TBI and treated with a single intravenous bolus infusion of 35b thirty minutes after trauma showed significantly improved motor and cognitive recovery (Faden et al., 2003).
In the present studies we sought determine (1) whether 35b had neuroprotective actions in another model of TBI (CCI) and in another species (mouse); (2) whether it had an inverted U-shaped dose–response curve like TRH or classical TRH analogs; (3) the therapeutic window of 35b; and (4) whether it had cognitive-enhancing properties (nootropic effects) in injured animals with an established lesion. We used a well-characterized CCI model in mice, which differs pathologically from fluid percussion injury. Dose–response studies showed a relatively flat, inverted U-shaped dose–response curve between 0.1 and 10 mg/kg, with the most effective dose being 1 mg/kg. These dose–response characteristics are similar to those we have reported for the N-terminal–substituted TRH analog YM14673, which shows potent neuroprotection in rat TBI and spinal cord injury models (Faden, 1989). The fact that significant neuroprotective effects were observed at 10 times the optimal dose, without apparent toxicity, suggests that 35b may have a good therapeutic index. Significantly improved functional recovery was observed even when initial treatment was delayed until 8 hours after trauma, which indicates a long therapeutic window for the compound. These improved behavioral effects were associated with a substantial reduction in lesion volumes, determined both histologically and with T2-weighted MRI.
To our knowledge, this is the first TBI study to directly compare histologic lesion volume measurements with those obtained using MRI; the results of these two methods were highly correlated. Use of MRI to measure lesion volumes has several potential advantages over standard histology: (1) lesion volumes can be followed noninvasively over time, so that one can examine effects of pharmacological interventions over the course of secondary injury development and longer-term tissue loss; (2) behavioral assessment and tissue loss can be correlated over time in the same animals; (3) potential postmortem artifacts are avoided; and (4) assessment can be done quite rapidly. In other studies, we have shown the utility of MRI application to evaluate both secondary injury and pharmacological actions (Faden et al., 2001). Whether this can be used as a surrogate marker for clinical head injury or pharmacological studies remains to be determined.
Importantly, administration of 35b before each water maze trial, on days 7 to 10 after trauma, resulted in remarkably improved spatial learning, with treated animals recovering to a level similar to uninjured (sham-injured) controls. This suggests that 35b may have nootropic properties and thus may have a role in the treatment of chronic head injury. The chemical structure of 35b suggests that it may also be administered orally—a highly desirable quality for chronic administration. It should be noted that some other diketopiperazines have shown cognitive-enhancing properties, although the TRH derivative cyclo-his-pro does not (Prasad, 1995).
The mechanisms by which 35b produces its therapeutic actions have not been identified, although it can attenuate in vitro models of both necrotic neuronal cell death (in vitro trauma, maitotoxin) and apoptotic cell death (staurosporine) (Faden et al., 2003). The class of compound also reduces glutamate or free radical–induced toxicity in vitro (S. Knoblach and A. I. Faden, unpublished observations, 2002), which suggests multipotential neuroprotective actions. Moreover, it attenuates the neuronal toxicity of β-amyloid in vitro (V. Movsesyan and A. I. Faden, unpublished observations, 2002), which, combined with its nootropic properties, may make this diketopiperazine a candidate for the treatment of Alzheimer disease.
Using the NIMH screening profile for receptor binding, 35b has shown very modest binding to various muscarinic receptors and no significant binding to a large number of classical neurotransmitter receptors or transporters. However, TRH has been shown to physiologically antagonize a number of factors implicated in secondary injury (endogenous opioids platelet activity factor, leukotrienes, glutamate), without having appreciable activity at their respective receptors (Faden, 1996). It is also possible that 35b may alter the regulation of genes or gene products involved in secondary injury. Such studies are now feasible using microarray technology.
CONCLUSIONS
In summary, we report the neuroprotective and nootropic effects of a novel diketopiperazine. Its relatively large therapeutic window and its ability to attenuate necrotic and apoptotic neuronal cell death suggest that this compound may have utility for the treatment of acute head injury. Moreover, its ability to improve cognitive function in animals with established lesions suggests that drugs of this class may also have a role in the treatment of chronic head-injured patients.
Footnotes
Acknowledgments:
The authors thank Sadia Aden and Anu Singh for their expert technical assistance.
