Abstract
During the last few years, the therapeutic use of stem and progenitor cells as a substitute for malfunctioning endogenous cell populations has received considerable attention. Unlike their current use in animal models, the introduction of therapeutic cells in patients will require techniques that can monitor their tissue biodistribution noninvasively. Among the different imaging modalities, magnetic resonance (MR) imaging offers both near-cellular (i.e., 25- to 50-μ) resolution and whole-body imaging capability. In order to be visualized, cells must be labeled with an intracellular tracer molecule that can be detected by MR imaging. Methods have now been developed that make it possible to incorporate sufficient amounts of superparamagnetic iron oxide into cells, enabling their detection in vivo using MR imaging. This is illustrated for (neural stem cell—derived) magnetically labeled oligodendroglial progenitors, transplanted in the central nervous system of dysmyelinated rats. Cells can be followed in vivo for at least 6 weeks after transplantation, with a good histopathologic correlation including the formation of myelin. Now that MR tracking of magnetically labeled cells appears feasible, it is anticipated that this technique may ultimately become an important tool for monitoring the efficacy of clinical (stem) cell transplantation protocols.
Keywords
USE OF STEM CELLS AS A SOURCE OF TRANSPLANTABLE NEURAL PRECURSORS FOR CENTRAL NERVOUS SYSTEM REPAIR
Currently, most treatments for injury or disease of the brain and spinal cord aim to relieve symptoms and limit further damage. Recent studies suggest that regeneration mechanisms do exist in the central nervous system (CNS), including the discovery of neural stem cells in the adult brain (Eriksson et al., 1998;Johansson et al., 1999). These findings raise hope that it may be possible to discover new treatments to repair CNS damage. Such treatment ideally would be based on the activation and recruitment of endogenous cells through growth factors that induce cell proliferation; at present, however, these factors are largely unknown. In addition, this approach would not work for inherited diseases where endogenous cells carry a genetic defect. Thus, for now, this repair strategy remains an elusive goal. An alternative approach is to administer exogenous cells in the form of stem cells or neural progenitor cells. Recent research suggests that the CNS is one of the few areas in which there is evidence that cell-replacement therapy can indeed restore lost function (McKay, 2000). It has recently been demonstrated, for instance, that transplantation of embryonic (ES) cells (differentiated into neural stem cells, or NSCs) in a rat model of spinal cord injury resulted in a significant improvement of hind limb support, including the ability to support weight (McDonald et al., 1999). This included the formation of new axons and myelin, although it is not clear whether this was a direct product of the transplanted cells or an indirect result from trophic molecules secreted by those cells. Another example is the transplantation of stem cells in animal models of dysmyelination. Brüstle et al. (1999) derived oligodendroglial precursor cells from mouse ES cells and showed that large areas in the spinal cord could be myelinated following intraventricular transplantation. Yandava et al. (1999) transplanted clonal NSCs in the shiverer mouse model, and demonstrated that these cells, after differentiation into oligodendroglial cells, can myelinate large areas in the CNS. Moreover, a significant number of recipient animals showed a decrease in their symptomatic tremor. Yet another example is the transplantation of (low doses of) mouse ES cells into the rat striatum of 6-OHDA lesioned animals, used as a model for Parkinson disease. The ES cell—derived dopamine neurons induced a sustained behavioral restoration of dopamine-mediated motor asymmetry (Björklund et al., 2002).
These initial animal studies indicate that an improvement of neural functioning can be established through stem cell transplantation. With the recent advances in human stem cell research, that is, the successful propagation of ES cell—derived (Thomson et al., 1998) and embryonic germ cell—derived human cells (Shamblott et al., 1998), and their derivation into a wide variety of cell lines, it may be possible in the future to achieve similar results in human disease. Several groups have reported the generation of enriched and expandable preparations of proliferating neural progenitors from human ES cells, capable in vitro of differentiating into progeny of the three neural lineages (i.e., astrocytes, oligodendrocytes, and mature neurons) (Reubinoff et al., 2001;Zhang et al., 2001). When transplanted into the ventricles of neonatal mice, these ES cells integrate in large numbers into the brain parenchyma, demonstrate a wide distribution, and differentiate into astrocytes and mature neurons. These results suggest that human ES cells are a source of transplantable neural precursors for possible nervous system repair. An alternative source of human ES cells is clones of neural stem cells isolated from human fetuses. After transplantation in the newborn mouse brain, these cells are capable of replacing deficient neurons, are able to express foreign genes, and participate in all aspects of normal brain development (Flax et al., 1998). Thus, stem cells harbor the potential of replacing malfunctioning endogenous cells in variety of CNS disorders.
TRANSPLANTATION OF OLIGODENDROGLIAL (PROGENITOR) CELLS IN MYELIN DISEASE
Myelin disorders form an important group of human neurologic diseases. Although the CNS appears to have only a limited capacity to remyelinate, remyelination by endogenous oligodendrocytes and Schwann cells has been reported in several clinical and experimental settings. In multiple sclerosis (MS), the most prevalent acquired myelin disease, remyelination has also been observed (Prineas et al., 1993;Raine and Scheinberg, 1981). Although recent studies suggest that significant numbers of oligodendrocyte progenitors can be present in and around MS lesions (Chang et al., 2002;Wolswijk, 2002), eventual remyelination is not complete, and other means of promoting repair are required to restore function in affected patients. One pragmatic approach is the transplantation of exogenous myelinating cells. For successful transplantation, cells should fulfill four requirements (Duncan, 1996): 1) cells should be capable of extensive migration and division; 2) cells must become associated with and myelinate axons; 3) cells must be nonimmunogenic; and 4) cells should be cryo-preservable and retain their properties on thawing before transplantation.
Stem cells and oligodendrocyte progenitors fulfill these requirements and, as such, are the transplantable cells of choice. Experimental transplantation has been carried out using two approaches. The first one is to transplant cells into persistently demyelinated lesions in the spinal cord that are created by focal irradiation and injection of gliotoxic chemicals (Blakemore and Franklin, 1991). In this model, it was found that transplanted glial cells migrate over a greater distance and remyelinate demyelinated lesions more rapidly than endogenous remyelinating cells (Blakemore et al., 2000). In the second approach, mutant animal models of dysmyelination have been used (Duncan et al., 1988;Duncan and Archer, 1993;Utzschneider et al., 1994;Tontsch et al., 1994;Zhang et al., 1999). To evaluate the migratory and myelinating capacity of transplanted cells, one must be able to discriminate these cells from host glia and myelin. In the shiverer mouse and myelin-deficient (md) rat, the presence of myelin basic protein and proteolipid protein—positive myelin, respectively, which is not detectable in nontransplanted animals, has been used as a marker for the transplanted cells (Lachapelle et al., 1983;Duncan et al., 1988). To detect grafted cells, it is possible to label them before transplantation with vital dyes (Baron-Van Evercooren et al., 1991;Espinosa de los Monteros et al., 1992;Gansmuller et al., 1992), tritiated thymidine (Lindsay and Raisman, 1984), and fast blue (Espinosa de los Monteros et al., 1993), in the latter case up to 6 months after transplantation. Another approach is to transplant male cells into recipient females and then use a Y-chromosome—specific probe for in situ hybridization (Blakemore et al., 1995;Harvey et al., 1992), or to insert a reporter gene such as LacZ. This gene has been transfected into transplanted oligodendrocyte progenitors or neural precursor cells (Franklin et al., 1995;Groves et al., 1993;Tontsch et al., 1994;Herrera et al., 1999;Learish et al., 1999), which can be visualized using X-gal staining. New myelination in the CNS by transplanted cells will be relevant to human demyelinating disease only if it results in restoration of nerve function. Transplantation of (cryopreserved) adult human Schwann cells (Kohama et al., 2001), adult human neural precursors cells (Akiyama et al., 2001), or pig olfactory ensheathing cells (Imaizumi et al., 2001) in rat models of chemical demyelination or transection has resulted in restoration of nerve conduction. For the md rat, transplanted glial cells have shown to be able to restore the nerve conduction velocity to near-normal values (Utzschneider et al., 1994). These data suggest that cell transplantation in demyelinated and dysmyelinated human disease may have significant potential for functional restoration. An animal model where the myelin defect is similar in scale to the human disease, MS, is the shaking pup; using this model, transplantation of oligodendrocyte progenitors resulted in repair of large areas similar in size to many MS plaques (Archer et al., 1997).
The aforementioned approaches for detecting transplanted cells require invasive and irreversible tissue removal procedures. The use of stem and progenitor cells in human clinical studies will require a technique that can monitor their migration and tissue biodistribution noninvasively. MR imaging has the capabilities of noninvasive whole-body in vivo imaging, with a resolution of 25 to 50 microns, approaching the resolution of single cells. For transplanted cells to be detectable by MR imaging, they need to be labeled with an MR contrast agent.
MR CONTRAST AGENTS
Two major classes of contrast agents exist. The first class of agents is the so-called paramagnetic agents, primarily based on Gd(III) that possess seven unpaired electrons, and that shorten the T1 and T2 relaxation by spin—spin– and spin—dipole—type interactions. Since they have relatively more effect on T1 relaxation—owing to the inherently longer T1 in most tissues—these agents are often referred to as “T1 agents,” and create hyperintense contrast on conventional T1-weighted spin-echo or spoiled gradient-echo MR sequences. The second class of contrast agents is based on superparamagnetic iron oxide (SPIO) particles. These particles have magnetic moments that, because of the small crystal size, are unhindered by lattice orientation and therefore do not exhibit hysteresis, hence the term “superparamagnetic.” In an applied magnetic field, the individual moments are free to align along the field, resulting in the formation of a single spin, with a net moment at least 4 orders of magnitude higher than a comparable ensemble of paramagnetic spins. This creates extremely large microscopic field gradients for dephasing nearby protons (Bulte and Brooks, 1997;Bulte et al., 1999a). This, in turn, dramatically shortens the nuclear magnetic resonance T2 relaxation time, over and far beyond the usual dipole—dipole relaxation mechanism that affects both T1 and T2, as in the case of paramagnetic contrast agents. Owing to the predominant T2 effect, these “T2 agents” usually create hypointense contrast on conventional spin-echo MR sequences, in particular when agglomerated within cells. Agglomeration or intracellular clustering of iron oxide nanoparticles dramatically reduces the T1 effect (Bulte et al., 2001). On gradient-echo images, where T2* effects dominate, these (intracellular) particles induce an even larger hypointense contrast effect. The microscopic inhomogeneities in the magnetic field induce a rapid dephasing of diffusing water protons, including those diffusing some distance away. This in turn leads to a “blooming effect,” that is, an amplification of signal changes.
Given the greater sensitivity of MR imaging for detecting superparamagnetic nanoparticles, these contrast agents are a natural choice for labeling cells. In addition, these agents are composed of biodegradable iron that can theoretically be recycled within the body. For instance, radiolabeled studies on the use of a liver-specific SPIO have shown that the iron is metabolized by liver Kupffer cells, with subsequent reuse and incorporation into the normal iron blood pool as well as erythrocytes (Weissleder et al., 1989). The normal adult body contains about 4 grams of iron. In contrast, when a paramagnetic Gd-chelate contrast agent is used for cell labeling, it is not clear how long the (toxic) metal remains chelated once it is incorporated within a cell, and how the cell will store or excrete the metal ion, given the larger doses of metal needed to induce sufficient contrast.
MAGNETIC LABELING OF CELLS
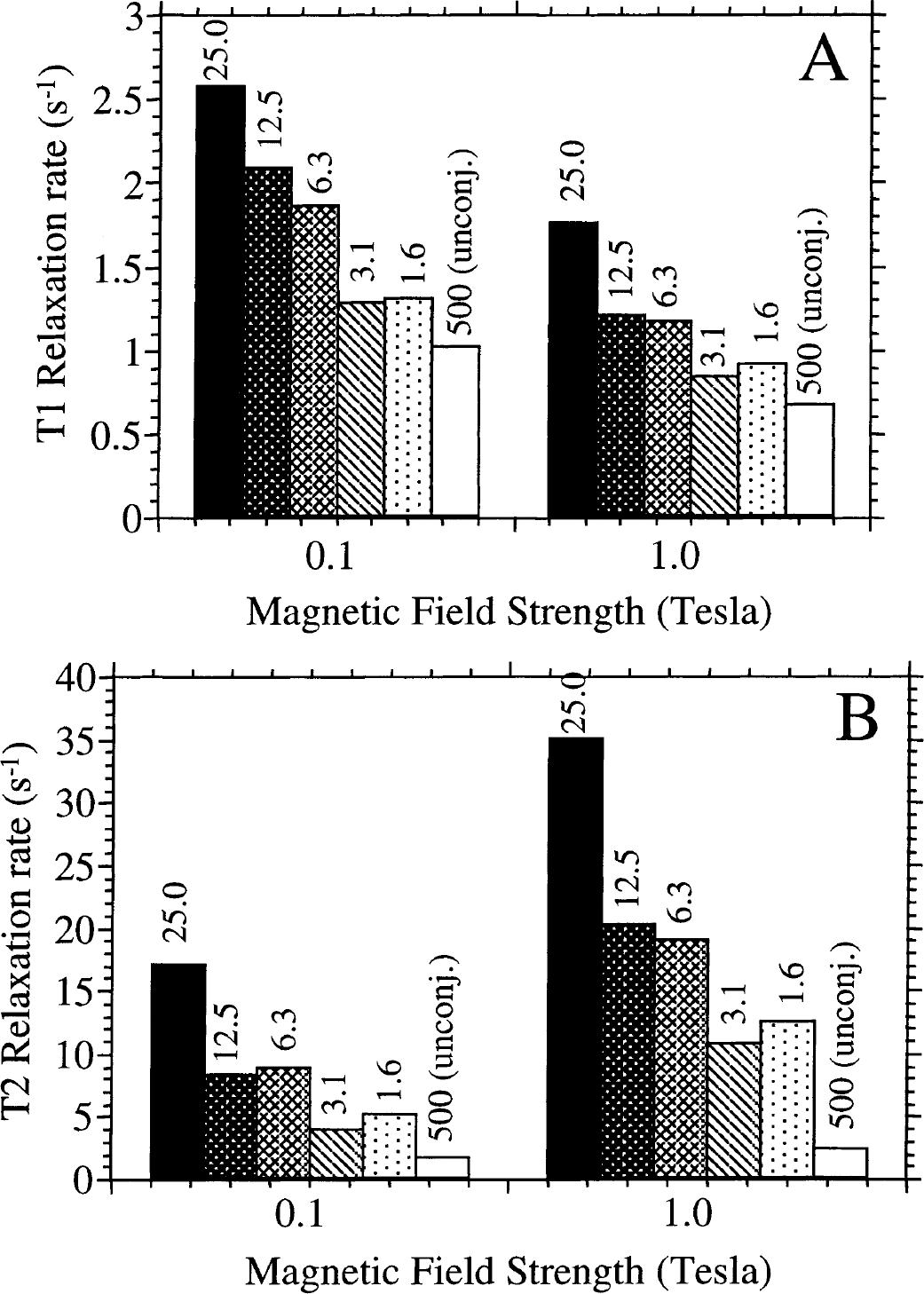
The history of tagging cells with an MR-detectable tracer dates back about a decade. Initially, attempts were made to label leukocytes, lymphocytes, and monocytes with superparamagnetic iron oxides. The rationale was that these labeled cells could be used for the study of immune trafficking. Strategies to prepare magnetically labeled cells included incubation with nonderivatized, dextran-coated iron oxide particles (Dodd et al., 1999, 2001;Schoepf et al., 1998;Sipe et al., 1999;Weissleder et al., 1997;Yeh et al., 1993, 1995), incubation with liposome-encapsulated iron oxide particles (Bulte et al., 1993), and lectin-mediated uptake (Bulte et al., 1996). In general, uptake was low, and further improvements in magnetic nanoparticle uptake were needed. In 1999, two groups reported on a modification of dextran-coated MION-46L magnetic nanoparticles (Shen et al., 1993) that resulted in efficient intracellular incorporation. The first group, Josephson et al. (1999) and Lewin et al. (2000), used a covalent construct of MION-46L and the HIV-1 tat-peptide. This peptide contains a membrane-translocating signal that efficiently shuttles MION nanoparticles into cells, and the particles accumulate in the nucleus. Magnetically labeled hematopoietic stem cells could be recovered following their homing to bone marrow, and could be visualized ex vivo by MR imaging of tubes containing bone marrow smears. Using MR imaging, homing of magnetically labeled lymphocytes could be visualized in the liver and spleen (Dodd et al., 2001), and in the pancreas after an adoptive transfer in diabetic mice (Moore et al., 2002). Our group used a covalent construct of OX-26, an internalizing antitransferrin receptor monoclonal antibody (Jefferies et al., 1984), and the dextran-coated MION-46L. On binding of the construct to the transferrin receptor, the complex is internalized and transported into intracellular endosomes, where the particles remain localized (i.e., are not further transported into the nucleus; Fig. 1) (Bulte et al., 1999b). CG-4 oligodendrocyte progenitors (Louis et al., 1992) were magnetically labeled by simply adding the MION-46L-OX-26 construct to the culture medium at doses ranging from 2 to 25 μg Fe/mL, and incubating for 48 hours (Fig. 2). This resulted in an efficient incorporation of the label into endosomes, as visualized using the Prussian blue histochemical staining for iron. Individual iron oxide particles could also be demonstrated using transmission electron microscopy (TEM), and a close association with the internalized cell membrane was observed. No uptake of iron oxide particles could be observed when MION-46L was incubated without conjugation of the OX-26 monoclonal antibody, even when a 20-fold higher incubation dose was used (Fig. 2). The T2 relaxation rates of labeled cells were highly increased as compared with nonlabeled cells, and it was found that the 1/T2 was dependent on the inter-echo time that was used. The longer we allowed for proton dephasing (i.e., the longer the echo time [TE]), the larger the effect on 1/T2. This effect cannot be observed for unbound (noncellular) SPIO particles in solution, and is important because for in vivo MR imaging longer TEs can be used, potentially increasing the sensitivity for detecting magnetically labeled cells. When cells were trypsinized and labeled in suspension for 2 to 6 hours (instead of labeling as monolayers for 48 hours at an 8-fold-lower incubation dose) the 1/T2 relaxation rate increased only marginally (Fig. 3). While the MION46L-OX-26 magnetic labeling procedure provides efficient labeling, however, a drawback is the fact that it is specific (i.e., monoclonal antibody-based) and suitable only for rat cells that express the transferrin receptor. A different antibody or labeling strategy would be needed to label, for instance, mouse or human cells.
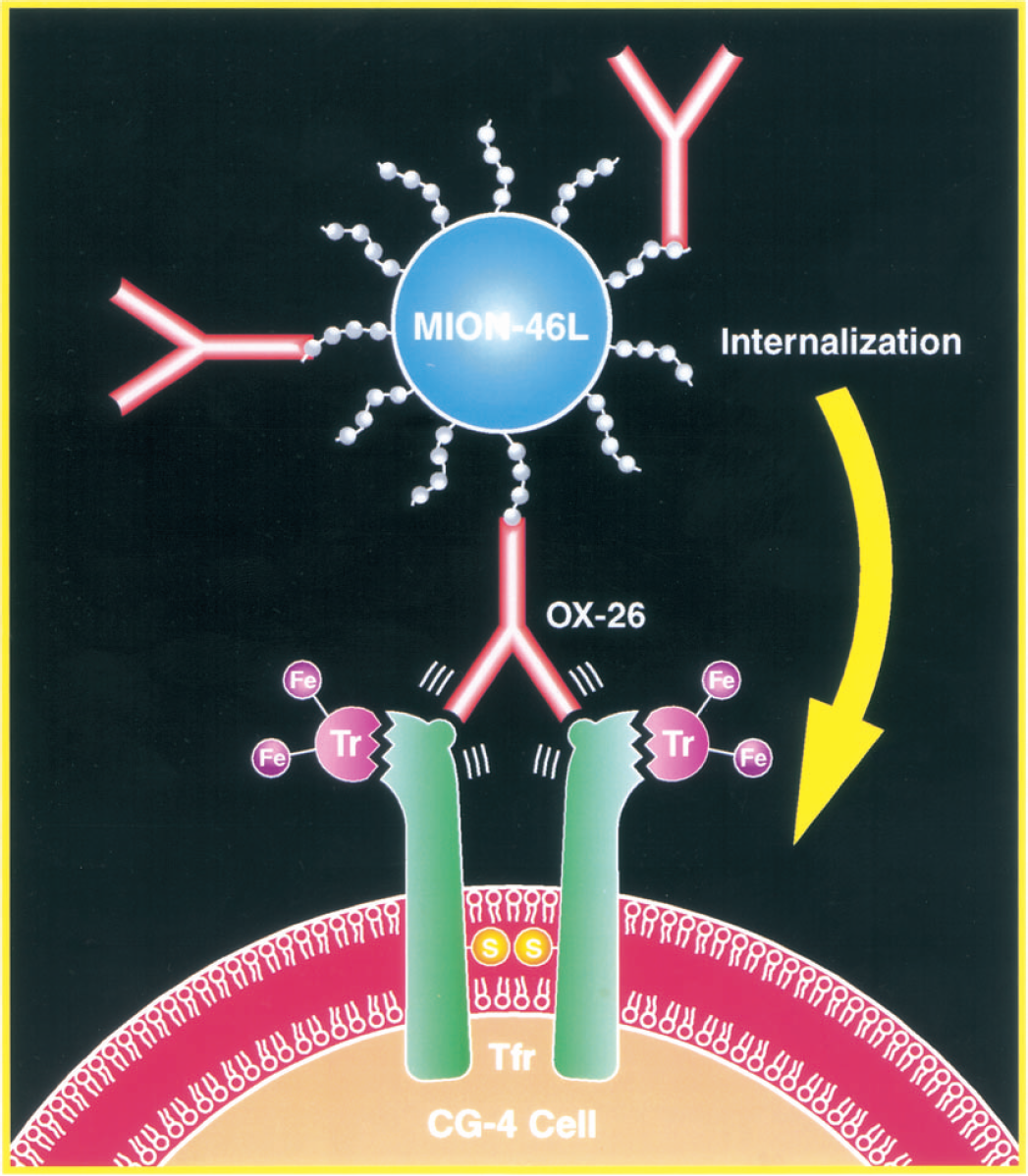
Internalization of an antitransferrin receptor mab-iron oxide particle construct. Oligodendrocyte progenitor (CG-4) cells express high numbers of the transferrin receptor (Tfr). After crosslinking of the Tfr by the specific OX-26 mab, the cell receives an internalizing signal to endocytose the Tfr-OX-26 complex plus the covalently linked MION-46L iron oxide nanoparticles used as magnetic label.
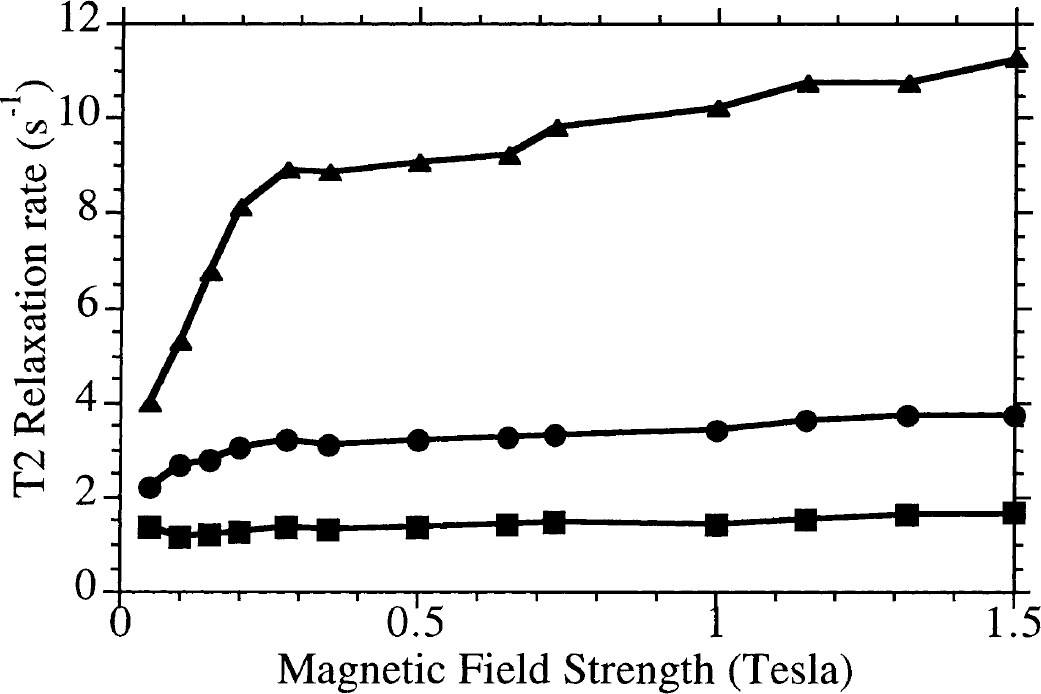
T2 (inter-echo time = 2 ms) relaxation rates of (squares) trypsinized oligodendrocyte progenitor (CG-4) cells labeled in suspension with 200 μg Fe/mL MION-46L-OX-26 for 2 hours, (circles) trypsinized CG-4 cells labeled in suspension with 200 μg Fe/mL MION-46L-OX-26 for 6 hours, and (triangles) nontrypsinized CG-4 cells labeled as monolayers with 25 μg Fe/mL MION-46L-OX-26 for 48 hours. All samples were adjusted to a density of 2 × 107 labeled cells/mL gel.
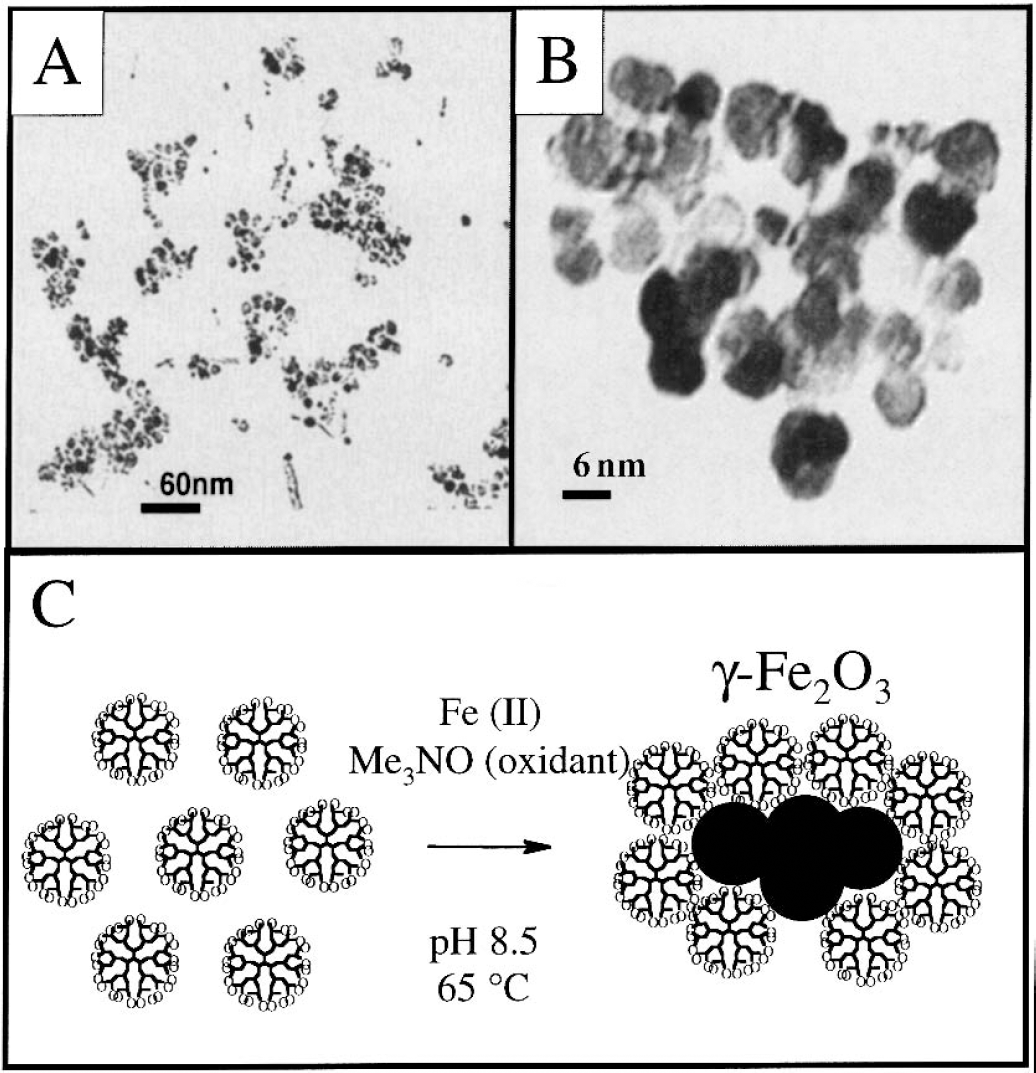
We recently developed a new magnetic labeling method based on the use of dendrimers as transfection agents (Bulte et al., 2001). Dendrimers are highly charged polymers that bind to cells nonspecifically and are internalized on membrane binding (Zhang and Smith, 2000). Dendrimers have been used successfully to shuttle or chaperone oligonucleotides into a wide variety of mammalian cells (Kukowska-Latallo et al., 1996;Tang et al., 1996;Plank et al., 1996;DeLong et al., 1997). We therefore sought to synthesize iron oxide—coated dendrimers, which can be taken up in a similar fashion, providing a universal, simple method of magnetically labeling cells regardless of their origin or species. These magnetodendrimers, MD-100 (so called because the dendrimers are initially loaded with 100 iron atoms per dendrimer), have now been synthesized (Strable et al., 2001), as shown in Fig. 4. TEM revealed an oligocrystalline structure of 7- to 8-nm crystals separated by a somewhat smaller distance. The particles were found to be highly magnetic, with a high saturation magnetization of 94 emu g/Fe, and no magnetic hysteresis at 300 K. These particles exhibit unusually high T2 relaxivities of 200 to 406 mmol/L−1 · s−1 (at 3 and 37°C, respectively), with a rapid approach to saturation at magnetic field strengths well below 1.5 tesla (T).
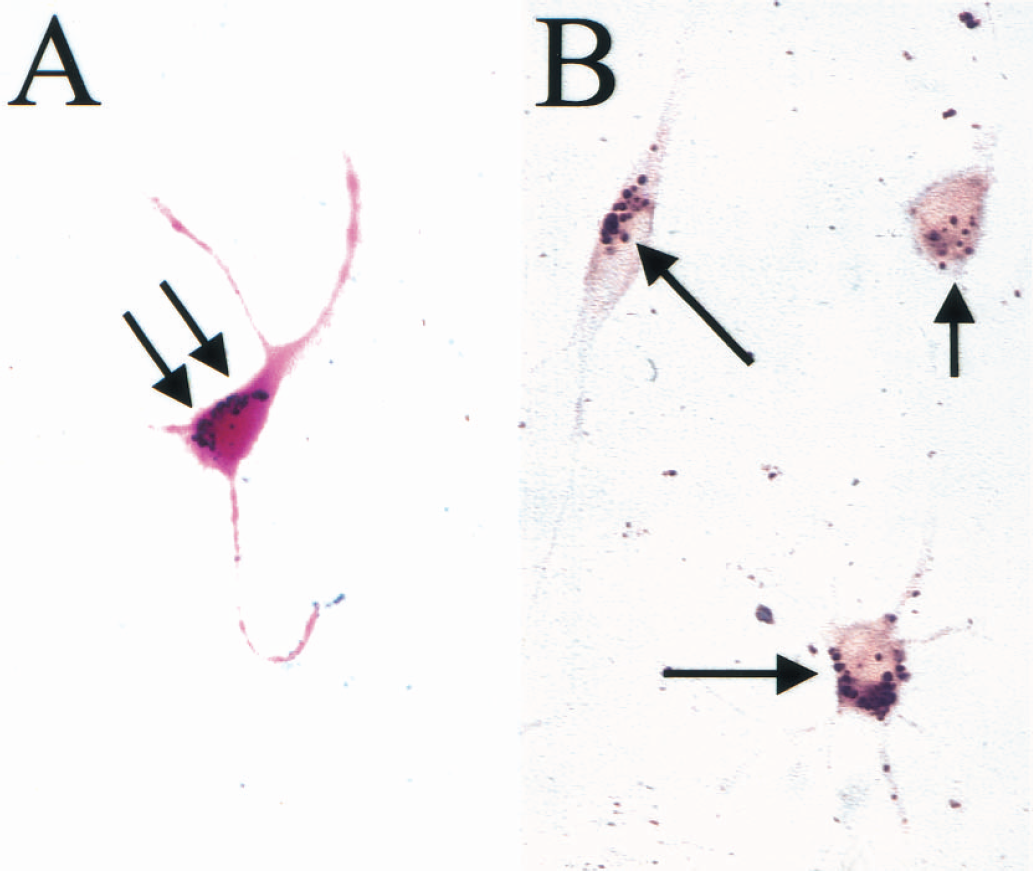
By simply adding MD-100 to the culture medium at concentrations of up to 25 μg Fe/mL and incubation periods of 1 to 2 days, a wide variety of mammalian cells can be highly labeled. This includes mouse ES cells, mouse 3T3 fibroblasts, mouse C2C12 muscle progenitor cells, rat CG-4 oligodendrocyte progenitors, rat NSC-derived oligodendroglial progenitors, human HeLa cervix carcinoma cells, human GLC-28 small cell lung carcinoma cells, human endothelial progenitor cells, human neural stem cells, human mesenchymal stem cells, and human ES cells. Prussian blue staining of magnetically tagged cells showed a remarkably high degree of intracellular labeling, with the cytoplasm containing large numbers of iron-containing vesicles or endosomes (Fig. 5). Achievement of intracellular labeling is a critical requirement, since a membrane-bound magnetic probe is likely to interfere with cell—tissue interactions (membrane-recognition processes), may detach easily from the cell membrane, or may be taken up and transferred to other cells in vivo. All cells show a comparable degree of uptake, demonstrating that the MD-100 uptake is nonspecific and not dependent on the cell type or species.
Non-enhanced
The total iron content of cells labeled with 25 μg Fe/mL MD-100 for 2 days is approximately 10 pg Fe/cell, whereas unlabeled cells contain about 1 pg Fe endogenous iron. Magnetic labeling with MD-100 appears irreversible, that is, the label is not excreted from the cells in vitro for at least 1 week after labeling. Labeled cells appear unaffected in their proliferation capacity and viability, and an MTT-based toxicity assay showed no difference between unlabeled cells and MD-100 labeled cells. Furthermore, MD-100–labeled human NSCs showed comparable formation of neuronal processes when replated and grown for an additional 10 days, and differentiated normally into glial cells and neurons.
Extrapolating from the idea of using a dendrimer transfection agent to shuttle SPIO particles into cells, as in the case of magnetodendrimers, we hypothesized that it may also be possible to use other transfection agent and SPIO combinations in a similar fashion. Indeed, we have shown that it is possible to coat the SPIO Feridex (Berlex Laboratories, Wayne, NJ, U.S.A.) with a variety of transfection agents such as heat-activated dendrimers, poly-l-lysine, and lipofectamine (Frank et al., in press). Following incubation of mouse, rat, or human cells at similar doses as that used with MD-100, we have found a comparable rate of uptake both in cellular localization (non-nuclear, in endosomes) and total amount of iron. This latest development has two major implications and advantages over other magnetic labeling techniques using SPIOs. First, since both the SPIO and transfection agent are commercially available, anyone interested in the magnetic labeling technology can have access to these compounds; and second, since an FDA-approved iron oxide (i.e., Feridex) can be used, it is expected to facilitate the introduction of MR tracking of magnetically labeled cells in a clinical setting.
MAGNETIC RESONANCE TRACKING OF MAGNETICALLY LABELED CELLS IN THE CENTRAL NERVOUS SYSTEM
Several groups have reported that it is possible to visualize magnetically labeled cells in the brain after transplantation. This includes rat fetal brain cells labeled with SPIO linked to the lectin wheat germ agglutinin (Norman et al., 1992), rat fetal brain tissue labeled with SPIO-containing reconstituted Sendai virus envelopes (Hawrylak et al., 1993), and CG-4 oligodendrocyte progenitor cells labeled with plain dextran-coated SPIO (Franklin et al., 1999). In these studies, the MR contrast remained primarily localized at the injection site, and no widespread migration of cells was observed.
We have labeled CG-4 oligodendrocyte progenitors according to the method shown in Fig. 1. Following transplantation of approximately 5 × 104 CG-4 cells (labeled with MION-46l-OX-26 at 12 μg Fe/mL for 48 hours) into the spinal cord of 7-day-old md rats (Bulte et al., 1999b), the cords were removed 10 or 14 days later and imaged ex vivo at 78-μm resolution using a 4.7-T imaging spectrometer. Cells were found to migrate primarily along the dorsal column, over a distance of up to 10 mm away from the injection site. Since multi-gradient-echo imaging can be performed under collection of a three-dimensional (3D) data set, one can not only analyze the tissue distribution of labeled cells in each of the three planes (x, y, and z), but also reconstruct a true 3D image (Fig. 6). A good agreement between the areas of MR contrast enhancement and histopathologic stainings for iron and newly formed myelin was demonstrated.
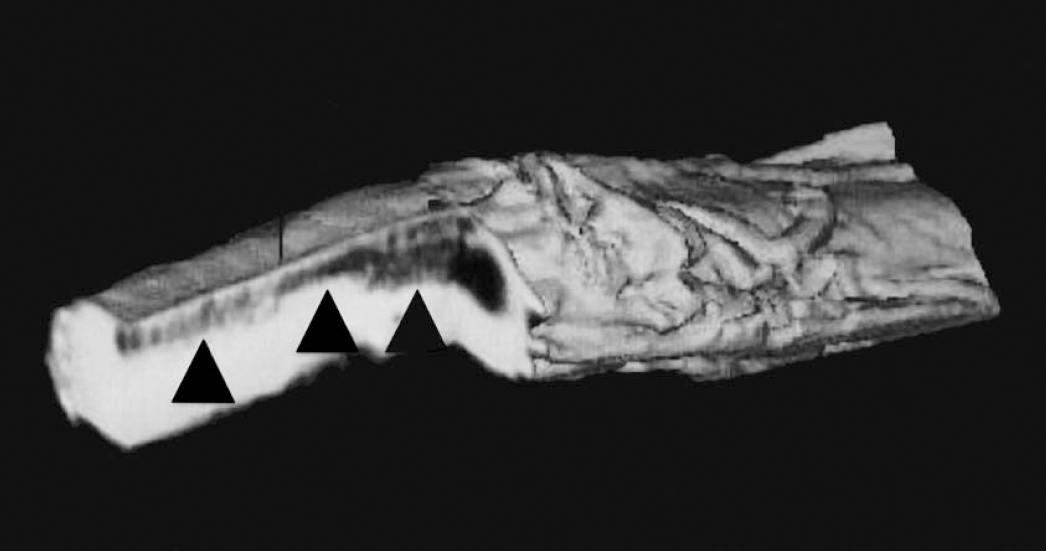
Three-dimensional reconstructed ex vivo magnetic resonance image (78-μm resolution) of myelin-deficient rat spinal cord showing distribution of magnetically tagged oligodendrocyte progenitors 10 days after transplantation. Note the migration along the dorsal column (arrowheads) away from the injection site.
Using MD-100, approximately 5 × 104 magnetically labeled NSC—derived rat oligodendroglial progenitors were transplanted into the ventricles of neonatal (P = 0) Long Evans shaker rats (Bulte et al., 2001). These animals are dysmyelinated mutants resulting from a defect in the gene encoding for myelin basic protein (O'Connor et al., 1999), with a life span of a few weeks longer than the md rat. Cells were co-transfected with the LacZ gene (encoding for the enzyme β-galactosidase), to track them histochemically by incubating with the X-gal enzyme substrate. Migration of labeled cells into the brain parenchyma could be observed at the earliest time points (between 2 and 3 weeks) throughout the latest time points of imaging (6 weeks after transplantation). There was a good gross anatomic correlation with the macroscopic distribution of β-galactosidase—expressing cells. Moreover, the transplanted and labeled cells were also found to be able to form new myelin that overlapped with the area of MR contrast. Figure 7 demonstrates an example of a 3D reconstructed MR image obtained in vivo at the latest time point of imaging (6 weeks). While this image was obtained at high resolution using a dedicated high-field (4.7-T) animal scanner, the labeled cells could also be readily identified in vivo using a conventional 1.5-T clinical imaging system. A comparison of several pulse sequences demonstrated that T2*-weighted gradient-echo imaging, which does not compensate for the induced dephasing of protons, was most sensitive to the presence of labeled cells. From the number of cells injected, and the overall area of contrast, we estimate that it should be possible to detect only a few cells when using T2*-weighted imaging techniques.
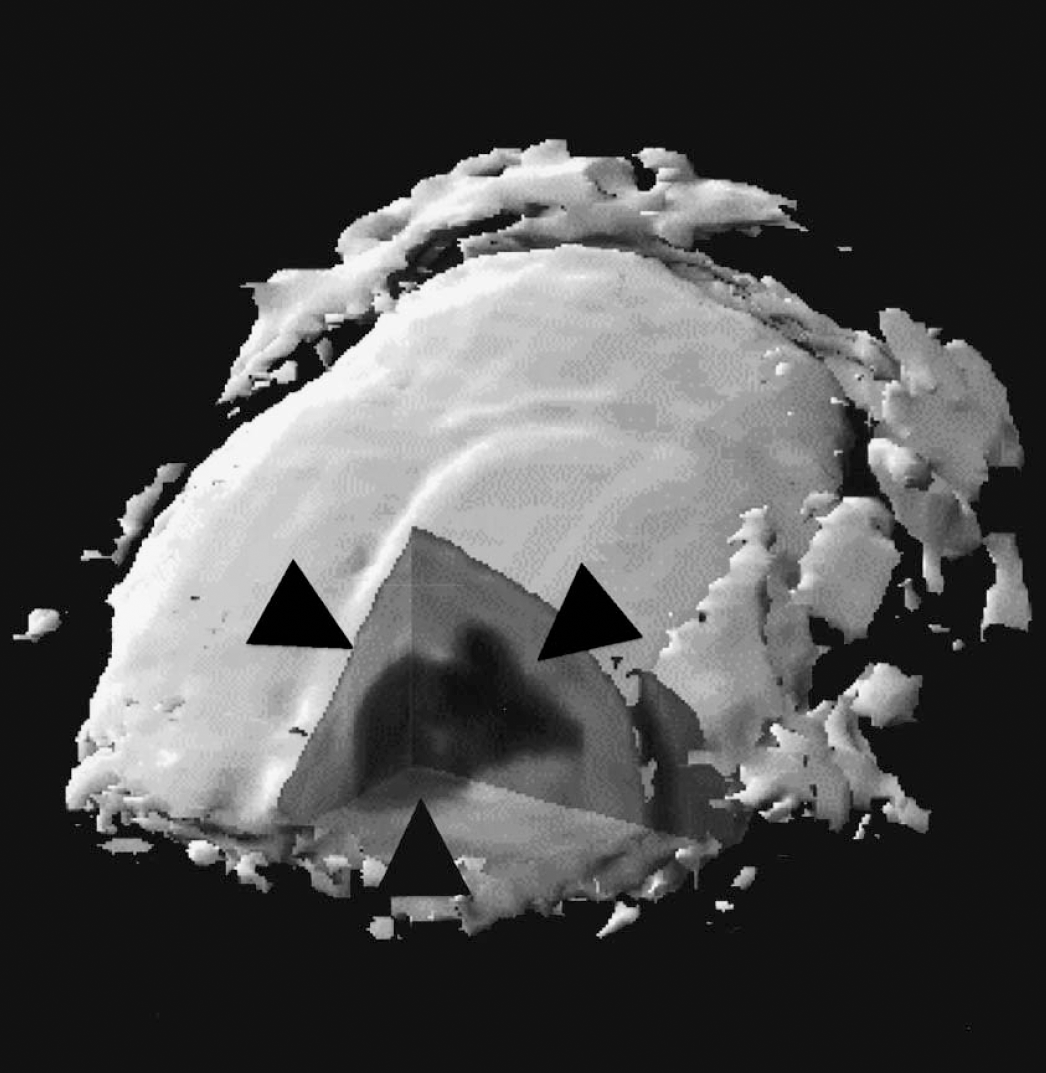
Three-dimensional reconstructed in vivo magnetic resonance image (313-μm resolution) of Long Evans shaker rat brain showing distribution of magnetically tagged oligodendroglial progenitors at 6 weeks after transplantation. Note the migration of cells into the parenchyma (arrowheads) away from the ventricle.
CONCLUSIONS
It has now been demonstrated that stem cells and progenitor cells can be magnetically labeled with derivatized SPIO particles in such a manner that they can be detected by MR imaging. This has allowed an accurate tracking of their tissue biodistribution following transplantation. Further studies are warranted to investigate the longevity and biodegradability of the magnetic probes, which may be different for different cell types. The exact number of cell divisions following which cells become undetectable will also require detailed further studies. In the meantime, the prospect of MR tracking of labeled cells appears attractive not only as a tool to perform longitudinal studies in the same animal, but also because of its potential to help guide future clinical studies involving the use of therapeutic stem and progenitor cells.
Footnotes
Acknowledgments
The authors are grateful to Trevor Douglas, Bobbi Lewis, Peter Van Gelderen, Brian Witwer, and Su-Chun Zhang; without their help the experiments described here could not have been conducted.
