Abstract
Magnetic resonance imaging (MRI) is based on a wide variety of physical parameters, which, in principle, can all influence the image contrast conditions. As these diverse variables are validated by independent physiological, metabolic, hemodynamic, and histological techniques, a physiological MRI evolves. This imaging modality has been successfully applied to experimental stroke studies, covering a broad range of raised questions. In the present review, we present an overview of possible physiological criteria to be studied by in vivo MRI and magnetic resonance spectroscopy, and critically analyze the present limits and future potential of the imaging technique for experimental stroke investigations. The documented applications cover the spectrum from morphological-structural details of the lesion to hemodynamic and metabolic alterations, inflammatory reaction, evaluation of thrombolytic treatment, studies on recovery of functional brain activation by functional MRI, and, finally, the most recent applications of exploring stem cells for regenerative therapy.
Introduction
Ischemic stroke is a sudden event, most often without prior warning. But after this primary onset, the disease has a very pronounced dynamical evolution, over the first few hours, during the so-called therapeutical window, but also during a long-term chronic phase (Back and Schuler, 2004; Fisher and Garcia, 1996; Garcia et al, 1996). To understand the spontaneous disease evolution as well as the effect of therapeutical interventions, a noninvasive imaging modality is highly desirable. This serves two purposes: the imaging aspect helps to localize and characterize the regional extent and spatial variability of the ischemic brain injury. The noninvasiveness of the technique is of paramount importance as it provides the opportunity to observe the dynamics of the pathophysiological processes within the individual, thus establishing a true and even individuum-based temporal profile. There are several imaging techniques available as of today, applied in the clinical setting of stroke diagnosis: Computed tomography (CT) is still the most frequently used imaging modality today, a fact that is related to its higher availability and lower costs compared with magnetic resonance imaging (MRI), or radio-tracer-based positron emission tomography (PET) and single-photon emission CT (SPECT) imaging. For the experimental investigations on small laboratory animals, in particular rodents, MRI has proved to be the dominantly used and preferable imaging modality due to its high spatial resolution in combination with the sensitivity and capability to depict various morphological and functional aspects.
With the rapid technological development, MRI has been applied extensively for experimental stroke studies since the late 80s. The real breakthrough came with the original presentation by Moseley et al (1990), showing the extreme sensitivity of diffusion-weighted MRI to detect the acute cerebral ischemia, within minutes after stroke onset. The aim of the present review is not an update of the understanding of experimental stroke using MRI. For this purpose, the readers are referred to reviews published over the last several years (Dijkhuizen and Nicolay, 2003a; Hoehn et al, 2001). Instead, the goal of the present review is a critical analysis of the value and contribution of MRI and magnetic resonance spectroscopy (MRS) for investigations into experimental stroke, both in terms of diagnostic use and in terms of understanding the pathophysiological developments and their therapeutical interventions. To further focus the topic, we have concentrated this review on experimental ischemic stroke investigations.
There are several animal models for experimental stroke studies available and characterized in the scientific literature (Kohno et al, 1995; Longa et al, 1989; Olah et al, 2000), covering various occlusion techniques of the middle cerebral artery (MCA) by electrocauterization, by vascular ligation, by the widely used intraluminal filament technique, and finally by thromboembolic clot occlusion. Furthermore, models exist for permanent middle cerebral artery occlusion (MCAO) as well as for transient focal cerebral ischemia (for review, see Hoehn et al, 2001; Hossmann, 1998; McAuley, 1995). Apart from the basic requirement of having a disease-relevant animal model, the experimental conditions under which the imaging data have been recorded are of great importance. To assure stable physiological conditions during the whole, and in some cases repetitive, imaging sessions, the animals need to be closely monitored for various essential life functions. Where necessary, the parameters need to be adjusted to remain within the normal range for physiologically relevant conditions. As the animals must be anesthetized during MRI sessions, selection of a well-tolerated anesthetic is important, as well as continuous recording and maintenance of normal body temperature. Further, blood pressure monitoring and regular blood gas analysis are indispensable to avoid hypotension or acidosis as confounding factors for the determination of the stroke development and its severity. In some particular investigations, combination of the imaging data with cerebral blood flow (CBF) measurement by laser-Doppler flowmetry (LDF) (Dirnagl et al, 1989) and with recording of electrocorticogram (Kohno et al, 1995; Norris et al, 1998) might be desirable. Only when these aspects, selection of the relevant model and monitoring of physiological functions, are assured can a meaningful assessment of the role and contribution of the applied imaging modality be made.
We will begin our review by analyzing the diagnostic potential of MRI for the dynamics of experimental stroke. Then, in the second part, assess the contribution experimental studies have made to the understanding of mechanistic aspects of the disease, and, in the third part, consider the role of MRI in the assessment of therapeutic strategies.
Diagnostic Aspects
Diffusion and Relaxometric Imaging
The most spectacular application of MRI to experimental (and probably also clinical) ischemic stroke is certainly the diffusion-weighted MRI (DWI). As this modality is sensitive to intercompartmental water shifts, it monitors disturbances of the water and ion homeostasis. This is one of the earliest events in the cascade of pathophysiological steps after interruption of the CBF. This change of the apparent diffusion coefficient (ADC) on intercompartmental water shifts is usually explained by the increased intracellular diffusion restriction and the fact that the ADC measures a volume-weighted average of the intra- and extracellular diffusion coefficients. Thus, since its first description by Moseley et al (1990), DWI has become a major tool for the detection of the ischemic territory and for the characterization of the acute and subacute development of stroke.
The diffusion change has been shown to depend on the severity of the ischemic lesion (Hoehn-Berlage et al, 1995, 1997). The possibility of an empirical determination of a diffusion coefficient threshold, to differentiate between the ischemic core and a surrounding, metastable penumbra region, has been shown by comparing quantitative diffusion magnetic resonance (MR) imaging with independent, invasive regional biochemical mapping techniques (Hoehn-Berlage et al, 1995; Kohno et al, 1995). However, an early study reported a CBF threshold reflecting the ischemic core necessary for a diffusion change to manifest itself and become detectable, thus contradicting the concept of combined core-penumbra diffusion changes (Busza et al, 1992). In support of the gradual ischemic changes of a penumbra concept, a relationship between CBF decrease and amount of diffusion coefficient change was reported (Miyabe et al, 1996). A similar situation had been described in a rat focal cerebral ischemia model (Hoehn-Berlage et al, 1995). These authors discussed the gradual shrinkage of the extracellular space with increasing ischemia severity (i.e. increasing CBF reduction) as a possible explanation for the empirical relationship between CBF decrease and ADC reduction. Thus, although there is fundamental agreement that the DWI is the most sensitive MR imaging technique to depict the ischemia affected territory very early-on, more investigations would be helpful to support the notion that quantitative diffusion coefficient changes can be successfully applied for the essential distinction between the ischemic core and a potentially salvageable penumbra zone.
For a long time, T1- and T2-weighted MR imaging was considered useful for the description of the mature lesion and of the vasogenic edema, but was not believed to be sensitive enough for the early detection of the acute ischemic tissue damage. Recent studies using quantitative relaxometric MR imaging (i.e. T1-, T2-mapping, and T1rho) indicate that these parameters are indeed much more sensitive for the early damage detection than anticipated (Calamante et al, 1999; Grohn et al, 2000), in particular for the early (intraischemia) determination of later damage developing during reperfusion, and becoming manifest on DWI only several hours later (Olah et al, 2000). Very recent studies by Wegener et al (2005a) showed the potential to use a temporal profile of T2 changes in the chronic phase to differentiate between pannecrosis and selective neuronal death in the subcortical tissues. Thus, careful analysis of the small relaxometric changes detectable only with quantitative parameter mapping points towards a revival of relaxometry for experimental stroke studies and an increasingly important role for T1- and T2-maps for the diagnosis of both early determined lesions and of late tissue damage severity (also relating to behavioral deficit data).
Blood Flow Measurements: Angiography and Perfusion Imaging
While the effect of vascular occlusion on tissue during thromboembolism is visualized by the ensuing diffusion changes in the ischemic territory, analysis of successful recanalization after induced thrombolysis requires the depiction of the vascular architecture in the brain. Magnetic resonance angiography (MRA) is capable of presenting the circle of Willis of rats and mice and even smaller side-branches of the MCA in 3D data sets within a few minutes (Beckmann et al, 1999; Besselmann et al, 2001; Reese et al, 1999). This allows in-vivo localization of the blood clot obstructing the blood flow. As MRA is achieved without use of contrast agent through the use of the time of flight (TOF) method and the recording time is short, repetitive data registration to obtain a temporal profile or to test response to vascular therapies (thrombolysis or mechanical clot destruction) becomes feasible (Hilger et al, 2002). Furthermore, using angiography, lysis of the blood clot and collateral flow as separate mechanisms for tissue perfusion improvement after therapeutical intervention can be distinguished. Also, lack of tissue recovery (determined by persistent DWI changes) despite observed tissue perfusion status can thus be explained by a too late onset of recanalization. This hemodynamic information is particularly helpful as ADC may also show various transient normalization patterns making early diagnosis of the lesion evolution and its recovery, respectively, rather difficult when based on ADC alone (Olah et al, 2000).
Due to the challenge of spatial resolution and signal-to-noise ratio, a characterization of the vascular patency beyond the binary statement ‘open’ or ‘occluded’ is presently difficult. Further improvement in sensitivity to distinguish between a completely or partially occluded MCA after thromboembolism would help to register ‘trickle flow’ through a narrowed lumen.
Assessment of tissue perfusion with MRI (perfusion-weighted imaging (PWI)) can be achieved with two alternative approaches: the contrast agent-based bolus track imaging and the spin label imaging method. As the former technique depends on addition of contrast agent, clearance from the blood stream must be awaited before injection of the next bolus to prevent systemic contrast agent saturation. Clearance in rats has been described within 30 mins of application (Rudin and Sauter, 1991), so that quantitative measurements should be possible on an hourly basis. The latter method is contrast agent independent and can be repeated continuously without loss in sensitivity. Both methods have reliably shown depiction of tissue perfusion and even differences in tissue perfusion between gray and white matter. On a qualitative level, both techniques can depict the area of perfusion deficit after stroke induction and also present perfusion improvement after recanalization of the formerly obstructed vessel (Hilger et al, 2002; Niessen et al, 2002). Gradual changes between severe perfusion deficit in the ischemic core and light perfusion reduction in the ischemic periphery have been reported using arterial spin-labeling methods (Busch et al, 1998). However, reliable quantification of reduced cerebral blood flow under ischemic conditions is difficult and has not been successfully shown—except in a validation study of bolus track imaging with CBF autoradiography (Wittlich et al, 1995). This problem is based on the fundamental issue that tissue regions of reduced flow exhibit also a reduced signal intensity so that with increasing perfusion deficit the detectability is rapidly compromised. Furthermore, reduced flow velocity leads to further modulation of the MR signal intensity, thus making quantification even more difficult. A further caveat relating specially to the ASL method is the need for repetitive quantitative T1 measurements during the evolution of the ischemic damage. This is necessary as the ASL-based CBF quantification requires the knowledge of T1 relaxation time, which however increases continually in the ischemic tissue during the acute phase. Nevertheless, despite such methodological problems of perfusion quantification, the techniques deserve their firm place in experimental ischemia studies: their strength lies in their registration in combination with other imaging modalities. Perfusion and diffusion imaging together will distinguish tissue regions with perfusion reduction, yet without serious tissue damage (no DWI change yet), thus showing ‘tissue-at-risk areas’. Further, perfusion imaging will characterize the effect and spatial extent of perfusion deficit after vascular obstruction or allow evaluation of thrombolytic therapy. In combination with both DWI and MRA, distinction between reperfusion of still viable tissue in penumbral areas (nutritive blood flow) and reperfusion of already dead tissue (nonnutritive blood flow) becomes feasible.
Proton Magnetic Resonance Spectroscopic Imaging and pH-Sensitive Magnetic Resonance Imaging
Changes in cerebral metabolism on experimental focal and global brain ischemia have been studied in vivo using longitudinal proton MR spectroscopy studies (Brulatout et al, 1996; Dreher et al, 2001; van der Toorn et al, 1994). With the appearance of fast techniques of proton magnetic resonance spectroscopic imaging (MRSI), higher spatial resolution became available and metabolic changes could be followed in the whole rodent brain. The most frequently assessed metabolites in proton MRSI are lactate, which accumulates as the end product of anaerobic glycolysis, and decreasing levels of N-acetyl-aspartate (NAA), which is considered to be dominantly a neuronal marker (Birken and Oldendorf, 1989; Simmons et al, 1991). Thus, reduction of NAA is often proposed to represent loss of neurons (Bruhn et al, 1989), but alternative explanations such as metabolic degradation of NAA or redistribution must be further analyzed (Franke et al, 2000). By combining DWI and MRSI, regional differences in lactate formation after infusion of 13C-labeled glucose could be detected after permanent MCAO in rats to better delineate potentially salvageable ischemic tissue (Dijkhuizen et al, 1999). No enrichment of 13C-labeled glucose was seen in the ischemic core, where the ADC already was dramatically reduced after 30 mins. In the border zone of the ischemic territory, the initial ADC decrease was not as pronounced as in the core region, but showed a further decline between 30 mins and 3 h. In contrast to the ischemic core, 13C was metabolized to lactate in the border zone, indicating metabolically impaired, but potentially salvageable, brain tissue.
Serial MRSI measurements were also used to study the effect of thrombolytic therapy with recombinant tissue-type plasminogen activator (rtPA) on metabolic changes in a thromboembolic rat stroke model (Franke et al, 2000). Successful reperfusion indicated on PWI in animals treated after 1.5 h resulted only in a slight decrease of the lactate concentration, while lactate concentration did not decline when treatment was started 3 or 4.5 h after embolism. Decreased NAA levels did not show a recovery after thrombolytic treatment during the whole observation period of 8 h after induction of embolism (Franke et al, 2000).
To achieve sufficiently good spatial and spectral resolution for application to spatially heterogeneous pathologies like stroke, the MRSI method must be fast enough. Otherwise, data acquisition (at mostly required signal averaging) at the low metabolite concentrations becomes prohibitively long. When combining ultrafast techniques with MRSI modules, even transient focal metabolic changes can be observed. Using UFLARE spectroscopic imaging, Norris et al (1998) achieved a temporal 4-mins resolution, while the voxel dimensions (32 × 32 matrix across the rat brain) were 1 mm × 1 mm × 3.5 mm. With such excellent temporal and spatial resolution, these authors were able to detect a transient lactate rise during spontaneously occurring spreading depressions (SD) in the ischemic periphery. The time profile of lactate rise and normalization coincided well with the transient diffusion coefficient decrease, earlier reported to be a reliable indicator of the passage of an SD wave.
It should be noted that spectroscopy and spectroscopic imaging both face the problem of quantification of metabolite concentration. Usually the statements are limited to relative concentration changes, in reference to preischemic values (if time series data are available). However, even relative changes often provide important information for the understanding of metabolic alterations and recovery, or even transient metabolic changes in response to pathophysiological events. Another unsolved limitation of MRS/MRSI is the few NMR-visible metabolites of relevance to ischemia studies and sufficient concentration for reliable detectability.
Recently, Zhou et al (2003b) reported a new, elegant and innovative approach to image noninvasively tissue acidosis using MRI and applied it to experimental stroke. The method, called amide proton transfer (APT) contrast MRI (Zhou et al, 2003a), is based on magnetization transfer effects between the water protons and amide protons of mobile intracellular proteins to detect intracellular pH changes. Although pH quantification showed a relatively wide pH range between 7.0 and 7.4 in normal brain areas, postischemic tissue acidosis (pH 6.52 ± 0.32) was clearly visible on pH-sensitive images and matched the area of decreased perfusion on PWI (Figure 1).
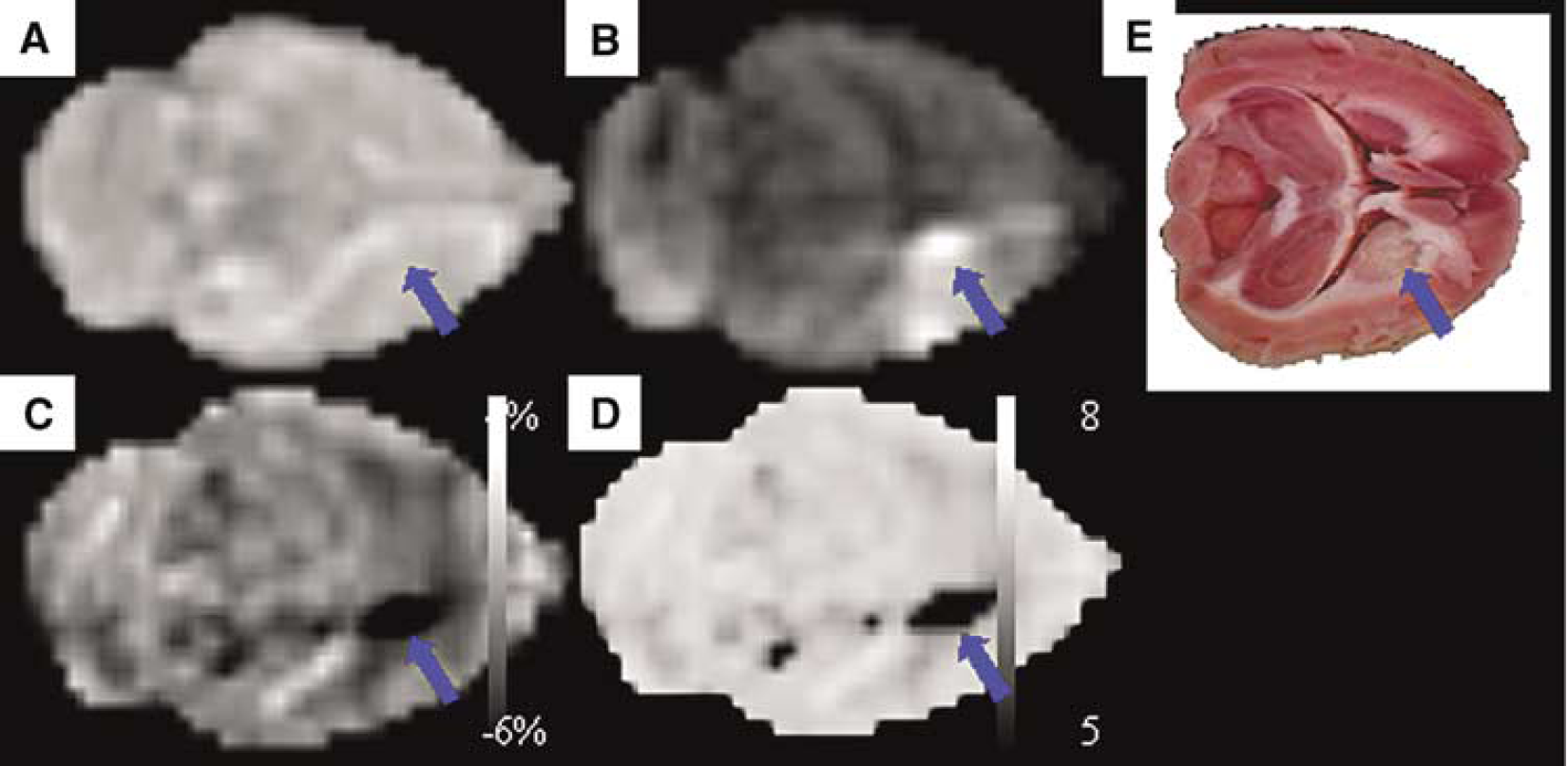
Comparison of horizontal T2-weighted image (
Mechanistic Aspects
Characterization of Stroke Evolution in the Hyperacute and Acute Phases: Diffusion-Weighted, 23Na and Manganese-Enhanced Magnetic Resonance Imaging
Multimodal MR imaging has become the standard diagnostic procedure to determine the location and extent of focal ischemic brain injury after occlusion of an arterial blood vessel. As stated above, DWI might be considered the gold standard MRI technique to visualize the acute ischemic brain tissue. The observed signal increase on DWI is caused by a decline in the ADC of water, presumably due to a reduction of free water diffusion. A major focus of research in the past years was to better characterize the temporal and spatial evolution of the ADC decline during the hyperacute and acute phases in animal stroke models and to relate the ADC changes to recognized pathophysiological, biochemical, and hemodynamic events.
A reduced ADC of water can be detected within minutes after the onset of ischemia (Davis et al, 1994; Hoehn-Berlage et al, 1995; Moseley et al, 1990) and is most likely caused by a cytotoxic edema through a shift of extracellular water into the damaged cells after the failure of Na+ /K+ ATPase ion pumps (for review, see Verheul et al, 1994; Hoehn et al, 2001). By combining serial ADC measurements with invasive techniques of ATP, glucose, and pH quantification, a distinction between a core region with a complete breakdown of energy metabolism and surrounding ischemic tissue with normal ATP distribution, but severe tissue acidosis, was established on quantification of the ADC. The ADC threshold for ATP depletion was found to be 77% after 1 or 2 h of MCAO and did not change with 1 or 10 h of reperfusion, whereas the ADC threshold for tissue acidosis was 86% directly after 1 h of MCAO and further decreased to 78% and 76% after 1 and 10 h of reperfusion, respectively (Hoehn-Berlage et al, 1995; Olah et al, 2001). This apparent lowering of the acidosis threshold is because in the beginning the ischemic core (depletion of energy rich metabolites) is surrounded by a region of normal ATP levels, but with anaerobic glycolysis. With increasing time the ischemic core extends into the periphery till it becomes identical with the whole ischemic territory (original core and periphery). Dependent on the duration of focal ischemia and reperfusion, a (pseudo-)normalization of the ADC (Knight et al, 1994) and secondary deterioration can be observed (Li et al, 1999, 2000a; Olah et al, 2000; Ringer et al, 2001). After a short transient ischemia of 10 mins, the initial ADC decline was reversed almost immediately after reperfusion and remained normal thereafter. Nevertheless, histology showed neuronal necrosis in the affected ischemic territory (Li et al, 2000a). On longer occlusion times between 30 and 60 mins, (pseudo-)normalized ADC values started to decrease again as early as 2 h (Li et al, 2000a, b; Olah et al, 2000), accompanied by a signal increase on T2-weighted images, although no hypoperfusion was seen on PWI (Olah et al, 2000). Histology showed again neuronal damage in the area of secondary ADC decline (Li et al, 1999, 2000b) and expression of the heat shock protein (HSP)-72 in the area of primary ADC decline (Ringer et al, 2001).
Owing to of the dynamic and variable temporal evolution of the ADC signal found in transient ischemic brain lesions, attempts were made to establish MRI-based methods to better delineate brain tissue, which will become infarcted from potentially salvageable brain tissue using high-resolution 23Na MRI (Lin et al, 2001; Thulborn et al, 1999) and manganese-enhanced MRI (Aoki et al, 2003, 2004).
The fact that Na+ ions accumulate in cells on ischemia-induced failure of the Na+ /K+ ATPase ion pumps was used by two groups to quantify tissue viability in rats (Lin et al, 2001), and in a small study with two nonhuman primates (Thulborn et al, 1999). Regions of increased 23Na correlated well with the triphenyltetrazolium chloride (TTC) measured infarct size from day 0.5 to day 14 in a transient three vessel occlusion MCAO model of the rat, and no transient normalization was observed in contrast to ADC and T2 maps (Lin et al, 2001). However, in contrast to DWI, 23Na MRI did not consistently detect the ischemic brain region in most of the animals 6 h after stroke induction. Thus, although 23Na MRI cannot replace DWI in the detection of hyperacute ischemia, it could have a place as predictor for irreversibly injured brain tissue, because no transient normalization was observed.
Besides Na+ accumulation, an influx of Ca2+ ions occurs during anoxic depolarization through Ca2+ channels among others. This influx through the Ca2+ channels was recently exploited for the early detection of stroke by manganese-enhanced MRI (Aoki et al, 2003, 2004). Intravenously applied Mn2+ ions can enter ischemic glial cells and neurons through these Ca2+ channels. The hyperintense area in the ischemic caudoputamen and cortex detected on T1-weighted MRI was much smaller compared with the area of reduced ADC in a model of permanent MCAO in the rat (Aoki et al, 2003). Unfortunately, no histological analysis was performed in this study to further investigate and interpret this discrepancy. One drawback of these studies lies in the need for intravenous application of osmotically active substances like mannitol to break the blood-brain barrier. The artificial disruption of the blood-brain barrier raises the risk of an aggravation of vasogenic edema formation and must therefore be considered with caution. However, a recent study by Lee et al (2005) has shown that MnCl2 can enter the intact brain via the ventricles and spread from there throughout the periventricular tissues.
Differentiation Between Complete and Incomplete Infarction in the Chronic Stroke Phase by Magnetic Resonance Imaging
Dependent on duration and severity of focal cerebral ischemia, two types of ischemic lesions can develop in the human and animal brain. The term ‘ischemic stroke’ is referred to an area of coagulation necrosis, later replaced by a cavity filled with cerebrospinal fluid (Garcia et al, 1996). In the acute stage of the developing ischemic infarct, an increase in T2 relaxation time is caused by the vasogenic edema (Neumann-Haefelin et al, 2000), which is fully developed after 24 h. Similar to the (pseudo-)normalization of the acute ADC decline, increased T2 values can transiently normalize in the subacute stage after transient MCAO, most likely because of a normalization of brain water content (Lin et al, 2002), but can subsequently increase again in the chronic stage due to cavitation formation. Thus, T2-weighted MRI has been used to quantify final infarction volume in animal stroke studies (Palmer et al, 2001; van Dorsten et al, 2002).
For short ischemic periods of minimal to moderate severity, incomplete infarction develops with delayed selective neuronal necrosis instead of pannecrosis and cavitation, a situation observed both in human and animal studies (Garcia et al, 1996, 1997). In recent years, longitudinal multimodal MR imaging has been used to characterize this selective neuronal necrosis in vivo after different occlusion times in transient focal ischemia models of the rat (Fujioka et al, 1999, 2003; Wegener et al, 2005b), emphasizing the advantage of longitudinal MR imaging in animal stroke studies to distinguish between the two ischemic lesion types. In Wistar rats subjected to 15 mins of MCA occlusion, a delayed hyperintense signal on T1-weighted images was observed in the ischemic striatum, matching with areas of selective neuronal necrosis and concomitant chronic glial and inflammatory reaction (Fujioka et al, 1999, 2003). Rats with such a delayed striatal neurodegeneration showed late-onset cognitive and/or behavioral declines.
A different MRI pattern was observed in Wistar rats subjected to a longer period (60 mins) of transient MCAO (Wegener et al, 2005a, b). In the group of rats with cortico-subcortical lesions, ischemic stroke develops and quantitative T2 values were strongly increased both 1 and 14 days after MCAO and these animals showed significant sensorimotor deficits in functional testing (Wegener et al, 2005b). Histology showed pannecrosis and cavitation formation. A second group of animals displaying only a subcortical lesion had increased T2 values in the striatum 1 day after MCAO, but T2 values had normalized again after 14 days and no functional deficit was observed at any time point in these animals, although histology showed selective neuronal necrosis, a strong glial and inflammatory response, but no cavitation formation. No PWI or DWI was performed in these aforementioned studies to address the important questions of the magnitude of perfusion deficit and the severity of acute lesion.
Magnetic Resonance Imaging Detection of Endothelial Activation and Macrophage Activity After Cerebral Ischemia
Ischemic brain injury not only leads to cell death through energy depletion but also to a postischemic inflammatory process which contributes to a delayed progression of the injury (Dirnagl et al, 1999). As a consequence, attempts were made to visualize the dynamic inflammatory response and coregister the MRI findings with invasive histological data.
Leukocyte infiltration of the ischemic brain is mediated by P- or E-selectin expression on activated endothelial cells. Using a new selective MR contrast agent, coupling the carbohydrate antigen Sialyl Lewisx, which is found on the surface of leukocytes, to Gd-DTPA, Barber et al (2004) were able to visualize the early endothelial activation on T1 maps and invasive fluorescence microscopy in the first hour after stroke induction.
Schroeter et al (2001) were not able to delineate areas of phagocyte accumulation with the help of a multiparametric MRI protocol, comprising ADC-mapping, T2-mapping, PWI, and Gd-DTPA-enhanced T1-weighted MRI in the first 14 days after photothrombotic stroke induction. However, combining high-resolution T2- or T*2-weighted MRI with the systemic administration of superparamagnetic iron oxide particles (SPIO), several groups were able to monitor the dynamics of the invasion of blood-borne macrophages into the ischemic brain tissue (Kleinschnitz et al, 2003; Rausch et al, 2001, 2002; Saleh et al, 2004): macrophages incorporate the SPIOs and store them in lysosomes, thus producing a strong contrast on T*2-weighted MRI. Validation with independent techniques (iron-sensitive histological Prussian blue staining and immunohistochemistry, using the antibody ED-1 for the identification of macrophages (Schroeter et al, 2004)) showed excellent spatial agreement with the hypointense regions on T*2-weighted MR images. Superparamagnetic iron oxide particle-labeled macrophages accumulated at the border zone of a photothrombotic-induced infarct as early as 5.5 days after stroke induction (Kleinschnitz et al, 2003). These results were confirmed by Saleh et al (2004), using ultrasmall SPIOs (USPIO) and a high-resolution 3D T*2-weighted MR imaging with an in-plane resolution of 78 μm. These authors also described a faint signal loss in ischemic control animals not receiving an injection of USPIOs. The origin of these endogenous susceptibility effects was further investigated in rats subjected to transient MCAO and not receiving USPIOs (Weber et al, 2005). A time-dependent accumulation of iron-rich macrophages was detected in the chronic phase (2–10 weeks after MCAO induction) on high-resolution 3D T*2-weighted MRI in excellent spatial correspondence with the corresponding histological tissue sections (Figure 2). Most of the iron-loaded macrophages were found in the neighborhood of blood vessels in the ischemic striatum, indicating delayed microhemorrhages due to a late onset of vascular degradation.
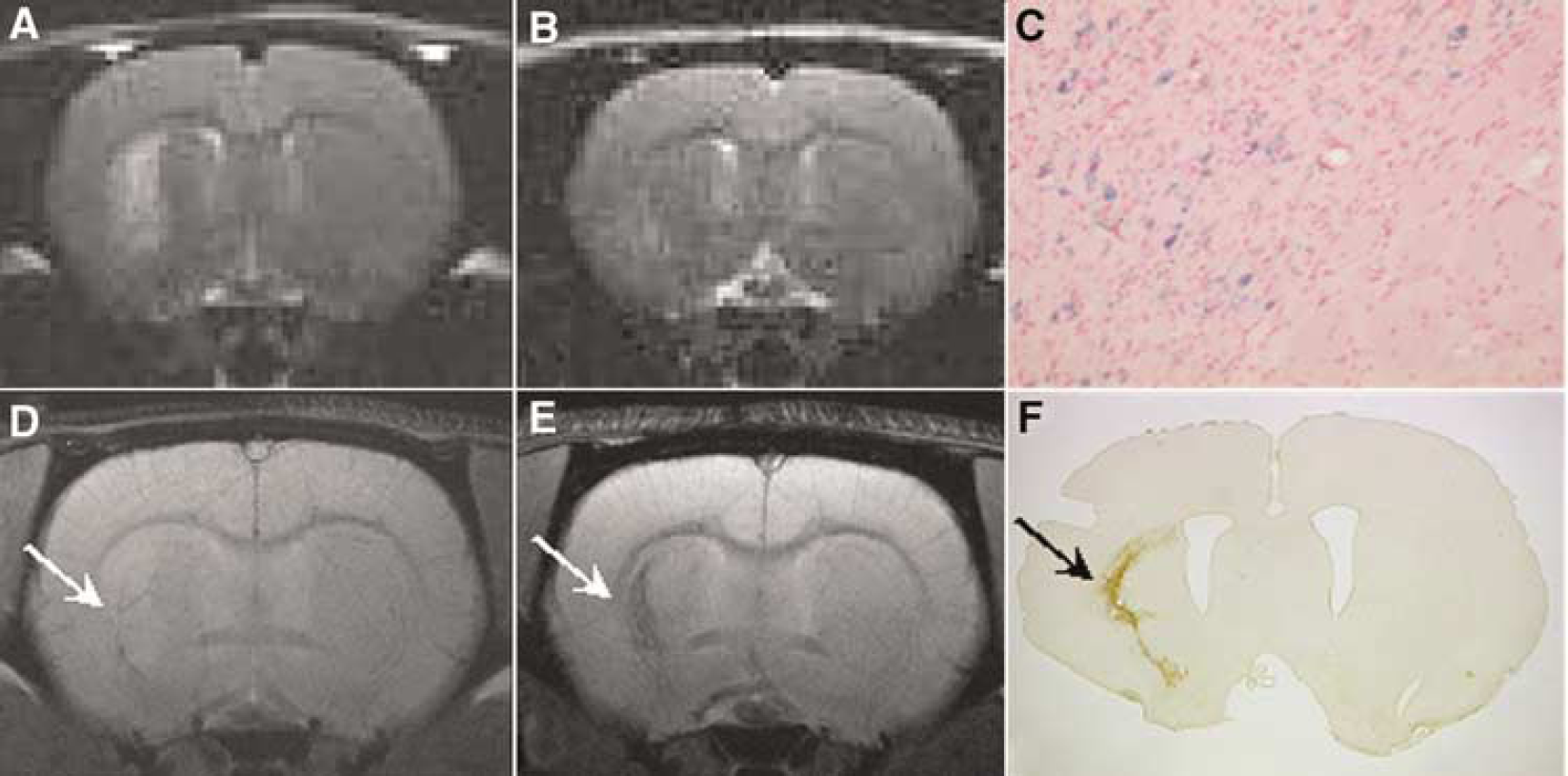
T2-weighted (
Functional Impairment
Besides MRI techniques assessing structural changes, functional MRI (fMRI) is increasingly used to obtain information about cerebral activity and spatial and temporal reorganization processes in animal models of ischemic brain injury. Combining multimodal MRI with fMRI and behavioral testing in animal stroke studies provides an elegant way of investigating the underlying mechanisms of ‘brain rehabilitation’.
On brain activation an increase in the regional CBF and cerebral blood volume (CBV) (‘neurovascular coupling’) can be observed on CBF-weighted or CBV-weighted MRI (Dijkhuizen and Nicolay, 2003a). A third method is based on detection of blood oxygen level dependent (BOLD) signal changes after activation (Logothetis and Pfeuffer, 2004; Ogawa et al, 1990). To date, all published fMRI studies in rodent stroke models have been performed in anesthetized animals, using mostly α-chloralose, since α-chloralose has been shown to best preserve vascular-metabolic coupling (Ueki et al, 1988, 1992). Anesthesia, however, is known to decrease both basal brain activity and brain activation on stimulation and to influence cerebral hemodynamics (Lindauer et al, 1993; Nakao et al, 2001). Nevertheless, the published fMRI studies in animal stroke models of the rat have shown the importance of including functional imaging modalities and behavioral studies when testing new treatment strategies for stroke in animal models.
Reese et al (2000) used a T*2-weighted imaging sequence with the intravascular contrast agent Endorem® to assess the CBV response in rats 24 h after permanent MCAO. Although the primary somatosensory cortex of the ischemic hemisphere did not show abnormalities on DWI- and T2-weighted images, it showed no CBV response after electric forepaw stimulation. In a subsequent study, functional recovery of the somatosensory cortex was assessed over a longer time period of up to 12 days (Sauter et al, 2002). Vehicle-treated rats did not show a recovery of the CBV response in the ipsilateral somatosensory cortex throughout the study, whereas three of five rats treated with the calcium antagonist isradipine showed a recovery of the CBV response after 5 days and five of 10 rats after 12 days. Again normal T2 and ADC values were measured in the somatosensory cortex of the recovered rats, but not all rats with normal T2 and ADC values showed restored functional activation.
In a permanent MCAO model involving directly the primary somatosensory cortex of rats, stimulation of the previously paretic hindlimb led to an activation of the ipsilateral primary sensorimotor cortex and a second region located lateral to the injured primary contralateral somatosensory cortex on CBV-weighted fMRI 3 weeks after lesion induction, when full behavioral recovery had occurred (Abo et al, 2001). Healthy control animals showed a normal activation pattern of the contralateral primary sensorimotor cortex.
Similar findings on functional reorganization in the chronic phase after both transient and permanent MCAO were recently reported in two studies by Dijkhuizen et al, (2001b, 2003b). At 3 days after permanent focal ischemia in rats, no activation was detected on CBV-weighted fMRI in the contralateral ischemic hemisphere on unilateral forelimb stimulation, but functional reorganization was seen as activation in the ipsilateral hemisphere (Dijkhuizen et al, 2001b). After 14 days, when behavioral testing showed a recovery to subnormal levels, activation responses were found again contralaterally, adjacent to the infarcted area, and the activation in the ipsilateral hemisphere had decreased. Similar reorganization patterns were found in rats subjected to transient MCAO (Dijkhuizen et al, 2003b).
In these studies, the use of α-chloralose, which is not suitable for survival protocols, or invasive physiological monitoring prohibited longitudinal fMRI investigation in the same animal. Rather the authors had to resort to different animal groups for different study times after stroke induction, thus reducing the attractiveness of the noninvasive imaging method substantially. Recently, we have therefore developed a completely noninvasive physiological monitoring (Ramos-Cabrer et al, 2005) and a new anesthesia protocol using the α2-adrenoreceptor agonist medetomidine to allow for uncompromised longitudinal fMRI studies after stroke. This new approach circumvents the earlier problems (side effects from α-chloralose anesthesia and noninvasive physiological monitoring) and permits characterization of intraindividual brain reorganization changes in longitudinal studies. As shown in Figure 3, it is thus possible to study activation patterns in the primary somatosensory cortex of the same animal before and at three different time points after 60 mins of MCAO.
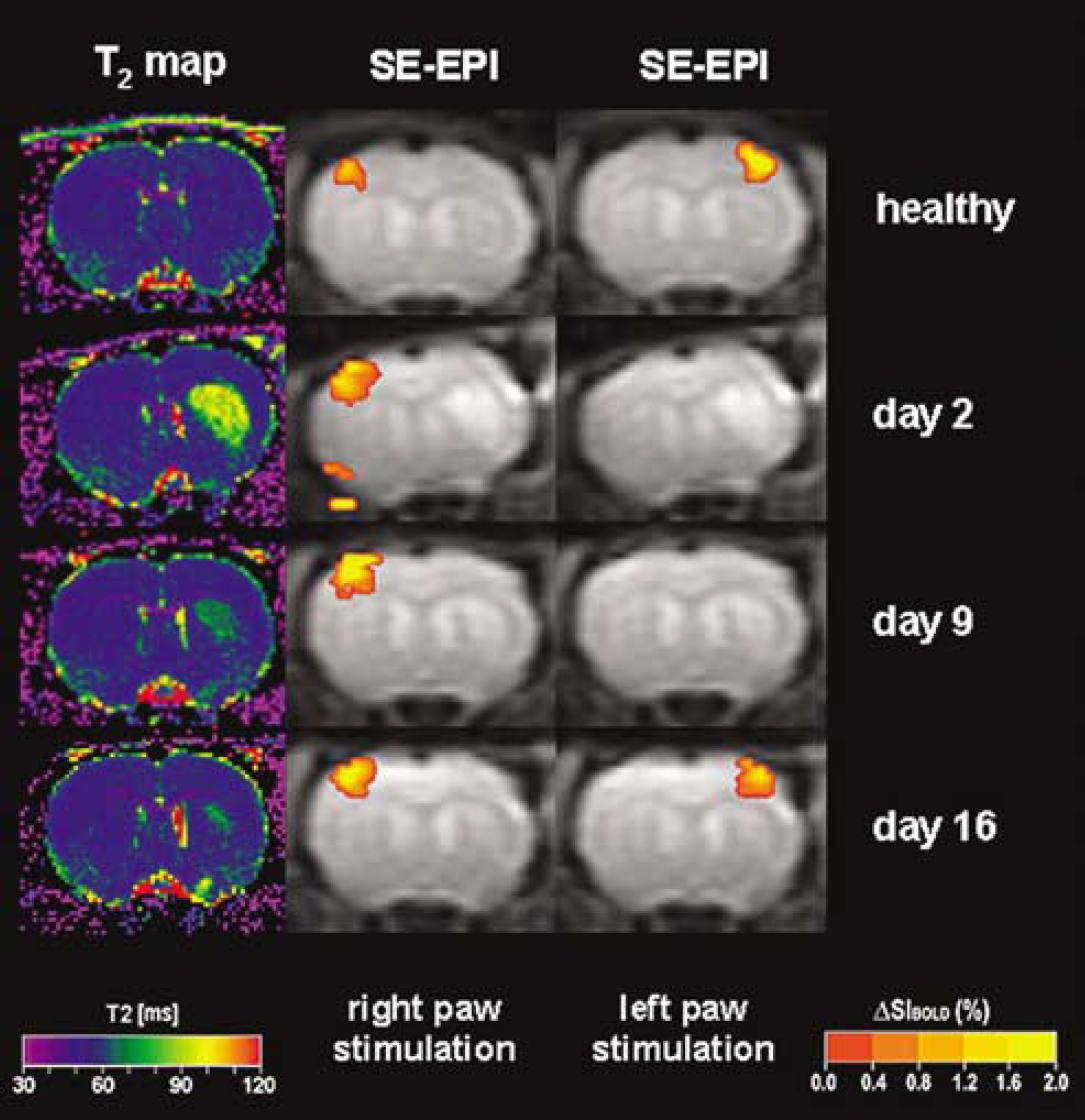
Longitudinal examination of a rat subjected to 60 mins of transient MCAO using T2-weighted MRI and SE-EPI BOLD imaging under sedation with medetomidine. In the first row, the healthy rat shows contralateral activation of the primary somatosensory cortex on either the right or left forepaw stimulation. T2 maps of the right striatum show increased T2 values after MCAO (row 2). At 2 and 9 days after MCAO, no activation can be observed in the right somatosensory cortex, although the cortex does not show evidence of ischemic damage on the corresponding T2 map. On day 16, unilateral activation is again observed in each somatosensory cortex (in response to stimulation of the opposite paw), as the hyperintensity on the T2 map in the right striatum has almost completely resolved (courtesy of R Weber and P Ramos-Cabrer, MPI for Neurological Research, Cologne, Germany).
Therapeutic Aspects
Cerebroprotection
Many drugs have been tested for cerebroprotective effectiveness over the years (reviewed by Fisher and Ratan, 2003; Hoehn et al, 2001) and it exceeds the scope of this review to discuss detailed results. The use of a noninvasive imaging modality such as DWI, T2-weighted, or T*2-weighted MRI will provide information about both the pharmacokinetics of the drug under investigation and the temporal response of the ischemic brain. Such imaging-based longitudinal studies of ischemic tissue volume give access to the growth curve of the affected tissue. This curve thus provides important data on the response time of the drug and on the mechanisms of action of the drug. It therefore allows a distinction between a slow-down of lesion expansion, a complete arrest of expansion (Hoehn-Berlage et al, 1997), or even (an unlikely) reversal of the affected tissue region. Such distinction is of great importance for the better understanding of the mechanisms of action and the effectiveness of a drug in ‘reducing’ the ischemic lesion volume.
Thrombolysis
Thrombolysis with rtPA is still the most promising treatment option in acute human stroke therapy, which has been shown to improve the outcome in a selected patient cohort (Hacke et al, 2004; Wardlaw et al, 2003). However, thrombolytic therapy is associated with an increased risk of hemorrhagic transformation (HT) (Hacke et al, 1995; Kwiatkowski et al, 1999). Thus, many MRI studies have been performed in animal stroke models to increase the rate of successful recanalization and decrease the rate of harmful postthrombolysis brain hemorrhage by identifying the tissue at risk for HT. For this purpose, new animal models of embolic stroke (Busch et al, 1998; Zhang et al, 1997) have been developed to study thrombolytic therapy with rtPA directly inside the magnet and to delineate the temporal and spatial time course of acute brain ischemia and reperfusion. Using one of these new models in rats, Niessen et al (2002) did not reveal any difference between intraarterial and intravenous application of rtPA administered 1 h after clot embolism. Both routes of rtPA application significantly improved blood flow on PWI and lesion volumes on ADC maps showed an inhibition of infarct growth in both groups. Nevertheless, the rate of HT was increased in both application routes compared with control animals. The recanalization rate in experimental thromboembolic stroke studies in rats is dependent on the clot composition, which has to be taken into account when performing thrombolysis studies (Niessen et al, 2003). As expected, a combined occlusion of the MCA and the circle of Willis, assessed by MR angiography, resulted in a poorer recanalization rate in rtPA-treated rats (Hilger et al, 2002).
The majority of recent MRI-based thrombolysis studies in animals had been performed to find reliable parameters to predict HT in thromboembolic stroke models and to improve patient selection for thrombolysis (Dijkhuizen et al, 2001a, 2002; Hilger et al, 2002; Neumann-Haefelin et al, 2002). An increased permeability of the blood-brain barrier, as assessed by a T1 contrast enhancement after GdDTPA application or by a T*2-sensitive signal decrease after injection of monocrytalline iron oxide nanoparticles, was found to be predictive of HT in both rats treated 3 (Neumann-Haefelin et al, 2002) and 6 h (Dijkhuizen et al, 2001a, 2002) after induction of thromboembolic stroke.
Regeneration After Cerebral Ischemia
Since neuroprotective agents have so far shown no benefits in human stroke trials, stem cell transplantation is one novel therapeutical approach for ischemic brain injury aiming at tissue replacement therapy. Moreover, it does not appear to have such a restricted therapeutical time window as the stem cells are implanted in the chronic disease phase. The selective migration and accumulation of different types of stem cells in animal models of cerebral ischemia can be monitored using in vivo MR imaging (Hoehn et al, 2002; Modo et al, 2004; Zhang et al, 2003). Cells have to be labeled in vitro with MR contrast agents to be detectable by MRI against the host tissue background (Hoehn and Himmelreich, 2005). To date, contrast agents based on iron oxide particles (Hoehn et al, 2002; Zhang et al, 2003) and gadolinium (Modo et al, 2002, 2004) have been used successfully in animal stroke studies. Murine embryonic stem cells were labeled by a lipofection procedure with USPIOs and 60,000 of these cells were implanted stereotactically in the parenchyma of the contralateral hemisphere in rats, 2 weeks after transient MCAO (Hoehn et al, 2002). Labeled cells migrate along the corpus callosum into the ischemic hemisphere and accumulate in the border zone of the ischemic brain tissue. This directed migration towards the ischemic border zone was observable on repetitive high-resolution 3D T*2-weighted MRI during 3 weeks after transplantation. A selective migration of iron-labeled adult neuronal stem cells from the ipsilateral ventricle system towards the ischemic parenchyma was also found after intracisternal implantation (Zhang et al, 2003), as shown in Figure 4. In this study, adult neuronal stem cells from the subventricular zone of rats were labeled with ferromagnetic particles using a ‘gene gun’ and implantation was conducted 2 days after MCAO. Moreover, rats transplanted with cells showed significant improvement on behavioral testing as early as 14 days after implantation. Recently, Modo et al (2002, 2004) used the bimodal contrast agent gadolinium-rhodamine dextran, which allowed the detection of implanted stem cells and their transhemispheric migration both by in vivo MRI and postmortem fluorescent microscopy.
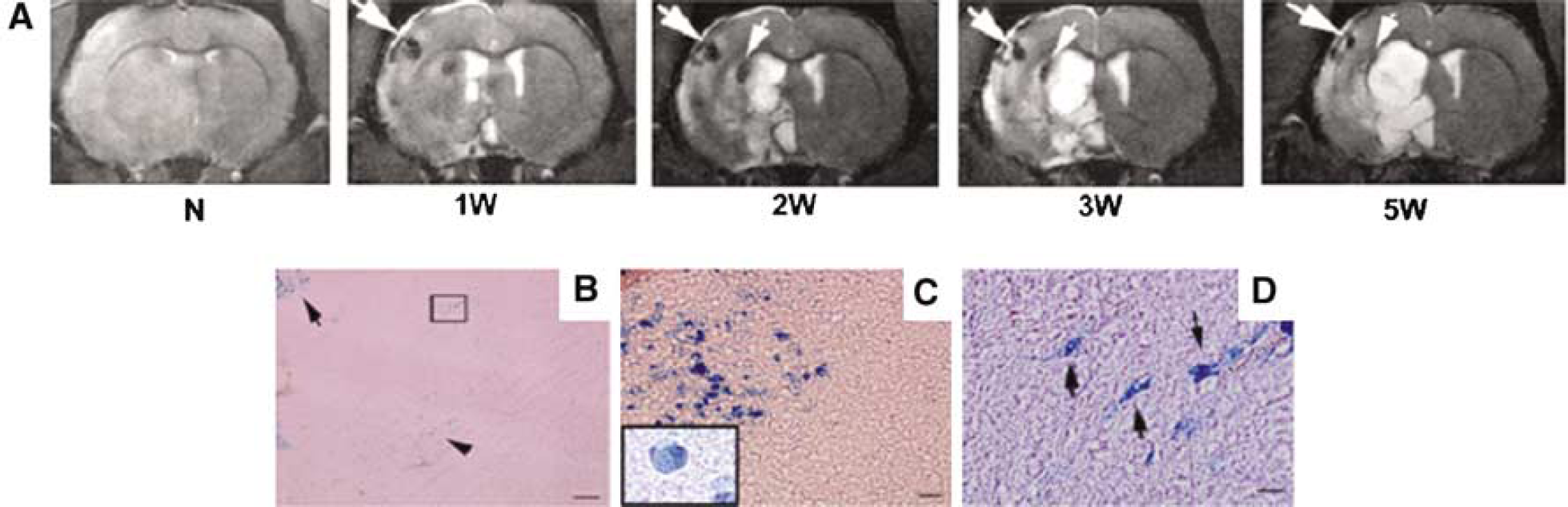
(
It should be cautioned here that for such experiments of cell tracking the cells generally need to be labeled. Then, careful evaluation of tolerance by the cells of both the labeling procedure and the label itself are necessary. Although in most cases no significant effect on viability or proliferation has been described, Kostura et al (2004) reported recently that human mesenchymal stem cells showed a marked inhibition of differentiation potential into chondrocytes (while adipogenic and osteogenic differentiation was not affected) due to Feridex incorporation. The general issue of assessment of labeling toxicity for MRI experiments of cells has broadly been discussed in a recent review (Modo et al, 2005).
Outlook
Over the years, many different modalities of MR imaging have been shown to successfully contribute to the study of cerebral stroke, the understanding of underlying mechanisms, and the effectiveness assessment of various therapeutical strategies. Magnetic resonance imaging with its multi-faceted features of morphological, metabolic, hemodynamic, and functional imaging information has become an indispensable tool for these studies. Of all those presently applied, the modality of the PWI would clearly profit from a reliable method for CBF quantification, in particular in areas of reduced blood flow (and consequently reduced MR signal inflow).
A new exciting field is the exploration of the therapeutical potential of stem cells. For this purpose, the emerging contribution of MRI as a ‘Molecular Imaging’ modality is finding its place among the other molecular imaging techniques. Successful labeling of stem cells and sensitive detection of their spatio-temporal dynamics at ultrahigh resolution and sufficiently high temporal resolution, compatible with tolerance of the animals for repetitive scanning sessions has recently been documented (Himmelreich et al, 2005; Hoehn et al, 2004).
A very innovative field is the development of responsive contrast agents to incorporate into the cells. As these agents will have to be activated by specifically selected enzymes expressed under specific conditions of the cells, this may lead to the imaging of functional cell status by imaging reporters of gene expression or enzyme activity. Another recent approach is the use of transgenic cell lines for transplantation, which have their own inbuilt MR contrast agent under specific gene control. Two such examples using the iron storage protein, ferritin, have recently been reported (Cohen et al, 2005; Genove et al, 2005). Application of these promising techniques will require close collaboration of contrast agent chemistry, molecular biology and imaging expertise together with that of stroke pathophysiology on experimental animal models. With such symbiosis of complementary disciplines, we may in future see an exciting role of MRI for new experiments elucidating the role of (stem) cells for stroke therapy.
Footnotes
Acknowledgements
Financial support by the Hertie Foundation (Functional Brain Imaging) is gratefully acknowledged.
