Abstract
Neural and oligodendrocyte progenitor cells in the adult brain express Ascl1 (also known as Mash1), a basic helix-loop-helix transcription factor. We examined the progeny and fate of this progenitor population in adult male
Keywords
Introduction
The adult rodent subventricular zone (SVZ) of the lateral ventricle is composed of migratory neuroblasts, actively proliferating progenitor cells, and quiescent neural stem cells (Lois and Alvarez-Buylla, 1994; Doetsch et al, 1997). Neuroblasts in the SVZ travel the rostral migratory stream to the olfactory bulb where they differentiate into granule and periglomerular neurons throughout adult life (Morshead et al, 1994). Subventricular zone neural stem cells generate oligodendrocyte progenitor cells and oligodendrocytes that distribute to the corpus callosum (Menn et al, 2006). Cerebral ischemia promotes proliferation of actively proliferating SVZ cells and recruits SVZ neuroblasts to the ischemic boundary regions (Jin et al, 2001; Zhang et al, 2001; Arvidsson et al, 2002; Parent et al, 2002). In contrast, the contribution of neural progenitor cells to oligodendrogenesis in the ischemic brain is understudied, although stroke triggers oligodendrogenesis (Gregersen et al, 2001; Dewar et al, 2003; Zhang et al, 2009). Moreover, molecular mechanisms underlying stroke-induced neurogenesis and oligodendrogenesis have not been extensively investigated.
Ascl1 (also known as Mash1) is a basic helix-loop-helix transcription factor, which is transiently expressed in neural progenitor cells (Guillemot et al, 1993; Battiste et al, 2007). During development, Ascl1 directs neurogenesis and oligodendrogenesis (Parras et al, 2007).
Materials and methods
All experimental procedures were approved by the Institutional Animal Care and Use Committee of the Henry Ford Hospital.
Bigenic Ascl1-CreER™; R26R-Stop-Yellow Fluorescent Protein Mice
A pair of
Animal Model of Stroke and Tamoxifen Treatment
Adult male
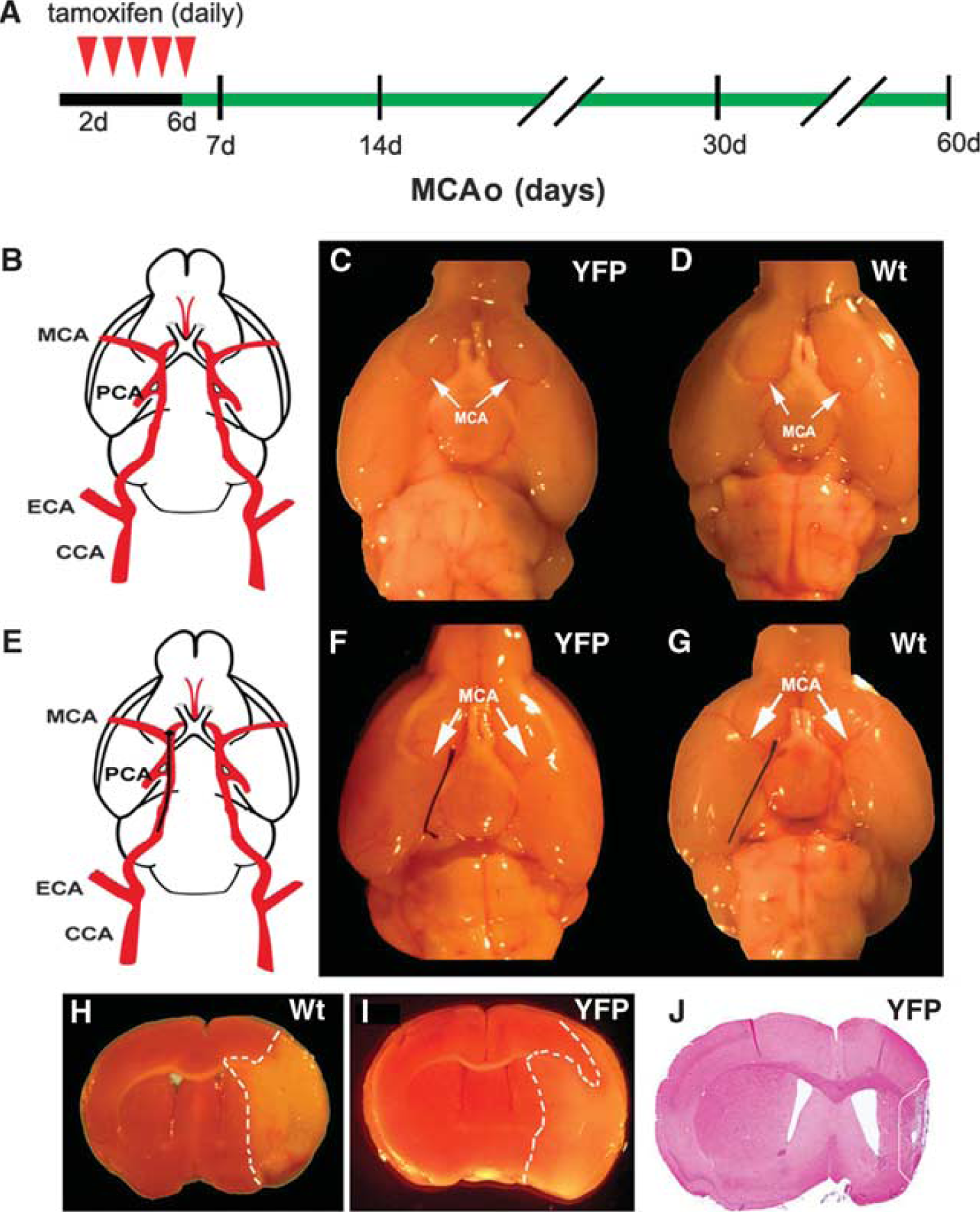
Experimental protocol and ischemic lesion. (
Bromodeoxyuridine Labeling
Bromodeoxyuridine (BrdU), the thymidine analog that is incorporated into the DNA of dividing cells during the S phase, was used for mitotic labeling (Sigma Chemical). Ischemic mice were intraperitoneally injected with BrdU (100 mg/kg) daily for 5 consecutive days starting 48 hours after stroke.
Brain Tissue Preparation and Immunohistochemistry
Animals were transcardially perfused with heparinized saline, followed by 4% paraformaldehyde. The brains were removed from the skull, fixed further in 4% formaldehyde for 4 hours at 4°C, and then transferred into 30% sucrose in phosphate-buffered saline for 24 hours. The brains were embedded and frozen in optimal cutting temperature compound. A series of 30-μm-thick brain coronal sections were cut in a cryostat from the bregma (1.18 mm to −0.82 mm) for the mouse (Franklin and Paxinos, 1997).
Every fifth section was used for immunohistochemistry, as described previously (Zhang et al, 2001). The following antibodies were used in this study: sheep anti-BrdU (1:100, Abcam, Cambridge, MA, USA), mouse anti-nestin (1:100, BD Bioscience, Franklin, NJ, USA), rabbit anti-Ki67 (1:300, Thermo, Fremont, CA, USA), goat anti-doublecortin (DCX, 1:200, Santa Cruz Biotechnology, Santa Cruz, CA, USA), got anti-Sox2 (1:500, Santa Cruz Biotechnology), mouse anti-NeuN (1:500, Chemicon/Millipore, Billerica, MA, USA), chicken anti-green fluorescent protein (1:500, Aves Labs, Tigard, OR, USA), rabbit anti-DARPP32 (1:200, Cell Signaling Technology, Danvers, MA, USA), mouse anti-calbindin (1:800, Swant/Fisher Scientific, Waltham, MA, USA), rabbit anti-GAD-47 (1:500, Sigma Chemical), rabbit-anti-GFAP (glial fibrillary acidic protein) (1:10,000, Dako, Carpinteria, CA, USA), rabbit anti-NG2 (1:800, Chemicon/Millipore), and mouse anti-2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase, 1:200, Chemicon/Millipore). Cell nuclei were stained with DAPI (4′, 6′-diamidino-2-phenylindole).
Double immunofluorescent images were acquired using a Zeiss (Zeiss, Thornwood, NY, USA) LSM 510 Meta-NLO system with Coherent Chameleon Ti: Sa laser. Three-color images were scanned using 488 nm argon, 543 HeNe, and Chameleon (750 nm for DAPI) lasers.
Quantification of Yellow Fluorescent Protein+ Cells
Stereological unbiased estimates of the total numbers of YFP+ cells within the regions of interest were obtained using a microcomputer imaging device stereology software (3D Fractionator, InterFocus Imaging, Cambridge, England), which drives the Ludl X-Y-Z-motorized stage of the actual microscope stage (Olympus BX61, Olympus, Center Valley, PA, USA) focus position. The YFP + cells were identified with antibodies against green fluorescent protein. In brief, using the automated optical fractionator method, we drew the corpus callosum, striatal, and SVZ areas on coronal sections at a × 4 objective. A higher power (a × 60 objective, NA 1.4) was then selected, and the system used random systematic sampling to sample 30% of the defined region. When the system moved to the first location within the region of interest, a counting frame was placed over the selected area. We then counted the number of immunostained cells by focusing up and down and marking targets within the counting frame. Data are presented as an estimate of the total number of green fluorescent protein + cells in defined regions.
Statistical Analysis
Data were evaluated for normality. Data transformation would be considered if data were abnormal. The average of the two measurements was taken for each mouse at each location at each time point. One-way analysis of variance was used to study the time effect on each region of interest. The analysis began with testing for the overall time effect, followed by pairwise group comparisons. All data are presented as mean ± s.e. Statistical significance was set at
Results
Occlusion of the Middle Cerebral Artery Results in Ischemic Lesion
To examine whether
Stroke did not Alter the Ascl1-Expressing Cell Profile in the Contralateral Hemisphere
Before tracking the progeny of Ascl1-expressing cells in the ischemic brain, we verified tamoxifen-inducible Cre-mediated recombination targeted at Ascl1-expressing cells in the young adult mice without MCAo.
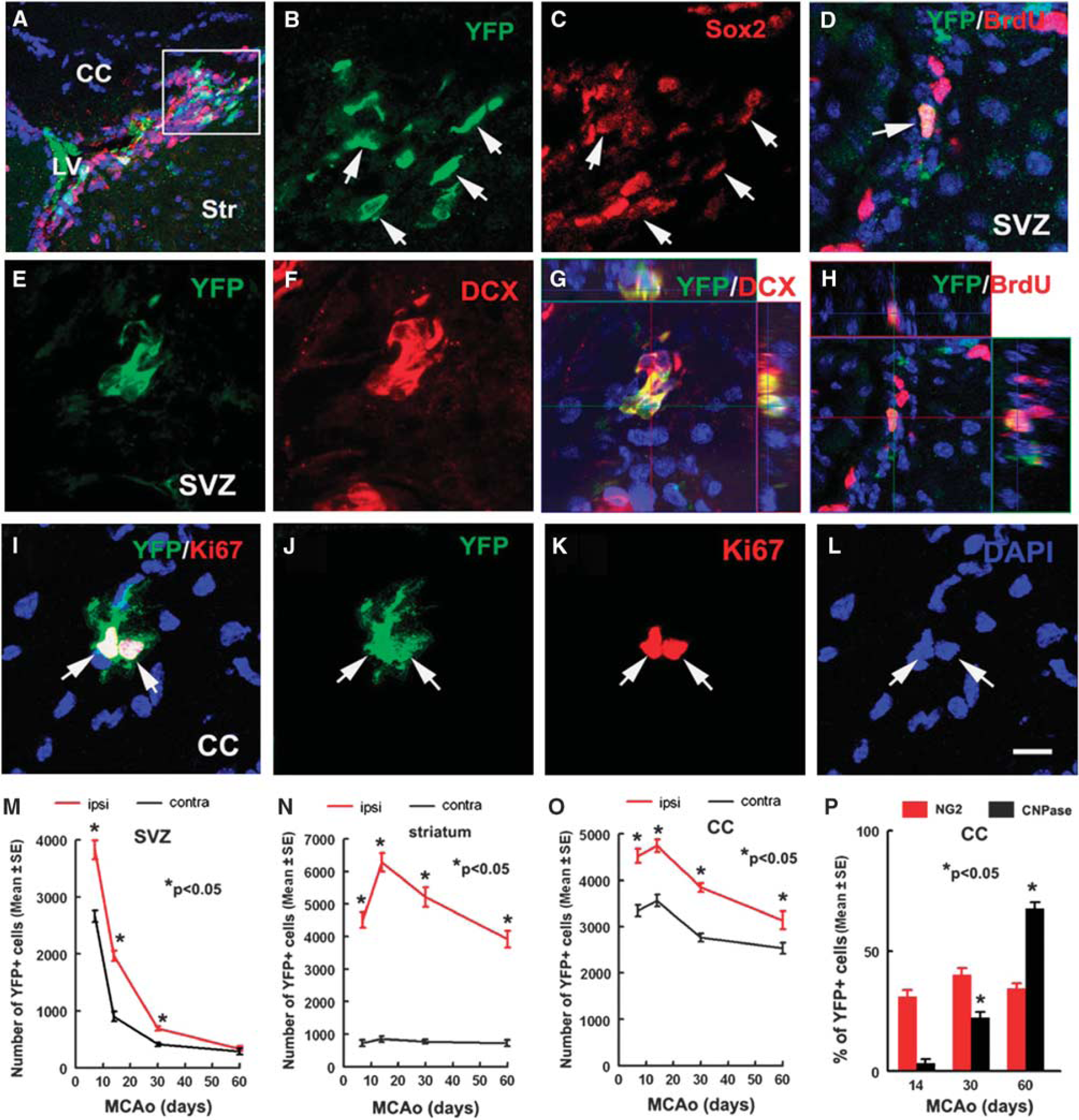
YFP+ cells are proliferating progenitor cells or neuroblasts in the SVZ and corpus callosum of nonischemic
Percentage of double immunoreactive YFP+ cells in nonischemic and ischemic brains
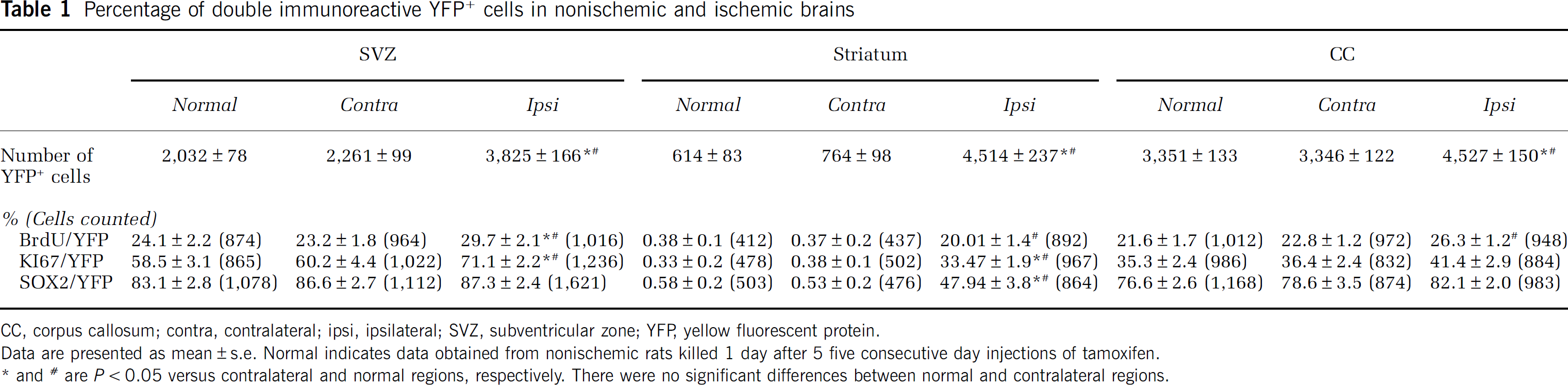
CC, corpus callosum; contra, contralateral; ipsi, ipsilateral; SVZ, subventricular zone; YFR yellow fluorescent protein.
Data are presented as mean ± s.e. Normal indicates data obtained from nonischemic rats killed 1 day after 5 five consecutive day injections of tamoxifen.
and # are
Double immunostaining revealed that many (86%) of the YFP+ cells in the SVZ were SOX2+ (Table 1, Figures 2A to 2C), a marker of neural progenitor cells, and ~63% of YFP+ cells were DCX+ (62.7 ± 3.7, Figures 2E to 2G), a marker of neuroblasts. The YFP+ cells were also BrdU+ after 24 hours exposure (Table 1, Figures 2D and 2H). These data indicate that Ascl1-expressing cells comprise transient amplifying cells and neuroblasts in the SVZ of the lateral ventricles, which is consistent with the published studies (Kim et al, 2007, 2008).
In the corpus callosum, many YFP+ cells appeared as doublets or clusters and were Ki67+ (Figures 2I to 2L, Table 1), suggesting that these YFP+ cells are actively proliferating
To examine whether stroke affects Ascl1-expressing cells in the contralateral hemisphere,
Percentage of YFP+ cells coexpressing neuronal and oligodendrocyte phenotypes in the ischemic striatum
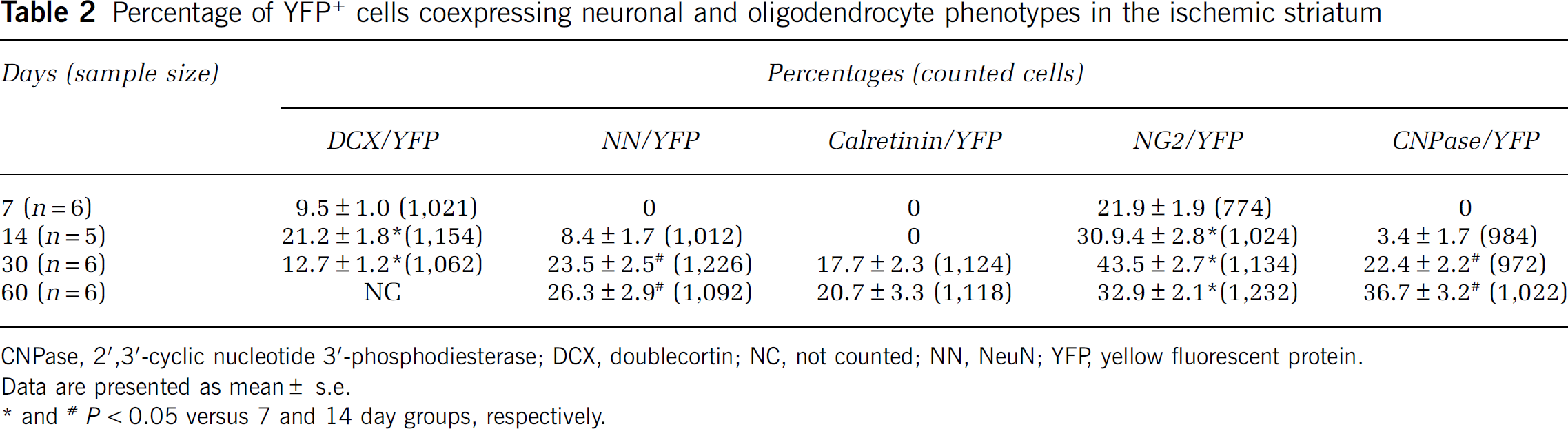
CNPase, 2′,3′-cyclic nucleotide 3′-phosphodiesterase; DCX, doublecortin; NC, not counted; NN, NeuN; YFP, yellow fluorescent protein.
Data are presented as mean ± s.e.
and #
Stroke Increases Ascl1 Lineage Cells in the Ipsilateral Subventricular Zone
To follow the Ascl1-expressing cells in the ischemic brain,
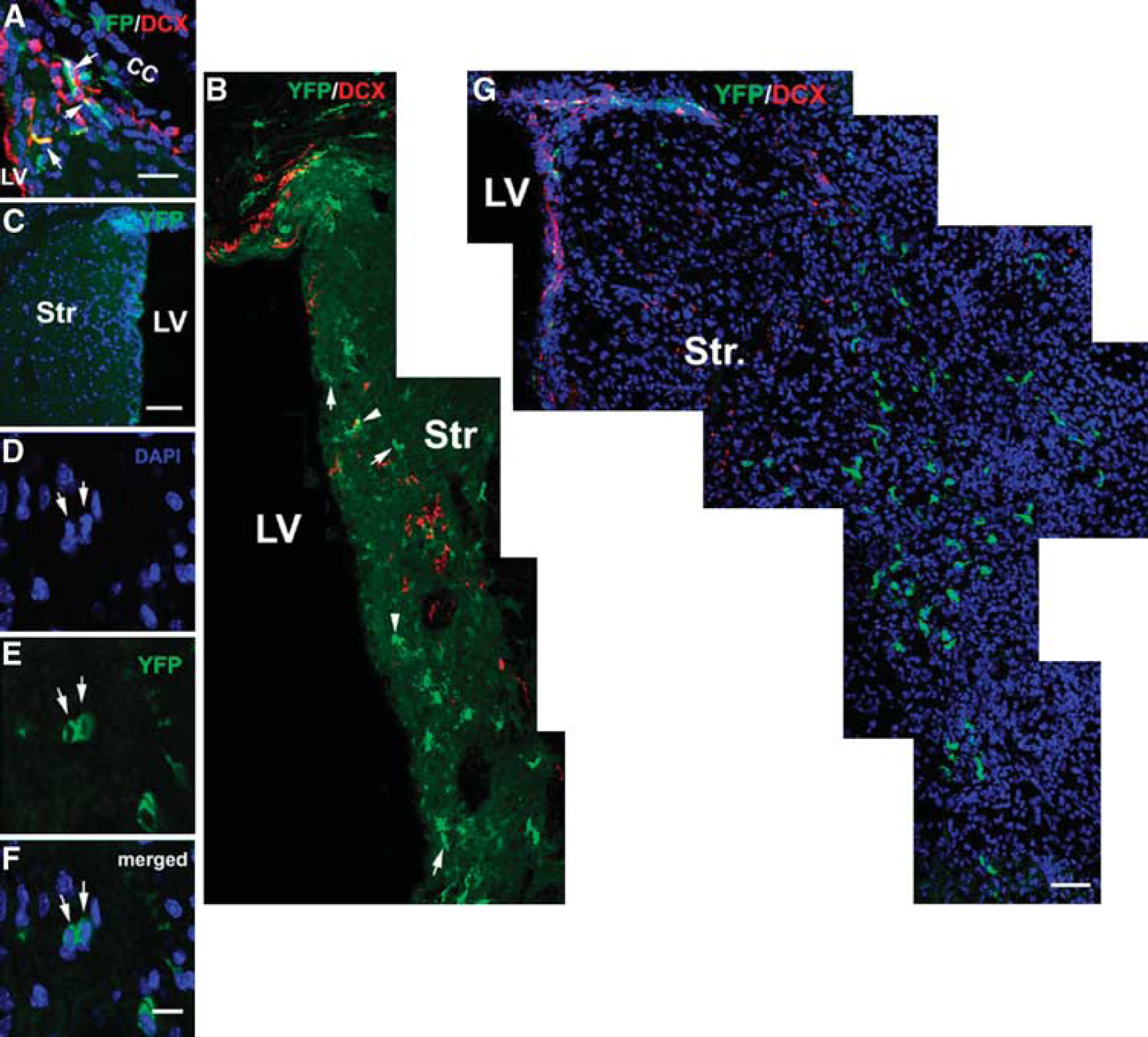
Distribution of YFP+ cells in the ischemic striatum. Double immunostaining shows YFP+ /DCX+ cells (
Stroke Increases Ascl1 Lineage Cells that Become Neurons and Oligodendrocyte Progenitor Cells in the Ischemic Striatum
In the ischemic striatum, 7 days after stroke, the number of YFP+ cells increased by 4.6-fold (
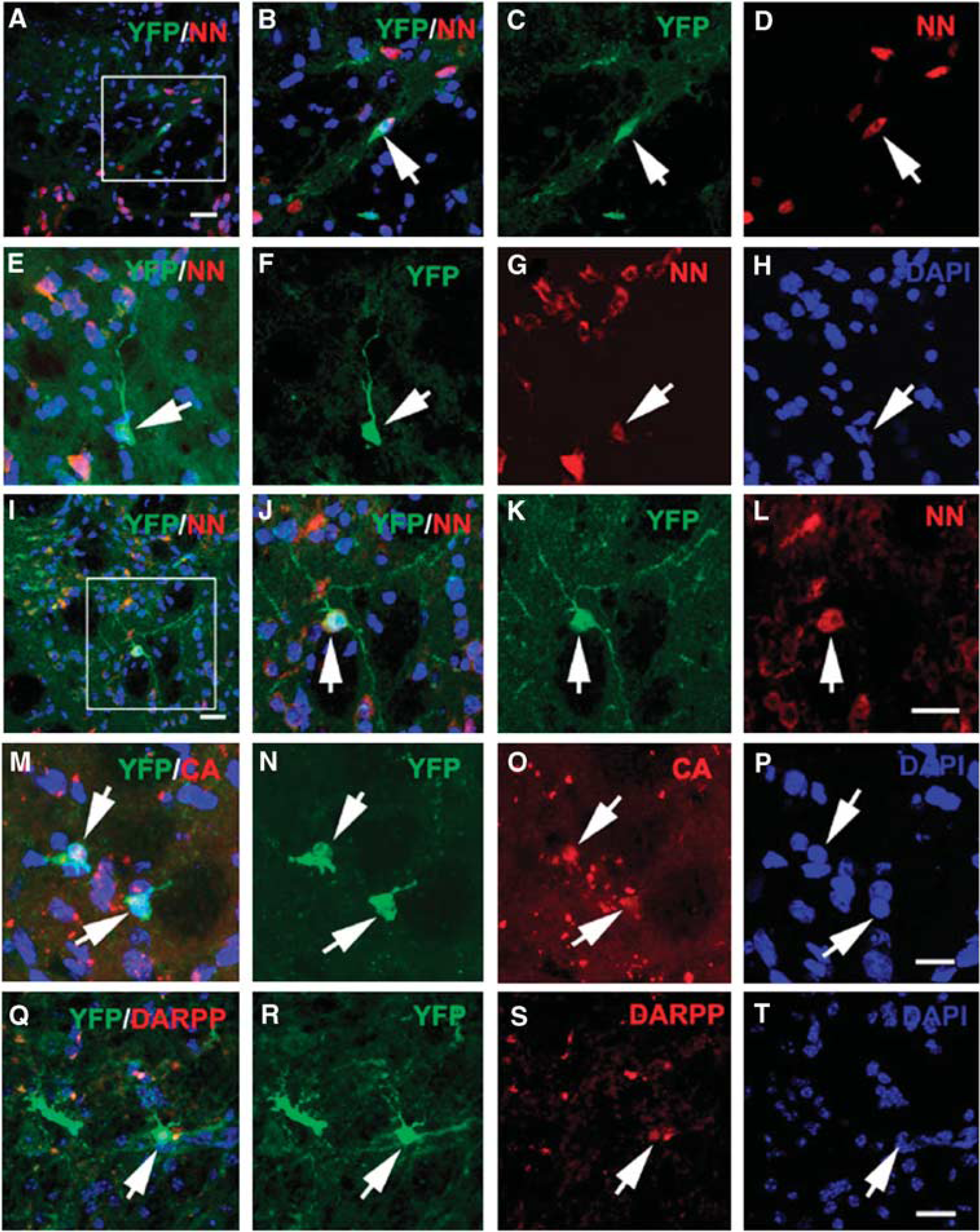
Neuronal phenotypes of YFP+ cells in the ischemic striatum. Double immuostaining shows YFP+/NN+ cells in the ischemic striatum 14 (
In the ischemic striatum, some of the YFP+ cells exhibited morphologic characteristics of oligodendrocyte progenitor cells and their cell bodies and processes were immunoreactive for the chondroitin sulfate proteoglycan NG2 (Table 2, Figure 5A). Some YFP+ cells exhibited faint NG2 immunostaining (Figures 5B to 5E) 30 days after stroke, and orthogonal reconstruction of confocal images revealed that the cell bodies of YFP+ cells with faint or strong NG2 immuostaining were within the imaged tissue (Figure 5E), indicating that cells with weak NG2 immunosignals are not owing to tissue cutting. This observation suggests that these YFP+ oligodendrocyte progenitor cells may differentiate into mature oligodendrocytes. By immunostaining mature oligodendrocytes with antibodies against CNPase and myelin basic protein (MBP), we found an increase in YFP+/CNPase+ and YFP+/MBP+ cells in the ischemic striatum during 30 to 60 days of stroke (Table 2, Figures 5F to 5M). These data indicate that in addition to neurons, Ascl1 lineage cells are fated to become oligodendrocyte progenitor cells and mature oligodendrocytes.
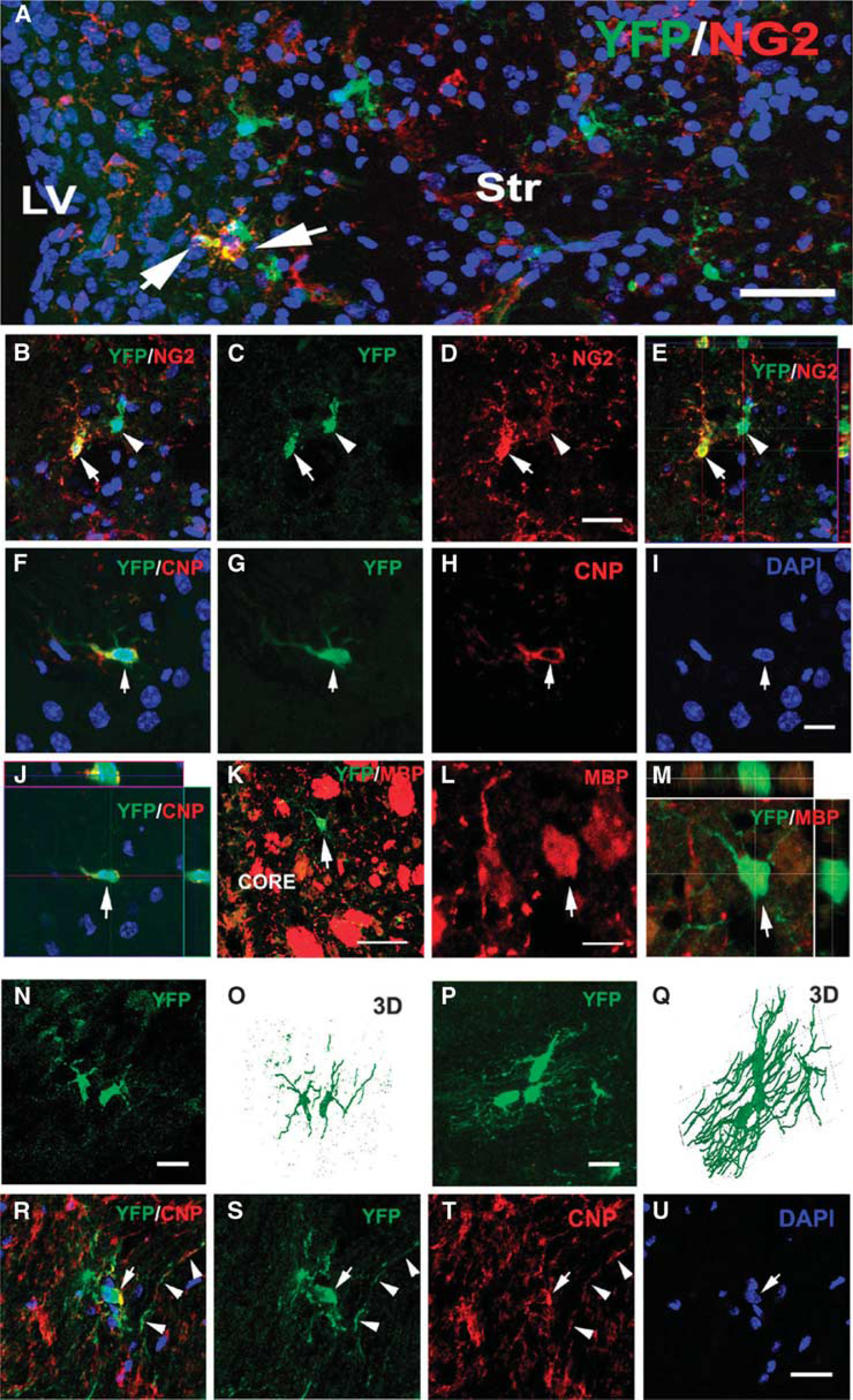
Oligodendrocyte phenotypes of YFP+ cells in the ischemic corpus callosum and striatum. Double immunostaining shows that YFP+ cells in the ischemic striatum were NG2+ (
Stroke Increases Ascl1 Lineage Cells that Become Oligodendrocytes in the Ischemic Corpus Callosum
Stroke significantly (
Discussion
This study provides
Ascl1 Lineage Cells Become Neurons in the Ischemic Striatum
Ascl1 is a pro-neural basic helix-loop-helix transcription factor that is transiently expressed in cells that are within the cell cycle (Guillemot et al, 1993; Battiste et al, 2007). Cells expressing Ascl1 in the
Ascl1 Lineage Cells Become Oligodendrocyte Progenitor Cells and Oligodendrocytes in the Corpus Callosum and Striatum
Oligodendrocytes are the myelin-forming glial cells in the adult brain (Levison and Goldman, 1993; Menn et al, 2006). Mature oligodendrocytes do not proliferate and new oligodendrocytes are derived from nonmyelinating oligodendrocyte progenitor cells (Carroll et al, 1990). The corpus callosum of the adult rodent contains heterogeneous oligodendrocyte progenitor cells (Gonzalez-Perez et al, 2009). Most NG2+ cells in the adult brain originate from Ascl1 lineage cells (Parras et al, 2007). Ascl1 could be the earliest oligodendrocyte marker in the corpus callosum based on its expression before Olig2 and Sox10 expression, two early markers of oligodendrocyte progenitor cells (Kim et al, 2007). Approximately 50% of oligodendrocyte progenitor cells in the corpus callosum are within the cell cycle (Rivers et al, 2008). This study confirms that Ascl1 lineage oligodendrocyte progenitor cells are actively proliferating and that these oligodendrocyte progenitor cells differentiate into myelinating oligodendrocytes. Oligodendrocytes are highly susceptible to ischemic insults and the loss of myelin leads to axonal damage after stroke (Pantoni et al, 1996). The contribution of progenitor cells to oligodendrogenesis in the ischemic brain has not been extensively investigated (Dewar et al, 2003). Oligodendrocyte progenitor cells exist in the corpus callosum, the striatum, and the SVZ (Levison and Goldman, 1993; Menn et al, 2006). Our lineage tracking experiments show that a considerable population of Ascl1 lineage cells contributes to NG2+ oligodendrocyte progenitor cells and mature oligodendrocytes in the ischemic corpus callosum and striatum, indicating that stroke augments oligodendrogenesis by Ascl1 lineage cells. We observed that in addition to the striatal Ascl1 lineage NG2+ cells, Ascl1-expressing cells extended from the SVZ to the ischemic striatum were NG2+, suggesting that stroke-induced oligodendrocytes in the striatum differentiate not only from the SVZ but also from parenchymal Ascl1 lineage NG2+ cells. These data indicate that both SVZ- and striatum-derived Ascl1 lineage cells are important for remyelination in the ischemic brain. However, oligodendrocytes in the corpus callosum likely differentiate from local Ascl1 lineage NG2+ cells. This study also revealed Ascl1 lineage oligodendrocytes in the ischemic striatum and corpus callosum, which is consistent with previous findings that Ascl1 lineage oligodendrocytes are heterogeneous and there are different sub-populations of oligodendrocytes in the brain (Parras et al, 2007). During stroke recovery, expansion of the white matter in the ischemic damaged areas has been observed (Li et al, 2009). It is possible that the newly formed Ascl1 lineage oligodendrocytes with myelin sheath observed in this study may function to remyelinate damaged and sprouting axons in the ischemic brain. Remyelination reinstates saltatory conduction and resolves functional deficits of axons (Franklin and Ffrench-Constant, 2008). Thus, amplification of Ascl1 lineage oligodendrogenesis could potentially promote axonal repair, leading to improvement in neurologic function.
The Ascl1 lineage cells gave rise to oligodendrocyte progenitor cells and to mature oligodendrocytes in the corpus callosum even after stroke, which is consistent with previous observations showing that the Ascl1 progeny do not differentiate into neurons in the corpus callosum (Kim et al, 2007, 2008). This is in contrast with Ascl1 lineage cells in the ischemic striatum that gave rise to neurons and oligodendrocytes, suggesting that differences in the local environment may be responsible. The fate determination of Ascl1 lineage cells is context dependent and additional molecular components are required for Ascl1 lineage cells to differentiate into neurons (Kim et al, 2007, 2008).
This study establishes the fate of Ascl1 lineage cells in the ischemic brain, which is consistent with essential regulatory functions of Ascl1 in directing both neurogenesis and oligodendrogenesis (Kim et al, 2007, 2008). Given the fact that recombination is only detected in ~10% of Ascl1-expressing cells in
Footnotes
Acknowledgements
The authors thank Dr Jane Johnson at the University of Texas Southwestern Medical Center for providing
The authors declare no conflict of interest.
