Abstract
There is evidence of marked variation in the brain distribution of specific subtypes of the GABA-benzodiazepine receptor and that particular subtypes mediate different functions. The α5-containing subtype is highly expressed in the hippocampus, and selective α5 inverse agonists (which decrease tonic GABA inhibition) are being developed as potential memory-enhancing agents. Evidence for such receptor localization and specialization in humans in vivo is lacking because the widely used probes for imaging the GABA-benzodiazepine receptors, [11C]flumazenil and [123I]iomazenil, appear to reflect binding to the α1 subtype, based on its distribution and affinity of flumazenil for this subtype. The authors characterized for positron emission tomography (PET) a radioligand from Ro15 4513, the binding of which has a marked limbic distribution in the rat and human brain in vivo. Competition studies in vivo in the rat revealed that radiolabeled Ro15 4513 uptake was reduced to nonspecific levels only by drugs that have affinity for the α5 subtype (flunitrazepam, RY80, Ro15 4513, L655,708), but not by the α1 selective agonist, zolpidem. Quantification of [11C]Ro15 4513 PET was performed in humans using a metabolite-corrected plasma input function. [11C]Ro15 4513 uptake was relatively greater in limbic areas compared with [11C]flumazenil, but lower in the occipital cortex and cerebellum. The authors conclude that [11C]Ro15 4513 PET labels in vivo the GABA-benzodiazepine receptor containing the α5 subtype in limbic structures and can be used to further explore the functional role of this subtype in humans.
Our understanding of the function and pharmacology of the GABA-benzodiazepine receptor is currently undergoing a major revolution through the use of knockout and knock-in techniques and the discovery of drugs selective for particular subtypes of the GABA-benzodiazepine receptor. The GABA-benzodiazepine receptor comprises five protein subunits drawn from several different families (e.g., α1–6, β1–3, γ, δ, and ρ) that come together to form an ion channel with binding sites or receptors for the endogenous transmitter GABA, benzodiazepines, and other agents such as neurosteroids and convulsants (Sieghart, 1995). When GABA binds with the GABA-benzodiazepine receptor complex, conformational changes increase the permeability of the central pore to chloride ions (Nutt and Malizia, 2001). The resulting chloride flux hyperpolarizes the neuron, reducing its excitability and producing a general inhibitory effect on neuronal activity. Other compounds, such as barbiturates and neurosteroids, also bind to this receptor complex to increase channel opening. Benzodiazepines, such as diazepam, also increase channel opening but do so through increasing the effectiveness of GABA rather than by directly opening the chloride channel.
There are profound differences in the distribution of the common receptor subunits in the brain, the α1 being most abundant, particularly in the cortex (Fritschy and Möhler, 1995; Pirker et al., 2000; Wisden et al., 1992). In contrast, the α2 and α5 types have a more limited distribution and predominate in the hippocampus. The thalamus is rich in α4 subunits, whereas the α6 subunit is almost exclusively found in cerebellum (Fritschy and Möhler et al., 1995; Pirker et al., 2000; Wisden et al., 1992). Such a distribution suggests that different subtypes may mediate different functions, and recently it has been proposed that the sedative but not anxiolytic effects of diazepam are mediated through the α1 subtype (Rudolph et al., 1999).
Given this definition of subtype functions in animal models, it is timely to conduct such characterization in humans. Benzodiazepine receptor function in humans can be assessed in vivo by measuring the effects of a benzodiazepine on sedation, anxiety, EEG, or saccadic eye movements (Roy-Byrne et al., 1993). However, the effects on a particular anatomical or subtype population of GABA-benzodiazepine receptors cannot be determined by any of these methods. Both single photon emission computed tomography and positron emission tomography (PET) have been used extensively to delineate the distribution and levels of the GABA-benzodiazepine receptor in a variety of neuropsychiatric disorders including anxiety, alcoholism, and epilepsy (Malizia and Richardson, 1995).
The commonly used radioligands for PET and single photon emission computed tomography are [11C]flumazenil and [123I]iomazenil (iodinated derivative of flumazenil), respectively. Flumazenil is a benzodiazepine antagonist that binds with high affinity to benzodiazepine receptors containing the α1, α2, α3, or α5, (Ki approximately 1 nmol/L) subunits and less so to those containing α4 or α6 subunits (Ki approximately 150 nmol/L) (Sieghart, 1995). Because of the high abundance of α1-containing receptors in the brain, a PET or single photon emission computed tomography image of benzodiazepine receptor binding appears to primarily reflect this population. Although other radioligands have been investigated (suriclone, flunitrazepam, triazolam), none has proved as useful, and also label a similar population of GABA-benzodiazepine receptors, as [11C]flumazenil or [123I]iomazenil (Pike et al., 1993). More recently, other radioligands—[3H]L655,708 and [3H]RY80—have been evaluated as putative PET ligands of the α5-containing GABA-benzodiazepine receptor, but were not sufficiently retained in the brain (Opacka-Juffry et al., 1999). Consequently, there is a paucity of probes for imaging selectively in vivo GABA-benzodiazepine receptor subtypes.
Ro15 4513 is a partial inverse agonist at the GABA-benzodiazepine receptor. [11C]Ro15 4513 was developed as a potential PET radioligand (Halldin et al., 1992; Inoue et al., 1992; Sazdot et al., 1989). Early studies revealed that [11C]Ro15 4513 bound with a very different regional brain profile to flumazenil, suggesting that it labeled a different population of GABA-benzodiazepine receptors (Halldin et al., 1992; Inoue et al., 1992; Onoe et al., 1996). Its use as a PET radiotracer has been limited because of the preeminence of [11C]flumazenil and the uncertainty about which GABA-benzodiazepine receptors were being labeled, though the α5-containing subtype was suggested but never demonstrated. In view of the recent knowledge concerning the subtypes of the GABA-benzodiazepine receptor and data suggesting that Ro15 4513 has relatively higher affinity for the α5-containing subtype (Hadingham et al., 1993), we decided to validate [11C]Ro15 4513 as a PET radioligand that could be used to characterize the α5 subtype in humans. We used our small animal PET study to characterize its binding in rats and tested its subtype selectivity by pretreating rats with compounds of known subtype selectivity, such as zolpidem (α1), flunitrazepam (α1, α2, α3, α5), RY80 (α5), and L655,708 (α5) (Hadingham et al., 1993; Quirk et al., 1996; Sieghart, 1995; Skolnick et al., 1997).
MATERIALS AND METHODS
[11C]Flumazenil was prepared with more than 95% radiochemical purity and with a specific radioactivity of approximately 24,000 MBq/μmol at the end of the synthesis by N-methylation of the corresponding N-desmethyl derivative with [11C]iodomethane, as described previously (Pike et al., 1993). [11C]Ro15 4513 was synthesized in a manner described by Inoue et al., (1992), by N-methylation of the corresponding N-desmethyl derivative with [11C]iodomethane. The product was purified by reverse phase HPLC on a column (Phenomenex Ultra-carb 7 ODS (30), 250 × 10-mm outer diameter) eluted with MeCN/0.03-mol/L MeCOONH4/AcOH (100/100/1 by volume). The specific radioactivity was approximately 14,000 MBq/μmol at end of synthesis.
All drugs and tritiated compounds were purchased from commercial sources with the exception of RY80, which was kindly provided by Drs. J. Cook and P. Skolnick. Adult male Sprague-Dawley rats (body weight 250 to 290 g) were used.
Nine healthy male volunteers without any serious medical or psychiatric illnesses were studied. Six volunteers underwent a [11C]flumazenil scan and three underwent a [11C]Ro15 4513 scan as control subjects in other studies. Informed consent was obtained before participation in the study, which had been approved by the local ethics committees and from the UK Administration of Radioactive Substances Advisory Committee.
Human studies
Scanning procedure for [11C]flumazenil.
[11C]Flumazenil scans were performed using an ECAT-953B PET scanner in three-dimensional mode (with the septa retracted; CTI/Siemens, Knoxville, TN, U.S.A.) using previously described performance characteristics (Spinks et al., 1992). The axial field of view was 10.6 cm. The final image resolution (using a Hann 0.5 reconstruction filter) was 8.0 × 8.0 × 4.3 mm at full-width half maximum.
A previously validated protocol was used (Richardson et al., 1996). Briefly, subjects were positioned such that the transaxial images were parallel to the intercommissural line (AC-PC line) and the majority of the cerebellum was scanned. A bolus injection of [11C]flumazenil (370 MBq, 10 mCi) in approximately 2 mL was administered through an intravenous cannula sited in the dominant antecubital fossa vein. Each subject had a radial arterial cannula in the nondominant wrist to allow continuous counting of blood radioactivity concentration with a bismuth germanate counter during the course of the experiment. Discrete samples were also taken at 4, 6, 8, 10, 20, 35, 50, and 65 minutes after injection to derive the metabolite-corrected plasma curve. Fractional concentrations of unchanged [11C]flumazenil and of less-lipophilic radioactive metabolites were determined by solid-phase extraction of plasma followed by HPLC (Luthra et al., 1993).
Twenty dynamic frames of data in three-dimensional mode were acquired over 90 minutes (1 × 30, 4 × 15, 4 × 60, 2 × 150, 2 × 300, 7 × 600 seconds) and reconstructed into dynamic images containing 31 contiguous slices.
Scanning procedure for [11C]Ro15 4513.
[11C]Ro15 4513 scans were performed using an ECAT EXACT3D HR++ PET scanner (CTI/Siemens) with performance characteristics as described previously (Spinks et al., 2000). Briefly, this scanner consists of six rings of standard detector blocks with 4.39 × 4.05 × 30-mm elements giving an axial field of view of 23.4 cm. Thus, data from the whole brain can be simultaneously acquired. The mean spatial resolution is 4.8 ± 0.2 mm full-width half maximum in-plane and 5.6 ± 0.5 mm axially. The same protocol used for [11C]flumazenil was followed, and 20 dynamic frames (1 × 30, 4 × 15, 4 × 60, 2 × 150, 2 × 300, 7 × 600 seconds) of data were acquired in three-dimensional mode over 90 minutes to produce images containing 78 contiguous slices. Blood sampling was performed as described for [11C]flumazenil to produce a metabolite-corrected plasma input function.
Measurement of radioactivity distribution in serial human plasma samples.
The distribution of radioactivity in serial human samples after intravenous injection of [11C]Ro15 4513 or [11C]flumazenil was measured using a semiautomated metabolite analysis system, consisting of an on-line solid phase sample enrichment column, a reverse-phase HPLC column, and radioactivity and ultraviolet absorbance detectors linked to a PC-based integrator. Although [11C]Ro15 4513 was produced as a PET radioligand more than a decade ago, only qualitative or semiquantitative analyses of its distribution were performed in humans (Inoue et al., 1992; Suhara et al., 1993). We performed fully quantitative analysis after showing in the rat that no radiolabeled metabolite crossed the blood—brain barrier, and unlike previous studies, a plasma-corrected input function was generated.
The blood samples were taken from the artery with heparinized syringes after intravenous injection of [11C]Ro15 4513 or [11C]flumazenil, and were rapidly centrifuged (2,000 g, 1 minute) to obtain plasma. Duplicate aliquots of plasma were taken for measurement of radioactivity and an aliquot of plasma (2 mL) was injected onto the solid-phase extraction reverse phase column (C18) that was washed with diammonium hydrogen phosphate (0.01 mol/L). The eluate from this column, referred to as the solid-phase extraction fraction, was collected, and duplicate aliquots (5 mL) were taken for measurement of radioactivity. The retained radioactivity on the solid-phase extraction column was then transferred onto the reverse-phase HPLC column (μ-Bondapak C18, 30 × 0.78-cm inner diameter, 10-μm particle size). The HPLC column was washed with the mobile phase (0.1-mol/L ammonium formate: methanol (45:55 volume/volume) at a flow rate of 3 mL/min. The HPLC eluate was monitored sequentially for radioactivity and ultraviolet absorbance at 254 nm. Both detectors were linked to a PC-based integrator that recorded the chromatogram and allowed the correction of the data for physical decay, background radioactivity, and integration of radioactive components. The amount of [11C]Ro15 4513 or [11C]flumazenil in plasma at a given time point was calculated as a percentage of all radioactive components separated by HPLC plus the radioactivity present in the solid-phase extraction fraction.
Image analysis.
Voxel-by-voxel parametric images of [11C]flumazenil and [11C]Ro15 4513 were produced using spectral analysis (Cunningham and Jones, 1993). Reconstructed [11C]flumazenil and [11C]Ro15 4513 parametric images were analyzed on a Sun SPARC workstation (Sun Microsystems, Mountain View, CA, U.S.A.) using AnalyzeAVW version 3.1 (Mayo Foundation;Robb and Hanson, 1991). Regions of interest were drawn on the volume-of-distribution map of the GABA-benzodiazepine receptors as labeled by [11C]flumazenil or [11C]Ro15 4513 with reference to a stereotaxic atlas (Talairach and Tournoux, 1988) on the individual brains, to obtain regional measures. The regions of interest were placed on the following regions: frontal, anterior cingulate, occipital and medial temporal cortices, thalamus, dorsal and ventral striatum, and cerebellum.
Animal studies
All work was performed by licensed investigators in accordance with the Home Office Guidance on the Operations of The Animals (Scientific Procedures) Act 1986. A tail vein and artery (the latter in those rats designated for blood sampling) were catheterized under isoflurane with nitrous oxide and oxygen anesthesia. The rats were allowed to recover in restrainers for approximately 2 hours before radioligand injection. Each rat was injected with approximately 10 MBq [11C]Ro15 4513 (as prepared for the human scans) or approximately 0.3 MBq [3H]Ro15 4513 in a volume of 0.2 mL, via the catheterized tail vein. There was no difference in uptake between the different radioligands and [3H]Ro15 4513 was used when the 11C-labeled ligand was unavailable.
Competition study.
In the competition experiments, rats were intravenously injected with one of the following drugs with known benzodiazepine receptor affinity: flunitrazepam (5 mg/kg), Ro15 4513 (2 mg/kg), zolpidem (5 mg/kg), L655, 708 (2 mg/kg), or RY80 (2 or 5 mg/kg) 10 minutes before injection of [11C]Ro15 4513. The doses were chosen to exploit the drugs' relatively higher affinity for GABA-benzodiazepine receptors containing particular subtypes (zolpidem for α1 and RY80 and L655,708 for α5). The rats were killed 60 minutes after radioligand injection. Immediately after euthanasia, the brains were rapidly removed and dissected. Fifteen brain tissues were sampled: olfactory bulbs and tubercles, frontopolar with cingulate cortex, septum, striata, thalamus, hypothalamus, hippocampus, occipital cortex, inferior colliculi, superior colliculi, medulla with pons, and cerebellum. Radioactivity content of brain tissues, blood, and plasma samples were measured using a γ ([11C]Ro15 4513) or β ([3H]Ro15 4513) scintillation counter. The data were normalized for radioactivity and body weight, giving “uptake units” defined as cpm per gram of tissue/cpm injected per gram body weight.
Measurement of radioactivity distribution in serial rat plasma samples.
To determine the distribution of radioactivity in blood and plasma after intravenous injection of [11C]Ro15 4513, blood samples (six per rat) were taken at graded times (1, 2, 5, 10, 20, 30, 40, or 50 minutes) from seven rats (but not at every time point) via the tail artery catheter and centrifuged to obtain plasma. The plasma was then processed using the same methods followed for the human plasma samples.
Measurement of radioactivity distribution in rat brain samples.
Brain samples were obtained at 30 minutes, cut into small pieces, and added to ice-cold methanol (15 mL). The samples were homogenized using a Polytron homogenizer set at half-maximum speed for 1 to 2 minutes, and the homogenate was centrifuged at 2000 g for 5 minutes. The resulting pellets and duplicate aliquots (100 μL) of methanol supernatant were taken for measurement of radioactivity. The remaining methanol supernatant containing the extracted radioactivity was rotary evaporated to dryness at 60°C, and the residue was collected during the HPLC mobile phase (3 mL) and filtered. Duplicate aliquots (100 μL) were taken for measurement of radioactivity. The processed sample (1 mL) was directly injected onto the automated metabolite analysis system. The conditions and the acquisition and processing of the data were the same as those described for the plasma samples. The amount of [11C]Ro15 4513 at a given time point was calculated using percentage of each radioactive component in each analyte.
Small-animal scanning.
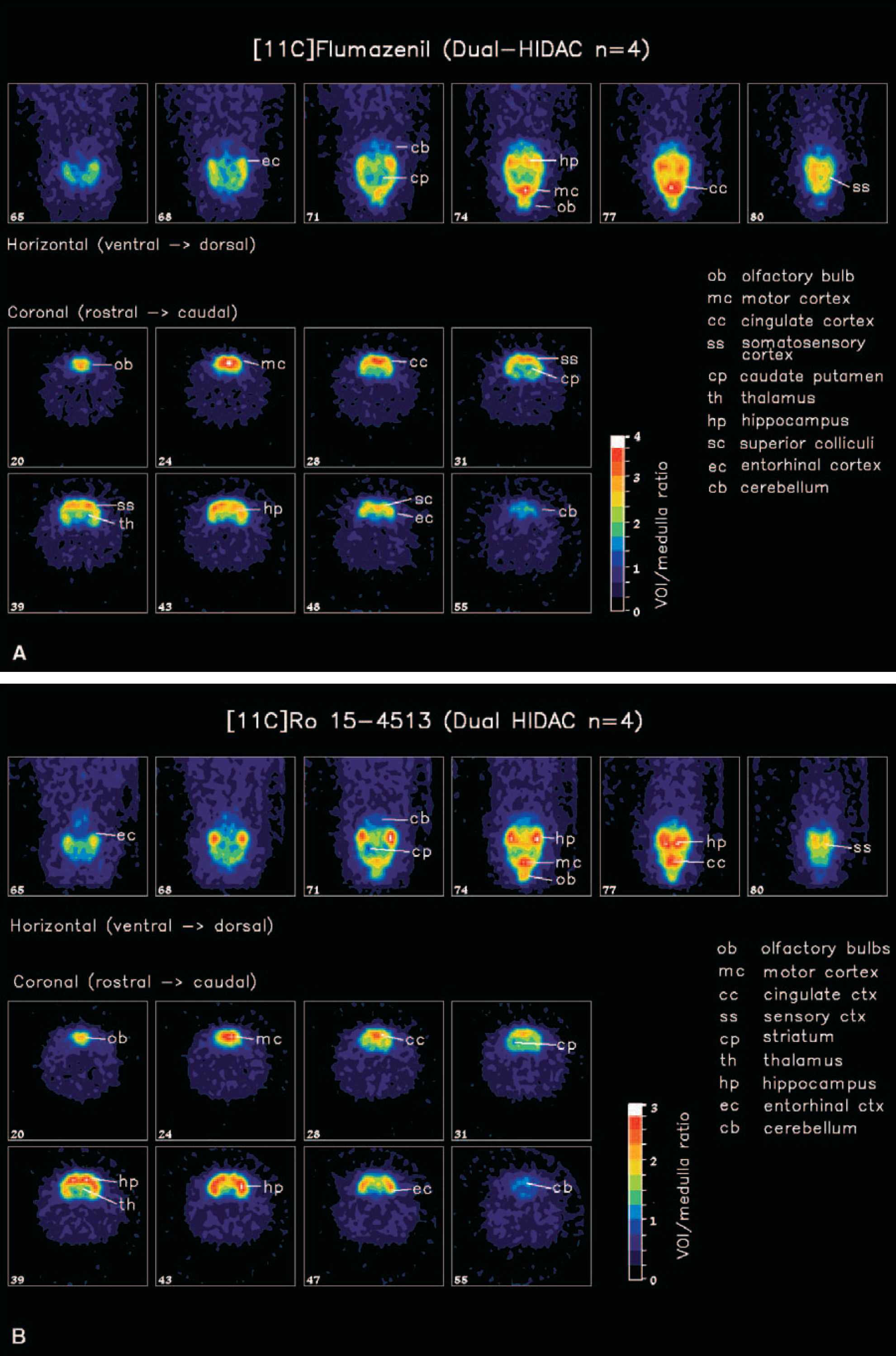
A dual-HIDAC (Oxford Positron Systems, Weston-on-the-Green, U.K.) scanner was used in a manner described elsewhere (Hume et al., 2001). Intravenous administration of [11C]flumazenil or [11C]Ro15 4513 was given to isoflurane-anesthetized rats. The mean (± SD) injectate was 11 ± 2 MBq per rat, with an associated stable content of 1.4 ± 0.5 nmol/kg. Each rat was scanned for 60 minutes, after which time the animal was killed. The scan sinograms were reconstructed into 0.5-mm cubic voxels (with a Hamming filter, 0.6 cut-off) and transferred into ANALYZE. A volume-of-interest template was constructed, based on the brain slices illustrated in Paxinos and Watson (1996), with 0.5-mm intervals. The template was projected onto each scan volume and data sampled from several volumes of interest, including frontal with cingulate cortex (452 voxels), hippocampus (264 voxels), and cerebellum (776 voxels). The whole template included volumes of interest representing all the tissues taken after death. Because of noise in the volume-of-interest data, we were unable to define the initial rapid-delivery phase (all volumes of interest) or to measure the tissue concentration after approximately 30 minutes in those volumes of interest with minimal specific binding. We obtained a measure of specific binding by using the volume-of-interest—medulla ratio, as used for the dissection protocol, during the period 20 to 60 minutes.
In addition, a dose-ranging study using intravenous L655,708 (0.01 to 2 mg/kg) was performed using both RATPET (Myers et al., 1996) and dual-HIDAC scanners (Hume et al., 2001). The dose of L655,708 was given intravenously 10 minutes before injection of [11C]Ro15 4513. In RATPET, data were acquired in three-dimensional mode in 21 temporal frames (3 × 5, 3 × 15, 4 × 60, 11 × 300 seconds) and the sinograms were reconstructed into 15 contiguous slices using a conventional filtered back projection algorithm, with Hamming filter and 0.5 cut-off. The data were transferred into ANALYZE and sampled using an regions-of-interest template to obtain radioactivity/time profiles for the hippocampus, prefrontal with cingulate cortex, and medulla with pons. Specific binding was estimated using a reference tissue compartmental model (Hume et al., 1992), with the medulla data set as the indirect input function. Data from the dual-HIDAC camera were acquired and analyzed using the previously described semiquantitative manner because full kinetic analysis has not yet been validated for this system.
RESULTS
Radioactive distribution in rat and human plasma
The clearances of [11C]Ro15 4513 from human and rat plasma were similar, though more rapid in the rat. The data are comparable with those reported by Suhara et al., (1993) in the mouse. Several radioactive metabolites were observed, but these were less lipophilic than [11C]Ro15 4513 and were not further identified. The percentage of radioactivity in human plasma represented by [11C]Ro15 4513 decreased from 83% at 3 minutes to 16% at 65 minutes, whereas in rat plasma, only 9% of the radioactivity was [11C]Ro15 4513 at 40 minutes. In both the human and rat plasma samples, the polar radioactive metabolites increased with time from less than 17% at 3 minutes to 89% of the radioactivity in plasma at 65 minutes.
Radioactive distribution in rat brain
The radioactivity in rat brain was essentially [11C]Ro15 4513. The percentages of radioactivity represented by [11C]Ro15 4513 in the cortex, hippocampus, and cerebellum were 97%, 100%, and 96% of the radioactivity in the tissues, respectively. These findings show that in the rat, radioactive metabolites do not cross the blood—brain barrier and that there is no significant metabolism of [11C]Ro15 4513 within brain tissue.
Positron emission tomography scanning
The scans showed that the patterns of distribution of [11C]Ro15 4513 binding in the human and rat brain are comparable, with the exception of the striatum. There were, however, marked differences in the distribution of [11C]Ro15 4513 and [11C]flumazenil in both the human and rat brain.
Human.
In the human brain, uptake of [11C]Ro15 4513 was particularly high in limbic regions compared with that of [11C]flumazenil (Fig. 1A and B and Table 1). The time—activity curves from regions of interest, the anterior cingulate cortex, occipital cortex, ventral striatum, and cerebellum are shown in Fig. 2. The highest levels of uptake were seen in the anterior cingulate cortex, with high levels also present in the hippocampus, insular cortex, septal region, and amygdala. In the cerebral cortex there was an anteroposterior gradient of decreasing levels of [11C]Ro15 4513 volumes of distribution (Vds), such that occipital Vds were the lowest (frontal, 6.27 ± 0.78; occipital, 5.46 ± 0.32). This is the reverse of that seen with [11C]flumazenil (frontal, 6.42 ± 1.25; occipital, 8.61 ± 0.81). In the striatum, greater levels of uptake of [11C]Ro15 4513 than [11C]flumazenil were found. In addition, a higher [11C]Ro15 4513 Vd was seen in the ventral striatum (5.2 ± 1.1) compared with the dorsal striatum (3.96 ± 0.63). Similar levels were seen in the thalamus for the two radioligands. A lower [11C]Ro15 4513 Vd was seen in the cerebellum compared with [11C]flumazenil (2.65 ± 0.21 vs. 4.97 ± 0.61;Table 1).
Human positron emission tomography data
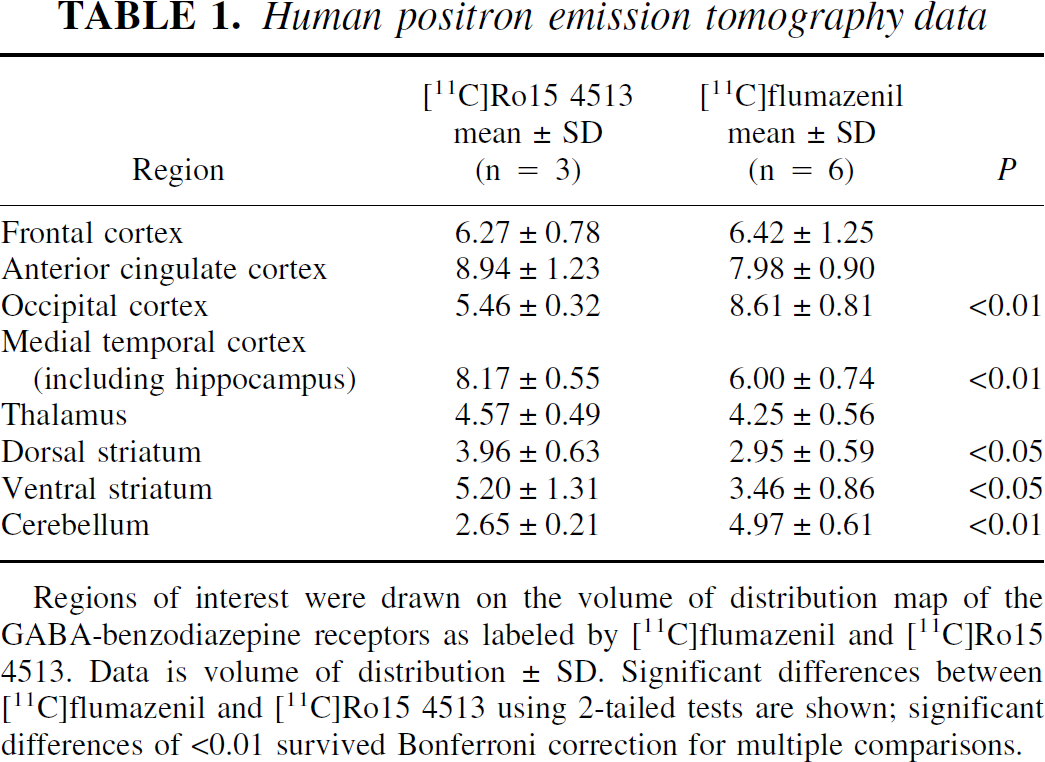
Regions of interest were drawn on the volume of distribution map of the GABA-benzodiazepine receptors as labeled by [11C]flumazenil and [11C]Ro15 4513. Data is volume of distribution ± SD. Significant differences between [11C]flumazenil and [11C]Ro15 4513 using 2-tailed tests are shown; significant differences of <0.01 survived Bonferroni correction for multiple comparisons.
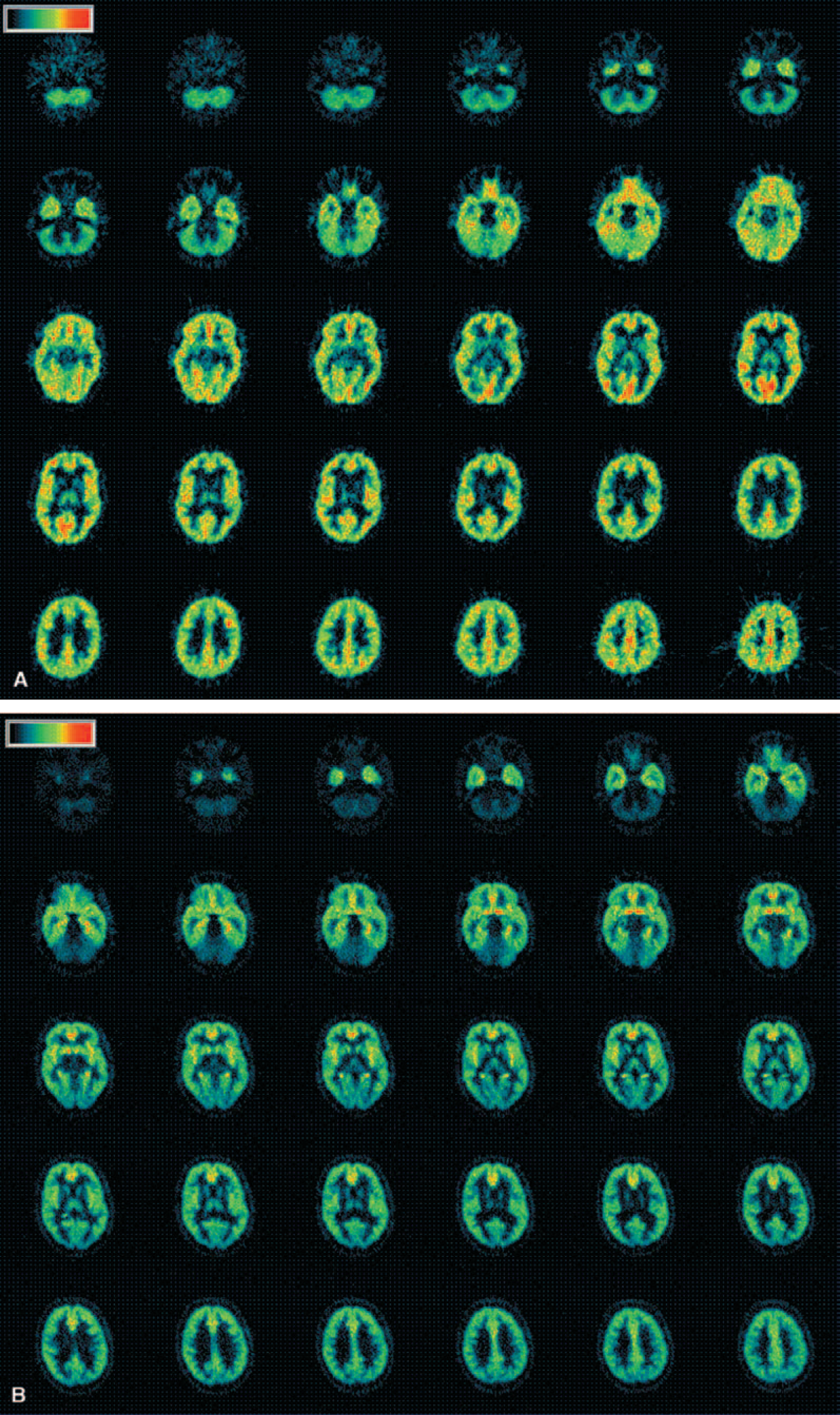
Human images. Typical
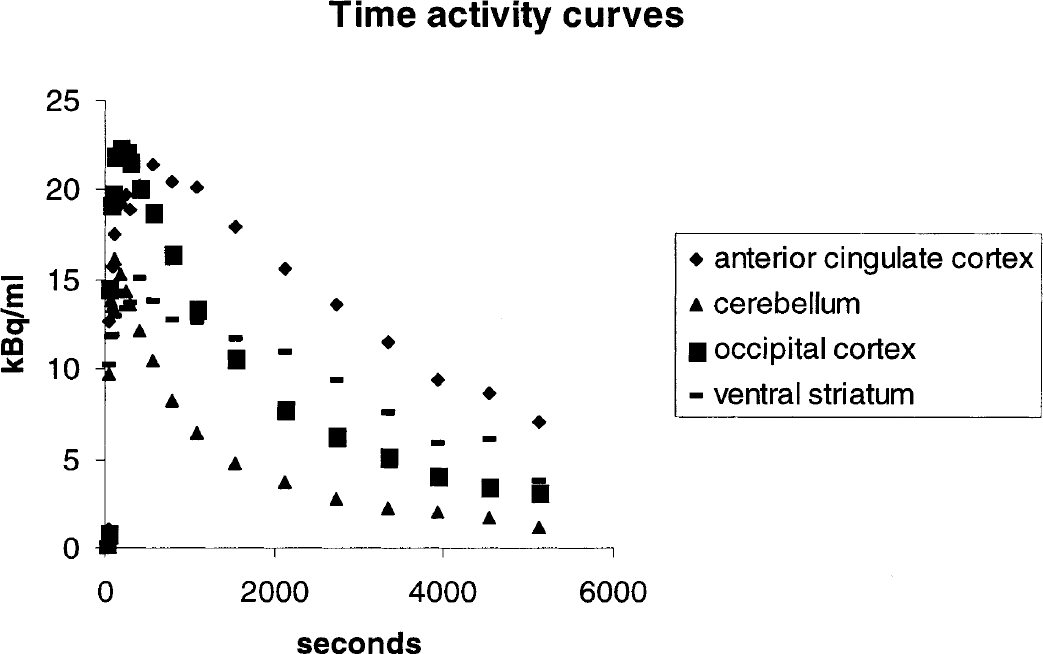
Time activity curves for [11C]Ro15 4513 uptake in four regions of interest, anterior cingulate cortex, cerebellum, occipital cortex and ventral striatum in man.
Rat.
The rat HIDAC-PET images (Fig. 3A and B) also reveal different patterns of distribution for the two radioligands. Using the medulla as a reference region (representing nonspecific binding for normalization), this semiquantitative analysis shows that specific binding of [11C]Ro15 4513 was lower than that for [11C]flumazenil, throughout the brain (Table 2). Binding of [11C]Ro15 4513 was particularly low in the cerebellum and retention of [11C]flumazenil was greater throughout the cortex. However, relative uptake of [11C]Ro15 4513 in the hippocampus (2.5 ± 0.2) compared with that in the frontal cortex (2.2 ± 0.4) was higher than that seen with [11C]flumazenil (3.4 ± 0.3 vs. 3.9 ± 0.5).
Dual-HIDAC scan data
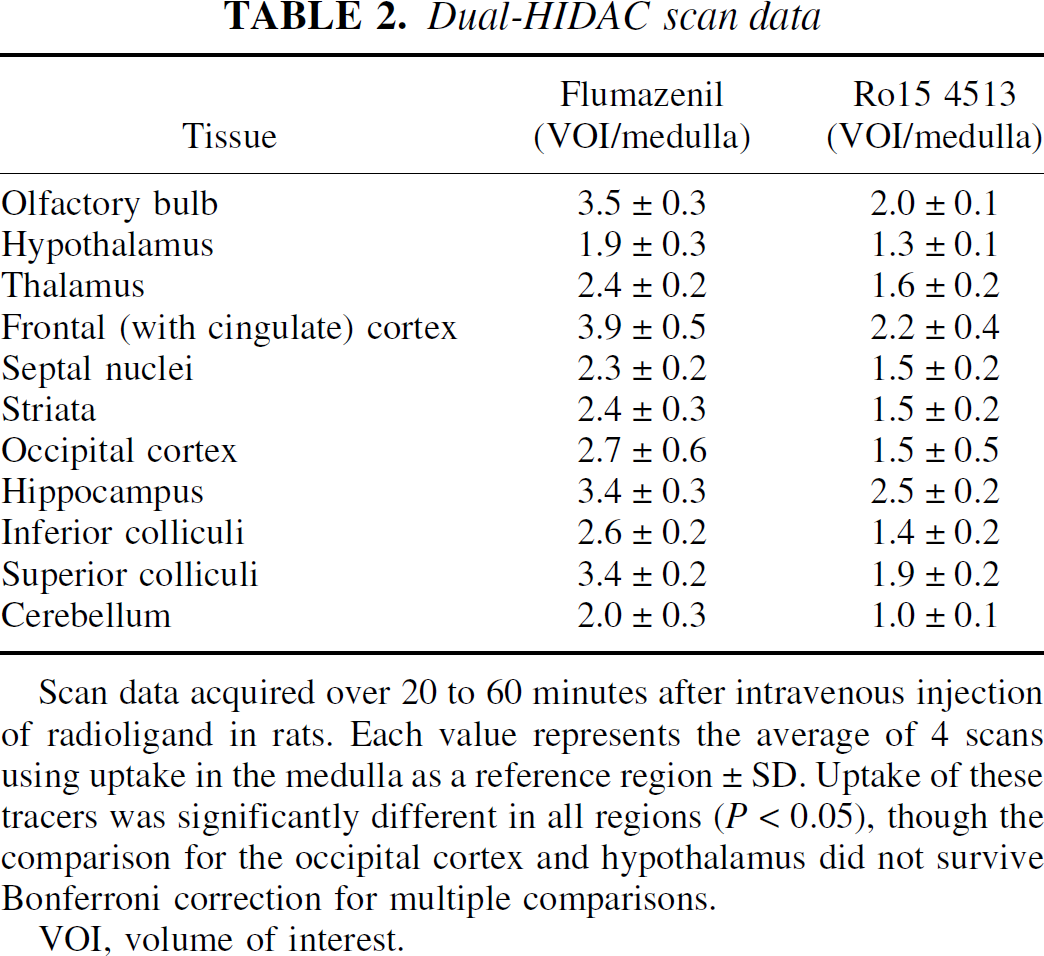
Scan data acquired over 20 to 60 minutes after intravenous injection of radioligand in rats. Each value represents the average of 4 scans using uptake in the medulla as a reference region ± SD. Uptake of these tracers was significantly different in all regions (P < 0.05), though the comparison for the occipital cortex and hypothalamus did not survive Bonferroni correction for multiple comparisons.
VOI, volume of interest.
Uptake of [11C]flumazenil
Human versus rat.
Although the patterns of [11C]Ro15 4513 uptake in rats and humans were similar, the most striking difference was observed in the striatum. Uptake was very low in the rat, at almost background levels, whereas in humans, uptake of [11C]Ro15 4513 in the striatum was higher than for [11C]flumazenil, particularly in the ventral striatum.
Dissection
Uptake of radiolabelled Ro15 4513.
After intravenous injection, [11C]Ro15 4513 was rapidly and highly extracted into rat brain, giving “uptake” values of 3 to 4 (cpm/g tissue)/(injected cpm/g body weight) 1 minute after injection (data not shown). Thereafter, the radioactivity content decreased in a tissue-dependent manner until a secular or transient equilibrium was achieved approximately 40 to 60 minutes after injection. The brain biodistribution in control (baseline) rats at 60 minutes after injection of [11C] or [3H]Ro15 4513 is given in Table 3, which also shows the tissue—medulla with pons ratios as a measure of total versus nonspecific binding. Medulla with pons was chosen as a “reference” tissue because its radioactivity content was unaffected by any of the treatment regimes, and to avoid the use of cerebellum, known to be rich in α1 subunit-containing GABAA receptors (Atack et al., 1999). Inoue et al. (1992) used the pons as a reference region of interest for [11C]Ro15 4513 PET studies in humans. The highest concentration of radiolabelled Ro15 4513 was in hippocampus (0.30 ± 0.08) with the olfactory bulbs and “frontopolar with cingulate” cortex showing approximately one half to one third of this level of uptake. Radioactivity in the occipital cortex (0.08 ± 0.03) was less than that in frontopolar cortex and similar to that in hypothalamus. The lowest levels were seen in medulla with pons, striatum, cerebellum, thalamus, and inferior colliculi (Table 3).
Rat binding data
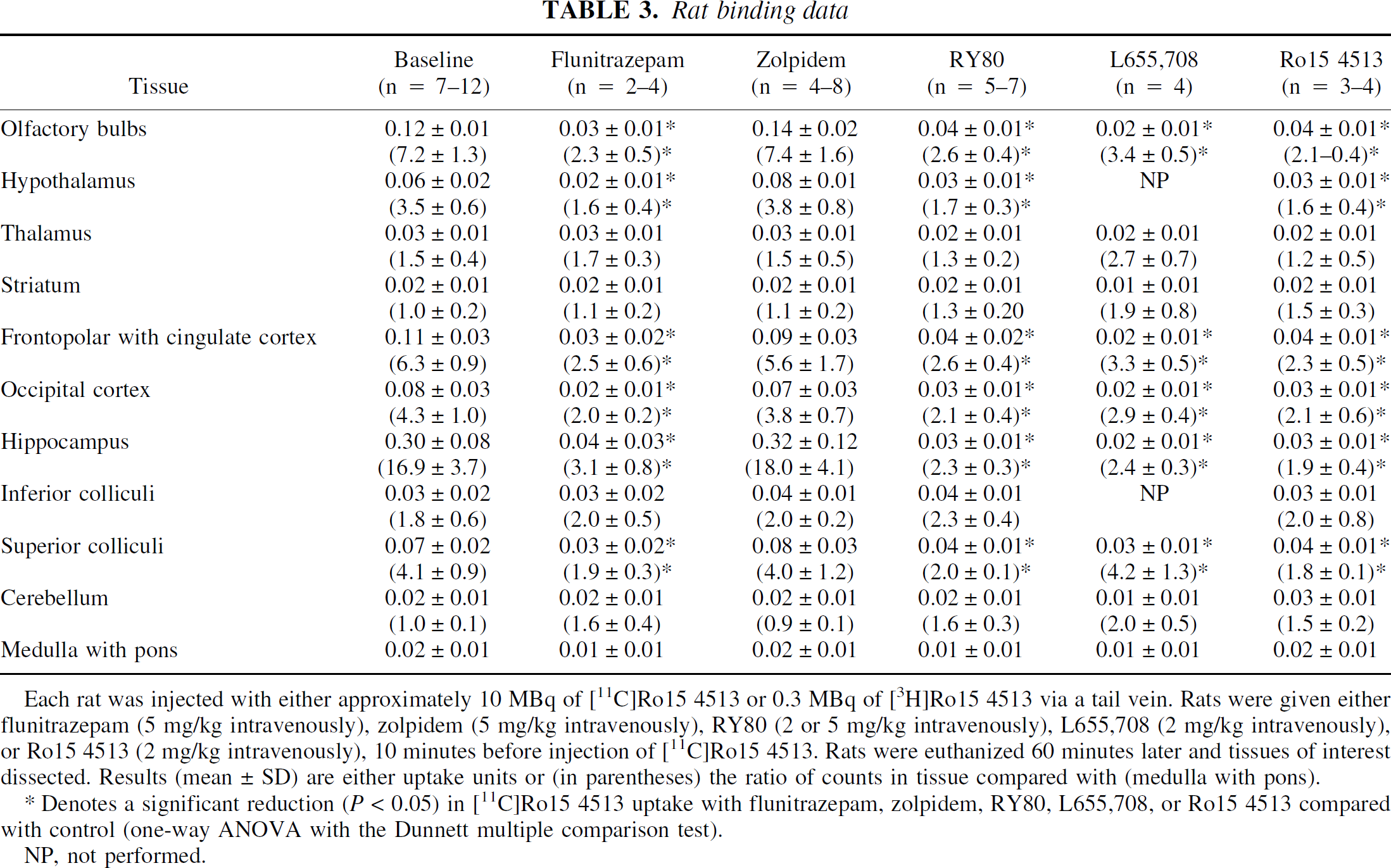
Each rat was injected with either approximately 10 MBq of [11C]Ro15 4513 or 0.3 MBq of [3H]Ro15 4513 via a tail vein. Rats were given either flunitrazepam (5 mg/kg intravenously), zolpidem (5 mg/kg intravenously), RY80 (2 or 5 mg/kg intravenously), L655,708 (2 mg/kg intravenously), or Ro15 4513 (2 mg/kg intravenously), 10 minutes before injection of [11C]Ro15 4513. Rats were euthanized 60 minutes later and tissues of interest dissected. Results (mean ± SD) are either uptake units or (in parentheses) the ratio of counts in tissue compared with (medulla with pons).
NP, not performed.
Denotes a significant reduction (P < 0.05) in [11C]Ro15 4513 uptake with flunitrazepam, zolpidem, RY80, L655,708, or Ro15 4513 compared with control (one-way ANOVA with the Dunnett multiple comparison test).
Competition studies
After pretreatment with unlabeled intravenous Ro15 4513 (2 mg/kg), the concentration of and tissue:medulla ratio of radiolabeled Ro15 4513 were significantly reduced in all regions studied, except in those tissues (e.g., striatum, thalamus, and cerebellum) where low uptake levels were seen in baseline conditions (Table 3). Intravenous pretreatment with the benzodiazepine receptor agonist flunitrazepam (2 mg/kg) similarly resulted in a significant reduction in radiolabeled Ro15 4513 uptake, to levels similar to those seen after pretreatment with unlabeled Ro15 4513. In contrast, intravenous pretreatment with the α1 relatively selective agonist, zolpidem (5 mg/kg), resulted in no significant decreases in either concentration or tissue:medulla ratio of radiolabeled Ro15 4513 in any of the tissues studied. Conversely, intravenous pretreatment with the α5 relatively selective inverse agonist RY80 (5 mg/kg) resulted in significantly reduced radiolabeled Ro15 4513 in the olfactory bulbs, hypothalamus, frontopolar and occipital cortices, hippocampus and superior colliculi, to nonspecific binding levels. Another more α5 selective ligand L655,708 also resulted in a similar pattern of significant reductions. There were no significantly different effects between flunitrazepam, RY80 and L655,708 (one-way analysis of variance with Bonferroni multiple comparison test) and each resulted in a distribution of radioactivity equivalent to that obtained by pretreatment with Ro15 4513.
To check that the dose of zolpidem used in the present series (5 mg/kg intravenously, 10 minutes before the radioligand) resulted in GABA-benzodiazepine receptor occupancy, [11C]flumazenil binding was measured in rat brain tissues 60 minutes after intravenous injection of the radioligand, with and without predosing. Zolpidem resulted in a statistically significant reduction in specific binding in frontopolar with cingulate cortex (50%), hypothalamus (48%), olfactory bulbs (53%), occipital cortex (46%), inferior colliculi (54%), and cerebellum (33%). In the other tissues dissected, no significant reduction of [11C]flumazenil binding was seen. In the same series of experiments, the α5-subunit containing GABAA receptor-selective compound, L655,708, caused a significant reduction in binding of [11C]flumazenil only in olfactory bulbs (50%), frontopolar with cingulate cortex (46%), and hippocampus (51%), reflecting the known localization of α5-subunit containing GABAA receptors in rat brain (Sur et al., 1999).
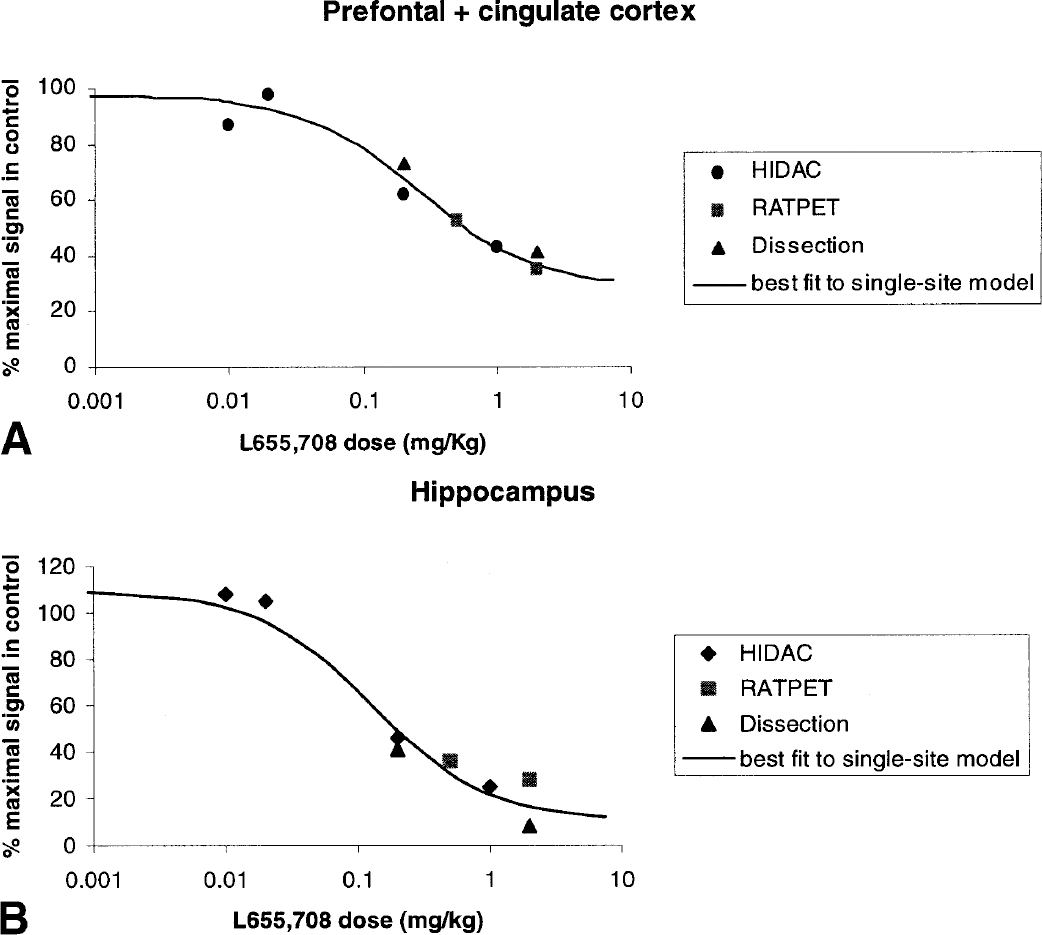
In a further study, blocking of [11C]Ro15 4513 binding with graded intravenous doses of L655,708 (0.01 to 2 mg/kg) was performed in scanned rats (RATPET and dual-HIDAC) and in ex vivo dissection experiments. Pooling the data enabled the quantification of a dose-dependent effect of L655,708 on [11C]Ro15 4513 binding. As can be seen from Fig. 4, increasing doses of L655,708 resulted in blocking of radiolabeled Ro15 4513 binding in both the hippocampus and frontal cortex. Although not statistically significant, the highest dose of L655,708 (2 mg) reduced Ro15 4513 labeling in the hippocampus to a greater extent than in the frontal cortex. This effect could be due to the relatively higher density of other α subunits (particularly α1) in the frontal cortex, to which Ro15 4513 is likely to be binding (Fig. 4).
Finally, the regional uptake of [11C]Ro15 4513 in our studies and levels of GABA-benzodiazepine receptor binding identified with another α5 selective ligand, [3H]L655,708 (Sur et al., 1999), are closely correlated (Fig. 5; r = 0.952). Using published kd values in the rat (Lui et al., 1996; Quirk et al., 1996; Sur et al., 1999), we have calculated regional relative Bmax values for [11C]Ro15 4513 and [11C]flumazenil and have found that the size of the specific signal obtained with [11C]Ro15 4513 is consistent with its binding to the α5 subtype (12 to 17%). Thus, at tracer quantities, [11C]Ro15 4513 labels α5-containing receptors because of its high affinity for this subtype, despite their low abundance.
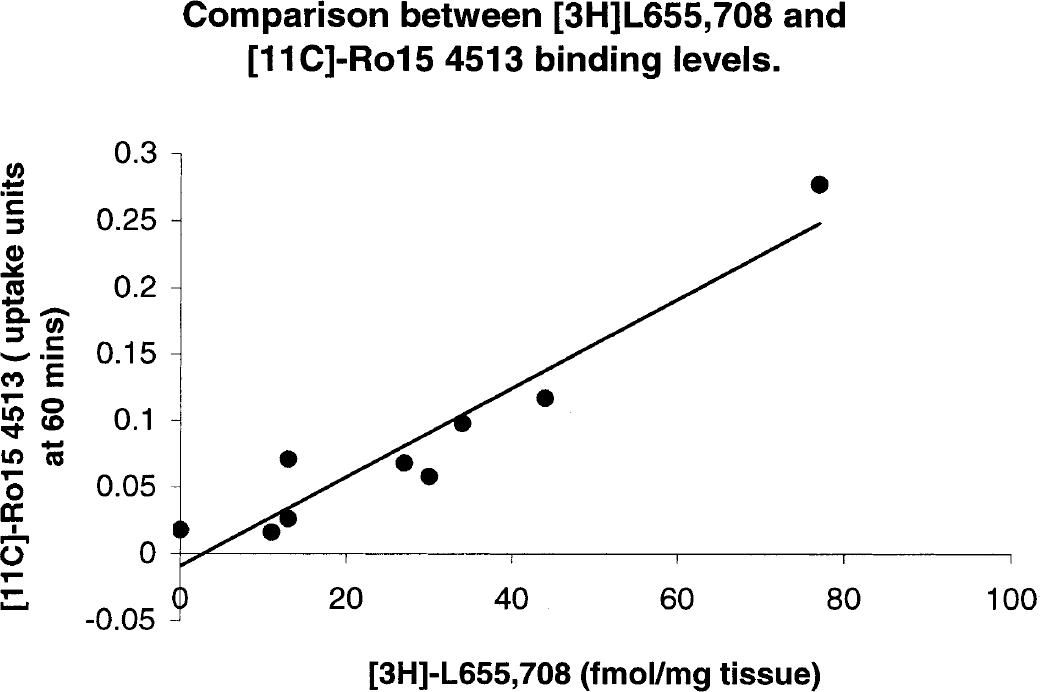
Comparison between [3H]L655,708 and [11C]Ro15 4513 binding levels in rats. The [3H]L655,708 data are taken from Sur et al. (1999). See Table 2 and Materials and Methods for a description of [11C]Ro15 4513 uptake.
DISCUSSION
We have characterized the PET ligand [11C]Ro15 4513 in rats and humans and showed that its uptake has a primarily limbic distribution, which is consistent with the view that it is labeling GABA-benzodiazepine receptors containing the α5 subunit. We performed fully quantitative analysis in man using a metabolite-corrected plasma input function. In man, [11C]Ro15 4513 uptake was relatively greater in limbic areas when compared with frontal cortical regions, and was lower in the occipital cortex and cerebellum than that seen with [11C]flumazenil. In rats, regional uptake of [11C]Ro15 4513 in our in vivo studies and GABA-benzodiazepine receptor density identified in vitro with an α5 selective ligand, [3H]L655,708 (Sur et al., 1999), were closely correlated.
In vivo blocking studies in rats showed that radiolabeled Ro15 4513 binding was reduced to nonspecific levels only by drugs that have affinity for the α5 subtype (flunitrazepam, RY80, L655,708, but not zolpidem). Regarding the zolpidem studies, Benavides et al., (1992) reported only a partial displacement of [3H]flumazenil by zolpidem in rat brain. A dose of 30 mg/kg given intraperitoneally 30 minutes before the radiotracer resulted in a 20% to 80% inhibition of binding density (as assessed with quantitative autoradiography), with maximal inhibition in cerebellum, a tissue enriched in α1-subunit—containing GABAA receptors. More recently, Atack et al., (1999) have reported an ID50 value of 10 mg/kg for zolpidem inhibition of binding of [3H]flumazenil in mouse cerebellum, when given intraperitoneally 30 minutes before the radiotracer. In a small supplementary study, we showed that the zolpidem dosing regime used in the present study investigating blockade of radiolabelled Ro15 4513 binding (5 mg/kg intravenously, 10 minutes before the radioligand injection) was sufficient to cause a significant reduction in binding of [11C]flumazenil in several tissues, including cerebellum. Taken together, these results therefore suggest that in vivo Ro15 4513 predominantly labels GABA-benzodiazepine receptors containing the α5 subunit because of its relatively higher affinity for such receptors, as previously shown in vitro.
It is timely to be able to image the α5-containing GABA-benzodiazepine receptor, because functions of the different subtypes of this receptor are being increasingly defined. This is important to inform development of subtype selective drugs to minimize unwanted side effects, such as sedation or tolerance. For instance, the α1-containing subtype may be involved with a number of functions, including sedation and the α2, with anxiolysis (Low et al., 2000; McKernan et al., 2000; Rudolph et al., 1999). Until recently, determining the function of the α5-containing subtype relied on relatively subtype-selective drugs, but a knockout mouse model is now available (Collinson et al., 2001). As reported, [11C]Ro15 4513 uptake is highest in the hippocampus in rats and humans, reflecting the high level of the α5 subunit in this region (Fritschy and Möhler et al., 1995; Pirker et al., 2000; Wisden et al., 1992). Because the hippocampus is critically involved in memory encoding and benzodiazepines are potent amnestic agents, their action may be mediated through enhancing GABA transmission in this region.
Benzodiazepine inverse agonists have actions opposite to those of agonists, in that they reduce the inhibitory effects of GABA and can lead to the development of anxiety, seizures, and enhanced memory (Polc et al., 1982; Sarter et al., 1988). Several selective α5 inverse agonists (e.g., RY24, S-8510) are promnestic yet not proconvulsant or anxiogenic in rodents (Huang et al., 2000; Kawasaki et al., 1996). More recently, an α5-knockout mouse has been developed and provides further evidence for a role of the α5-containing receptor in memory, because these mice perform better than their wild-type counterparts but are not more anxious (Collinson et al., 2001).
Although only 28% of GABA-benzodiazepine receptors in the human hippocampus contain α5 (compared with 25% in the rat), this part of the brain contains the highest concentration of these receptors (Fritschy and Möhler 1995; Pirker et al., 2000; Sur et al., 1999; Wisden et al., 1992). A recent study (Howell et al., 2000) found no reduction in α5 receptors in the hippocampus of Alzheimer patients at postmortem, so these receptors may be an ideal therapeutic target for memory enhancement. Because clinical interest in the use of α5 selective inverse agents in Alzheimer disease is increasing, it would be of considerable interest to be able to image this receptor subtype in humans. Moreover, a radioligand for this purpose could help optimize the dosing and pharmacokinetics of new α5 selective agents.
The α5-containing benzodiazepine receptor may also be involved in benzodiazepine tolerance. Long-term exposure to benzodiazepines leads to reduced levels of α5-containing receptors, specifically those in the CA1 region of the hippocampus (Li et al., 2000). Studies have shown that α5-containing receptors are expressed particularly in the dendritic tree of CA1 to CA3 pyramidal neurons, molecular layer of the dentate gyrus, and entorhinal cortex (Fritschy and Möhler 1995, Sur et 1999). Dendritic α5-containing receptors play a key role in monitoring excitatory inputs impinging on pyramidal cell dendrites (Nusser et al., 1996). Consequently, downregulation of these could lead to the hyperexcitable state seen in benzodiazepine withdrawal.
In the human cerebral cortex, [11C]Ro15 4513 uptake appears to be prominent in the deeper layers, as is the case in rat cerebral cortex (Fritschy and Möhler, 1995). Although it has been reported that α5-containing receptors represent only 4.2% of receptors in the rat cortex (Quirk et al., 1996), [11C]Ro15 4513 uptake in the human cerebral cortex in the present study suggests that more α5-containing receptors are present. Although [11C]Ro15 4513 predominantly labels the α5 subtype, other subtypes of the GABA-benzodiazepine receptor, particularly the α1-containing subtype, are present in higher levels; hence, some contribution is likely from binding to non-α5 subtypes. Although competition studies in the rat showed that α1-selective zolpidem did not significantly reduce the concentration of radiolabelled Ro15 4513 in the cortex, a slight reduction was observed. The results of the rat studies using both dissection and PET, with graded doses of L655,708 to block radiolabelled Ro15 4513 binding showed that uptake was reduced to background levels in the hippocampus, but not in the frontal cortex (Figure 3). This finding implies a contribution from the predominant α1-containing subtype to the PET image of [11C]Ro15 4513. Thus, the higher-than-expected cortical signal in both rats and humans may be due to some labeling of the α1-containing GABA-benzodiazepine receptor by [11C]Ro15 4513.
Although Ro15 4513, unlike other benzodiazepines, binds with relatively high affinity to α6-containing GABA-benzodiazepine receptors (Ki 3.8;Sieghart, 1995) that are found predominantly in the cerebellum, little binding was observed here in vivo. Presumably the 10-fold—lower affinity of Ro15 4513 for the α6 (Ki 3.8;Huang et al., 2000) compared with the α5 (Ki 0.3;Huang et al., 2000) subtype and their comparatively low abundance accounts for the relative lack of α6 labeling.
The anteroposterior gradient of [11C]Ro15 4513 uptake in the human cortex with lowest levels in the occipital cortex is the reverse of that seen with [11C]flumazenil. The distribution of the α5-containing GABA-benzodiazepine receptor in rats does not show such a gradation, and no comparable study has been performed in humans. The functional impact of this differential distribution of α1-containing and α5-containing GABA-benzodiazepine receptors is unknown.
A marked difference was seen between rats and humans regarding the uptake of [11C]Ro15 4513 in the striatum. Onoe et al., (1996) previously noted that in nonhuman primates, [11C]Ro15 4513 intensely labeled the nucleus accumbens. In humans, [11C]Ro15 4513 labeling of the ventral striatum was greater than that in the dorsal striatum, suggesting that the nucleus accumbens is particularly labeled. In the rat, [11C]Ro15 4513 uptake throughout the striatum was very low, at almost background levels. The nucleus accumbens is a key part of the mesolimbic reward system and GABA interneurons modulate dopaminergic function (Koob and Nestler, 1997). Such a difference in distribution of the GABA-benzodiazepine receptors containing the α5 subunit will have a significant impact on the effects of subtype-selective drugs, and rat studies are unlikely to reflect effects of such drugs in humans.
In summary, we have characterized a fully quantitative PET method for labeling the α5 subtype of the GABA-benzodiazepine receptors in vivo. This PET radioligand will allow us to explore the role of this particular GABA-benzodiazepine receptor subtype in humans. Such a development is timely given our rapidly expanding knowledge about which subtypes mediate specific effects of benzodiazepines in rodents. The finding that humans have a different distribution of these receptors than rodents is clearly important when applying results of experiments using rat models to humans. Moreover, because the α5-containing GABA-benzodiazepine receptors are predominantly located in the limbic system, this suggests they play a role in emotional states, memory, and in disease processes such as dementia and anxiety.
Footnotes
Acknowledgments
The authors thank Drs. P. Skolnick and J. Wood for their gift of RY80 and all the staff at IRSL (formerly the MRC Cyclotron Unit), particularly Dr. Ralph Myers, Claire Townsend, Shaun Creasey, and Andy Blythe, for their help with this study.
