Abstract
Water homeostasis in the brain is of central physiologic and clinical importance. Neuronal activity and ion water homeostasis are inextricably coupled. For example, the clearance of K+ from areas of high neuronal activity is associated with a concomitant water flux. Furthermore, cerebral edema, a final common pathway of numerous neurologic diseases, including stroke, may rapidly become life threatening because of the rigid encasement of the brain. A water channel family, the aquaporins, facilitates water flux through the plasma membrane of many cell types. In rodent brain, several recent studies have demonstrated the presence of different types of aquaporins. Aquaporin 1 (AQP1) was detected on epithelial cells in the choroid plexus whereas AQP4, AQP5 and AQP9 were localized on astrocytes and ependymal cells. In rodent brain, AQP4 is present on astrocytic end-feet in contact with brain vessels, and AQP9 is found on astrocytic processes and cell bodies. In basal physiologic conditions, AQP4 and AQP9 appear to be implicated in brain homeostasis and in central plasma osmolarity regulation. Aquaporin 4 may also play a role in pathophysiologic conditions, as shown by the reduced edema formation observed after water intoxication and focal cerebral ischemia in AQP4-knockout mice. Furthermore, pathophysiologic conditions may modulate AQP4 and AQP9 expression. For example, AQP4 and AQP9 were shown to be upregulated after ischemia or after traumatic injuries. Taken together, these recent reports suggest that water homeostasis in the brain is maintained by regulatory processes that, by control of aquaporin expression and distribution, induce and organize water movements. Facilitation of these movements may contribute to the development of edema formation after acute cerebral insults such as ischemia or traumatic injury.
Water is quantitatively the major component of all the organisms that maintain high water flux through plasma membranes. In the human body, the exchanged volume is believed to be approximatively 100 L per day (see Heymann et al., 1998). Because water movement across cell membranes is a fundamental property of life, it is no surprise that the initial cloning of aquaporin 1 (AQP1), the archetypal water channel, boosted research for the study of water transport and osmoregulation in mammals. Forty years ago, studies on red blood cell membranes showed that water crosses plasma membranes by a diffusion process that, in contrast to simple diffusion through pure lipid bilayers, does not need high-energy activation. This result raised the hypothesis of the presence of water pores in plasma membranes of red blood cells (Sidel and Solomon, 1957). Multiple observations provided clues for the presence of water channels in a variety of specialized membranes; their molecular identity, however, remained unknown until the 1990s. A protein highly expressed in plasma membranes of red blood cells and renal proximal tubules was suspected to be a water channel (Agre et al., 1987; Denker et al., 1988). Characterization of this protein (Preston and Agre, 1991; van Hoek et al., 1991) showed high homology with a membrane-intrinsic protein of the lens fiber (Gorin et al., 1984), the major integral protein (MIP) of the eye lens, which was later named AQP0. DNA transfection in oocytes has shown that this protein increases water flux through the plasma membrane. Indeed, in hypoosmostic medium, cell swelling is more rapid in transfected than in nontransfected oocytes. After this demonstration, the authors named this protein aquaporin (Agre, 1997).
The aquaporin family contains 11 subtypes ubiquitously distributed in tissues and organisms. Taking into account the presumed role of aquaporins in water homeostasis, the regulation of expression and activity was largely studied in different physiologic and pathophysiologic conditions. After a rapid description of the aquaporin family, we will focus this review on hypotheses concerning its physiologic roles in the brain and consequences in brain disorders.
WATER CHANNELS: THE AQUAPORIN FAMILY
Aquaporin subtypes and classification
As previously mentioned, the aquaporins are a growing family of molecular water channels, first identified in the early 1990s. Aquaporin 1 was the first specific water channel to be characterized in red blood cells (Preston and Agre, 1991; Preston et al., 1992). Presently, the aquaporin family comprises 11 subtypes, AQP0 to AQP10. The molecular weight of approximatively 30 kd could vary with different glycosylations on the extracellular domains.
Despite a common molecular structure (see Molecular Structure of the Aquaporins), mammalian aquaporins have now been subdivided into three functional groups, according to permeability characteristics (Verkman and Mitra, 2000): the aquaporins, including AQP0, AQP1, AQP2, AQP4, AQP5, AQP6, are permeable to water; the aquaglyceroporins, including AQP3, AQP7, AQP8, are permeable to water, glycerol, and urea; the neutral solute channels, including AQP9, are permeable to water, glycerol, urea, purines, pyrimidines, and monocarboxylates. Aquaporin 10 was recently cloned and is highly expressed in duodenum and jejunum (Hatakeyama et al., 2001). Like AQP9, AQP10 is permeable to water and neutral solutes, but, surprisingly, it is not permeable to urea and glycerol (Hatakeyama et al., 2001). Aquaporin 10 showed amino acid sequence homology with AQP3 (53%), AQP9 (52%) and AQP7 (43%). The structural explanation and the physiologic relevance of the differences between the aquaporins are not yet known.
The aquaporins are ubiquitously distributed in mammalian tissues. To date, 6 aquaporin subtypes have been described in rodent brain, AQP1, AQP3, AQP4, AQP5, AQP8, AQP9 (Badaut et al., 2001; Elkjaer et al., 2000; Nielsen et al., 1997; Venero et al., 1999; Yamamoto et al., 2001b).
Molecular structure of the aquaporins
The configuration of AQP1 reflects the common structural features of the aquaporin family. They are formed by two tandem repeats of three membrane-spanning α helices, with cytoplasmic carboxyl- and amino-terminals. Two connecting loops, each containing an Asn-Pro-Ala motif, are believed to determine water selectivity (Murata et al., 2000) (Fig. 1). The essential role of these proteins is suggested by their presence in bacteria through plants to mammals, possibly as water and small neutral solute–transporting channels. Model studies suggest that in lipid bilayers the protein presents a pore with a diameter between 3 and 6 Å (for review see, Heymann et al., 1998). The specificity for water seems to be due to the diameter of the pore (Gutierrez et al., 1995). The absence of ionic permeability may be due to the size of hydrated ions (Heymann et al., 1998). This explanation is not entirely satisfying, however, because AQP3, AQP7, and AQP8 are permeable to molecules like glycerol and urea whereas AQPO, AQP1, AQP2, AQP4, AQP5, and AQP6 are permeable to water (for review see King and Agre, 1996). Furthermore, AQP6 has been recently shown to conduct anions as well as water (Yasui et al., 1999).
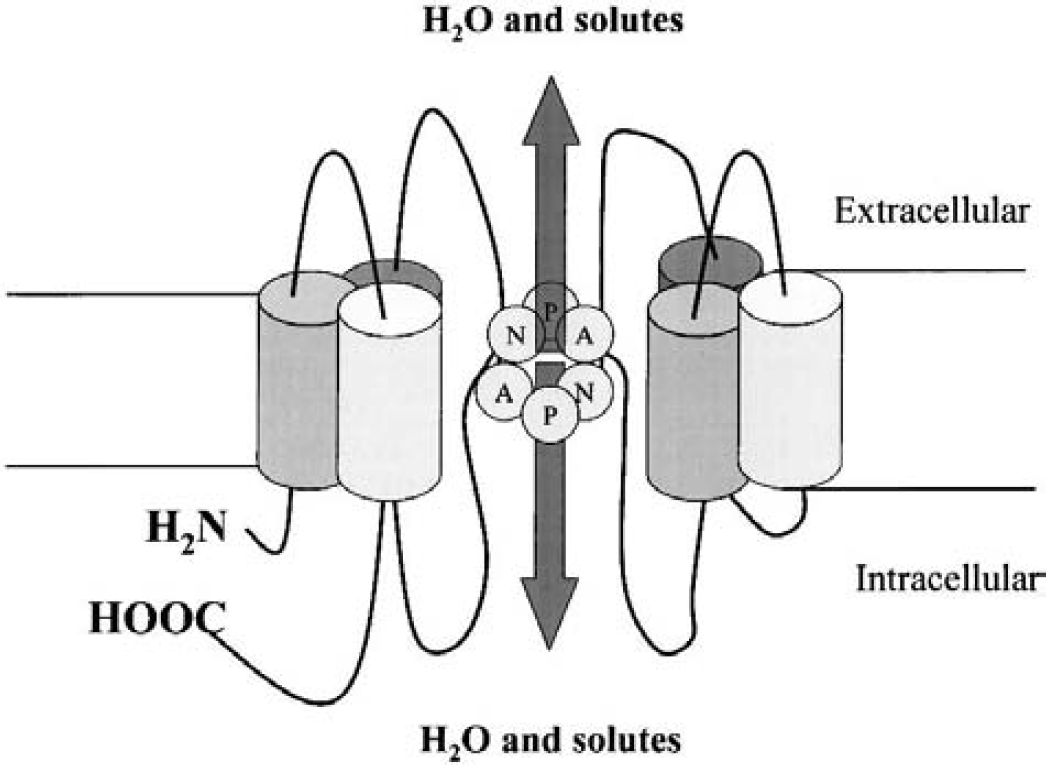
Schematic representation of the common aquaporin structure. The aquaporins are formed by two tandem repeats of three membrane-spanning α helices. Two connecting loops, each containing an Asn-Pro-Ala (NPA) motif, are supposed to form pore in plasma membrane.
AQUAPORINS IN THE BRAIN
Of the six aquaporin subtypes (AQP1, AQP3, AQP4, AQP5, AQP8, AQP9) described in the rodent brain, much of the data come from studies on AQP4, one of the first subtypes described in the CNS. More recently, AQP3 and AQP8 messenger RNAs (mRNAs) were found in glial and neuronal cells of the rat brain. Patterns of brain aquaporin distribution are described in Aquaporin 1: Implication in Cerebrospinal Fluid Formation, Aquaporin 4: Brain Distribution and Putative Funtions, and Aquaporin 9: Brain Distribution and Putative Functions.
Aquaporin 1: Implication in cerebrospinal fluid formation
In rodents, AQP1 is found on epithelial cells of choroid plexus (Hasegawa et al., 1993, 1994; Masseguin et al., 2000; Nielsen et al., 1993), where it was concentrated at the apical pole (Masseguin et al., 2000; Nielsen et al., 1993). The polarized distribution and the general expression are downregulated in response to altered gravity, as shown after 14-day spaceflight (Masseguin et al., 2000). The epithelial localization in choroid plexus and the effects of low gravity are consistent with the role of AQP1 in cerebrospinal fluid (CSF) formation. This protein was proposed to be the major water-transporting protein in choroid plexus.
Whereas capillary endothelial cells in the systemic circulation show immunostaining to AQP1 (Nielsen et al., 1993), capillaries in the rat cerebrovascular system remained unstained (Kobayashi et al., 2001). Conversely, vascular AQP1 immunolabeling was observed in various tumor cell lines (mammary carcinomas and glioblastomas) implanted in the rat brain (Endo et al., 1999). Tumor vascularization is known to be characterized by an impaired blood–brain barrier (BBB; Vajkoczy and Menger, 2000). The absence of AQP1 expression in brain capillaries might be a specific phenotype related to impaired barrier.
Aquaporin 4: Brain distribution and putative functions
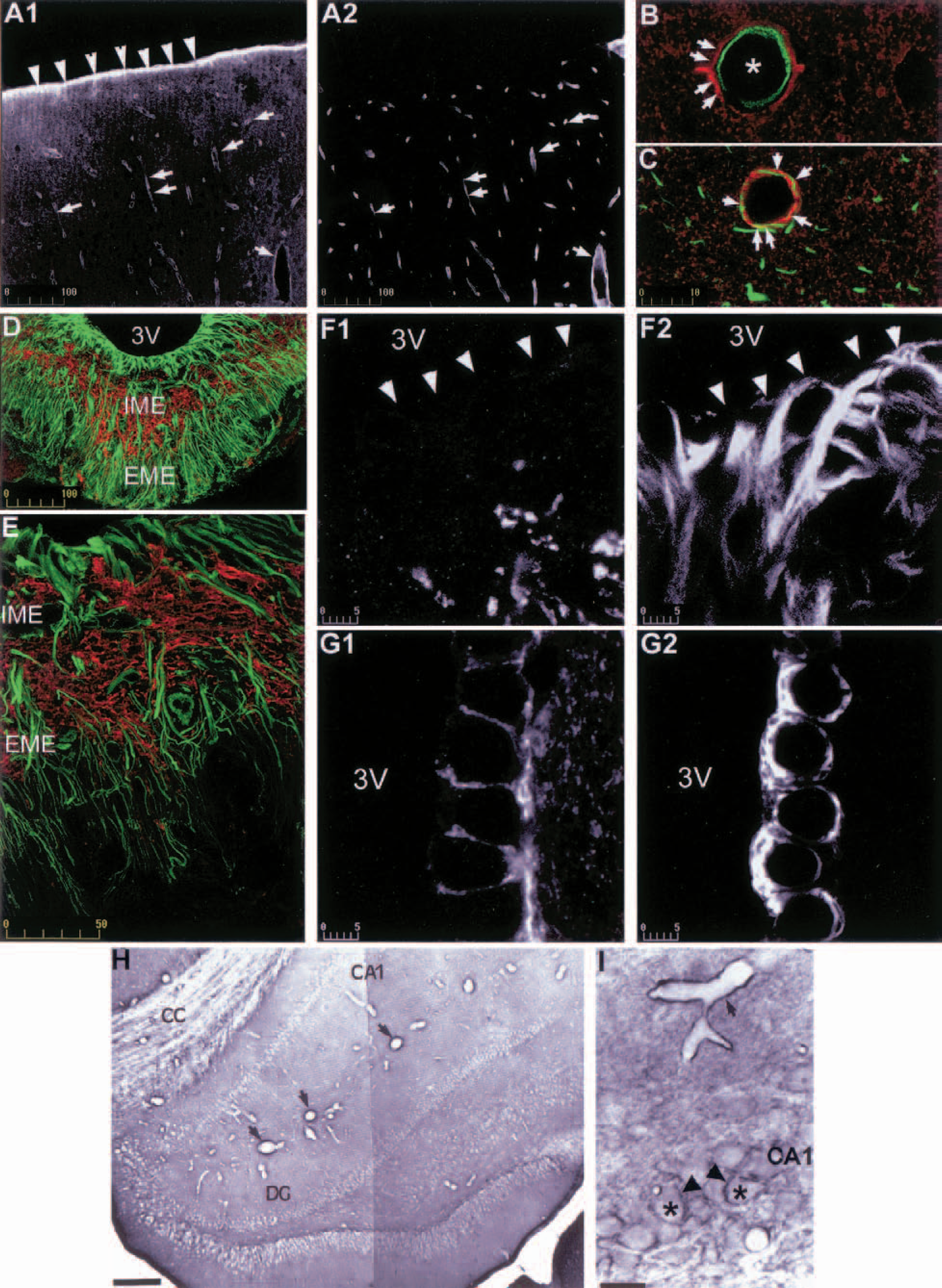
A hybridization study in 1994 showed for the first time AQP4 expression in the rat brain (Jung et al., 1994). Aquaporin 4 mRNA was detected in glia limitans, ependymal lining system, cerebellum, hippocampal dentate gyrus, and in the supraoptic and paraventricular nuclei of the hypothalamus (Jung et al., 1994). In addition, a low but significant AQP4 mRNA hybridization was also reported in neocortex, in the hippocampal areas CA1-CA3, in the nucleus of stria terminalis, and in medial habenular nucleus (Venero et al., 1999). Our immunohistochemical studies confirmed this regional distribution (Figs. 2A1, 2H, 2I, and 3E), and the immunolabeling study of Nielsen et al. (1997) showed that, in the supraoptic nuclei, neocortex, and cerebellum, AQP4 was present on astrocyte processes. The expression and distribution of AQP4 has been mainly performed in rodent brains. All immunologic studies are consistent with AQP4 expression in astrocytes and ependymal cells and with the absence of neuronal expression.
Confocal images of double immunolabeling—aquaporin 4 (AQP4) with endothelial brain antigen (EBA) (
Confocal pictures of double immunolabeling—aquaporin 4 (AQP4) with endothelial brain antigen (EBA) (
Several observations suggest that, in brain and kidney, AQP4 molecules are clustered in geometric structures named orthogonal arrays of particles (Rash and Yasumura, 1999; Rash et al., 1998; Verbavatz et al., 1997). In agreement with these observations, recent results demonstrate that AQP4 ontogenesis coincides with the formation of orthogonal arrays of particles (Nico et al., 2001). After cryofracture, these microscopic structures showed square shapes included in cytoplasmic membranes of astrocytic end-feet (for review, see Wolburg, 1995). The physiologic function of these geometric arrangements is not yet clearly determined but it may be related to the astrocytic potassium-buffer function (Grange-Messent et al., 1996). Aquaporin 4 expression during brain development might be related to the formation of the BBB. In accord with this hypothesis, a significant increase in AQP4 expression was shown in the 14-day postnatal cerebellum (Wen et al., 1999). More recently, Nico et al. (2001) observed that expression of AQP4 and BBB phenotype coincide during development. Furthermore, interactions between AQP4 in astrocytes and BBB phenotype in endothelium could persist after development, because pathologic conditions such as traumatic lesions producing BBB breakdown disclosed a decrease of AQP4 expression (Ke et al., 2001). However, whether the two factors (AQP4 and BBB) were independently affected or not by the lesion remains to be determined.
Two mice strains with deficiencies in gene expression have provided models to study the role of perivascular AQP4. In the perivascular space, dystrophin deficiency, as seen in X-chromosome–linked muscular dystrophy (mdx) mice, results in a marked decrease of AQP4 immunolabeling and a swelling of astrocytic end-feet (Frigeri et al., 2001). In this model of Duchenne muscular dystrophy, a reduction of AQP4 signal was also shown in skeletal muscle fibers. These results, together with results showing the absence of AQP4 on perivascular astrocyte end-feet in α1-syntrophin (a protein linked to dystrophin)–knockout mice (Neely et al., 2001; Yokota et al., 2000), suggest that the dystrophin complex may play a role in AQP4 assembly on the plasma membrane of various cell types. Another clue to the function of perivascular AQP4 was given by platelet-derived growth factor B (PDGF-B)-knockout mice, which showed abnormal vascular morphogenesis resulting in the absence of pericytes, and the presence of endothelial hyperplasia (Hellstrom et al., 2001). These knockout mice showed an increase in AQP4 concentration and swelling of astrocytes (Hellstrom et al., 2001; Neely et al., 2001; Yokota et al., 2000). Studies on primary astrocyte cultures demonstrating that volume changes in response to osmotic gradients were mediated by AQP4 water channels (Nicchia et al., 2000) suggest that astrocyte swelling and modification of AQP4 expression are likely to be associated in the two mice strains. In mdx mice, astrocyte swelling may be due to impaired water elimination resulting from AQP4-disturbed organization on plasma membrane. In PDGF-B–knockout mice, AQP4 overexpression may be a response to astrocyte swelling resulting from vascular deficiencies. The high AQP4 content of the perivascular space highlights the need for rapid water movements in this region. Aquaporin 4 on astrocyte end-feet, together with the absence of AQP1 on the endothelium (see Aquaporin 1: Implication in Cerebrospinal Fluid Formation), may make water movements from the extracellular space easier with astrocytes than with the blood compartment through the BBB. The pattern of distribution of aquaporins within the perivascular space might be related to the control of the perivascular volume, a function that may be crucial for maintenance of cerebral blood perfusion (Badaut et al., 2000a).
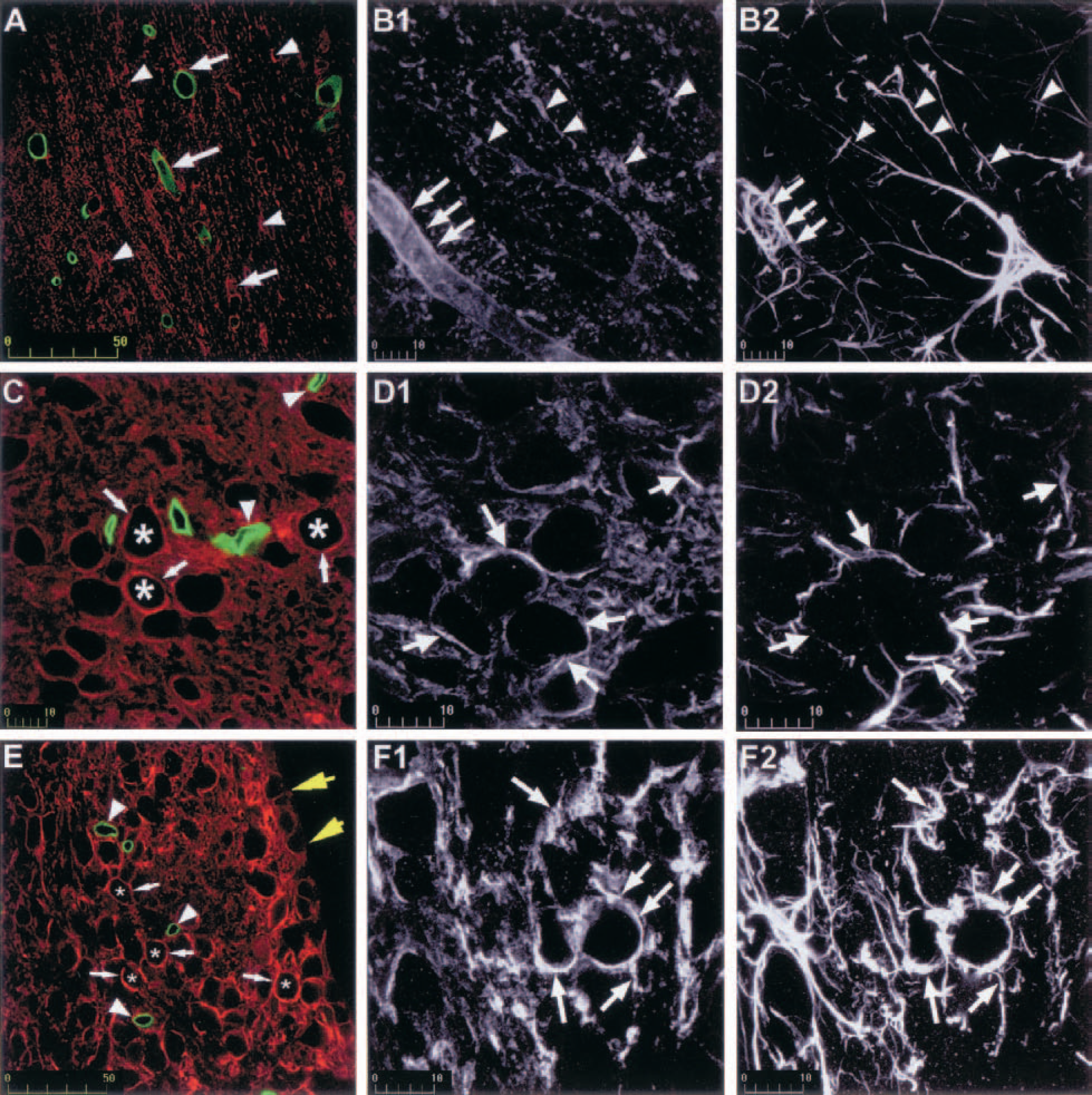
In the rat, several brain regions show a strong immunoreactive signal to AQP4 (Badaut et al., 2000a; Venero et al., 1999, 2001) that may be due to the presence of the channel out of the perivascular space on astrocyte processes apposed to neuronal cells (Badaut et al., 2000a; Nielsen et al., 1997; Venero et al., 2001). We performed a systematic study throughout the brain that revealed the presence of AQP4 on astrocyte processes in glia limitans (Badaut et al., 2000b) (Fig. 2A1). It was also seen in all white matter tracts and in various brain regions: olfactory areas in the cortex, medial habenular nucleus, the bed nucleus of stria terminalis, tenia tecta, pontine nucleus, CA1/CA3 layers of the hippocampus, cortical cerebellum, and several thalamic and hypothalamic areas. A recent in situ hybridization study performed in the rat brain showed mRNA staining in most of the same regions (Jung et al., 1994; Venero et al., 1999, 2001; Vizuete et al., 1999). However, in contrast to immunologic studies that did not report neuronal staining (Figs. 2I, 3C, 3D1, 3D2, 3E, 3F1, and 3F2), AQP4 mRNA was detected in CA1-CA3 hippocampal pyramidal cells, granular cells of dentate gyrus, and neuronal cells of the cortex (Venero et al., 1999). In addition, the semicircular arrangement of AQP4-mRNA in the bed nucleus of stria terminalis and medial habenular nucleus, suggested expression in neuronal cells (Venero et al., 1999, 2001). By double immunolabeling, we observed a systematic colocalization of AQP4 with glial fibrillary acidic protein in all AQP4-immunoreactive regions, including the parietal cortex (Fig. 2C), the corpus callosum (Figs. 3B1 and 3B2), the medial habenular nucleus (Figs. 3F1 and 3F2), and the suprachiasmatic nucleus (Figs. 3D1 and 3D2). The absence of staining on neurons containing AQP4 mRNA might be due to low protein density, not detectable by immunohistochemical methods.
The AQP4 distribution within the brain shows striking similarities with the distribution previously described for connexin-43 (Yamamoto and Sasaki, 1998) and connexin-43 mRNA (Micevych and Abelson, 1991). High concentrations of the two proteins were observed in brain regions such as piriform cortex, magnocellular nuclei of the hypothalamus, habenular nucleus, and the Purkinje cell layer of the cerebellum. In white matter, both AQP4 and connexin-43 were organized as puncta, parallel to myelinated fibers (Badaut et al., 2000a; Nagy et al., 1997). Because connexin-43 is the major component of the astrocyte gap junction, brain regions highly enriched with this protein may be endowed with a powerful K+ spatial buffering (Orkand et al., 1966). In these regions AQP4 may help astrocytes to take up excess K+ by allowing the resulting water transfer to be rapid. Astrocytes might take up K+ through various transporters and inwardly rectifying K+ (Kirs) channels (Newman, 1995). In rat retinal Müller cells, the distribution of Kir4.1 was described as following the pattern of AQP4 distribution along different plasma membrane domains (Nagelhus et al., 1999). The study by Yakushigawa et al. (1998) showed that Kir4.1 was concentrated in astrocytes around blood vessels in the optic nerve of the Japanese macaque (Macaca fuscata) (Yakushigawa et al., 1998). However, a recent study of Kir4.1 distribution in the adult rat CNS showed a poor correlation with our own study on AQP4 repartition (Poopalasundaram et al., 2000). Kir4.1 is therefore unlikely to be systematically associated with AQP4 in the uptake of K+ in astrocytes, suggesting the contribution of other systems that remain to be established. The comparative study of AQP4 expression among different brain regions showed that all these regions were characterized by very compacted neuronal cell bodies. The concentration of AQP4 may be related to the size of the extracellular space. In agreement with this hypothesis, the CA1 stratum pyramidale, which shows a high AQP4 labeling, exhibits a very low extracellular volume fraction, smaller than that observed in the cortex, where AQP4 was not seen around pyramidal cells (McBain et al., 1990; Perez-Pinzon et al., 1995). During neuronal excitation in physiologic conditions cell volume changes can be observed in hippocampal slices (Andrew and MacVicar, 1994). A low extracellular space may result in high K+ concentrations, particularly in pathologic conditions like ischemia (Perez-Pinzon et al., 1995). In view of its water permeability, AQP4 may play a role in these regions in maintaining low K+ concentration in the extracellular space. The presence of AQP4 may also be due to the sensitivity of these regions towards an expansion of the extracellular space. In accord with this hypothesis, a hyperosmotic increase in the extracellular volume was shown to greatly reduce the propensity of the CA1 stratum pyramidale to develop seizures (Traynelis and Dingledine, 1989). The susceptibility to physical uncoupling in regions with compacted cell bodies might be explained by the abundance of intercellular junctions. For example, a higher concentration of neuroligin 1, which forms intercellular junctions between neurones, has been shown in the CA1 layer of the hippocampus (Song et al., 1999). The presence of AQP4 may help maintain intercellular junctions by rapidly clearing excess water resulting from alterations in ion concentrations in the extracellular space. The same may be true in white matter tracts, where perinodal astrocytes contribute to the maintenance of intercellular junctions at the nodes of Ranvier by preventing activity-related swelling of the extracellular space.
As shown by in vitro studies on astrocyte cultures, AQP4 mediates volume changes (Nicchia et al., 2000). This characteristic gives rise to a role for AQP4 in the control of extracellular homeostasis by astrocytes. When exposed to hypotonic conditions, the astrocytes swell, and subsequently return to near-normal volume by a process of regulatory volume decrease. Regulatory volume decrease is an active, calcium-dependent mechanism that is accomplished by extrusion of intracellular osmotically active solutes, such as taurine. The release of osmotically active solutes is accompanied by water and could involve AQP4 water channels.
Like astrocytes, ependymal cells are endowed with an AQP4-polarized labeling on the basal side of the plasma membrane (Figs. 2G1 and 2G2). However, tanycytes (a category of ependymal cells found along the third ventricle in the internal median eminence [ME]) do not express AQP4 (Figs. 2D, 2E, 2G1, and 2G2). The ME can be divided into external and internal areas (Figs. 2D and 2E). The internal ME is composed of a tract of neuronal axons that come from the supraoptic and paraventricular nuclei and project into the neurohypophysis; the external ME is a region of axon ending on capillaries of the portal circulation of the hypophysis. While the external part of ME is not stained by anti-AQP4, the internal part is highly labeled and the distribution is comparable to the distribution in white matter tracts (Figs. 2D and 2E; for functional interpretation, see previous Aquaporin 4 and Control of the Extracellular Space). The aforementioned description, based on light-microscopy observations, emphasizes AQP4 concentration near or close to water compartments like plasma and CSF. Despite differences, high water permeability may be a common feature of all ependymal cells, including tanycytes. Indeed, the study by Elkjaer et al. (2000) in the rat brain suggests a shift of aquaporin expression from AQP4 to AQP9 in tanycytes.
Aquaporin 9: Brain distribution and putative functions
The AQP9 gene was cloned in 1998 both in the rat and in the human (Ishibashi et al., 1998; Tsukaguchi et al., 1998, 1999). Although AQP9 mRNA was detected in astrocytes (Tsukaguchi et al., 1998), the presence of the protein in brain was only recently demonstrated by Elkjaer et al. (2000). In a recent study we have shown that AQP9 was expressed in mouse brain (Badaut et al., 2001). In this work, AQP9 immunolabeling was found on astrocyte processes in the periventricular region of parenchyma and in the glia limitans, bordering the subarachnoid space (Badaut et al., 2001) (Figs. 4A2, 4A3, 4B2, and 4B3). Aquaporin 4 is also present in these regions in the mouse, suggesting that AQP9 contributes with AQP4 to the facilitation of water movements between CSF and brain parenchyma. In addition, AQP4 and AQP9 labeling on astrocytes was found in white matter tracts (corpus callosum, anterior commissure, and optic chiasm), hippocampus (Fig. 4C), septum (Fig. 4B2 and 4B3), and several hypothalamic nuclei including magnocellular nuclei (Figs. 4D and 4E) (Badaut et al., 2001). Similarities of distribution pattern within the brain between AQP4 and AQP9 in mice and between AQP4 in rats and AQP9 in mice suggest that the two proteins mediate common functions, at least in rodents, and that they can act in synergy.
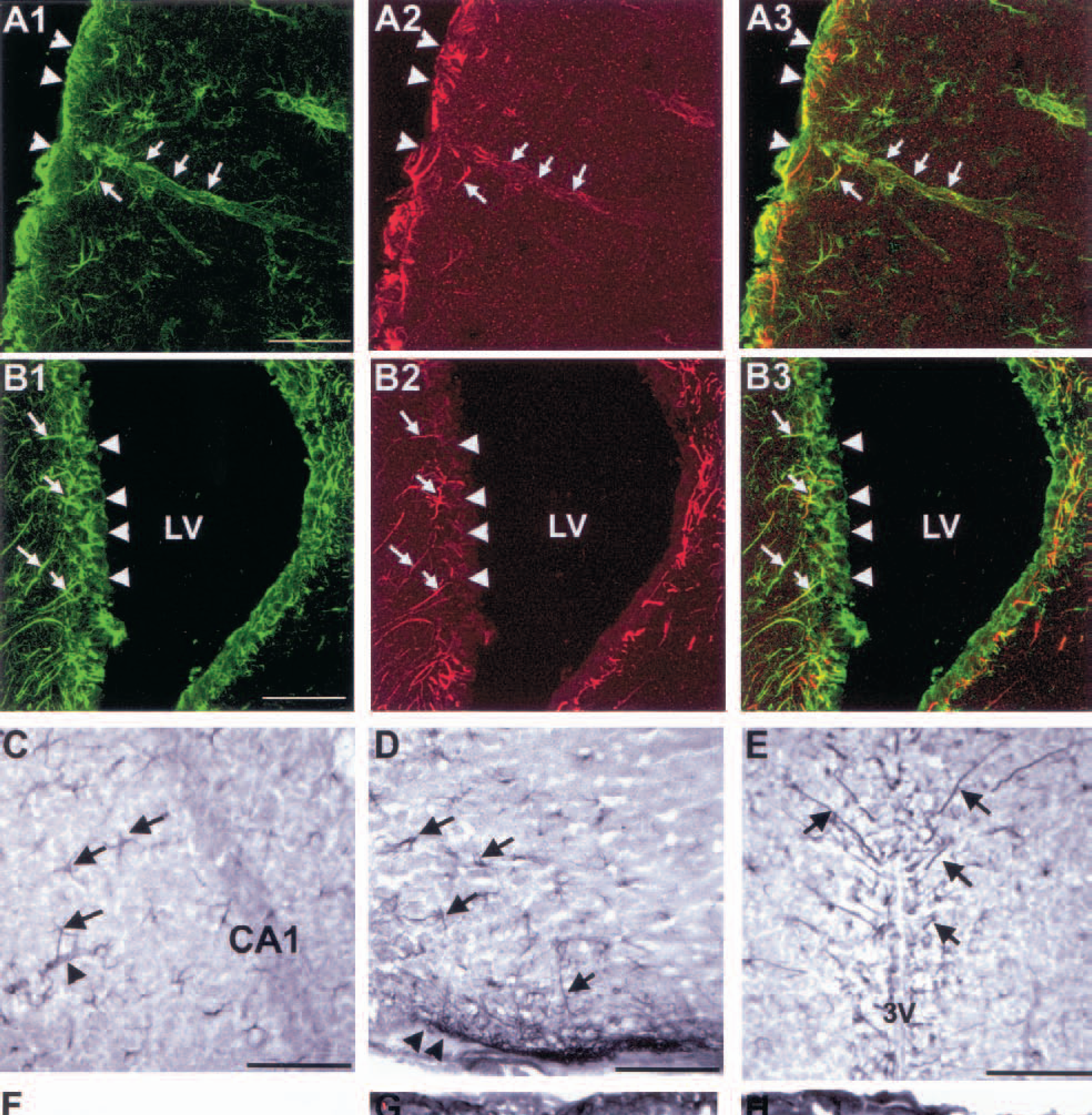
Confocal pictures of anti-GFAP (glial fibrillary acidic protein) and anti-AQP9 (aquaporin 9) in two mouse brain regions: parietal cortex (
Aquaporin 3, −5, and −8: Newly identified aquaporins in the brain
Recent results by reverse transcription–polymerase chain reaction showed expression of AQP3, AQP5, and AQP8 in neuronal primary cultures and astrocyte cultures. Whereas microglial cells did not express AQP in rat brain, AQP8 was observed on oligodendrocytes, and AQP5 on astrocytes (Yamamoto et al., 2001b). The physiologic role of these proteins remains to be determined.
AQUAPORINS IN BRAIN DISORDERS
Variation of aquaporin levels in brain disorders
As suggested by morphologic studies on aquaporins in the rodent brain, water channels may play a crucial role in extracellular homeostasis. A profound perturbation of brain environment may induce regional cerebral edema, which can be divided into two types: vasogenic and cytotoxic edema. The first is a consequence of BBB opening with extravasation of plasma autacoids into the extracellular space. Although the origin of cellular swelling in cytotoxic edema is unclear (for review see Kimelberg, 1995), it is attributed mainly to astrocytic swelling. Although soma and processes of astrocytes have been noted to swell, the most prominent swelling was observed in perivascular end-feet. After ischemia, the astrocytic uptake of osmolytes such as Na+, K+, Cl− and neurotransmitters from the extracellular space is followed by the concomitant passage of water into the intracellular space (Kimelberg, 1995). Therefore, water channels are likely to be involved in brain edema formation. Different studies have shown variations in aquaporin expression after brain injury; however, opposite directions were observed. Vizuete et al. (1999) have shown an increase of AQP4 mRNA in rat brain after mechanical trauma or intrastriatal injection of quinolinic acid. The authors concluded that the induction of AQP4 mRNA expression is related to disruption of the BBB. An increase in AQP4 mRNA was also described in response to permanent focal brain ischemia in the rat. In this model, the AQP4 mRNA response followed the decrease in edema observed by magnetic resonance imaging (Taniguchi et al., 2000). Time-dependent changes in AQP4 mRNA expression have been studied in different models of vasogenic and cytotoxic edema. After traumatic brain injury a decrease of AQP4 immunostaining was observed 24 hours later—specifically, in regions affected by BBB breakdown—whereas an increase appeared on the border of the insult 3 days after injury (Ke et al., 2001). The opposite effects observed in this study may be due, for one part, to transient vasogenic edema, and, for another, to long-term cytotoxic edema. The decrease of AQP4 after brain trauma may be linked to a possible protective effect against vasogenic edema formation (Ke et al., 2001), whereas the increase could be essential for cytotoxic edema resolution (Ke et al., 2001; Taniguchi et al., 2000). AQP4 deletion in knockout mice was shown to reduce brain edema after acute water intoxication (cytotoxic edema without BBB disruption) and after ischemic stroke produced by permanent middle cerebral artery occlusion (Manley et al., 2000). Interestingly, neurologic recovery was also improved in these AQP4-knockout mice (Manley et al., 2000). As previously indicated, AQP9, AQP5, AQP3, and AQP8 are also expressed in the rodent brain. Recent results showing variations in the expression after ischemia (Badaut et al., 2001; Yamamoto et al., 2001b) suggest that in AQP4-knockout mice, these alternative water channels can balance the absence of AQP4. Expression of all the aquaporins, therefore, should be considered in determining the contribution of AQP4 in edema formation and resolution.
We have reported an increase of the immunohistochemical signal to AQP9 on astrocytes bordering on the infarct after transient middle cerebral artery occlusion in mice (Badaut et al., 2001). The increased signal was observed on reactive astrocytes, mainly in the cortex, ventral pallidum, and nuclei of the amygdala. The specific permeability of AQP9 to lactate and water may be important under pathologic conditions such as brain ischemia. Interestingly, AQP9 permeability to lactate increases 4-fold when pH decreases to 5.5 (Tsukaguchi et al., 1998). Therefore, AQP9 could contribute to lactate clearing from extracellular space under pathologic conditions resulting in lactic acidosis, such as ischemia. Lactate movement into astrocytes is likely to be followed by water flux involving aquaporins. In addition to AQP4 and AQP9 responses, an induction of AQP5 expression was found near the ischemia-induced infarct border in the rat brain (Yamamoto et al., 2001b).
Astrocyte cultures were used to study the effects of hypoxia on AQP expression. Whereas AQP4, AQP5, and AQP9 expression was transiently decreased, AQP3 and AQP8 expression did not change. After restoration of oxygenation AQP4 and AQP9 returned to basal values and AQP5 was upregulated (Yamamoto et al., 2001b). It has been suggested that expression of aquaporins 4, −5 and −9 is regulated by the transcription factor AP-1 (Yamamoto et al., 2001b).
Whereas AQP3, AQP4, AQP5, and AQP9 were affected by brain injury, two recent reports disclosed overexpression of AQP1 in glioblastoma multiforme. A heterogeneous AQP1 immunolabeling was observed in tumor cell lines implanted in the rat brain (Endo et al., 1999), and comparison by differential gene expression of glioblastoma multiforme to normal brain tissue from patients with intractable epilepsy showed AQP1 overexpression. This difference was confirmed by immunohistochemistry (Markert et al., 2001). Endo et al. (1999) suggested that AQP1 overexpression could result from the high level of growth factors such as platelet-derived growth factor and fibroblast growth factor in the tumors. The significance of this abnormal upregulation remains unknown. The authors suggested that AQP1 might allow glioma cells to shrink by extruding water, thereby permitting invasion of the surrounding brain through the extracellular matrix (Endo et al., 1999). The expression of AQP1 in tumors may also reflect a role of this water channel in the development of edema due to changes in hydrostatic or oncotic pressure in the tumor.
Cellular and molecular regulation of aquaporin synthesis and activity in the brain
Most of the reports on regulation of AQP activity concern AQP2 in the kidney. In the collecting duct AQP2 translocates in response to vasopressin from cytoplasm vesicles to the apical membrane and induces an increase in water permeability (for review see Lee et al., 1997). Regulatory mechanisms of aquaporins have not yet been clearly identified in the brain.
A decrease of AQP4 and AQP9 mRNA and protein was observed in astrocyte cultures in response to stimulation of the protein kinase C pathway (Nakahama et al., 1999). As previously described, AQP5 level transiently decreases in hypoxia-induced astrocyte cultures (Yamamoto et al., 2001b); this protein, however, is not regulated by the protein kinase C pathway, suggesting that other regulatory mechanisms can influence its expression (Yamamoto et al., 2001a). The occurrence of a regulatory mechanism affecting AQP5 was also suggested by studies on parotid gland showing that AQP5 translocation was induced by activation of the M3 muscarinic-acetylcholine receptor via a cytosolic calcium pathway (Ishikawa et al., 1998). The presence of AQP5 and muscarinic receptors on astrocytes raises the possibility of a comparable regulatory system in the CNS (Badaut et al., 2000a). Furthermore, we have shown in the rat brain that AQP4 colocalized with muscarinic receptors on astrocytic processes and ependymal cells (Badaut et al., 2000a). Therefore, we suggest that muscarinic receptors play a role in the regulation of aquaporin activity.
In addition to molecular studies a recent work on cortical slices demonstrated that brain vasopressin through V1a-receptor activation induced water flux through the astrocytic syncytium. In addition to water flux V1a-receptor stimulation also induced activation of protein kinase C. Owing to the rapid time-course of the volume changes, the authors suggest that the effect is mediated by water channels. These molecular and physiologic data demonstrate that research on regulatory mechanisms may be useful for developing therapeutic targets in brain edema.
CONCLUSIONS
Appropriate regulation of water fluxes through membranes is a central requirement for all living organisms. The discovery of a family of water channels revealed that membrane proteins constitute an important pathway for water transport, which had long been considered to occur entirely by diffusion through plasma membranes. Data obtained during the past ten years support roles for aquaporins in CNS, both in physiologic and in pathophysiologic conditions such as brain edema. Brain swelling plays a critical role in the pathophysiology and morbidity of a wide variety of nervous system disorders (head trauma, tumors, ischemic and hemorrhagic stroke, infections, metabolic diseases). An increasing number of aquaporins have been identified in the CNS, but the functional role of the more recently discovered proteins remains to be established. The need for functional studies is essential because permeability properties of these aquaporins are specific and involve not only water but also different neutral solutes.
Footnotes
Acknowledgments:
The authors thank Elisabeth Waltisperger and Dr. Lorenz Hirt for their collaboration in this work.
