Abstract
Brain edema is frequently shown after cerebral ischemia. It is an expansion of brain volume because of increasing water content in brain. It causes to increase mortality after stroke. Agmatine, formed by the decarboxylation of L-arginine by arginine decarboxylase, has been shown to be neuroprotective in trauma and ischemia models. The purpose of this study was to investigate the effect of agmatine for brain edema in ischemic brain damage and to evaluate the expression of aquaporins (AQPs). Results showed that agmatine significantly reduced brain swelling volume 22 h after 2 h middle cerebral artery occlusion in mice. Water content in brain tissue was clearly decreased 24 h after ischemic injury by agmatine treatment. Blood–brain barrier (BBB) disruption was diminished with agmatine than without. The expressions of AQPs-1 and -9 were well correlated with brain edema as water channels, were significantly decreased by agmatine treatment. It can thus be suggested that agmatine could attenuate brain edema by limitting BBB disruption and blocking the accumulation of brain water content through lessening the expression of AQP-1 after cerebral ischemia.
Introduction
Agmatine, formed by the decarboxylation of L-arginine by arginine decarboxylase, was first discovered in 1910. It is hydrolyzed to putrescine and urea by agmatinase (Yang and Reis, 1999). Recently, agmatine, arginine decarboxylase, and agmatinase were found in mammalian brain (Li et al, 1994). Agmatine is an endogenous clonidine-displacing substance, an agonist for the two adrenergic and imidazoline receptors, and an antagonist at
Stroke is one of the leading causes of death in most of the developed countries with its incidence slowly increasing worldwide (Bonita, 1992; Chalela et al, 2004). Edema is frequently observed in ischemic stroke. Brain edema, defined as an abnormal increase in brain water content, which leads to an expansion of brain volume, has a crucial impact on morbidity and mortality after stroke, in that it increases intracranial pressure, favors herniations, and contributes to additional ischemic injuries (Klatzo, 1985). Aquaporins (AQPs) are a family of water channel proteins that facilitate the diffusion of water through the plasma membrane (Agre et al, 2002). In the rodent brain, three AQPs have been clearly identified, AQPs-1, -4, and -9 (Agre et al, 2004; Badaut et al, 2002). AQP-1 has been detected in epithelial cells of the choroid plexus (Nielsen et al, 1993), AQP-4 in astrocytes with a polarization on astrocyte endfeet (Nielsen et al, 1997), and AQP-9 in astrocytes of the white matter and in catecholaminergic neurons (Badaut et al, 2001, 2004). AQP-1 and AQP-4 are permeable only to water and are presumed to be involved in cerebrospinal fluid formation and brain water homeostasis (Amiry-Moghaddam and Ottersen, 2003). AQP-9 is an aquaglyceroporin, a subgroup of the AQP family, and is permeable to water and also glycerol, monocarboxylates, and urea (Badaut and Regli, 2004). These three channels may be implicated in water movements occurring during the formation and resolution of cerebral edema after ischemia.
On the basis of the above available reports, it was hypothesized that agmatine may have neuroprotective effect on brain edema. The purpose of this study was to investigate the protective effect of agmatine, if any, for brain edema in ischemic brain damage and also to investigate the expression of AQPs.
Materials and methods
Animals
ICR mice from Sam (Osan, Korea) were used for this study. All animal procedures were performed according to a protocol approved by the Yonsei University Animal Care and Use Committee in accordance with NIH guidelines.
Stroke Model
Male ICR mice weighing 36 ± 2 g were subjected to transient middle cerebral artery occlusion (MCAO,
Treatment
Agmatine was dissolved in normal saline (100 mg/kg IP, Sigma, St Louis, MO, USA) and administered after the suture was removed (agmatine,
Assessment of Brain Edema and Infarct Volume
Brain swelling and infarct volumes were determined by triphenyl tetrazolium chloride staining, using a computer-assisted image analysis system (Optimas ver 6.1, Optimas, Bothell, WA, USA), and corrected for the presence of edema using earlier published methods (Kim et al, 2004; Lee et al, 2001). The volume of infarct was expressed as a percentage of the total area of ipsilateral hemisphere.
Brain Water Content
Mice were killed 22 h after reperfusion, at the time point of maximal brain edema formation. Brains were removed. Hemispheres were separated and weighed to assess the wet weight (WW). Thereafter, the hemispheres were dried for 24 h at 110°C and the dry weight (DW) was determined (Groger et al, 2005). Hemispheric water content (%) was calculated using the following formula: ((WW–DW)/WW) × 100 (%).
Blood–Brain Barrier Disruption
The integrity of the blood–brain barrier (BBB) was investigated using Evans blue extravasation (Chan et al, 1991; Uyama et al, 1988). Evans blue dissolved at 2% in saline (100 μL) was injected in the tail vein and allowed to circulate for 90 mins. The chest wall was then opened under chloral hydrate anesthesia (400 mg/kg, intraperitoneally). Blood sample was obtained from the heart. Animals were perfused transcardially with saline until blue color was washed out from the effluent. Brains were removed immediately and were separated into cortex, hippocampus, striatum, and oligemia (the border area of cerebral infarct) with weights taken. They were homogenized in 500 μL of 50% trichloroacetic acid (weight/volume), and centrifuged (10,000 r.p.m., 20 mins). The supernatant obtained was measured at 445 nm using ELISA reader. Evans blue content of the plasma was similarly determined and the ratio of tissue to plasma Evans blue content was calculated as tissue Evans blue (μg/g wet weight)/plasma Evans blue (μg/g).
Immunohistochemical Staining for AQPs
Brains were fixed with 4% paraformaldehyde, and embedded in paraffin. Brain sections were microtomed into 10 μm. Sections were immunostained with antibodies against AQP-1 (Abcam, Cambridgeshire, UK), AQP-4, or AQP-9 (Chemicon, Temecula, CA, USA), followed by an appropriate fluorescein-conjugated secondary antibody for double-labeled fluorescent immunohistochemistry. Double-labeled immunostaining was evaluated using a fluorescence microscope (LSM 510 META, Carl Zeiss, Jena, Germany). Immunostained controls were also prepared simultaneously without primary antibodies. All incubation steps were performed in a humidified chamber.
Immunoblotting of AQPs
The expressions of AQPs-1, -4, and -9 proteins in ischemic injured brain were estimated by immunoblotting. Immunoblotting was performed using anti-AQPs and anti-actin (Santa Cruz, Santa Cruz, CA, USA) antibodies. Equal amounts of protein (100 μg) per condition were separated on an 8% polyacrylamide gel and electrotransferred onto Immobilon-NC membrane (Millipore, Bedford, MA, USA). Immunoreactive bands were visualized with the ECL detection system using Kodak X-AR film (Kim et al, 2004).
Statistical Analysis
Statistical tests to determine the differences between groups were performed with Student's
Results
Agmatine Reduced the Brain Edema and the Infarct Volume After Cerebral Ischemia
To investigate the effect of agmatine in ischemic damage, brain swelling volume was assessed in serial coronal sections of the mouse brain. The data were summarized in Figure 1. The average total brain swelling volume (cortical plus subcortical areas) in experimental control group was 117.11% ± 2.37% after 2 h MCAO and 22 h reperfusion. In agmatine treatment group, the average total brain swelling volume was 102.73% ± 0.16% after 2 h MCAO and 22 h reperfusion. Agmatine significantly reduced brain swelling volume (14.38% ± 2.21%,
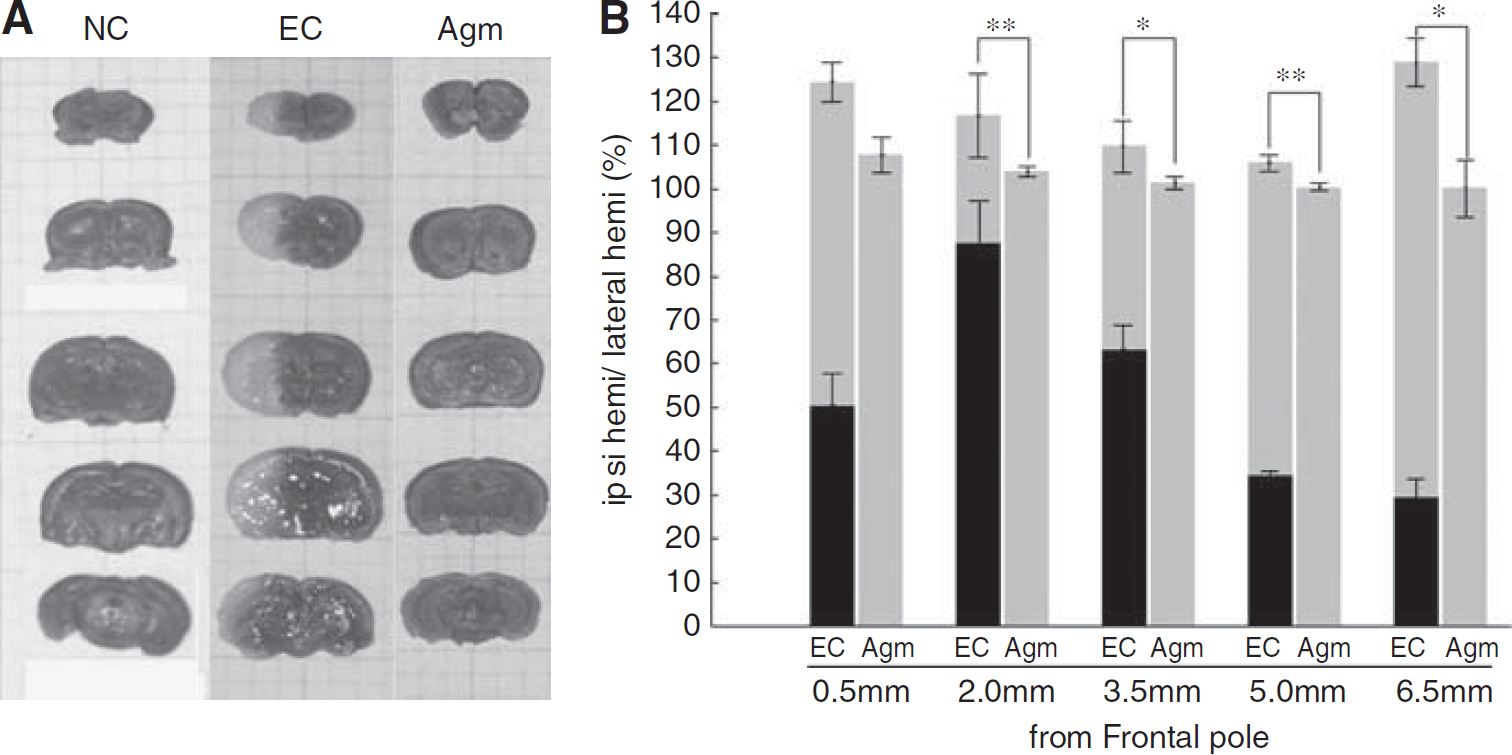
Brain edema and infarct volume on cerebral ischemia. Agmatine reduced infarct volume and brain swelling after ischemic injury. (
Agmatine Decreased the Water Content in Ischemic Injured Brain
The water content in ischemic injured mouse brains at 22 h reperfusion was shown in Figure 2. In normal control group, water content averaged 79.63% ± 1.10% in the ipsilateral hemispheres. Ischemia led to a significant increase in water content in the ipsilateral hemispheres (88.23% ± 0.59%; Figure 2). However, in agmatine treatment group, water content was significantly decreased in the ipsilateral hemispheres (80.61% ± 1.33%,
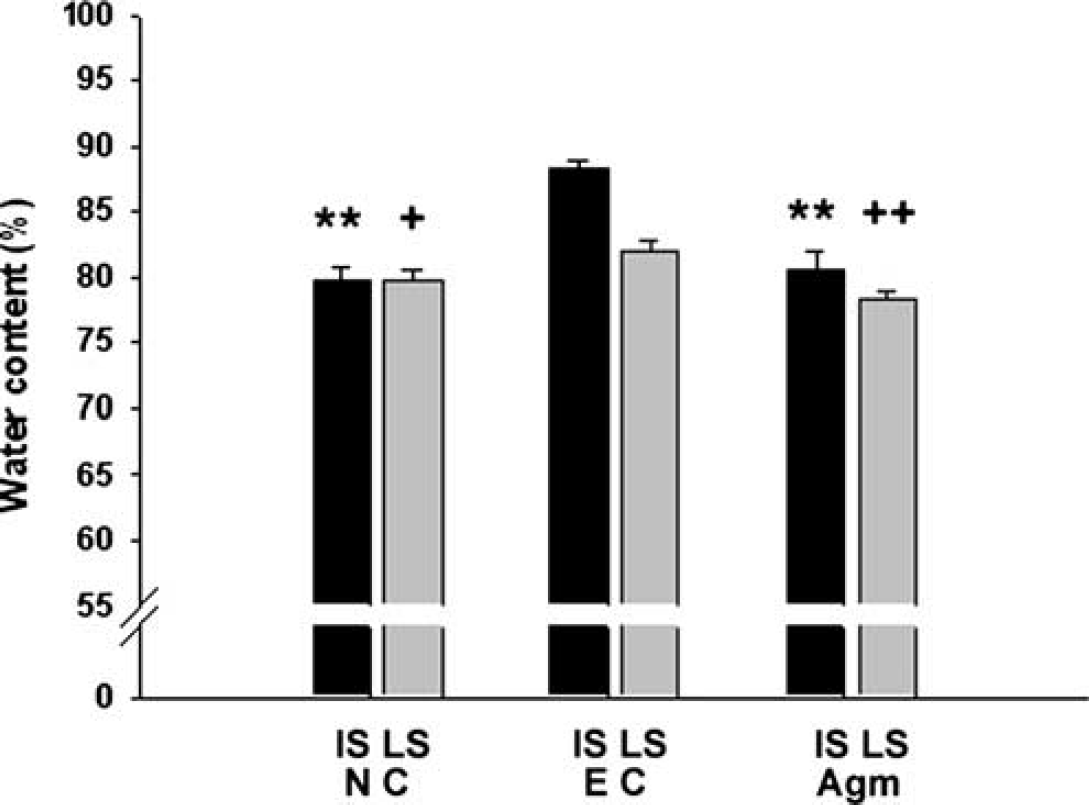
Brain water content on cerebral ischemia. Brain water content was analyzed as a measure of brain edema of the ischemic hemisphere 22 h after 2 h MCAO. Agmatine decreased the total water content of ischemic injured brain to normal level (**
Agmatine Limited BBB Disruption
It is needed to confirm whether the brain edema is vasogenic accompanied by BBB disruption or cytotoxic without BBB disruption. At 24 h after ischemic injury, Evans blue contents in the striatal, hippocampal, and cerebral cortical area of agmatine treatment group (2.74 ± 0.307; 3.08 ± 0.134; 1.14 ± 0.042) were significantly less than that of experimental control group (4.90 ± 0.120 in striatum; 4.24 ± 0.135 in hippocampus; 1.96 ± 0.110 in cerebral cortex,
EB ratio (Evans blue (μg/g wet weight)/plasma Evans blue (μg/g))
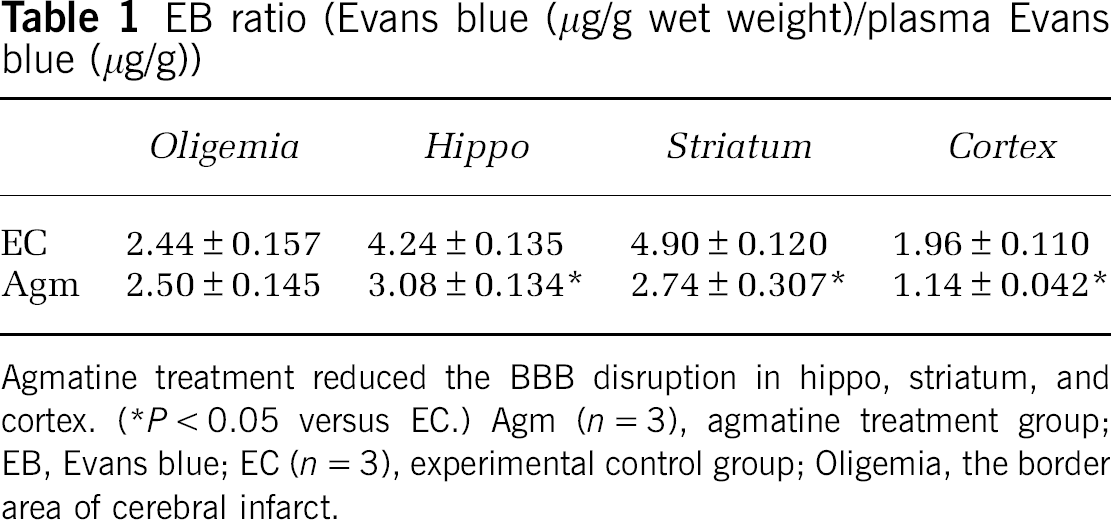
Agmatine treatment reduced the BBB disruption in hippo, striatum, and cortex. (*
Agmatine Lessened the Expression of AQPs 22 h After 2 h of MCAO
It was well known that AQP-1 was expressed only in epithelial cells of choroid plexus under normal condition, but AQP-1 was strongly expressed in endothelial cells 22 h after MCAO without agmatine. AQP-1-positive endothelial cells were shown in cortex (Figure 3A) and striatum (Figure 3B) except oligemia (data not shown). The expression of AQP-1 was reduced in endothelial cells of cortex (Figure 3A) and striatum (Figure 3B) in agmatine treatment group. Agmatine treatment also decreased the expression of AQP-1 in blood vessels of the choroid plexus (Figure 3C). Immunoblotting results showed significantly less expression of AQP-1 in agmatine treatment group compared with experimental control at 22 h after 2 h MCAO (
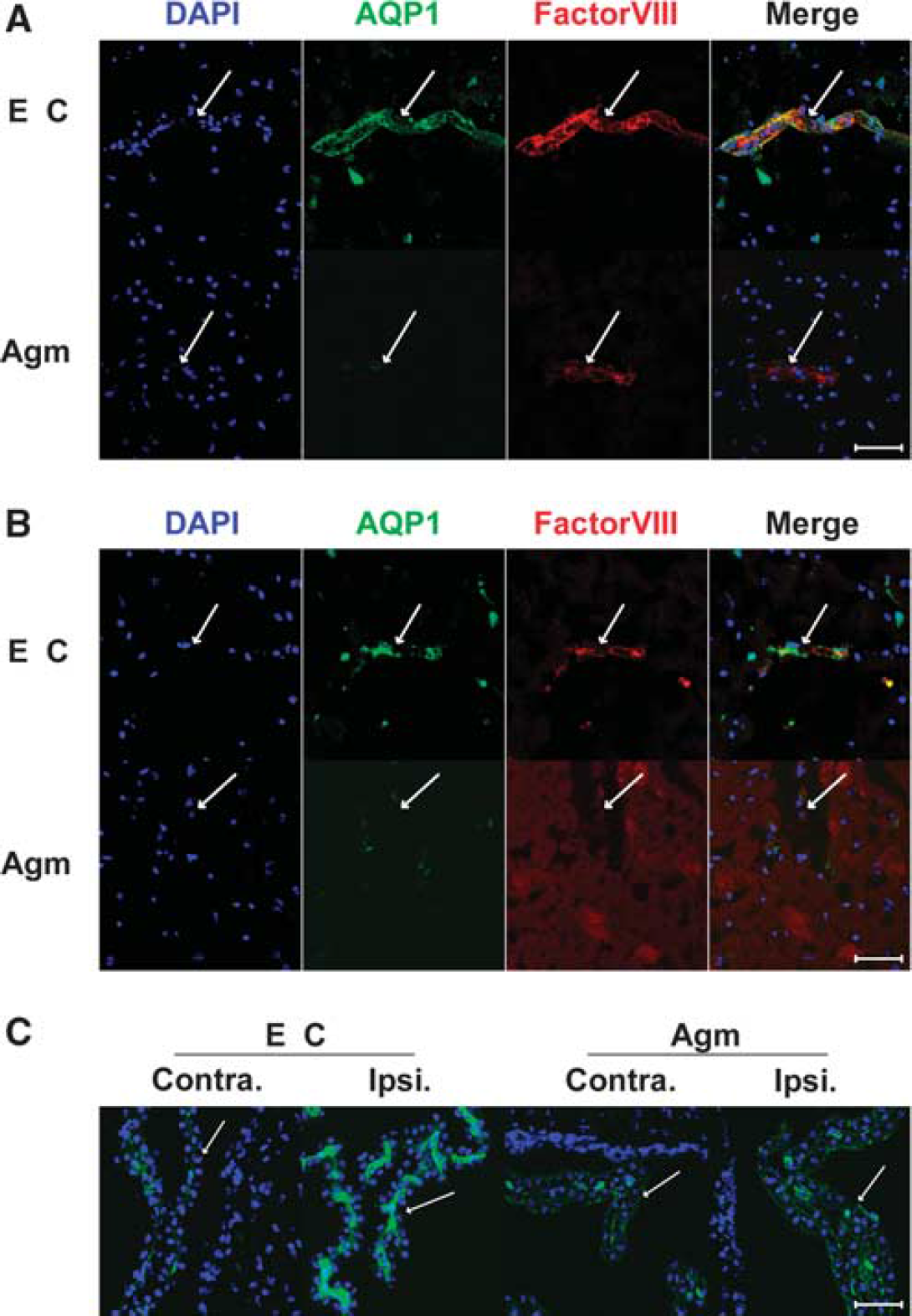
Macrographs of AQP-1 immunofluorescence in the ischemic injured brain 22 h after 2 h MCAO with or without agmatine. Blood vessel marked with factorVIII (red) was stained with AQP-1 (green) in cortex (
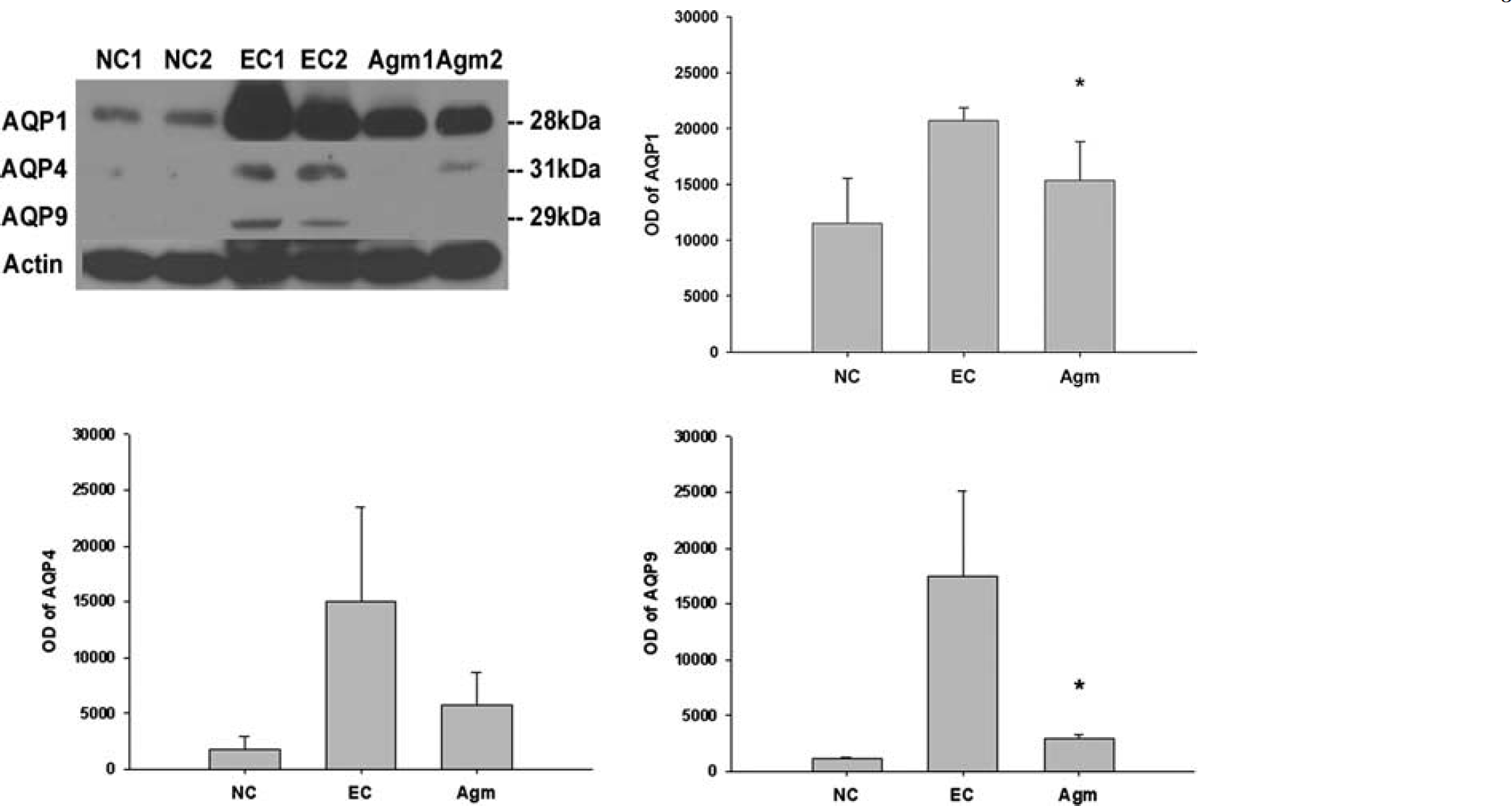
The expression of AQPs in the ischemic injured brain 22 h after 2 h MCAO with or without agmatine by immunoblotting. Optical densites (OD) of AQPs-1, -4, and -9 are expressed as the band density of each group. Data are expressed as mean ± s.d. Agmatine treatment significantly decreased the expression of AQPs-1 and -9 compared with experimental control (*
Discussion
It was shown that agmatine reduced brain swelling and brain edema in experimental stroke in this study. This effect of agmatine on the brain edema is associated with the decrease in AQP-1 expression and with the limitation of BBB disruption. It also appears that agmatine lessens the expression of AQP-1 in endothelial cells in cerebral ischemia.
There are two types of brain edema, cytotoxic edema and vasogenic edema after brain injury, such as brain trauma and stroke. Cytotoxic edema occurs without the BBB disruption, but vasogenic edema with the BBB disruption (The Korean Neurosurgical Society, 2005). The disruption of BBB was observed 22 h after 2 h MCAO in this study (Table 1). AQP-1 and AQP-4 are the two members of AQP family known to be expressed in the central nervous system, and it is possible that these proteins contribute to water transport across the BBB (Dolman et al, 2005). In a study of AQP-1 expression in human brain, a small number of microvessels were positively stained, but they were markedly upregulated in endothelium in astrocytomas and metastatic carcinomas (Verkman, 2002). Function of BBB is known to be impaired in such brain tumors, leading to formation of edema (Saadoun et al, 2002). Downregulation of the tight-junction proteins claudin and occludin has also been demonstrated in microvessels in glioblastoma multiforme (Liebner et al, 2000). Thus, loss of BBB function and the expression of AQP-1 may both be regarded as downregulation of BBB phenotype (Dolman et al, 2005). BBB disruption was reduced with agmatine treatment compared with experimental control. Earlier we had reported that agmatine decreased the expression of matrix metalloproteinase-2 and the expression of matrix metalloproteinase-9 in cerebral ischemia (Kim et al, 2008) and it was known that the early expression of matrix metalloproteinases-2 or -9 is associated with BBB disruption and the formation of vasogenic edema after transient focal cerebral ischemia (Fujimura et al, 1999; Gasche et al, 1999; Heo et al, 1999; Rosenberg et al, 1998). These reports can explain the possible mechanism of agmatine to limit BBB disruption in this study.
Our present investigation showed that AQP-1 was expressed in endothelial cells but not in endfoot of astrocytes surrounding capillary vessel in ischemic injured brain (Supplementary Figure 1). As of now, this might be the first report that AQP-1 is highly expressed in endothelial cells of choroid plexus in the case of vasogenic edema after cerebral ischemia to our knowledge. In normal rat brain, AQP-1 transcript and protein expression are restricted to the ventricular facing surface of choroid plexus and AQP-1 is related to cerebrospinal fluid secretion (Hasegawa et al, 1993; Masseguin et al, 2000). The expression of AQP-1 in the ventricular facing surface of choroid plexus was reduced in contralateral side but was not reduced in ipsilateral side of ischemic injured hemisphere compared with normal brain. This phenomenon may be because of the controlled cerebrospinal fluid secretion through dropping the expression of AQP-1 in the ventricular facing surface of choroid plexus to decrease the ascension of intracranial pressure caused by ipsilateral swelling. In agmatine treatment group, its expression in ipsilateral and contralateral side was similar to normal group, but the expression of AQP-1 was little increased in endothelial cells of choroid plexus in ipsilateral side compared with contralateral side of ischemic injured hemisphere. On the basis of these results, agmatine treatment decreased the expression of AQP-1 in endothelial cells of whole injured brain tissue, such as cortex, striatum, and choroid plexus.
The expression of AQP-4 mRNA is strongly induced when BBB is preserved. However, the expression of AQP-4 mRNA is reduced in astrocytes when BBB is disrupted after brain trauma (Kim et al, 2006). It explains that the expression of AQP-4 is much less than that of AQP-1 in the brain tissue after MCAO injury in this study (Figure 4 and Supplementary Figure 2). The expression of AQP-4 was not significantly changed but a reducing trend was obtained in agmatine treatment group than in experimental control group. Gunnarson et al (2008) reported that AQP-4 phosphorylation is related to nitric oxide in astrocyte. It is possible that agmatine would suppress the expression of AQP-4 in astrocyte by inhibiting nitric oxide synthesis (Auguet et al, 1995; Galea et al, 1996).
It was published that AQP-9 protein is upregulated on reactive astrocytes in the border of the infarct after transient MCAO in mice (Badaut et al, 2001) and AQP-9 showed a significant induction at 24 h, with expression increasing gradually with time, without correlation to swelling (Ribeiro et al, 2006). These reports suggest that the decreased expression of AQP-9 in agmatine treatment group is not directly related to the reduced brain edema after cerebral ischemia by agmatine treatment.
In conclusion, agmatine attenuates brain edema by limiting BBB disruption and blocking the accumulation of brain water content through lessening the expression of AQP-1 in endothelial cells in cortex, striatum, and choroid plexus after cerebral ischemia, suggesting that agmatine may have therapeutic potential in treatment of brain edema after brain injury, such as stroke and brain trauma.
Footnotes
Acknowledgements
This work was supported by the Korea Science and Engineering Foundation (KOSEF) grant funded by the Korea government (MOST) (R01-2007-000-10357-0).
The authors declare no conflict of interest.
