Abstract
It is not known whether acute brain injury results in a systemic acute phase response (APR) or whether an APR influences outcome after an insult to the CNS. The present study sought to establish whether brain injury elicits a systemic or local APR. The expression of acute phase protein (APP) mRNA in liver and brain tissues was measured by Taqman reverse transcriptase-polymerase chain reaction after an excitotoxic lesion in the striatum or challenge with a proinflammatory cytokine. N-methyl-D-aspartate (NMDA)–induced brain lesion did not elicit a systemic APR. In contrast, proinflammatory challenge with mouse recombinant interleukin-1β (mrIL-1β) resulted in a significant hepatic APP mRNA expression within 6 hours. Thus, an inflammatory challenge that results in a meningitis leads to a hepatic APR, whereas acute brain injury alone, with no evidence of a meningitis, does not produce an APR. This is surprising because NMDA leads to an increase in endogenous IL-1β synthesis. This suggests that the brain has an endogenous antiinflammatory mechanism, which protects against the spread of inflammation after an acute injury. In the brain, both excitotoxic lesions and proinflammatory challenge resulted in a profound parenchymal upregulation of APP mRNA after 6 and 12 hours in the injected hemisphere. These results suggest that the local APR may play a role as an antiinflammatory mechanism. These findings indicate a potentially pivotal role for peripheral and local APP production on outcome after brain injury.
Local inflammation, resulting from injury or infection, may be accompanied by systemic responses, including leukocytosis, fever and changes in serum levels of electrolytes, metal ions, clotting agents, complement, and glucocorticoids. In addition, there may be profound changes in serum titers of liver-derived acute phase proteins (APPs). These changes are referred to as the acute phase response (APR) (Baumann and Gauldie, 1994; Steel and Whitehead, 1993). Hepatic APPs are involved in protease inhibition, opsonization of pathogens and debris, neutrophil activation, metal ion transport, clotting and tissue repair (Steel and Whitehead, 1993). Synthesis of complement proteins, particularly C3, is also elevated in the liver during an APR. These proteins have cytolytic, opsonic and proinflammatory functions (Bickerstaff et al., 1999; Robey et al., 1984). Although the APPs are structurally and functionally diverse, their concerted effects mediate a return to local homeostasis by assisting in the removal of the inflammatory stimuli, attenuating local inflammation, and promoting tissue repair and regeneration.
The hepatic synthesis of APPs is orchestrated by a range of inflammatory mediators, including cytokines, glucocorticoids, and anaphylotoxins, in a complex inter-endocrine fashion (Jensen and Whitehead, 1998b; Koj, 1998; Steel and Whitehead, 1993; Szalai et al., 2000). However, the proinflammatory cytokines interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) have been shown to play a central role in APR initiation (Jensen and Whitehead, 1998b; Koj, 1996; Kushner, 1993).
After an acute brain insult, such as a stroke or traumatic brain injury, local inflammation in the brain parenchyma can exacerbate neuronal loss as a result of bystander damage. Infiltrating macrophages release proinflammatory cytokines (Feuerstein et al., 1997), including IL-1β, that have been shown to contribute to neuronal death (Lawrence et al., 1998; Relton and Rothwell, 1992). The respiratory burst of neutrophils may also result in indiscriminate damage to surrounding tissues, and it has been shown in animal models of brain injury that neutrophil depletion has beneficial affects (Clark et al., 1996). Recently, tissue plasminogen activator has been implicated in glutamate receptor–mediated excitotoxic cell death via its lytic effects on the extracellular matrix (Tsirka et al., 1996). Moreover, components of the complement cascade are thought to be deleterious in chronic neurologic conditions such as multiple sclerosis (Morgan and Gasque, 1996).
Considering the detrimental effects that inflammatory responses may have on brain tissue and, in light of the role that the APR plays in peripheral inflammation, the contribution of the APR to brain injury has received scant attention. It is well documented that acute brain injury results in the production of inflammatory cytokines, including IL-1, IL-6, and TNFα (Taupin et al., 1993). Studies of patients with head trauma have shown increased serum and CSF levels of proinflammatory cytokines, including IL-1 and IL-6 (Kossmann et al., 1995,1996; McClain et al., 1991). These changes in cytokine expression are strongly associated with increased hepatic expression of APPs, including CRP and α1-antitrypsin (Kossmann et al., 1995). However, from these studies it is impossible to disassociate the effects of accompanying peripheral tissue damage from the brain insult. It remains unknown whether brain injury alone leads to an APR and what influence, if any, an APR has on outcome.
The present investigation seeks to establish whether a brain injury, in the absence of significant incidental tissue trauma, results in either a systemic or local APR. The use of an animal model has permitted precise focal lesions to be induced within the brain parenchyma, with minimal incidental trauma, and provides a route to investigate whether brain injury per se is associated with an APR.
MATERIALS AND METHODS
Experimental animals
Female C57/BL6 mice (15 to 20 g) were obtained from Harlan-Olac (Bicester, U.K.), housed under standard conditions and fed with pelleted food and water ad libitum. All animal experiments were performed under Home Office Licence (U.K.).
Stereotaxic injections
Animals were anesthetized under isoflurane (1.5% in 100% O2), held in a stereotaxic frame and injected with 1 ng of mouse recombinant interleukin-1β (mrIL-1β; R&D Systems, Abingdon, U.K.), or 60 nmol of N-methyl-
Tissue preparation
At 6 or 12 hours after surgery, animals were deeply anesthetized with sodium pentabarbitone and liver samples (approximately 2 mm3) were taken, immediately snap frozen in liquid nitrogen and stored at −80°C. The animals were then transcardially perfused with heparinized saline, and brain samples, containing the site of injection in the striatum, were quickly removed and snap frozen and stored as for liver.
RNA extraction
Total RNA was extracted using Qiagen RNeasy mini columns (Qiagen, Crawley, U.K.). Contaminating DNA was degraded with Qiagen Dnasel according to the manufacturer's instructions. Yields were determined spectraphotometrically at λ 260 nm. Typical RNA yields per 10 mg of tissue were 40 μg for liver, 8 μg for brain.
Taqman reverse transcription polymerase chain reaction assay
All equipment and reagents were supplied by Applied Biosystems (Warrington, U.K.) unless otherwise stated. Taqman PCR primers and probes were designed using “Primer Express” software according to published sequences for murine serum amyloid A protein isoform 2 (SAA2), (Lowell et al., 1986) serum amyloid P component (SAP) (Whitehead and Rits, 1989), complement protein component C3 (Wiebauer et al., 1982) and the serine protease inhibitor, α2-antiplasmin (α2-AP) (Menoud et al., 1996). Primers and probes for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were supplied in kit form. Primer and probe sequences for APP genes are listed in Table 1. Probes were labeled at the 5′ end with a 6′-carboxyfluorescein reporter dye and at the 3′ end with a 6′-carboxy-tetramethyl rhodamine quencher dye. As an additional control measure, where possible, probes were designed to span an intron–exon boundary in order that they were specific for complementary DNA (cDNA) and did not bind genomic DNA.
Mouse acute phase protein Taqman primer and probe sequences

SAA, serum amyloid A protein; SAP, serum amyloid P component; C3, protein complement C3; α2-AP, α2-antiplasmin.
Primer pairs were tested using conventional reverse transcription polymerase chain reaction (RT-PCR) followed by gel electrophoresis to show single bands of the expected size. Amplified products were confirmed by direct sequencing of the PCR reaction (Big Dye) and diagnostic digestion with suitable restriction enzymes (results not shown).
For Taqman PCR, cDNA was synthesized using 200 ng total RNA in 10 μl reaction volumes using RT-Gold reagents according to the manufacturer's instructions.
Taqman PCR was performed with 0.5 μl to 1.0 μl (equivalent to 10 to 20 ng of RNA input) of cDNA template in a 25-μl PCR reaction containing 100-nM primers and 50-nM probe made in Universal Taqman Master mix. Cycling conditions were: 50°C for 2 minutes (for activation of AmpErase enzyme, contained in the master-mix, to remove any contaminating carry-over Taqman PCR products before the run), 95 °C for 10 minutes (to activate the Amplitaq Gold DNA polymerase) followed by a 40-cycle amplification phase of 95 °C for 15 seconds and 60°C for 1 minute on the Applied Biosystems PRISM 7700 Sequence Detection System. Samples were assayed in duplicate and quantified against a standard curve, generated as described below. The principles and methods for Taqman PCR have been described in detail elsewhere (Bustin, 2000).
Absolute RNA standards and RNA quantification
Single-species RNA templates were made in vitro. Standard PCR products, amplified using the gene-specific Taqman primer pairs, were gel-purified and recovered using Qiaquick Gel Extraction kits (Qiagen) and subsequently ligated to a T7 phage promoter sequence (Lig'n Scribe; Ambion, Huntingdon, U.K.). The ligated product was then amplified by conventional PCR using a gene-specific primer and a T7-specific primer. Amplified products were incubated at 37°C overnight in the presence of T7 RNA polymerase enzyme (MEGAshortscript Enzyme Mix; Ambion) according to the manufacturer's instructions. Transcripts were then gel-purified to remove unincorporated nucleotides, prematurely terminated products and enzymes on a 10% polyacrylamide gel containing 3 mol/L urea. Discrete, single bands of the expected size were excised and eluted in buffer containing 10 mmol/L magnesium acetate, 1 mmol/L ethylenediaminetetraacetic acid, 0.5 mol/L ammonium acetate, and 10% sodium dodecyl sulphate (pH 8.0) overnight at 37°C and ethanol precipitated. Eluted RNA was DNase1 treated (Promega, Southampton, U.K.) to remove template, quantified using a RiboGreen RNA quantification kit (Molecular Probes, Cambridge, U.K.), and diluted to give standards in the range 10 pg to 0.1 fg. These dilutions were expressed as log molecules, calculated according to Avogadro's number (i.e., 6.022 × 1023 molecules = 1 mole). The molecular weight for each transcript was calculated according to the formula: mW = (An × 328.2) + (Un × 305.2) + (Cn × 304.2) + (Gn × 344.2) + 159, where A, C, U, and G are nucleotides. RNA templates were subsequently used in Taqman RT-PCR. Thus, results are expressed as the number of copies per ng of total RNA, normalized to the measurement of the housekeeping gene GAPDH. Statistical analysis was performed using analysis of variance (ANOVA) with Fischer's post hoc test.
RESULTS
Excitotoxic brain lesion does not induce hepatic APP expression
The hepatic response to a focal excitotoxic lesion was assessed by the intraparenchymal injection of the glutamate receptor agonist NMDA and subsequent measurement of APP gene expression in the liver. The anesthetic, surgery, and intrastriatal injection of vehicle resulted in a marked systemic inflammatory response, compared with naïve mouse liver samples (not shown). There was no increase in the expression of any of the four APP genes measured 6 hours after NMDA injection when compared to the vehicle-injected controls. NMDA-injected mice did show a very slight reduction in α2-AP messenger RNA (mRNA) in the liver at 6 hours. Measurements taken 12 hours after NMDA injection remained at control levels (Fig. 1) and no difference was seen in α2-AP expression at this time-point. Acute neuronal degeneration and the associated acute inflammatory response therefore failed to evoke a systemic APR.
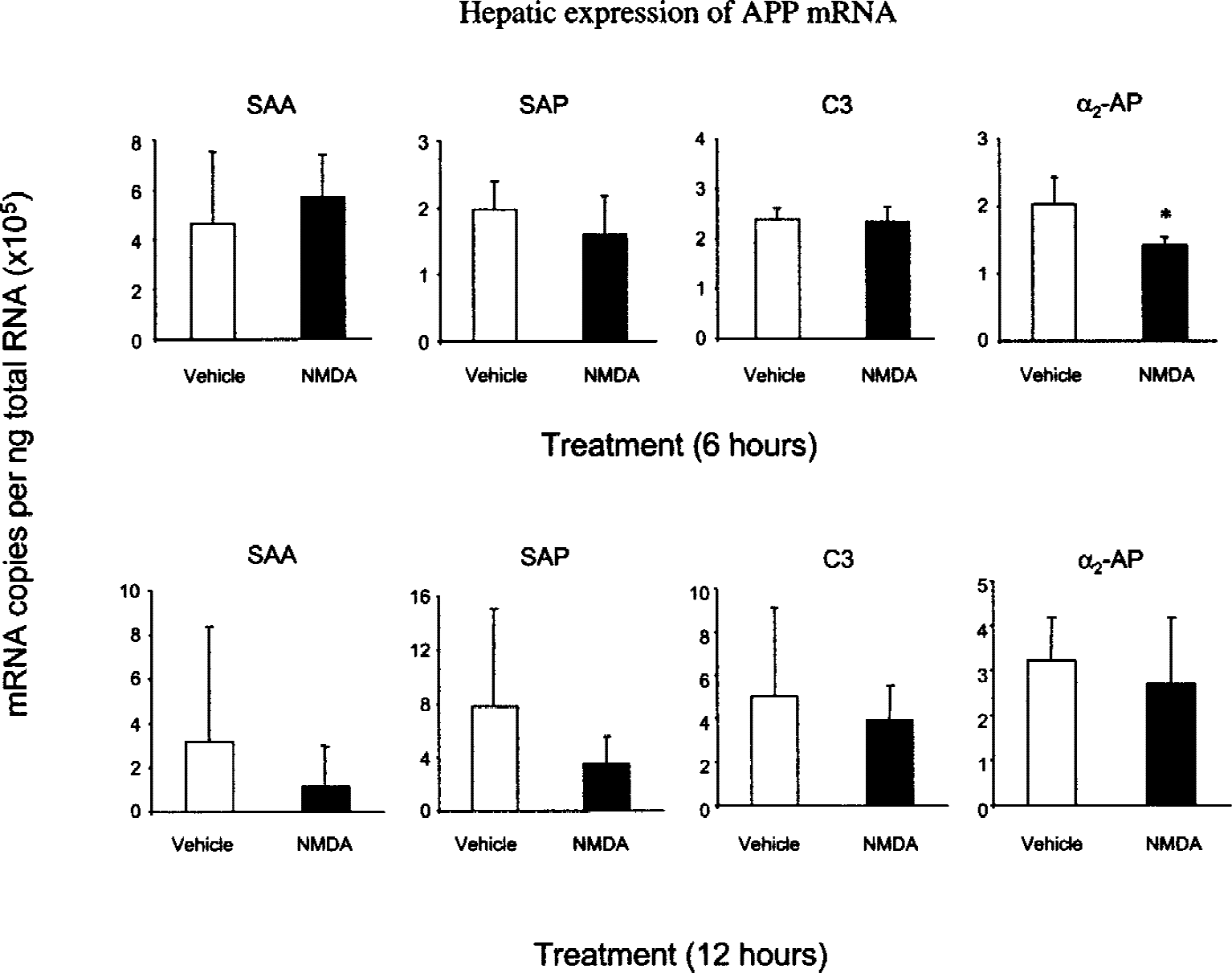
N-methyl-D-aspartate (NMDA)–mediated brain injury does not result in a peripheral acute phase response. Sixty nmol NMDA was injected into the striatum of mice and hepatic acute phase protein (APP) gene expression measured after 6 and 12 hours. No significant increases were seen between treatment and vehicle-injected control groups (ANOVA with Fischer's post hoc test). Reduced expression of α2- antiplasmin (α2-AP) mRNA was seen after 6 hours. Bars represent mean ± 1 SD (N = 4). *Significance value P < 0.05 (ANOVA with Fischer's post hoc test). SAA, serum amyloid A protein; SAP, serum amyloid P component; C3, protein complement C3.
Hepatic expression of APPs is evoked by intrastriatal injection of interleukin-1β
To investigate whether a proinflammatory challenge results in a systemic immune response, mice were intra-striatally injected with the proinflammatory cytokine mrIL-1β and changes in hepatic APP gene expression were measured.
At 6 hours after injection of mrIL-1β the expression of hepatic SAA mRNA was significantly increased (P < 0.001), approximately five-fold higher than vehicle-injected animals (Fig. 2). SAP expression was also elevated above vehicle control levels (P < 0.05) whereas C3 and α2-AP expression were not statistically different in the mrIL-1β–treated animals from controls. However, although the data for C3 and α2-AP did not achieve statistical significance, a trend towards increased mRNA expression at 6 hours after injection was evident.
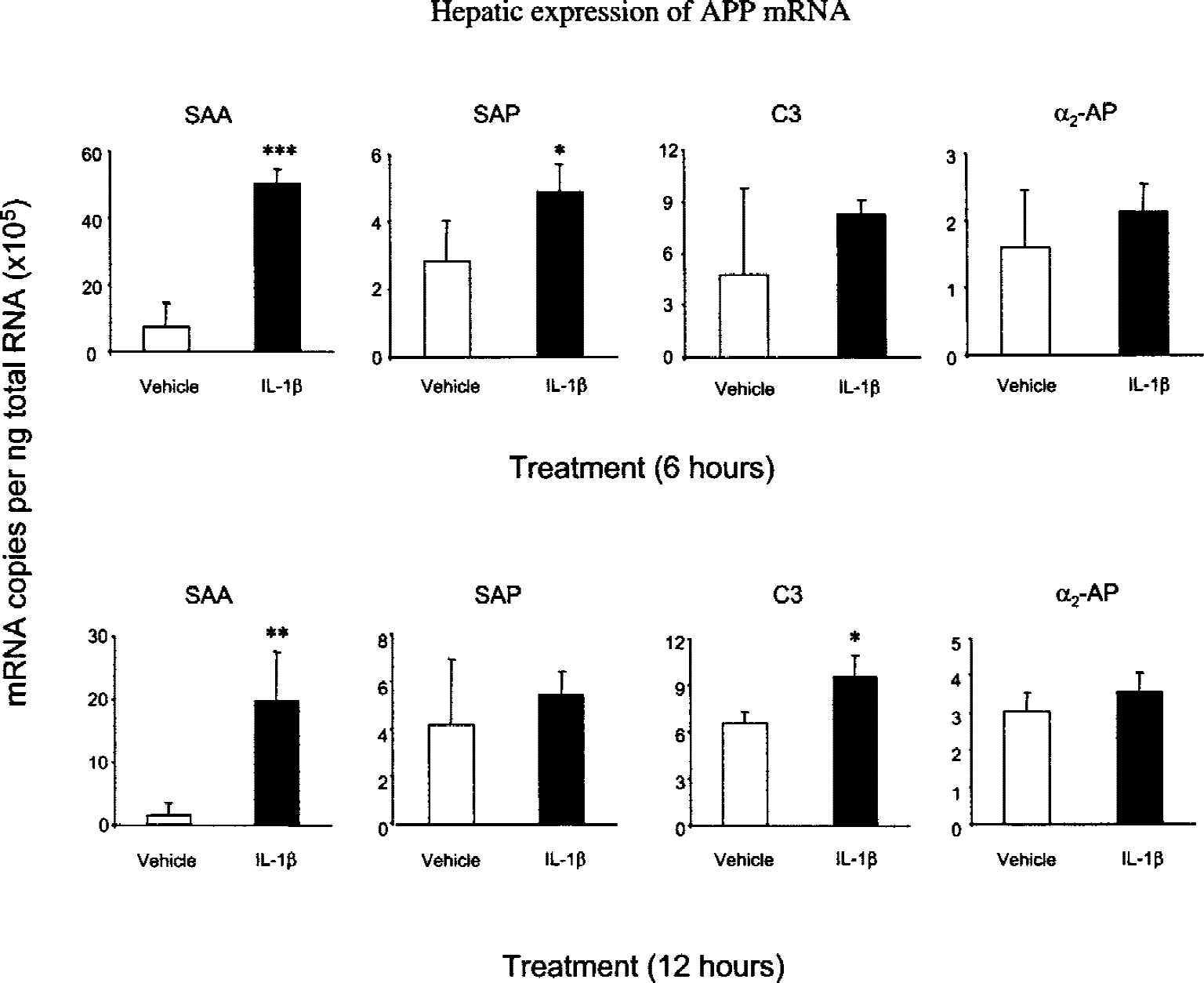
Intrastriatal proinflammatory challenge with mouse recombinant interleukin-1β (mrIL-1β) produces a profound systemic acute phase response, indicated by increased hepatic acute phase protein (APP) gene expression. One ng mrIL-1β was injected into the striatum and APP gene expression measured in liver samples taken at 6 hours and 12 hours. Transcripts for serum amyloid A protein (SAA) and serum amyloid P component (SAP), major APPs in the mouse, were found to be significantly increased after IL-1β challenge at 6 hours. After 12 hours, SAA remained elevated with respect to the vehicle controls. At this time-point, SAP mRNA levels in the vehicle controls were slightly increased and the difference between treatment and controls nonsignificant. Protein complement C3 showed moderate increases in expression at this time, whereas α2-antiplasmin (α2-AP) was not significantly different from vehicle controls. Bars show mean ± 1 SD. Significance values: ***P < 0.001; *P < 0.05 (ANOVA with Fischer's post hoc test; N = 4).
Hepatic APP mRNA for SAA measured 12 hours after intrastriatal injection of mrIL-1β showed a slight reduction in transcript numbers compared with those measured at 6 hours (Fig. 2). For SAA, both IL-1β–injected and control groups showed raised levels when compared to naïve animals, but reduced levels compared to 6 hours, although the difference between the vehicle and mrIL-1β groups was still significant (P < 0.01). At 12 hours, the number of SAP transcripts in the mrIL-1β treatment group was unchanged compared to those measured at 6 hours, whereas the vehicle control levels were slightly elevated and the difference between the treatment and controls nonsignificant. Transcripts for C3 and α2-AP were marginally increased in both treatment and controls, although, at this time point, only C3 showed statistically significant differences between control and treatment groups (P < 0.05).
Excitotoxic brain lesions evoke APR in the brain parenchyma
The expression of all mRNA species in the brains of naïve animals assayed approached the detection limits of the assay (results not shown). Transcripts for SAP were invariably undetected in brain samples. Measurements of vehicle-injected control samples were also extremely low (Fig. 3). After NMDA injection, SAA, C3, and α2- AP genes showed increased expression in the injected hemisphere compared with controls at 6 hours (SAA, P < 0.01; C3, P < 0.05; α2-AP, P < 0.05). Twelve hours after NMDA injection, APP mRNA levels remained above controls. However, at this time-point, increased variation in the mRNA expression of SAA and C3 genes rendered the differences statistically nonsignificant (Fig. 3). At this time-point, only α2-AP expression was significantly different to controls (P < 0.05). The uninjected hemispheres also showed elevated mRNA expression compared with the negligible levels found in naïve brains. Thus, although NMDA injection failed to induce a systemic APR the acute neurodegeneration did induce a local APR.
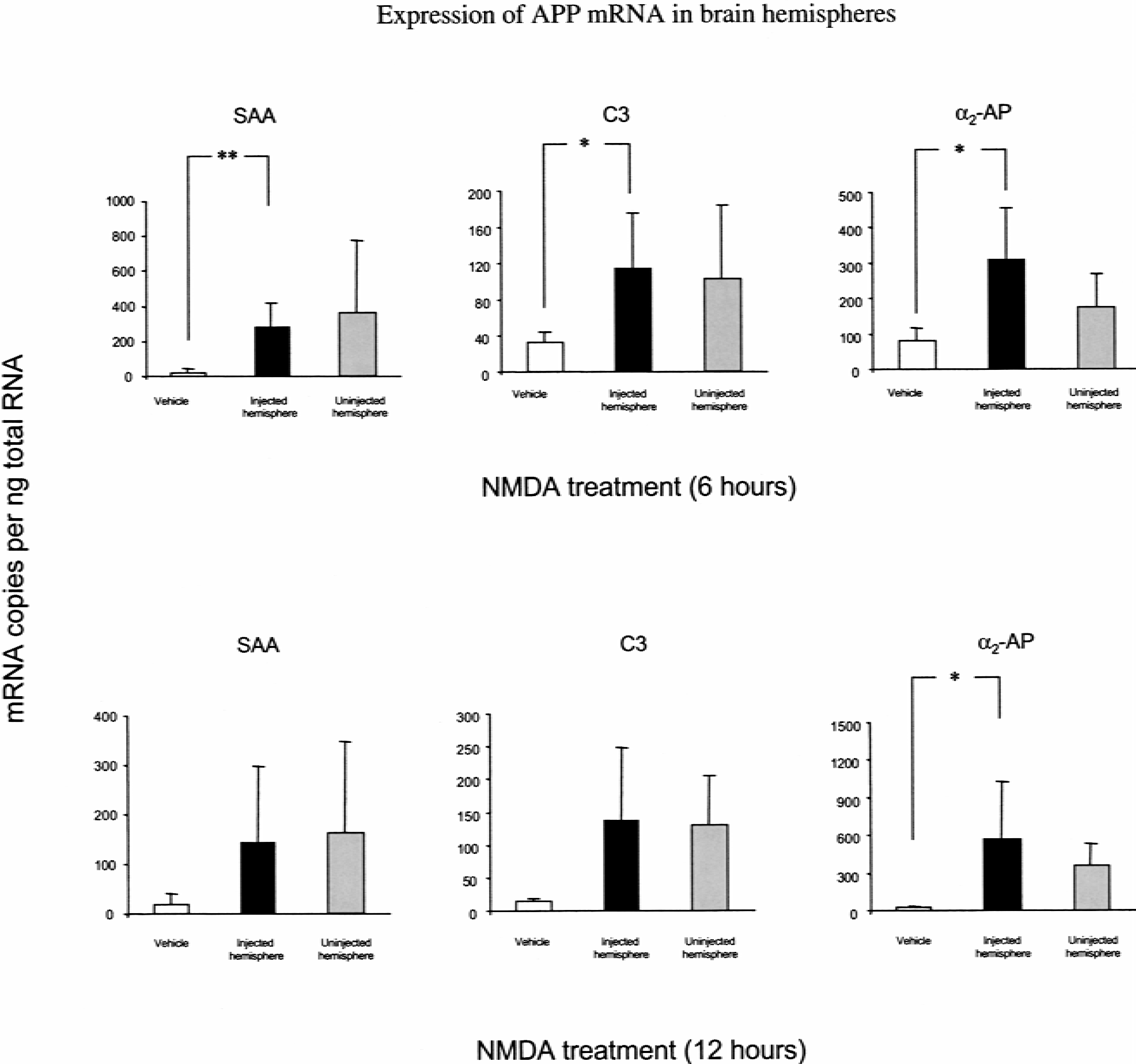
The brain expresses acute phase protein (APPs) after excitotoxic brain injury. Striatal samples from NMDA-injected mice were taken from the injected and uninjected hemispheres at 6 and 12 hours and APP gene expression measured. After 6 hours marked increases in serum amyloid A protein (SAA), protein complement C3, and α2-antiplasmin (α2-AP) copies were found, compared with vehicle controls. Serum amyloid P component (SAP) was never detected in any striatal samples throughout the investigation. After 12 hours, SAA mRNA levels were reduced, whereas C3 and α2-AP were marginally increased. Despite these changes, only α2-AP was statistically different from controls at 12 hours. The uninjected hemispheres also showed a trend towards increased gene expression for all APP at both time-points. Bars show mean ± 1 SD. Significance values: **P < 0.01; *P < 0.05 (ANOVA with Fischer's post hoc test; N = 4).
The brain expresses APP mRNA after interleukin-1β injection
Six hours after mrIL-1β injection there was a dramatic increase in SAA, (40-fold, P < 0.01), C3 (30-fold, P < 0.001), and α2-AP (P < 0.05) within the brain parenchyma (Fig. 4).
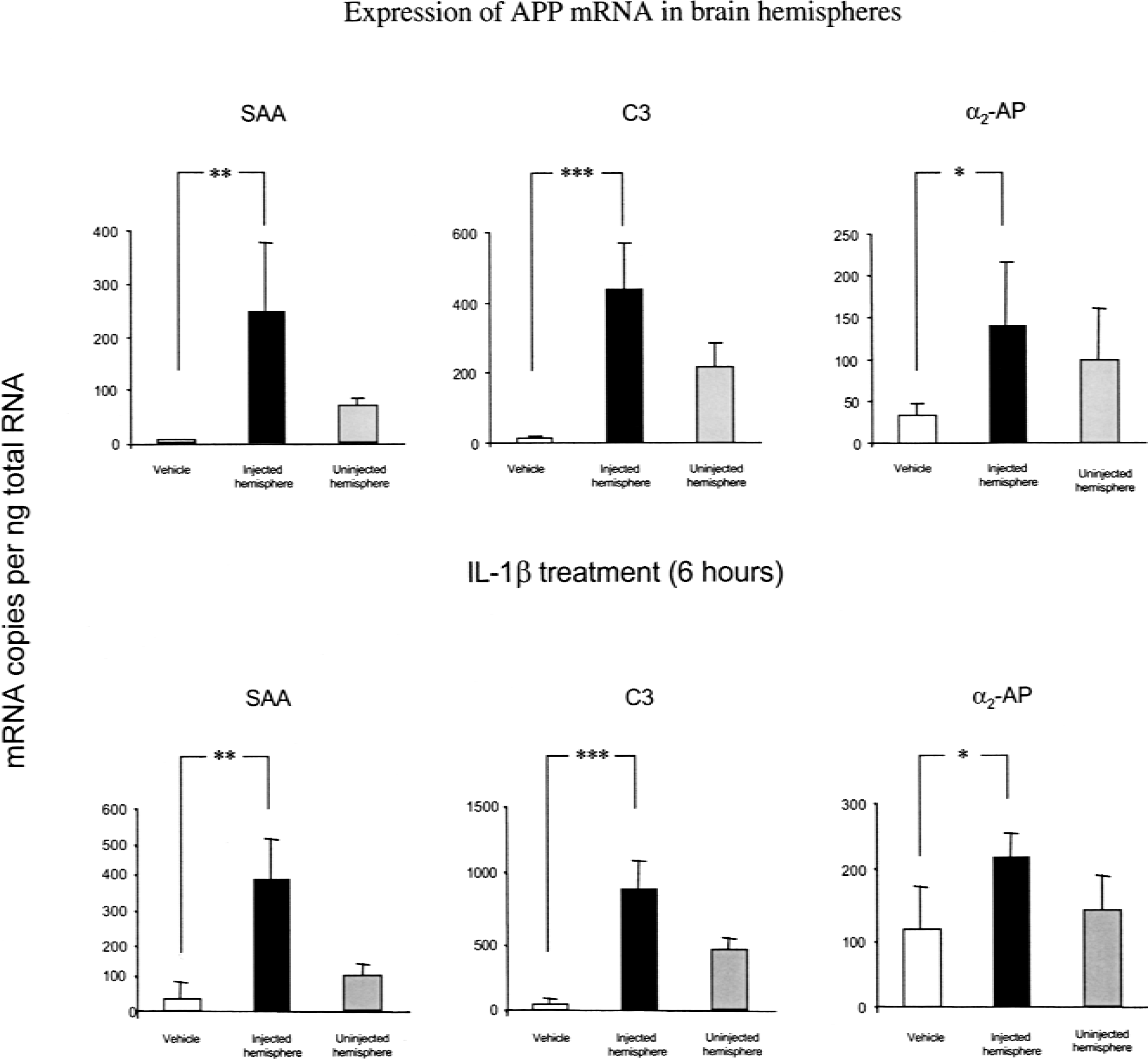
A proinflammatory challenge in the brain parenchyma elicits local acute phase protein (APP) gene expression. Injection of 1 ng mouse recombinant interleukin-1β (mrIL-1 β) into the striatum was followed by the measurement of mRNA expression in the injected and uninjected hemispheres at 6 and 12 hours. After 6 hours, serum amyloid A protein (SAA), protein complement C3, and α2-antiplasmin (α2-AP) expression was increased with respect to vehicle controls, which remained at basal levels. After 12 hours, APP mRNA expression remained significantly elevated compared with controls, which were marginally higher than at 6 hours. At both time-points the APP expression in uninjected hemispheres showed a trend towards increased mRNA expression. Bars show mean ± 1 SD. Significance values: ***P < 0.001; **P < 0.01; *P < 0.05 (ANOVA with Fischer's post hoc test; N = 4).
After 12 hours the mrIL-1β–injected hemispheres continued to express SAA mRNA at levels higher than those measured in vehicle controls (10-fold, P < 0.01) (Fig. 4). Similarly, C3 (20-fold, P < 0.001) and α2-AP (P < 0.05) mRNA continued to show elevated expression at 12 hours. Measurements from the uninjected hemispheres at both time-points showed a trend towards increased expression of APP genes compared with the negligible levels expressed in naïve animals.
DISCUSSION
The APR of mice has been extensively investigated and characterized after intraperitoneal injection of bacterial lipopolysaccharide or thioglycollate; measurements of APP serum titers or hepatic mRNA levels by northern blotting or competitive PCR have been performed (Jensen and Whitehead, 1998a; Zahedi and Whitehead, 1989). The use of Taqman RT-PCR in the present study permitted the rapid and reliable quantitation of four murine mRNA transcripts encoding the major APPs, SAA and SAP, a serine protease inhibitor (α2-AP) and complement protein (C3), in the liver and brain in two contrasting models of acute brain insults.
Using this technology we have shown that brain injury resulting from intrastriatal injection of the excitotoxin, NMDA, does not elicit a hepatic APR. An unexpected reduction in α2-AP expression was seen 6 hours after NMDA injection. However, this change was not evident at 12 hours. Given that none of the other APR genes, particularly for the major APPs, showed significant changes after NMDA injection, it is clear that an APR did not follow the excitotoxic injury. In contrast, intraparenchymal injection of the proinflammatory cytokine IL-1β resulted in a systemic inflammatory response, indicated by increased hepatic synthesis of APP mRNA. After IL-1β challenge, SAA mRNA showed a dramatic increase in the liver, whereas the amount of SAP and C3 transcripts showed slight increases after 6 and 12 hours respectively. α2-AP mRNA levels were unchanged at each time-point. However, comparison of the APP mRNA levels from vehicle control animals with naïve animals showed that surgery per se results in a strong APR. Previous studies have shown that SAA and SAP are the major APPs in the mouse and have the capacity to increase their expression 100- to 1000-fold from basal levels (Steel and Whitehead, 1993). In contrast, the positive response of C3 and α2-AP (which increase only 2- to 3-fold from basal levels) may have reached a maximum in response to the surgery alone and, consequently, upregulation of these genes after administration of mrIL-1β may have been masked.
The present study represents the first demonstration of a parenchymal proinflammatory challenge, using physiologically relevant levels of IL-1β, that results in a systemic APR. The peripheral effects of intracerebroventricularly administered IL-1 have been described previously (De Simoni et al., 1990,1993) and the transport of proinflammatory cytokines from the ventricles to the blood has been clearly demonstrated (Banks et al., 1991; Chen et al., 1997). It has been postulated that CSF drains via the superior sagittal sinus, or along the perineural sheaths of the cranial nerves that pass through the cribriform plate and terminate in the nasal mucosa where macromolecules are taken up at the vascular bed or drain into the lymphatic system (Bradbury et al., 1981; Cserr et al., 1992; Szentistvanyi et al., 1984; Yamada et al., 1991). Whatever the system of transport, it is well documented that a communicating pathway between the brain and periphery exists. Therefore, IL-1 upregulation in the brain would be expected to effect a peripheral APR. Indeed, an APR has been shown after intracerebroventricular injection of this cytokine (Di Santo et al., 1999). Measurements of serum IL-6 after IL-1β injection intracerebroventricularly suggest that peak titers of this cytokine occur at about 2 hours (Di Santo, et al., 1999). Increases in SAA and SAP mRNA have been shown within two hours of peripheral thioglycollate challenge (Zahedi and Whitehead, 1989). In the present study a profound APR, as shown by increased hepatic SAA and SAP mRNA levels, was seen only after 6 hours after intraparenchymal injection of mrIL-1β. Before this time there were no observable differences in any of the APP mRNA investigated (results not shown). It is possible that the apparent delay in onset of hepatic APP gene transcription after mrIL-1β injection resulted from the time taken for the signal to relay between the brain and the liver.
We used NMDA to produce an acute, excitotoxic neuronal degeneration in the parenchyma of the brain. In contrast to IL-1β injections that resulted in increased SAA and SAP gene transcription, NMDA induced neurodegeneration did not induce APP mRNA upregulation in the liver. The reason for this disparity in systemic inflammatory responses evoked by NMDA and IL-1β is unclear. It has been shown in rats that there is rapid induction of IL-1β and that the endogenous production of IL-1β after NMDA injection is comparable with IL-1β production after the delivery of exogenous IL-1β (Blond et al., 2000). In the same study, it was shown also that both IL-1β and NMDA induced similar levels and temporal profiles of the IL-1 receptor antagonist (IL-1ra). A notable difference between IL-1β and NMDA challenges is that intraparenchymal IL-1β injection results in robust meningitis in the injected hemisphere (Bolton and Perry, 1998) whereas NMDA injection does not (Anthony et al., 1997). Considering these observations, it is possible that the transduction of inflammatory signals from the brain to peripheral tissues, after NMDA-induced endogenous production of IL-1β, is regulated by local antiinflammatory mechanisms. In contrast, the same processes initiated after IL-1β injection, which augments further IL-1β synthesis, are insufficient to prevent the transduction of cytokine signals to the liver and subsequent APP release. It is also possible that the meningitis and hepatic responses resulting from delivery of exogenous IL-1β are due to the temporal differences between injury-driven cytokine production and the topical delivery of the cytokine
Several acute phase proteins have been reported in brain tissues (Coria et al., 1988; Duong et al., 1989; Hawkins et al., 1990; Kordula et al., 2000; Licastro et al., 1999; Morgan and Gasque, 1996). Despite these reports, the present study is the first to investigate the expression of several APPs in the brain after an acute challenge in the context of an APR. Given that the blood–brain barrier restricts the movement of large molecules from the blood to the brain parenchyma, the existence of an intraparenchymal APR may play a crucial role in the early inflammatory response to acute brain injury. It is shown here that, whereas both NMDA and IL-1β injection result in the parenchymal production of APP mRNA, only IL-1β produces a peripheral APR. As alluded to earlier, it is known also that IL-1β, but not NMDA, injection results in a meningitis (Anthony, et al., 1997; Bolton and Perry, 1998). Clearly, given that NMDA-mediated neurodegeneration results in proinflammatory cytokine release, the spread of endogenously produced proinflammatory cytokines to the meninges and periphery is being prevented. In this regard it is tempting to speculate that components of the parenchymal APP production may be responsible for down-regulating the effects of endogenous IL-1β or aiding its clearance or degradation after NMDA injection.
Upregulation of APP mRNAs was shown also in the uninjected hemisphere of lesioned animals; when compared with the extremely low levels of APP mRNA in naïve brain samples an upregulation is apparent. This indicates that communication between the two hemispheres for the induction of acute proinflammatory signals exists. The extent to which the two challenges (i.e., IL-1β and NMDA) were effective in causing an APP increase was not the same. Interleukin-1β–injected animals showed less induction of APP in the uninjected hemispheres than did NMDA. The reasons and significance (if any) for these interhemispheric affects is unknown, although it has been shown in rats that, after IL-1β injection or NMDA injection into the striatum, IL-1ra levels are increased in the uninjected hemisphere (Blond et al., 2000). Because APP production is cytokine-dependent, and NMDA results in bilateral APP production, it is clear that, after excitotoxic cell death, APP-inducing signals spread to both brain hemispheres. The failure of IL-1β, known to be synthesized after NMDA injection (Blond et al., 2000), to elicit a meningitis or hepatic APR may further support the notion that the brain APPs are acting in an antiinflammatory fashion. Further work will focus also on this potential role of the parenchymal APPs.
Although it is now well established that brain damage can initiate a strong APR, the function of this response in terms of brain injury and outcome has never been established. The use of an animal model in the present study has enabled us to minimize incidental tissue trauma and create a reproducible brain lesion from which we could assess the contribution of the brain injury alone to a systemic APR. This model can now be used to investigate the influence of systemic inflammation on acute brain damage.
In summary, it is clear that several markers of acute peripheral inflammation are expressed after intrastriatal IL-1β injection, used as a model of brain inflammation. Furthermore, it was shown that, in addition to the hepatic expression of APP mRNA, the brain parenchyma has the capacity to increase the gene expression of major APPs, complement proteins, and serine protease inhibitors in response to excitotoxic lesions or proinflammatory cytokines. Further investigations will seek to identify whether these locally produced and systemic responses influence neurologic outcome. Such studies may provide valuable insights into the affect of the APR on brain-injured patients.
