Abstract
The adult central nervous system parenchyma is resistant to inflammation, but in juvenile rats the injection of inflammatory mediators, interleukin-1β for example, gives rise to extensive neutrophil recruitment and neutrophil-dependent blood–brain barrier breakdown. The factors that confer this resistant phenotype are unknown. In this study, the authors demonstrate that E- and P-selectin expression is increased to a similar extent in adult and juvenile brain after the intracerebral injection of IL-1β. Thus, the refractory nature of the brain parenchyma cannot be attributed to an absence of selectin expression. However, in injuries where the resistant characteristic of the brain parenchyma is compromised, and neutrophil recruitment occurs, selectin blockade may be an advantage. The authors investigated the contribution that selectins make to neutrophil recruitment during acute inflammation in the brain. The authors examined neutrophil recruitment by immunohistochemistry on brain sections of juvenile rats killed four hours after the intracerebral injection of IL-1β and the intravenous injection of neutralizing anti-selectin monoclonal antibodies (mAb). The administration of the P-selectin blocking mAb inhibited neutrophil recruitment by 85% compared with controls. Surprisingly, E-selectin blockade had no effect on neutrophil recruitment to the brain parenchyma. Thus, P-selectin appears to play a pivotal role in mediating neutrophil recruitment to the brain parenchyma during acute inflammation.
Studies on humans and in animal models show that inflammation can have a deleterious effect on the outcome of acute brain injury, such as stroke and head injury. In the periphery, neutrophils are the first circulating leukocytes to arrive at the site of injury. When they accumulate in blood vessels, neutrophils may bring about a reduction in blood flow and occlusion of microvessels (del Zoppo et al., 1991). The release of lysosomal enzymes and free radicals by neutrophils also produces an increase in vascular permeability, which gives rise to edema (Weiss, 1989). Although mechanisms appear to have evolved to restrict neutrophil entry to the brain parenchyma, it is clear that neutrophil recruitment is a feature of acute brain injury (stroke or trauma). Numerous studies using the permanent, or transient, focal ischemia model in mice and rats have shown that neutrophil depletion decreases cerebral tissue injury (Jean et al., 1998). In models of traumatic brain injury, there is also a correlation between neutrophil accumulation and the development of cerebral edema (Schoettle et al., 1990). These studies highlight two important issues: first, what factors are responsible for restricting neutrophil recruitment to the brain; and second, when the brain is compromised after a severe injury, which molecules become important mediators of neutrophil recruitment.
It is often stated that the presence of the blood–brain barrier (BBB), which restricts the entry of polar solutes into the brain, as a consequence of the tight junctions between the endothelial cells, poses an impenetrable obstacle to leukocytes attempting to exit the vasculature. Moreover, it is assumed that when the BBB is damaged, leukocyte recruitment is enabled. However, recent evidence in animal models shows that this is not the case. Circumstances under which leukocytes enter the brain under normal conditions and during inflammation have been reviewed recently (Perry et al., 1997). It is becoming increasingly clear that factors other than the BBB, such as adhesion molecule or chemokine expression, must be responsible for the atypical nature of the inflammatory response in the brain.
An essential requirement for leukocyte recruitment from the circulation is the expression of adhesion molecules on the endothelium. The arrest of freely flowing leukocytes and their subsequent rolling along the endothelium is thought to be mediated by the selectins, which bind to carbohydrate ligands (Vestweber and Blanks, 1999). Both E-selectin and P-selectin are expressed on activated endothelium, and P-selectin also is present on platelets. They interact with the sialyl Lewis-X (sLex) carbohydrate associated with CD15 on the surface of neutrophils. Their function is to slow the circulating neutrophils as they roll along the endothelium. Tethered neutrophils then recognize chemokines presented on the surface of the activated endothelium (Springer, 1995). This interaction leads to the up-regulation of the affinity of integrins such as very late activation antigen-4 (VLA-4) and leukocyte function antigen-1 (LFA-1) on the surface of the neutrophils for their cognate ligands, members of the immunoglobulin family of transmembrane glycoproteins such as vascular cell adhesion molecule-1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-1), respectively, on the endothelium (Gahmberg, 1997, 1998; Gonzalez-Amaro and Sanchez-Madrid, 1999; Issekutz, 1993). In the central nervous system (CNS), ICAM-1 and VCAM-1 are readily up-regulated on brain endothelium after injury, but currently, it is still unclear how selectin expression might affect neutrophil recruitment in the CNS.
Previous studies have demonstrated that although the injection of interleukin-1β (IL-1β) into the brain parenchyma of adult rats does not induce the recruitment of leukocytes at the site of injection, a similar injection in juvenile rats will give rise to an inflammatory response more typical of the response in peripheral tissues (Anthony et al., 1997). The IL-1β–induced inflammation is characterized by the recruitment of neutrophils to the brain parenchyma by 4 hours with concomitant increase in BBB permeability. This BBB breakdown is neutrophil-dependent as demonstrated by immunohistochemistry and magnetic resonance imaging on neutrophil-depleted animals (Anthony et al., 1997; Blamire et al., 2000). This “window of susceptibility” model provides a tool to study the refractory nature of the brain parenchyma, where the adult rat brain mimics a normal resistant brain and the juvenile rat brain mimics a brain compromised by acute injury where neutrophil recruitment occurs. A differential expression of adhesion molecules or chemokines may be responsible for the lack of neutrophil recruitment to the adult brain parenchyma after IL-1β injection. Currently, no difference in the endothelial expression of adhesion molecules belonging to the immunoglobulin superfamily, ICAM-1, VCAM-1, and PECAM-1 has been detected between the two age groups. However, E- and P-selectin expression has never been studied in this model. In this study, the authors tested the hypothesis that a differential expression of selectins may account for the refractory nature of the adult brain. The authors sought to determine whether E-selectin or P-selectin are up-regulated after an acute inflammatory challenge in the CNS and whether the pattern of selectin induction could account for the increased susceptibility of juvenile animals. The authors also sought to establish how E-selectin and P-selectin expression might contribute to the recruitment of neutrophils into the CNS.
MATERIALS AND METHODS
Animals
Male Lewis rats aged either 3 to 4 weeks (40 to 50 g, juveniles) or 10 weeks (200 to 250 g, adults) were obtained from Charles River (Margate, Kent, U.K.). All animals were housed under standard conditions with pelleted food and water available ad libitum. Home office approval was obtained for all of the animal experiments described in this study.
Stereotaxic surgery
Stereotaxic surgery was performed as previously described (Matyszak and Perry, 1995). Briefly, anesthetized animals (Avertin 1 mL/100 g) were held in a stereotaxic frame and the skull and brain were exposed. One nanogram of rat recombinant IL-1β diluted in phosphate-buffered saline (PBS) containing 0.1% bovine serum albumin (BSA) (n = 3 at each time point, total n = 12) or vehicle (n = 3 at each time point, total n = 12) were injected into the striatum, in a volume of 1 μL, with a finely drawn glass capillary using the following coordinates: bregma +1.0 mm, lateral 3.0 mm, and depth 4.0 mm. Injections were made over a period of 5 minutes and the animals were left to recover for 2, 4, 6, or 8 hours.
Neutrophil depletion
Three-week-old animals were gamma irradiated with 9Gy (Gulmay medical 225 kV orthovoltage irradiator, dose rate 3 Gy/min) to deplete blood leukocytes (n = 3). These animals were maintained for 3 days with oxytetracycline (10 mg/100 mL) added to the drinking water before the cytokine injections were performed. The animals were perfused 6 hours after IL-1β injection. The efficiency of the depletion treatment was assessed on brain sections of depleted animals by neutrophil-specific immunostaining using an in-house polyclonal antibody (Anthony et al., 1998b).
Selectin blockade
The individual contribution of E-selectins and P-selectins to neutrophil emigration in juvenile rats was studied by specifically blocking the selectin proteins with anti-E (RME-1) (n = 6), anti-P-selectin (RMP-1) (n = 6), or a combination of both monoclonal antibodies (n = 3) that have been characterized previously (Walter et al., 1997a, 1997b). The antibodies were administered to juvenile rats intravenously (400 μg/animal) immediately before intracerebral injection of cytokine. At 4 or 6 hours, the animals were deeply anesthetized and perfused. The effect of the blocking antibodies on neutrophil recruitment was assessed by immunocytochemistry with the same neutrophil-specific antibody described above.
Perfusion and tissue preparation
The animals were deeply anesthetized after 2, 4, 6, or 8 hours with sodium pentobarbitone (Sagatal, Rhone Merieux Ltd., Harlow, Essex, U.K.) and transcardially perfused with heparinized 0.9% saline solution. The brains were removed immediately and quickly frozen in Tissue-Tek OCT embedding compound (Sakura Finetek Europe B.V., Zoeterwoude, The Netherlands). The frozen brains were stored at −20°C until use.
Immunohistochemistry
Frozen 10-μm coronal serial sections of fresh-frozen tissue were cut on a cryostat and mounted on 3-aminopropyltriethoxysilane-coated slides. The sections were air-dried and fixed in ice-cold absolute ethanol for 10 minutes before staining. The primary antibodies (anti-E-selectin mAb RME-1, 10 μg/mL, anti-P-selectin mAb RP-2, 10 μg/mL, anti-GPIIb/IIIa mAb p55, 10 μg/mL, anti-laminin polyclonal antibody, 1/500, or anti-neutrophil serum, 1/2000) were incubated on the sections for 2 hours and detected using a biotinylated secondary antibody and the ABC Elite detection kit from Vector laboratories (Peterborough, U.K.) with 3,3′-diaminobenzidine (DAB) as the substrate. Sections used for counting blood vessels were not counterstained. Sections used for photographic purposes were counterstained with hematoxylin.
Platelets immunostaining
Platelets were detected on 10-μm brain sections by immunohistochemistry using the anti-rat GPIIb/IIIa p55 monoclonal antibody. Blood smears from normal animals were used as positive controls.
Analysis
For each animal, five fields immediately adjacent to the site of injection were chosen in the injected hemisphere (E-selectin, P-selectin, laminin) and five fields were chosen randomly in the uninjected hemisphere (laminin staining only). The number of positively-stained vessels were counted using the Leica Qwin image analysis software (Leica Imaging Systems Ltd., Cambridge, U.K.). All vessels, whether longitudinal, transverse, or branched, were each counted as one single blood vessel. The number of E-selectin and P-selectin positive vessels per mm2 were expressed as a percentage of the laminin positive vessels to account for variations between animals.
Neutrophils were counted in sections immediately adjacent to the injection site. Five nonoverlapping fields, containing the highest density of recruited cells within the parenchyma, were chosen and the number of neutrophils were calculated as an average number per mm2 for each animal.
The minimum transverse diameters of E- or P-selectin immunostained vessels were measured using the Leica Qwin image analysis software. Capillaries were identified as being less than 7.5 μm in diameter, whereas precapillary and postcapillary vessels ranged from 7.5 to 30 μm in diameter.
Statistics
Data are presented as the mean ± (SD), and where statistical analysis has been used, Student's t-test or analysis of variance test with Bonferroni adjustment was applied.
Reagents and antibodies
The anti-E-Selectin (clone RME-1) and anti-P-Selectin (clones RP-2, RMP-1) mAbs generated by Dr. A. Issekutz have been previously described (Walter et al., 1997a, 1997b). The anti-rat GPIIb/IIIa mAb p55 reactive with platelets was a kind gift from Dr H. Miyazaki (Kirin Brewery, Gunma, Japan). The anti-laminin polyclonal antibody was purchased from Sigma (Cat N° L9393, 1/500; Poole, Dorset, U.K.). Rat recombinant IL-1β was provided by the National Institute for Biological Standards and Controls (NIBSC; Potters Bar, U.K.). The anti-neutrophil polyclonal antibody was raised against rat neutrophils in house. Unless otherwise stated, all chemicals were purchased from Merck (Poole, Dorset, U.K.) and were of AnalaR grade.
RESULTS
Laminin staining
For each animal, the authors compared the number of laminin-positive vessels in the injected and uninjected hemispheres (Fig. 1A). The authors also compared the number of laminin-positive vessels at each time point after IL-1β injection in the adult and juvenile animals. There was no significant difference between the number of laminin-positive vessels in the injected and uninjected hemispheres (data not shown). Furthermore, no difference was found in the number of laminin-positive vessels at each time point and between the two age groups. However, because slight variations were observed between individual animals, the number of selectin-positive vessels for each animal was expressed as a percentage of the laminin-positive vessels.
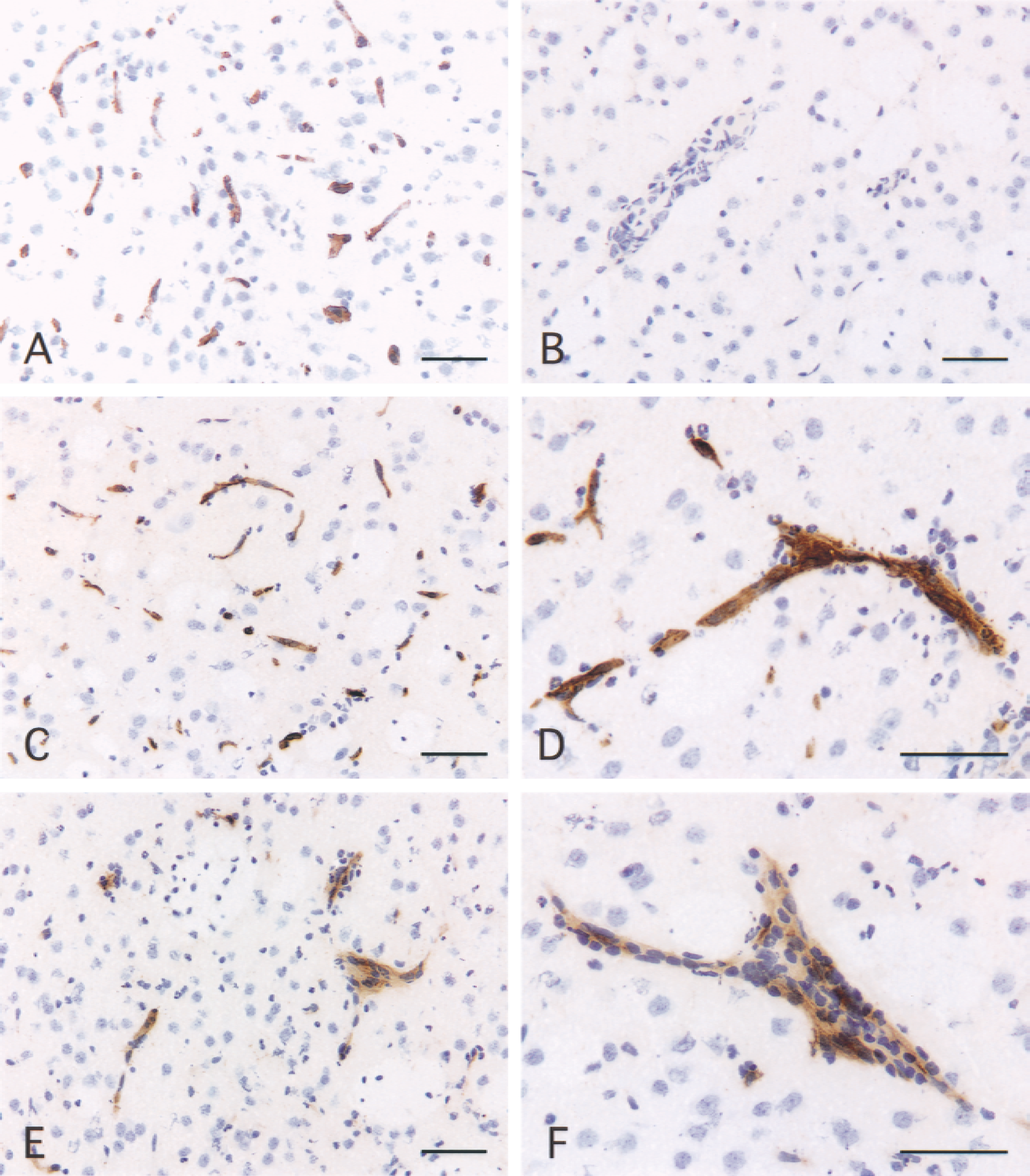
Expression of E- and P-selectin in juvenile rat brain after injection of IL-1β. Coronal sections of brain were immunostained as described in Materials and Methods.
E-selectin expression in adult and juvenile rats
Sections from control animals of either age injected with PBS-BSA showed no positive E-selectin staining (Fig. 1B). The injection of IL-1β induced the expression of E-selectin on vascular endothelium in adult and juvenile animals (Figs. 1C, 1D, and 2A) between 4 and 8 hours in the injected hemisphere. In both age groups, no E-selectin expression could be detected in the uninjected hemisphere at the various time points. In adult rats, the expression of E-selectin peaked at 4 hours when 76% of the vessels were positive and decreased rapidly to 30% at 8 hours, although no neutrophil recruitment was observed at these time points. In juvenile animals, E-selectin expression could be detected at 4 hours but peaked at 6 hours when 90% of the vasculature was positive and declined at 8 hours. Vessels in both adult and juvenile animals expressed E-selectin, but there was significantly more E-selectin in adults at 4 hours (unpaired Student's t-test, P = 0.0256) and more E-selectin–positive vessels in juveniles at 8 hours (unpaired Student's t-test, P = 0.0237). Although there were more E-selectin-positive vessels in the juveniles than in the adults at 6 hours, this difference was not statistically significant (unpaired Student's t-test, P = 0.2024). E-selectin staining was detected on capillary microvessels (diameter < 7.5 μm) as well as on pre-and postcapillary venules (diameter between 7.5 μm and 30 μm). Only 13% of the E-selectin–positive vessels were involved in the recruitment of neutrophils in juvenile animals, and the mean diameter of these vessels was 12.17 μm. E-selectin expression also could be observed on vessels of the meninges and choroid plexus.
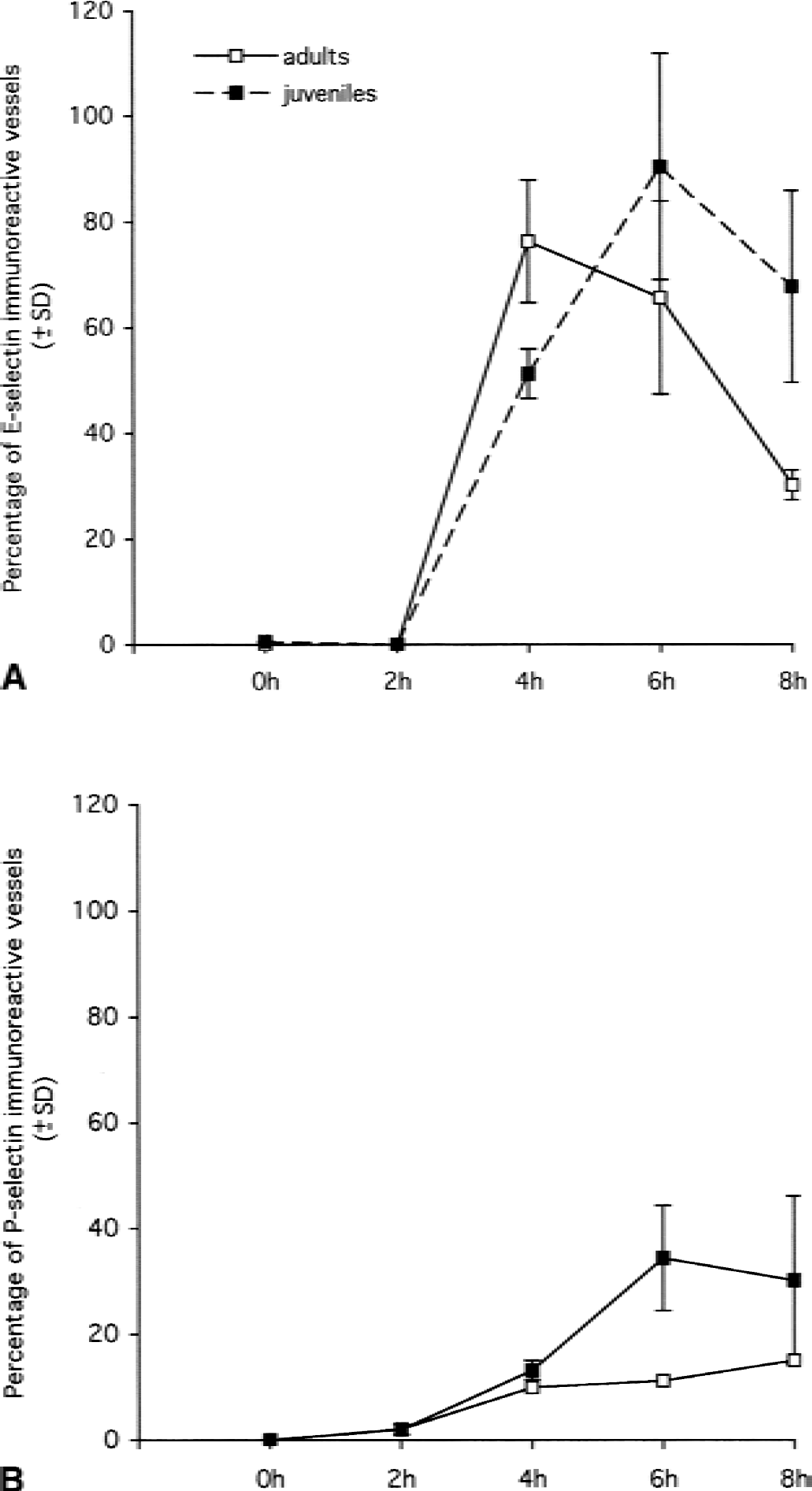
Temporal expression of E- and P-selectin on cerebral endothelium in the hemisphere injected with IL-1β in adult and juvenile animals. Coronal sections of brain were immunostained for E- and P-selectin, and the number of immunoreactive vessels was counted as described in Materials and Methods.
P-selectin expression in adult and juvenile rats
Sections from control animals, adults, and juveniles injected with PBS-BSA showed no positive staining for P-selectin. The intracerebral injection of IL-1β induced the up-regulation of P-selectin between 2 and 8 hours (Figs. 1E, 1F, and 2B). This up-regulation of P-selectin expression could not be attributed to the aggregation of platelets as none could be detected by immunocytochemistry with an anti-platelet monoclonal antibody. Like E-selectin, no expression of P-selectin could be detected in the uninjected hemisphere at these time points either in adult or juvenile animals. In adult rats, P-selectin expression appeared at 2 hours when only 3% of the vessels were positive and this increased up to 15% at 8 hours. In juvenile rats, P-selectin also could be detected from 2 hours, but expression peaked at 6 hours when 35% of the vessels were positive; this then decreased slightly at 8 hours. At 6 hours, there was significantly more P-selectin expression in juveniles than in adults (unpaired Student's t-test, P = 0.0158) and there was more P-selectin expression in juveniles than in adults at 4 and 8 hours, but these differences were not statistically significant (unpaired Student's t-test, 4 hours, P = 0.0608, 8 hours, P = 0.1753). Immunostaining with an anti-platelet mAb (anti-GPIIIb/IIa, p55) showed no platelets bound to the vasculature both in adult and in juvenile animals. Therefore, the P-selectin expression observed on the cerebral endothelium can only be attributed to the endothelial cells (data not shown). Like E-selectin, P-selectin immunoreactivity was detected on capillaries and venules, as well as on a few larger vessels (diameter > 30 μm). There were significantly more vessels stained for E-selectin than for P-selectin both in adults and in juveniles at 2, 4, 6, and 8 hours (Fig. 2). In juvenile rats, 37% of the P-selectin–positive vessels were involved in neutrophil recruitment. The mean diameter of these vessels was 15 μm. Like E-selectin, P-selectin immunoreactivity also was detected on vessels of the meninges and choroid plexus.
Effect of neutrophil depletion on selectin expression
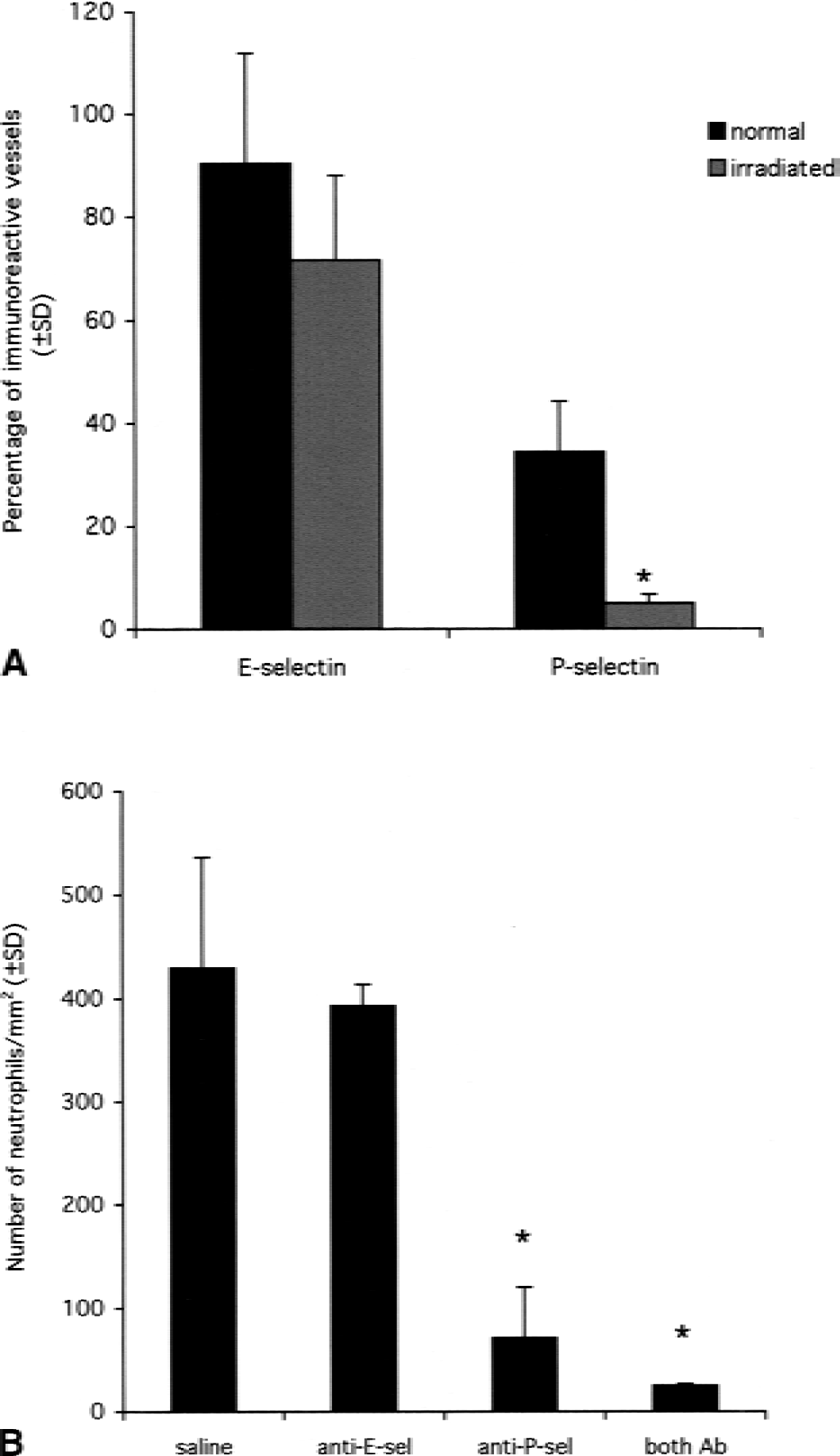
After observing that juvenile animals expressed significantly more selectins than their adult counterparts, the authors studied whether the neutrophils that were recruited in the juveniles, but not in the adults, could lead to an increase in selectin expression. For this purpose, juvenile animals were irradiated to deplete the circulating leukocytes. In irradiated juvenile rats, neutrophil infiltration was not seen after IL-1β injection, as assessed by anti-neutrophil serum staining (data not shown). The irradiation had no effect on the number of laminin-positive vessels present in the brain (data not shown). Six hours after IL-1β injection, 72% of the laminin-immunoreactive vessels were positive for E-selectin but only 5% of the vessels were positive for P-selectin. There was no statistical difference between the number of E-selectin–positive vessels in irradiated and in normal juvenile rats (unpaired Student's t-test, P = 0.1091). However, there were significantly fewer P-selectin–positive vessels in irradiated animals as compared with control animals (unpaired Student's t-test, P = 0.0051) (Fig. 3A).
Effect of selectin blockade on neutrophil recruitment
To study the functional importance of both selectins, the authors specifically blocked each selectin using anti-E-selectin (RME-1) or anti-P-selectin (RMP-1) antibodies administered intravenously before the intracerebral injection of IL-1β in juvenile animals. Synergy between E- and P-selectin also was studied by injecting both blocking antibodies simultaneously. Animals were killed after 4 and 6 hours. The detection of E- and P-selectin staining on vessels in sections where no primary antibody had been applied demonstrates that the intravenous injections had led to antibody binding to the cerebral vasculature. In response to IL-1β, control rats injected intravenously with saline (n = 9) showed extensive neutrophil recruitment to the brain parenchyma in the injected hemisphere at 4 and 6 hours (Fig. 3B). However, the P-selectin–blocked rats showed only a small number of neutrophils in the brain parenchyma at 4 hours (Figs. 3B and 4). Monoclonal antibody RMP-1 blocked the neutrophil emigration by 85% compared with saline controls at 4 hours (ANOVA, P < 0.0001). Although RME-1 decreased neutrophil emigration by 8.5% compared with saline controls at 4 hours, this did not reach statistical significance. By 6 hours, the number of neutrophils recruited to the brain parenchyma in RME-1–blocked animals and in controls was virtually identical (Fig. 4B). The combination of RME-1 and RMP-1 did not significantly increase the neutrophil emigration blockade (94%) compared with RMP-1 alone.
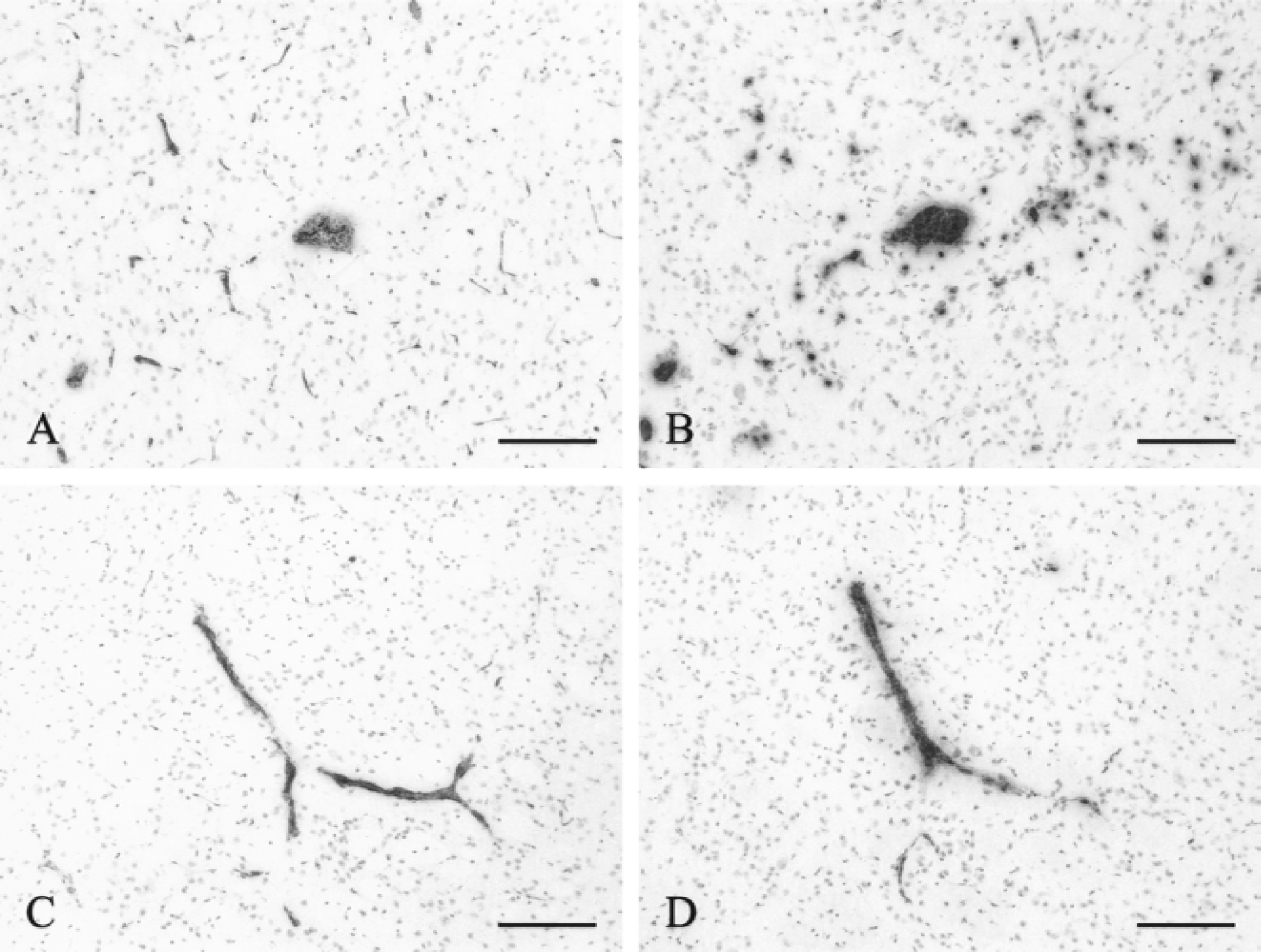
Effect of selectin blockade on neutrophil recruitment to the brain parenchyma. Neutrophil recruitment was examined by immunohistochemistry on brain sections of juvenile rats killed 6 hours after the intracerebral injection of IL-1β and the intravenous injection of mAbs blocking selectin function.
DISCUSSION
The principal findings of this study may be summarized as follows. First, the refractory nature of the adult brain can not be attributed to a lack of expression of the adhesion molecules belonging to the selectin family, as both adult and juvenile rats express E- and P-selectins after intrastriatal injection of IL-1β. Second, the neutrophil contributes to the induction of P-selectin on cerebral blood vessels because the depletion of circulating neutrophils in juvenile rats significantly decreased the number of P-selectin positive vessels compared with nonirradiated controls. Third, neutrophil recruitment to the brain parenchyma depends almost solely on P-selectin expression, because blocking of P-selectin function alone significantly reduced the migration of neutrophils to injected hemisphere.
The authors used the laminin staining to ensure that the intracerebral injection of saline or IL-1β did not induce a variation in the number of vessels present at and around the site of injection. At the early time points that were studied, no neovascularization was detected, making laminin a suitable blood vessel marker.
Expression of E-selectins and P-selectins on adult and juvenile cerebral endothelium
The expression of E- and P-selectins on cerebral blood vessels has been described previously in models of permanent or transient middle cerebral artery occlusion in rats (Zhang et al., 1998) and in animal models of head trauma (Whalen et al., 1998).
All studies have been performed on adult or adolescent animals, but the expression of selectins in juvenile animals has never been studied. To see if the differential neutrophil recruitment observed between adult and juvenile rats after a proinflammatory stimulus could be explained by a difference in selectin expression, the authors quantified by immunohistochemistry the number of E-selectin and P-selectin–positive vessels in both age groups after intrastriatal injection of IL-1β. In agreement with all the studies described earlier, constitutive expression of E-selectin or P-selectin was not detected in the control animals. In peripheral tissues, P-selectin is constitutively expressed in endothelial cells and stored in Weibel-Palade bodies along with the von Willebrand factor. This constitutes a readily available store of P-selectin, which is released almost instantly upon endothelial cell activation. Unlike other tissues, the cerebral endothelium lacks this constitutive expression of P-selectin (Gotsch et al., 1994). The current results demonstrate that the temporal and quantitative expression of E-selectin and P-selectin was similar in adult and juvenile animals in response to IL-1β. At 4 hours, there was no difference in the number of P-selectin immunoreactive vessels between the two age groups. Unexpectedly, there were more E-selectin immunoreactive vessels in the adults than in the juveniles at 4 hours. Therefore, the lack of recruitment of neutrophils to the brain parenchyma in the adult animals cannot be attributed to a lack of expression of the selectin adhesion molecules at this early time point. However, the authors observed significantly more E-selectin and P-selectin, at 6 and 8 hours, respectively, in the juveniles than in the adults, and this difference may be relevant to the limited neutrophil recruitment in the adult brain after IL-1β injection.
It is generally agreed that the emigration of leukocytes from the blood vessels to the site of inflammation follows a three-step paradigm: leukocyte rolling along the activated endothelium, firm adhesion, and transendothelial migration. The selectins are responsible for the slow rolling of the leukocytes, whereas adhesion molecules of the immunoglobulin and integrin superfamilies are responsible for the firm adhesion. Although the rolling and the adhesion are usually considered as two very distinct stages in the recruitment of white blood cells, there is a certain level of overlap in adhesion molecule function. It appears that ICAM-1 and VCAM-1 are capable of mediating capture and rolling of leukocytes as well as firm adhesion in non-CNS tissue (Alon et al., 1995; Johnston et al., 1996). A lack of ICAM-1, VCAM-1, or platelet/endothelial cell adhesion molecule-1 (PECAM-1) expression on the cerebral endothelium of adult rats may explain the lack of neutrophil recruitment in these animals. However, previous studies by the authors' group have demonstrated the expression of ICAM-1 on cerebral vessels of adult rats and of VCAM-1 and PECAM-1 on murine cerebral endothelium (Bell and Perry, 1995; Bolton et al., 1998). Although the neutrophils are not recruited to the brain parenchyma of adult rats at the earliest time point studied, they are present in the meninges and the choroid plexus (Anthony et al., 1997). Therefore, the lack of neutrophil emigration can not be explained by a lack of expression of adhesion molecules such as L-selectin on their surface. The differential recruitment of neutrophils could be explained by a different expression of chemokines in the adult and juvenile brain. However, the authors have observed that the intracerebral injection of IL-1β leads to similar levels of transcription and expression of the chemokines CINC-1 and MIP-2 in the adult and the juvenile brains as demonstrated by quantitative reverse transcriptase-polymerase chain reaction and ELISA measurements (Anthony et al., 1998a). Therefore, the differential neutrophil recruitment between the two age groups can not be explained by a lack of chemokine expression; but the intrastriatal injection of recombinant chemokines produced a more intense neutrophil response in young rats than in adults. The IL-1β–induced BBB breakdown in young rats could be attenuated by an anti-CINC-1 neutralizing antibody suggesting that the immature CNS is more susceptible to the chemotactic effect of CXC chemokines.
Neutrophils contribute to the expression of P-selectin on cerebral endothelial cells
As described above, the authors consistently detected more E- and P-selectin in juveniles than in adults at 6 and 8 hours. To know whether the recruitment of neutrophils itself could lead to an up-regulation of the selectins on the cerebral endothelium, the authors depleted juvenile rats of circulating neutrophils by gamma irradiation and studied selectin expression at 6 hours post IL-1β injection, time of peak neutrophil recruitment, and selectin expression. The irradiation had no significant effect on the number of E-selectin immunoreactive vessels. However, the depletion of circulating neutrophils lead to a significant decrease of P-selectin-positive vessels suggesting that the neutrophils themselves can induce the expression of P-selectin on the endothelium. The decrease in P-selectin expression cannot be attributed to a decrease in the number of platelets binding to the endothelium as immunocytochemistry has demonstrated no platelet deposition on the vasculature in control nonirradiated animals injected with IL-1β. This observation suggests that two distinct pathways are involved in the expression of E- and P-selectin on cerebral endothelium. First, with stimulation by IL-1β, endothelial cells will express E-selectin and P-selectin, which could allow for neutrophil rolling along the endothelium. Second, P-selectin alone is further up-regulated by the neutrophils. On the one hand, activated neutrophils could secrete a cytokine that selectively up-regulates P-selectin expression. On the other hand, binding of E- and P-selectin to their respective ligands may trigger different signaling pathways, which may lead to the up-regulation of P-selectin but not E-selectin. These two hypotheses will be discussed in turn.
With activation by proinflammatory cytokines or LPS, neutrophils are capable of producing secondary cytokines. These include IL-1β, tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and transforming growth factor-β (TGF-β). Both IL-1β and TNFα are capable of inducing the expression of E- and P-selectin on BMECs in vivo and in vitro (Gotsch et al., 1994). When porcine aortic endothelial cells are stimulated by TNF-α in the presence of IL-4, there is enhanced P-selectin expression with a parallel reduction in E-selectin expression (Stocker et al., 2000). IL-6 also has been reported to induce the expression of E-selectin on immortalized human brain capillary endothelial cells, but P-selectin expression was not studied (Prudhomme et al., 1996). Interestingly, TGF-β has been shown to down-regulate the expression of E-selectin on endothelium, whereas blocking of TGF-β activity with antibodies significantly inhibited P-selectin gene expression in a rat model of hypertension (Gamble et al., 1993).
Although the extracellular domains of the selectins are well conserved, there is little homology between the intracellular domains of E- and P-selectin, suggesting that the two molecules use different signaling pathways and have different functions (Johnston et al., 1989). The regulation of P-selectin expression after leukocyte adhesion has not been studied, but Yoshida and coworkers have shown that the E-selectin cytoplasmic domain is constitutively phosphorylated and, upon ligation, becomes dephosphorylated in vitro (Yoshida et al., 1998). Although there is a biochemical modification of the protein in response to ligation, no variation in the amount of extracellular or intracellular E-selectin was observed suggesting a lack of transcriptional regulation. These results are in agreement with the authors' own results that show that E-selectin expression is not modified after neutrophil adhesion.
Neutrophil recruitment to the brain parenchyma depends mainly on selectin expression
The current results show that P-selectin plays a particular role in the recruitment of neutrophils to the brain parenchyma. To study more specifically the respective function of each selectin in the recruitment of neutrophils, the authors selectively blocked rat E- and P-selectin with mAbs shown previously to block adhesion function of these selectins in vitro (Walter, et al., 1997a, 1997b) and in vivo (Walter and Issekutz, 1997). Neutrophil recruitment then was investigated at 4 hours after IL-1β injection, when polymorphonuclear leukocytes are still being recruited. The intracardial perfusion of the rats removes all nonbinding leukocytes from the vasculature. However, after perfusion, neutrophils could still be observed attached to the venules demonstrating that, even in the presence of blocking antibodies, the neutrophils can attach to the cerebral endothelium by an E- and P-selectin–independent mechanism. Compared with saline-injected control animals, blocking of P-selectin decreased by 85% the number of neutrophils recruited into the brain parenchyma. However, the E-selectin antibody did not inhibit the emigration of neutrophils. To see if neutrophil emigration was simply delayed in E-selectin–blocked rats, the blockade experiment was repeated at 6 hours. At this later time point, no further blocking was observed. Simultaneous blocking of E- and P-selectins did not significantly decrease the number of recruited neutrophil compared with the animals where P-selectin alone was blocked.
Studies looking at the role played by E- and P-selectin during leukocyte recruitment and acute inflammation in peripheral tissue have led to conflicting results. P-selectin knockout mice show a partial defect in neutrophil recruitment to peripheral tissues, whereas neutrophil emigration is not impaired in E-selectin-deficient mice (Bullard et al., 1996). In peripheral organs, E- and P-selectin have redundant functions during the recruitment of neutrophils (Homeister et al., 1998). In the brain, the study of the role of selectins has mainly been performed using the rodent focal cerebral ischemia model. These studies looked either at E-selectin or at P-selectin, but rarely studied the individual contribution of each selectin in the same model of inflammation. The administration of a synthetic oligopeptide corresponding to the lectin domain of E-selectin, and therefore blocking the adhesion of leukocytes, decreases the size of ischemic injury after transient focal cerebral ischemia, suggesting that E-selectin plays a role in recruiting neutrophils in this model (Morikawa et al., 1996). However, the oligopeptide may have interfered with binding of PSGL-1 to P-selectin as well as to E-selectin ligands. A different study showed that neutrophil accumulation in the ischemic cortex of wild-type mice is significantly greater than in P-selectin knockout mice, suggesting that P-selectin also plays a significant part in neutrophil recruitment (Connolly et al., 1997). In the same study, the blocking of P-selectin in wild-type mice improved the stroke outcome. Recently, Carrithers and colleagues have observed that an anti–P-selectin monoclonal antibody could inhibit by 49% the recruitment of activated T lymphocytes across the BBB (Carrithers et al., 2000). The current study shows that P-selectin plays a key role in neutrophil emigration into the brain parenchyma during acute inflammation. The results that the authors observed can not be attributed to a neutropenic effect of the anti-P-selectin antibody as no such effect was observed with the same blocking antibody in normal rats (Walter and Issekutz, 1997). The residual recruitment observed after blockade of selectin function may be attributed to P-selectin–independent mechanisms such as E-selectin, L-selectin, VCAM-1, or ICAM-1. The preeminent role of P-selectin may arise from the fact that P-selectin is a far more extended structure than E-selectin and can protrude beyond the glycocalyx, allowing for more efficient interaction between leukocytes and endothelial cells (Patel et al., 1995). It may also be attributed to the fact that E- and P-selectins have slightly different functions during neutrophil recruitment. In the periphery, E-selectin mediates slow rolling of the neutrophils, whereas P-selectin is capable of mediating both capture and slow rolling (Labow et al., 1994). Although E-selectin seems to play only a secondary role in the recruitment of neutrophils to the brain parenchyma during acute inflammation, it may have a more important function during the recruitment of other leukocytes during other types of inflammation.
The observation that 85% of the neutrophil recruitment induced by a proinflammatory cytokine is selectin-dependent may have important implications for the treatment of acute brain injuries such as stroke or head trauma, where neutrophils are known to participate to cerebral tissue injury by the occlusion of blood vessels and the release of enzymes and free radicals. In the spinal cord where inflammation is significantly more florid than in the brain, it is likely that neutrophils may cause even more damage (Schnell et al., 1999). Blockade of selectin-mediated neutrophil recruitment in these acute injuries to the CNS offers a valuable route to reduce secondary tissue damage.
Footnotes
Acknowledgments:
The authors thank Dr. Tim Illidge and Mr. Richard Reid from the Tenovus Research Laboratory for technical assistance with the irradiator.
