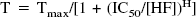Abstract
Fasudil hydrochloride (AT877, hexahydro-1-(5-isoquinolinesulfonyl)-1H-1,4-diazepine hydrochloride, identical to HA1077) inhibits cerebral vasospasm after subarachnoid hemorrhage in experimental animals and humans. In the current study, the vasorelaxing mechanism of hydroxyfasudil, a hydroxylated metabolite of fasudil hydrochloride, was determined in the rabbit basilar artery. The effects of hydroxyfasudil on tension, intracellular Ca2+ concentration ([Ca2+]i), and phosphorylation of the myosin light chain were examined using the isolated and intact or permeabilized rabbit basilar artery without endothelium in vitro. In the intact rabbit basilar artery, hydroxyfasudil elicited a concentration-dependent relaxation of the artery precontracted with 1 nmol/L endothelin-1 (ET-1) plus 20 mmol/L KCl without any significant decrease in [Ca2+]i as determined by fura-2 microfluorometry (IC50: 5.1 ± 4.6 μmol/L). The relaxation induced by hydroxyfasudil was accompanied with dephosphorylation of the myosin light chain. In the permeabilized preparation, hydroxyfasudil inhibited the contraction induced by ET-1, guanosine 5′-O-(3-thiotriphosphate), or the catalytic subunit of rho-associated kinase, but it did not inhibit Ca2+-induced contraction under the condition of inhibited myosin light chain phosphatase. Hydroxyfasudil showed a greater relaxant effect under decreased adenosine triphosphate (ATP) levels. The present study indicated that hydroxyfasudil relaxes the rabbit basilar artery mainly by disinhibiting myosin light chain phosphatase through the inhibition of rho-associated kinase and that this effect depends on the intracellular ATP concentration.
Keywords
Fasudil hydrochloride is dramatically effective for directly releasing cerebral vasospasm after subarachnoid hemorrhage (SAH) in experimental animals and humans (Takayasu et al., 1986; Shibuya et al., 1992). It was initially synthesized as an inhibitor of myosin light chain kinase (MLCK). However, it was soon recognized that fasudil hydrochloride is a potent inhibitor of rho-associated kinase (RK) and protein kinase C (PKC) rather than MLCK in the cell-free in vitro study (Nagumo et al., 2000). In addition, it is well known that all of these kinases—MLCK, RK and PKC—are playing important roles in the excitation–contraction coupling in the smooth muscle (Somlyo and Somlyo, 2000). Therefore, it is of particular importance to determine the mechanism of action of fasudil hydrochloride not only for the treatment but also for the understanding of pathophysiology of cerebral vasospasm after aneurysmal SAH.
Contraction and relaxation of smooth muscle cells are primarily regulated by the changes of intracellular Ca2+ concentration ([Ca2+]i) and subsequent phosphorylation and dephosphorylation of the myosin light chain (MLC20) catalyzed by MLCK and myosin light chain phosphatase (MLCP), respectively (Somlyo and Somlyo, 1994). Recently, however, it was shown that the Ca2+ sensitivity of the contractile apparatus could be modulated by the inhibition of MLCP through rho A–RK pathway or PKC–CPI-17, a 17 kDa protein substrate, pathway in smooth muscle cells (Somlyo and Somlyo, 2000). Therefore, it is possible that smooth muscle cells can contract and relax without any change in [Ca2+]i through the activation and inactivation of RK or PKC. Because it has been reported that [Ca2+]i did not increase during cerebral vasospasm after SAH (Yamada et al., 1994), there is the temptation to speculate that fasudil hydrochloride may prevent cerebral vasospasm after SAH exclusively through the inhibition of RK, or PKC, or both. However, the direct evidence showing the inhibition of RK by fasudil hydrochloride in the in situ or in vivo study (not in the cell-free in vitro study) is still lacking.
In the current study, the mechanism underlying the relaxation induced by fasudil hydrochloride in the rabbit basilar artery was investigated. Hydroxyfasudil rather than fasudil hydrochloride was examined in the current study because fasudil hydrochloride is cleared quickly from the blood with a half-life of less than 15 minutes, whereas a detectable amount (10 to 100 nmol/L) of its hydroxylated metabolite, hydroxyfasudil, remains in the blood for as long as 8 hours after infusion (Nakashima et al., 1992; Shibuya et al., 1992). The results clearly indicate that hydroxyfasudil relaxes the rabbit basilar artery presumably through the inhibition of RK and subsequent disinhibition of MLCP without changing [Ca2+]i.
MATERIALS AND METHODS
Tissue preparation
This study followed a protocol approved by the Animal Care and Committee of Research Institute of Angiocardiology, Graduate School of Medical Sciences, Kyushu University. Adult male, Japanese white rabbits (2.5 to 3.0 kg) were killed by intravenous injection of pentobarbital sodium (400 mg) and exsanguinated from the carotid artery. The basilar artery was rapidly excised and put into ice-cold physiologic salt solution (PSS). The connective tissues around the arteries were removed under a binocular microscope, and the arteries were prepared for use by being cut into small rings measuring 500 μm in length. In all of the rings, the endothelium was mechanically removed by rubbing the intimal surface with a human hair about 10 times after the basilar arteries were cut into rings. In the current study, the effects of hydroxyfasudil were examined using intact or permeabilized preparations of the basilar artery smooth muscle.
Experiments
Intact smooth muscle study.
Measurement of tension.
Each arterial ring was mounted horizontally between 2 tungsten wires for tension measurement in an organ bath with a 2 mL working volume of PSS, which was kept at 37°C and aerated with a mixture of 95% O2 and 5% CO2 (pH 7.4 to 7.5). One wire was fixed and the other was attached to a force transducer (U Gauge; Minebea, Nagano, Japan). The preparation was allowed to equilibrate at 37°C for 60 minutes before use. During the equilibrium period, the rings were stimulated with 118 mmol/L K+-PSS every 15 minutes with increasing the resting load in a stepwise manner. The optimal resting load yielding the maximum tension development in response to depolarization with 118 mmol/L K+-PSS was thus determined in each arterial ring. The optimal resting load so obtained ranged from 250 mg to 350 mg. Before commencing each protocol, the lack of functional endothelium was confirmed by the observation that the addition of 1 μmol/L carbachol during contraction induced by 118 mmol/L K+-depolarization had not induced relaxation. For studying the relaxation responses, vessels were precontracted with 1 nmol/L endothelin-1 (ET-1) plus 20 mmol/L K+-depolarization. After the contraction had become stable, hydroxyfasudil (0.1 to 100 μmol/L) was added. The developed tension was expressed as a percentage, assigning the values in normal (5.9 mmol/L KCl) and 118 mmol/L K+-PSS to be 0% and 100%, respectively.
Microfluorometry of fura-2.
Rabbit basilar arterial rings were loaded with fura-2 through incubation in Dulbecco's Modified Eagle's Medium containing 25 μmol/L fura-2/AM (an acetoxymethyl ester form of fura-2) and 5% fetal bovine serum for 4 hours at 37°C under aeration with a mixture of 95% O2 and 5% CO2. After fura-2 loading, rings were incubated in PSS for at least 1 hour at 37°C before the measurements were started, to remove the dye in the extracellular space and for equilibration. Thereafter, the arterial ring was mounted between two tungsten wires in an organ bath specifically designed for an inverted fluorescence microscope (TMD 56; Nikon, Tokyo, Japan) equipped with a spectrophotometer (CAM 220; Japan Spectroscope, Tokyo, Japan) to perform the simultaneous measurements of fura-2 microfluorometry and tension. As for the [Ca2+]i measurement, the ratio of the fluorescence (500 nm) intensities at alternating 348-nm (F348) and 380-nm (F380) excitation wavelengths was monitored. The fluorescence ratio was expressed as a percentage, assigning the values in normal (5.9 mmol/L KCl) and 118 mmol/L K+-PSS to be 0% and 100%, respectively. Although the absolute values of [Ca2+]i of the arterial rings could be calculated, the obtained [Ca2+]i values are only an approximation to the true [Ca2+]i values; therefore, the calibration of the absolute levels of [Ca2+]i at the end of the experiments is likely to be uncertain. Therefore, a statistical analysis of the [Ca2+]i signal was performed using the values of percent fluorescence ratio. The effects of hydroxyfasudil on [Ca2+]i were examined while monitoring the change in tension.
Measurement of MLC20 phosphorylation.
The extent of MLC20 phosphorylation was determined using the urea-glycerol gel electrophoresis technique, followed by immunoblot detection with a specific mouse monoclonal anti-MLC20 antibody. The details were described previously (Zhou et al., 1999).
Permeabilized preparation study.
Permeabilization of the smooth muscle with staphylococcal α-toxin was performed as previously reported (Nishimura et al., 1988; Chen et al., 1997). Briefly, the small rings (500 μm in length) of the rabbit basilar artery were incubated in Ca2+-free cytosolic substitution solution (CSS) for 1 minute and then permeabilized by the addition of 5,000 U/mL staphylococcal α-toxin in Ca2+-free CSS for 30 minutes at 25°C. Measurement of isometric tension was performed as described previously (Nishimura et al., 1988; Chen et al., 1997; Zhou et al., 1999). Briefly, the arterial ring was mounted onto two tungsten wires bathed in wells filled with Ca2+-free CSS on a plate by passing the tungsten wires through the lumen of the arterial ring. One of the wires was fixed and the other was connected to a force transducer (U Gauge; Minebea, Nagano, Japan). The arterial ring then was stretched to twice its resting diameter, as in the authors' previous study (Zhou et al., 1999), and was allowed to relax completely in Ca2+-free CSS for 30 minutes. The extent of tension development was expressed as a percentage, assigning values of the tension in Ca2+-free CSS (resting state) and 10 μmol/L Ca2+ CSS (maximum contraction) to be 0% and 100%, respectively. The effects of hydroxyfasudil were examined against the contraction induced by applying excitatory agonists or by increasing the Ca2+ as described previously (Yoshii et al., 1999).
In examining the effects of hydroxyfasudil on RK-induced contraction, the arterial ring was permeabilized with Triton X-100. For permeabilization with Triton X-100, 0.5% Triton X-100 was applied to the pCa 6.0 solution for 20 minutes at 25°C in the presence of 1 μmol/L calmodulin, which was included in all solutions used after Triton X-100 permeabilization. All experiments on α-toxin or Triton X-100-permeabilized rings were performed at 25°C.
Drugs and solutions
Hydroxyfasudil, hexahydro-1-(1-hydroxy-5-isoquinolinesulfonyl)-1H-1,4-diazepine, was kindly provided by Asahi Chemical Industry (Shizuoka, Japan) and was dissolved in distilled water. Ethylene glycol bis(β-aminoethyl ether) N, N, N', N'-tetraacetic acid (EGTA) and fura-2/AM were purchased from Dojindo (Kumamoto, Japan). Na2ATP was from Kohjin (Tokyo, Japan), whereas creatine phosphate and Tris were from WAKO Pure Chemical Industries (Osaka, Japan). Phorbol 12, 13-dibutyrate (PDBu), staphylococcal α-toxin, and carbachol were from Sigma (St. Louis, MO, U.S.A.); guanosine 5′-O-(3-thiotriphosphate) (GTPγS) and adenosine 5′-O-(3-thiotriphosphate) (ATPγS) from Boehringer-Mannheim (Mannheim, Germany); ET-1 from Peptide Institute (Osaka, Japan); Wortmannin from Kyowa Hakko (Tokyo, Japan); Triton X-100 from Katayama Chemicals (Osaka, Japan); and calmodulin from Seikagaku Kogyo (Tokyo, Japan).
The catalytic subunit of recombinant human RK (RK-CAT; 1–452 amino acids) was expressed as a hexahistidine-tagged protein in Sf9 cells with a baculovirus system, according to the manufacturer's instructions (Life Technologies, Rockville, MD, U.S.A.). Purification of expressed protein was as described previously (Zhou et al., 1999).
The composition of PSS was 123 mmol/L NaCl, 4.7 mmol/L KCl, 15.5 mmol/L NaHCO3, 1.2 mmol/L KH2PO4, 1.2 mmol/L MgCl2, 1.25 mmol/L CaCl2, and 11.5 mmol/L d-glucose (pH 7.4). High KCl-PSS was identical to normal PSS, except for an equimolar substitution of KCl for NaCl. The composition of CSS was 10 mmol/L EGTA, 100 mmol/L K-metanesulfonate, 3.38 mmol/L MgCl2, 2.2 mmol/L Na2ATP, 10 mmol/L creatine phosphate, 20 mmol/L Tris maleate (pH 6.8), and the indicated concentration of free Ca2+. The Ca2+ CSS containing the indicated concentration of free Ca2+ was prepared by adding an appropriate amount of CaCl2, using the EGTA-Ca2+-binding constant of 106 mol/L−1 (Saida and Nonomura, 1978). When changing the adenosine triphosphate (ATP) concentration of CSS, the MgCl2 concentration also was changed to maintain the Mg2+ concentration at 1 mmol/L, using the binding constant 4 × 103 mol/L−1 for the MgATP complex (Nishimura and van Breemen, 1989, 1991). The rigor solution contained no Ca2+, no ATP, and no creatine phosphate.
Statistical analysis
All data were collected by a computerized data acquisition system (MacLab; Analog Digital Instruments, Castle Hill, Australia) running on a Macintosh computer (Apple Computer, Cupertino, CA, U.S.A.). Data were expressed as the mean ± SD. One ring obtained from one animal was used for each experiment; therefore, the number of experiments (n) indicates the number of animals. IC50 values—that is, drug concentrations eliciting 50% of the maximum inhibition—were determined from the concentration-response curve fitted according to a four-parameter logistic model
where T is developed tension, Tmax is the maximum tension (which corresponds in this case to the steady-state level of contraction before administering hydroxyfasudil), [HF] is the concentration of hydroxyfasudil, and H is the Hill coefficient. The curve fitting was performed using KaleidaGraph software (Synergy Software, Reading, PA, U.S.A.). Data were compared by Student's t-test. As for comparison of tension between hydroxyfasudil-treated samples and time-matched controls, the authors used repeated measures analysis of variance and contrast analysis. Statistical analysis was performed using STATISTICA software (StatSoft, Tulsa, OK, U.S.A.). P < 0.05 was considered to be statistically significant.
RESULTS
Effects of hydroxyfasudil in the intact rabbit basilar artery
To determine the relaxant effect, hydroxyfasudil was applied at the steady state of contraction induced by 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization, because both ET-1 (Juvela, 2000) and high K+-depolarization (Hubschmann and Kornhauser, 1982) have been implicated in the pathogenesis of cerebral vasospasm after SAH. The developed tension induced by 118 mmol/L K+-depolarization was 956.5 ± 312.8 mg (n = 12). The increase in tension and [Ca2+]i induced by 20 mmol/L K+-depolarization was 26.5% ± 18.2% and 60.6% ± 18.4% of 118 mmol/L K+-depolarization, respectively (n = 12). The level of tension and [Ca2+]i induced by 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization was 90.8% ± 16.3% and 94.1% ± 16.3% of 118 mmol/L K+-depolarization (n = 12), respectively. Hydroxyfasudil elicited a concentration-dependent relaxation of the rabbit basilar arteries precontracted with 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization (Fig. 1). The IC50 value for hydroxyfasudil-induced relaxation was 5.1 ± 4.6 μmol/L (n = 4).
Concentration-response curve for the effects of hydroxyfasudil (HF) on contraction induced by 1 nmol/L endothelin-1 (ET-1) plus 20 mmol/L K+-depolarization in the intact rabbit basilar artery (n = 4). Hydroxyfasudil was administered after the contraction became stable. In this figure, in particular, the steady-state level of contraction induced by 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization was assigned to be 100% tension to obtain IC50 values of hydroxyfasudil.
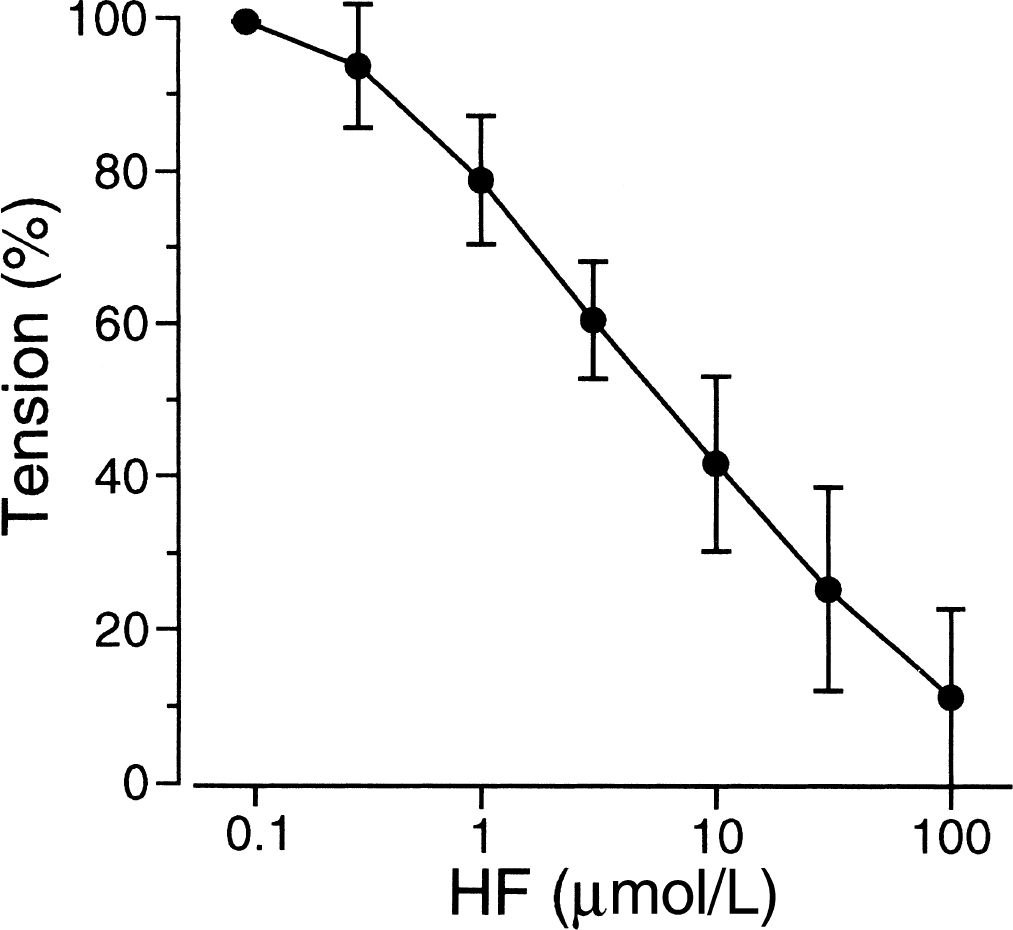
As shown in Fig. 2, the addition of 1 μmol/L hydroxyfasudil to the arterial ring precontracted with 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization decreased the tension from 86.7% ± 18.5% to 71.4% ± 19.9% (n = 5, P < 0.01). The increase in [Ca2+]i was not affected significantly by 1 μmol/L hydroxyfasudil (from 96.2% ± 15.2% to 95.1% ± 16.1%, n = 5, P > 0.1). When the effect of hydroxyfasudil on [Ca2+]I was examined, a concentration greater than 1 μmol/L could not be used because hydroxyfasudil at this concentration interfered with the measurement of fura-2 fluorescence because of its fluorescent property.
Change in fluorescence signals of fura-2 (F348, F380, and ratio) and tension induced by 1 nmol/L endothelin-1 (ET-1) plus 20 mmol/L K+-depolarization and 1 μmol/L hydroxyfasudil (HF). Hydroxyfasudil was administered after the contraction became stable. Hydroxyfasudil (1 μmol/L) did not interfere with the fluorescence signals of fura-2. Traces shown are representative of five similar experiments.
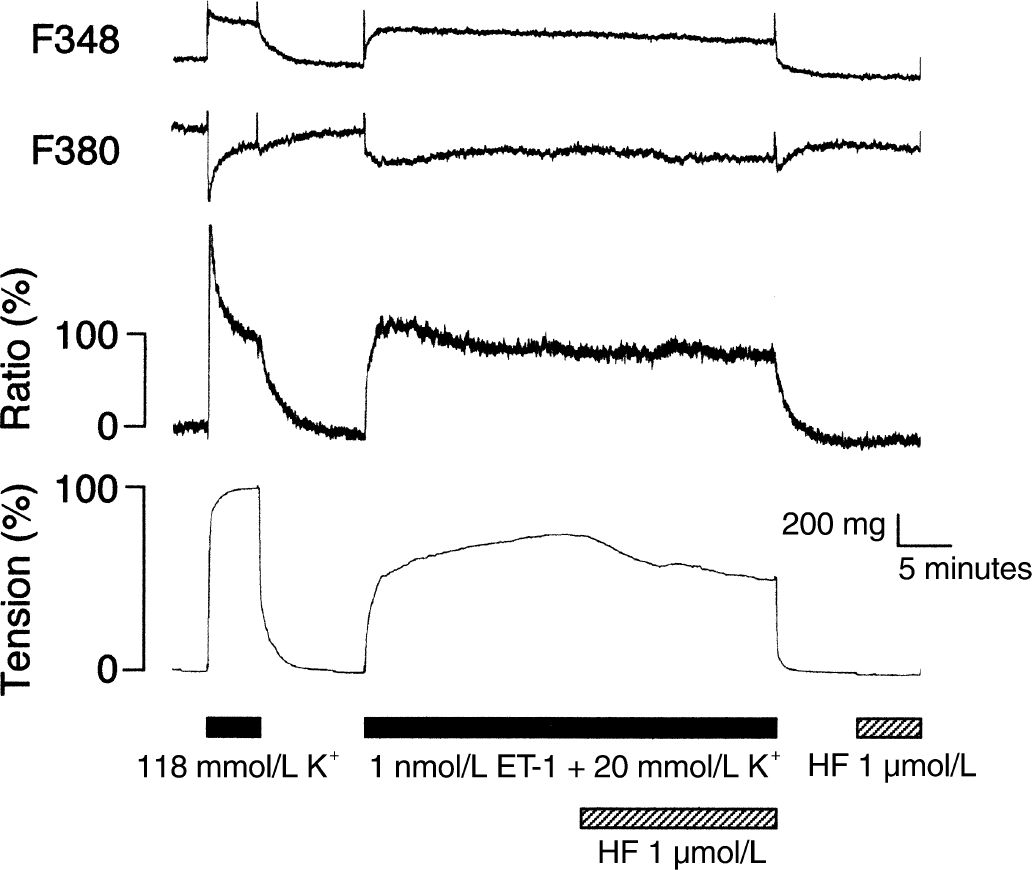
The effect of hydroxyfasudil on MLC20 phosphorylation was examined next. At the resting state (time zero), phosphorylated MLC20 was 15.4% ± 13.7% of total MLC20 (n = 5). The level of phosphorylated MLC20 was 59.2% ± 29.2% at the time point of 20 minutes after the ring was stimulated with 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization. As shown in Fig. 3, administration of 30 μmol/L hydroxyfasudil for additional 20 minutes accelerated dephosphorylation of MLC20 compared with the time-matched control.
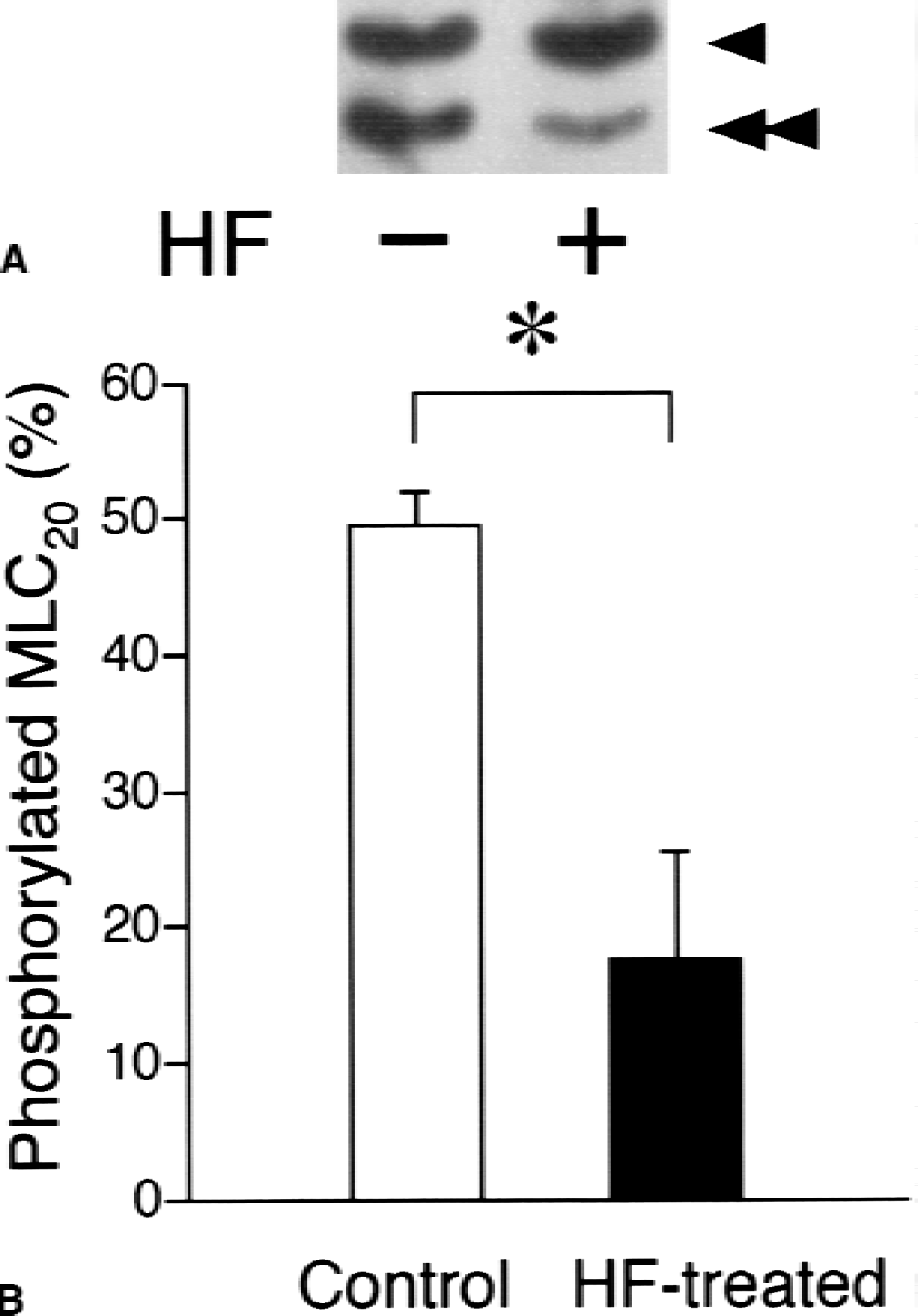
Effect of hydroxyfasudil (HF) on myosin light chain (MLC20) phosphorylation. Hydroxyfasudil (30 μmol/L) was administered 20 minutes after the stimulation with 1 nmol/L endothelin-1 (ET-1) plus 20 mmol/L K+-depolarization, and the reaction was stopped 20 minutes after administration of hydroxyfasudil.
Effects of hydroxyfasudil in the permeabilized preparation
Because the results obtained in the intact smooth muscle study might indicate that hydroxyfasudil decreases the Ca2+ sensitivity of the contractile apparatus, the authors further explored the effect of hydroxyfasudil on the Ca2+ sensitivity of the contractile apparatus by using the permeabilized preparations. Greater concentrations of hydroxyfasudil can be used and the effect of hydroxyfasudil on the Ca2+ sensitivity of the contractile apparatus can be evaluated more directly in the experiments using the permeabilized preparations. The developed tension induced by pCa 5.0 CSS was 1824.3 ± 610.1 mg in α-toxin-permeabilized preparations (n = 10). The stable contraction induced by pCa 6.7 CSS was 21.9% ± 12.2% of the maximum contraction induced by pCa 5.0 CSS (n = 10). ET-1 (100 nmol/L) induced an additional contraction at a fixed free Ca2+ concentration of pCa 6.7 in α-toxin-permeabilized preparations. The developed tension achieved 61.5% ± 7.3% (n = 10) of the maximum contraction. Although the contraction induced by ET-1 declined spontaneously, a comparison between the hydroxyfasudil-treated group and the time-matched control (the vehicle) group revealed that a concentration of hydroxyfasudil of 10 μmol/L or more significantly decreased the tension development induced by 100 nmol/L ET-1 (Fig. 4).
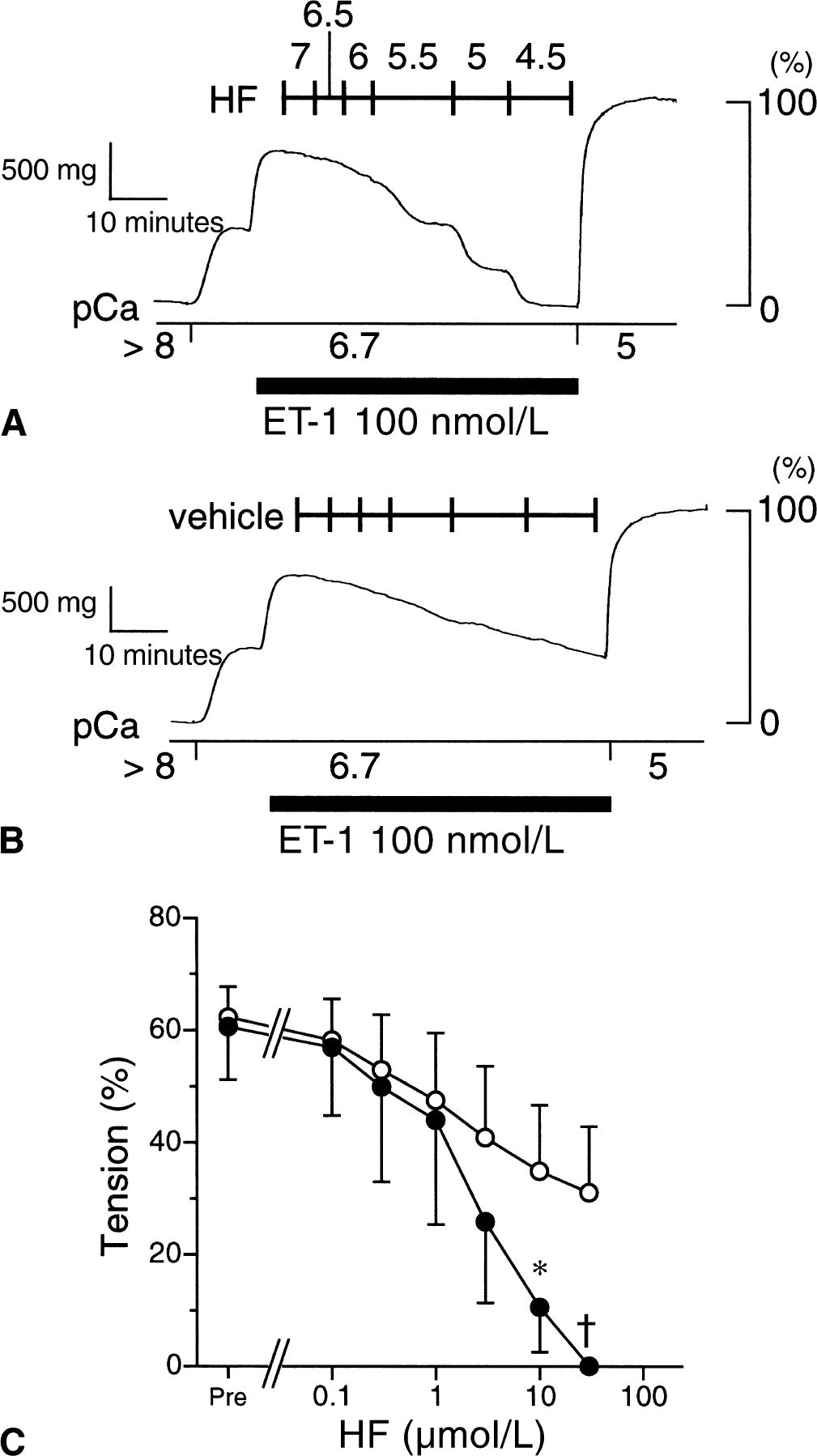
Effect of hydroxyfasudil (HF) on 100 nmol/L endothelin-1 (ET-1)-induced contraction in the α-toxin-permeabilized rabbit basilar artery at pCa 6.7. When 100 nmol/L ET-1-induced contraction reached the peak level at pCa 6.7, hydroxyfasudil was added cumulatively at the concentration shown by −log[molar concentration]
Administration of GTPγS (10 μmol/L), a nonhydrolyzable GTP analogue, at pCa 6.7 increased the tension to 64.8% ± 7.7% (n = 10) of the maximum contraction at pCa 5.0. Although the GTPγS-induced tension development declined spontaneously, as did ET-1, a concentration of hydroxyfasudil of 3 μmol/L or more significantly accelerated relaxation (Fig. 5).
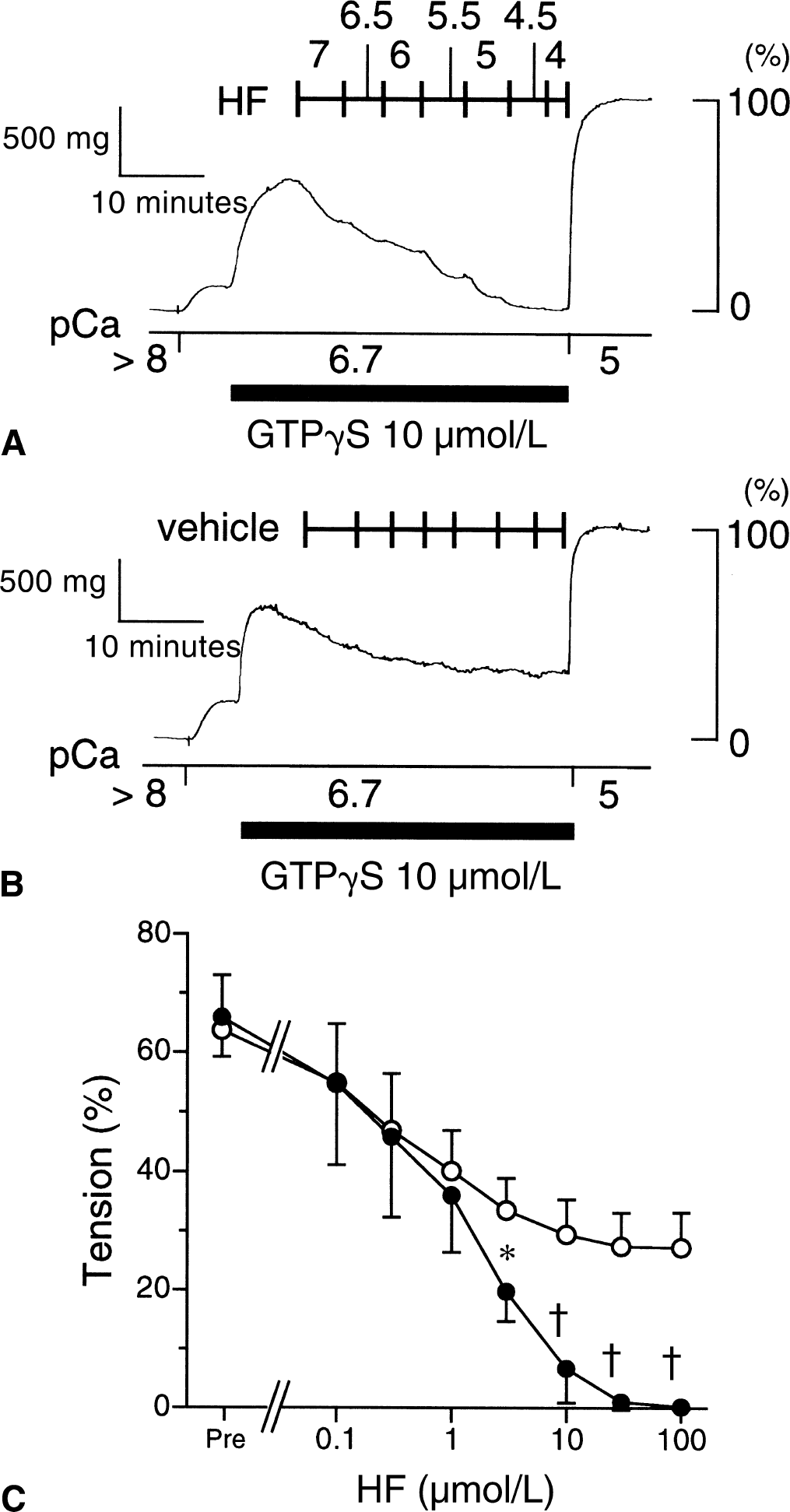
Effect of hydroxyfasudil (HF) on 10 μmol/L guanosine 5′-O-(3-thiotriphosphate) (GTPγS)-induced contraction in the α-toxin-permeabilized rabbit basilar artery at pCa 6.7. When 10 μmol/L GTPγS-induced contraction reached the peak level at pCa 6.7, hydroxyfasudil was added cumulatively at the concentration shown by −log[molar concentration]
Although GTPγS is supposed to activate rho A and subsequently RK, the activation is not specific for RK. Therefore, the effect of hydroxyfasudil on contraction induced by RK was directly examined using the catalytic subunit of RK, RK-CAT. The developed tension induced by pCa 5.0 CSS was 215.5 ± 91.4 mg in Triton-X-100-permeabilized preparations (n = 6). Introduction of 2.0 μg/mL RK-CAT into the cytosol provoked a sustained contraction at a constant cytosolic Ca2+ (pCa 6.5) (88.8 ± 3.6% of the contraction at pCa 5.0, n = 3). Administration of 30 μmol/L hydroxyfasudil decreased the developed tension to 58.5% ± 13.6% of the contraction at pCa 5.0 (n = 3, P < 0.05; Fig. 6).
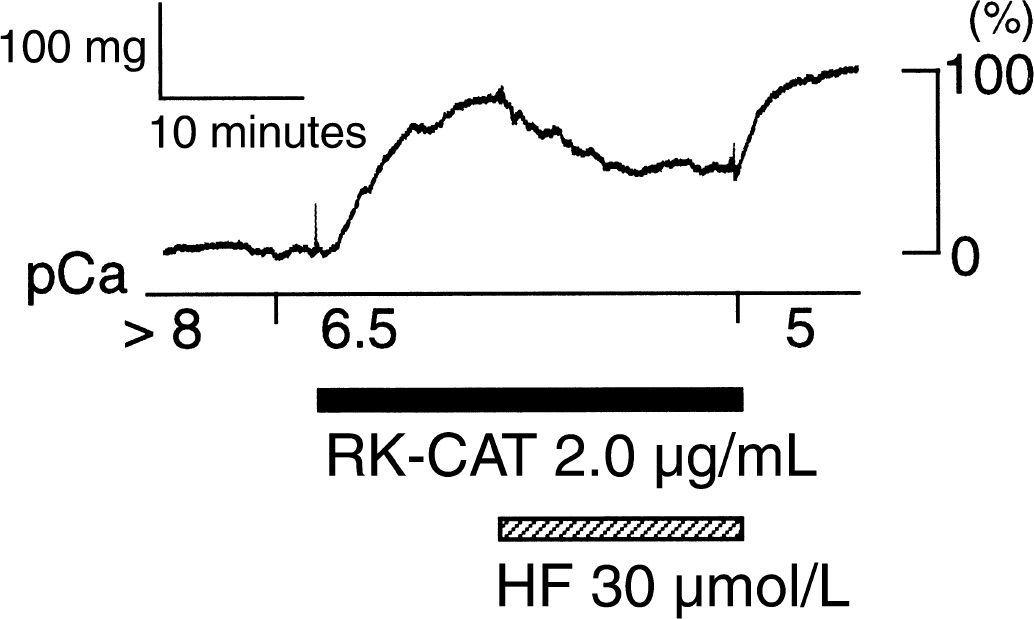
Effect of hydroxyfasudil (HF) on RK-CAT-induced contraction in the Triton X-100-permeabilized rabbit basilar artery. Hydroxyfasudil (30 μmol/L) was administered after the 2.0 μg/mL RK-CAT-induced contraction became stable. Developed tension was normalized to maximal Ca2+ (pCa 5.0)-induced contraction. Traces shown are representative of three similar experiments.
To examine the effect of hydroxyfasudil on PKC, the authors used PDBu, a PKC activator. PDBu (100 nmol/L) gradually increased the tension to 67.5% ± 10.9% of the maximum contraction at pCa 5.0 (n = 10). After the contraction had become stable, hydroxyfasudil or the vehicle was cumulatively added to the arterial rings. Although hydroxyfasudil concentration dependently relaxed the ring, the inhibitory effect of hydroxyfasudil on PDBu-induced contraction was less than that of ET-1-or GTPγS-induced contraction (Fig. 7).
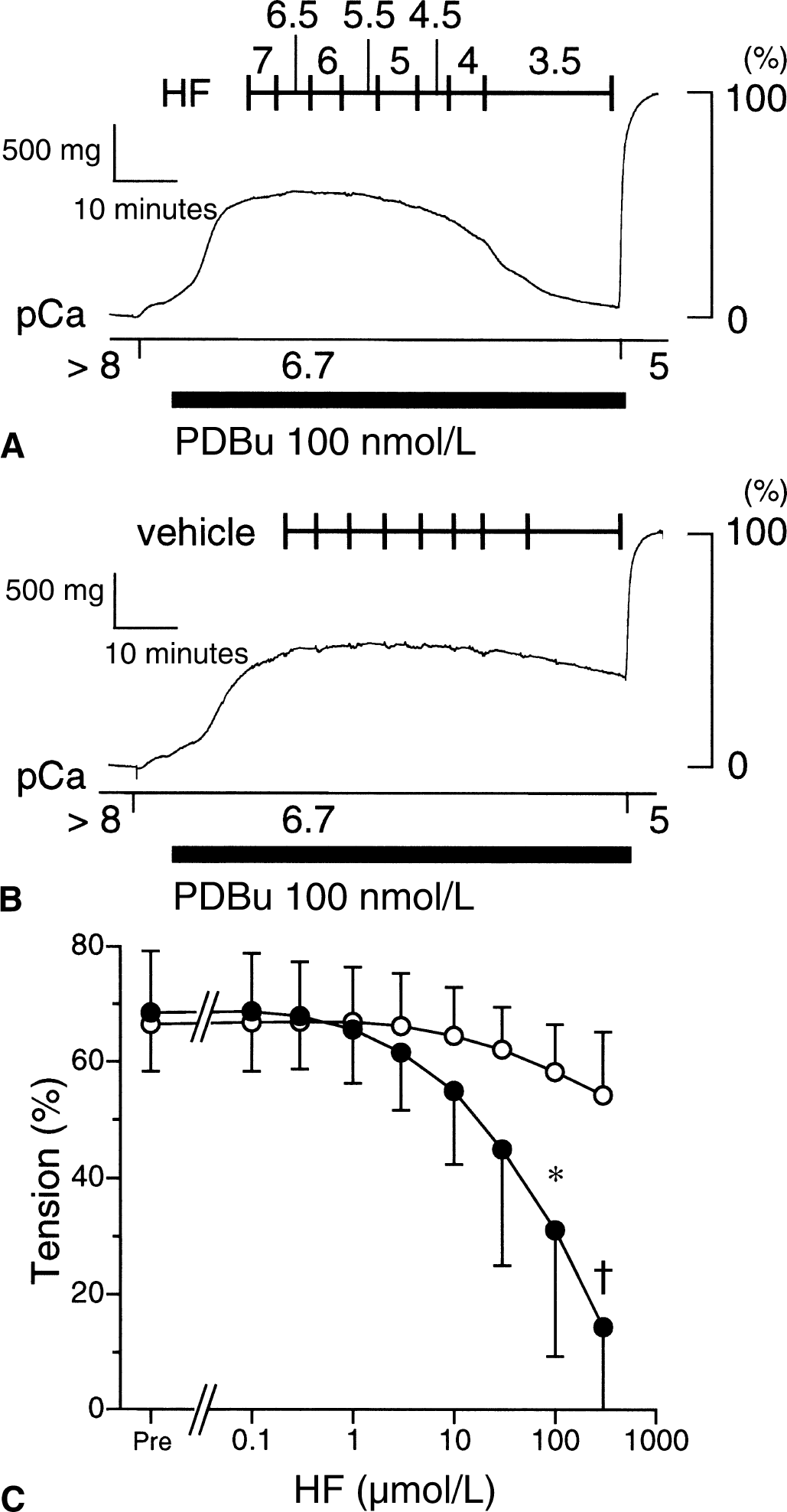
Effect of hydroxyfasudil (HF) on 100 nmol/L phorbol 12, 13-dibutyrate (PDBu)-induced contraction in the α-toxin-permeabilized rabbit basilar artery at pCa 6.7. When 100 nmol/L PDBu-induced contraction reached the peak level at pCa 6.7, hydroxyfasudil was added cumulatively at the concentration shown by −log[molar concentration]
ATPγS is reported to thiophosphorylate MLC20 and the myosin binding subunit of MLCP, which leads to the inhibition of MLCP (Trinkle-Mulcahy et al., 1995). To examine the effect of hydroxyfasudil under the condition of inhibited MLCP, the permeabilized arterial ring was first incubated in rigor solution for 15 minutes, and then switched to pCa 7.0 CSS containing 2 mmol/L ATPγS instead of ATP with or without hydroxyfasudil. As shown in Fig. 8, in pCa 7.0 CSS containing 2 mmol/L ATPγS, the tension increased both in the rings with or without hydroxyfasudil. There was no difference in the developed tension between these two groups. The time to reach half-maximal force (t1/2) was not different in either group (7.5 ± 2.6 minutes in 10 μmol/L hydroxyfasudil-treated group, n = 5; 8.6 ± 2.0 minutes in 100 μmol/L hydroxyfasudil-treated group, n = 5; and 7.1 ± 1.6 minutes in the control group, n = 5). In the rings treated with 50 μmol/L wortmannin, an MLCK inhibitor, the contraction was significantly smaller, and it took a longer time to reach the peak level compared with the control (t1/2 = 20.9 ± 1.9 minutes, n = 4;P < 0.001 vs. the control group; Fig. 8).
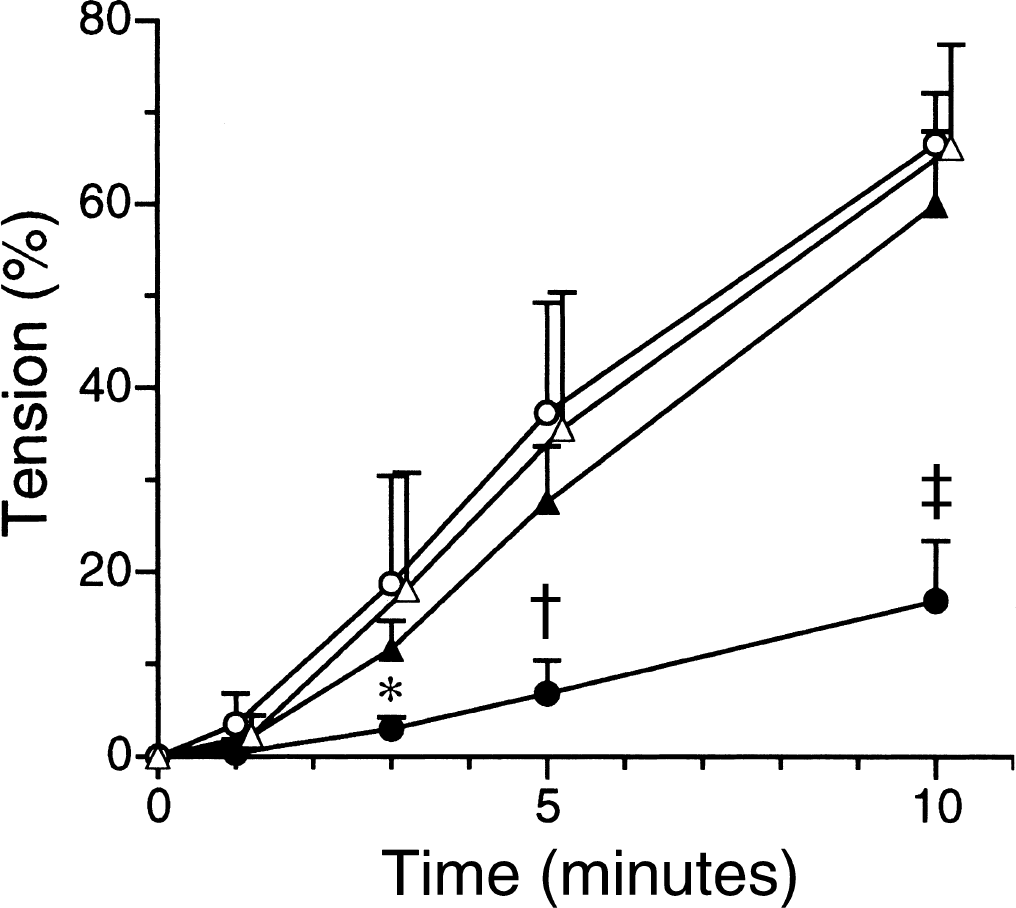
Effect of hydroxyfasudil on Ca2+-induced contraction under the inhibition of myosin light chain phosphatase (MLCP) in the α-toxin-permeabilized rabbit basilar artery. Rings were incubated in Ca2+ (0.1 μmol/L) alone (control), Ca2+ + hydroxyfasudil (10 μmol/L), Ca2+ + hydroxyfasudil (100 μmol/L), or Ca2+ + wortmannin (50 μmol/L) in the presence of 2 mmol/L adenosine 5′-O-(3-thiotriphosphate) (ATPγS), after incubation in rigor solution for 15 minutes. Time courses of changes in tension after changing the solution are summarized for the control (open circles, n = 5), 10 μmol/L hydroxyfasudil-treated (open triangles, n = 5), 100 μmol/L hydroxyfasudil-treated (closed triangles, n = 5), and 50 μmol/L wortmannin-treated rings (closed circles, n = 4). Developed tension was normalized to maximal Ca2+ (pCa 5.0)-induced contraction. * P < 0.05, †P < 0.01, ‡P < 0.001 vs. the control (repeated measures analysis of variance and contrast analysis).
Finally, the effect of the ATP concentration on the hydroxyfasudil-induced relaxation was examined. In pCa 6.5 CSS containing 2 mmol/L MgATP, 3 μmol/L hydroxyfasudil decreased the tension from 58.8% ± 16.8% to 37.0% ± 20.4% (n = 4, P < 0.01). When the MgATP concentration was decreased to 0.1 mmol/L, hydroxyfasudil (3 μmol/L) induced greater relaxation (from 29.5% ± 12.2% to 6.8% ± 5.3%; Fig. 9A to 9C). However, when the MgATP concentration was increased to 10 mmol/L, the relaxant effect of hydroxyfasudil was not observed (from 42.3% ± 8.1% to 39.3% ± 7.9%; Fig. 9D to 9F).
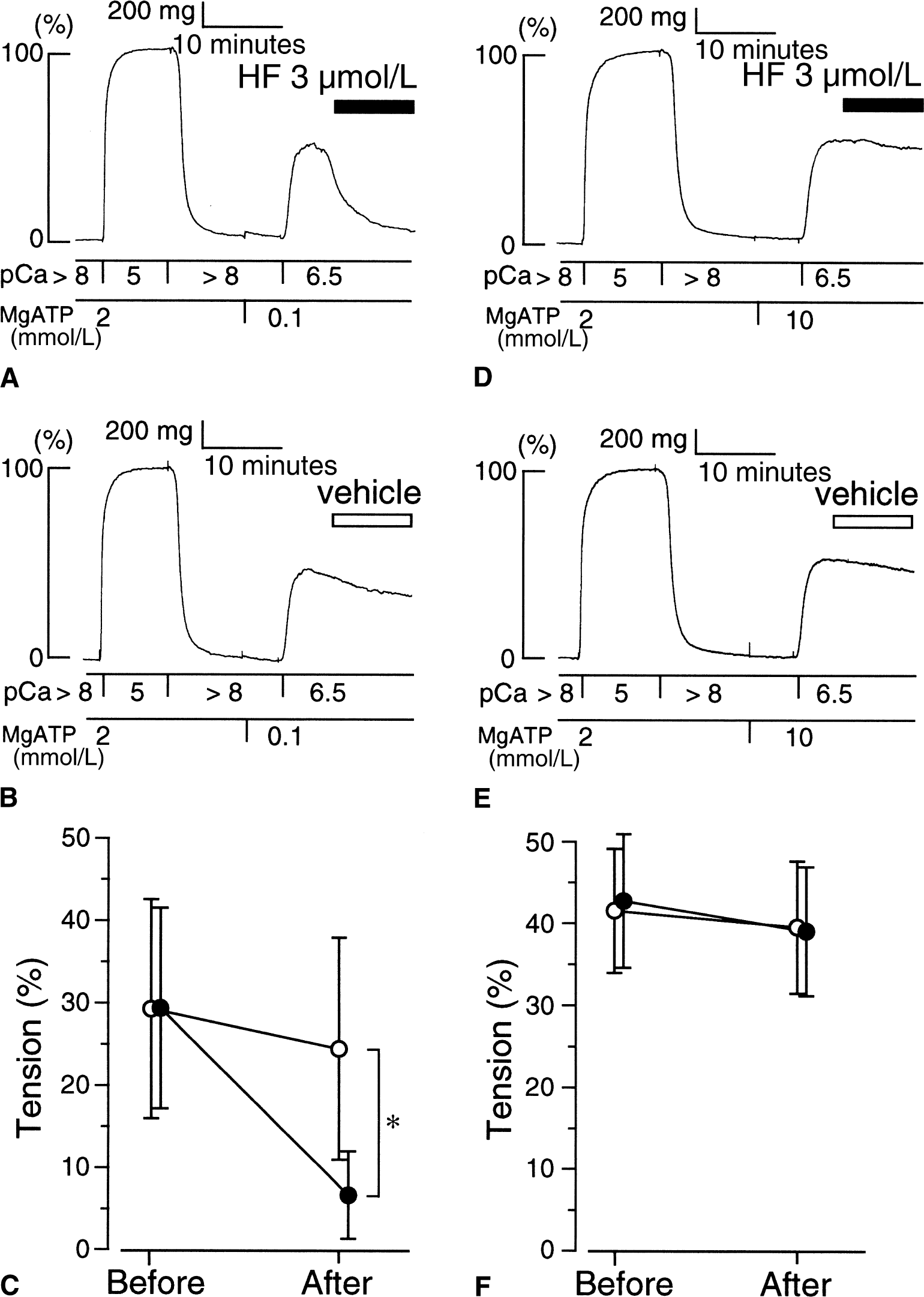
Effect of the adenosine triphosphate (ATP) concentration on hydroxyfasudil (HF)-induced relaxation in the α-toxin-permeabilized rabbit basilar artery. Hydroxyfasudil (
DISCUSSION
In the current study, the authors investigated the mechanism underlying the hydroxyfasudil-induced relaxation of the rabbit basilar artery without endothelium. The major findings are as follows: (1) hydroxyfasudil relaxed the intact rabbit basilar artery contracted with 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization without decreasing [Ca2+]i levels; (2) hydroxyfasudil inhibited the contraction induced by ET-1, GTPγS, RK, or PDBu in permeabilized preparations at constant pCa levels; (3) hydroxyfasudil did not inhibit Ca2+-induced contraction, when MLCP was inhibited by ATPγS; and (4) the effect of hydroxyfasudil depended on the intracellular ATP concentration.
In the intact basilar artery study, ET-1 plus KCl was used to contract the vessel for two reasons. First, both ET-1 (Juvela, 2000) and high K+-depolarization (Hubschmann and Kornhauser, 1982) are considered to be involved in the development of vasospasm after SAH. Second, the contraction induced by ET-1 alone often fluctuates in the rabbit basilar artery. Once the contraction fluctuates, it becomes difficult to evaluate the relaxant effect of hydroxyfasudil. It was reported that fluctuating myogenic contraction is because of the interaction between increased [Ca2+]i and K+ channels in the rabbit basilar artery (Omote and Mizusawa, 1996). Therefore, the authors administered ET-1 in 20 mmol/L K+-PSS, which made the contraction stable. The authors found that the precontraction induced by ET-1 in 20 mmol/L K+-PSS might be the best way to evaluate the relaxant effect by the vasodilators in the rabbit basilar artery. Although the mechanism of contraction induced by ET-1 and KCl may not be the same and this may complicate the interpretation of the results, this may be an appropriate model to examine the effect of hydroxyfasudil on [Ca2+]i-tension relationship.
The activation of smooth muscle cells induces the increase in [Ca2+]i and subsequent activation of Ca2+-calmodulin dependent MLCK to phosphorylate MLC20. The phosphorylation of MLC20 at serine 19 increases the actin-activated ATPase activity of myosin to induce contraction (Somlyo and Somlyo, 1994). In addition, contraction and relaxation of the vascular smooth muscle are regulated by the agonist-and G-protein-mediated modulation of the [Ca2+]i-tension relation, that is, the Ca2+ sensitivity of the contractile apparatus (Nishimura et al., 1988; Kitazawa et al., 1989). Hydroxyfasudil relaxed the rabbit basilar artery precontracted with 1 nmol/L ET-1 plus 20 mmol/L K+-depolarization (Fig. 1) without significant changes in [Ca2+]i (Fig. 2). According to a previous study, fasudil hydrochloride, a source of hydroxyfasudil, inhibited Ca2+ influx in the rat aorta (Takizawa et al., 1993). This discrepancy may be explained by the difference in the concentration of the drug used, or by the difference in the chemical structure (that is, fasudil hydrochloride vs. hydroxyfasudil), or both. Although the possibility that high concentrations of hydroxyfasudil inhibit Ca2+ influx could not be ruled out, the results obtained in the current study suggest that the relaxant effect of hydroxyfasudil could not be explained solely by the inhibition of calcium influx, and also that a decrease in the Ca2+ sensitivity of the contractile apparatus plays an important role in the relaxation. The decrease in the Ca2+ sensitivity of the contractile apparatus is, so far, explained by the decrease in phosphorylated MLC20 through activation of MLCP, or inactivation of MLCK (Somlyo and Somlyo, 2000), or both. The results shown in Fig. 3 confirmed this, because hydroxyfasudil decreased the MLC20 phosphorylation level.
To further investigate how hydroxyfasudil induces relaxation at a constant [Ca2+]i, a receptor coupled permeabilization by α-toxin was used. It has been reported that MLCP activity is inhibited by RK-catalyzed phosphorylation of the myosin binding subunit of MLCP (Kimura et al., 1996) and PKC-catalyzed phosphorylation of CPI-17, a 17 kDa protein substrate of PKC that becomes a potent inhibitor of MLCP after phosphorylation (Li et al., 1998; Kitazawa et al., 2000). Thus, the authors expected that hydroxyfasudil should induce relaxation of the contraction induced by the activation of these pathways if hydroxyfasudil-induced relaxation involves these pathways. As shown in Fig. 5, hydroxyfasudil inhibited the increase in the Ca2+ sensitivity of the contractile apparatus induced by GTPγS, a potent activator of rho A and RK. This result indicates that hydroxyfasudil may inhibit RK to disinhibit MLCP. In other words, hydroxyfasudil might inhibit RK and induce activation of MLCP, which has been inhibited by receptor-mediated activation of rho A–RK pathway. To further identify the site of action, recombinant RK-CAT was used for the activation. As shown in Fig. 6, the contraction induced by RK-CAT could be relaxed by hydroxyfasudil, clearly indicating the inhibition of RK by hydroxyfasudil. These findings confirmed the results obtained in the cell-free in vitro study showing the inhibitory effect of hydroxyfasudil on RK (Shimokawa et al., 1999).
Hydroxyfasudil also inhibited the increase in the Ca2+ sensitivity of the contractile apparatus induced by PDBu, a potent activator of PKC (Fig. 7). However, the inhibition of PKC induced by hydroxyfasudil may not play a major role, because the relaxant effect on the PDBu-induced contraction was much less than that on the GTPγS-induced contraction.
The possibility that hydroxyfasudil inhibits MLCK to decrease the Ca2+ sensitivity of the contractile apparatus was assessed by the experiments shown in Fig. 8. The rate of contraction induced by Ca2+ in the presence of ATPγS should reflect the MLCK activity because of the following reasons. When ATPγS is used as the substrate instead of ATP, MLC20 is thiophosphorylated in a Ca2+-dependent manner (Hoar et al., 1979). This thiophosphorylated MLC20 is a poor substrate for MLCP. ATPγS treatment of the permeabilized smooth muscle also thiophosphorylates the myosin binding subunit of MLCP, which leads to the inhibition of MLCP (Trinkle-Mulcahy et al., 1995). Thus, ATPγS treatment should result in the inhibition of MLCP. As shown in Fig. 8, hydroxyfasudil did not inhibit ATPγS-induced contraction, whereas wortmannin, an MLCK inhibitor, did inhibit this contraction. This result indicates that the inhibition of MLCK by hydroxyfasudil may not play a major role in the relaxation.
The effect of hydroxyfasudil depended on the intracellular ATP concentration (Fig. 9). The relaxant effect of hydroxyfasudil was augmented by decreasing the ATP concentration and antagonized by increasing the ATP concentration. The current results support the previous report that isoquinoline derivatives, including hydroxyfasudil, inhibit the activity of several serine/threonine protein kinases by interacting with the ATP-binding sites through its isoquinoline ring (Takizawa et al., 1993). From the clinical point of view, the fact that hydroxyfasudil shows a stronger relaxant effect under decreased ATP levels appears to be important, because decreased ATP levels have been documented in the smooth muscle cells of the vasospastic artery after SAH (Yoshimoto et al., 1993).
In conclusion, the present study indicated that hydroxyfasudil, an active metabolite of fasudil hydrochloride in vivo, relaxed the rabbit basilar artery mainly by disinhibiting MLCP through the inhibition of RK. The dramatic therapeutic effect of fasudil hydrochloride on cerebral vasospasm after aneurysmal SAH may suggest the involvement of RK in the pathogenesis of cerebral vasospasm in human aneurysmal SAH.